Triamterene And Hydrochlorothiazide Tablets, Usp 75 Mg/50 Mg
a5525a2a-d2c7-411b-9ffc-0cc4fe77f7c7
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
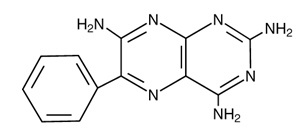
DESCRIPTION: Triamterene and hydrochlorothiazide tablets, USP combine triamterene, a potassium-conserving diuretic, with the natriuretic agent, hydrochlorothiazide. Each Triamterene and hydrochlorothiazide tablet, USP contains: Triamterene, USP ............................................................................... 75 mg Hydrochlorothiazide, USP .................................................................. 50 mg OR Triamterene, USP ................................................................................ 37.5 mg Hydrochlorothiazide, USP .................................................................. 25 mg Triamterene and hydrochlorothiazide tablets, USP for oral administration contain the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, D&C Yellow No. 10 Aluminum Lake, Hypromellose, magnesium stearate and microcrystalline cellulose. Triamterene is 2,4,7-triamino-6-phenylpteridine. Triamterene is practically insoluble in water, benzene, chloroform, ether and dilute alkali hydroxides. It is soluble in formic acid and sparingly soluble in methoxyethanol. Triamterene is very slightly soluble in acetic acid, alcohol and dilute mineral acids. Its molecular weight is 253.27. Its structural formula is: Hydrochlorothiazide is 6-chloro-3,4-dihydro-2 H -1,2,4,benzothiadiazine-7- sulfonamide 1,1-dioxide. Hydrochlorothiazide is slightly soluble in water and freely soluble in sodium hydroxide solution, n-butylamine and dimethylformamide. It is sparingly soluble in methanol and insoluble in ether, chloroform and dilute mineral acids. Its molecular weight is 297.73. Its structural formula is:
Medication Information
Recent Major Changes
WARNINGS: Hyperkalemia:Abnormal elevation of serum potassium levels (greater than or equal to 5.5 mEq/liter) can occur with all potassium-conserving diuretic combinations, including triamterene and hydrochlorothiazide. Hyperkalemia is more likely to occur in patients with renal impairment, diabetes (even without evidence of renal impairment), or elderly or severely ill patients. Since uncorrected hyperkalemia may be fatal, serum potassium levels must be monitored at frequent intervals especially in patients first receiving triamterene and hydrochlorothiazide, when dosages are changed or with any illness that may influence renal function.
Indications and Usage
INDICATIONS AND USAGE: This fixed combination drug is not indicated for the initial therapy of edema or hypertension except in individuals in whom the development of hypokalemia cannot be risked.
- Triamterene and hydrochlorothiazide is indicated for the treatment of hypertension or edema in patients who develop hypokalemia on hydrochlorothiazide alone.
- Triamterene and hydrochlorothiazide is also indicated for those patients who require a thiazide diuretic and in whom the development of hypokalemia cannot be risked (e.g., patients on concomitant digitalis preparations, or with a history of cardiac arrhythmias, etc.).
Triamterene and hydrochlorothiazide may be used alone or in combination with other antihypertensive drugs, such as beta-blockers. Since triamterene and hydrochlorothiazide may enhance the actions of these drugs, dosage adjustments may be necessary.
Dosage and Administration
DOSAGE AND ADMINISTRATION:The usual dose of triamterene and hydrochlorothiazide tablets 37.5mg/25mg is one or two tablets daily, given as a single dose, with appropriate monitoring of serum potassium (see WARNINGS). The usual dose of triamterene and hydrochlorothiazide tablets 75mg/ 50 mg is one tablet daily, with appropriate monitoring of serum potassium (see WARNINGS). There is no experience with the use of more than one triamterene and hydrochlorothiazide tablet 75mg/50mg daily or more than two triamterene and hydrochlorothiazide tablets 37.5mg/25mg daily. Clinical experience with the administration of two triamterene and hydrochlorothiazide tablets 37.5mg/25mg daily in divided doses (rather than as a single dose) suggests an increased risk of electrolyte imbalance and renal dysfunction.
Patients receiving 50 mg of hydrochlorothiazide who become hypokalemic may be transferred to triamterene and hydrochlorothiazide tablets 75mg/50mg directly. Patients receiving 25 mg hydrochlorothiazide who become hypokalemic may be transferred to triamterene and hydrochlorothiazide tablets 37.5mg/25mg directly.
In patients requiring hydrochlorothiazide therapy and in whom hypokalemia cannot be risked therapy may be initiated with triamterene and hydrochlorothiazide tablets 37.5mg/25mg. If an optimal blood pressure response is not obtained with triamterene and hydrochlorothiazide tablets 37.5mg/25mg, the dose should be increased to two triamterene and hydrochlorothiazide tablets 37.5mg/25mg daily as a single dose, or one triamterene and hydrochlorothiazide tablet 75mg/25mg daily. If blood pressure still is not controlled, another antihypertensive agent may be added (see PRECAUTIONS: Drug Interactions).
Clinical studies have shown that patients taking less bioavailable formulations of triamterene and hydrochlorothiazide in daily doses of 25 mg to 50 mg hydrochlorothiazide and 50 mg to 100 mg triamterene may be safely changed to one triamterene and hydrochlorothiazide tablets 37.5mg/25mg daily. All patients changed from less bioavailable formulations to triamterene and hydrochlorothiazide tablets 75mg/50mg should be monitored clinically and for serum potassium after the transfer.
Dosage Forms and Strengths
HOW SUPPLIED:Triamterene and hydrochlorothiazide tablets, USP 75mg/50 mg are yellow colored, oval shaped tablets, debossed with “Λ 135” one side and scored on the other side. Each tablet contains 75 mg of triamterene, USP and 50 mg of hydrochlorothiazide, USP. They are supplied as follows:
NDC 72888-095-30 bottle of 30
NDC 72888-095-01 bottles of 100
NDC 72888-095-05 bottles of 500
NDC 72888-095-00 bottles of 1000
Triamterene and hydrochlorothiazide tablets, USP 37.5mg/25 mg are yellow colored, oval shaped tablets, debossed with “Λ 134” one side and scored on the other side. Each tablet contains 37.5 mg of triamterene, USP and 25 mg of hydrochlorothiazide, USP. They are supplied as follows:
NDC 72888-094-30 bottle of 30
NDC 72888-094-01 bottles of 100
NDC 72888-094-05 bottles of 500
NDC 72888-094-00 bottles of 1000
Store at 20º to 25ºC (68º to 77ºF) [see USP Controlled Room Temperature.]
Protect from light.
Dispense in a tight, light-resistant container as defined in the USP using a child- resistant closure.
Manufactured by:
Rubicon Research Ltd.,
Thane 421506, India.
Distributed by:
Advagen Pharma Ltd.,
East Windsor, NJ 08520, USA.
Rev. 01/2026
Contraindications
CONTRAINDICATIONS: Hyperkalemia:Triamterene and hydrochlorothiazide should not be used in the presence of elevated serum potassium levels (greater than or equal to 5.5 mEq/liter). If hyperkalemia develops, this drug should be discontinued and a thiazide alone should be substituted.
Description
DESCRIPTION:Triamterene and hydrochlorothiazide tablets, USP combine triamterene, a potassium-conserving diuretic, with the natriuretic agent, hydrochlorothiazide.
Each Triamterene and hydrochlorothiazide tablet, USP contains:
Triamterene, USP ............................................................................... 75 mg
Hydrochlorothiazide, USP .................................................................. 50 mg
OR
Triamterene, USP ................................................................................ 37.5 mg
Hydrochlorothiazide, USP .................................................................. 25 mg
Triamterene and hydrochlorothiazide tablets, USP for oral administration contain the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, D&C Yellow No. 10 Aluminum Lake, Hypromellose, magnesium stearate and microcrystalline cellulose.
Triamterene is 2,4,7-triamino-6-phenylpteridine. Triamterene is practically insoluble in water, benzene, chloroform, ether and dilute alkali hydroxides. It is soluble in formic acid and sparingly soluble in methoxyethanol. Triamterene is very slightly soluble in acetic acid, alcohol and dilute mineral acids. Its molecular weight is 253.27. Its structural formula is:
Hydrochlorothiazide is 6-chloro-3,4-dihydro-2 H-1,2,4,benzothiadiazine-7- sulfonamide 1,1-dioxide. Hydrochlorothiazide is slightly soluble in water and freely soluble in sodium hydroxide solution, n-butylamine and dimethylformamide. It is sparingly soluble in methanol and insoluble in ether, chloroform and dilute mineral acids. Its molecular weight is 297.73. Its structural formula is:
Section 34073-7
Drug Interactions:Thiazides may add to or potentiate the action of other antihypertensive drugs.
The thiazides may decrease arterial responsiveness to norepinephrine. This diminution is not sufficient to preclude effectiveness of the pressor agent for therapeutic use. Thiazides have also been shown to increase the responsiveness to tubocurarine.
Lithium generally should not be given with diuretics because they reduce its renal clearance and add a high risk of lithium toxicity. Refer to the package insert on lithium before use of such concomitant therapy.
Acute renal failure has been reported in a few patients receiving indomethacin and formulations containing triamterene and hydrochlorothiazide. Caution is therefore advised when administering non-steroidal anti-inflammatory agents with triamterene and hydrochlorothiazide.
Potassium-sparing agents should be used very cautiously, if at all, in conjunction with angiotensin-converting enzyme (ACE) inhibitors due to a greatly increased risk of hyperkalemia. Serum potassium should be monitored frequently.
Section 34078-6
Nonteratogenic Effects: Thiazides and triamterene have been shown to cross the placental barrier and appear in cord blood. The use of thiazides and triamterene in pregnant women requires that the anticipated benefits be weighed against possible hazards to the fetus. These hazards include fetal or neonatal jaundice, pancreatitis, thrombocytopenia, and possibly other adverse reactions that have occurred in the adult.
Section 34080-2
Nursing Mothers:Thiazides and triamterene in combination have not been studied in nursing mothers. Triamterene appears in animal milk and this may occur in humans. Thiazides are excreted in human breast milk. If use of the combination drug product is deemed essential, the patient should stop nursing.
Section 34081-0
Pediatric Use:Safety and effectiveness in pediatric patients have not been established.
Information for Patients: Non-melanoma Skin Cancer: Instruct patients taking hydrochlorothiazide to protect skin from the sun and undergo regular skin cancer screening.
Section 34084-4
ADVERSE REACTIONS:Side effects observed in association with the use of triamterene and hydrochlorothiazide, other combination products containing triamterene/hydrochlorothiazide, and products containing triamterene or hydrochlorothiazide include the following:
Gastrointestinal:jaundice (intrahepatic cholestatic jaundice), pancreatitis, nausea, appetite disturbance, taste alteration, vomiting, diarrhea, constipation, anorexia, gastric irritation, cramping.
Central Nervous System:drowsiness and fatigue, insomnia, headache, dizziness, dry mouth, depression, anxiety, vertigo, restlessness, paresthesias.
Cardiovascular:tachycardia, shortness of breath and chest pain, orthostatic hypotension (may be aggravated by alcohol, barbiturates or narcotics).
Renal:acute renal failure, acute interstitial nephritis, renal stones composed of triamterene in association with other calculus materials, urine discoloration.
Hematologic:leukopenia, agranulocytosis, thrombocytopenia, aplastic anemia, hemolytic anemia and megaloblastosis.
Ophthalmic:xanthopsia, transient blurred vision.
Hypersensitivity:anaphylaxis, photosensitivity, rash, urticaria, purpura, necrotizing angiitis (vasculitis, cutaneous vasculitis), fever, respiratory distress including pneumonitis.
Other:muscle cramps and weakness, decreased sexual performance and sialadenitis.
Whenever adverse reactions are moderate to severe, therapy should be reduced or withdrawn.
Altered Laboratory Findings:
Serum Electrolytes: hyperkalemia, hypokalemia, hyponatremia, hypomagnesemia, hypochloremia (see WARNINGSand PRECAUTIONS).
Creatinine, Blood Urea Nitrogen: Reversible elevations in BUN and serum creatinine have been observed in hypertensive patients treated with triamterene and hydrochlorothiazide.
Glucose: hyperglycemia, glycosuria and diabetes mellitus (see PRECAUTIONS).
Serum Uric Acid, PBI and Calcium: (see PRECAUTIONS).
Other: Elevated liver enzymes have been reported in patients receiving triamterene and hydrochlorothiazide.
Postmarketing Experience: Non-melanoma Skin Cancer: Hydrochlorothiazide is associated with an increased risk of non-melanoma skin cancer. In a study conducted in the Sentinel System, increased risk was predominantly for squamous cell carcinoma (SCC) and in white patients taking large cumulative doses. The increased risk for SCC in the overall population was approximately 1 additional case per 16,000 patients per year, and for white patients taking a cumulative dose of ≥ 50,000 mg the risk increase was approximately 1 additional SCC case for every 6,700 patients per year.
Section 34088-5
OVERDOSAGE:No specific data are available regarding triamterene and hydrochlorothiazide overdosage in humans and no specific antidote is available.
Fluid and electrolyte imbalances are the most important concern. Excessive doses of the triamterene component may elicit hyperkalemia, dehydration, nausea, vomiting and weakness and possibly hypotension. Overdosing with hydrochlorothiazide has been associated with hypokalemia, hypochloremia, hyponatremia, dehydration, lethargy (may progress to coma) and gastrointestinal irritation. Treatment is symptomatic and supportive. Therapy with triamterene and hydrochlorothiazide should be discontinued. Induce emesis or institute gastric lavage. Monitor serum electrolyte levels and fluid balance. Institute supportive measures as required to maintain hydration, electrolyte balance, respiratory, cardiovascular and renal function.
Section 34090-1
CLINICAL PHARMACOLOGY:Triamterene and hydrochlorothiazide tablets are diuretic, antihypertensive drug product, principally due to its hydrochlorothiazide component; the triamterene component of triamterene and hydrochlorothiazide tablets reduces the excessive potassium loss which may occur with hydrochlorothiazide use.
Section 42229-5
Hydrochlorothiazide:Hydrochlorothiazide is a diuretic and antihypertensive agent. It blocks the renal tubular absorption of sodium and chloride ions. This natriuresis and diuresis is accompanied by a secondary loss of potassium and bicarbonate. Onset of hydrochlorothiazide’s diuretic effect occurs within 2 hours and the peak action takes place in 4 hours. Diuretic activity persists for approximately 6 to 12 hours.
The exact mechanism of hydrochlorothiazide’s antihypertensive action is not known although it may relate to the excretion and redistribution of body sodium. Hydrochlorothiazide does not affect normal blood pressure.
Following oral administration, peak hydrochlorothiazide plasma levels are attained in approximately 2 hours. It is excreted rapidly and unchanged in the urine.
Well controlled studies have demonstrated that doses of hydrochlorothiazide as low as 25 mg given once daily are effective in treating hypertension, but the dose-response has not been clearly established.
Package Label Principal Display Panel
Triamterene and Hydrochlorothiazide Tablets, USP 37.5mg/25mg - NDC 72888-094-30 - 30s Label
Triamterene and Hydrochlorothiazide Tablets, USP 37.5mg/25 mg - NDC 72888-094-01 - 100s Label
Triamterene and Hydrochlorothiazide Tablets, USP 37.5mg/25 mg - NDC 72888-094-05 - 500s Label
Triamterene and Hydrochlorothiazide Tablets, USP 37.5mg/25 mg - NDC 72888-094-00 - 1000s Label
Triamterene and Hydrochlorothiazide Tablets, USP 75mg/50 mg - NDC 72888-095-30 - 30s Label
Triamterene and Hydrochlorothiazide Tablets, USP 75mg/50 mg - NDC 72888-095-01 - 100s Label
Triamterene and Hydrochlorothiazide Tablets, USP 75mg/50 mg - NDC 72888-095-05 - 500s Label
Triamterene and Hydrochlorothiazide Tablets, USP 75mg/50 mg - NDC 72888-095-00 - 1000s Label
Structured Label Content
Recent Major Changes (34066-1)
WARNINGS: Hyperkalemia:Abnormal elevation of serum potassium levels (greater than or equal to 5.5 mEq/liter) can occur with all potassium-conserving diuretic combinations, including triamterene and hydrochlorothiazide. Hyperkalemia is more likely to occur in patients with renal impairment, diabetes (even without evidence of renal impairment), or elderly or severely ill patients. Since uncorrected hyperkalemia may be fatal, serum potassium levels must be monitored at frequent intervals especially in patients first receiving triamterene and hydrochlorothiazide, when dosages are changed or with any illness that may influence renal function.
Indications and Usage (34067-9)
INDICATIONS AND USAGE: This fixed combination drug is not indicated for the initial therapy of edema or hypertension except in individuals in whom the development of hypokalemia cannot be risked.
- Triamterene and hydrochlorothiazide is indicated for the treatment of hypertension or edema in patients who develop hypokalemia on hydrochlorothiazide alone.
- Triamterene and hydrochlorothiazide is also indicated for those patients who require a thiazide diuretic and in whom the development of hypokalemia cannot be risked (e.g., patients on concomitant digitalis preparations, or with a history of cardiac arrhythmias, etc.).
Triamterene and hydrochlorothiazide may be used alone or in combination with other antihypertensive drugs, such as beta-blockers. Since triamterene and hydrochlorothiazide may enhance the actions of these drugs, dosage adjustments may be necessary.
Dosage and Administration (34068-7)
DOSAGE AND ADMINISTRATION:The usual dose of triamterene and hydrochlorothiazide tablets 37.5mg/25mg is one or two tablets daily, given as a single dose, with appropriate monitoring of serum potassium (see WARNINGS). The usual dose of triamterene and hydrochlorothiazide tablets 75mg/ 50 mg is one tablet daily, with appropriate monitoring of serum potassium (see WARNINGS). There is no experience with the use of more than one triamterene and hydrochlorothiazide tablet 75mg/50mg daily or more than two triamterene and hydrochlorothiazide tablets 37.5mg/25mg daily. Clinical experience with the administration of two triamterene and hydrochlorothiazide tablets 37.5mg/25mg daily in divided doses (rather than as a single dose) suggests an increased risk of electrolyte imbalance and renal dysfunction.
Patients receiving 50 mg of hydrochlorothiazide who become hypokalemic may be transferred to triamterene and hydrochlorothiazide tablets 75mg/50mg directly. Patients receiving 25 mg hydrochlorothiazide who become hypokalemic may be transferred to triamterene and hydrochlorothiazide tablets 37.5mg/25mg directly.
In patients requiring hydrochlorothiazide therapy and in whom hypokalemia cannot be risked therapy may be initiated with triamterene and hydrochlorothiazide tablets 37.5mg/25mg. If an optimal blood pressure response is not obtained with triamterene and hydrochlorothiazide tablets 37.5mg/25mg, the dose should be increased to two triamterene and hydrochlorothiazide tablets 37.5mg/25mg daily as a single dose, or one triamterene and hydrochlorothiazide tablet 75mg/25mg daily. If blood pressure still is not controlled, another antihypertensive agent may be added (see PRECAUTIONS: Drug Interactions).
Clinical studies have shown that patients taking less bioavailable formulations of triamterene and hydrochlorothiazide in daily doses of 25 mg to 50 mg hydrochlorothiazide and 50 mg to 100 mg triamterene may be safely changed to one triamterene and hydrochlorothiazide tablets 37.5mg/25mg daily. All patients changed from less bioavailable formulations to triamterene and hydrochlorothiazide tablets 75mg/50mg should be monitored clinically and for serum potassium after the transfer.
Dosage Forms and Strengths (34069-5)
HOW SUPPLIED:Triamterene and hydrochlorothiazide tablets, USP 75mg/50 mg are yellow colored, oval shaped tablets, debossed with “Λ 135” one side and scored on the other side. Each tablet contains 75 mg of triamterene, USP and 50 mg of hydrochlorothiazide, USP. They are supplied as follows:
NDC 72888-095-30 bottle of 30
NDC 72888-095-01 bottles of 100
NDC 72888-095-05 bottles of 500
NDC 72888-095-00 bottles of 1000
Triamterene and hydrochlorothiazide tablets, USP 37.5mg/25 mg are yellow colored, oval shaped tablets, debossed with “Λ 134” one side and scored on the other side. Each tablet contains 37.5 mg of triamterene, USP and 25 mg of hydrochlorothiazide, USP. They are supplied as follows:
NDC 72888-094-30 bottle of 30
NDC 72888-094-01 bottles of 100
NDC 72888-094-05 bottles of 500
NDC 72888-094-00 bottles of 1000
Store at 20º to 25ºC (68º to 77ºF) [see USP Controlled Room Temperature.]
Protect from light.
Dispense in a tight, light-resistant container as defined in the USP using a child- resistant closure.
Manufactured by:
Rubicon Research Ltd.,
Thane 421506, India.
Distributed by:
Advagen Pharma Ltd.,
East Windsor, NJ 08520, USA.
Rev. 01/2026
Contraindications (34070-3)
CONTRAINDICATIONS: Hyperkalemia:Triamterene and hydrochlorothiazide should not be used in the presence of elevated serum potassium levels (greater than or equal to 5.5 mEq/liter). If hyperkalemia develops, this drug should be discontinued and a thiazide alone should be substituted.
Section 34073-7 (34073-7)
Drug Interactions:Thiazides may add to or potentiate the action of other antihypertensive drugs.
The thiazides may decrease arterial responsiveness to norepinephrine. This diminution is not sufficient to preclude effectiveness of the pressor agent for therapeutic use. Thiazides have also been shown to increase the responsiveness to tubocurarine.
Lithium generally should not be given with diuretics because they reduce its renal clearance and add a high risk of lithium toxicity. Refer to the package insert on lithium before use of such concomitant therapy.
Acute renal failure has been reported in a few patients receiving indomethacin and formulations containing triamterene and hydrochlorothiazide. Caution is therefore advised when administering non-steroidal anti-inflammatory agents with triamterene and hydrochlorothiazide.
Potassium-sparing agents should be used very cautiously, if at all, in conjunction with angiotensin-converting enzyme (ACE) inhibitors due to a greatly increased risk of hyperkalemia. Serum potassium should be monitored frequently.
Section 34078-6 (34078-6)
Nonteratogenic Effects: Thiazides and triamterene have been shown to cross the placental barrier and appear in cord blood. The use of thiazides and triamterene in pregnant women requires that the anticipated benefits be weighed against possible hazards to the fetus. These hazards include fetal or neonatal jaundice, pancreatitis, thrombocytopenia, and possibly other adverse reactions that have occurred in the adult.
Section 34080-2 (34080-2)
Nursing Mothers:Thiazides and triamterene in combination have not been studied in nursing mothers. Triamterene appears in animal milk and this may occur in humans. Thiazides are excreted in human breast milk. If use of the combination drug product is deemed essential, the patient should stop nursing.
Section 34081-0 (34081-0)
Pediatric Use:Safety and effectiveness in pediatric patients have not been established.
Information for Patients: Non-melanoma Skin Cancer: Instruct patients taking hydrochlorothiazide to protect skin from the sun and undergo regular skin cancer screening.
Section 34084-4 (34084-4)
ADVERSE REACTIONS:Side effects observed in association with the use of triamterene and hydrochlorothiazide, other combination products containing triamterene/hydrochlorothiazide, and products containing triamterene or hydrochlorothiazide include the following:
Gastrointestinal:jaundice (intrahepatic cholestatic jaundice), pancreatitis, nausea, appetite disturbance, taste alteration, vomiting, diarrhea, constipation, anorexia, gastric irritation, cramping.
Central Nervous System:drowsiness and fatigue, insomnia, headache, dizziness, dry mouth, depression, anxiety, vertigo, restlessness, paresthesias.
Cardiovascular:tachycardia, shortness of breath and chest pain, orthostatic hypotension (may be aggravated by alcohol, barbiturates or narcotics).
Renal:acute renal failure, acute interstitial nephritis, renal stones composed of triamterene in association with other calculus materials, urine discoloration.
Hematologic:leukopenia, agranulocytosis, thrombocytopenia, aplastic anemia, hemolytic anemia and megaloblastosis.
Ophthalmic:xanthopsia, transient blurred vision.
Hypersensitivity:anaphylaxis, photosensitivity, rash, urticaria, purpura, necrotizing angiitis (vasculitis, cutaneous vasculitis), fever, respiratory distress including pneumonitis.
Other:muscle cramps and weakness, decreased sexual performance and sialadenitis.
Whenever adverse reactions are moderate to severe, therapy should be reduced or withdrawn.
Altered Laboratory Findings:
Serum Electrolytes: hyperkalemia, hypokalemia, hyponatremia, hypomagnesemia, hypochloremia (see WARNINGSand PRECAUTIONS).
Creatinine, Blood Urea Nitrogen: Reversible elevations in BUN and serum creatinine have been observed in hypertensive patients treated with triamterene and hydrochlorothiazide.
Glucose: hyperglycemia, glycosuria and diabetes mellitus (see PRECAUTIONS).
Serum Uric Acid, PBI and Calcium: (see PRECAUTIONS).
Other: Elevated liver enzymes have been reported in patients receiving triamterene and hydrochlorothiazide.
Postmarketing Experience: Non-melanoma Skin Cancer: Hydrochlorothiazide is associated with an increased risk of non-melanoma skin cancer. In a study conducted in the Sentinel System, increased risk was predominantly for squamous cell carcinoma (SCC) and in white patients taking large cumulative doses. The increased risk for SCC in the overall population was approximately 1 additional case per 16,000 patients per year, and for white patients taking a cumulative dose of ≥ 50,000 mg the risk increase was approximately 1 additional SCC case for every 6,700 patients per year.
Section 34088-5 (34088-5)
OVERDOSAGE:No specific data are available regarding triamterene and hydrochlorothiazide overdosage in humans and no specific antidote is available.
Fluid and electrolyte imbalances are the most important concern. Excessive doses of the triamterene component may elicit hyperkalemia, dehydration, nausea, vomiting and weakness and possibly hypotension. Overdosing with hydrochlorothiazide has been associated with hypokalemia, hypochloremia, hyponatremia, dehydration, lethargy (may progress to coma) and gastrointestinal irritation. Treatment is symptomatic and supportive. Therapy with triamterene and hydrochlorothiazide should be discontinued. Induce emesis or institute gastric lavage. Monitor serum electrolyte levels and fluid balance. Institute supportive measures as required to maintain hydration, electrolyte balance, respiratory, cardiovascular and renal function.
Description (34089-3)
DESCRIPTION:Triamterene and hydrochlorothiazide tablets, USP combine triamterene, a potassium-conserving diuretic, with the natriuretic agent, hydrochlorothiazide.
Each Triamterene and hydrochlorothiazide tablet, USP contains:
Triamterene, USP ............................................................................... 75 mg
Hydrochlorothiazide, USP .................................................................. 50 mg
OR
Triamterene, USP ................................................................................ 37.5 mg
Hydrochlorothiazide, USP .................................................................. 25 mg
Triamterene and hydrochlorothiazide tablets, USP for oral administration contain the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, D&C Yellow No. 10 Aluminum Lake, Hypromellose, magnesium stearate and microcrystalline cellulose.
Triamterene is 2,4,7-triamino-6-phenylpteridine. Triamterene is practically insoluble in water, benzene, chloroform, ether and dilute alkali hydroxides. It is soluble in formic acid and sparingly soluble in methoxyethanol. Triamterene is very slightly soluble in acetic acid, alcohol and dilute mineral acids. Its molecular weight is 253.27. Its structural formula is:
Hydrochlorothiazide is 6-chloro-3,4-dihydro-2 H-1,2,4,benzothiadiazine-7- sulfonamide 1,1-dioxide. Hydrochlorothiazide is slightly soluble in water and freely soluble in sodium hydroxide solution, n-butylamine and dimethylformamide. It is sparingly soluble in methanol and insoluble in ether, chloroform and dilute mineral acids. Its molecular weight is 297.73. Its structural formula is:
Section 34090-1 (34090-1)
CLINICAL PHARMACOLOGY:Triamterene and hydrochlorothiazide tablets are diuretic, antihypertensive drug product, principally due to its hydrochlorothiazide component; the triamterene component of triamterene and hydrochlorothiazide tablets reduces the excessive potassium loss which may occur with hydrochlorothiazide use.
Section 42229-5 (42229-5)
Hydrochlorothiazide:Hydrochlorothiazide is a diuretic and antihypertensive agent. It blocks the renal tubular absorption of sodium and chloride ions. This natriuresis and diuresis is accompanied by a secondary loss of potassium and bicarbonate. Onset of hydrochlorothiazide’s diuretic effect occurs within 2 hours and the peak action takes place in 4 hours. Diuretic activity persists for approximately 6 to 12 hours.
The exact mechanism of hydrochlorothiazide’s antihypertensive action is not known although it may relate to the excretion and redistribution of body sodium. Hydrochlorothiazide does not affect normal blood pressure.
Following oral administration, peak hydrochlorothiazide plasma levels are attained in approximately 2 hours. It is excreted rapidly and unchanged in the urine.
Well controlled studies have demonstrated that doses of hydrochlorothiazide as low as 25 mg given once daily are effective in treating hypertension, but the dose-response has not been clearly established.
Package Label Principal Display Panel (PACKAGE LABEL-PRINCIPAL DISPLAY PANEL)
Triamterene and Hydrochlorothiazide Tablets, USP 37.5mg/25mg - NDC 72888-094-30 - 30s Label
Triamterene and Hydrochlorothiazide Tablets, USP 37.5mg/25 mg - NDC 72888-094-01 - 100s Label
Triamterene and Hydrochlorothiazide Tablets, USP 37.5mg/25 mg - NDC 72888-094-05 - 500s Label
Triamterene and Hydrochlorothiazide Tablets, USP 37.5mg/25 mg - NDC 72888-094-00 - 1000s Label
Triamterene and Hydrochlorothiazide Tablets, USP 75mg/50 mg - NDC 72888-095-30 - 30s Label
Triamterene and Hydrochlorothiazide Tablets, USP 75mg/50 mg - NDC 72888-095-01 - 100s Label
Triamterene and Hydrochlorothiazide Tablets, USP 75mg/50 mg - NDC 72888-095-05 - 500s Label
Triamterene and Hydrochlorothiazide Tablets, USP 75mg/50 mg - NDC 72888-095-00 - 1000s Label
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:52:03.824731 · Updated: 2026-03-14T22:42:38.249942