Varubi
a52896cd-4a98-49b8-82db-bf1985a64d97
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
VARUBI ® is indicated in combination with other antiemetic agents in adults for the prevention of delayed nausea and vomiting associated with initial and repeat courses of emetogenic cancer chemotherapy, including, but not limited to, highly emetogenic chemotherapy.
Dosage and Administration
The recommended dosage of VARUBI in adults in combination with a 5-HT 3 receptor antagonist and dexamethasone for the prevention of nausea and vomiting with emetogenic cancer chemotherapy is shown in Table 1 . There is no drug interaction between rolapitant and dexamethasone, so no dosage adjustment for dexamethasone is required. Administer a dexamethasone dose of 20 mg on Day 1 [see Clinical Pharmacology (12.3) ] . Administer VARUBI prior to the initiation of each chemotherapy cycle, but at no less than 2 week intervals. Administer VARUBI without regards to meals. Table 1: Recommended Dosing Regimen of VARUBI Day 1 Day 2 Day 3 Day 4 Prevention of Nausea and Vomiting Associated with Cisplatin-Based Highly Emetogenic Cancer Chemotherapy VARUBI 180 mg as a single dose orally within 2 hours prior to initiation of chemotherapy None Dexamethasone 20 mg; 30 min prior to initiation of chemotherapy 8 mg twice daily 8 mg twice daily 8 mg twice daily 5-HT 3 receptor antagonist See the prescribing information for the co-administered 5-HT 3 receptor antagonist for appropriate dosing information. None Prevention of Nausea and Vomiting Associated with Moderately Emetogenic Cancer Chemotherapy and Combinations of Anthracycline and Cyclophosphamide VARUBI 180 mg as a single dose orally within 2 hours prior to initiation of chemotherapy None Dexamethasone 20 mg; 30 min prior to initiation of chemotherapy None 5-HT 3 receptor antagonist See the prescribing information for the co-administered 5-HT 3 receptor antagonist for appropriate dosing information. See the prescribing information for the co-administered 5-HT 3 receptor antagonist for appropriate dosing information.
Contraindications
VARUBI is contraindicated in patients taking CYP2D6 substrates with a narrow therapeutic index, such as thioridazine and pimozide. VARUBI can significantly increase the plasma concentrations of thioridazine and pimozide, which may result in QT prolongation and Torsades de Pointes [see Warnings and Precautions (5.1) ] . VARUBI is contraindicated in pediatric patients less than 2 years of age because of irreversible impairment of sexual development and fertility observed in juvenile rats at clinically relevant dosages [see Use in Specific Populations (8.4) ] .
Warnings and Precautions
CYP2D6 Substrates : Rolapitant is a moderate inhibitor of CYP2D6 and significantly increases the plasma concentrations of CYP2D6 substrates for at least 28 days following single dose administration of VARUBI. Before starting VARUBI, consider if patients require: thioridazine or pimozide; if so, use an alternative antiemetic to VARUBI or an alternative to thioridazine or pimozide that is not metabolized by CYP2D6. other CYP2D6 substrates; if so, consult the prescribing information for the CYP2D6 substrate for additional information about interactions with CYP2D6 inhibitors. ( 4 , 5.1 , 7 )
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling: Interaction with CYP2D6 Substrates [see Contraindications (4) , Warnings and Precautions (5.1) ]
Drug Interactions
Rolapitant is a moderate CYP2D6 inhibitor. The inhibition of CYP2D6 persisted on Day 28 with a 2.3-fold increase in dextromethorphan concentrations, the last time point measured. The inhibitory effect of rolapitant on CYP2D6 is expected to persist beyond 28 days for an unknown duration following administration of VARUBI [see Clinical Pharmacology (12.3) ] . Oral rolapitant is an inhibitor of Breast-Cancer-Resistance Protein (BCRP) and p-glycoprotein (P-gp). Rolapitant, given as a single oral dose, is not an inhibitor or inducer of CYP3A4 [see Clinical Pharmacology (12.3) ] . Therefore, no dosage adjustment for dexamethasone (CYP3A4 substrate) is needed when co-administered with VARUBI [see Dosage and Administration (2) ] . Table 4 and Table 5 include drugs with clinically important drug interactions when administered concomitantly with VARUBI and instructions for preventing or managing them. Table 4: Clinically Relevant Interactions Affecting Drugs Co-Administered with VARUBI CYP2D6 Substrates Narrow Therapeutic Index Drugs (Thioridazine and Pimozide) Clinical Impact: Increased plasma concentrations of thioridazine and pimozide are associated with serious and/or life-threatening events of QT prolongation and Torsades de Pointes. Intervention: VARUBI is contraindicated in patients taking CYP2D6 substrates with a narrow therapeutic index such as thioridazine and pimozide. If patients require these drugs, use an alternative antiemetic to VARUBI or an alternative to thioridazine or pimozide that is not metabolized by CYP2D6 [see Contraindications (4) , Warnings and Precautions (5.1) ]. Other CYP2D6 Substrates Clinical Impact: VARUBI can increase plasma concentrations of other CYP2D6 substrates for at least 28 days following administration of VARUBI and may result in adverse reactions. Before starting treatment with VARUBI consult the prescribing information of CYP2D6 substrates to obtain further information about interactions with CYP2D6 inhibitors [see Clinical Pharmacology (12.3) ]. Intervention: Before starting treatment with VARUBI consult the prescribing information of CYP2D6 substrates to obtain further information about interactions with CYP2D6 inhibitors [see Warnings and Precautions (5.1) ]. BCRP Substrates with a Narrow Therapeutic Index (e.g., methotrexate, topotecan, or irinotecan) Clinical Impact: Increased plasma concentrations of BCRP substrates (e.g., methotrexate, topotecan, or irinotecan) may result in potential adverse reactions [see Clinical Pharmacology (12.3) ]. Intervention: Monitor for adverse reactions related to the concomitant drug if use of VARUBI cannot be avoided. Use the lowest effective dose of rosuvastatin (see prescribing information for additional information on recommended dosing). P-gp Substrates with a Narrow Therapeutic Index (e.g. digoxin) Clinical Impact: Increased plasma concentrations of P-gp substrates (e.g., digoxin) may result in potential adverse reactions [see Clinical Pharmacology (12.3) ]. Intervention: Monitor digoxin concentrations with concomitant use of VARUBI and adjust the dosage as needed to maintain therapeutic concentrations. Monitor for adverse reactions if concomitant use of VARUBI with other P-gp substrates with a narrow therapeutic index cannot be avoided. Warfarin Clinical Impact: Although co-administration of intravenous rolapitant (VARBI is not approved for intravenous use), which has a higher C max than oral VARUBI, with warfarin did not substantially increase the systemic exposure to S-warfarin, the active enantiomer, the effects on INR and prothrombin time were not studied [see Clinical Pharmacology (12.3) ] . Intervention: Monitor INR and prothrombin time and adjust the dosage of warfarin, as needed with concomitant use of VARUBI, to maintain the target INR range. Table 5: Clinically Relevant Interactions Affecting Rolapitant When Co-Administered with Other Drugs Strong CYP3A4 Inducers (e.g. rifampin) Clinical Impact: Co-administration of VARUBI with rifampin can significantly reduce the plasma concentrations of rolapitant and decrease the efficacy of VARUBI [see Clinical Pharmacology (12.3) ] . Intervention: Avoid the use of VARUBI in patients who require chronic administration of strong CYP3A4 inducers.
How Supplied
VARUBI is available as film-coated, capsule shaped, blue tablets, debossed with T0101 on one side and 100 on the other side. Each tablet contains 90 mg rolapitant. VARUBI is packaged in an Aclar blister shell with aluminum foil backing and supplied as follows: NDC 70720-101-02 A single dose child-resistant wallet (2 tablets as one set of twinned blisters) Store at 20ºC to 25ºC (68ºF to 77ºF); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Storage and Handling
VARUBI is available as film-coated, capsule shaped, blue tablets, debossed with T0101 on one side and 100 on the other side. Each tablet contains 90 mg rolapitant. VARUBI is packaged in an Aclar blister shell with aluminum foil backing and supplied as follows: NDC 70720-101-02 A single dose child-resistant wallet (2 tablets as one set of twinned blisters) Store at 20ºC to 25ºC (68ºF to 77ºF); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Description
Dosage and Administration ( 2 ) 8/2020 Contraindications ( 4 ) 8/2020
Medication Information
Warnings and Precautions
CYP2D6 Substrates : Rolapitant is a moderate inhibitor of CYP2D6 and significantly increases the plasma concentrations of CYP2D6 substrates for at least 28 days following single dose administration of VARUBI. Before starting VARUBI, consider if patients require: thioridazine or pimozide; if so, use an alternative antiemetic to VARUBI or an alternative to thioridazine or pimozide that is not metabolized by CYP2D6. other CYP2D6 substrates; if so, consult the prescribing information for the CYP2D6 substrate for additional information about interactions with CYP2D6 inhibitors. ( 4 , 5.1 , 7 )
Indications and Usage
VARUBI ® is indicated in combination with other antiemetic agents in adults for the prevention of delayed nausea and vomiting associated with initial and repeat courses of emetogenic cancer chemotherapy, including, but not limited to, highly emetogenic chemotherapy.
Dosage and Administration
The recommended dosage of VARUBI in adults in combination with a 5-HT 3 receptor antagonist and dexamethasone for the prevention of nausea and vomiting with emetogenic cancer chemotherapy is shown in Table 1 . There is no drug interaction between rolapitant and dexamethasone, so no dosage adjustment for dexamethasone is required. Administer a dexamethasone dose of 20 mg on Day 1 [see Clinical Pharmacology (12.3) ] . Administer VARUBI prior to the initiation of each chemotherapy cycle, but at no less than 2 week intervals. Administer VARUBI without regards to meals. Table 1: Recommended Dosing Regimen of VARUBI Day 1 Day 2 Day 3 Day 4 Prevention of Nausea and Vomiting Associated with Cisplatin-Based Highly Emetogenic Cancer Chemotherapy VARUBI 180 mg as a single dose orally within 2 hours prior to initiation of chemotherapy None Dexamethasone 20 mg; 30 min prior to initiation of chemotherapy 8 mg twice daily 8 mg twice daily 8 mg twice daily 5-HT 3 receptor antagonist See the prescribing information for the co-administered 5-HT 3 receptor antagonist for appropriate dosing information. None Prevention of Nausea and Vomiting Associated with Moderately Emetogenic Cancer Chemotherapy and Combinations of Anthracycline and Cyclophosphamide VARUBI 180 mg as a single dose orally within 2 hours prior to initiation of chemotherapy None Dexamethasone 20 mg; 30 min prior to initiation of chemotherapy None 5-HT 3 receptor antagonist See the prescribing information for the co-administered 5-HT 3 receptor antagonist for appropriate dosing information. See the prescribing information for the co-administered 5-HT 3 receptor antagonist for appropriate dosing information.
Contraindications
VARUBI is contraindicated in patients taking CYP2D6 substrates with a narrow therapeutic index, such as thioridazine and pimozide. VARUBI can significantly increase the plasma concentrations of thioridazine and pimozide, which may result in QT prolongation and Torsades de Pointes [see Warnings and Precautions (5.1) ] . VARUBI is contraindicated in pediatric patients less than 2 years of age because of irreversible impairment of sexual development and fertility observed in juvenile rats at clinically relevant dosages [see Use in Specific Populations (8.4) ] .
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling: Interaction with CYP2D6 Substrates [see Contraindications (4) , Warnings and Precautions (5.1) ]
Drug Interactions
Rolapitant is a moderate CYP2D6 inhibitor. The inhibition of CYP2D6 persisted on Day 28 with a 2.3-fold increase in dextromethorphan concentrations, the last time point measured. The inhibitory effect of rolapitant on CYP2D6 is expected to persist beyond 28 days for an unknown duration following administration of VARUBI [see Clinical Pharmacology (12.3) ] . Oral rolapitant is an inhibitor of Breast-Cancer-Resistance Protein (BCRP) and p-glycoprotein (P-gp). Rolapitant, given as a single oral dose, is not an inhibitor or inducer of CYP3A4 [see Clinical Pharmacology (12.3) ] . Therefore, no dosage adjustment for dexamethasone (CYP3A4 substrate) is needed when co-administered with VARUBI [see Dosage and Administration (2) ] . Table 4 and Table 5 include drugs with clinically important drug interactions when administered concomitantly with VARUBI and instructions for preventing or managing them. Table 4: Clinically Relevant Interactions Affecting Drugs Co-Administered with VARUBI CYP2D6 Substrates Narrow Therapeutic Index Drugs (Thioridazine and Pimozide) Clinical Impact: Increased plasma concentrations of thioridazine and pimozide are associated with serious and/or life-threatening events of QT prolongation and Torsades de Pointes. Intervention: VARUBI is contraindicated in patients taking CYP2D6 substrates with a narrow therapeutic index such as thioridazine and pimozide. If patients require these drugs, use an alternative antiemetic to VARUBI or an alternative to thioridazine or pimozide that is not metabolized by CYP2D6 [see Contraindications (4) , Warnings and Precautions (5.1) ]. Other CYP2D6 Substrates Clinical Impact: VARUBI can increase plasma concentrations of other CYP2D6 substrates for at least 28 days following administration of VARUBI and may result in adverse reactions. Before starting treatment with VARUBI consult the prescribing information of CYP2D6 substrates to obtain further information about interactions with CYP2D6 inhibitors [see Clinical Pharmacology (12.3) ]. Intervention: Before starting treatment with VARUBI consult the prescribing information of CYP2D6 substrates to obtain further information about interactions with CYP2D6 inhibitors [see Warnings and Precautions (5.1) ]. BCRP Substrates with a Narrow Therapeutic Index (e.g., methotrexate, topotecan, or irinotecan) Clinical Impact: Increased plasma concentrations of BCRP substrates (e.g., methotrexate, topotecan, or irinotecan) may result in potential adverse reactions [see Clinical Pharmacology (12.3) ]. Intervention: Monitor for adverse reactions related to the concomitant drug if use of VARUBI cannot be avoided. Use the lowest effective dose of rosuvastatin (see prescribing information for additional information on recommended dosing). P-gp Substrates with a Narrow Therapeutic Index (e.g. digoxin) Clinical Impact: Increased plasma concentrations of P-gp substrates (e.g., digoxin) may result in potential adverse reactions [see Clinical Pharmacology (12.3) ]. Intervention: Monitor digoxin concentrations with concomitant use of VARUBI and adjust the dosage as needed to maintain therapeutic concentrations. Monitor for adverse reactions if concomitant use of VARUBI with other P-gp substrates with a narrow therapeutic index cannot be avoided. Warfarin Clinical Impact: Although co-administration of intravenous rolapitant (VARBI is not approved for intravenous use), which has a higher C max than oral VARUBI, with warfarin did not substantially increase the systemic exposure to S-warfarin, the active enantiomer, the effects on INR and prothrombin time were not studied [see Clinical Pharmacology (12.3) ] . Intervention: Monitor INR and prothrombin time and adjust the dosage of warfarin, as needed with concomitant use of VARUBI, to maintain the target INR range. Table 5: Clinically Relevant Interactions Affecting Rolapitant When Co-Administered with Other Drugs Strong CYP3A4 Inducers (e.g. rifampin) Clinical Impact: Co-administration of VARUBI with rifampin can significantly reduce the plasma concentrations of rolapitant and decrease the efficacy of VARUBI [see Clinical Pharmacology (12.3) ] . Intervention: Avoid the use of VARUBI in patients who require chronic administration of strong CYP3A4 inducers.
Storage and Handling
VARUBI is available as film-coated, capsule shaped, blue tablets, debossed with T0101 on one side and 100 on the other side. Each tablet contains 90 mg rolapitant. VARUBI is packaged in an Aclar blister shell with aluminum foil backing and supplied as follows: NDC 70720-101-02 A single dose child-resistant wallet (2 tablets as one set of twinned blisters) Store at 20ºC to 25ºC (68ºF to 77ºF); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
How Supplied
VARUBI is available as film-coated, capsule shaped, blue tablets, debossed with T0101 on one side and 100 on the other side. Each tablet contains 90 mg rolapitant. VARUBI is packaged in an Aclar blister shell with aluminum foil backing and supplied as follows: NDC 70720-101-02 A single dose child-resistant wallet (2 tablets as one set of twinned blisters) Store at 20ºC to 25ºC (68ºF to 77ºF); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Description
Dosage and Administration ( 2 ) 8/2020 Contraindications ( 4 ) 8/2020
Section 42229-5
Narrow Therapeutic Index Drugs (Thioridazine and Pimozide)
VARUBI is contraindicated in patients taking CYP2D6 substrates with a narrow therapeutic index such as thioridazine and pimozide. Increased plasma concentrations of thioridazine and pimozide are associated with serious and/or life-threatening events of QT prolongation and Torsades de Pointes [see Contraindications (4)].
Before starting treatment with VARUBI, consider whether patients require treatment with thioridazine or pimozide. If patients require these drugs, use an alternative antiemetic to VARUBI or an alternative to thioridazine or pimozide that is not metabolized by CYP2D6.
Section 42230-3
|
This Patient Information has been approved by the U.S. Food and Drug Administration. |
Revised: 8/2020 |
| PATIENT INFORMATION VARUBI ® (vuh ROO bee) (rolapitant) tablets, for oral use |
|
What is VARUBI?
|
|
Do not take VARUBI if you:
|
|
Before taking VARUBI, tell your doctor about all of your medical conditions, including if you:
|
|
|
Tell your doctor about all the medicines you take or stop taking, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Keep a list of your medicines to show your doctor and pharmacist when you get a new medicine. VARUBI and other medicines may affect each other and could cause serious side effects. |
|
How should I take VARUBI?
|
|
|
What are the possible side effects of VARUBI?
VARUBI may cause serious side effects, including:
|
|
|
The most common side effects of VARUBI in people who take VARUBI and receive Cisplatin chemotherapy medicine include: low white blood cell count, hiccups, and stomach (abdominal) pain. The most common side effects of VARUBI in people who take VARUBI and receive Anthracycline and Cyclophosphamide chemotherapy medicines include: decreased appetite, low white blood cell count, dizziness, indigestion, urinary tract infection, mouth sores, and low red blood cell count. These are not all the possible side effects of VARUBI. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store VARUBI?
|
|
| Keep VARUBI and all medicines out of the reach of children. | |
|
General information about the safe and effective use of VARUBI.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use VARUBI for a condition for which it was not prescribed. Do not give VARUBI to other people, even if they have the same symptoms that you have. It may harm them. You can ask your doctor or pharmacist for information about VARUBI that is written for health professionals. |
|
|
What are the ingredients in VARUBI?
Active ingredient: rolapitant Inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, lactose monohydrate, magnesium stearate, microcrystalline cellulose, povidone, and pregelatinized starch. The tablets are coated in non-functional blue and clear coats. The tablet coating comprises the following inactive ingredients: FD&C Blue No. 2-Indigo Carmine Lake, polyethylene glycol, polysorbate 80, polyvinyl alcohol, talc, and titanium dioxide. Manufactured for: TerSera Therapeutics LLC, Deerfield, IL 60015 For more information, go to www.varubirx.com or call 1-844-334-4035. |
Section 51945-4
PRINCIPAL DISPLAY PANEL - 90 mg Tablet Blister Wallet
NDC 70720-101-02
Single dose
180 mg total dose (2 x 90 mg tablets)
VARUBI
®
(rolapitant) tablets
90 mg* per tablet
Contains two 90 mg tablets
equal to a single dose of 180 mg
*(equivalent to 100 mg rolapitant hydrochloride)
Rx only
For oral use.
Keep this and all drugs out of the reach of
children. This package is child-resistant.
Store at 20°C - 25°C (68°F - 77°F);
excursions permitted to 15°C - 30°C
(59°F - 86°F) [see USP Controlled
Room Temperature].
TerSera
®
therapeutics
Manufactured for TerSera Therapeutics LLC
Deerfield, IL 60015
See package insert for dosage information.
10 Overdosage
There are no data on overdose with VARUBI.
There is no antidote for VARUBI overdose. Discontinue VARUBI in the event of overdose, and institute general supportive measures and close observation.
11 Description
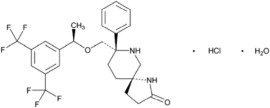
VARUBI contains rolapitant, a substance P/neurokinin 1 (NK1) receptor antagonist. Rolapitant hydrochloride is chemically described as (5S,8S)-8- { [(1R)-1-[3,5-bis(trifluoromethyl)phenyl]ethoxy]methyl]}-8-phenyl-1,7-diazaspiro[4.5]decan-2-one hydrochloride. Its empirical formula is C25H26F6N2O2. HCl.H2O, and its structural formula is:
| rolapitant hydrochloride |
Rolapitant hydrochloride is a white to off-white powder, with a molecular weight of 554.95. Solubility of rolapitant hydrochloride in aqueous solution is pH-dependent and is more soluble at lower pH. Rolapitant has good solubility in common pharmaceutical solvents such as ethanol, propylene glycol and 40% hydroxypropyl beta-cyclodextrin.
Each tablet contains 90 mg rolapitant (equivalent to 100 mg rolapitant hydrochloride) and the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, lactose monohydrate, magnesium stearate, microcrystalline cellulose, povidone, and pregelatinized starch. The tablets are coated in non-functional blue and clear coats. The tablet coating comprises the following inactive ingredients: FD&C Blue No. 2-Indigo Carmine Lake, polyethylene glycol, polysorbate 80, polyvinyl alcohol, talc, and titanium dioxide.
8.4 Pediatric Use
The safety and effectiveness of VARUBI have not been established in pediatric patients. VARUBI is contraindicated in pediatric patients less than 2 years of age [see Contraindications(4)]. Rolapitant administration in juvenile rats (human age equivalent of birth to 2 years) resulted in abnormal ovarian and uterine development, early sexual development in females, delayed sexual development in males, and impaired fertility.
8.5 Geriatric Use
Of the 1294 subjects treated with VARUBI, 25% were 65 years and over, while 5% were 75 and over. No overall differences in safety or efficacy were reported between the elderly subjects and younger subjects, but greater sensitivity of some older individuals cannot be ruled out.
4 Contraindications
VARUBI is contraindicated in patients taking CYP2D6 substrates with a narrow therapeutic index, such as thioridazine and pimozide. VARUBI can significantly increase the plasma concentrations of thioridazine and pimozide, which may result in QT prolongation and Torsades de Pointes [see Warnings and Precautions (5.1)].
VARUBI is contraindicated in pediatric patients less than 2 years of age because of irreversible impairment of sexual development and fertility observed in juvenile rats at clinically relevant dosages [see Use in Specific Populations (8.4)].
6 Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling:
- Interaction with CYP2D6 Substrates [see Contraindications (4), Warnings and Precautions (5.1)]
7 Drug Interactions
Rolapitant is a moderate CYP2D6 inhibitor. The inhibition of CYP2D6 persisted on Day 28 with a 2.3-fold increase in dextromethorphan concentrations, the last time point measured. The inhibitory effect of rolapitant on CYP2D6 is expected to persist beyond 28 days for an unknown duration following administration of VARUBI [see Clinical Pharmacology (12.3)].
Oral rolapitant is an inhibitor of Breast-Cancer-Resistance Protein (BCRP) and p-glycoprotein (P-gp). Rolapitant, given as a single oral dose, is not an inhibitor or inducer of CYP3A4 [see Clinical Pharmacology (12.3)]. Therefore, no dosage adjustment for dexamethasone (CYP3A4 substrate) is needed when co-administered with VARUBI [see Dosage and Administration (2)].
Table 4 and Table 5 include drugs with clinically important drug interactions when administered concomitantly with VARUBI and instructions for preventing or managing them.
| CYP2D6 Substrates | |
| Narrow Therapeutic Index Drugs (Thioridazine and Pimozide) | |
| Clinical Impact: | Increased plasma concentrations of thioridazine and pimozide are associated with serious and/or life-threatening events of QT prolongation and Torsades de Pointes. |
| Intervention: | VARUBI is contraindicated in patients taking CYP2D6 substrates with a narrow therapeutic index such as thioridazine and pimozide. If patients require these drugs, use an alternative antiemetic to VARUBI or an alternative to thioridazine or pimozide that is not metabolized by CYP2D6 [see Contraindications (4), Warnings and Precautions (5.1)]. |
| Other CYP2D6 Substrates | |
| Clinical Impact: | VARUBI can increase plasma concentrations of other CYP2D6 substrates for at least 28 days following administration of VARUBI and may result in adverse reactions. Before starting treatment with VARUBI consult the prescribing information of CYP2D6 substrates to obtain further information about interactions with CYP2D6 inhibitors [see Clinical Pharmacology (12.3)]. |
| Intervention: | Before starting treatment with VARUBI consult the prescribing information of CYP2D6 substrates to obtain further information about interactions with CYP2D6 inhibitors [see Warnings and Precautions (5.1)]. |
| BCRP Substrates with a Narrow Therapeutic Index (e.g., methotrexate, topotecan, or irinotecan) | |
| Clinical Impact: | Increased plasma concentrations of BCRP substrates (e.g., methotrexate, topotecan, or irinotecan) may result in potential adverse reactions [see Clinical Pharmacology (12.3)]. |
| Intervention: | Monitor for adverse reactions related to the concomitant drug if use of VARUBI cannot be avoided. Use the lowest effective dose of rosuvastatin (see prescribing information for additional information on recommended dosing). |
| P-gp Substrates with a Narrow Therapeutic Index (e.g. digoxin) | |
| Clinical Impact: | Increased plasma concentrations of P-gp substrates (e.g., digoxin) may result in potential adverse reactions [see Clinical Pharmacology (12.3)]. |
| Intervention: | Monitor digoxin concentrations with concomitant use of VARUBI and adjust the dosage as needed to maintain therapeutic concentrations. Monitor for adverse reactions if concomitant use of VARUBI with other P-gp substrates with a narrow therapeutic index cannot be avoided. |
| Warfarin | |
| Clinical Impact: | Although co-administration of intravenous rolapitant (VARBI is not approved for intravenous use), which has a higher Cmax than oral VARUBI, with warfarin did not substantially increase the systemic exposure to S-warfarin, the active enantiomer, the effects on INR and prothrombin time were not studied [see Clinical Pharmacology (12.3)]. |
| Intervention: | Monitor INR and prothrombin time and adjust the dosage of warfarin, as needed with concomitant use of VARUBI, to maintain the target INR range. |
| Strong CYP3A4 Inducers (e.g. rifampin) | |
| Clinical Impact: | Co-administration of VARUBI with rifampin can significantly reduce the plasma concentrations of rolapitant and decrease the efficacy of VARUBI [see Clinical Pharmacology (12.3)]. |
| Intervention: | Avoid the use of VARUBI in patients who require chronic administration of strong CYP3A4 inducers. |
8.6 Hepatic Impairment
No dosage adjustment is needed in patients with mild (Child-Pugh Class A) or moderate (Child-Pugh Class B) hepatic impairment. There are no clinical or pharmacokinetic data in patients with severe hepatic impairment (Child-Pugh Class C). Avoid use of VARUBI in patients with severe hepatic impairment. If use cannot be avoided, monitor patients for adverse reactions related to rolapitant [see Adverse Reactions (6.1)].
1 Indications and Usage
VARUBI® is indicated in combination with other antiemetic agents in adults for the prevention of delayed nausea and vomiting associated with initial and repeat courses of emetogenic cancer chemotherapy, including, but not limited to, highly emetogenic chemotherapy.
12.1 Mechanism of Action
Rolapitant is a selective and competitive antagonist of human substance P/NK1 receptors. Rolapitant does not have significant affinity for the NK2 or NK3 receptors or for a battery of other receptors, transporters, enzymes and ion channels. Rolapitant is also active in animal models of chemotherapy-induced emesis.
5 Warnings and Precautions
CYP2D6 Substrates: Rolapitant is a moderate inhibitor of CYP2D6 and significantly increases the plasma concentrations of CYP2D6 substrates for at least 28 days following single dose administration of VARUBI. Before starting VARUBI, consider if patients require:
- thioridazine or pimozide; if so, use an alternative antiemetic to VARUBI or an alternative to thioridazine or pimozide that is not metabolized by CYP2D6.
- other CYP2D6 substrates; if so, consult the prescribing information for the CYP2D6 substrate for additional information about interactions with CYP2D6 inhibitors. (4, 5.1, 7)
2 Dosage and Administration
The recommended dosage of VARUBI in adults in combination with a 5-HT3 receptor antagonist and dexamethasone for the prevention of nausea and vomiting with emetogenic cancer chemotherapy is shown in Table 1. There is no drug interaction between rolapitant and dexamethasone, so no dosage adjustment for dexamethasone is required. Administer a dexamethasone dose of 20 mg on Day 1 [see Clinical Pharmacology (12.3)].
Administer VARUBI prior to the initiation of each chemotherapy cycle, but at no less than 2 week intervals.
Administer VARUBI without regards to meals.
| Day 1 | Day 2 | Day 3 | Day 4 | |
| Prevention of Nausea and Vomiting Associated with Cisplatin-Based Highly Emetogenic Cancer Chemotherapy | ||||
| VARUBI | 180 mg as a single dose orally within 2 hours prior to initiation of chemotherapy | None | ||
| Dexamethasone | 20 mg; 30 min prior to initiation of chemotherapy | 8 mg twice daily | 8 mg twice daily | 8 mg twice daily |
| 5-HT3 receptor antagonist | See the prescribing information for the co-administered 5-HT3 receptor antagonist for appropriate dosing information. | None | ||
| Prevention of Nausea and Vomiting Associated with Moderately Emetogenic Cancer Chemotherapy and Combinations of Anthracycline and Cyclophosphamide | ||||
| VARUBI | 180 mg as a single dose orally within 2 hours prior to initiation of chemotherapy | None | ||
| Dexamethasone | 20 mg; 30 min prior to initiation of chemotherapy | None | ||
| 5-HT3 receptor antagonist | See the prescribing information for the co-administered 5-HT3 receptor antagonist for appropriate dosing information. | See the prescribing information for the co-administered 5-HT3 receptor antagonist for appropriate dosing information. |
3 Dosage Forms and Strengths
Tablets: 90 mg rolapitant; film-coated, blue capsule shaped, debossed with T0101 on one side and 100 on the other side.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
In 4 controlled clinical trials in patients receiving emetogenic cancer chemotherapy, VARUBI was given in combination with a 5-HT3 receptor antagonist and dexamethasone. On Day 1 of Cycle 1 of chemotherapy, 1567 patients were treated with VARUBI and 1198 of these patients continued into the optional multiple cycle extension for up to 6 cycles of chemotherapy. The median number of cycles administered 180 mg of VARUBI was four. VARUBI 180 mg was administered to 1294 patients.
In Cycle 1 adverse reactions were reported in approximately 7% of patients treated with VARUBI compared with approximately 6% of patients treated with control therapy. The most common adverse reactions reported with an incidence of ≥3% and greater than control are listed in Table 2 and Table 3.
|
* all reactions occurring at ≥3% in the VARUBI group and for which the rate for VARUBI exceeds the rate for control |
||
|
VARUBI Regimen
(VARUBI, Dexamethasone, and 5-HT 3 Receptor Antagonist) N = 624 |
Control
(Placebo, Dexamethasone, and 5-HT 3 Receptor Antagonist) N = 627 |
|
| Neutropenia | 9% | 8% |
| Hiccups | 5% | 4% |
| Abdominal Pain | 3% | 2% |
|
*all reactions occurring at ≥3% in the VARUBI group and for which the rate for VARUBI exceeds the rate for control. |
||
|
VARUBI Regimen
(VARUBI, Dexamethasone, and 5-HT 3 Receptor Antagonist) N = 670 |
Control
(Placebo, Dexamethasone, and 5-HT 3 Receptor Antagonist) N = 674 |
|
| Decreased appetite | 9% | 7% |
| Neutropenia | 7% | 6% |
| Dizziness | 6% | 4% |
| Dyspepsia | 4% | 2% |
| Urinary tract infection | 4% | 3% |
| Stomatitis | 4% | 2% |
| Anemia | 3% | 2% |
Adverse reactions in the multiple-cycle extensions of highly and moderately emetogenic chemotherapy studies for up to 6 cycles of chemotherapy were generally similar to that observed in Cycle 1.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling
VARUBI is available as film-coated, capsule shaped, blue tablets, debossed with T0101 on one side and 100 on the other side. Each tablet contains 90 mg rolapitant. VARUBI is packaged in an Aclar blister shell with aluminum foil backing and supplied as follows:
| NDC 70720-101-02 | A single dose child-resistant wallet (2 tablets as one set of twinned blisters) |
Store at 20ºC to 25ºC (68ºF to 77ºF); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
5.1 Interaction With Cyp2d6 Substrates
Rolapitant is a moderate inhibitor of CYP2D6. Exposure to dextromethorphan, a CYP2D6 substrate, following a single dose of rolapitant increased about 3-fold on Days 8 and Day 22. The inhibition of CYP2D6 persisted on Day 28 with a 2.3-fold increase in dextromethorphan (CYP2D6 substrate) concentrations, the last time point measured. The inhibitory effect of rolapitant on CYP2D6 is expected to persist beyond 28 days for an unknown duration following administration of VARUBI [see Drug Interactions (7), Clinical Pharmacology (12.3)].
Principal Display Panel Ndc: 70720 101 02 Blister Backing Label
Structured Label Content
Section 42229-5 (42229-5)
Narrow Therapeutic Index Drugs (Thioridazine and Pimozide)
VARUBI is contraindicated in patients taking CYP2D6 substrates with a narrow therapeutic index such as thioridazine and pimozide. Increased plasma concentrations of thioridazine and pimozide are associated with serious and/or life-threatening events of QT prolongation and Torsades de Pointes [see Contraindications (4)].
Before starting treatment with VARUBI, consider whether patients require treatment with thioridazine or pimozide. If patients require these drugs, use an alternative antiemetic to VARUBI or an alternative to thioridazine or pimozide that is not metabolized by CYP2D6.
Section 42230-3 (42230-3)
|
This Patient Information has been approved by the U.S. Food and Drug Administration. |
Revised: 8/2020 |
| PATIENT INFORMATION VARUBI ® (vuh ROO bee) (rolapitant) tablets, for oral use |
|
What is VARUBI?
|
|
Do not take VARUBI if you:
|
|
Before taking VARUBI, tell your doctor about all of your medical conditions, including if you:
|
|
|
Tell your doctor about all the medicines you take or stop taking, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Keep a list of your medicines to show your doctor and pharmacist when you get a new medicine. VARUBI and other medicines may affect each other and could cause serious side effects. |
|
How should I take VARUBI?
|
|
|
What are the possible side effects of VARUBI?
VARUBI may cause serious side effects, including:
|
|
|
The most common side effects of VARUBI in people who take VARUBI and receive Cisplatin chemotherapy medicine include: low white blood cell count, hiccups, and stomach (abdominal) pain. The most common side effects of VARUBI in people who take VARUBI and receive Anthracycline and Cyclophosphamide chemotherapy medicines include: decreased appetite, low white blood cell count, dizziness, indigestion, urinary tract infection, mouth sores, and low red blood cell count. These are not all the possible side effects of VARUBI. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store VARUBI?
|
|
| Keep VARUBI and all medicines out of the reach of children. | |
|
General information about the safe and effective use of VARUBI.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use VARUBI for a condition for which it was not prescribed. Do not give VARUBI to other people, even if they have the same symptoms that you have. It may harm them. You can ask your doctor or pharmacist for information about VARUBI that is written for health professionals. |
|
|
What are the ingredients in VARUBI?
Active ingredient: rolapitant Inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, lactose monohydrate, magnesium stearate, microcrystalline cellulose, povidone, and pregelatinized starch. The tablets are coated in non-functional blue and clear coats. The tablet coating comprises the following inactive ingredients: FD&C Blue No. 2-Indigo Carmine Lake, polyethylene glycol, polysorbate 80, polyvinyl alcohol, talc, and titanium dioxide. Manufactured for: TerSera Therapeutics LLC, Deerfield, IL 60015 For more information, go to www.varubirx.com or call 1-844-334-4035. |
Section 51945-4 (51945-4)
PRINCIPAL DISPLAY PANEL - 90 mg Tablet Blister Wallet
NDC 70720-101-02
Single dose
180 mg total dose (2 x 90 mg tablets)
VARUBI
®
(rolapitant) tablets
90 mg* per tablet
Contains two 90 mg tablets
equal to a single dose of 180 mg
*(equivalent to 100 mg rolapitant hydrochloride)
Rx only
For oral use.
Keep this and all drugs out of the reach of
children. This package is child-resistant.
Store at 20°C - 25°C (68°F - 77°F);
excursions permitted to 15°C - 30°C
(59°F - 86°F) [see USP Controlled
Room Temperature].
TerSera
®
therapeutics
Manufactured for TerSera Therapeutics LLC
Deerfield, IL 60015
See package insert for dosage information.
10 Overdosage (10 OVERDOSAGE)
There are no data on overdose with VARUBI.
There is no antidote for VARUBI overdose. Discontinue VARUBI in the event of overdose, and institute general supportive measures and close observation.
11 Description (11 DESCRIPTION)
VARUBI contains rolapitant, a substance P/neurokinin 1 (NK1) receptor antagonist. Rolapitant hydrochloride is chemically described as (5S,8S)-8- { [(1R)-1-[3,5-bis(trifluoromethyl)phenyl]ethoxy]methyl]}-8-phenyl-1,7-diazaspiro[4.5]decan-2-one hydrochloride. Its empirical formula is C25H26F6N2O2. HCl.H2O, and its structural formula is:
| rolapitant hydrochloride |
Rolapitant hydrochloride is a white to off-white powder, with a molecular weight of 554.95. Solubility of rolapitant hydrochloride in aqueous solution is pH-dependent and is more soluble at lower pH. Rolapitant has good solubility in common pharmaceutical solvents such as ethanol, propylene glycol and 40% hydroxypropyl beta-cyclodextrin.
Each tablet contains 90 mg rolapitant (equivalent to 100 mg rolapitant hydrochloride) and the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, lactose monohydrate, magnesium stearate, microcrystalline cellulose, povidone, and pregelatinized starch. The tablets are coated in non-functional blue and clear coats. The tablet coating comprises the following inactive ingredients: FD&C Blue No. 2-Indigo Carmine Lake, polyethylene glycol, polysorbate 80, polyvinyl alcohol, talc, and titanium dioxide.
8.4 Pediatric Use
The safety and effectiveness of VARUBI have not been established in pediatric patients. VARUBI is contraindicated in pediatric patients less than 2 years of age [see Contraindications(4)]. Rolapitant administration in juvenile rats (human age equivalent of birth to 2 years) resulted in abnormal ovarian and uterine development, early sexual development in females, delayed sexual development in males, and impaired fertility.
8.5 Geriatric Use
Of the 1294 subjects treated with VARUBI, 25% were 65 years and over, while 5% were 75 and over. No overall differences in safety or efficacy were reported between the elderly subjects and younger subjects, but greater sensitivity of some older individuals cannot be ruled out.
4 Contraindications (4 CONTRAINDICATIONS)
VARUBI is contraindicated in patients taking CYP2D6 substrates with a narrow therapeutic index, such as thioridazine and pimozide. VARUBI can significantly increase the plasma concentrations of thioridazine and pimozide, which may result in QT prolongation and Torsades de Pointes [see Warnings and Precautions (5.1)].
VARUBI is contraindicated in pediatric patients less than 2 years of age because of irreversible impairment of sexual development and fertility observed in juvenile rats at clinically relevant dosages [see Use in Specific Populations (8.4)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in greater detail in other sections of the labeling:
- Interaction with CYP2D6 Substrates [see Contraindications (4), Warnings and Precautions (5.1)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Rolapitant is a moderate CYP2D6 inhibitor. The inhibition of CYP2D6 persisted on Day 28 with a 2.3-fold increase in dextromethorphan concentrations, the last time point measured. The inhibitory effect of rolapitant on CYP2D6 is expected to persist beyond 28 days for an unknown duration following administration of VARUBI [see Clinical Pharmacology (12.3)].
Oral rolapitant is an inhibitor of Breast-Cancer-Resistance Protein (BCRP) and p-glycoprotein (P-gp). Rolapitant, given as a single oral dose, is not an inhibitor or inducer of CYP3A4 [see Clinical Pharmacology (12.3)]. Therefore, no dosage adjustment for dexamethasone (CYP3A4 substrate) is needed when co-administered with VARUBI [see Dosage and Administration (2)].
Table 4 and Table 5 include drugs with clinically important drug interactions when administered concomitantly with VARUBI and instructions for preventing or managing them.
| CYP2D6 Substrates | |
| Narrow Therapeutic Index Drugs (Thioridazine and Pimozide) | |
| Clinical Impact: | Increased plasma concentrations of thioridazine and pimozide are associated with serious and/or life-threatening events of QT prolongation and Torsades de Pointes. |
| Intervention: | VARUBI is contraindicated in patients taking CYP2D6 substrates with a narrow therapeutic index such as thioridazine and pimozide. If patients require these drugs, use an alternative antiemetic to VARUBI or an alternative to thioridazine or pimozide that is not metabolized by CYP2D6 [see Contraindications (4), Warnings and Precautions (5.1)]. |
| Other CYP2D6 Substrates | |
| Clinical Impact: | VARUBI can increase plasma concentrations of other CYP2D6 substrates for at least 28 days following administration of VARUBI and may result in adverse reactions. Before starting treatment with VARUBI consult the prescribing information of CYP2D6 substrates to obtain further information about interactions with CYP2D6 inhibitors [see Clinical Pharmacology (12.3)]. |
| Intervention: | Before starting treatment with VARUBI consult the prescribing information of CYP2D6 substrates to obtain further information about interactions with CYP2D6 inhibitors [see Warnings and Precautions (5.1)]. |
| BCRP Substrates with a Narrow Therapeutic Index (e.g., methotrexate, topotecan, or irinotecan) | |
| Clinical Impact: | Increased plasma concentrations of BCRP substrates (e.g., methotrexate, topotecan, or irinotecan) may result in potential adverse reactions [see Clinical Pharmacology (12.3)]. |
| Intervention: | Monitor for adverse reactions related to the concomitant drug if use of VARUBI cannot be avoided. Use the lowest effective dose of rosuvastatin (see prescribing information for additional information on recommended dosing). |
| P-gp Substrates with a Narrow Therapeutic Index (e.g. digoxin) | |
| Clinical Impact: | Increased plasma concentrations of P-gp substrates (e.g., digoxin) may result in potential adverse reactions [see Clinical Pharmacology (12.3)]. |
| Intervention: | Monitor digoxin concentrations with concomitant use of VARUBI and adjust the dosage as needed to maintain therapeutic concentrations. Monitor for adverse reactions if concomitant use of VARUBI with other P-gp substrates with a narrow therapeutic index cannot be avoided. |
| Warfarin | |
| Clinical Impact: | Although co-administration of intravenous rolapitant (VARBI is not approved for intravenous use), which has a higher Cmax than oral VARUBI, with warfarin did not substantially increase the systemic exposure to S-warfarin, the active enantiomer, the effects on INR and prothrombin time were not studied [see Clinical Pharmacology (12.3)]. |
| Intervention: | Monitor INR and prothrombin time and adjust the dosage of warfarin, as needed with concomitant use of VARUBI, to maintain the target INR range. |
| Strong CYP3A4 Inducers (e.g. rifampin) | |
| Clinical Impact: | Co-administration of VARUBI with rifampin can significantly reduce the plasma concentrations of rolapitant and decrease the efficacy of VARUBI [see Clinical Pharmacology (12.3)]. |
| Intervention: | Avoid the use of VARUBI in patients who require chronic administration of strong CYP3A4 inducers. |
8.6 Hepatic Impairment
No dosage adjustment is needed in patients with mild (Child-Pugh Class A) or moderate (Child-Pugh Class B) hepatic impairment. There are no clinical or pharmacokinetic data in patients with severe hepatic impairment (Child-Pugh Class C). Avoid use of VARUBI in patients with severe hepatic impairment. If use cannot be avoided, monitor patients for adverse reactions related to rolapitant [see Adverse Reactions (6.1)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
VARUBI® is indicated in combination with other antiemetic agents in adults for the prevention of delayed nausea and vomiting associated with initial and repeat courses of emetogenic cancer chemotherapy, including, but not limited to, highly emetogenic chemotherapy.
12.1 Mechanism of Action
Rolapitant is a selective and competitive antagonist of human substance P/NK1 receptors. Rolapitant does not have significant affinity for the NK2 or NK3 receptors or for a battery of other receptors, transporters, enzymes and ion channels. Rolapitant is also active in animal models of chemotherapy-induced emesis.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
CYP2D6 Substrates: Rolapitant is a moderate inhibitor of CYP2D6 and significantly increases the plasma concentrations of CYP2D6 substrates for at least 28 days following single dose administration of VARUBI. Before starting VARUBI, consider if patients require:
- thioridazine or pimozide; if so, use an alternative antiemetic to VARUBI or an alternative to thioridazine or pimozide that is not metabolized by CYP2D6.
- other CYP2D6 substrates; if so, consult the prescribing information for the CYP2D6 substrate for additional information about interactions with CYP2D6 inhibitors. (4, 5.1, 7)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
The recommended dosage of VARUBI in adults in combination with a 5-HT3 receptor antagonist and dexamethasone for the prevention of nausea and vomiting with emetogenic cancer chemotherapy is shown in Table 1. There is no drug interaction between rolapitant and dexamethasone, so no dosage adjustment for dexamethasone is required. Administer a dexamethasone dose of 20 mg on Day 1 [see Clinical Pharmacology (12.3)].
Administer VARUBI prior to the initiation of each chemotherapy cycle, but at no less than 2 week intervals.
Administer VARUBI without regards to meals.
| Day 1 | Day 2 | Day 3 | Day 4 | |
| Prevention of Nausea and Vomiting Associated with Cisplatin-Based Highly Emetogenic Cancer Chemotherapy | ||||
| VARUBI | 180 mg as a single dose orally within 2 hours prior to initiation of chemotherapy | None | ||
| Dexamethasone | 20 mg; 30 min prior to initiation of chemotherapy | 8 mg twice daily | 8 mg twice daily | 8 mg twice daily |
| 5-HT3 receptor antagonist | See the prescribing information for the co-administered 5-HT3 receptor antagonist for appropriate dosing information. | None | ||
| Prevention of Nausea and Vomiting Associated with Moderately Emetogenic Cancer Chemotherapy and Combinations of Anthracycline and Cyclophosphamide | ||||
| VARUBI | 180 mg as a single dose orally within 2 hours prior to initiation of chemotherapy | None | ||
| Dexamethasone | 20 mg; 30 min prior to initiation of chemotherapy | None | ||
| 5-HT3 receptor antagonist | See the prescribing information for the co-administered 5-HT3 receptor antagonist for appropriate dosing information. | See the prescribing information for the co-administered 5-HT3 receptor antagonist for appropriate dosing information. |
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tablets: 90 mg rolapitant; film-coated, blue capsule shaped, debossed with T0101 on one side and 100 on the other side.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
In 4 controlled clinical trials in patients receiving emetogenic cancer chemotherapy, VARUBI was given in combination with a 5-HT3 receptor antagonist and dexamethasone. On Day 1 of Cycle 1 of chemotherapy, 1567 patients were treated with VARUBI and 1198 of these patients continued into the optional multiple cycle extension for up to 6 cycles of chemotherapy. The median number of cycles administered 180 mg of VARUBI was four. VARUBI 180 mg was administered to 1294 patients.
In Cycle 1 adverse reactions were reported in approximately 7% of patients treated with VARUBI compared with approximately 6% of patients treated with control therapy. The most common adverse reactions reported with an incidence of ≥3% and greater than control are listed in Table 2 and Table 3.
|
* all reactions occurring at ≥3% in the VARUBI group and for which the rate for VARUBI exceeds the rate for control |
||
|
VARUBI Regimen
(VARUBI, Dexamethasone, and 5-HT 3 Receptor Antagonist) N = 624 |
Control
(Placebo, Dexamethasone, and 5-HT 3 Receptor Antagonist) N = 627 |
|
| Neutropenia | 9% | 8% |
| Hiccups | 5% | 4% |
| Abdominal Pain | 3% | 2% |
|
*all reactions occurring at ≥3% in the VARUBI group and for which the rate for VARUBI exceeds the rate for control. |
||
|
VARUBI Regimen
(VARUBI, Dexamethasone, and 5-HT 3 Receptor Antagonist) N = 670 |
Control
(Placebo, Dexamethasone, and 5-HT 3 Receptor Antagonist) N = 674 |
|
| Decreased appetite | 9% | 7% |
| Neutropenia | 7% | 6% |
| Dizziness | 6% | 4% |
| Dyspepsia | 4% | 2% |
| Urinary tract infection | 4% | 3% |
| Stomatitis | 4% | 2% |
| Anemia | 3% | 2% |
Adverse reactions in the multiple-cycle extensions of highly and moderately emetogenic chemotherapy studies for up to 6 cycles of chemotherapy were generally similar to that observed in Cycle 1.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
VARUBI is available as film-coated, capsule shaped, blue tablets, debossed with T0101 on one side and 100 on the other side. Each tablet contains 90 mg rolapitant. VARUBI is packaged in an Aclar blister shell with aluminum foil backing and supplied as follows:
| NDC 70720-101-02 | A single dose child-resistant wallet (2 tablets as one set of twinned blisters) |
Store at 20ºC to 25ºC (68ºF to 77ºF); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
5.1 Interaction With Cyp2d6 Substrates (5.1 Interaction with CYP2D6 Substrates)
Rolapitant is a moderate inhibitor of CYP2D6. Exposure to dextromethorphan, a CYP2D6 substrate, following a single dose of rolapitant increased about 3-fold on Days 8 and Day 22. The inhibition of CYP2D6 persisted on Day 28 with a 2.3-fold increase in dextromethorphan (CYP2D6 substrate) concentrations, the last time point measured. The inhibitory effect of rolapitant on CYP2D6 is expected to persist beyond 28 days for an unknown duration following administration of VARUBI [see Drug Interactions (7), Clinical Pharmacology (12.3)].
Principal Display Panel Ndc: 70720 101 02 Blister Backing Label (PRINCIPAL DISPLAY PANEL - NDC: 70720-101-02 - Blister Backing Label)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:35:30.321855 · Updated: 2026-03-14T21:43:34.600459