These Highlights Do Not Include All The Information Needed To Use Dexrazoxane For Injection Safely And Effectively. See Full Prescribing Information For Dexrazoxane For Injection.
a7c01089-85f5-40aa-b679-f53a425b4d68
34391-3
Human Prescription Drug Label
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dexrazoxane for injection is indicated for reducing the incidence and severity of cardiomyopathy associated with doxorubicin administration in women with metastatic breast cancer who have received a cumulative doxorubicin dose of 300 mg/m 2 and who will continue to receive doxorubicin therapy to maintain tumor control. Do not use with the initiation of doxorubicin therapy [see Warnings and Precautions (5.2) ].
Indications and Usage
Dexrazoxane for injection is indicated for reducing the incidence and severity of cardiomyopathy associated with doxorubicin administration in women with metastatic breast cancer who have received a cumulative doxorubicin dose of 300 mg/m 2 and who will continue to receive doxorubicin therapy to maintain tumor control. Do not use with the initiation of doxorubicin therapy [see Warnings and Precautions (5.2) ].
Dosage and Administration
Reconstitute vial contents and dilute before use. ( 2.3 ) Administer dexrazoxane for injection by intravenous infusion over 15 minutes. DO NOT ADMINISTER VIA AN INTRAVENOUS PUSH. ( 2.1 , 2.3 ) The recommended dosage ratio of dexrazoxane for injection to doxorubicin is 10:1 (e.g., 500 mg/m 2 dexrazoxane for injection to 50 mg/m 2 doxorubicin). Do not administer doxorubicin before dexrazoxane for injection. ( 2.1 ) Reduce dose by 50% for patients with creatinine clearance <40 mL/min. ( 2.2 , 8.6 )
Warnings and Precautions
Myelosuppression: Dexrazoxane may increase the myelosuppresive effects of chemotherapeutic agents. Perform hematological monitoring. ( 5.1 ) Embryo-Fetal Toxicity: Can cause fetal harm. Advise patients of the potential risk to a fetus and to use effective contraception. ( 5.5 , 8.1 , 8.3 )
Contraindications
Do not use dexrazoxane with non-anthracycline chemotherapy regimens.
Adverse Reactions
In clinical studies, dexrazoxane was administered to patients also receiving chemotherapeutic agents for cancer. Pain on injection was observed more frequently in patients receiving dexrazoxane versus placebo. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Novadoz Pharmaceuticals LLC at 1-855-668-2369 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
No drug interactions have been identified [see Clinical Pharmacology (12.3) ].
Storage and Handling
Dexrazoxane for injection is available in the following strengths as a sterile, off white to pale yellow pyrogen-free lyophilized powder in a single-dose vials for reconstitution. Strength Pack Style NDC Code 250 mg Carton of single-dose vial with a blue flip-top seal 72205-247-01 500 mg Carton of single-dose vial with a blue flip-top seal 72205-248-01 Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Follow special handling and disposal procedures. 1 Discard unused solutions.
How Supplied
Dexrazoxane for injection is available in the following strengths as a sterile, off white to pale yellow pyrogen-free lyophilized powder in a single-dose vials for reconstitution. Strength Pack Style NDC Code 250 mg Carton of single-dose vial with a blue flip-top seal 72205-247-01 500 mg Carton of single-dose vial with a blue flip-top seal 72205-248-01 Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Follow special handling and disposal procedures. 1 Discard unused solutions.
Medication Information
Warnings and Precautions
Myelosuppression: Dexrazoxane may increase the myelosuppresive effects of chemotherapeutic agents. Perform hematological monitoring. ( 5.1 ) Embryo-Fetal Toxicity: Can cause fetal harm. Advise patients of the potential risk to a fetus and to use effective contraception. ( 5.5 , 8.1 , 8.3 )
Indications and Usage
Dexrazoxane for injection is indicated for reducing the incidence and severity of cardiomyopathy associated with doxorubicin administration in women with metastatic breast cancer who have received a cumulative doxorubicin dose of 300 mg/m 2 and who will continue to receive doxorubicin therapy to maintain tumor control. Do not use with the initiation of doxorubicin therapy [see Warnings and Precautions (5.2) ].
Dosage and Administration
Reconstitute vial contents and dilute before use. ( 2.3 ) Administer dexrazoxane for injection by intravenous infusion over 15 minutes. DO NOT ADMINISTER VIA AN INTRAVENOUS PUSH. ( 2.1 , 2.3 ) The recommended dosage ratio of dexrazoxane for injection to doxorubicin is 10:1 (e.g., 500 mg/m 2 dexrazoxane for injection to 50 mg/m 2 doxorubicin). Do not administer doxorubicin before dexrazoxane for injection. ( 2.1 ) Reduce dose by 50% for patients with creatinine clearance <40 mL/min. ( 2.2 , 8.6 )
Contraindications
Do not use dexrazoxane with non-anthracycline chemotherapy regimens.
Adverse Reactions
In clinical studies, dexrazoxane was administered to patients also receiving chemotherapeutic agents for cancer. Pain on injection was observed more frequently in patients receiving dexrazoxane versus placebo. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Novadoz Pharmaceuticals LLC at 1-855-668-2369 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
No drug interactions have been identified [see Clinical Pharmacology (12.3) ].
Storage and Handling
Dexrazoxane for injection is available in the following strengths as a sterile, off white to pale yellow pyrogen-free lyophilized powder in a single-dose vials for reconstitution. Strength Pack Style NDC Code 250 mg Carton of single-dose vial with a blue flip-top seal 72205-247-01 500 mg Carton of single-dose vial with a blue flip-top seal 72205-248-01 Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Follow special handling and disposal procedures. 1 Discard unused solutions.
How Supplied
Dexrazoxane for injection is available in the following strengths as a sterile, off white to pale yellow pyrogen-free lyophilized powder in a single-dose vials for reconstitution. Strength Pack Style NDC Code 250 mg Carton of single-dose vial with a blue flip-top seal 72205-247-01 500 mg Carton of single-dose vial with a blue flip-top seal 72205-248-01 Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Follow special handling and disposal procedures. 1 Discard unused solutions.
Description
Dexrazoxane for injection is indicated for reducing the incidence and severity of cardiomyopathy associated with doxorubicin administration in women with metastatic breast cancer who have received a cumulative doxorubicin dose of 300 mg/m 2 and who will continue to receive doxorubicin therapy to maintain tumor control. Do not use with the initiation of doxorubicin therapy [see Warnings and Precautions (5.2) ].
10 Overdosage
There are no data on overdosage in the cardioprotective trials; the maximum dose administered during the cardioprotective trials was 1000 mg/m2 every three weeks.
Disposition studies with dexrazoxane have not been conducted in cancer patients undergoing dialysis, but retention of a significant dose fraction (>0.4) of the unchanged drug in the plasma pool, minimal tissue partitioning or binding, and availability of greater than 90% of the systemic drug levels in the unbound form suggest that it could be removed using conventional peritoneal or hemodialysis.
There is no known antidote for dexrazoxane. Instances of suspected overdose should be managed with good supportive care until resolution of myelosuppression and related conditions is complete. Management of overdose should include treatment of infections, fluid regulation, and maintenance of nutritional requirements.
15 References
1. “OSHA Hazardous Drugs.” OSHA http://www.osha.gov/SLTC/hazardousdrugs/index.html.
8.1 Pregnancy
Risk Summary
Based on findings from animal studies and its mechanism of action, dexrazoxane can cause fetal harm when administered to pregnant women [see Clinical Pharmacology (12.1)]. Available human data with dexrazoxane use in pregnant women is not sufficient to inform the drug-associated risk. In animal reproduction studies, dexrazoxane administration resulted in maternal toxicity and teratogenicity in rats and rabbits at doses significantly lower than the clinically recommended dose (see Data). Advise pregnant women and females of reproductive potential of the potential risk to a fetus.
The background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
Dexrazoxane resulted in maternal toxicity in rats at doses of ≥2 mg/kg (1/40 the human dose on a mg/m2 basis) and embryotoxicity and teratogenicity at 8 mg/kg (approximately 1/10 the human dose on a mg/m2 basis) when given daily to pregnant rats during the period of organogenesis. Teratogenic effects in the rat included imperforate anus, microphthalmia, and anophthalmia. In offspring allowed to develop to maturity, fertility was impaired in the male and female rats treated in utero during organogenesis at 8 mg/kg. In rabbits, doses of ≥5 mg/kg (approximately 1/10 the human dose on a mg/m2 basis) daily during the period of organogenesis caused maternal toxicity and doses of 20 mg/kg (1/2 the human dose on a mg/m2 basis) were embryotoxic and teratogenic. Teratogenic effects in the rabbit included several skeletal malformations such as short tail, rib and thoracic malformations, and soft tissue variations including subcutaneous, eye and cardiac hemorrhagic areas, as well as agenesis of the gallbladder and of the intermediate lobe of the lung.
8.2 Lactation
Risk Summary
There are no data on the presence of dexrazoxane or its metabolites in human milk, the effects on a breastfeed child, or the effects on milk production. Because of the potential for serious adverse reactions in a breastfed child, advise women not to breastfeed during treatment with dexrazoxane and for 2 weeks after the last dose.
11 Description
Dexrazoxane for injection, a cardioprotective agent for use in conjunction with doxorubicin, is a sterile, pyrogen-free lyophilizate intended for intravenous administration.
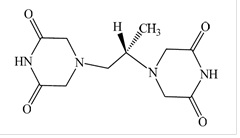
Chemically, dexrazoxane is (S)-4,4'-(1-methyl-1,2-ethanediyl)bis-2,6-piperazinedione. The structural formula is as follows:
C11H16N4O4 M.W. 268.28
Dexrazoxane, an intracellular chelating agent, is a derivative of EDTA. Dexrazoxane is a white to off-white crystalline powder that melts at 191° to 197°C. It is sparingly soluble in 0.1 N HCl, slightly soluble in water, very slightly soluble in methanol, and insoluble in cyclohexane. The pKa is 2.2. Dexrazoxane has an octanol/water partition coefficient of (-) 1.43 and degrades rapidly above a pH of 7.0.
Each 250 mg vial contains dexrazoxane hydrochloride equivalent to 250 mg dexrazoxane. Hydrochloric Acid, NF is added for pH adjustment. When reconstituted as directed with 25 mL of Sterile Water for Injection, USP, each mL contains: 10 mg dexrazoxane. The pH of the resultant solution is 1.0 to 3.0.
Each 500 mg vial contains dexrazoxane hydrochloride equivalent to 500 mg dexrazoxane. Hydrochloric Acid, NF is added for pH adjustment. When reconstituted as directed with 50 mL of Sterile Water for Injection, USP, each mL contains: 10 mg dexrazoxane. The pH of the resultant solution is 1.0 to 3.0.
The reconstituted dexrazoxane for injection, solutions prepared from Sterile Water for Injection, USP, are intended for further dilution with Lactated Ringer’s Injection, USP, for rapid intravenous drip infusion. DO NOT ADMINISTER VIA AN INTRAVENOUS PUSH [see Dosage and Administration (2.1, 2.3)].
8.4 Pediatric Use
The safety and effectiveness of dexrazoxane in pediatric patients have not been established [see Warnings and Precautions (5.4)].
8.5 Geriatric Use
Clinical studies of dexrazoxane did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently than younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
14 Clinical Studies
The ability of dexrazoxane to prevent/reduce the incidence and severity of doxorubicin-induced cardiomyopathy was evaluated in three prospectively randomized placebo-controlled studies. In these studies, patients were treated with a doxorubicin-containing regimen and either dexrazoxane or placebo starting with the first course of chemotherapy. There was no restriction on the cumulative dose of doxorubicin. Cardiac function was assessed by measurement of the LVEF, utilizing resting multigated nuclear medicine (MUGA) scans, and by clinical evaluations. Patients receiving dexrazoxane had significantly smaller mean decreases from baseline in LVEF and lower incidences of congestive heart failure than the control group; however, in the largest study, patients with advanced breast cancer receiving FAC with dexrazoxane had a lower response rate (48% vs. 63%) and a shorter time to progression than patients who received FAC versus placebo.
In the clinical trials, patients who were initially randomized to receive placebo were allowed to receive dexrazoxane after a cumulative dose of doxorubicin above 300 mg/m2. Retrospective historical analyses showed that the risk of experiencing a cardiac event (see Table 3 for definition) at a cumulative dose of doxorubicin above 300 mg/m2 was greater in the patients who did not receive dexrazoxane beginning with their seventh course of FAC than in the patients who did receive dexrazoxane (HR=13.08; 95% CI: 3.72, 46.03; p<0.001). Overall, 3% of patients treated with dexrazoxane developed CHF compared with 22% of patients not receiving dexrazoxane.
Table 3: Definition of Cardiac Events:
- Development of congestive heart failure, defined as having two or more of the following:
a. Cardiomegaly by X-ray
b. Basilar Rales
c. S3 Gallop
d. Paroxysmal nocturnal dyspnea and/or orthopnea and/or significant dyspnea on exertion. - Decline from baseline in LVEF by ≥10% and to below the lower limit of normal for the institution.
- Decline in LVEF by ≥20% from baseline value.
- Decline in LVEF to ≥5% below lower limit of normal for the institution.
Figure 1 shows the number of patients still on treatment at increasing cumulative doses.
Figure 1
Cumulative Number of Patients On Treatment FAC vs. FAC/Dexrazoxane Patients
Patients Receiving at Least Seven Courses of Treatment
4 Contraindications
Do not use dexrazoxane with non-anthracycline chemotherapy regimens.
6 Adverse Reactions
In clinical studies, dexrazoxane was administered to patients also receiving chemotherapeutic agents for cancer. Pain on injection was observed more frequently in patients receiving dexrazoxane versus placebo. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Novadoz Pharmaceuticals LLC at 1-855-668-2369 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions
No drug interactions have been identified [see Clinical Pharmacology (12.3)].
2.1 Recommended Dose
Administer dexrazoxane for injection via intravenous infusion over 15 minutes. DO NOT ADMINISTER VIA AN INTRAVENOUS PUSH.
The recommended dosage ratio of dexrazoxane for injection to doxorubicin is 10:1 (e.g., 500 mg/m2 dexrazoxane for injection to 50 mg/m2 doxorubicin). Do not administer doxorubicin before dexrazoxane for injection. Administer doxorubicin within 30 minutes after the completion of dexrazoxane for injection infusion.
5.1 Myelosuppression
Dexrazoxane may add to the myelosuppression caused by chemotherapeutic agents. Obtain a complete blood count prior to and during each course of therapy, and administer dexrazoxane and chemotherapy only when adequate hematologic parameters are met.
5.3 Cardiac Toxicity
Treatment with dexrazoxane does not completely eliminate the risk of anthracycline-induced cardiac toxicity. Monitor cardiac function before and periodically during therapy to assess left ventricular ejection fraction (LVEF). In general, if test results indicate deterioration in cardiac function associated with doxorubicin, the benefit of continued therapy should be carefully evaluated against the risk of producing irreversible cardiac damage.
8.6 Renal Impairment
Greater exposure to dexrazoxane may occur in patients with compromised renal function. Reduce the dexrazoxane dose by 50% in patients with creatinine clearance values <40 mL/min [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
The pharmacokinetics of dexrazoxane have been studied in advanced cancer patients with normal renal and hepatic function. The pharmacokinetics of dexrazoxane can be adequately described by a two-compartment open model with first-order elimination. Dexrazoxane has been administered as a 15 minute infusion over a dose range of 60 to 900 mg/m2 with 60 mg/m2 of doxorubicin, and at a fixed dose of 500 mg/m2 with 50 mg/m2 doxorubicin. The disposition kinetics of dexrazoxane are dose-independent, as shown by linear relationship between the area under plasma concentration-time curves and administered doses ranging from 60 to 900 mg/m2. The mean peak plasma concentration of dexrazoxane was 36.5 µg/mL at 15- minute after intravenous administration of 500 mg/m2 dose of dexrazoxane over 15 to 30 minutes prior to the 50 mg/m2 doxorubicin dose.
The important pharmacokinetic parameters of dexrazoxane are summarized in Table 2:
Table 2: SUMMARY OF MEAN (%CVa) DEXRAZOXANE
PHARMACOKINETIC PARAMETERS AT A DOSAGE RATIO OF
10:1 OF DEXRAZOXANE:DOXORUBICIN
| Dose Doxorubicin (mg/m2) |
Dose Dexrazoxane (mg/m2) |
Number of Subjects |
Elimination Half-Life (h) |
Plasma Clearance (L/h/m2) |
Renal Clearance (L/h/m2) |
bVolume of Distribution (L/m2) |
| 50 |
500 |
10 |
2.5 (16) |
7.88 (18) |
3.35 (36) |
22.4 (22) |
| 60 |
600 |
5 |
2.1 (29) |
6.25 (31) |
— |
22.0 (55) |
a Coefficient of variation
b Steady-state volume of distribution
Distribution
Following a rapid distributive phase (0.2 to 0.3 hours), dexrazoxane reaches post-distributive equilibrium within two to four hours. The estimated mean steady-state volume of distribution of dexrazoxane is 22.4 L/m2 after 500 mg/m2 of dexrazoxane dose followed by 50 mg/m2 of doxorubicin, suggesting distribution throughout total body water (25 L/m2).
In vitro studies have shown that dexrazoxane is not bound to plasma proteins.
Metabolism
Qualitative metabolism studies with dexrazoxane have confirmed the presence of unchanged drug, a diacid-diamide cleavage product, and two monoacid-monoamide ring products in the urine of animals and man. The metabolite levels were not measured in the pharmacokinetic studies.
Excretion
Urinary excretion plays an important role in the elimination of dexrazoxane. Forty-two percent of a 500 mg/m2 dose of dexrazoxane was excreted in the urine. Renal clearance averages 3.35 L/h/m2 after the 500 mg/m2 dexrazoxane dose followed by 50 mg/m2 of doxorubicin.
Specific Populations
Pediatric
Pharmacokinetics following dexrazoxane administration have not been evaluated in pediatric patients.
Effect of Renal Impairment
The pharmacokinetics of dexrazoxane were assessed following a single 15-minute IV infusion of 150 mg/m2 of dexrazoxane. Dexrazoxane clearance was reduced in subjects with renal dysfunction. Compared with controls, the mean AUC0-inf value was two-fold greater in subjects with moderate (CLCR 30-50 mL/min) to severe (CLCR <30 mL/min) renal dysfunction. Modeling demonstrated that equivalent exposure (AUC-inf) could be achieved if dosing were reduced by 50% in subjects with creatinine clearance values <40 mL/min compared with control subjects (CLCR >80 mL/min) [see Use in Specific Populations (8.6) and Dosage and Administration (2.2)].
Effect of Hepatic Impairment
Pharmacokinetics following dexrazoxane administration have not been evaluated in patients with hepatic impairment. The dexrazoxane dose is dependent upon the dose of doxorubicin [seeDosage and Administration (2.2)].
Drug Interactions
There was no significant change in the pharmacokinetics of doxorubicin (50 mg/m2) and its predominant metabolite, doxorubicinol, in the presence of dexrazoxane (500 mg/m2) in a crossover study in cancer patients.
2.2 Dose Modifications
Dosing in Patients with Renal Impairment
Reduce dexrazoxane for injection dosage in patients with moderate to severe renal impairment (creatinine clearance values less than 40 mL/min) by 50% (Dexrazoxane for injection to doxorubicin ratio reduced to 5:1; such as 250 mg/m2 dexrazoxane for injection to 50 mg/m2 doxorubicin) [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
Dosing in Patients with Hepatic Impairment
Since a doxorubicin dose reduction is recommended in the presence of hyperbilirubinemia, reduce the dexrazoxane for injection dosage proportionately (maintaining the 10:1 ratio) in patients with hepatic impairment.
1 Indications and Usage
Dexrazoxane for injection is indicated for reducing the incidence and severity of cardiomyopathy associated with doxorubicin administration in women with metastatic breast cancer who have received a cumulative doxorubicin dose of 300 mg/m2 and who will continue to receive doxorubicin therapy to maintain tumor control. Do not use with the initiation of doxorubicin therapy [see Warnings and Precautions (5.2)].
12.1 Mechanism of Action
The mechanism by which dexrazoxane exerts its cytoprotective activity is not fully understood. Dexrazoxane is a cyclic derivative of EDTA that penetrates cell membranes. Results of laboratory studies suggest that dexrazoxane is converted intracellularly to a ring-opened chelating agent that interferes with iron-mediated free radical generation thought to be responsible, in part, for anthracycline-induced cardiomyopathy.
5.5 Embryo Fetal Toxicity
Based on finding from animal studies and its mechanism of action, dexrazoxane can cause fetal harm when administered to pregnant women [see Clinical Pharmacology (12.1)]. In animal reproduction studies, dexrazoxane administration during the period of organogenesis resulted in maternal toxicity and teratogenicity in rats and rabbits at doses significantly lower than the clinically recommended dose.
Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with dexrazoxane and for 6 months after the last dose. Advise male patients with female partners of reproductive potential to use effective contraception during treatment with dexrazoxane and for 3 months after the last dose [see Use in Specific Populations (8.1, 8.3)].
5 Warnings and Precautions
5.4 Secondary Malignancies
Secondary malignancies such as acute myeloid leukemia (AML) and myelodysplastic syndrome (MDS) have been reported in studies of pediatric patients who have received dexrazoxane in combination with chemotherapy. Dexrazoxane is not indicated for use in pediatric patients. Some adult patients who received dexrazoxane in combination with anti-cancer agents known to be carcinogenic have also developed secondary malignancies, including AML and MDS.
Razoxane is the racemic mixture, of which dexrazoxane is the S(+)-enantiomer. Secondary malignancies (primarily acute myeloid leukemia) have been reported in patients treated chronically with oral razoxane. In these patients, the total cumulative dose of razoxane ranged from 26 to 480 grams and the duration of treatment was from 42 to 319 weeks. One case of T-cell lymphoma, one case of B-cell lymphoma, and six to eight cases of cutaneous basal cell or squamous cell carcinoma have also been reported in patients treated with razoxane. Long-term administration of razoxane to rodents was associated with the development of malignancies [see Nonclinical Toxicology (13.1)].
2 Dosage and Administration
- Reconstitute vial contents and dilute before use. (2.3)
- Administer dexrazoxane for injection by intravenous infusion over 15 minutes.
- DO NOT ADMINISTER VIA AN INTRAVENOUS PUSH. (2.1, 2.3)
- The recommended dosage ratio of dexrazoxane for injection to doxorubicin is 10:1 (e.g., 500 mg/m2 dexrazoxane for injection to 50 mg/m2 doxorubicin). Do not administer doxorubicin before dexrazoxane for injection. (2.1)
- Reduce dose by 50% for patients with creatinine clearance <40 mL/min. (2.2, 8.6)
3 Dosage Forms and Strengths
For injection: 250 mg or 500 mg of dexrazoxane as a sterile, off white to pale yellow lyophilized powder in a single-dose vials for reconstitution.
5.2 Concomitant Chemotherapy
Only use dexrazoxane in those patients who have received a cumulative doxorubicin dose of 300 mg/m2 and are continuing with doxorubicin therapy. Do not use with chemotherapy initiation as dexrazoxane may interfere with the antitumor activity of the chemotherapy regimen. In a trial conducted in patients with metastatic breast cancer who were treated with fluorouracil, doxorubicin, and cyclophosphamide (FAC) with or without dexrazoxane starting with their first cycle of FAC therapy, patients who were randomized to receive dexrazoxane had a lower response rate (48% vs. 63%) and shorter time to progression than patients who were randomized to receive placebo.
8 Use in Specific Populations
- Lactation: Advise not to breastfeed. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reaction rates observed cannot be directly compared to rates in other trials and may not reflect the rates observed in clinical practice.
The adverse reaction profile described in this section was identified from randomized, placebo- controlled, double-blind studies in patients with metastatic breast cancer who received the combination of the FAC chemotherapy regimen with or without dexrazoxane. The dose of doxorubicin was 50 mg/m2 in each of these trials. Treatment was administered every three weeks until disease progression or cardiac toxicity.
Patients in clinical trials who received FAC with dexrazoxane experienced more severe leukopenia, granulocytopenia, and thrombocytopenia than patients receiving FAC without dexrazoxane [see Warnings and Precautions (5.1)].
Table 1 below lists the incidence of adverse reactions for patients receiving FAC with either dexrazoxane or placebo in the breast cancer studies. Adverse experiences occurring during courses 1 through 6 are displayed for patients receiving dexrazoxane or placebo with FAC beginning with their first course of therapy (columns 1 and 3, respectively). Adverse experiences occurring at course 7 and beyond for patients who received placebo with FAC during the first six courses and who then received either dexrazoxane or placebo with FAC are also displayed (columns 2 and 4, respectively).
The adverse reactions listed below in Table 1 demonstrate that the frequency of adverse reaction “Pain on Injection” has been greater for dexrazoxane arm, as compared to placebo.
Table 1
| Adverse Reaction |
Percentage (%) of Breast Cancer Patients With Adverse Reaction |
|||
|---|---|---|---|---|
| FAC + Dexrazoxane |
FAC + Placebo |
|||
| Courses 1-6 N = 413 |
Courses ≥ 7 N = 102 |
Courses 1-6 N = 458 |
Courses ≥ 7 N = 99 |
|
| Alopecia |
94 |
100 |
97 |
98 |
| Nausea |
77 |
51 |
84 |
60 |
| Vomiting |
59 |
42 |
72 |
49 |
| Fatigue/Malaise |
61 |
48 |
58 |
55 |
| Anorexia |
42 |
27 |
47 |
38 |
| Stomatitis |
34 |
26 |
41 |
28 |
| Fever |
34 |
22 |
29 |
18 |
| Infection |
23 |
19 |
18 |
21 |
| Diarrhea |
21 |
14 |
24 |
7 |
| Pain on Injection |
12 |
13 |
3 |
0 |
| Sepsis |
17 |
12 |
14 |
9 |
| Neurotoxicity |
17 |
10 |
13 |
5 |
| Streaking/Erythema |
5 |
4 |
4 |
2 |
| Phlebitis |
6 |
3 |
3 |
5 |
| Esophagitis |
6 |
3 |
7 |
4 |
| Dysphagia |
8 |
0 |
10 |
5 |
| Hemorrhage |
2 |
3 |
2 |
1 |
| Extravasation |
1 |
3 |
1 |
2 |
| Urticaria |
2 |
2 |
2 |
0 |
| Recall Skin Reaction |
1 |
1 |
2 |
0 |
17 Patient Counseling Information
Myelosuppression
Treatment with dexrazoxane is associated with leukopenia, neutropenia, and thrombocytopenia. Perform hematological monitoring [see Warnings and Precautions (5.1)].
Embryo-Fetal Toxicity
Advise female patients of the potential risk to a fetus and to inform their healthcare provider of a known or suspected pregnancy. Advise females of reproductive potential to use effective contraception during treatment with dexrazoxane and for 6 months after the last dose. Advise male patients with female partners of reproductive potential to use effective contraception during treatment with dexrazoxane and for 3 months after the last dose [seeWarnings and Precautions (5.5) and Use in Specific Populations (8.1, 8.3)].
Lactation
Advise women not to breastfeed during treatment with dexrazoxane and for 2 weeks after the last dose [see Use in Specific Populations (8.2)].
Infertility
Advise males of reproductive potential that dexrazoxane may impair fertility [see Use in Specific Populations (8.3)].
This product’s label may have been updated. For current full prescribing information, please visit www.msnlabs.com.
Manufactured by:
MSN Laboratories Private Limited
Telangana – 509 228,
INDIA
Distributed by:
Novadoz Pharmaceuticals LLC
Piscataway, NJ 08854 -3714
Revised: 07/2025
2.3 Preparation and Administration
Preparation and Handling of Infusion Solution
Reconstitute dexrazoxane for injection with Sterile Water for Injection, USP. Reconstitute with 25 mL for a dexrazoxane for injection 250 mg vial and 50 mL for a dexrazoxane for injection 500 mg vial to give a concentration of 10 mg/mL. Dilute the reconstituted solution further with Lactated Ringer’s Injection, USP to a concentration of 1.3 to 3.0 mg/mL in intravenous infusion bags for intravenous infusion.
Following reconstitution with Sterile Water for Injection, USP, dexrazoxane for injection is stable for 30 minutes at room temperature or if storage is necessary, up to 3 hours from the time of reconstitution when stored under refrigeration, 2° to 8°C (36° to 46°F). The pH of the resultant solution is 1.0 to 3.0. DISCARD UNUSED SOLUTIONS. The diluted infusion solutions are stable for one hour at room temperature or if storage is necessary, up to 4 hours when stored under refrigeration, 2° to 8°C (36° to 46°F). The infusion solutions have a pH of 3.5 to 5.5. DISCARD UNUSED SOLUTIONS.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Solutions containing a precipitate should be discarded.
Use caution when handling and preparing the reconstituted solution. The use of gloves is recommended. If dexrazoxane for injection powder or solutions contact the skin or mucosae, wash exposed area immediately and thoroughly with soap and water. Follow special handling and disposal procedures.1
Administration
Do not mix dexrazoxane for injection with other drugs.
Administer the final diluted solution of dexrazoxane for injection by intravenous infusion over 15 minutes before the administration of doxorubicin. DO NOT ADMINISTER VIA AN INTRAVENOUS PUSH. Administer doxorubicin within 30 minutes after the completion of dexrazoxane for injection infusion.
16 How Supplied/storage and Handling
Dexrazoxane for injection is available in the following strengths as a sterile, off white to pale yellow pyrogen-free lyophilized powder in a single-dose vials for reconstitution.
| Strength
|
Pack Style
|
NDC Code
|
| 250 mg |
Carton of single-dose vial with a blue flip-top seal |
72205-247-01 |
| 500 mg |
Carton of single-dose vial with a blue flip-top seal |
72205-248-01 |
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Follow special handling and disposal procedures.1
Discard unused solutions.
Package Label.principal Display Panel
Dexrazoxane for injection 250 mg/Vial Carton Label
Dexrazoxane for injection 250 mg/Vial Vial Label
Dexrazoxane for injection 500 mg/Vial Carton Label
Dexrazoxane for injection 500 mg/Vial Vial Label
8.3 Females and Males of Reproductive Potential
Dexrazoxane can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)].
Pregnancy Testing
Verify pregnancy status in females of reproductive potential prior to starting treatment with dexrazoxane.
Contraception
Females
Advise females of reproductive potential to use effective contraception during treatment with dexrazoxane and for 6 months after the last dose. Males Because of the potential for genotoxicity, advise male patients with female partners of reproductive potential to use effective contraception during treatment with dexrazoxane and for 3 months after the last dose [see Nonclinical Toxicology (13.1)].
Infertility Based on findings from animal studies, dexrazoxane may impair male fertility [see Nonclinical Toxicology (13.1)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term carcinogenicity studies have been carried out with dexrazoxane in animals. Nevertheless, a study by the National Cancer Institute has reported that long-term dosing with razoxane (the racemic mixture of dexrazoxane, ICRF-187, and its enantiomer ICRF-186) is associated with the development of malignancies in rats and possibly in mice [see Warnings and Precautions (5.4)].
Dexrazoxane was not mutagenic in the bacterial reverse mutation (Ames) test, but was found to be clastogenic to human lymphocytes in vitro and to mouse bone marrow erythrocytes in vivo (micronucleus test).
Dexrazoxane has the potential to impair fertility in male patients based on effects in repeat-dose toxicology studies. Testicular atrophy was seen with dexrazoxane administration at doses as low as 30 mg/kg weekly for 6 weeks in rats (1/3 the human dose on a mg/m2 basis) and as low as 20 mg/kg weekly for 13 weeks in dogs (approximately equal to the human dose on a mg/m2 basis).
Structured Label Content
10 Overdosage (10 OVERDOSAGE)
There are no data on overdosage in the cardioprotective trials; the maximum dose administered during the cardioprotective trials was 1000 mg/m2 every three weeks.
Disposition studies with dexrazoxane have not been conducted in cancer patients undergoing dialysis, but retention of a significant dose fraction (>0.4) of the unchanged drug in the plasma pool, minimal tissue partitioning or binding, and availability of greater than 90% of the systemic drug levels in the unbound form suggest that it could be removed using conventional peritoneal or hemodialysis.
There is no known antidote for dexrazoxane. Instances of suspected overdose should be managed with good supportive care until resolution of myelosuppression and related conditions is complete. Management of overdose should include treatment of infections, fluid regulation, and maintenance of nutritional requirements.
15 References (15 REFERENCES)
1. “OSHA Hazardous Drugs.” OSHA http://www.osha.gov/SLTC/hazardousdrugs/index.html.
8.1 Pregnancy
Risk Summary
Based on findings from animal studies and its mechanism of action, dexrazoxane can cause fetal harm when administered to pregnant women [see Clinical Pharmacology (12.1)]. Available human data with dexrazoxane use in pregnant women is not sufficient to inform the drug-associated risk. In animal reproduction studies, dexrazoxane administration resulted in maternal toxicity and teratogenicity in rats and rabbits at doses significantly lower than the clinically recommended dose (see Data). Advise pregnant women and females of reproductive potential of the potential risk to a fetus.
The background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
Dexrazoxane resulted in maternal toxicity in rats at doses of ≥2 mg/kg (1/40 the human dose on a mg/m2 basis) and embryotoxicity and teratogenicity at 8 mg/kg (approximately 1/10 the human dose on a mg/m2 basis) when given daily to pregnant rats during the period of organogenesis. Teratogenic effects in the rat included imperforate anus, microphthalmia, and anophthalmia. In offspring allowed to develop to maturity, fertility was impaired in the male and female rats treated in utero during organogenesis at 8 mg/kg. In rabbits, doses of ≥5 mg/kg (approximately 1/10 the human dose on a mg/m2 basis) daily during the period of organogenesis caused maternal toxicity and doses of 20 mg/kg (1/2 the human dose on a mg/m2 basis) were embryotoxic and teratogenic. Teratogenic effects in the rabbit included several skeletal malformations such as short tail, rib and thoracic malformations, and soft tissue variations including subcutaneous, eye and cardiac hemorrhagic areas, as well as agenesis of the gallbladder and of the intermediate lobe of the lung.
8.2 Lactation
Risk Summary
There are no data on the presence of dexrazoxane or its metabolites in human milk, the effects on a breastfeed child, or the effects on milk production. Because of the potential for serious adverse reactions in a breastfed child, advise women not to breastfeed during treatment with dexrazoxane and for 2 weeks after the last dose.
11 Description (11 DESCRIPTION)
Dexrazoxane for injection, a cardioprotective agent for use in conjunction with doxorubicin, is a sterile, pyrogen-free lyophilizate intended for intravenous administration.
Chemically, dexrazoxane is (S)-4,4'-(1-methyl-1,2-ethanediyl)bis-2,6-piperazinedione. The structural formula is as follows:
C11H16N4O4 M.W. 268.28
Dexrazoxane, an intracellular chelating agent, is a derivative of EDTA. Dexrazoxane is a white to off-white crystalline powder that melts at 191° to 197°C. It is sparingly soluble in 0.1 N HCl, slightly soluble in water, very slightly soluble in methanol, and insoluble in cyclohexane. The pKa is 2.2. Dexrazoxane has an octanol/water partition coefficient of (-) 1.43 and degrades rapidly above a pH of 7.0.
Each 250 mg vial contains dexrazoxane hydrochloride equivalent to 250 mg dexrazoxane. Hydrochloric Acid, NF is added for pH adjustment. When reconstituted as directed with 25 mL of Sterile Water for Injection, USP, each mL contains: 10 mg dexrazoxane. The pH of the resultant solution is 1.0 to 3.0.
Each 500 mg vial contains dexrazoxane hydrochloride equivalent to 500 mg dexrazoxane. Hydrochloric Acid, NF is added for pH adjustment. When reconstituted as directed with 50 mL of Sterile Water for Injection, USP, each mL contains: 10 mg dexrazoxane. The pH of the resultant solution is 1.0 to 3.0.
The reconstituted dexrazoxane for injection, solutions prepared from Sterile Water for Injection, USP, are intended for further dilution with Lactated Ringer’s Injection, USP, for rapid intravenous drip infusion. DO NOT ADMINISTER VIA AN INTRAVENOUS PUSH [see Dosage and Administration (2.1, 2.3)].
8.4 Pediatric Use
The safety and effectiveness of dexrazoxane in pediatric patients have not been established [see Warnings and Precautions (5.4)].
8.5 Geriatric Use
Clinical studies of dexrazoxane did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently than younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
14 Clinical Studies (14 CLINICAL STUDIES)
The ability of dexrazoxane to prevent/reduce the incidence and severity of doxorubicin-induced cardiomyopathy was evaluated in three prospectively randomized placebo-controlled studies. In these studies, patients were treated with a doxorubicin-containing regimen and either dexrazoxane or placebo starting with the first course of chemotherapy. There was no restriction on the cumulative dose of doxorubicin. Cardiac function was assessed by measurement of the LVEF, utilizing resting multigated nuclear medicine (MUGA) scans, and by clinical evaluations. Patients receiving dexrazoxane had significantly smaller mean decreases from baseline in LVEF and lower incidences of congestive heart failure than the control group; however, in the largest study, patients with advanced breast cancer receiving FAC with dexrazoxane had a lower response rate (48% vs. 63%) and a shorter time to progression than patients who received FAC versus placebo.
In the clinical trials, patients who were initially randomized to receive placebo were allowed to receive dexrazoxane after a cumulative dose of doxorubicin above 300 mg/m2. Retrospective historical analyses showed that the risk of experiencing a cardiac event (see Table 3 for definition) at a cumulative dose of doxorubicin above 300 mg/m2 was greater in the patients who did not receive dexrazoxane beginning with their seventh course of FAC than in the patients who did receive dexrazoxane (HR=13.08; 95% CI: 3.72, 46.03; p<0.001). Overall, 3% of patients treated with dexrazoxane developed CHF compared with 22% of patients not receiving dexrazoxane.
Table 3: Definition of Cardiac Events:
- Development of congestive heart failure, defined as having two or more of the following:
a. Cardiomegaly by X-ray
b. Basilar Rales
c. S3 Gallop
d. Paroxysmal nocturnal dyspnea and/or orthopnea and/or significant dyspnea on exertion. - Decline from baseline in LVEF by ≥10% and to below the lower limit of normal for the institution.
- Decline in LVEF by ≥20% from baseline value.
- Decline in LVEF to ≥5% below lower limit of normal for the institution.
Figure 1 shows the number of patients still on treatment at increasing cumulative doses.
Figure 1
Cumulative Number of Patients On Treatment FAC vs. FAC/Dexrazoxane Patients
Patients Receiving at Least Seven Courses of Treatment
4 Contraindications (4 CONTRAINDICATIONS)
Do not use dexrazoxane with non-anthracycline chemotherapy regimens.
6 Adverse Reactions (6 ADVERSE REACTIONS)
In clinical studies, dexrazoxane was administered to patients also receiving chemotherapeutic agents for cancer. Pain on injection was observed more frequently in patients receiving dexrazoxane versus placebo. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Novadoz Pharmaceuticals LLC at 1-855-668-2369 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions (7 DRUG INTERACTIONS)
No drug interactions have been identified [see Clinical Pharmacology (12.3)].
2.1 Recommended Dose
Administer dexrazoxane for injection via intravenous infusion over 15 minutes. DO NOT ADMINISTER VIA AN INTRAVENOUS PUSH.
The recommended dosage ratio of dexrazoxane for injection to doxorubicin is 10:1 (e.g., 500 mg/m2 dexrazoxane for injection to 50 mg/m2 doxorubicin). Do not administer doxorubicin before dexrazoxane for injection. Administer doxorubicin within 30 minutes after the completion of dexrazoxane for injection infusion.
5.1 Myelosuppression
Dexrazoxane may add to the myelosuppression caused by chemotherapeutic agents. Obtain a complete blood count prior to and during each course of therapy, and administer dexrazoxane and chemotherapy only when adequate hematologic parameters are met.
5.3 Cardiac Toxicity
Treatment with dexrazoxane does not completely eliminate the risk of anthracycline-induced cardiac toxicity. Monitor cardiac function before and periodically during therapy to assess left ventricular ejection fraction (LVEF). In general, if test results indicate deterioration in cardiac function associated with doxorubicin, the benefit of continued therapy should be carefully evaluated against the risk of producing irreversible cardiac damage.
8.6 Renal Impairment
Greater exposure to dexrazoxane may occur in patients with compromised renal function. Reduce the dexrazoxane dose by 50% in patients with creatinine clearance values <40 mL/min [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
The pharmacokinetics of dexrazoxane have been studied in advanced cancer patients with normal renal and hepatic function. The pharmacokinetics of dexrazoxane can be adequately described by a two-compartment open model with first-order elimination. Dexrazoxane has been administered as a 15 minute infusion over a dose range of 60 to 900 mg/m2 with 60 mg/m2 of doxorubicin, and at a fixed dose of 500 mg/m2 with 50 mg/m2 doxorubicin. The disposition kinetics of dexrazoxane are dose-independent, as shown by linear relationship between the area under plasma concentration-time curves and administered doses ranging from 60 to 900 mg/m2. The mean peak plasma concentration of dexrazoxane was 36.5 µg/mL at 15- minute after intravenous administration of 500 mg/m2 dose of dexrazoxane over 15 to 30 minutes prior to the 50 mg/m2 doxorubicin dose.
The important pharmacokinetic parameters of dexrazoxane are summarized in Table 2:
Table 2: SUMMARY OF MEAN (%CVa) DEXRAZOXANE
PHARMACOKINETIC PARAMETERS AT A DOSAGE RATIO OF
10:1 OF DEXRAZOXANE:DOXORUBICIN
| Dose Doxorubicin (mg/m2) |
Dose Dexrazoxane (mg/m2) |
Number of Subjects |
Elimination Half-Life (h) |
Plasma Clearance (L/h/m2) |
Renal Clearance (L/h/m2) |
bVolume of Distribution (L/m2) |
| 50 |
500 |
10 |
2.5 (16) |
7.88 (18) |
3.35 (36) |
22.4 (22) |
| 60 |
600 |
5 |
2.1 (29) |
6.25 (31) |
— |
22.0 (55) |
a Coefficient of variation
b Steady-state volume of distribution
Distribution
Following a rapid distributive phase (0.2 to 0.3 hours), dexrazoxane reaches post-distributive equilibrium within two to four hours. The estimated mean steady-state volume of distribution of dexrazoxane is 22.4 L/m2 after 500 mg/m2 of dexrazoxane dose followed by 50 mg/m2 of doxorubicin, suggesting distribution throughout total body water (25 L/m2).
In vitro studies have shown that dexrazoxane is not bound to plasma proteins.
Metabolism
Qualitative metabolism studies with dexrazoxane have confirmed the presence of unchanged drug, a diacid-diamide cleavage product, and two monoacid-monoamide ring products in the urine of animals and man. The metabolite levels were not measured in the pharmacokinetic studies.
Excretion
Urinary excretion plays an important role in the elimination of dexrazoxane. Forty-two percent of a 500 mg/m2 dose of dexrazoxane was excreted in the urine. Renal clearance averages 3.35 L/h/m2 after the 500 mg/m2 dexrazoxane dose followed by 50 mg/m2 of doxorubicin.
Specific Populations
Pediatric
Pharmacokinetics following dexrazoxane administration have not been evaluated in pediatric patients.
Effect of Renal Impairment
The pharmacokinetics of dexrazoxane were assessed following a single 15-minute IV infusion of 150 mg/m2 of dexrazoxane. Dexrazoxane clearance was reduced in subjects with renal dysfunction. Compared with controls, the mean AUC0-inf value was two-fold greater in subjects with moderate (CLCR 30-50 mL/min) to severe (CLCR <30 mL/min) renal dysfunction. Modeling demonstrated that equivalent exposure (AUC-inf) could be achieved if dosing were reduced by 50% in subjects with creatinine clearance values <40 mL/min compared with control subjects (CLCR >80 mL/min) [see Use in Specific Populations (8.6) and Dosage and Administration (2.2)].
Effect of Hepatic Impairment
Pharmacokinetics following dexrazoxane administration have not been evaluated in patients with hepatic impairment. The dexrazoxane dose is dependent upon the dose of doxorubicin [seeDosage and Administration (2.2)].
Drug Interactions
There was no significant change in the pharmacokinetics of doxorubicin (50 mg/m2) and its predominant metabolite, doxorubicinol, in the presence of dexrazoxane (500 mg/m2) in a crossover study in cancer patients.
2.2 Dose Modifications
Dosing in Patients with Renal Impairment
Reduce dexrazoxane for injection dosage in patients with moderate to severe renal impairment (creatinine clearance values less than 40 mL/min) by 50% (Dexrazoxane for injection to doxorubicin ratio reduced to 5:1; such as 250 mg/m2 dexrazoxane for injection to 50 mg/m2 doxorubicin) [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
Dosing in Patients with Hepatic Impairment
Since a doxorubicin dose reduction is recommended in the presence of hyperbilirubinemia, reduce the dexrazoxane for injection dosage proportionately (maintaining the 10:1 ratio) in patients with hepatic impairment.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Dexrazoxane for injection is indicated for reducing the incidence and severity of cardiomyopathy associated with doxorubicin administration in women with metastatic breast cancer who have received a cumulative doxorubicin dose of 300 mg/m2 and who will continue to receive doxorubicin therapy to maintain tumor control. Do not use with the initiation of doxorubicin therapy [see Warnings and Precautions (5.2)].
12.1 Mechanism of Action
The mechanism by which dexrazoxane exerts its cytoprotective activity is not fully understood. Dexrazoxane is a cyclic derivative of EDTA that penetrates cell membranes. Results of laboratory studies suggest that dexrazoxane is converted intracellularly to a ring-opened chelating agent that interferes with iron-mediated free radical generation thought to be responsible, in part, for anthracycline-induced cardiomyopathy.
5.5 Embryo Fetal Toxicity (5.5 Embryo-Fetal Toxicity)
Based on finding from animal studies and its mechanism of action, dexrazoxane can cause fetal harm when administered to pregnant women [see Clinical Pharmacology (12.1)]. In animal reproduction studies, dexrazoxane administration during the period of organogenesis resulted in maternal toxicity and teratogenicity in rats and rabbits at doses significantly lower than the clinically recommended dose.
Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with dexrazoxane and for 6 months after the last dose. Advise male patients with female partners of reproductive potential to use effective contraception during treatment with dexrazoxane and for 3 months after the last dose [see Use in Specific Populations (8.1, 8.3)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
5.4 Secondary Malignancies
Secondary malignancies such as acute myeloid leukemia (AML) and myelodysplastic syndrome (MDS) have been reported in studies of pediatric patients who have received dexrazoxane in combination with chemotherapy. Dexrazoxane is not indicated for use in pediatric patients. Some adult patients who received dexrazoxane in combination with anti-cancer agents known to be carcinogenic have also developed secondary malignancies, including AML and MDS.
Razoxane is the racemic mixture, of which dexrazoxane is the S(+)-enantiomer. Secondary malignancies (primarily acute myeloid leukemia) have been reported in patients treated chronically with oral razoxane. In these patients, the total cumulative dose of razoxane ranged from 26 to 480 grams and the duration of treatment was from 42 to 319 weeks. One case of T-cell lymphoma, one case of B-cell lymphoma, and six to eight cases of cutaneous basal cell or squamous cell carcinoma have also been reported in patients treated with razoxane. Long-term administration of razoxane to rodents was associated with the development of malignancies [see Nonclinical Toxicology (13.1)].
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Reconstitute vial contents and dilute before use. (2.3)
- Administer dexrazoxane for injection by intravenous infusion over 15 minutes.
- DO NOT ADMINISTER VIA AN INTRAVENOUS PUSH. (2.1, 2.3)
- The recommended dosage ratio of dexrazoxane for injection to doxorubicin is 10:1 (e.g., 500 mg/m2 dexrazoxane for injection to 50 mg/m2 doxorubicin). Do not administer doxorubicin before dexrazoxane for injection. (2.1)
- Reduce dose by 50% for patients with creatinine clearance <40 mL/min. (2.2, 8.6)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
For injection: 250 mg or 500 mg of dexrazoxane as a sterile, off white to pale yellow lyophilized powder in a single-dose vials for reconstitution.
5.2 Concomitant Chemotherapy
Only use dexrazoxane in those patients who have received a cumulative doxorubicin dose of 300 mg/m2 and are continuing with doxorubicin therapy. Do not use with chemotherapy initiation as dexrazoxane may interfere with the antitumor activity of the chemotherapy regimen. In a trial conducted in patients with metastatic breast cancer who were treated with fluorouracil, doxorubicin, and cyclophosphamide (FAC) with or without dexrazoxane starting with their first cycle of FAC therapy, patients who were randomized to receive dexrazoxane had a lower response rate (48% vs. 63%) and shorter time to progression than patients who were randomized to receive placebo.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Lactation: Advise not to breastfeed. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reaction rates observed cannot be directly compared to rates in other trials and may not reflect the rates observed in clinical practice.
The adverse reaction profile described in this section was identified from randomized, placebo- controlled, double-blind studies in patients with metastatic breast cancer who received the combination of the FAC chemotherapy regimen with or without dexrazoxane. The dose of doxorubicin was 50 mg/m2 in each of these trials. Treatment was administered every three weeks until disease progression or cardiac toxicity.
Patients in clinical trials who received FAC with dexrazoxane experienced more severe leukopenia, granulocytopenia, and thrombocytopenia than patients receiving FAC without dexrazoxane [see Warnings and Precautions (5.1)].
Table 1 below lists the incidence of adverse reactions for patients receiving FAC with either dexrazoxane or placebo in the breast cancer studies. Adverse experiences occurring during courses 1 through 6 are displayed for patients receiving dexrazoxane or placebo with FAC beginning with their first course of therapy (columns 1 and 3, respectively). Adverse experiences occurring at course 7 and beyond for patients who received placebo with FAC during the first six courses and who then received either dexrazoxane or placebo with FAC are also displayed (columns 2 and 4, respectively).
The adverse reactions listed below in Table 1 demonstrate that the frequency of adverse reaction “Pain on Injection” has been greater for dexrazoxane arm, as compared to placebo.
Table 1
| Adverse Reaction |
Percentage (%) of Breast Cancer Patients With Adverse Reaction |
|||
|---|---|---|---|---|
| FAC + Dexrazoxane |
FAC + Placebo |
|||
| Courses 1-6 N = 413 |
Courses ≥ 7 N = 102 |
Courses 1-6 N = 458 |
Courses ≥ 7 N = 99 |
|
| Alopecia |
94 |
100 |
97 |
98 |
| Nausea |
77 |
51 |
84 |
60 |
| Vomiting |
59 |
42 |
72 |
49 |
| Fatigue/Malaise |
61 |
48 |
58 |
55 |
| Anorexia |
42 |
27 |
47 |
38 |
| Stomatitis |
34 |
26 |
41 |
28 |
| Fever |
34 |
22 |
29 |
18 |
| Infection |
23 |
19 |
18 |
21 |
| Diarrhea |
21 |
14 |
24 |
7 |
| Pain on Injection |
12 |
13 |
3 |
0 |
| Sepsis |
17 |
12 |
14 |
9 |
| Neurotoxicity |
17 |
10 |
13 |
5 |
| Streaking/Erythema |
5 |
4 |
4 |
2 |
| Phlebitis |
6 |
3 |
3 |
5 |
| Esophagitis |
6 |
3 |
7 |
4 |
| Dysphagia |
8 |
0 |
10 |
5 |
| Hemorrhage |
2 |
3 |
2 |
1 |
| Extravasation |
1 |
3 |
1 |
2 |
| Urticaria |
2 |
2 |
2 |
0 |
| Recall Skin Reaction |
1 |
1 |
2 |
0 |
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Myelosuppression
Treatment with dexrazoxane is associated with leukopenia, neutropenia, and thrombocytopenia. Perform hematological monitoring [see Warnings and Precautions (5.1)].
Embryo-Fetal Toxicity
Advise female patients of the potential risk to a fetus and to inform their healthcare provider of a known or suspected pregnancy. Advise females of reproductive potential to use effective contraception during treatment with dexrazoxane and for 6 months after the last dose. Advise male patients with female partners of reproductive potential to use effective contraception during treatment with dexrazoxane and for 3 months after the last dose [seeWarnings and Precautions (5.5) and Use in Specific Populations (8.1, 8.3)].
Lactation
Advise women not to breastfeed during treatment with dexrazoxane and for 2 weeks after the last dose [see Use in Specific Populations (8.2)].
Infertility
Advise males of reproductive potential that dexrazoxane may impair fertility [see Use in Specific Populations (8.3)].
This product’s label may have been updated. For current full prescribing information, please visit www.msnlabs.com.
Manufactured by:
MSN Laboratories Private Limited
Telangana – 509 228,
INDIA
Distributed by:
Novadoz Pharmaceuticals LLC
Piscataway, NJ 08854 -3714
Revised: 07/2025
2.3 Preparation and Administration
Preparation and Handling of Infusion Solution
Reconstitute dexrazoxane for injection with Sterile Water for Injection, USP. Reconstitute with 25 mL for a dexrazoxane for injection 250 mg vial and 50 mL for a dexrazoxane for injection 500 mg vial to give a concentration of 10 mg/mL. Dilute the reconstituted solution further with Lactated Ringer’s Injection, USP to a concentration of 1.3 to 3.0 mg/mL in intravenous infusion bags for intravenous infusion.
Following reconstitution with Sterile Water for Injection, USP, dexrazoxane for injection is stable for 30 minutes at room temperature or if storage is necessary, up to 3 hours from the time of reconstitution when stored under refrigeration, 2° to 8°C (36° to 46°F). The pH of the resultant solution is 1.0 to 3.0. DISCARD UNUSED SOLUTIONS. The diluted infusion solutions are stable for one hour at room temperature or if storage is necessary, up to 4 hours when stored under refrigeration, 2° to 8°C (36° to 46°F). The infusion solutions have a pH of 3.5 to 5.5. DISCARD UNUSED SOLUTIONS.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Solutions containing a precipitate should be discarded.
Use caution when handling and preparing the reconstituted solution. The use of gloves is recommended. If dexrazoxane for injection powder or solutions contact the skin or mucosae, wash exposed area immediately and thoroughly with soap and water. Follow special handling and disposal procedures.1
Administration
Do not mix dexrazoxane for injection with other drugs.
Administer the final diluted solution of dexrazoxane for injection by intravenous infusion over 15 minutes before the administration of doxorubicin. DO NOT ADMINISTER VIA AN INTRAVENOUS PUSH. Administer doxorubicin within 30 minutes after the completion of dexrazoxane for injection infusion.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Dexrazoxane for injection is available in the following strengths as a sterile, off white to pale yellow pyrogen-free lyophilized powder in a single-dose vials for reconstitution.
| Strength
|
Pack Style
|
NDC Code
|
| 250 mg |
Carton of single-dose vial with a blue flip-top seal |
72205-247-01 |
| 500 mg |
Carton of single-dose vial with a blue flip-top seal |
72205-248-01 |
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Follow special handling and disposal procedures.1
Discard unused solutions.
Package Label.principal Display Panel (PACKAGE LABEL.PRINCIPAL DISPLAY PANEL)
Dexrazoxane for injection 250 mg/Vial Carton Label
Dexrazoxane for injection 250 mg/Vial Vial Label
Dexrazoxane for injection 500 mg/Vial Carton Label
Dexrazoxane for injection 500 mg/Vial Vial Label
8.3 Females and Males of Reproductive Potential
Dexrazoxane can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)].
Pregnancy Testing
Verify pregnancy status in females of reproductive potential prior to starting treatment with dexrazoxane.
Contraception
Females
Advise females of reproductive potential to use effective contraception during treatment with dexrazoxane and for 6 months after the last dose. Males Because of the potential for genotoxicity, advise male patients with female partners of reproductive potential to use effective contraception during treatment with dexrazoxane and for 3 months after the last dose [see Nonclinical Toxicology (13.1)].
Infertility Based on findings from animal studies, dexrazoxane may impair male fertility [see Nonclinical Toxicology (13.1)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term carcinogenicity studies have been carried out with dexrazoxane in animals. Nevertheless, a study by the National Cancer Institute has reported that long-term dosing with razoxane (the racemic mixture of dexrazoxane, ICRF-187, and its enantiomer ICRF-186) is associated with the development of malignancies in rats and possibly in mice [see Warnings and Precautions (5.4)].
Dexrazoxane was not mutagenic in the bacterial reverse mutation (Ames) test, but was found to be clastogenic to human lymphocytes in vitro and to mouse bone marrow erythrocytes in vivo (micronucleus test).
Dexrazoxane has the potential to impair fertility in male patients based on effects in repeat-dose toxicology studies. Testicular atrophy was seen with dexrazoxane administration at doses as low as 30 mg/kg weekly for 6 weeks in rats (1/3 the human dose on a mg/m2 basis) and as low as 20 mg/kg weekly for 13 weeks in dogs (approximately equal to the human dose on a mg/m2 basis).
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:44.482747 · Updated: 2026-03-14T22:30:44.173822