Venipuncture Px1
a76e1cee-f89e-481e-adcc-0a4e3414a8be
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Rx only
Indications and Usage
Stop use and ask a doctor if: Irritation or redness develops condition persists for more than 72 hours Cleansing of an injection site
Dosage and Administration
When Lidocaine HCl 2% Jelly is used concomitantly with other products containing lidocaine, the total dose contributed by all formulations must be kept in mind. The dosage varies and depends upon the area to be anesthetized, vascularity of the tissues, individual tolerance, and the technique of anesthesia. The lowest dosage needed to provide effective anesthesia should be administered. Dosages should be reduced for children and for elderly and debilitated patients. Although the incidence of adverse effects with Lidocaine HCl 2% Jelly is quite low, caution should be exercised, particularly when employing large amounts, since the incidence of adverse effects is directly proportional to the total dose of local anesthetic agent administered.
Contraindications
Lidocaine is contraindicated in patients with a known history of hypersensitivity to local anesthetics of the amide type or to other components of Lidocaine HCl 2% Jelly.
Adverse Reactions
Adverse experiences following the administration of lidocaine are similar in nature to those observed in other amide local anesthetic agents. These adverse experiences are, in general, dose-related and may result from high plasma levels caused by excessive dosage or rapid absorption, or may result from a hypersensitivity, idiosyncrasy, or diminished tolerance on the part of the patient. Serious adverse experiences are generally systemic in nature. The following types are those most commonly reported: There have been rare reports of endotracheal tube occlusion associated with the presence of dried jelly residue in the inner lumen of the tube. (See also WARNINGS and DOSAGE AND ADMINISTRATION .)
How Supplied
Lidocaine HCI 2% Jelly is supplied in the listed dosage forms. NDC 17478-840-30 30 mL aluminum tube NDC 17478-840-05 5 mL aluminum tube A detachable applicator cone and a key for expressing the contents are included in the 30 mL carton.
Medication Information
Indications and Usage
Lidocaine HCl 2% Jelly is indicated for prevention and control of pain in procedures involving the male and female urethra, for topical treatment of painful urethritis, and as an anesthetic lubricant for endotracheal intubation (oral and nasal).
Dosage and Administration
When Lidocaine HCl 2% Jelly is used concomitantly with other products containing lidocaine, the total dose contributed by all formulations must be kept in mind.
The dosage varies and depends upon the area to be anesthetized, vascularity of the tissues, individual tolerance, and the technique of anesthesia. The lowest dosage needed to provide effective anesthesia should be administered. Dosages should be reduced for children and for elderly and debilitated patients. Although the incidence of adverse effects with Lidocaine HCl 2% Jelly is quite low, caution should be exercised, particularly when employing large amounts, since the incidence of adverse effects is directly proportional to the total dose of local anesthetic agent administered.
Contraindications
Lidocaine is contraindicated in patients with a known history of hypersensitivity to local anesthetics of the amide type or to other components of Lidocaine HCl 2% Jelly.
Adverse Reactions
Adverse experiences following the administration of lidocaine are similar in nature to those observed in other amide local anesthetic agents. These adverse experiences are, in general, dose-related and may result from high plasma levels caused by excessive dosage or rapid absorption, or may result from a hypersensitivity, idiosyncrasy, or diminished tolerance on the part of the patient. Serious adverse experiences are generally systemic in nature. The following types are those most commonly reported:
There have been rare reports of endotracheal tube occlusion associated with the presence of dried jelly residue in the inner lumen of the tube. (See also WARNINGS and DOSAGE AND ADMINISTRATION.)
How Supplied
Lidocaine HCI 2% Jelly is supplied in the listed dosage forms.
NDC 17478-840-30 30 mL aluminum tube
NDC 17478-840-05 5 mL aluminum tube
A detachable applicator cone and a key for expressing the contents are included in the 30 mL carton.
Description
Lidocaine HCl 2% Jelly is a sterile, aqueous product that contains a local anesthetic agent and is administered topically. (See INDICATIONS for specific uses.)
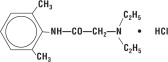
Lidocaine HCl 2% Jelly contains lidocaine HCl which is chemically designated as acetamide, 2-(diethylamino)-N-(2,6-dimethylphenyl)-, monohydrochloride and has the following structural formula:
Its molecular formula is C14H22N2O • HCl and its molecular weight is 270.80.
Lidocaine HCl 2% Jelly also contains hypromellose, and the resulting mixture maximizes contact with mucosa and provides lubrication for instrumentation. The unused portion should be discarded after initial use.
Composition of Lidocaine HCl 2% Jelly 30 mL and 5 mL tubes: Each mL contains 20 mg of lidocaine HCl. The formulation also contains methylparaben, propylparaben, hypromellose, and sodium hydroxide and/or hydrochloric acid to adjust pH between 6.0 to 7.0.
Section 34072-9
General: The safety and effectiveness of lidocaine depend on proper dosage, correct technique, adequate precautions, and readiness for emergencies. (See WARNINGS and ADVERSE REACTIONS.) The lowest dosage that results in effective anesthesia should be used to avoid high plasma levels and serious adverse effects. Repeated doses of lidocaine may cause significant increases in blood levels with each repeated dose because of slow accumulation of the drug or its metabolites. Tolerance to elevated blood levels varies with the status of the patient. Debilitated, elderly patients, acutely ill patients, and children should be given reduced doses commensurate with their age and physical status. Lidocaine should also be used with caution in patients with severe shock or heart block.
Lidocaine HCl 2% Jelly should be used with caution in patients with known drug sensitivities. Patients allergic to para-aminobenzoic acid derivatives (procaine, tetracaine, benzocaine, etc.) have not shown cross sensitivity to lidocaine.
Many drugs used during the conduct of anesthesia are considered potential triggering agents for familial malignant hyperthermia. Since it is not known whether amide-type local anesthetics may trigger this reaction and since the need for supplemental general anesthesia cannot be predicted in advance, it is suggested that a standard protocol for management should be available. Early unexplained signs of tachycardia, tachypnea, labile blood pressure, and metabolic acidosis may precede temperature elevation. Successful outcome is dependent on early diagnosis, prompt discontinuance of the suspect triggering agent(s) and institution of treatment, including oxygen therapy, indicated supportive measures and dantrolene (consult dantrolene sodium intravenous package insert before using).
Section 34076-0
Information for Patients: When topical anesthetics are used in the mouth, the patient should be aware that the production of topical anesthesia may impair swallowing and thus enhance the danger of aspiration. For this reason, food should not be ingested for 60 minutes following use of local anesthetic preparations in the mouth or throat area. This is particularly important in children because of their frequency of eating.
Numbness of the tongue or buccal mucosa may enhance the danger of unintentional biting trauma. Food or chewing gum should not be taken while the mouth or throat area is anesthetized.
Section 34079-4
Labor and Delivery: Lidocaine is not contraindicated in labor and delivery. Should Lidocaine HCl 2% Jelly be used concomitantly with other products containing lidocaine, the total dose contributed by all formulations must be kept in mind.
Section 34080-2
Nursing Mothers: Lidocaine is secreted in human milk. The clinical significance of this observation is unknown. Caution should be exercised when lidocaine is administered to a nursing woman.
Section 34081-0
Pediatric Use: Although, the safety and effectiveness of Lidocaine 2% Jelly in pediatric patients have not been established, a study of 19 premature neonates (gestational age <33 weeks) found no correlation between the plasma concentration of lidocaine or monoethylglycinexylidide and infant body weight when moderate amounts of lidocaine (i.e. 0.3 mL/kg of lidocaine gel 20 mg/mL) were used for lubricating both intranasal and endotracheal tubes. No neonate had plasma levels of lidocaine above 750 mcg/L. Dosages in children should be reduced, commensurate with age, body weight, and physical condition. (See DOSAGE AND ADMINISTRATION.)
Section 42229-5
Rx only
Section 43679-0
Mechanism of Action: Lidocaine stabilizes the neuronal membrane by inhibiting the ionic fluxes required for the initiation and conduction of impulses, thereby effecting local anesthetic action.
Section 43682-4
Pharmacokinetics and Metabolism: Lidocaine may be absorbed following topical administration to mucous membranes, its rate and extent of absorption depending upon concentration and total dose administered, the specific site of application, and duration of exposure. In general, the rate of absorption of local anesthetic agents following topical application occurs most rapidly after intratracheal administration. Lidocaine is also well-absorbed from the gastrointestinal tract, but little intact drug may appear in the circulation because of biotransformation in the liver.
Lidocaine is metabolized rapidly by the liver and metabolites and unchanged drug are excreted by the kidneys. Biotransformation includes oxidative N-dealkylation, ring hydroxylation, cleavage of the amide linkage, and conjugation. N-dealkylation, a major pathway of biotransformation, yields the metabolites monoethylglycinexylidide and glycinexylidide. The pharmacological/toxicological actions of these metabolites are similar to, but less potent than, those of lidocaine. Approximately 90% of lidocaine administered is excreted in the form of various metabolites, and less than 10% is excreted unchanged. The primary metabolite in urine is a conjugate of 4-hydroxy-2, 6-dimethylaniline.
The plasma binding of lidocaine is dependent on drug concentration, and the fraction bound decreases with increasing concentration. At concentrations of 1 to 4 mcg of free base per mL 60 to 80 percent of lidocaine is protein bound. Binding is also dependent on the plasma concentration of the alpha-l-acid glycoprotein.
Lidocaine crosses the blood-brain and placental barriers, presumably by passive diffusion.
Studies of lidocaine metabolism following intravenous bolus injections have shown that the elimination half-life of this agent is typically 1.5 to 2.0 hours. Because of the rapid rate at which lidocaine is metabolized, any condition that effects liver function may alter lidocaine kinetics. The half-life may be prolonged twofold or more in patients with liver dysfunction. Renal dysfunction does not affect lidocaine kinetics but may increase the accumulation of metabolites.
Factors such as acidosis and the use of CNS stimulants and depressants affect the CNS levels of lidocaine required to produce overt systemic effects. Objective adverse manifestations become increasingly apparent with increasing venous plasma levels above 6 mcg free base per mL. In the rhesus monkey arterial blood levels of 18 to 21 mcg/mL have been shown to be threshold for convulsive activity.
Section 44425-7
Storage: Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
PREMIERProRx®
Manufactured by:
Akorn, Inc.
Lake Forest, IL 60045
PremierProRx® is a registered trademark of Premier Inc., used under license.
PLD00N Rev. 10/15
Section 55106-9
Active Ingredient Purpose
Isopropyl Alcohol 70% v/v Antiseptic
Warnings
EXCESSIVE DOSAGE, OR SHORT INTERVALS BETWEEN DOSES, CAN RESULT IN HIGH PLASMA LEVELS AND SERIOUS ADVERSE EFFECTS. PATIENTS SHOULD BE INSTRUCTED TO STRICTLY ADHERE TO THE RECOMMENDED DOSAGE AND ADMINISTRATION GUIDELINES AS SET FORTH IN THIS PACKAGE INSERT. THE MANAGEMENT OF SERIOUS ADVERSE REACTIONS MAY REQUIRE THE USE OF RESUSCITATIVE EQUIPMENT, OXYGEN AND OTHER RESUSCITATIVE DRUGS.
Lidocaine HCl 2% Jelly should be used in extreme caution in the presence of sepsis or severely traumatized mucosa in the area of application since under such conditions there is the potential for rapid systemic absorption.
When used for endotracheal tube lubrication care should be taken to avoid introducing the product into the lumen of the tube. Do not use the jelly to lubricate the endotracheal stylettes. If allowed into the inner lumen, the jelly may dry on the inner surface leaving a residue which tends to clump with flexion, narrowing the lumen. There have been rare reports in which this residue has caused the lumen to occlude. (See also ADVERSE REACTIONS and DOSAGE AND ADMINISTRATION.)
Warnings:
- For external use only
- Flammable, keep away from flame or fire
- Not for use with electrocautinary devices or procedures
- Do not use in eyes
- Sterile unless package is damaged or open.
Overdosage
Acute emergencies from local anesthetics are generally related to high plasma levels encountered during therapeutic use of local anesthetics. (See ADVERSE REACTIONS, WARNINGS, and PRECAUTIONS.)
Directions:
Wipe injection site vigorously and discard
Precautions
Maximum Dosage
No more than 600 mg of lidocaine HCl should be given in any 12 hour period.
Venipuncture Px1
Contents
1- Bio-Hazard Specimen Transport Bag
1-Pair of Sterile Vinyl Exam Gloves (Medium), Latex Free
2-Sheer Adhesive Bandages, Spots
1- Basic conforming Stretch Gauze, Non-sterile
1- Cohesive Wrap Bandage
2- Alcohol Prep Pads
1- Tourniquet, Latex free
1- Vacutainer Single Use Holder
1- Vacutainer Blood Collection Needle , Butterfly W/7"
Tubing, 23GX 3/4"
1- Vacutainer Eclipse Blood Collection Needle 21G X 1.25
3-Red Vacutainer SST Blood collection Tubes, 8.5 mL
1-Lavender Vacutainer EDTA Blood collection Tube 4.0 mL
1- 2% Lidocaine Hydrochloride Jelly, USP- 5.0 mL
Use: Alcohol Prep
For preparation of the skin prior to injection.
Other Information:
- Store at room temperature: 15 deg C to 30 deg C 59 deg F to 86 deg F
- avoid excessive heat
Inactive Ingredient
Inactive Ingredient
- Water
Packaging Kit Label
Clinical Pharmacology
Indications and Usage:
Stop use and ask a doctor if:
- Irritation or redness develops
- condition persists for more than 72 hours
- Cleansing of an injection site
Keep Out of Reach of Children.
In case of accidental ingestion, seek professional assistance or consult a poison control center immediately.
Packaging Kit Components Labeling
Teratogenic Effects: Pregnancy Category B.
Reproduction studies for lidocaine have been performed in both rats and rabbits. There was no evidence of harm to the fetus at subcutaneous doses of up to 50 mg/kg lidocaine (300 mg/m2 on a body surface area basis) in the rat model. In the rabbit model, there was no evidence of harm to the fetus at a dose of 5 mg/kg, s.c. (60 mg/m2 on a body surface area basis). Treatment of rabbits with 25 mg/kg (300 mg/m2) produced evidence of maternal toxicity and evidence of delayed fetal development, including a non-significant decrease in fetal weight (7%) and an increase in minor skeletal anomalies (skull and sternebral defect, reduced ossification of the phalanges). The effect of lidocaine on post-natal development was examined in rats by treating pregnant female rats daily subcutaneously at doses of 2, 10, and 50 mg/kg (12, 60, and 300 mg/m2) from day 15 of pregnancy and up to 20 days post partum. No signs of adverse effects were seen either in dams or in the pups up to and including the dose of 10 mg/kg (60 mg/m2); however, the number of surviving pups was reduced at 50 mg/kg (300 mg/m2), both at birth and the duration of lactation period, the effect most likely being secondary to maternal toxicity. No other effects on litter size, litter weight, abnormalities in the pups and physical developments of the pups were seen in this study.
A second study examined the effects of lidocaine on post-natal development in the rat that included assessment of the pups from weaning to sexual maturity. Rats were treated for 8 months with 10 or 30 mg/kg, s.c. lidocaine (60 mg/m2 and 180 mg/m2 on a body surface area basis, respectively). This time period encompassed 3 mating periods. There was no evidence of altered post-natal development in any offspring; however, both doses of lidocaine significantly reduced the average number of pups per litter surviving until weaning of offspring from the first 2 mating period.
There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Structured Label Content
Section 34072-9 (34072-9)
General: The safety and effectiveness of lidocaine depend on proper dosage, correct technique, adequate precautions, and readiness for emergencies. (See WARNINGS and ADVERSE REACTIONS.) The lowest dosage that results in effective anesthesia should be used to avoid high plasma levels and serious adverse effects. Repeated doses of lidocaine may cause significant increases in blood levels with each repeated dose because of slow accumulation of the drug or its metabolites. Tolerance to elevated blood levels varies with the status of the patient. Debilitated, elderly patients, acutely ill patients, and children should be given reduced doses commensurate with their age and physical status. Lidocaine should also be used with caution in patients with severe shock or heart block.
Lidocaine HCl 2% Jelly should be used with caution in patients with known drug sensitivities. Patients allergic to para-aminobenzoic acid derivatives (procaine, tetracaine, benzocaine, etc.) have not shown cross sensitivity to lidocaine.
Many drugs used during the conduct of anesthesia are considered potential triggering agents for familial malignant hyperthermia. Since it is not known whether amide-type local anesthetics may trigger this reaction and since the need for supplemental general anesthesia cannot be predicted in advance, it is suggested that a standard protocol for management should be available. Early unexplained signs of tachycardia, tachypnea, labile blood pressure, and metabolic acidosis may precede temperature elevation. Successful outcome is dependent on early diagnosis, prompt discontinuance of the suspect triggering agent(s) and institution of treatment, including oxygen therapy, indicated supportive measures and dantrolene (consult dantrolene sodium intravenous package insert before using).
Section 34076-0 (34076-0)
Information for Patients: When topical anesthetics are used in the mouth, the patient should be aware that the production of topical anesthesia may impair swallowing and thus enhance the danger of aspiration. For this reason, food should not be ingested for 60 minutes following use of local anesthetic preparations in the mouth or throat area. This is particularly important in children because of their frequency of eating.
Numbness of the tongue or buccal mucosa may enhance the danger of unintentional biting trauma. Food or chewing gum should not be taken while the mouth or throat area is anesthetized.
Section 34079-4 (34079-4)
Labor and Delivery: Lidocaine is not contraindicated in labor and delivery. Should Lidocaine HCl 2% Jelly be used concomitantly with other products containing lidocaine, the total dose contributed by all formulations must be kept in mind.
Section 34080-2 (34080-2)
Nursing Mothers: Lidocaine is secreted in human milk. The clinical significance of this observation is unknown. Caution should be exercised when lidocaine is administered to a nursing woman.
Section 34081-0 (34081-0)
Pediatric Use: Although, the safety and effectiveness of Lidocaine 2% Jelly in pediatric patients have not been established, a study of 19 premature neonates (gestational age <33 weeks) found no correlation between the plasma concentration of lidocaine or monoethylglycinexylidide and infant body weight when moderate amounts of lidocaine (i.e. 0.3 mL/kg of lidocaine gel 20 mg/mL) were used for lubricating both intranasal and endotracheal tubes. No neonate had plasma levels of lidocaine above 750 mcg/L. Dosages in children should be reduced, commensurate with age, body weight, and physical condition. (See DOSAGE AND ADMINISTRATION.)
Section 42229-5 (42229-5)
Rx only
Section 43679-0 (43679-0)
Mechanism of Action: Lidocaine stabilizes the neuronal membrane by inhibiting the ionic fluxes required for the initiation and conduction of impulses, thereby effecting local anesthetic action.
Section 43682-4 (43682-4)
Pharmacokinetics and Metabolism: Lidocaine may be absorbed following topical administration to mucous membranes, its rate and extent of absorption depending upon concentration and total dose administered, the specific site of application, and duration of exposure. In general, the rate of absorption of local anesthetic agents following topical application occurs most rapidly after intratracheal administration. Lidocaine is also well-absorbed from the gastrointestinal tract, but little intact drug may appear in the circulation because of biotransformation in the liver.
Lidocaine is metabolized rapidly by the liver and metabolites and unchanged drug are excreted by the kidneys. Biotransformation includes oxidative N-dealkylation, ring hydroxylation, cleavage of the amide linkage, and conjugation. N-dealkylation, a major pathway of biotransformation, yields the metabolites monoethylglycinexylidide and glycinexylidide. The pharmacological/toxicological actions of these metabolites are similar to, but less potent than, those of lidocaine. Approximately 90% of lidocaine administered is excreted in the form of various metabolites, and less than 10% is excreted unchanged. The primary metabolite in urine is a conjugate of 4-hydroxy-2, 6-dimethylaniline.
The plasma binding of lidocaine is dependent on drug concentration, and the fraction bound decreases with increasing concentration. At concentrations of 1 to 4 mcg of free base per mL 60 to 80 percent of lidocaine is protein bound. Binding is also dependent on the plasma concentration of the alpha-l-acid glycoprotein.
Lidocaine crosses the blood-brain and placental barriers, presumably by passive diffusion.
Studies of lidocaine metabolism following intravenous bolus injections have shown that the elimination half-life of this agent is typically 1.5 to 2.0 hours. Because of the rapid rate at which lidocaine is metabolized, any condition that effects liver function may alter lidocaine kinetics. The half-life may be prolonged twofold or more in patients with liver dysfunction. Renal dysfunction does not affect lidocaine kinetics but may increase the accumulation of metabolites.
Factors such as acidosis and the use of CNS stimulants and depressants affect the CNS levels of lidocaine required to produce overt systemic effects. Objective adverse manifestations become increasingly apparent with increasing venous plasma levels above 6 mcg free base per mL. In the rhesus monkey arterial blood levels of 18 to 21 mcg/mL have been shown to be threshold for convulsive activity.
Section 44425-7 (44425-7)
Storage: Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
PREMIERProRx®
Manufactured by:
Akorn, Inc.
Lake Forest, IL 60045
PremierProRx® is a registered trademark of Premier Inc., used under license.
PLD00N Rev. 10/15
Section 55106-9 (55106-9)
Active Ingredient Purpose
Isopropyl Alcohol 70% v/v Antiseptic
Warnings (WARNINGS)
EXCESSIVE DOSAGE, OR SHORT INTERVALS BETWEEN DOSES, CAN RESULT IN HIGH PLASMA LEVELS AND SERIOUS ADVERSE EFFECTS. PATIENTS SHOULD BE INSTRUCTED TO STRICTLY ADHERE TO THE RECOMMENDED DOSAGE AND ADMINISTRATION GUIDELINES AS SET FORTH IN THIS PACKAGE INSERT. THE MANAGEMENT OF SERIOUS ADVERSE REACTIONS MAY REQUIRE THE USE OF RESUSCITATIVE EQUIPMENT, OXYGEN AND OTHER RESUSCITATIVE DRUGS.
Lidocaine HCl 2% Jelly should be used in extreme caution in the presence of sepsis or severely traumatized mucosa in the area of application since under such conditions there is the potential for rapid systemic absorption.
When used for endotracheal tube lubrication care should be taken to avoid introducing the product into the lumen of the tube. Do not use the jelly to lubricate the endotracheal stylettes. If allowed into the inner lumen, the jelly may dry on the inner surface leaving a residue which tends to clump with flexion, narrowing the lumen. There have been rare reports in which this residue has caused the lumen to occlude. (See also ADVERSE REACTIONS and DOSAGE AND ADMINISTRATION.)
Warnings:
- For external use only
- Flammable, keep away from flame or fire
- Not for use with electrocautinary devices or procedures
- Do not use in eyes
- Sterile unless package is damaged or open.
Overdosage (OVERDOSAGE)
Acute emergencies from local anesthetics are generally related to high plasma levels encountered during therapeutic use of local anesthetics. (See ADVERSE REACTIONS, WARNINGS, and PRECAUTIONS.)
Description (DESCRIPTION)
Lidocaine HCl 2% Jelly is a sterile, aqueous product that contains a local anesthetic agent and is administered topically. (See INDICATIONS for specific uses.)
Lidocaine HCl 2% Jelly contains lidocaine HCl which is chemically designated as acetamide, 2-(diethylamino)-N-(2,6-dimethylphenyl)-, monohydrochloride and has the following structural formula:
Its molecular formula is C14H22N2O • HCl and its molecular weight is 270.80.
Lidocaine HCl 2% Jelly also contains hypromellose, and the resulting mixture maximizes contact with mucosa and provides lubrication for instrumentation. The unused portion should be discarded after initial use.
Composition of Lidocaine HCl 2% Jelly 30 mL and 5 mL tubes: Each mL contains 20 mg of lidocaine HCl. The formulation also contains methylparaben, propylparaben, hypromellose, and sodium hydroxide and/or hydrochloric acid to adjust pH between 6.0 to 7.0.
Directions:
Wipe injection site vigorously and discard
Precautions (PRECAUTIONS)
How Supplied (HOW SUPPLIED)
Lidocaine HCI 2% Jelly is supplied in the listed dosage forms.
NDC 17478-840-30 30 mL aluminum tube
NDC 17478-840-05 5 mL aluminum tube
A detachable applicator cone and a key for expressing the contents are included in the 30 mL carton.
Maximum Dosage (MAXIMUM DOSAGE)
No more than 600 mg of lidocaine HCl should be given in any 12 hour period.
Venipuncture Px1
Contents
1- Bio-Hazard Specimen Transport Bag
1-Pair of Sterile Vinyl Exam Gloves (Medium), Latex Free
2-Sheer Adhesive Bandages, Spots
1- Basic conforming Stretch Gauze, Non-sterile
1- Cohesive Wrap Bandage
2- Alcohol Prep Pads
1- Tourniquet, Latex free
1- Vacutainer Single Use Holder
1- Vacutainer Blood Collection Needle , Butterfly W/7"
Tubing, 23GX 3/4"
1- Vacutainer Eclipse Blood Collection Needle 21G X 1.25
3-Red Vacutainer SST Blood collection Tubes, 8.5 mL
1-Lavender Vacutainer EDTA Blood collection Tube 4.0 mL
1- 2% Lidocaine Hydrochloride Jelly, USP- 5.0 mL
Adverse Reactions (ADVERSE REACTIONS)
Adverse experiences following the administration of lidocaine are similar in nature to those observed in other amide local anesthetic agents. These adverse experiences are, in general, dose-related and may result from high plasma levels caused by excessive dosage or rapid absorption, or may result from a hypersensitivity, idiosyncrasy, or diminished tolerance on the part of the patient. Serious adverse experiences are generally systemic in nature. The following types are those most commonly reported:
There have been rare reports of endotracheal tube occlusion associated with the presence of dried jelly residue in the inner lumen of the tube. (See also WARNINGS and DOSAGE AND ADMINISTRATION.)
Contraindications (CONTRAINDICATIONS)
Lidocaine is contraindicated in patients with a known history of hypersensitivity to local anesthetics of the amide type or to other components of Lidocaine HCl 2% Jelly.
Use: Alcohol Prep (Use: alcohol_prep)
For preparation of the skin prior to injection.
Other Information: (Other information:)
- Store at room temperature: 15 deg C to 30 deg C 59 deg F to 86 deg F
- avoid excessive heat
Inactive Ingredient
Inactive Ingredient
- Water
Packaging Kit Label (Packaging-Kit Label)
Clinical Pharmacology (CLINICAL PHARMACOLOGY)
Indications and Usage (INDICATIONS AND USAGE)
Lidocaine HCl 2% Jelly is indicated for prevention and control of pain in procedures involving the male and female urethra, for topical treatment of painful urethritis, and as an anesthetic lubricant for endotracheal intubation (oral and nasal).
Indications and Usage:
Stop use and ask a doctor if:
- Irritation or redness develops
- condition persists for more than 72 hours
- Cleansing of an injection site
Dosage and Administration (DOSAGE AND ADMINISTRATION)
When Lidocaine HCl 2% Jelly is used concomitantly with other products containing lidocaine, the total dose contributed by all formulations must be kept in mind.
The dosage varies and depends upon the area to be anesthetized, vascularity of the tissues, individual tolerance, and the technique of anesthesia. The lowest dosage needed to provide effective anesthesia should be administered. Dosages should be reduced for children and for elderly and debilitated patients. Although the incidence of adverse effects with Lidocaine HCl 2% Jelly is quite low, caution should be exercised, particularly when employing large amounts, since the incidence of adverse effects is directly proportional to the total dose of local anesthetic agent administered.
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of accidental ingestion, seek professional assistance or consult a poison control center immediately.
Packaging Kit Components Labeling (Packaging- Kit Components Labeling)
Teratogenic Effects: Pregnancy Category B.
Reproduction studies for lidocaine have been performed in both rats and rabbits. There was no evidence of harm to the fetus at subcutaneous doses of up to 50 mg/kg lidocaine (300 mg/m2 on a body surface area basis) in the rat model. In the rabbit model, there was no evidence of harm to the fetus at a dose of 5 mg/kg, s.c. (60 mg/m2 on a body surface area basis). Treatment of rabbits with 25 mg/kg (300 mg/m2) produced evidence of maternal toxicity and evidence of delayed fetal development, including a non-significant decrease in fetal weight (7%) and an increase in minor skeletal anomalies (skull and sternebral defect, reduced ossification of the phalanges). The effect of lidocaine on post-natal development was examined in rats by treating pregnant female rats daily subcutaneously at doses of 2, 10, and 50 mg/kg (12, 60, and 300 mg/m2) from day 15 of pregnancy and up to 20 days post partum. No signs of adverse effects were seen either in dams or in the pups up to and including the dose of 10 mg/kg (60 mg/m2); however, the number of surviving pups was reduced at 50 mg/kg (300 mg/m2), both at birth and the duration of lactation period, the effect most likely being secondary to maternal toxicity. No other effects on litter size, litter weight, abnormalities in the pups and physical developments of the pups were seen in this study.
A second study examined the effects of lidocaine on post-natal development in the rat that included assessment of the pups from weaning to sexual maturity. Rats were treated for 8 months with 10 or 30 mg/kg, s.c. lidocaine (60 mg/m2 and 180 mg/m2 on a body surface area basis, respectively). This time period encompassed 3 mating periods. There was no evidence of altered post-natal development in any offspring; however, both doses of lidocaine significantly reduced the average number of pups per litter surviving until weaning of offspring from the first 2 mating period.
There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:36:41.940720 · Updated: 2026-03-14T21:47:24.245918