These Highlights Do Not Include All The Information Needed To Use Vermox™ Chewable Safely And Effectively. See Full Prescribing Information For Vermox™ Chewable.
a720c424-517a-461b-8e13-6c37caf8aa3f
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dosing and Administration ( 2 ) 9/2021
Indications and Usage
VERMOX™ CHEWABLE is indicated for the treatment of patients one year of age and older with gastrointestinal infections caused by Ascaris lumbricoides (roundworm) and Trichuris trichiura (whipworm).
Dosage and Administration
The recommended dosage in patients one year of age and older is one VERMOX™ CHEWABLE 500 mg tablet taken as a single dose. Chew VERMOX™ CHEWABLE 500 mg tablet completely before swallowing. Do not swallow the tablet whole. For patients who have difficulty chewing the tablet, approximately 2 mL to 3 mL of drinking water can be added to a suitably sized spoon and the VERMOX™ CHEWABLE 500 mg tablet placed into the water. Within 2 minutes, the tablet absorbs the water and turns into a soft mass with semi-solid consistency, which can then be swallowed. VERMOX™ CHEWABLE 500 mg tablet can be taken without regard to food intake [see Clinical Pharmacology (12.3) ].
Warnings and Precautions
Risk of Convulsions: Convulsions in infants below the age of 1 year have been reported ( 5.1 ) Hematologic Effects: Neutropenia and agranulocytosis have been reported in patients receiving mebendazole at higher doses and for prolonged duration. Monitor blood counts in these patients ( 5.2 ) Metronidazole and Serious Skin Reactions: Stevens-Johnson syndrome/toxic epidermal necrolysis (SJS/TEN) have been reported with the concomitant use of mebendazole and metronidazole. Avoid concomitant use of mebendazole and metronidazole ( 5.3 )
Contraindications
VERMOX™ CHEWABLE is contraindicated in persons with a known hypersensitivity to the drug or its excipients.
Adverse Reactions
Adverse reactions reported in clinical trials were anorexia, abdominal pain, diarrhea, flatulence, nausea, vomiting and rash. ( 6.1 ). To report SUSPECTED ADVERSE REACTIONS, contact Janssen Pharmaceuticals, Inc. at 1-800-JANSSEN (1-800-526-7736) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Concomitant use of mebendazole and metronidazole should be avoided [see Warnings and Precautions (5.3) ] .
Storage and Handling
VERMOX™ CHEWABLE tablets are supplied as 500 mg, round, flat radius-edged white to yellowish chewable tablets that are debossed with "M/500" on one side and "J" on the other side. They are supplied as follows: Bottles of 200 tablets NDC 50458-675-20
How Supplied
VERMOX™ CHEWABLE tablets are supplied as 500 mg, round, flat radius-edged white to yellowish chewable tablets that are debossed with "M/500" on one side and "J" on the other side. They are supplied as follows: Bottles of 200 tablets NDC 50458-675-20
Medication Information
Warnings and Precautions
Risk of Convulsions: Convulsions in infants below the age of 1 year have been reported ( 5.1 ) Hematologic Effects: Neutropenia and agranulocytosis have been reported in patients receiving mebendazole at higher doses and for prolonged duration. Monitor blood counts in these patients ( 5.2 ) Metronidazole and Serious Skin Reactions: Stevens-Johnson syndrome/toxic epidermal necrolysis (SJS/TEN) have been reported with the concomitant use of mebendazole and metronidazole. Avoid concomitant use of mebendazole and metronidazole ( 5.3 )
Indications and Usage
VERMOX™ CHEWABLE is indicated for the treatment of patients one year of age and older with gastrointestinal infections caused by Ascaris lumbricoides (roundworm) and Trichuris trichiura (whipworm).
Dosage and Administration
The recommended dosage in patients one year of age and older is one VERMOX™ CHEWABLE 500 mg tablet taken as a single dose. Chew VERMOX™ CHEWABLE 500 mg tablet completely before swallowing. Do not swallow the tablet whole. For patients who have difficulty chewing the tablet, approximately 2 mL to 3 mL of drinking water can be added to a suitably sized spoon and the VERMOX™ CHEWABLE 500 mg tablet placed into the water. Within 2 minutes, the tablet absorbs the water and turns into a soft mass with semi-solid consistency, which can then be swallowed. VERMOX™ CHEWABLE 500 mg tablet can be taken without regard to food intake [see Clinical Pharmacology (12.3) ].
Contraindications
VERMOX™ CHEWABLE is contraindicated in persons with a known hypersensitivity to the drug or its excipients.
Adverse Reactions
Adverse reactions reported in clinical trials were anorexia, abdominal pain, diarrhea, flatulence, nausea, vomiting and rash. ( 6.1 ). To report SUSPECTED ADVERSE REACTIONS, contact Janssen Pharmaceuticals, Inc. at 1-800-JANSSEN (1-800-526-7736) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Concomitant use of mebendazole and metronidazole should be avoided [see Warnings and Precautions (5.3) ] .
Storage and Handling
VERMOX™ CHEWABLE tablets are supplied as 500 mg, round, flat radius-edged white to yellowish chewable tablets that are debossed with "M/500" on one side and "J" on the other side. They are supplied as follows: Bottles of 200 tablets NDC 50458-675-20
How Supplied
VERMOX™ CHEWABLE tablets are supplied as 500 mg, round, flat radius-edged white to yellowish chewable tablets that are debossed with "M/500" on one side and "J" on the other side. They are supplied as follows: Bottles of 200 tablets NDC 50458-675-20
Description
Dosing and Administration ( 2 ) 9/2021
Section 34077-8
Animal Data
Embryo-fetal developmental toxicity studies in rats revealed no adverse effects on dams or their progeny at doses up to 2.5 mg/kg/day on gestation days 6–15 (the period of organogenesis). Dosing at ≥10 mg/kg/day resulted in a lowered body weight gain and a decreased pregnancy rate. Maternal toxicity, including body weight loss in one animal and maternal death in 11 of 20 animals, was seen at 40 mg/kg/day. At 10 mg/kg/day, increased embryo-fetal resorption (100% were resorbed at 40 mg/kg/day), decreased pup weight and increased incidence of malformations (primarily skeletal) were observed. Mebendazole was also embryotoxic and teratogenic in pregnant rats at single oral doses during organogenesis as low as 10 mg/kg (approximately 0.2-fold the MRHD, based on mg/m2).
In embryo-fetal developmental toxicity studies in mice dosed on gestation days 6–15, doses of 10 mg/kg/day and higher resulted in decreased body weight gain at 10 and 40 mg/kg/day and a higher mortality rate at 40 mg/kg/day. At doses of 10 mg/kg/day (approximately 0.1-fold the MRHD, based on mg/m2) and higher, embryo-fetal resorption increased (100% at 40 mg/kg) and fetal malformations, including skeletal, cranial, and soft tissue anomalies, were present. Dosing of hamsters and rabbits did not result in embryotoxicity or teratogenicity at doses up to 40 mg/kg/day (0.6 to 1.6-fold the MRHD, based on mg/m2).
In a peri- and post-natal toxicity study in rats, mebendazole did not adversely affect dams or their progeny at 20 mg/kg/day. At 40 mg/kg (0.8-fold the MRHD, based on mg/m2), a reduction of the number of live pups was observed and there was no survival at weaning. No abnormalities were found on gross and radiographic examination of pups at birth.
Section 42229-5
Clinical Studies with Mebendazole Chewable 500 mg Tablet
The safety profile of mebendazole chewable 500 mg tablets administered as a single dose was evaluated in 677 pediatric subjects aged 1 to 16 years and in 34 adults. The safety profile was consistent with the known safety profile of mebendazole.
Section 43683-2
| Dosing and Administration (2) | 9/2021 |
Section 44425-7
Store below 30°C. Keep container tightly closed. Unused tablets should be discarded 1 month after the bottle is first opened. When the bottle is first opened this Discard After date should be written on the bottle label in the place provided.
10 Overdosage
In patients treated at dosages substantially higher than recommended or for prolonged periods of time, the following adverse reactions have been reported: alopecia, reversible transaminase elevations, hepatitis, agranulocytosis, neutropenia, and glomerulonephritis.
8.6 Adult Use
The safety and effectiveness of VERMOX™ CHEWABLE 500 mg tablets have been established in adults for the treatment of gastrointestinal infections by T. trichiura and A. lumbricoides. Use of VERMOX™ CHEWABLE 500 mg tablets in adults for these indications is supported by evidence from an adequate and well-controlled trial in pediatric patients ages 1 to 16 years [see Clinical Studies (14.1)], safety data in adults [see Adverse Reactions (6.1)], pharmacokinetic data in adults [see Clinical Pharmacology (12.3)], and the evidence from published literature.
11 Description
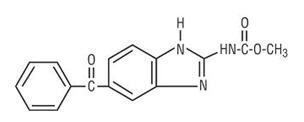
VERMOX™ CHEWABLE (mebendazole chewable tablets) is an orally administered anthelmintic. Chemically, it is methyl 5-benzoylbenzimidazole-2-carbamate. Its molecular formula is C16H13N3O3. Its molecular weight is 295.30. It has the following chemical structure:
Mebendazole exhibits polymorphism. The polymorph used in VERMOX™ CHEWABLE is polymorph form C. Mebendazole is a white to almost white powder. It is practically insoluble in water, in ethanol (96%) and in methylene chloride.
Each round, flat radius-edged white to yellowish chewable tablet contains 500 mg of mebendazole and is debossed with "M/500" on one side and "J" on the other side.
Inactive ingredients consist of: crospovidone, magnesium stearate, microcrystalline cellulose, povidone, purified water, strawberry flavor and sucralose.
8.4 Pediatric Use
The safety and effectiveness of VERMOX™ CHEWABLE 500 mg tablets have been established in pediatric patients 1 to 16 years of age. Use of VERMOX™ CHEWABLE 500 mg tablets in children is supported by evidence from adequate and well-controlled studies of VERMOX™ CHEWABLE 500 mg tablets [see Clinical Studies (14)].
The safety and effectiveness of mebendazole, including VERMOX™ CHEWABLE have not been established in pediatric patients less than one year of age. Convulsions have been reported with mebendazole use in this age group [see Warnings and Precautions (5.1) and Adverse Reactions (6.2)].
8.5 Geriatric Use
Clinical studies of mebendazole did not include sufficient numbers of subjects aged 65 and older to determine whether they respond differently from younger subjects.
14 Clinical Studies
The efficacy of VERMOX™ CHEWABLE 500 mg tablets was evaluated in a double-blind, randomized, placebo controlled trial conducted in Africa in 295 pediatric patients between the ages of 1 year to 16 years of age with A. lumbricoides and/or T. trichiura infections. Patients were stratified by worm type and randomized to receive either VERMOX™ CHEWABLE 500 mg tablet (N=149) or placebo (N=146) at the baseline visit (double-blind period). After the 19 day double-blind period, all subjects received a single VERMOX™ CHEWABLE 500 mg tablet (open-label period).
Clinical cure was defined as zero egg count (A. lumbricoides and/or T. trichiura) at the end of the double-blind period (Day 19) in patients with positive egg count for the respective worm(s) at baseline. Patients with missing stool sample at Day 19 were considered not cured (Table 4).
| Infection Type | VERMOX™ CHEWABLE 500 mg All Patients=149 Some patients had mixed infection.
|
Placebo All Patients=146 |
Difference Difference in cure rates, expressed in percentages, and based on Mantel Haenszel methods to account for stratification by site.
(95% CI) |
|---|---|---|---|
| A. lumbricoides |
N=86 n (%) |
N=81 n (%) |
|
| Cure | 72 (83.7) | 9 (11.1) |
72.6 (62.3, 82.7) P-value <0.001 based on the Cochran-Mantel-Haenszel test, controlling for the effect of site.
|
| Failure Failures include patients who tested positive for the worm at Visit 3 (Day 19, i.e. test-of-cure).
|
9 (10.5) | 67 (82.7) | |
| Missing Patients with missing stool sample at Day 19.
|
5 (5.8) | 5 (6.2) | |
| T. trichiura |
N=124 n (%) |
N=119 n (%) |
|
| Cure | 42 (33.9) | 9 (7.6) | 26.2 (16.7, 35.6) |
| Failure | 76 (61.3) | 103 (86.6) | |
| Missing | 6 (4.8) | 7 (5.8) |
In patients treated with VERMOX™ CHEWABLE 500 mg, egg count reduction rate at the end of the double-blind period (Day 19) in patients with A. lumbricoides and/or T. trichiura was statistically significant (p<0.001) compared to placebo, 100% compared to 30.0% for A. lumbricoides, respectively, and 81.2% compared to 27.4% for T. trichiura, respectively.
4 Contraindications
VERMOX™ CHEWABLE is contraindicated in persons with a known hypersensitivity to the drug or its excipients.
6 Adverse Reactions
Adverse reactions reported in clinical trials were anorexia, abdominal pain, diarrhea, flatulence, nausea, vomiting and rash. (6.1).
To report SUSPECTED ADVERSE REACTIONS, contact Janssen Pharmaceuticals, Inc. at 1-800-JANSSEN (1-800-526-7736) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions
Concomitant use of mebendazole and metronidazole should be avoided [see Warnings and Precautions (5.3)].
6.1 Clinical Studies
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of mebendazole was evaluated in 6276 adult and pediatric subjects one year of age and older who participated in 39 clinical trials for treatment of single or mixed parasitic infections of the gastrointestinal tract. In these trials, the formulations, dosages and duration of mebendazole treatment varied. Adverse reactions reported in mebendazole-treated subjects from the 39 clinical trials are shown in Table 1 below.
| Adverse Reaction(s) |
|---|
| Gastrointestinal Disorders |
| Anorexia |
| Abdominal Pain |
| Diarrhea |
| Flatulence |
| Nausea |
| Vomiting |
| Skin and Subcutaneous Tissue Disorders |
| Rash |
1 Indications and Usage
VERMOX™ CHEWABLE is indicated for the treatment of patients one year of age and older with gastrointestinal infections caused by Ascaris lumbricoides (roundworm) and Trichuris trichiura (whipworm).
5.1 Risk of Convulsions
Convulsions have been reported in infants below the age of 1 year during post-marketing experience with mebendazole [see Adverse Reactions (6.2)].
5.2 Hematologic Effects
Agranulocytosis and neutropenia have been reported with mebendazole use at higher doses and for more prolonged durations than is recommended for the treatment of soil-transmitted helminth infections. Monitor blood counts if VERMOX™ CHEWABLE is used at higher doses or for prolonged duration.
12.1 Mechanism of Action
Mebendazole, a benzimidazole, is an anthelmintic [see Microbiology (12.4)].
5 Warnings and Precautions
- Risk of Convulsions: Convulsions in infants below the age of 1 year have been reported (5.1)
- Hematologic Effects: Neutropenia and agranulocytosis have been reported in patients receiving mebendazole at higher doses and for prolonged duration. Monitor blood counts in these patients (5.2)
- Metronidazole and Serious Skin Reactions: Stevens-Johnson syndrome/toxic epidermal necrolysis (SJS/TEN) have been reported with the concomitant use of mebendazole and metronidazole. Avoid concomitant use of mebendazole and metronidazole (5.3)
2 Dosage and Administration
The recommended dosage in patients one year of age and older is one VERMOX™ CHEWABLE 500 mg tablet taken as a single dose.
Chew VERMOX™ CHEWABLE 500 mg tablet completely before swallowing. Do not swallow the tablet whole.
For patients who have difficulty chewing the tablet, approximately 2 mL to 3 mL of drinking water can be added to a suitably sized spoon and the VERMOX™ CHEWABLE 500 mg tablet placed into the water. Within 2 minutes, the tablet absorbs the water and turns into a soft mass with semi-solid consistency, which can then be swallowed.
VERMOX™ CHEWABLE 500 mg tablet can be taken without regard to food intake [see Clinical Pharmacology (12.3)].
3 Dosage Forms and Strengths
Chewable Tablet: 500 mg round, flat radius-edged white to yellowish chewable tablet that is debossed with "M/500" on one side and "J" on the other side.
6.2 Postmarketing Experience
The following adverse reactions have been identified in adult and pediatric patients postmarketing with mebendazole formulations and dosages other than the VERMOX™ CHEWABLE 500 mg tablet. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
| Adverse Reaction(s) | |
|---|---|
| Blood and Lymphatic System Disorders | Agranulocytosis, Neutropenia |
| Immune System Disorders | Hypersensitivity including anaphylactic reactions |
| Nervous System Disorders | Convulsions, Dizziness |
| Hepatobiliary Disorders | Hepatitis, Abnormal liver tests |
| Renal and Urinary Disorders | Glomerulonephritis |
| Skin and Subcutaneous Tissue Disorders | Toxic epidermal necrolysis, Stevens-Johnson syndrome, Exanthema, Angioedema, Urticaria, Alopecia |
17 Patient Counseling Information
Advise patients that:
- VERMOX™ CHEWABLE tablet must be chewed completely before swallowing. For patients who have difficulty chewing the tablet, VERMOX™ CHEWABLE tablet can be turned into a soft mass with semi-solid consistency by adding 2 mL to 3 mL of drinking water to a spoon then placing the tablet into the water, which can then be swallowed. [see Dosage and Administration (2)]
- VERMOX™ CHEWABLE tablet must not be swallowed whole. [see Dosage and Administration (2)]
- VERMOX™ CHEWABLE tablet can be taken with or without food. [see Dosage and Administration (2)]
- Taking VERMOX™ CHEWABLE tablet and metronidazole together may cause serious skin reactions and should be avoided. [see Warnings and Precautions (5.3)]
16 How Supplied/storage and Handling
VERMOX™ CHEWABLE tablets are supplied as 500 mg, round, flat radius-edged white to yellowish chewable tablets that are debossed with "M/500" on one side and "J" on the other side. They are supplied as follows:
| Bottles of 200 tablets | NDC 50458-675-20 |
Principal Display Panel 500 Mg Tablet Bottle Label
NDC 50458-675-20
Vermox™ Chewable
(mebendazole chewable tablets)
500 mg
Each chewable tablet contains 500 mg
of mebendazole.
200 tablets
Rx only
USUAL DOSAGE: See package insert for
full Prescribing Information.
Chew tablets completely before swallowing.
Do not swallow tablets whole.
Store below 30°C.
Discard after _/_/_.
Discard unused portion
1 month after first
opening.
USA-AW_112506
© Janssen Pharmaceuticals, Inc. 2016
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In carcinogenicity tests of mebendazole in mice and rats, no carcinogenic effects were seen at doses as high as 40 mg/kg (0.4 to 0.8-fold the MRHD, based on mg/m2) given daily over two years. No mutagenic activity was observed with mebendazole in a bacterial reverse gene mutation test. Mebendazole was mutagenic in the absence of S-9 when tested using a continuous (24 hour) treatment incubation period in the mouse lymphoma thymidine kinase assay. Mebendazole was aneugenic in vitro in mammalian somatic cells. In the in vivo mouse micronucleus assay, orally administered mebendazole induced an increased frequency of micronucleated polychromatic erythrocytes with evidence suggestive of aneugenicity. Doses up to 40 mg/kg in rats (0.8-fold the MRHD, based on mg/m2), given to males for 60 days and to females for 14 days prior to gestation, had no effect upon fetuses and offspring.
5.3 Metronidazole Drug Interaction and Serious Skin Reactions
Stevens-Johnson syndrome/toxic epidermal necrolysis (SJS/TEN) have been reported with the concomitant use of mebendazole and metronidazole. Avoid concomitant use of mebendazole and metronidazole.
Structured Label Content
Section 34077-8 (34077-8)
Animal Data
Embryo-fetal developmental toxicity studies in rats revealed no adverse effects on dams or their progeny at doses up to 2.5 mg/kg/day on gestation days 6–15 (the period of organogenesis). Dosing at ≥10 mg/kg/day resulted in a lowered body weight gain and a decreased pregnancy rate. Maternal toxicity, including body weight loss in one animal and maternal death in 11 of 20 animals, was seen at 40 mg/kg/day. At 10 mg/kg/day, increased embryo-fetal resorption (100% were resorbed at 40 mg/kg/day), decreased pup weight and increased incidence of malformations (primarily skeletal) were observed. Mebendazole was also embryotoxic and teratogenic in pregnant rats at single oral doses during organogenesis as low as 10 mg/kg (approximately 0.2-fold the MRHD, based on mg/m2).
In embryo-fetal developmental toxicity studies in mice dosed on gestation days 6–15, doses of 10 mg/kg/day and higher resulted in decreased body weight gain at 10 and 40 mg/kg/day and a higher mortality rate at 40 mg/kg/day. At doses of 10 mg/kg/day (approximately 0.1-fold the MRHD, based on mg/m2) and higher, embryo-fetal resorption increased (100% at 40 mg/kg) and fetal malformations, including skeletal, cranial, and soft tissue anomalies, were present. Dosing of hamsters and rabbits did not result in embryotoxicity or teratogenicity at doses up to 40 mg/kg/day (0.6 to 1.6-fold the MRHD, based on mg/m2).
In a peri- and post-natal toxicity study in rats, mebendazole did not adversely affect dams or their progeny at 20 mg/kg/day. At 40 mg/kg (0.8-fold the MRHD, based on mg/m2), a reduction of the number of live pups was observed and there was no survival at weaning. No abnormalities were found on gross and radiographic examination of pups at birth.
Section 42229-5 (42229-5)
Clinical Studies with Mebendazole Chewable 500 mg Tablet
The safety profile of mebendazole chewable 500 mg tablets administered as a single dose was evaluated in 677 pediatric subjects aged 1 to 16 years and in 34 adults. The safety profile was consistent with the known safety profile of mebendazole.
Section 43683-2 (43683-2)
| Dosing and Administration (2) | 9/2021 |
Section 44425-7 (44425-7)
Store below 30°C. Keep container tightly closed. Unused tablets should be discarded 1 month after the bottle is first opened. When the bottle is first opened this Discard After date should be written on the bottle label in the place provided.
10 Overdosage (10 OVERDOSAGE)
In patients treated at dosages substantially higher than recommended or for prolonged periods of time, the following adverse reactions have been reported: alopecia, reversible transaminase elevations, hepatitis, agranulocytosis, neutropenia, and glomerulonephritis.
8.6 Adult Use
The safety and effectiveness of VERMOX™ CHEWABLE 500 mg tablets have been established in adults for the treatment of gastrointestinal infections by T. trichiura and A. lumbricoides. Use of VERMOX™ CHEWABLE 500 mg tablets in adults for these indications is supported by evidence from an adequate and well-controlled trial in pediatric patients ages 1 to 16 years [see Clinical Studies (14.1)], safety data in adults [see Adverse Reactions (6.1)], pharmacokinetic data in adults [see Clinical Pharmacology (12.3)], and the evidence from published literature.
11 Description (11 DESCRIPTION)
VERMOX™ CHEWABLE (mebendazole chewable tablets) is an orally administered anthelmintic. Chemically, it is methyl 5-benzoylbenzimidazole-2-carbamate. Its molecular formula is C16H13N3O3. Its molecular weight is 295.30. It has the following chemical structure:
Mebendazole exhibits polymorphism. The polymorph used in VERMOX™ CHEWABLE is polymorph form C. Mebendazole is a white to almost white powder. It is practically insoluble in water, in ethanol (96%) and in methylene chloride.
Each round, flat radius-edged white to yellowish chewable tablet contains 500 mg of mebendazole and is debossed with "M/500" on one side and "J" on the other side.
Inactive ingredients consist of: crospovidone, magnesium stearate, microcrystalline cellulose, povidone, purified water, strawberry flavor and sucralose.
8.4 Pediatric Use
The safety and effectiveness of VERMOX™ CHEWABLE 500 mg tablets have been established in pediatric patients 1 to 16 years of age. Use of VERMOX™ CHEWABLE 500 mg tablets in children is supported by evidence from adequate and well-controlled studies of VERMOX™ CHEWABLE 500 mg tablets [see Clinical Studies (14)].
The safety and effectiveness of mebendazole, including VERMOX™ CHEWABLE have not been established in pediatric patients less than one year of age. Convulsions have been reported with mebendazole use in this age group [see Warnings and Precautions (5.1) and Adverse Reactions (6.2)].
8.5 Geriatric Use
Clinical studies of mebendazole did not include sufficient numbers of subjects aged 65 and older to determine whether they respond differently from younger subjects.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of VERMOX™ CHEWABLE 500 mg tablets was evaluated in a double-blind, randomized, placebo controlled trial conducted in Africa in 295 pediatric patients between the ages of 1 year to 16 years of age with A. lumbricoides and/or T. trichiura infections. Patients were stratified by worm type and randomized to receive either VERMOX™ CHEWABLE 500 mg tablet (N=149) or placebo (N=146) at the baseline visit (double-blind period). After the 19 day double-blind period, all subjects received a single VERMOX™ CHEWABLE 500 mg tablet (open-label period).
Clinical cure was defined as zero egg count (A. lumbricoides and/or T. trichiura) at the end of the double-blind period (Day 19) in patients with positive egg count for the respective worm(s) at baseline. Patients with missing stool sample at Day 19 were considered not cured (Table 4).
| Infection Type | VERMOX™ CHEWABLE 500 mg All Patients=149 Some patients had mixed infection.
|
Placebo All Patients=146 |
Difference Difference in cure rates, expressed in percentages, and based on Mantel Haenszel methods to account for stratification by site.
(95% CI) |
|---|---|---|---|
| A. lumbricoides |
N=86 n (%) |
N=81 n (%) |
|
| Cure | 72 (83.7) | 9 (11.1) |
72.6 (62.3, 82.7) P-value <0.001 based on the Cochran-Mantel-Haenszel test, controlling for the effect of site.
|
| Failure Failures include patients who tested positive for the worm at Visit 3 (Day 19, i.e. test-of-cure).
|
9 (10.5) | 67 (82.7) | |
| Missing Patients with missing stool sample at Day 19.
|
5 (5.8) | 5 (6.2) | |
| T. trichiura |
N=124 n (%) |
N=119 n (%) |
|
| Cure | 42 (33.9) | 9 (7.6) | 26.2 (16.7, 35.6) |
| Failure | 76 (61.3) | 103 (86.6) | |
| Missing | 6 (4.8) | 7 (5.8) |
In patients treated with VERMOX™ CHEWABLE 500 mg, egg count reduction rate at the end of the double-blind period (Day 19) in patients with A. lumbricoides and/or T. trichiura was statistically significant (p<0.001) compared to placebo, 100% compared to 30.0% for A. lumbricoides, respectively, and 81.2% compared to 27.4% for T. trichiura, respectively.
4 Contraindications (4 CONTRAINDICATIONS)
VERMOX™ CHEWABLE is contraindicated in persons with a known hypersensitivity to the drug or its excipients.
6 Adverse Reactions (6 ADVERSE REACTIONS)
Adverse reactions reported in clinical trials were anorexia, abdominal pain, diarrhea, flatulence, nausea, vomiting and rash. (6.1).
To report SUSPECTED ADVERSE REACTIONS, contact Janssen Pharmaceuticals, Inc. at 1-800-JANSSEN (1-800-526-7736) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions (7 DRUG INTERACTIONS)
Concomitant use of mebendazole and metronidazole should be avoided [see Warnings and Precautions (5.3)].
6.1 Clinical Studies
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of mebendazole was evaluated in 6276 adult and pediatric subjects one year of age and older who participated in 39 clinical trials for treatment of single or mixed parasitic infections of the gastrointestinal tract. In these trials, the formulations, dosages and duration of mebendazole treatment varied. Adverse reactions reported in mebendazole-treated subjects from the 39 clinical trials are shown in Table 1 below.
| Adverse Reaction(s) |
|---|
| Gastrointestinal Disorders |
| Anorexia |
| Abdominal Pain |
| Diarrhea |
| Flatulence |
| Nausea |
| Vomiting |
| Skin and Subcutaneous Tissue Disorders |
| Rash |
1 Indications and Usage (1 INDICATIONS AND USAGE)
VERMOX™ CHEWABLE is indicated for the treatment of patients one year of age and older with gastrointestinal infections caused by Ascaris lumbricoides (roundworm) and Trichuris trichiura (whipworm).
5.1 Risk of Convulsions
Convulsions have been reported in infants below the age of 1 year during post-marketing experience with mebendazole [see Adverse Reactions (6.2)].
5.2 Hematologic Effects
Agranulocytosis and neutropenia have been reported with mebendazole use at higher doses and for more prolonged durations than is recommended for the treatment of soil-transmitted helminth infections. Monitor blood counts if VERMOX™ CHEWABLE is used at higher doses or for prolonged duration.
12.1 Mechanism of Action
Mebendazole, a benzimidazole, is an anthelmintic [see Microbiology (12.4)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Risk of Convulsions: Convulsions in infants below the age of 1 year have been reported (5.1)
- Hematologic Effects: Neutropenia and agranulocytosis have been reported in patients receiving mebendazole at higher doses and for prolonged duration. Monitor blood counts in these patients (5.2)
- Metronidazole and Serious Skin Reactions: Stevens-Johnson syndrome/toxic epidermal necrolysis (SJS/TEN) have been reported with the concomitant use of mebendazole and metronidazole. Avoid concomitant use of mebendazole and metronidazole (5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
The recommended dosage in patients one year of age and older is one VERMOX™ CHEWABLE 500 mg tablet taken as a single dose.
Chew VERMOX™ CHEWABLE 500 mg tablet completely before swallowing. Do not swallow the tablet whole.
For patients who have difficulty chewing the tablet, approximately 2 mL to 3 mL of drinking water can be added to a suitably sized spoon and the VERMOX™ CHEWABLE 500 mg tablet placed into the water. Within 2 minutes, the tablet absorbs the water and turns into a soft mass with semi-solid consistency, which can then be swallowed.
VERMOX™ CHEWABLE 500 mg tablet can be taken without regard to food intake [see Clinical Pharmacology (12.3)].
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Chewable Tablet: 500 mg round, flat radius-edged white to yellowish chewable tablet that is debossed with "M/500" on one side and "J" on the other side.
6.2 Postmarketing Experience
The following adverse reactions have been identified in adult and pediatric patients postmarketing with mebendazole formulations and dosages other than the VERMOX™ CHEWABLE 500 mg tablet. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
| Adverse Reaction(s) | |
|---|---|
| Blood and Lymphatic System Disorders | Agranulocytosis, Neutropenia |
| Immune System Disorders | Hypersensitivity including anaphylactic reactions |
| Nervous System Disorders | Convulsions, Dizziness |
| Hepatobiliary Disorders | Hepatitis, Abnormal liver tests |
| Renal and Urinary Disorders | Glomerulonephritis |
| Skin and Subcutaneous Tissue Disorders | Toxic epidermal necrolysis, Stevens-Johnson syndrome, Exanthema, Angioedema, Urticaria, Alopecia |
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise patients that:
- VERMOX™ CHEWABLE tablet must be chewed completely before swallowing. For patients who have difficulty chewing the tablet, VERMOX™ CHEWABLE tablet can be turned into a soft mass with semi-solid consistency by adding 2 mL to 3 mL of drinking water to a spoon then placing the tablet into the water, which can then be swallowed. [see Dosage and Administration (2)]
- VERMOX™ CHEWABLE tablet must not be swallowed whole. [see Dosage and Administration (2)]
- VERMOX™ CHEWABLE tablet can be taken with or without food. [see Dosage and Administration (2)]
- Taking VERMOX™ CHEWABLE tablet and metronidazole together may cause serious skin reactions and should be avoided. [see Warnings and Precautions (5.3)]
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
VERMOX™ CHEWABLE tablets are supplied as 500 mg, round, flat radius-edged white to yellowish chewable tablets that are debossed with "M/500" on one side and "J" on the other side. They are supplied as follows:
| Bottles of 200 tablets | NDC 50458-675-20 |
Principal Display Panel 500 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 500 mg Tablet Bottle Label)
NDC 50458-675-20
Vermox™ Chewable
(mebendazole chewable tablets)
500 mg
Each chewable tablet contains 500 mg
of mebendazole.
200 tablets
Rx only
USUAL DOSAGE: See package insert for
full Prescribing Information.
Chew tablets completely before swallowing.
Do not swallow tablets whole.
Store below 30°C.
Discard after _/_/_.
Discard unused portion
1 month after first
opening.
USA-AW_112506
© Janssen Pharmaceuticals, Inc. 2016
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In carcinogenicity tests of mebendazole in mice and rats, no carcinogenic effects were seen at doses as high as 40 mg/kg (0.4 to 0.8-fold the MRHD, based on mg/m2) given daily over two years. No mutagenic activity was observed with mebendazole in a bacterial reverse gene mutation test. Mebendazole was mutagenic in the absence of S-9 when tested using a continuous (24 hour) treatment incubation period in the mouse lymphoma thymidine kinase assay. Mebendazole was aneugenic in vitro in mammalian somatic cells. In the in vivo mouse micronucleus assay, orally administered mebendazole induced an increased frequency of micronucleated polychromatic erythrocytes with evidence suggestive of aneugenicity. Doses up to 40 mg/kg in rats (0.8-fold the MRHD, based on mg/m2), given to males for 60 days and to females for 14 days prior to gestation, had no effect upon fetuses and offspring.
5.3 Metronidazole Drug Interaction and Serious Skin Reactions
Stevens-Johnson syndrome/toxic epidermal necrolysis (SJS/TEN) have been reported with the concomitant use of mebendazole and metronidazole. Avoid concomitant use of mebendazole and metronidazole.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:36:19.750489 · Updated: 2026-03-14T21:45:57.149460