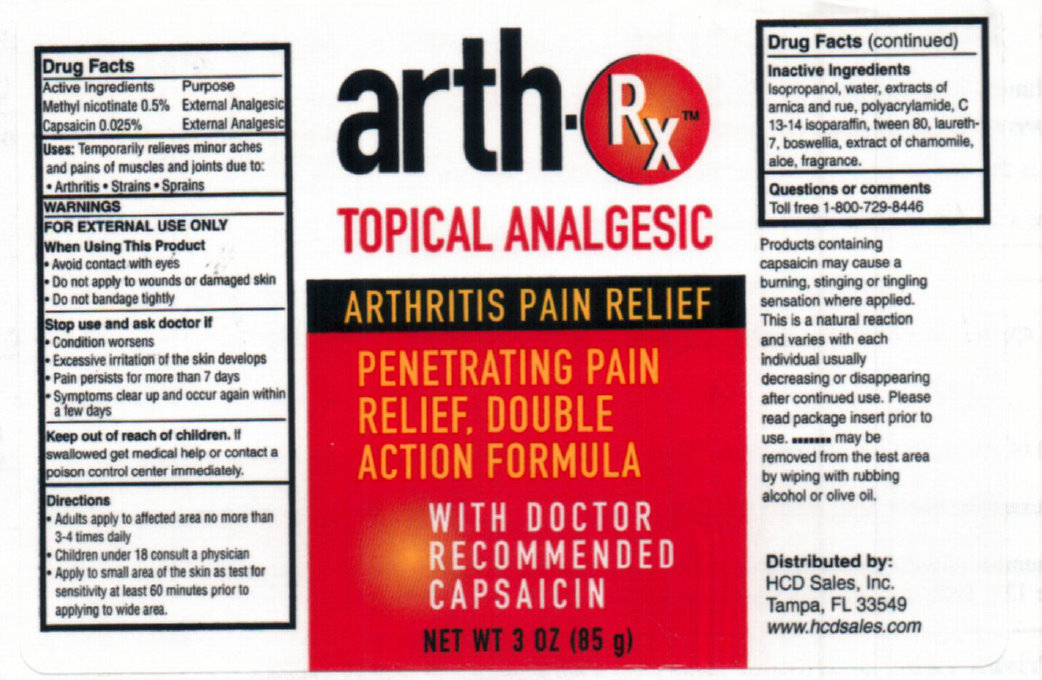
Arth Rx Topical Analgesic
a7b3b9b4-b1c4-4e51-8e3a-c84f64c75609
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active Ingredients Purpose Methyl nicotinate 0.5% External Analgesic Capsaicin 0.025 % External Analgesic
Medication Information
Warnings and Precautions
WARNINGS
FOR EXTERNAL USE ONLY
Indications and Usage
When Using This Product
- Avoid contact with eyes - Do not apply to wounds or damaged skin - Do not bandage tightly
Dosage and Administration
Directions - Adults apply to affected area no more than 3-4 times daily - Children under 18 consult a physician - Apply to small area of the skin as test for sensitivity at least 60 minutes prior to applying to wide area.
Description
Products containing capsaicin may cause a burning, stinging or tingling sensation where applied. This is a natural reaction and varies with each individual usually decreasing or disappearing after continued use. Please read package insert prior to use. .......... may be removed from the test area by wiping or olive oil.
Distributed by: HCD sales, Inc, Tampa, FL 33549 www.hcdsales.com
Section 50565-1
Keep out of reach of children. If swallowed get medical help or contact a poison control center immediately.
Section 50566-9
Stop use and ask doctor if
- Condition worsens - Excessive irritation of the skin develops - Pain persists for more than 7 days - Symptoms clear up and occur again within a few days
Section 51727-6
Inactive Ingredients
Isopropanol, water, extracts of arnica and rue, polyacrylamide, C 13-14 isoparaffin. tween 80, laureth-7, boswellia, extract of chamomile, aloe, fragrance.
Section 51945-4
arth.Rx TOPICAL ANALGESIC ARTHRITIS PAIN RELIEF PENETRATING PAIN RELIEF, DOUBLE ACTION FORMULA WITH DOCTOR RECOMMENDED CAPSAICIN NEW WT 3 OZ (85 g)
Section 53413-1
Questions or comments
Toll free 1-800-729-8446
Section 55105-1
Uses: Temporarily relieves minor aches and pains of muscles and joints due to:
- Arthritis - Strains - Sprains
Section 55106-9
Active Ingredients Purpose Methyl nicotinate 0.5% External Analgesic Capsaicin 0.025 % External Analgesic
Structured Label Content
Indications and Usage (34067-9)
When Using This Product
- Avoid contact with eyes - Do not apply to wounds or damaged skin - Do not bandage tightly
Dosage and Administration (34068-7)
Directions - Adults apply to affected area no more than 3-4 times daily - Children under 18 consult a physician - Apply to small area of the skin as test for sensitivity at least 60 minutes prior to applying to wide area.
Warnings and Precautions (34071-1)
WARNINGS
FOR EXTERNAL USE ONLY
Description (34089-3)
Products containing capsaicin may cause a burning, stinging or tingling sensation where applied. This is a natural reaction and varies with each individual usually decreasing or disappearing after continued use. Please read package insert prior to use. .......... may be removed from the test area by wiping or olive oil.
Distributed by: HCD sales, Inc, Tampa, FL 33549 www.hcdsales.com
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed get medical help or contact a poison control center immediately.
Section 50566-9 (50566-9)
Stop use and ask doctor if
- Condition worsens - Excessive irritation of the skin develops - Pain persists for more than 7 days - Symptoms clear up and occur again within a few days
Section 51727-6 (51727-6)
Inactive Ingredients
Isopropanol, water, extracts of arnica and rue, polyacrylamide, C 13-14 isoparaffin. tween 80, laureth-7, boswellia, extract of chamomile, aloe, fragrance.
Section 51945-4 (51945-4)
arth.Rx TOPICAL ANALGESIC ARTHRITIS PAIN RELIEF PENETRATING PAIN RELIEF, DOUBLE ACTION FORMULA WITH DOCTOR RECOMMENDED CAPSAICIN NEW WT 3 OZ (85 g)
Section 53413-1 (53413-1)
Questions or comments
Toll free 1-800-729-8446
Section 55105-1 (55105-1)
Uses: Temporarily relieves minor aches and pains of muscles and joints due to:
- Arthritis - Strains - Sprains
Section 55106-9 (55106-9)
Active Ingredients Purpose Methyl nicotinate 0.5% External Analgesic Capsaicin 0.025 % External Analgesic
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:14.287862 · Updated: 2026-03-14T23:01:41.296752