These Highlights Do Not Include All The Information Needed To Use Regadenoson Injection Safely And Effectively. See Full Prescribing Information For Regadenoson Injection.
a550b451-fb30-4c1c-a60e-6e40fba20c88
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Regadenoson Injection is a pharmacologic stress agent indicated for radionuclide myocardial perfusion imaging (MPI) in patients unable to undergo adequate exercise stress.
Indications and Usage
Regadenoson Injection is a pharmacologic stress agent indicated for radionuclide myocardial perfusion imaging (MPI) in patients unable to undergo adequate exercise stress.
Dosage and Administration
The recommended dose of regadenoson injection is 5 mL (0.4 mg regadenoson) administered as an intravenous injection within 10 seconds. Patients should be instructed to avoid consumption of any products containing methylxanthines, including caffeinated coffee, tea or other caffeinated beverages, caffeine-containing drug products, aminophylline and theophylline for at least 12 hours before a scheduled radionuclide MPI [see Drug Interactions ( 7.1 ) and Clinical Pharmacology ( 12.2 )]. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Do not administer regadenoson injection if it contains particulate matter or is discolored. Administer regadenoson injection as an intravenous injection within 10 seconds into a peripheral vein using a 22 gauge or larger catheter or needle. Administer a 5 mL saline flush immediately after the injection of regadenoson injection. Administer the radionuclide myocardial perfusion imaging agent 10–20 seconds after the saline flush. The radionuclide may be injected directly into the same catheter as regadenoson injection.
Warnings and Precautions
Myocardial Ischemia. Fatal cardiac events have occurred. Avoid use in patients with symptoms or signs of acute myocardial ischemia, for example unstable angina or cardiovascular instability, who may be at greater risk. Cardiac resuscitation equipment and trained staff should be available before administration ( 5.1 ). Sinoatrial (SA) and Atrioventricular (AV) Nodal Block. Adenosine receptor agonists, including regadenoson, can depress the SA and AV nodes and may cause first-, second- or third-degree AV block, or sinus bradycardia ( 5.2 ). Atrial Fibrillation/Atrial Flutter. New-onset or recurrent atrial fibrillation with rapid ventricular response and atrial flutter have been reported ( 5.3 ). Hypersensitivity, including anaphylaxis, angioedema, cardiac or respiratory arrest, respiratory distress, decreased oxygen saturation, hypotension, throat tightness, urticaria, and rashes have occurred. Have personnel and resuscitative equipment immediately available ( 5.4 ). Hypotension. Adenosine receptor agonists, including regadenoson, induce vasodilation and hypotension. The risk of serious hypotension may be higher in patients with autonomic dysfunction, stenotic valvular heart disease, pericarditis or pericardial effusions, stenotic carotid artery disease with cerebrovascular insufficiency, or hypovolemia ( 5.5 ). Hypertension. Adenosine receptor agonists, including regadenoson, may induce clinically significant increases in blood pressure particularly in patients with a history of hypertension and when the MPI includes low level exercise ( 5.6 ). Bronchoconstriction. Adenosine receptor agonists, including regadenoson, may induce dyspnea, bronchoconstriction and respiratory compromise in patients with chronic obstructive pulmonary disease (COPD) or asthma. Resuscitative measures should be available ( 5.7 ). Seizure. Regadenoson may lower the seizure threshold. New onset or recurrence of convulsive seizures has occurred. Some seizures are prolonged and require urgent anticonvulsive management. Methylxanthine use is not recommended in patients who experience a seizure in association with regadenoson ( 5.8 ). Cerebrovascular Accident (Stroke). Hemorrhagic and ischemic cerebrovascular accidents have occurred ( 5.9 ).
Contraindications
Do not administer regadenoson to patients with: Second- or third-degree AV block, or sinus node dysfunction unless these patients have a functioning artificial pacemaker [see Warnings and Precautions ( 5.2 )].
Adverse Reactions
The following adverse reactions are discussed in more detail in other sections of the labeling. Myocardial Ischemia [see Warnings and Precautions ( 5.1 )] Sinoatrial and Atrioventricular Nodal Block [see Warnings and Precautions ( 5.2 )] Atrial Fibrillation/Atrial Flutter [see Warnings and Precautions ( 5.3 )] Hypersensitivity, Including Anaphylaxis [see Warnings and Precautions ( 5.4 )] Hypotension [see Warnings and Precautions ( 5.5 )] Hypertension [see Warnings and Precautions ( 5.6 )] Bronchoconstriction [see Warnings and Precautions ( 5.7 )] Seizure [see Warnings and Precautions ( 5.8 )] Cerebrovascular Accident (Stroke) [see Warnings and Precautions ( 5.9 )]
Drug Interactions
No formal pharmacokinetic drug interaction studies have been conducted with regadenoson.
Storage and Handling
Regadenoson Injection, a clear, colorless solution, is supplied as follows: NDC Regadenoson Injection (0.08 mg per mL) Package Factor 71288- 201 -85 0.4 mg regadenoson per 5 mL Single-Dose Pre-filled Syringe 1 syringe per carton Store at 20° to 25°C (68° to 77°F); excursions permitted 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature.] Discard unused portion. Sterile, Nonpyrogenic, Preservative-free. The container closure is not made with natural rubber latex.
How Supplied
Regadenoson Injection, a clear, colorless solution, is supplied as follows: NDC Regadenoson Injection (0.08 mg per mL) Package Factor 71288- 201 -85 0.4 mg regadenoson per 5 mL Single-Dose Pre-filled Syringe 1 syringe per carton Store at 20° to 25°C (68° to 77°F); excursions permitted 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature.] Discard unused portion. Sterile, Nonpyrogenic, Preservative-free. The container closure is not made with natural rubber latex.
Medication Information
Warnings and Precautions
Myocardial Ischemia. Fatal cardiac events have occurred. Avoid use in patients with symptoms or signs of acute myocardial ischemia, for example unstable angina or cardiovascular instability, who may be at greater risk. Cardiac resuscitation equipment and trained staff should be available before administration ( 5.1 ). Sinoatrial (SA) and Atrioventricular (AV) Nodal Block. Adenosine receptor agonists, including regadenoson, can depress the SA and AV nodes and may cause first-, second- or third-degree AV block, or sinus bradycardia ( 5.2 ). Atrial Fibrillation/Atrial Flutter. New-onset or recurrent atrial fibrillation with rapid ventricular response and atrial flutter have been reported ( 5.3 ). Hypersensitivity, including anaphylaxis, angioedema, cardiac or respiratory arrest, respiratory distress, decreased oxygen saturation, hypotension, throat tightness, urticaria, and rashes have occurred. Have personnel and resuscitative equipment immediately available ( 5.4 ). Hypotension. Adenosine receptor agonists, including regadenoson, induce vasodilation and hypotension. The risk of serious hypotension may be higher in patients with autonomic dysfunction, stenotic valvular heart disease, pericarditis or pericardial effusions, stenotic carotid artery disease with cerebrovascular insufficiency, or hypovolemia ( 5.5 ). Hypertension. Adenosine receptor agonists, including regadenoson, may induce clinically significant increases in blood pressure particularly in patients with a history of hypertension and when the MPI includes low level exercise ( 5.6 ). Bronchoconstriction. Adenosine receptor agonists, including regadenoson, may induce dyspnea, bronchoconstriction and respiratory compromise in patients with chronic obstructive pulmonary disease (COPD) or asthma. Resuscitative measures should be available ( 5.7 ). Seizure. Regadenoson may lower the seizure threshold. New onset or recurrence of convulsive seizures has occurred. Some seizures are prolonged and require urgent anticonvulsive management. Methylxanthine use is not recommended in patients who experience a seizure in association with regadenoson ( 5.8 ). Cerebrovascular Accident (Stroke). Hemorrhagic and ischemic cerebrovascular accidents have occurred ( 5.9 ).
Indications and Usage
Regadenoson Injection is a pharmacologic stress agent indicated for radionuclide myocardial perfusion imaging (MPI) in patients unable to undergo adequate exercise stress.
Dosage and Administration
The recommended dose of regadenoson injection is 5 mL (0.4 mg regadenoson) administered as an intravenous injection within 10 seconds. Patients should be instructed to avoid consumption of any products containing methylxanthines, including caffeinated coffee, tea or other caffeinated beverages, caffeine-containing drug products, aminophylline and theophylline for at least 12 hours before a scheduled radionuclide MPI [see Drug Interactions ( 7.1 ) and Clinical Pharmacology ( 12.2 )]. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Do not administer regadenoson injection if it contains particulate matter or is discolored. Administer regadenoson injection as an intravenous injection within 10 seconds into a peripheral vein using a 22 gauge or larger catheter or needle. Administer a 5 mL saline flush immediately after the injection of regadenoson injection. Administer the radionuclide myocardial perfusion imaging agent 10–20 seconds after the saline flush. The radionuclide may be injected directly into the same catheter as regadenoson injection.
Contraindications
Do not administer regadenoson to patients with: Second- or third-degree AV block, or sinus node dysfunction unless these patients have a functioning artificial pacemaker [see Warnings and Precautions ( 5.2 )].
Adverse Reactions
The following adverse reactions are discussed in more detail in other sections of the labeling. Myocardial Ischemia [see Warnings and Precautions ( 5.1 )] Sinoatrial and Atrioventricular Nodal Block [see Warnings and Precautions ( 5.2 )] Atrial Fibrillation/Atrial Flutter [see Warnings and Precautions ( 5.3 )] Hypersensitivity, Including Anaphylaxis [see Warnings and Precautions ( 5.4 )] Hypotension [see Warnings and Precautions ( 5.5 )] Hypertension [see Warnings and Precautions ( 5.6 )] Bronchoconstriction [see Warnings and Precautions ( 5.7 )] Seizure [see Warnings and Precautions ( 5.8 )] Cerebrovascular Accident (Stroke) [see Warnings and Precautions ( 5.9 )]
Drug Interactions
No formal pharmacokinetic drug interaction studies have been conducted with regadenoson.
Storage and Handling
Regadenoson Injection, a clear, colorless solution, is supplied as follows: NDC Regadenoson Injection (0.08 mg per mL) Package Factor 71288- 201 -85 0.4 mg regadenoson per 5 mL Single-Dose Pre-filled Syringe 1 syringe per carton Store at 20° to 25°C (68° to 77°F); excursions permitted 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature.] Discard unused portion. Sterile, Nonpyrogenic, Preservative-free. The container closure is not made with natural rubber latex.
How Supplied
Regadenoson Injection, a clear, colorless solution, is supplied as follows: NDC Regadenoson Injection (0.08 mg per mL) Package Factor 71288- 201 -85 0.4 mg regadenoson per 5 mL Single-Dose Pre-filled Syringe 1 syringe per carton Store at 20° to 25°C (68° to 77°F); excursions permitted 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature.] Discard unused portion. Sterile, Nonpyrogenic, Preservative-free. The container closure is not made with natural rubber latex.
Description
Regadenoson Injection is a pharmacologic stress agent indicated for radionuclide myocardial perfusion imaging (MPI) in patients unable to undergo adequate exercise stress.
Section 42229-5
ECG Abnormalities
The frequency of rhythm or conduction abnormalities following regadenoson or Adenoscan is shown in Table 2 [see Warnings and Precautions (5.2)].
|
* 12-lead ECGs were recorded before and for up to 2 hours after dosing. |
||
|
† includes rhythm abnormalities (PACs, PVCs, atrial fibrillation/flutter, wandering atrial pacemaker, supraventricular or ventricular arrhythmia) or conduction abnormalities, including AV block. |
||
|
Regadenoson
N / N evaluable (%) |
Adenoscan
N / N evaluable (%) |
|
| Rhythm or conduction abnormalities† | 332/1275 (26%) | 192/645 (30%) |
| Rhythm abnormalities | 260/1275 (20%) | 131/645 (20%) |
| PACs | 86/1274 (7%) | 57/645 (9%) |
| PVCs | 179/1274 (14%) | 79/645 (12%) |
| First-degree AV block (PR prolongation > 220 msec) | 34/1209 (3%) | 43/618 (7%) |
| Second-degree AV block | 1/1209 (0.1%) | 9/618 (1%) |
| AV conduction abnormalities (other than AV blocks) | 1/1209 (0.1%) | 0/618 (0%) |
| Ventricular conduction abnormalities | 64/1152 (6%) | 31/581 (5%) |
5.8 Seizure
Regadenoson may lower the seizure threshold; obtain a seizure history. New-onset or recurrence of convulsive seizures has occurred following regadenoson injection. Some seizures are prolonged and require emergent anticonvulsive management. Aminophylline may increase the risk of seizures associated with regadenoson injection. Methylxanthine use is not recommended in patients who experience a seizure in association with regadenoson administration.
10 Overdosage
Regadenoson overdosage may result in serious reactions [see Warnings and Precautions (5)]. In a study of healthy volunteers, symptoms of flushing, dizziness and increased heart rate were assessed as intolerable at regadenoson doses greater than 0.02 mg/kg.
11 Description
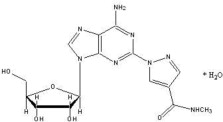
Regadenoson is an A2A adenosine receptor agonist that is a coronary vasodilator [see Clinical Pharmacology (12.1)]. Regadenoson is chemically described as adenosine, 2-[4-[(methylamino)carbonyl]-1H-pyrazol-1-yl]-, monohydrate. Its structural formula is:
The molecular formula for regadenoson is C15H18N8O5 • H2O and its molecular weight is 408.37. Regadenoson Injection is a sterile, nonpyrogenic solution for intravenous injection. The solution is clear and colorless. Each 1 mL in the 5 mL pre-filled syringe contains 0.084 mg of regadenoson monohydrate, corresponding to 0.08 mg regadenoson on an anhydrous basis, 8.7 mg dibasic sodium phosphate anhydrous, 5.4 mg monobasic sodium phosphate monohydrate, 150 mg propylene glycol, 1 mg edetate disodium dihydrate, and water for injection, with pH between 6.3 and 7.7.
5.5 Hypotension
Adenosine receptor agonists, including regadenoson, induce arterial vasodilation and hypotension. In clinical trials, decreased systolic blood pressure (> 35 mm Hg) was observed in 7% of patients and decreased diastolic blood pressure (> 25 mm Hg) was observed in 4% of patients within 45 minutes of regadenoson administration. The risk of serious hypotension may be higher in patients with autonomic dysfunction, hypovolemia, left main coronary artery stenosis, stenotic valvular heart disease, pericarditis or pericardial effusions, or stenotic carotid artery disease with cerebrovascular insufficiency. In post-marketing experience, syncope, transient ischemic attacks and seizures have been observed [see Adverse Reactions (6.2)].
5.6 Hypertension
Administration of adenosine receptor agonists, including regadenoson, may result in clinically significant increases in blood pressure in some patients. Among patients who experienced an increase in blood pressure in clinical trials, the increase was observed within minutes of regadenoson administration. Most increases resolved within 10 to 15 minutes, but in some cases, increases were observed at 45 minutes following administration [see Clinical Pharmacology (12.2)]. In post-marketing experience, cases of potentially clinically significant hypertension have been reported, particularly with underlying hypertension and when low-level exercise was included in the MPI [see Adverse Reactions (6.2)].
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Of the 1,337 patients receiving regadenoson in Studies 1 and 2, 56% were 65 years of age and over and 24% were 75 years of age and over. Older patients (≥ 75 years of age) had a similar adverse event profile compared to younger patients (< 65 years of age), but had a higher incidence of hypotension (2% vs. ≤ 1%).
4 Contraindications
Do not administer regadenoson to patients with:
- Second- or third-degree AV block, or
- sinus node dysfunction
unless these patients have a functioning artificial pacemaker [see Warnings and Precautions (5.2)].
6 Adverse Reactions
The following adverse reactions are discussed in more detail in other sections of the labeling.
- Myocardial Ischemia [see Warnings and Precautions (5.1)]
- Sinoatrial and Atrioventricular Nodal Block [see Warnings and Precautions (5.2)]
- Atrial Fibrillation/Atrial Flutter [see Warnings and Precautions (5.3)]
- Hypersensitivity, Including Anaphylaxis [see Warnings and Precautions (5.4)]
- Hypotension [see Warnings and Precautions (5.5)]
- Hypertension [see Warnings and Precautions (5.6)]
- Bronchoconstriction [see Warnings and Precautions (5.7)]
- Seizure [see Warnings and Precautions (5.8)]
- Cerebrovascular Accident (Stroke) [see Warnings and Precautions (5.9)]
7 Drug Interactions
No formal pharmacokinetic drug interaction studies have been conducted with regadenoson.
8.6 Renal Impairment
No dose adjustment is needed in patients with renal impairment including patients with end stage renal disease and/or dependent on dialysis [see Pharmacokinetics (12.3)].
12.3 Pharmacokinetics
In healthy subjects, the regadenoson plasma concentration-time profile is multi-exponential in nature and best characterized by 3-compartment model. The maximal plasma concentration of regadenoson is achieved within 1 to 4 minutes after injection of regadenoson and parallels the onset of the pharmacodynamic response. The half-life of this initial phase is approximately 2 to 4 minutes. An intermediate phase follows, with a half-life on average of 30 minutes coinciding with loss of the pharmacodynamic effect. The terminal phase consists of a decline in plasma concentration with a half-life of approximately 2 hours [see Clinical Pharmacology (12.2)]. Within the dose range of 0.3–20 mcg/kg in healthy subjects, clearance, terminal half-life or volume of distribution do not appear dependent upon the dose.
A population pharmacokinetic analysis including data from subjects and patients demonstrated that regadenoson clearance decreases in parallel with a reduction in creatinine clearance and clearance increases with increased body weight. Age, gender, and race have minimal effects on the pharmacokinetics of regadenoson.
1 Indications and Usage
Regadenoson Injection is a pharmacologic stress agent indicated for radionuclide myocardial perfusion imaging (MPI) in patients unable to undergo adequate exercise stress.
5.1 Myocardial Ischemia
Fatal and nonfatal myocardial infarction (MI), ventricular arrhythmias, and cardiac arrest have occurred following regadenoson injection. Avoid use in patients with symptoms or signs of acute myocardial ischemia, for example unstable angina or cardiovascular instability; these patients may be at greater risk of serious cardiovascular reactions to regadenoson. Cardiac resuscitation equipment and trained staff should be available before administering regadenoson. Adhere to the recommended duration of injection [see Dosage and Administration (2)]. As noted in an animal study, longer injection times may increase the duration and magnitude of increase in coronary blood flow [see Clinical Pharmacology (12.2)]. If serious reactions to regadenoson occur, consider the use of aminophylline, an adenosine antagonist, to shorten the duration of increased coronary blood flow induced by regadenoson [see Overdosage (10)].
5.7 Bronchoconstriction
Adenosine receptor agonists, including regadenoson, may cause dyspnea, bronchoconstriction, and respiratory compromise. Appropriate bronchodilator therapy and resuscitative measures should be available prior to and following regadenoson administration [see Adverse Reactions (6.1), Clinical Pharmacology (12.2), Overdosage (10) and Patient Counseling Information (17)].
12.1 Mechanism of Action
Regadenoson is a low affinity agonist (Ki ≈ 1.3 μM) for the A2A adenosine receptor, with at least 10-fold lower affinity for the A1 adenosine receptor (Ki > 16.5 μM), and weak, if any, affinity for the A2B and A3 adenosine receptors. Activation of the A2A adenosine receptor by regadenoson produces coronary vasodilation and increases coronary blood flow (CBF).
5 Warnings and Precautions
- Myocardial Ischemia. Fatal cardiac events have occurred. Avoid use in patients with symptoms or signs of acute myocardial ischemia, for example unstable angina or cardiovascular instability, who may be at greater risk. Cardiac resuscitation equipment and trained staff should be available before administration (5.1).
- Sinoatrial (SA) and Atrioventricular (AV) Nodal Block. Adenosine receptor agonists, including regadenoson, can depress the SA and AV nodes and may cause first-, second- or third-degree AV block, or sinus bradycardia (5.2).
- Atrial Fibrillation/Atrial Flutter. New-onset or recurrent atrial fibrillation with rapid ventricular response and atrial flutter have been reported (5.3).
- Hypersensitivity, including anaphylaxis, angioedema, cardiac or respiratory arrest, respiratory distress, decreased oxygen saturation, hypotension, throat tightness, urticaria, and rashes have occurred. Have personnel and resuscitative equipment immediately available (5.4).
- Hypotension. Adenosine receptor agonists, including regadenoson, induce vasodilation and hypotension. The risk of serious hypotension may be higher in patients with autonomic dysfunction, stenotic valvular heart disease, pericarditis or pericardial effusions, stenotic carotid artery disease with cerebrovascular insufficiency, or hypovolemia (5.5).
- Hypertension. Adenosine receptor agonists, including regadenoson, may induce clinically significant increases in blood pressure particularly in patients with a history of hypertension and when the MPI includes low level exercise (5.6).
- Bronchoconstriction. Adenosine receptor agonists, including regadenoson, may induce dyspnea, bronchoconstriction and respiratory compromise in patients with chronic obstructive pulmonary disease (COPD) or asthma. Resuscitative measures should be available (5.7).
- Seizure. Regadenoson may lower the seizure threshold. New onset or recurrence of convulsive seizures has occurred. Some seizures are prolonged and require urgent anticonvulsive management. Methylxanthine use is not recommended in patients who experience a seizure in association with regadenoson (5.8).
- Cerebrovascular Accident (Stroke). Hemorrhagic and ischemic cerebrovascular accidents have occurred (5.9).
2 Dosage and Administration
The recommended dose of regadenoson injection is 5 mL (0.4 mg regadenoson) administered as an intravenous injection within 10 seconds.
- Patients should be instructed to avoid consumption of any products containing methylxanthines, including caffeinated coffee, tea or other caffeinated beverages, caffeine-containing drug products, aminophylline and theophylline for at least 12 hours before a scheduled radionuclide MPI [see Drug Interactions (7.1) and Clinical Pharmacology (12.2)].
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Do not administer regadenoson injection if it contains particulate matter or is discolored.
- Administer regadenoson injection as an intravenous injection within 10 seconds into a peripheral vein using a 22 gauge or larger catheter or needle.
- Administer a 5 mL saline flush immediately after the injection of regadenoson injection.
- Administer the radionuclide myocardial perfusion imaging agent 10–20 seconds after the saline flush. The radionuclide may be injected directly into the same catheter as regadenoson injection.
3 Dosage Forms and Strengths
- Single-dose pre-filled syringe: clear, colorless solution containing regadenoson 0.4 mg per 5 mL (0.08 mg per mL).
6.2 Post Marketing Experience
The following adverse reactions have been reported from worldwide marketing experience with regadenoson. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
During clinical development, 1,651 patients were exposed to regadenoson, with most receiving 0.4 mg as a rapid (≤ 10 seconds) intravenous injection. Most of these patients received regadenoson in two clinical studies that enrolled patients who had no history of bronchospastic lung disease as well as no history of a cardiac conduction block of greater than first-degree AV block, except for patients with functioning artificial pacemakers. In these studies (Studies 1 and 2), 2,015 patients underwent myocardial perfusion imaging after administration of regadenoson (N = 1,337) or Adenoscan® (N = 678). The population was 26–93 years of age (median 66 years), 70% male and primarily Caucasian (76% Caucasian, 7% African American, 9% Hispanic, 5% Asian). Table 1 shows the most frequently reported adverse reactions.
Overall, any adverse reaction occurred at similar rates between the study groups (80% for the regadenoson group and 83% for the Adenoscan group). Aminophylline was used to treat the reactions in 3% of patients in the regadenoson group and 2% of patients in the Adenoscan group. Most adverse reactions began soon after dosing, and generally resolved within approximately 15 minutes, except for headache which resolved in most patients within 30 minutes.
|
Regadenoson
N = 1,337 |
Adenoscan
N = 678 |
|
| Dyspnea | 28% | 26% |
| Headache | 26% | 17% |
| Flushing | 16% | 25% |
| Chest Discomfort | 13% | 18% |
| Angina Pectoris or ST Segment Depression | 12% | 18% |
| Dizziness | 8% | 7% |
| Chest Pain | 7% | 10% |
| Nausea | 6% | 6% |
| Abdominal Discomfort | 5% | 2% |
| Dysgeusia | 5% | 7% |
| Feeling Hot | 5% | 8% |
16 How Supplied/storage and Handling
Regadenoson Injection, a clear, colorless solution, is supplied as follows:
| NDC | Regadenoson Injection (0.08 mg per mL) | Package Factor |
| 71288-201-85 | 0.4 mg regadenoson per 5 mL Single-Dose Pre-filled Syringe | 1 syringe per carton |
Store at 20° to 25°C (68° to 77°F); excursions permitted 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature.]
Discard unused portion.
Sterile, Nonpyrogenic, Preservative-free.
The container closure is not made with natural rubber latex.
5.9 Cerebrovascular Accident (stroke)
5.3 Atrial Fibrillation/atrial Flutter
New-onset or recurrent atrial fibrillation with rapid ventricular response and atrial flutter have been reported following regadenoson injection [see Adverse Reactions (6.2)].
7.2 Effect of Regadenoson On Other Drugs
Regadenoson does not inhibit the metabolism of substrates for CYP1A2, CYP2C8, CYP2C9, CYP2C19, CYP2D6, or CYP3A4 in human liver microsomes, indicating that it is unlikely to alter the pharmacokinetics of drugs metabolized by these cytochrome P450 enzymes.
7.1 Effects of Other Drugs On Regadenoson
- Methylxanthines (e.g., caffeine, aminophylline and theophylline) are non-specific adenosine receptor antagonists that interfere with the vasodilation activity of regadenoson [see Clinical Pharmacology (12.2) and Patient Counseling Information (17)]. Patients should avoid consumption of any products containing methylxanthines as well as any drugs containing theophylline or aminophylline for at least 12 hours before regadenoson administration. Aminophylline may be used to attenuate severe or persistent adverse reactions to regadenoson [see Overdosage (10)].
- In clinical studies, regadenoson was administered to patients taking other cardioactive drugs (i.e., β-blockers, calcium channel blockers, ACE inhibitors, nitrates, cardiac glycosides, and angiotensin receptor blockers) without reported adverse reactions or apparent effects on efficacy.
- Dipyridamole may change the effects of regadenoson. When possible, withhold dipyridamole for at least two days prior to regadenoson administration.
5.4 Hypersensitivity, Including Anaphylaxis
Anaphylaxis, angioedema, cardiac or respiratory arrest, respiratory distress, decreased oxygen saturation, hypotension, throat tightness, urticaria and rashes have occurred. In clinical trials, hypersensitivity reactions were reported in fewer than 1 percent of patients [see Adverse Reactions (6.1)]. Have personnel and resuscitative equipment immediately available.
5.2 Sinoatrial and Atrioventricular Nodal Block
Adenosine receptor agonists, including regadenoson, can depress the SA and AV nodes and may cause first-, second- or third-degree AV block, or sinus bradycardia requiring intervention. In clinical trials first-degree AV block (PR prolongation > 220 msec) developed in 3% of patients within 2 hours of regadenoson administration; transient second-degree AV block with one dropped beat was observed in one patient receiving regadenoson. In post-marketing experience, third-degree heart block and asystole within minutes of regadenoson administration have occurred [see Adverse Reactions (6.2)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Regadenoson was negative in the Ames bacterial mutation assay, chromosomal aberration assay in Chinese hamster ovary (CHO) cells, and mouse bone marrow micronucleus assay.
Long-term animal studies have not been conducted to evaluate regadenoson's carcinogenic potential or potential effects on fertility.
Principal Display Panel – Regadenoson Injection 5 Ml Carton
NDC 71288-201-85
1 x 5 mL Single-Dose Pre-filled Syringe
Discard unused portion
Rx only
Regadenoson Injection
0.4 mg per 5 mL
(0.08 mg per mL)
For Intravenous Use Only
Inject 5 mL intravenously within 10 seconds.
Follow immediately with saline flush and radiopharmaceutical.
Principal Display Panel – Regadenoson Injection 5 Ml Syringe Label
NDC 71288-201-85
Rx only
Regadenoson Injection
0.4 mg per 5 mL
(0.08 mg per mL)
For Intravenous Use Only
5 mL Single-Dose Pre-filled Syringe
Discard unused portion
Structured Label Content
Section 42229-5 (42229-5)
ECG Abnormalities
The frequency of rhythm or conduction abnormalities following regadenoson or Adenoscan is shown in Table 2 [see Warnings and Precautions (5.2)].
|
* 12-lead ECGs were recorded before and for up to 2 hours after dosing. |
||
|
† includes rhythm abnormalities (PACs, PVCs, atrial fibrillation/flutter, wandering atrial pacemaker, supraventricular or ventricular arrhythmia) or conduction abnormalities, including AV block. |
||
|
Regadenoson
N / N evaluable (%) |
Adenoscan
N / N evaluable (%) |
|
| Rhythm or conduction abnormalities† | 332/1275 (26%) | 192/645 (30%) |
| Rhythm abnormalities | 260/1275 (20%) | 131/645 (20%) |
| PACs | 86/1274 (7%) | 57/645 (9%) |
| PVCs | 179/1274 (14%) | 79/645 (12%) |
| First-degree AV block (PR prolongation > 220 msec) | 34/1209 (3%) | 43/618 (7%) |
| Second-degree AV block | 1/1209 (0.1%) | 9/618 (1%) |
| AV conduction abnormalities (other than AV blocks) | 1/1209 (0.1%) | 0/618 (0%) |
| Ventricular conduction abnormalities | 64/1152 (6%) | 31/581 (5%) |
5.8 Seizure
Regadenoson may lower the seizure threshold; obtain a seizure history. New-onset or recurrence of convulsive seizures has occurred following regadenoson injection. Some seizures are prolonged and require emergent anticonvulsive management. Aminophylline may increase the risk of seizures associated with regadenoson injection. Methylxanthine use is not recommended in patients who experience a seizure in association with regadenoson administration.
10 Overdosage (10 OVERDOSAGE)
Regadenoson overdosage may result in serious reactions [see Warnings and Precautions (5)]. In a study of healthy volunteers, symptoms of flushing, dizziness and increased heart rate were assessed as intolerable at regadenoson doses greater than 0.02 mg/kg.
11 Description (11 DESCRIPTION)
Regadenoson is an A2A adenosine receptor agonist that is a coronary vasodilator [see Clinical Pharmacology (12.1)]. Regadenoson is chemically described as adenosine, 2-[4-[(methylamino)carbonyl]-1H-pyrazol-1-yl]-, monohydrate. Its structural formula is:
The molecular formula for regadenoson is C15H18N8O5 • H2O and its molecular weight is 408.37. Regadenoson Injection is a sterile, nonpyrogenic solution for intravenous injection. The solution is clear and colorless. Each 1 mL in the 5 mL pre-filled syringe contains 0.084 mg of regadenoson monohydrate, corresponding to 0.08 mg regadenoson on an anhydrous basis, 8.7 mg dibasic sodium phosphate anhydrous, 5.4 mg monobasic sodium phosphate monohydrate, 150 mg propylene glycol, 1 mg edetate disodium dihydrate, and water for injection, with pH between 6.3 and 7.7.
5.5 Hypotension
Adenosine receptor agonists, including regadenoson, induce arterial vasodilation and hypotension. In clinical trials, decreased systolic blood pressure (> 35 mm Hg) was observed in 7% of patients and decreased diastolic blood pressure (> 25 mm Hg) was observed in 4% of patients within 45 minutes of regadenoson administration. The risk of serious hypotension may be higher in patients with autonomic dysfunction, hypovolemia, left main coronary artery stenosis, stenotic valvular heart disease, pericarditis or pericardial effusions, or stenotic carotid artery disease with cerebrovascular insufficiency. In post-marketing experience, syncope, transient ischemic attacks and seizures have been observed [see Adverse Reactions (6.2)].
5.6 Hypertension
Administration of adenosine receptor agonists, including regadenoson, may result in clinically significant increases in blood pressure in some patients. Among patients who experienced an increase in blood pressure in clinical trials, the increase was observed within minutes of regadenoson administration. Most increases resolved within 10 to 15 minutes, but in some cases, increases were observed at 45 minutes following administration [see Clinical Pharmacology (12.2)]. In post-marketing experience, cases of potentially clinically significant hypertension have been reported, particularly with underlying hypertension and when low-level exercise was included in the MPI [see Adverse Reactions (6.2)].
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Of the 1,337 patients receiving regadenoson in Studies 1 and 2, 56% were 65 years of age and over and 24% were 75 years of age and over. Older patients (≥ 75 years of age) had a similar adverse event profile compared to younger patients (< 65 years of age), but had a higher incidence of hypotension (2% vs. ≤ 1%).
4 Contraindications (4 CONTRAINDICATIONS)
Do not administer regadenoson to patients with:
- Second- or third-degree AV block, or
- sinus node dysfunction
unless these patients have a functioning artificial pacemaker [see Warnings and Precautions (5.2)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in more detail in other sections of the labeling.
- Myocardial Ischemia [see Warnings and Precautions (5.1)]
- Sinoatrial and Atrioventricular Nodal Block [see Warnings and Precautions (5.2)]
- Atrial Fibrillation/Atrial Flutter [see Warnings and Precautions (5.3)]
- Hypersensitivity, Including Anaphylaxis [see Warnings and Precautions (5.4)]
- Hypotension [see Warnings and Precautions (5.5)]
- Hypertension [see Warnings and Precautions (5.6)]
- Bronchoconstriction [see Warnings and Precautions (5.7)]
- Seizure [see Warnings and Precautions (5.8)]
- Cerebrovascular Accident (Stroke) [see Warnings and Precautions (5.9)]
7 Drug Interactions (7 DRUG INTERACTIONS)
No formal pharmacokinetic drug interaction studies have been conducted with regadenoson.
8.6 Renal Impairment
No dose adjustment is needed in patients with renal impairment including patients with end stage renal disease and/or dependent on dialysis [see Pharmacokinetics (12.3)].
12.3 Pharmacokinetics
In healthy subjects, the regadenoson plasma concentration-time profile is multi-exponential in nature and best characterized by 3-compartment model. The maximal plasma concentration of regadenoson is achieved within 1 to 4 minutes after injection of regadenoson and parallels the onset of the pharmacodynamic response. The half-life of this initial phase is approximately 2 to 4 minutes. An intermediate phase follows, with a half-life on average of 30 minutes coinciding with loss of the pharmacodynamic effect. The terminal phase consists of a decline in plasma concentration with a half-life of approximately 2 hours [see Clinical Pharmacology (12.2)]. Within the dose range of 0.3–20 mcg/kg in healthy subjects, clearance, terminal half-life or volume of distribution do not appear dependent upon the dose.
A population pharmacokinetic analysis including data from subjects and patients demonstrated that regadenoson clearance decreases in parallel with a reduction in creatinine clearance and clearance increases with increased body weight. Age, gender, and race have minimal effects on the pharmacokinetics of regadenoson.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Regadenoson Injection is a pharmacologic stress agent indicated for radionuclide myocardial perfusion imaging (MPI) in patients unable to undergo adequate exercise stress.
5.1 Myocardial Ischemia
Fatal and nonfatal myocardial infarction (MI), ventricular arrhythmias, and cardiac arrest have occurred following regadenoson injection. Avoid use in patients with symptoms or signs of acute myocardial ischemia, for example unstable angina or cardiovascular instability; these patients may be at greater risk of serious cardiovascular reactions to regadenoson. Cardiac resuscitation equipment and trained staff should be available before administering regadenoson. Adhere to the recommended duration of injection [see Dosage and Administration (2)]. As noted in an animal study, longer injection times may increase the duration and magnitude of increase in coronary blood flow [see Clinical Pharmacology (12.2)]. If serious reactions to regadenoson occur, consider the use of aminophylline, an adenosine antagonist, to shorten the duration of increased coronary blood flow induced by regadenoson [see Overdosage (10)].
5.7 Bronchoconstriction
Adenosine receptor agonists, including regadenoson, may cause dyspnea, bronchoconstriction, and respiratory compromise. Appropriate bronchodilator therapy and resuscitative measures should be available prior to and following regadenoson administration [see Adverse Reactions (6.1), Clinical Pharmacology (12.2), Overdosage (10) and Patient Counseling Information (17)].
12.1 Mechanism of Action
Regadenoson is a low affinity agonist (Ki ≈ 1.3 μM) for the A2A adenosine receptor, with at least 10-fold lower affinity for the A1 adenosine receptor (Ki > 16.5 μM), and weak, if any, affinity for the A2B and A3 adenosine receptors. Activation of the A2A adenosine receptor by regadenoson produces coronary vasodilation and increases coronary blood flow (CBF).
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Myocardial Ischemia. Fatal cardiac events have occurred. Avoid use in patients with symptoms or signs of acute myocardial ischemia, for example unstable angina or cardiovascular instability, who may be at greater risk. Cardiac resuscitation equipment and trained staff should be available before administration (5.1).
- Sinoatrial (SA) and Atrioventricular (AV) Nodal Block. Adenosine receptor agonists, including regadenoson, can depress the SA and AV nodes and may cause first-, second- or third-degree AV block, or sinus bradycardia (5.2).
- Atrial Fibrillation/Atrial Flutter. New-onset or recurrent atrial fibrillation with rapid ventricular response and atrial flutter have been reported (5.3).
- Hypersensitivity, including anaphylaxis, angioedema, cardiac or respiratory arrest, respiratory distress, decreased oxygen saturation, hypotension, throat tightness, urticaria, and rashes have occurred. Have personnel and resuscitative equipment immediately available (5.4).
- Hypotension. Adenosine receptor agonists, including regadenoson, induce vasodilation and hypotension. The risk of serious hypotension may be higher in patients with autonomic dysfunction, stenotic valvular heart disease, pericarditis or pericardial effusions, stenotic carotid artery disease with cerebrovascular insufficiency, or hypovolemia (5.5).
- Hypertension. Adenosine receptor agonists, including regadenoson, may induce clinically significant increases in blood pressure particularly in patients with a history of hypertension and when the MPI includes low level exercise (5.6).
- Bronchoconstriction. Adenosine receptor agonists, including regadenoson, may induce dyspnea, bronchoconstriction and respiratory compromise in patients with chronic obstructive pulmonary disease (COPD) or asthma. Resuscitative measures should be available (5.7).
- Seizure. Regadenoson may lower the seizure threshold. New onset or recurrence of convulsive seizures has occurred. Some seizures are prolonged and require urgent anticonvulsive management. Methylxanthine use is not recommended in patients who experience a seizure in association with regadenoson (5.8).
- Cerebrovascular Accident (Stroke). Hemorrhagic and ischemic cerebrovascular accidents have occurred (5.9).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
The recommended dose of regadenoson injection is 5 mL (0.4 mg regadenoson) administered as an intravenous injection within 10 seconds.
- Patients should be instructed to avoid consumption of any products containing methylxanthines, including caffeinated coffee, tea or other caffeinated beverages, caffeine-containing drug products, aminophylline and theophylline for at least 12 hours before a scheduled radionuclide MPI [see Drug Interactions (7.1) and Clinical Pharmacology (12.2)].
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Do not administer regadenoson injection if it contains particulate matter or is discolored.
- Administer regadenoson injection as an intravenous injection within 10 seconds into a peripheral vein using a 22 gauge or larger catheter or needle.
- Administer a 5 mL saline flush immediately after the injection of regadenoson injection.
- Administer the radionuclide myocardial perfusion imaging agent 10–20 seconds after the saline flush. The radionuclide may be injected directly into the same catheter as regadenoson injection.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- Single-dose pre-filled syringe: clear, colorless solution containing regadenoson 0.4 mg per 5 mL (0.08 mg per mL).
6.2 Post Marketing Experience (6.2 Post-Marketing Experience)
The following adverse reactions have been reported from worldwide marketing experience with regadenoson. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
During clinical development, 1,651 patients were exposed to regadenoson, with most receiving 0.4 mg as a rapid (≤ 10 seconds) intravenous injection. Most of these patients received regadenoson in two clinical studies that enrolled patients who had no history of bronchospastic lung disease as well as no history of a cardiac conduction block of greater than first-degree AV block, except for patients with functioning artificial pacemakers. In these studies (Studies 1 and 2), 2,015 patients underwent myocardial perfusion imaging after administration of regadenoson (N = 1,337) or Adenoscan® (N = 678). The population was 26–93 years of age (median 66 years), 70% male and primarily Caucasian (76% Caucasian, 7% African American, 9% Hispanic, 5% Asian). Table 1 shows the most frequently reported adverse reactions.
Overall, any adverse reaction occurred at similar rates between the study groups (80% for the regadenoson group and 83% for the Adenoscan group). Aminophylline was used to treat the reactions in 3% of patients in the regadenoson group and 2% of patients in the Adenoscan group. Most adverse reactions began soon after dosing, and generally resolved within approximately 15 minutes, except for headache which resolved in most patients within 30 minutes.
|
Regadenoson
N = 1,337 |
Adenoscan
N = 678 |
|
| Dyspnea | 28% | 26% |
| Headache | 26% | 17% |
| Flushing | 16% | 25% |
| Chest Discomfort | 13% | 18% |
| Angina Pectoris or ST Segment Depression | 12% | 18% |
| Dizziness | 8% | 7% |
| Chest Pain | 7% | 10% |
| Nausea | 6% | 6% |
| Abdominal Discomfort | 5% | 2% |
| Dysgeusia | 5% | 7% |
| Feeling Hot | 5% | 8% |
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Regadenoson Injection, a clear, colorless solution, is supplied as follows:
| NDC | Regadenoson Injection (0.08 mg per mL) | Package Factor |
| 71288-201-85 | 0.4 mg regadenoson per 5 mL Single-Dose Pre-filled Syringe | 1 syringe per carton |
Store at 20° to 25°C (68° to 77°F); excursions permitted 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature.]
Discard unused portion.
Sterile, Nonpyrogenic, Preservative-free.
The container closure is not made with natural rubber latex.
5.9 Cerebrovascular Accident (stroke) (5.9 Cerebrovascular Accident (Stroke))
5.3 Atrial Fibrillation/atrial Flutter (5.3 Atrial Fibrillation/Atrial Flutter)
New-onset or recurrent atrial fibrillation with rapid ventricular response and atrial flutter have been reported following regadenoson injection [see Adverse Reactions (6.2)].
7.2 Effect of Regadenoson On Other Drugs (7.2 Effect of Regadenoson on Other Drugs)
Regadenoson does not inhibit the metabolism of substrates for CYP1A2, CYP2C8, CYP2C9, CYP2C19, CYP2D6, or CYP3A4 in human liver microsomes, indicating that it is unlikely to alter the pharmacokinetics of drugs metabolized by these cytochrome P450 enzymes.
7.1 Effects of Other Drugs On Regadenoson (7.1 Effects of Other Drugs on Regadenoson)
- Methylxanthines (e.g., caffeine, aminophylline and theophylline) are non-specific adenosine receptor antagonists that interfere with the vasodilation activity of regadenoson [see Clinical Pharmacology (12.2) and Patient Counseling Information (17)]. Patients should avoid consumption of any products containing methylxanthines as well as any drugs containing theophylline or aminophylline for at least 12 hours before regadenoson administration. Aminophylline may be used to attenuate severe or persistent adverse reactions to regadenoson [see Overdosage (10)].
- In clinical studies, regadenoson was administered to patients taking other cardioactive drugs (i.e., β-blockers, calcium channel blockers, ACE inhibitors, nitrates, cardiac glycosides, and angiotensin receptor blockers) without reported adverse reactions or apparent effects on efficacy.
- Dipyridamole may change the effects of regadenoson. When possible, withhold dipyridamole for at least two days prior to regadenoson administration.
5.4 Hypersensitivity, Including Anaphylaxis
Anaphylaxis, angioedema, cardiac or respiratory arrest, respiratory distress, decreased oxygen saturation, hypotension, throat tightness, urticaria and rashes have occurred. In clinical trials, hypersensitivity reactions were reported in fewer than 1 percent of patients [see Adverse Reactions (6.1)]. Have personnel and resuscitative equipment immediately available.
5.2 Sinoatrial and Atrioventricular Nodal Block
Adenosine receptor agonists, including regadenoson, can depress the SA and AV nodes and may cause first-, second- or third-degree AV block, or sinus bradycardia requiring intervention. In clinical trials first-degree AV block (PR prolongation > 220 msec) developed in 3% of patients within 2 hours of regadenoson administration; transient second-degree AV block with one dropped beat was observed in one patient receiving regadenoson. In post-marketing experience, third-degree heart block and asystole within minutes of regadenoson administration have occurred [see Adverse Reactions (6.2)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Regadenoson was negative in the Ames bacterial mutation assay, chromosomal aberration assay in Chinese hamster ovary (CHO) cells, and mouse bone marrow micronucleus assay.
Long-term animal studies have not been conducted to evaluate regadenoson's carcinogenic potential or potential effects on fertility.
Principal Display Panel – Regadenoson Injection 5 Ml Carton (PRINCIPAL DISPLAY PANEL – Regadenoson Injection 5 mL Carton)
NDC 71288-201-85
1 x 5 mL Single-Dose Pre-filled Syringe
Discard unused portion
Rx only
Regadenoson Injection
0.4 mg per 5 mL
(0.08 mg per mL)
For Intravenous Use Only
Inject 5 mL intravenously within 10 seconds.
Follow immediately with saline flush and radiopharmaceutical.
Principal Display Panel – Regadenoson Injection 5 Ml Syringe Label (PRINCIPAL DISPLAY PANEL – Regadenoson Injection 5 mL Syringe Label)
NDC 71288-201-85
Rx only
Regadenoson Injection
0.4 mg per 5 mL
(0.08 mg per mL)
For Intravenous Use Only
5 mL Single-Dose Pre-filled Syringe
Discard unused portion
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:36:48.701910 · Updated: 2026-03-14T21:47:42.884935