Pepto Relief Bismuth Subsalicylate 262 Mg
a6f383b4-47f9-4837-93e2-2ca114fb20b9
34390-5
HUMAN OTC DRUG LABEL
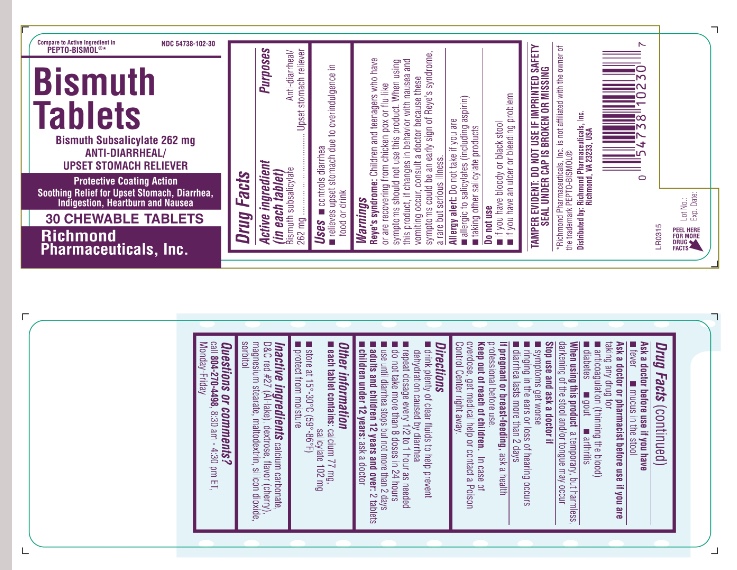
Drug Facts
Composition & Product
Identifiers & Packaging
Description
.
Purpose
Anti-diarrheal/Upset stomach reliever
Medication Information
Purpose
Anti-diarrheal/Upset stomach reliever
Description
.
Uses
- Controls diarrhea
- Relieves upset stomach due to overindulgence in food or drink
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Warnings
Reye’s syndrome: Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye’s syndrome, a rare but serious illness.
Allergy alert: Do not take if you are
- allergic to salicylates (including aspirin)
- taking other salicylate products
Do not use
- If you have bloody or black stool
- If you have an ulcer or bleeding problem
Ask a doctor before use if you have
- Fever
- Mucus in the stool
Ask a doctor or pharmacist before use if you are
- taking any drug for
- anticoagulation (thinning the blood)
- diabetes
- gout
- arthritis
when using this product a temporary, but harmless, darkening of the stool and/or tongue may occur
stop use and ask a doctor if
- symptoms get worse
- ringing in the ears or loss of hearing occurs
diarrhea lasts more than 2 days
If pregnant or breast-feeding, ask a health professional before use.
Directions
- drink plenty of clear fluids to help prevent dehydration caused by diarrhea
- repeat dosage every 1/2 to 1 hour as needed
- do not take more than 8 doses in 24 hours
- use until diarrhea stops but not more than 2 days
- adults and children 12 years and over: 2 tablets
- children under 12 years: ask a doctor
Drug Facts
.
Package Label
NDC: 54738-102-30
– 30 TABLETS
NDC: 54738-102-48- 48 TABLETS
Active Ingredient
(in each tablet)
Bismuth subsalicylate 262 mg
Other Information
- Each tablet contains: calcium 77 mg, salicylate 102 mg
- store at 15°-30°C (59°-86°F)
- protect from moisture
Inactive Ingredient
calcium carbonate, D&C red # 27 (Al-lake), dextrose, flavor (cherry), magnesium stearate, maltodextrin, silicon dioxide, sorbitol
Questions Or Comments
TAMPER EVIDENT: DO NOT USE IF IMPRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
Call 804-270-4498, 8.30 am – 4.30 pm EST Monday - Friday
Structured Label Content
Uses
- Controls diarrhea
- Relieves upset stomach due to overindulgence in food or drink
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Purpose
Anti-diarrheal/Upset stomach reliever
Warnings
Reye’s syndrome: Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye’s syndrome, a rare but serious illness.
Allergy alert: Do not take if you are
- allergic to salicylates (including aspirin)
- taking other salicylate products
Do not use
- If you have bloody or black stool
- If you have an ulcer or bleeding problem
Ask a doctor before use if you have
- Fever
- Mucus in the stool
Ask a doctor or pharmacist before use if you are
- taking any drug for
- anticoagulation (thinning the blood)
- diabetes
- gout
- arthritis
when using this product a temporary, but harmless, darkening of the stool and/or tongue may occur
stop use and ask a doctor if
- symptoms get worse
- ringing in the ears or loss of hearing occurs
diarrhea lasts more than 2 days
If pregnant or breast-feeding, ask a health professional before use.
Directions
- drink plenty of clear fluids to help prevent dehydration caused by diarrhea
- repeat dosage every 1/2 to 1 hour as needed
- do not take more than 8 doses in 24 hours
- use until diarrhea stops but not more than 2 days
- adults and children 12 years and over: 2 tablets
- children under 12 years: ask a doctor
Drug Facts
.
Package Label
NDC: 54738-102-30
– 30 TABLETS
NDC: 54738-102-48- 48 TABLETS
Active Ingredient
(in each tablet)
Bismuth subsalicylate 262 mg
Other Information
- Each tablet contains: calcium 77 mg, salicylate 102 mg
- store at 15°-30°C (59°-86°F)
- protect from moisture
Inactive Ingredient (INACTIVE INGREDIENT)
calcium carbonate, D&C red # 27 (Al-lake), dextrose, flavor (cherry), magnesium stearate, maltodextrin, silicon dioxide, sorbitol
Questions Or Comments (Questions or Comments)
TAMPER EVIDENT: DO NOT USE IF IMPRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
Call 804-270-4498, 8.30 am – 4.30 pm EST Monday - Friday
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:26.108991 · Updated: 2026-03-14T22:54:54.026912