loperamide hydrochloride
a732a67c-bce1-4521-9df4-69709fdf537d
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Anti-diarrheal
Description
Loperamide HCl 2 mg
Medication Information
Warnings
Allergy alert: Do not use if you have ever had a rash or other allergic reaction to loperamide HCl
Purpose
Anti-diarrheal
Directions
- drink plenty of clear fluids to help prevent dehydration caused by diarrhea
- find right dose on chart. If possible, use weight to dose; otherwise, use age.
| adults and children 12 years and over | 2 caplets after the first loose stool; 1 caplet after each subsequent loose stool; but no more than 4 caplets in 24 hours |
| children 9-11 years (60-95 lbs) | 1 caplet after the first loose stool; 1/2 caplet after each subsequent loose stool; but no more than 3 caplets in 24 hours |
| children 6-8 years (48-59 lbs) | 1 caplet after the first loose stool; 1/2 caplet after each subsequent loose stool; but no more than 2 caplets in 24 hours |
| children under 6 years (up to 47 lbs) | ask a doctor |
Other Information
- store at 20°-25°C (68°-77°F)
- do not use if printed foil under cap is broken or missing
- see end panel for lot number and expiration date
Inactive Ingredients
anhydrous lactose, carnauba wax, D&C yellow no. 10 aluminum lake, FD&C blue no. 1 aluminum lake, hypromellose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, pregelatinized starch
Description
Loperamide HCl 2 mg
Use
controls symptoms of diarrhea, including Travelers' Diarrhea
Do Not Use
if you have bloody or black stool
When Using This Product
- tiredness, drowsiness or dizziness may occur. Be careful when driving or operating machinery.
Stop Use and Ask A Doctor If
- symptoms get worse
- diarrhea lasts for more than 2 days
- you get abdominal swelling or bulging. These may be signs of a serious condition.
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
Active Ingredient (in Each Caplet)
Loperamide HCl 2 mg
Ask A Doctor Before Use If You Have
- fever
- mucus in the stool
- a history of liver disease
Ask A Doctor Or Pharmacist Before Use If You Are
taking antibiotics
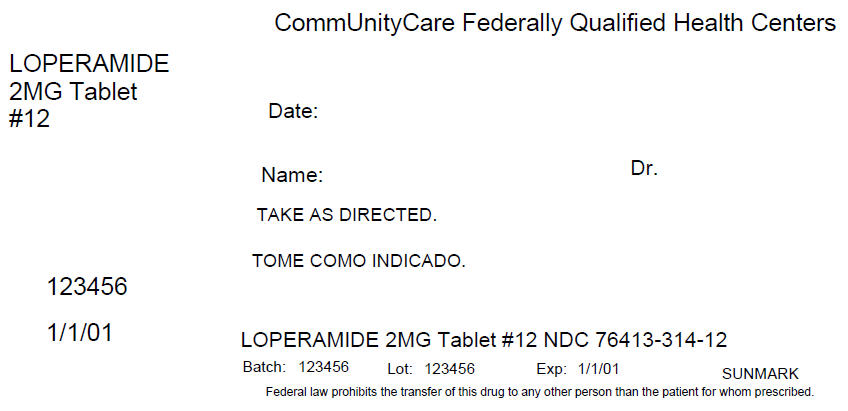
Principal Display Panel 2 Mg Tablet Blister Pack Carton Label
CommUnityCare Federally Qualified Health Centers
LOPERAMIDE
2MG Tablet
#12
Date:
Name:
Dr.
TAKE AS DIRECTED.
123456
1/1/01
LOPERAMIDE 2MG Tablet #12 NDC 76413-314-12
Batch: 123456
Lot: 123456
Exp: 1/1/01
SUNMARK
Federal law prohibits the transfer of this drug to any other person than the patient for whom prescribed.
Structured Label Content
Warnings
Allergy alert: Do not use if you have ever had a rash or other allergic reaction to loperamide HCl
Purpose
Anti-diarrheal
Directions
- drink plenty of clear fluids to help prevent dehydration caused by diarrhea
- find right dose on chart. If possible, use weight to dose; otherwise, use age.
| adults and children 12 years and over | 2 caplets after the first loose stool; 1 caplet after each subsequent loose stool; but no more than 4 caplets in 24 hours |
| children 9-11 years (60-95 lbs) | 1 caplet after the first loose stool; 1/2 caplet after each subsequent loose stool; but no more than 3 caplets in 24 hours |
| children 6-8 years (48-59 lbs) | 1 caplet after the first loose stool; 1/2 caplet after each subsequent loose stool; but no more than 2 caplets in 24 hours |
| children under 6 years (up to 47 lbs) | ask a doctor |
Other Information (Other information)
- store at 20°-25°C (68°-77°F)
- do not use if printed foil under cap is broken or missing
- see end panel for lot number and expiration date
Inactive Ingredients (Inactive ingredients)
anhydrous lactose, carnauba wax, D&C yellow no. 10 aluminum lake, FD&C blue no. 1 aluminum lake, hypromellose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, pregelatinized starch
Use
controls symptoms of diarrhea, including Travelers' Diarrhea
Do Not Use (Do not use)
if you have bloody or black stool
When Using This Product (When using this product)
- tiredness, drowsiness or dizziness may occur. Be careful when driving or operating machinery.
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- symptoms get worse
- diarrhea lasts for more than 2 days
- you get abdominal swelling or bulging. These may be signs of a serious condition.
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
Active Ingredient (in Each Caplet) (Active ingredient (in each caplet))
Loperamide HCl 2 mg
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- fever
- mucus in the stool
- a history of liver disease
Ask A Doctor Or Pharmacist Before Use If You Are (Ask a doctor or pharmacist before use if you are)
taking antibiotics
Principal Display Panel 2 Mg Tablet Blister Pack Carton Label (PRINCIPAL DISPLAY PANEL - 2 MG Tablet Blister Pack Carton Label)
CommUnityCare Federally Qualified Health Centers
LOPERAMIDE
2MG Tablet
#12
Date:
Name:
Dr.
TAKE AS DIRECTED.
123456
1/1/01
LOPERAMIDE 2MG Tablet #12 NDC 76413-314-12
Batch: 123456
Lot: 123456
Exp: 1/1/01
SUNMARK
Federal law prohibits the transfer of this drug to any other person than the patient for whom prescribed.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:06.374117 · Updated: 2026-03-14T22:54:19.785916