Pecgen®
a627824c-5e3d-4672-a047-6c801b50b432
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dextromethorphan HBr, 10 mg Guaifenesin, 187 mg
Purpose
Antitussive Expectorant
Medication Information
Purpose
Antitussive Expectorant
Description
Dextromethorphan HBr, 10 mg Guaifenesin, 187 mg
Uses
• suppresses cough due to minor throat and bronchial irritation associated with a cold or inhaled irritants.
• helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passages of bothersome mucus, drain bronchial tubes, and make coughs more productive.
Section 42229-5
Contains the same active ingredients as Trispec® DMX*
Sugar, Alcohol, Dye and Gluten FREE
CHERRY RASPBERRY FLAVOR
Manufactured in the USA for Kramer Novis
*Trispec® DMX is a registered trademark of Deliz Pharmaceutical Corp. This product is not manufactured, distributed or marketed by Deliz Pharmaceutical Corp.
Section 50565-1
Keep out of reach of children. In case of accidental overdose, get medical help or contact a Poison Control Center right away.
Purposes
Antitussive
Expectorant
Warnings
Do not use if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
• cough that occurs with too much phlegm (mucus)
• a cough that lasts or is chronic such as occurs with smoking, asthma, chronic bronchitis or emphysema.
When using this product, do not use more than directed.
Stop use and ask a doctor if
• symptoms do not get better within 7 days or are accompanied by fever
• cough lasts more than 7 days, comes back, or is accompanied by fever, rash or persistent headache. These could be signs of a serious condition.
If pregnant or breast-feeding, ask a health professional before use.
Packaging
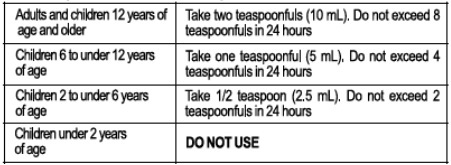
Directions
Take every 4 hours, or as directed by a doctor.
Drug Facts
Other Information
• Store at controlled room temperature 15°-30°C (59°-86°F).
• Avoid excessive heat or humidity.
• Tamper Evident Feature: Do not use if inner seal is torn, broken or missing.
Inactive Ingredients
Citric acid, flavors, menthol, methylparaben, polyethylene glycol, propylene glycol, propylparaben, purified water, sodium citrate, sucralose.
Questions Or Comments?
Call weekdays from 8 AM to 4 PM AST at 1-787-767-2072. San Juan, PR 00917
www.kramernovis.com
Active Ingredients (in Each 5 Ml Tsp)
Dextromethorphan HBr, 10 mg
Guaifenesin, 187 mg
Structured Label Content
Uses
• suppresses cough due to minor throat and bronchial irritation associated with a cold or inhaled irritants.
• helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passages of bothersome mucus, drain bronchial tubes, and make coughs more productive.
Section 42229-5 (42229-5)
Contains the same active ingredients as Trispec® DMX*
Sugar, Alcohol, Dye and Gluten FREE
CHERRY RASPBERRY FLAVOR
Manufactured in the USA for Kramer Novis
*Trispec® DMX is a registered trademark of Deliz Pharmaceutical Corp. This product is not manufactured, distributed or marketed by Deliz Pharmaceutical Corp.
Section 50565-1 (50565-1)
Keep out of reach of children. In case of accidental overdose, get medical help or contact a Poison Control Center right away.
Purposes
Antitussive
Expectorant
Warnings
Do not use if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
• cough that occurs with too much phlegm (mucus)
• a cough that lasts or is chronic such as occurs with smoking, asthma, chronic bronchitis or emphysema.
When using this product, do not use more than directed.
Stop use and ask a doctor if
• symptoms do not get better within 7 days or are accompanied by fever
• cough lasts more than 7 days, comes back, or is accompanied by fever, rash or persistent headache. These could be signs of a serious condition.
If pregnant or breast-feeding, ask a health professional before use.
Packaging
Directions
Take every 4 hours, or as directed by a doctor.
Drug Facts
Other Information (Other information)
• Store at controlled room temperature 15°-30°C (59°-86°F).
• Avoid excessive heat or humidity.
• Tamper Evident Feature: Do not use if inner seal is torn, broken or missing.
Inactive Ingredients (Inactive ingredients)
Citric acid, flavors, menthol, methylparaben, polyethylene glycol, propylene glycol, propylparaben, purified water, sodium citrate, sucralose.
Questions Or Comments? (Questions or comments?)
Call weekdays from 8 AM to 4 PM AST at 1-787-767-2072. San Juan, PR 00917
www.kramernovis.com
Active Ingredients (in Each 5 Ml Tsp) (Active Ingredients (in each 5 mL tsp))
Dextromethorphan HBr, 10 mg
Guaifenesin, 187 mg
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:16.160531 · Updated: 2026-03-14T23:10:31.490874