Moxifloxacin Injection
a4e28b09-714b-46e7-b4e6-0163cad78fc5
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Moxifloxacin Injection is a fluoroquinolone antibacterial drug indicated for treating infections in adults ≥ 18 years of age caused by designated, susceptible bacteria. ( 1 , 12.4 ) Community Acquired Pneumonia ( 1.1 ) Skin and Skin Structure Infections: Uncomplicated ( 1.2 ) and Complicated ( 1.3 ) Complicated Intra-Abdominal Infections ( 1.4 ) Acute Bacterial Sinusitis ( 1.5 ) Acute Bacterial Exacerbation of Chronic Bronchitis ( 1.6 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of Moxifloxacin Injection and other antibacterial drugs, Moxifloxacin Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. ( 1.7 )
Dosage and Administration
Type of Infection Dose Every 24 hours Duration (days) Community Acquired Pneumonia ( 1.1 ) 400 mg 7 to 14 Uncomplicated Skin and Skin Structure Infections (SSSI) ( 1.2 ) 400 mg 7 Complicated SSSI ( 1.3 ) 400 mg 7 to 21 Complicated Intra-Abdominal Infections ( 1.4 ) 400 mg 5 to 14 Acute Bacterial Sinusitis ( 1.5 ) 400 mg 10 Acute Bacterial Exacerbation of Chronic Bronchitis ( 1.6 ) 400 mg 5 No dosage adjustment in patients with renal or hepatic impairment. ( 8.6 , 8.7 ) Moxifloxacin Injection: Slow Intravenous infusion over 60 minutes. Avoid rapid or bolus Intravenous infusion. ( 2.2 ) Do not mix with other medications in intravenous bag or in intravenous line. ( 2.2 )
Contraindications
Moxifloxacin is contraindicated in persons with a history of hypersensitivity to moxifloxacin or any member of the quinolone class of antimicrobial agents.
Warnings and Precautions
Prolongation of the QT interval and isolated cases of torsades de pointes has been reported. Avoid use in patients with known prolongation, hypokalemia, and with drugs that prolong the QT interval. ( 5.6 , 7.4 , 8.5 ). Use caution in patients with proarrhythmic conditions such as clinically significant bradycardia or acute myocardial ischemia. ( 5.6 ) Serious and sometimes fatal hypersensitivity reactions, including anaphylactic reactions, may occur after first or subsequent doses. Discontinue moxifloxacin at the first sign of skin rash, jaundice or any other sign of hypersensitivity. ( 5.7 , 5.8 ) Clostridioides difficile -associated diarrhea: Evaluate if diarrhea occurs. ( 5.10 ) High sodium load: each unit dose contains 52.5 mEq (1,207 mg) of sodium. Avoid in patients with sodium restriction. ( 5.11 )
Adverse Reactions
Other serious and sometimes fatal adverse reactions, some due to hypersensitivity, and some due to uncertain etiology, have been reported rarely in patients receiving therapy with quinolones, including moxifloxacin. These events may be severe and generally occur following the administration of multiple doses. Clinical manifestations may include one or more of the following: Fever, rash, or severe dermatologic reactions (for example, toxic epidermal necrolysis, Stevens-Johnson syndrome) Vasculitis; arthralgia; myalgia; serum sickness Allergic pneumonitis Interstitial nephritis; acute renal insufficiency or failure Hepatitis; jaundice; acute hepatic necrosis or failure Anemia, including hemolytic and aplastic; thrombocytopenia, including thrombotic thrombocytopenic purpura; leukopenia; agranulocytosis; pancytopenia; and/or other hematologic abnormalities Discontinue Moxifloxacin Injection immediately at the first appearance of a skin rash, jaundice, or any other sign of hypersensitivity and supportive measures instituted [see Patient Counseling Information ( 17 ) and Adverse Reactions ( 6.2 )].
Drug Interactions
Interacting Drug Interaction Warfarin Anticoagulant effect of warfarin may be enhanced. Monitor prothrombin time/INR, watch for bleeding. ( 6.2 , 7.1 , 12.3 ) Class IA and Class III antiarrhythmics: Proarrhythmic effect may be enhanced. Avoid concomitant use. ( 5.6 , 7.4 ) Antidiabetic agents Carefully monitor blood glucose. ( 5.13 , 7.2 )
Description
Indications and Usage ( 1 ) 3/2020 Dosage and Administration ( 2 ) 3/2020 Warnings and Precautions ( 5 ) 3/2020
Medication Information
Warnings and Precautions
Prolongation of the QT interval and isolated cases of torsades de pointes has been reported. Avoid use in patients with known prolongation, hypokalemia, and with drugs that prolong the QT interval. ( 5.6 , 7.4 , 8.5 ). Use caution in patients with proarrhythmic conditions such as clinically significant bradycardia or acute myocardial ischemia. ( 5.6 ) Serious and sometimes fatal hypersensitivity reactions, including anaphylactic reactions, may occur after first or subsequent doses. Discontinue moxifloxacin at the first sign of skin rash, jaundice or any other sign of hypersensitivity. ( 5.7 , 5.8 ) Clostridioides difficile -associated diarrhea: Evaluate if diarrhea occurs. ( 5.10 ) High sodium load: each unit dose contains 52.5 mEq (1,207 mg) of sodium. Avoid in patients with sodium restriction. ( 5.11 )
Indications and Usage
Moxifloxacin Injection is a fluoroquinolone antibacterial drug indicated for treating infections in adults ≥ 18 years of age caused by designated, susceptible bacteria. ( 1 , 12.4 ) Community Acquired Pneumonia ( 1.1 ) Skin and Skin Structure Infections: Uncomplicated ( 1.2 ) and Complicated ( 1.3 ) Complicated Intra-Abdominal Infections ( 1.4 ) Acute Bacterial Sinusitis ( 1.5 ) Acute Bacterial Exacerbation of Chronic Bronchitis ( 1.6 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of Moxifloxacin Injection and other antibacterial drugs, Moxifloxacin Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. ( 1.7 )
Dosage and Administration
Type of Infection Dose Every 24 hours Duration (days) Community Acquired Pneumonia ( 1.1 ) 400 mg 7 to 14 Uncomplicated Skin and Skin Structure Infections (SSSI) ( 1.2 ) 400 mg 7 Complicated SSSI ( 1.3 ) 400 mg 7 to 21 Complicated Intra-Abdominal Infections ( 1.4 ) 400 mg 5 to 14 Acute Bacterial Sinusitis ( 1.5 ) 400 mg 10 Acute Bacterial Exacerbation of Chronic Bronchitis ( 1.6 ) 400 mg 5 No dosage adjustment in patients with renal or hepatic impairment. ( 8.6 , 8.7 ) Moxifloxacin Injection: Slow Intravenous infusion over 60 minutes. Avoid rapid or bolus Intravenous infusion. ( 2.2 ) Do not mix with other medications in intravenous bag or in intravenous line. ( 2.2 )
Contraindications
Moxifloxacin is contraindicated in persons with a history of hypersensitivity to moxifloxacin or any member of the quinolone class of antimicrobial agents.
Adverse Reactions
Other serious and sometimes fatal adverse reactions, some due to hypersensitivity, and some due to uncertain etiology, have been reported rarely in patients receiving therapy with quinolones, including moxifloxacin. These events may be severe and generally occur following the administration of multiple doses. Clinical manifestations may include one or more of the following: Fever, rash, or severe dermatologic reactions (for example, toxic epidermal necrolysis, Stevens-Johnson syndrome) Vasculitis; arthralgia; myalgia; serum sickness Allergic pneumonitis Interstitial nephritis; acute renal insufficiency or failure Hepatitis; jaundice; acute hepatic necrosis or failure Anemia, including hemolytic and aplastic; thrombocytopenia, including thrombotic thrombocytopenic purpura; leukopenia; agranulocytosis; pancytopenia; and/or other hematologic abnormalities Discontinue Moxifloxacin Injection immediately at the first appearance of a skin rash, jaundice, or any other sign of hypersensitivity and supportive measures instituted [see Patient Counseling Information ( 17 ) and Adverse Reactions ( 6.2 )].
Drug Interactions
Interacting Drug Interaction Warfarin Anticoagulant effect of warfarin may be enhanced. Monitor prothrombin time/INR, watch for bleeding. ( 6.2 , 7.1 , 12.3 ) Class IA and Class III antiarrhythmics: Proarrhythmic effect may be enhanced. Avoid concomitant use. ( 5.6 , 7.4 ) Antidiabetic agents Carefully monitor blood glucose. ( 5.13 , 7.2 )
Description
Indications and Usage ( 1 ) 3/2020 Dosage and Administration ( 2 ) 3/2020 Warnings and Precautions ( 5 ) 3/2020
Section 42229-5
Moxifloxacin Injection Solution for Infusion
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Moxifloxacin Injection should be administered by intravenous infusion only. It is not intended for intra-arterial, intramuscular, intrathecal, intraperitoneal, or subcutaneous administration.
Moxifloxacin Injection should be administered by intravenous infusion over a period of 60 minutes by direct infusion or through a Y-type intravenous infusion set which may already be in place. Caution: rapid or bolus intravenous infusion must be avoided.
Because only limited data are available on the compatibility of moxifloxacin intravenous injection with other intravenous substances, additives or other medications should not be added to Moxifloxacin Injection or infused simultaneously through the same intravenous line. If the same intravenous line or a Y-type line is used for sequential infusion of other drugs, or if the “piggyback” method of administration is used, the line should be flushed before and after infusion of Moxifloxacin Injection with an infusion solution compatible with moxifloxacin injection as well as with other drug(s) administered via this common line.
Moxifloxacin Injection is compatible with the following intravenous solutions at ratios from 1:10 to 10:1
| 0.9% Sodium Chloride Injection, USP | Sterile Water for Injection, USP |
| 1 molar Sodium Chloride Injection | 10% Dextrose for Injection, USP |
| 5% Dextrose Injection, USP | Lactated Ringer's for Injection |
Section 42231-1
|
This Medication Guide has been approved by the U.S. Food and Drug Administration |
Revised:10/2022 |
|
|
Medication Guide
MOXIFLOXACIN (mox i FLOX a sin) (in jek shun) injection, for intravenous use |
||
| Read the Medication Guide that comes with moxifloxacin injection before you start receiving it and each time you receive it. There may be new information. This Medication Guide does not take the place of talking to your healthcare provider about your medical condition or your treatment. | ||
|
What is the most important information I should know about moxifloxacin injection?
|
||
|
2. Changes in sensation and possible nerve damage (peripheral neuropathy).
Damage to the nerves in arms, hands, legs, or feet can happen in people who take fluoroquinolones, including moxifloxacin. Stop receiving moxifloxacin immediately and talk to your healthcare provider right away if you get any of the following symptoms of peripheral neuropathy in your arms, hands, legs, or feet: |
||
|
|
|
| Moxifloxacin may need to be stopped to prevent permanent nerve damage. 3. Central Nervous System (CNS) effects. Seizures have been reported in people who take fluoroquinolone antibiotic medicines, including moxifloxacin. Tell your healthcare provider if you have a history of seizures before you start taking moxifloxacin. CNS side effects may happen as soon as after taking the first dose of moxifloxacin. Stop taking moxifloxacin immediately and talk to your healthcare provider right away if you get any of these side effects, or other changes in mood or behavior: |
||
|
|
|
|
4. Worsening of myasthenia gravis (a disease which causes muscle weakness). Fluoroquinolones like moxifloxacin injection may cause worsening of myasthenia gravis symptoms, including muscle weakness and breathing problems. Tell your healthcare provider if you have a history of myasthenia gravis. Moxifloxacin should not be used in people who have a history of myasthenia gravis. Call your healthcare provider right away if you have any worsening muscle weakness or breathing problems. See the section “ What are the possible side effects of moxifloxacin injection? ” for more information about side effects. |
||
What is moxifloxacin injection?
|
||
| Call your healthcare provider if you think your condition is not getting better while you are receiving moxifloxacin injection. | ||
|
Who should not receive moxifloxacin injection? Do not receive moxifloxacin injection if you have ever had an allergic reaction to moxifloxacin, other fluoroquinolone antibiotics, or any of the ingredients in moxifloxacin injection. Ask your healthcare provider if you are not sure. See the end of this Medication Guide for a complete list of ingredients in moxifloxacin injection. |
||
What should I tell my healthcare provider before receiving moxifloxacin injection?
|
||
|
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, herbal and dietary supplements. Moxifloxacin injection and other medicines can affect each other causing side effects. Especially tell your healthcare provider if you take:
|
||
|
Ask your healthcare provider if you are not sure if any of your medicines are listed above. Know the medicines you take. Keep a list of your medicines and show it to your healthcare provider and pharmacist when you get a new medicine. |
||
How should I receive moxifloxacin injection?
|
||
|
What should I avoid while receiving moxifloxacin injection?
|
||
|
What are the possible side effects of moxifloxacin injection?
|
||
|
|
|
|
||
| The most common side effects of moxifloxacin injection include: | ||
|
|
|
| These are not all the possible side effects of moxifloxacin injection. Tell your healthcare provider about any side effect that bothers you or that does not go away. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA- 1088. | ||
|
General Information about the safe and effective use of moxifloxacin injection.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. This Medication Guide summarizes the most important information about moxifloxacin injection. If you would like more information about moxifloxacin injection, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about moxifloxacin injection that is written for health professionals |
||
|
What are the ingredients in moxifloxacin injection? Lake Zurich, IL 60047 www.fresenius-kabi.com/us For more information, call 1-800-551-7176. 451327G |
Section 43683-2
Section 51945-4
PACKAGE LABEL - PRINCIPAL DISPLAY - Moxifloxacin 250 mL Bag Label
NDC 63323-850-04
850174
Moxifloxacin Injection
400 mg per 250 mL (1.6 mg per mL)
For Intravenous Infusion Rx Only
USE IMMEDIATELY ONCE REMOVED FROM THE OVERWRAP.
INFUSE OVER 60 MINUTES.
1.7 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Moxifloxacin Injection and other antibacterial drugs, Moxifloxacin Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
7.1 Warfarin
Quinolones, including moxifloxacin, have been reported to enhance the anticoagulant effects of warfarin or its derivatives in the patient population. In addition, infectious disease and its accompanying inflammatory process, age, and general status of the patient are risk factors for increased anticoagulant activity. Therefore, the prothrombin time, International Normalized Ratio (INR), or other suitable anticoagulation tests should be closely monitored if a quinolone is administered concomitantly with warfarin or its derivatives [see Adverse Reactions (6, 6.1,), Clinical Pharmacology (12.3), and Patient Counseling Information (17)].
10 Overdosage
Single oral overdoses up to 2.8 g were not associated with any serious adverse events.
In the event of acute overdose, the stomach should be emptied and adequate hydration maintained. ECG monitoring is recommended due to the possibility of QT interval prolongation. The patient should be carefully observed and given supportive treatment. The administration of activated charcoal as soon as possible after oral overdose may prevent excessive increase of systemic moxifloxacin exposure. About 3% and 9% of the dose of moxifloxacin, as well as about 2% and 4.5% of its glucuronide metabolite are removed by continuous ambulatory peritoneal dialysis and hemodialysis, respectively.
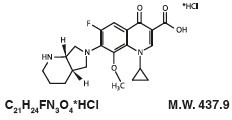
11 Description
Moxifloxacin is a synthetic broad spectrum antibacterial agent for intravenous administration. Moxifloxacin, a fluoroquinolone, is available as a buffered monohydrochloride salt of 1-cyclopropyl-7-[(S,S)-2,8-diazabicyclo[4.3.0]non-8-yl]-6- fluoro-8-methoxy-1,4-dihydro-4-oxo-3 quinoline carboxylic acid. It is a slightly yellow to yellow crystalline substance. Its chemical structure is as follows:
Moxifloxacin Injection is sterile solution for infusion in a ready-to-use, single-dose flexible bag.
8.4 Pediatric Use
Effectiveness in pediatric patients and adolescents less than 18 years of age has not been established. Moxifloxacin causes arthropathy in juvenile animals. Limited information on the safety of Moxifloxacin in 301 pediatric patients is available from the cIAI trial [see Boxed Warning, Warnings and Precautions (5.9) and Nonclinical Toxicology (13.2)].
8.5 Geriatric Use
Geriatric patients are at increased risk for developing severe tendon disorders including tendon rupture when being treated with a fluoroquinolone such as Moxifloxacin Injection. This risk is further increased in patients receiving concomitant corticosteroid therapy. Tendinitis or tendon rupture can involve the Achilles, hand, shoulder, or other tendon sites and can occur during or after completion of therapy; cases occurring up to several months after fluoroquinolone treatment have been reported. Caution should be used when prescribing Moxifloxacin Injection to elderly patients especially those on corticosteroids. Patients should be informed of this potential side effect and advised to discontinue Moxifloxacin Injection and contact their healthcare provider if any symptoms of tendinitis or tendon rupture occur [see Boxed Warning, Warnings and Precautions (5.1, 5.2), and Adverse Reactions (6.2)].
Epidemiologic studies report an increased rate of aortic aneurysm and dissection within two months following use of fluoroquinolones, particularly in elderly patients [see Warnings and Precautions (5.9)].
Moxifloxacin Injection contains 1,207 mg (52.5 mEq) of sodium per unit dose. The geriatric population may respond with a blunted natriuresis to salt loading. This may be clinically important with regard to such diseases as congestive heart failure [see Warnings and Precautions (5.11)].
In controlled multiple-dose clinical trials, 23% of patients receiving oral moxifloxacin were greater than or equal to 65 years of age and 9% were greater than or equal to 75 years of age. The clinical trial data demonstrate that there is no difference in the safety and efficacy of oral moxifloxacin in patients aged 65 or older compared to younger adults.
In trials of intravenous use, 42% of moxifloxacin patients were greater than or equal to 65 years of age, and 23% were greater than or equal to 75 years of age. The clinical trial data demonstrate that the safety of intravenous moxifloxacin in patients aged 65 or older was similar to that of comparator-treated patients. In general, elderly patients may be more susceptible to drug-associated effects of the QT interval. Therefore, Moxifloxacin Injection should be avoided in patients taking drugs that can result in prolongation of the QT interval (for example, Class IA or Class III antiarrhythmics) or in patients with risk factors for torsades de pointes (for example, known QT prolongation, uncorrected hypokalemia) [see Warnings and Precautions (5.6), Drug Interactions (7.4), and Clinical Pharmacology (12.3)].
4 Contraindications
Moxifloxacin is contraindicated in persons with a history of hypersensitivity to moxifloxacin or any member of the quinolone class of antimicrobial agents.
5.6 Qt Prolongation
Moxifloxacin has been shown to prolong the QT interval of the electrocardiogram in some patients. Following oral dosing with 400 mg of moxifloxacin the mean (± SD) change in QTc from the pre-dose value at the time of maximum drug concentration was 6 msec (± 26) (n = 787). Following a course of daily intravenous dosing (400 mg; 1 hour infusion each day) the mean change in QTc from the Day 1 pre-dose value was 10 msec (± 22) on Day 1 (n = 667) and 7 msec (± 24) on Day 3 (n = 667).
The drug should be avoided in patients with known prolongation of the QT interval, patients with uncorrected hypokalemia and patients receiving Class IA (for example, quinidine, procainamide) or Class III (for example, amiodarone, sotalol) antiarrhythmic agents, due to the lack of clinical experience with the drug in these patient populations.
Pharmacokinetic studies between moxifloxacin and other drugs that prolong the QT interval such as cisapride, erythromycin, antipsychotics, and tricyclic antidepressants have not been performed. An additive effect of moxifloxacin and these drugs cannot be excluded; therefore caution should be exercised when moxifloxacin is given concurrently with these drugs. In premarketing clinical trials, the rate of cardiovascular adverse events was similar in 798 moxifloxacin and 702 comparator treated patients who received concomitant therapy with drugs known to prolong the QTc interval.
Moxifloxacin should be used with caution in patients with ongoing proarrhythmic conditions, such as clinically significant bradycardia, acute myocardial ischemia. The magnitude of QT prolongation may increase with increasing concentrations of the drug or increasing rates of infusion of the intravenous formulation. Therefore the recommended dose or infusion rate should not be exceeded. QT prolongation may lead to an increased risk for ventricular arrhythmias including torsades de pointes. No excess in cardiovascular morbidity or mortality attributable to QTc prolongation occurred with moxifloxacin treatment in over 15,500 patients in controlled clinical studies, including 759 patients who were hypokalemic at the start of treatment, and there was no increase in mortality in over 18,000 moxifloxacin tablet treated patients in a postmarketing observational study in which ECGs were not performed. Elderly patients using Moxifloxacin Injection may be more susceptible to drug-associated QT prolongation [see Use in Specific Populations (8.5)]. In addition, moxifloxacin should be used with caution in patients with mild, moderate, or severe liver cirrhosis [see Clinical Pharmacology (12.3) and Patient Counseling Information (17)].
6 Adverse Reactions
The following serious and otherwise important adverse reactions are discussed in greater detail in the Warnings and Precautions section of the label:
- Disabling and Potentially Irreversible Serious Adverse Reactions Including Tendinitis and Tendon Rupture, Peripheral Neuropathy, and Central Nervous System Effects [see Warnings and Precautions (5.1)]
- Tendinitis and Tendon Rupture [see Warnings and Precautions (5.2)]
- Peripheral Neuropathy [see Warnings and Precautions (5.3)]
- Central Nervous System Effects [see Warnings and Precautions (5.4)]
- Exacerbation of Myasthenia Gravis [see Warnings and Precautions (5.5)]
- QT Prolongation [see Warnings and Precautions (5.6)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.7)]
- Other Serious and Sometimes Fatal Adverse Reactions [see Warnings and Precautions (5.8)]
- Clostridioides Difficile-Associated Diarrhea [see Warnings and Precautions (5.10)]
- Blood Glucose Disturbances [see Warnings and Precautions (5.13)]
- Photosensitivity/Phototoxicity [see Warnings and Precautions (5.14)]
- Development of Drug Resistant Bacteria [see Warnings and Precautions (5.15)]
7 Drug Interactions
| Interacting Drug | Interaction |
| Warfarin | Anticoagulant effect of warfarin may be enhanced. Monitor prothrombin time/INR, watch for bleeding. (6.2, 7.1, 12.3) |
| Class IA and Class III antiarrhythmics: | Proarrhythmic effect may be enhanced. Avoid concomitant use. (5.6, 7.4) |
| Antidiabetic agents | Carefully monitor blood glucose. (5.13, 7.2) |
8.6 Renal Impairment
The pharmacokinetic parameters of moxifloxacin are not significantly altered in mild, moderate, severe, or end-stage renal disease. No dosage adjustment is necessary in patients with renal impairment, including those patients requiring hemodialysis (HD) or continuous ambulatory peritoneal dialysis (CAPD) [see Dosage and Administration (2), and Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
The mean (± SD) pharmacokinetic parameters of moxifloxacin following single and multiple dose of 400 mg moxifloxacin given by 1 hour intravenous infusion are summarized in Table 7. The mean (± SD) elimination half-life from plasma is 12 ± 1.3 hours; steady-state is achieved after at least three days with a 400 mg once daily regimen. The absolute bioavailability of moxifloxacin is approximately 90 percent. When switching from intravenous to oral formulation, no dosage adjustment is necessary [see Dosage and Administration (2.1)].
|
a Range of means from different studies |
|||
|
b Expected Cmax (concentration obtained around the time of the end of the infusion) |
|||
|
Cmax
(mg/L) |
AUC
(mg•h/L) |
Half-life (hr) | |
| Single Dose IV | |||
| Healthy young male/female (n = 56) | 3.9 ± 0.9 | 39.3 ± 8.6 | 8.2 to 15.4a |
| Patients (n = 118) | |||
| Male (n = 64) | 4.4 ± 3.7 | ||
| Female (n = 54) | 4.5 ± 2 | ||
| < 65 years (n = 58) | 4.6 ± 4.2 | ||
| ≥ 65 years (n = 60) | 4.3 ± 1.3 | ||
| Multiple Dose IV | |||
| Healthy young male (n = 8) Healthy elderly (n =12; 8 male, 4 female) |
4.2 ± 0.8 6.1 ± 1.3 |
38 ± 4.7 48.2 ± 0.9 |
14.8 ± 2.2 10.1 ± 1.6 |
| Patientsb (n = 107) | |||
| Male (n = 58) | 4.2 ± 2.6 | ||
| Female (n = 49) | 4.6 ± 1.5 | ||
| < 65 years (n = 52) | 4.1 ± 1.4 | ||
| ≥ 65 years (n = 55) | 4.7 ± 2.7 |
5.11 High Sodium Load
8.7 Hepatic Impairment
No dosage adjustment is recommended for mild, moderate, or severe hepatic insufficiency (Child-Pugh Classes A, B, or C). However, due to metabolic disturbances associated with hepatic insufficiency, which may lead to QT prolongation, moxifloxacin should be used with caution in these patients [see Warnings and Precautions (5.6), and Clinical Pharmacology (12.3)].
1 Indications and Usage
Moxifloxacin Injection is a fluoroquinolone antibacterial drug indicated for treating infections in adults ≥ 18 years of age caused by designated, susceptible bacteria. (1, 12.4)
- Community Acquired Pneumonia (1.1)
- Skin and Skin Structure Infections: Uncomplicated (1.2) and Complicated (1.3)
- Complicated Intra-Abdominal Infections (1.4)
- Acute Bacterial Sinusitis (1.5)
- Acute Bacterial Exacerbation of Chronic Bronchitis (1.6)
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Moxifloxacin Injection and other antibacterial drugs, Moxifloxacin Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. (1.7)
7.2 Antidiabetic Agents
Disturbances of blood glucose, including hyperglycemia and hypoglycemia, have been reported in patients treated concomitantly with fluoroquinolones and an antidiabetic agent. Therefore, careful monitoring of blood glucose is recommended when these agents are co-administered. If a hypoglycemic reaction occurs, moxifloxacin should be discontinued and appropriate therapy should be initiated immediately [see Warnings and Precautions (5.13), Adverse Reactions (6.1), and Patient Counseling Information (17)].
12.1 Mechanism of Action
Moxifloxacin is a member of the fluoroquinolone class of antibacterial agents [see Microbiology (12.4)].
5.3 Peripheral Neuropathy
Fluoroquinolones, including moxifloxacin, have been associated with an increased risk of peripheral neuropathy. Cases of sensory or sensorimotor axonal polyneuropathy affecting small and/or large axons resulting in paresthesias, hypoesthesias, dysesthesias and weakness have been reported in patients receiving fluoroquinolones including moxifloxacin. Symptoms may occur soon after initiation of moxifloxacin and may be irreversible in some patients [see Warnings and Precautions (5.1) and Adverse Reactions (6, 6.1, 6.2)].
Discontinue moxifloxacin immediately if the patient experiences symptoms of peripheral neuropathy including pain, burning, tingling, numbness, and/or weakness or other alterations of sensation including light touch, pain, temperature, position sense, and vibratory sensation. Avoid fluoroquinolones, including moxifloxacin, in patients who have previously experienced peripheral neuropathy [see Adverse Reactions (6.1, 6.2) and Patient Counseling Information (17)].
7.4 Drugs That Prolong Qt
There is limited information available on the potential for a pharmacodynamic interaction in humans between moxifloxacin and other drugs that prolong the QTc interval of the electrocardiogram. Sotalol, a Class III antiarrhythmic, has been shown to further increase the QTc interval when combined with high doses of intravenous (IV) moxifloxacin in dogs. Therefore, moxifloxacin should be avoided with Class IA and Class III antiarrhythmics [see Warnings and Precautions (5.6), Nonclinical Toxicology (13.2), and Patient Counseling Information (17)].
5 Warnings and Precautions
- Prolongation of the QT interval and isolated cases of torsades de pointes has been reported. Avoid use in patients with known prolongation, hypokalemia, and with drugs that prolong the QT interval. (5.6, 7.4, 8.5). Use caution in patients with proarrhythmic conditions such as clinically significant bradycardia or acute myocardial ischemia. (5.6)
- Serious and sometimes fatal hypersensitivity reactions, including anaphylactic reactions, may occur after first or subsequent doses. Discontinue moxifloxacin at the first sign of skin rash, jaundice or any other sign of hypersensitivity. (5.7, 5.8)
- Clostridioides difficile-associated diarrhea: Evaluate if diarrhea occurs. (5.10)
- High sodium load: each unit dose contains 52.5 mEq (1,207 mg) of sodium. Avoid in patients with sodium restriction. (5.11)
2 Dosage and Administration
|
Type of Infection |
Dose Every 24 hours | Duration (days) |
| Community Acquired Pneumonia (1.1) | 400 mg | 7 to 14 |
| Uncomplicated Skin and Skin Structure Infections (SSSI) (1.2) |
400 mg |
7 |
| Complicated SSSI (1.3) | 400 mg | 7 to 21 |
| Complicated Intra-Abdominal Infections (1.4) | 400 mg | 5 to 14 |
| Acute Bacterial Sinusitis (1.5) | 400 mg | 10 |
| Acute Bacterial Exacerbation of Chronic Bronchitis (1.6) |
400 mg |
5 |
2.1 Dosage in Adult Patients
The dose of Moxifloxacin Injection is 400 mg intravenously once every 24 hours. The duration of therapy depends on the type of infection as described in Table 1.
|
a Due to the designated pathogens [see Indications and Usage (1), for IV use, see Use in Specific Populations (8.5)]. |
||
|
b Sequential therapy (intravenous to oral) may be instituted at the discretion of the physician. |
||
|
Type of Infection a |
Dose Every 24 hours | Duration b (days) |
| Community Acquired Pneumonia (1.1) | 400 mg | 7 to 14 |
| Uncomplicated Skin and Skin Structure Infections (SSSI) (1.2) | 400 mg | 7 |
| Complicated SSSI (1.3) | 400 mg | 7 to 21 |
| Complicated Intra-Abdominal Infections (1.4) | 400 mg | 5 to 14 |
| Acute Bacterial Sinusitis (1.5) | 400 mg | 10 |
| Acute Bacterial Exacerbation of Chronic Bronchitis (1.6) | 400 mg | 5 |
When switching from intravenous to oral formulation, no dosage adjustment is necessary [see Clinical Pharmacology (12.4)]. Patients whose therapy is started with Moxifloxacin Injection may be switched to moxifloxacin tablets when clinically indicated at the discretion of the physician.
3 Dosage Forms and Strengths
Injection: 400 mg moxifloxacin in 250 mL single-dose flexible bag. (3.1)
6.2 Postmarketing Experience
Table 4 lists adverse reactions that have been identified during post-approval use of moxifloxacin. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
| System/Organ Class | Adverse Reaction |
| Blood and Lymphatic System Disorders | Agranulocytosis Pancytopenia [see Warnings and Precautions (5.8)] |
| Cardiac Disorders | Ventricular tachyarrhythmias (including in very rare cases cardiac arrest and torsades de pointes, and usually in patients with concurrent severe underlying proarrhythmic conditions) |
| Ear and Labyrinth Disorders | Hearing impairment, including deafness (reversible in majority of cases) |
| Eye Disorders | Vision loss (especially in the course of CNS reactions, transient in majority of cases) |
| Hepatobiliary Disorders | Hepatitis (predominantly cholestatic) Hepatic failure (including fatal cases) Jaundice Acute hepatic necrosis [see Warnings and Precautions (5.8)] |
| Immune System Disorders | Anaphylactic reaction Anaphylactic shock Angioedema (including laryngeal edema) [see Warnings and Precautions (5.7, 5.8)] |
| Musculoskeletal and Connective Tissue Disorders | Tendon rupture [see Warnings and Precautions (5.2)] |
| Nervous System Disorders | Altered coordination Abnormal gait [see Warnings and Precautions (5.3)] Myasthenia gravis (exacerbation of) [see Warnings and Precautions (5.5)] Muscle weakness Peripheral neuropathy (that may be irreversible), polyneuropathy [see Warnings and Precautions (5.3)] |
| Psychiatric Disorders | Psychotic reaction (very rarely culminating in self- injurious behavior, such as suicidal ideation/thoughts or suicide attempts [see Warnings and Precautions (5.4)] |
| Renal and Urinary Disorders | Renal dysfunction Interstitial nephritis [see Warnings and Precautions (5.8)] |
| Respiratory, Thoracic and Mediastinal Disorders | Allergic pneumonitis [see Warnings and Precautions (5.8)] |
| Skin and Subcutaneous Tissue Disorders | Photosensitivity/phototoxicity reaction [see Warnings and Precautions (5.14)] Stevens-Johnson syndrome Toxic epidermal necrolysis [see Warnings and Precautions (5.8)] |
1.5 Acute Bacterial Sinusitis
Moxifloxacin Injection is indicated in adults (18 years of age or older) for the treatment of Acute Bacterial Sinusitis (ABS) caused by susceptible isolates of Streptococcus pneumoniae, Haemophilus influenzae, or Moraxella catarrhalis [see Clinical Studies (14.4)].
Because fluoroquinolones, including Moxifloxacin Injection, have been associated with serious adverse reactions [see Warnings and Precautions (5.1 to 5.14)] and for some patients ABS is self-limiting, reserve Moxifloxacin Injection for treatment of ABS in patients who have no alternative treatment options.
5.10 Clostridioides Difficile
Clostridioides difficile-associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including moxifloxacin, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibiotic use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated [see Adverse Reactions (6.1) and Patient Counseling Information (17)].
8 Use in Specific Populations
14.4 Acute Bacterial Sinusitis
In a controlled double-blind study conducted in the US, moxifloxacin tablets (400 mg once daily for ten days) were compared with cefuroxime axetil (250 mg twice daily for ten days) for the treatment of acute bacterial sinusitis. The trial included 457 patients valid for the efficacy analysis. Clinical success (cure plus improvement) at the 7 to 21 day post-therapy test of cure visit was 90% for moxifloxacin and 89% for cefuroxime.
An additional non-comparative study was conducted to gather bacteriological data and to evaluate microbiological eradication in adult patients treated with moxifloxacin 400 mg once daily for seven days. All patients (n = 336) underwent antral puncture in this study. Clinical success rates and eradication/presumed eradication rates at the 21 to 37 day follow-up visit were 97% (29 out of 30) for Streptococcus pneumoniae, 83% (15 out of 18) for Moraxella catarrhalis, and 80% (24 out of 30) for Haemophilus influenzae.
5.7 Hypersensitivity Reactions
Serious anaphylactic reactions, some following the first dose, have been reported in patients receiving fluoroquinolone therapy, including moxifloxacin. Some reactions were accompanied by cardiovascular collapse, loss of consciousness, tingling, pharyngeal or facial edema, dyspnea, urticaria, and itching. Discontinue Moxifloxacin Injection at the first appearance of a skin rash or any other sign of hypersensitivity [see Warnings and Precautions (5.7), Adverse Reactions (6) and Patient Counseling Information (17)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below reflect exposure to moxifloxacin in 14,981 patients in 71 active controlled Phase II - IV clinical trials in different indications [see Indications and Usage (1)]. The population studied had a mean age of 50 years (approximately 73% of the population was < 65 years of age), 50% were male, 63% were Caucasian, 12% were Asian and 9% were Black. Patients received moxifloxacin 400 mg once daily PO, IV, or sequentially (IV followed by PO). Treatment duration was usually 6 to 10 days, and the mean number of days on therapy was 9 days.
Discontinuation of moxifloxacin due to adverse events occurred in 5% of patients overall, 4.1% of patients treated with 400 mg PO, 3.9% with 400 mg IV and 8.2% with sequential therapy 400 mg PO/IV. The most common adverse events leading to discontinuation with the 400 mg PO doses were nausea (0.8%), diarrhea (0.5%), dizziness (0.5%), and vomiting (0.4%). The most common adverse event leading to discontinuation with the 400 mg IV dose was rash (0.5%). The most common adverse events leading to discontinuation with the 400 mg IV/PO sequential dose were diarrhea (0.5%) and pyrexia (0.4%).
Adverse reactions occurring in ≥ 1% of moxifloxacin-treated patients and less common adverse reactions, occurring in 0.1 to < 1% of moxifloxacin-treated patients, are shown in Table 2 and Table 3, respectively. The most common adverse drug reactions (≥ 3%) are nausea, diarrhea, headache, and dizziness.
|
a MedDRA Version 12.0 |
||
|
System Organ Class |
Adverse Reactions a |
% (N=14,981) |
| Blood and Lymphatic System Disorders | Anemia | 1.1 |
| Gastrointestinal Disorders | Nausea | 6.9 |
| Diarrhea | 6 | |
| Vomiting | 2.4 | |
| Constipation | 1.9 | |
| Abdominal pain | 1.5 | |
| Abdominal pain upper | 1.1 | |
| Dyspepsia | 1 | |
| General Disorders and Administration Site Conditions | Pyrexia | 1.1 |
| Investigations | Alanine aminotransferase increased | 1.1 |
| Metabolism and Nutritional Disorder | Hypokalemia | 1 |
| Nervous System Disorders | Headache | 4.2 |
| Dizziness | 3 | |
| Psychiatric Disorders | Insomnia | 1.9 |
|
a MedDRA Version 12.0 |
|
| System Organ Class | Adverse Reactions a |
| Blood and Lymphatic System Disorders | Thrombocythemia Eosinophilia Neutropenia Thrombocytopenia Leukopenia Leukocytosis |
| Cardiac Disorders | Atrial fibrillation Palpitations Tachycardia Cardiac failure congestive Angina pectoris Cardiac failure Cardiac arrest Bradycardia |
| Ear and Labyrinth Disorders | Vertigo Tinnitus |
| Eye Disorders | Vision blurred |
| Gastrointestinal Disorders | Dry mouth Abdominal discomfort Flatulence Abdominal distention Gastritis Gastroesophageal reflux disease |
| General Disorders and Administration Site Conditions | Fatigue Chest pain Asthenia Edema peripheral Pain Malaise |
| System Organ Class | Adverse Reactions a |
| Infusion site extravasation Edema Chills Chest discomfort Facial pain |
|
| Hepatobiliary Disorders | Hepatic function abnormal |
| Infections and Infestations | Vulvovaginal candidiasis Oral candidiasis Vulvovaginal mycotic infection Candidiasis Vaginal infection Oral fungal infection Fungal infection Gastroenteritis |
| Investigations | Aspartate aminotransferase increased Gamma-glutamyltransferase increased Blood alkaline phosphatase increased Hepatic enzyme increased Electrocardiogram QT prolonged Blood lactate dehydrogenase increased Platelet count increased Blood amylase increased Blood glucose increased Lipase increased Hemoglobin decreased Blood creatinine increased Transaminases increased White blood cell count increased Blood urea increased Liver function test abnormal Hematocrit decreased Prothrombin time prolonged Eosinophil count increased Activated partial thromboplastin time prolonged Blood bilirubin increased Blood triglycerides increased Blood uric acid increased Blood pressure increased |
| Metabolism and Nutrition Disorders | Hyperglycemia Anorexia Hypoglycemia Hyperlipidemia Decreased appetite Dehydration |
| Musculoskeletal and Connective Tissue Disorders | Back pain Pain in extremity Arthralgia Myalgia Muscle spasms Musculoskeletal chest pain Musculoskeletal pain |
| Nervous System Disorders | Dysgeusia Somnolence Tremor Lethargy |
| System Organ Class | Adverse Reactions a |
| Paresthesia Tension headache Hypoesthesia Syncope |
|
| Psychiatric Disorders | Anxiety Confusional state Agitation Depression Nervousness Restlessness Hallucination Disorientation |
| Renal and Urinary Disorders | Renal failure Dysuria Renal failure acute |
| Reproductive System and Breast Disorders | Vulvovaginal pruritus |
| Respiratory, Thoracic, and Mediastinal Disorders | Dyspnea Asthma Wheezing Bronchospasm |
| Skin and Subcutaneous Tissue Disorders | Rash Pruritus Hyperhidrosis Erythema Urticaria Dermatitis allergic Night sweats |
| Vascular Disorders | Hypertension Hypotension Phlebitis |
5.13 Blood Glucose Disturbances
As with all fluoroquinolones, disturbances in blood glucose, including both hypoglycemia and hyperglycemia have been reported with moxifloxacin. In moxifloxacin-treated patients, dysglycemia occurred predominantly in elderly diabetic patients receiving concomitant treatment with an oral hypoglycemic agent (for example, sulfonylurea) or with insulin. Severe cases of hypoglycemia resulting in coma or death have been reported. In diabetic patients, careful monitoring of blood glucose is recommended [see Adverse Reactions (6.1)]. If a hypoglycemic reaction occurs, discontinue moxifloxacin and initiate appropriate therapy immediately [see Adverse Reactions (6.1), Drug Interactions (7.2) and Patient Counseling Information (17)].
1.1 Community Acquired Pneumonia
Moxifloxacin Injection is indicated in adults (18 years of age or older) for the treatment of Community Acquired Pneumonia caused by susceptible isolates of Streptococcus pneumoniae (including multi-drug resistant isolates*), Haemophilus influenzae, Moraxella catarrhalis, methicillin-susceptible Staphylococcus aureus, Klebsiella pneumoniae, Mycoplasma pneumoniae, or Chlamydophila pneumoniae.
* MDRSP, Multi-drug resistant Streptococcus pneumoniae includes isolates previously known as PRSP (Penicillin-resistant S. pneumoniae), and are isolates resistant to two or more of the following antibiotics: penicillin (minimum inhibitory concentrations [MIC] ≥ 2 mcg/mL), 2nd generation cephalosporins (for example, cefuroxime), macrolides, tetracyclines, and trimethoprim/sulfamethoxazole [see Clinical Studies (14.2)].
14.2 Community Acquired Pneumonia
A randomized, double-blind, controlled clinical trial was conducted in the US to compare the efficacy of moxifloxacin tablets (400 mg once daily) to that of high-dose clarithromycin (500 mg twice daily) in the treatment of patients with clinically and radiologically documented community acquired pneumonia. This study enrolled 474 patients (382 of whom were valid for the efficacy analysis conducted at the 14 to 35 day follow-up visit). Clinical success for clinically evaluable patients was 95% (184/194) for moxifloxacin and 95% (178/188) for high dose clarithromycin.
A randomized, double-blind, controlled trial was conducted in the US and Canada to compare the efficacy of sequential IV/PO moxifloxacin 400 mg QD for 7 to 14 days to an IV/PO fluoroquinolone control (trovafloxacin or levofloxacin) in the treatment of patients with clinically and radiologically documented community acquired pneumonia. This study enrolled 516 patients, 362 of whom were valid for the efficacy analysis conducted at the 7 to 30 day post-therapy visit. The clinical success rate was 86% (157/182) for moxifloxacin therapy and 89% (161/180) for the fluoroquinolone comparators.
An open-label ex-US study that enrolled 628 patients compared moxifloxacin to sequential IV/PO amoxicillin/clavulanate (1.2 g IV q8h/625 mg PO q8h) with or without high-dose IV/PO clarithromycin (500 mg BID). The intravenous formulations of the comparators are not FDA approved. The clinical success rate at Day 5 to 7 for moxifloxacin therapy was 93% (241/258) and demonstrated superiority to amoxicillin/clavulanate ± clarithromycin (85%, 239/280) [95% C.I. of difference in success rates between moxifloxacin and comparator (2.9%, 13.2%)]. The clinical success rate at the 21 to 28 days post-therapy visit for moxifloxacin was 84% (216/258), which also demonstrated superiority to the comparators (74%, 208/280) [95% C.I. of difference in success rates between moxifloxacin and comparator (2.6%, 16.3%)].
The clinical success rates by pathogen across four CAP studies are presented in Table 10.
| Pathogen | Moxifloxacin | |
| Streptococcus pneumoniae | 80/85 | (94%) |
| Staphylococcus aureus | 17/20 | (85%) |
| Klebsiella pneumoniae | 11/12 | (92%) |
| Haemophilus influenzae | 56/61 | (92%) |
| Chlamydophila pneumoniae | 119/128 | (93%) |
| Mycoplasma pneumoniae | 73/76 | (96%) |
| Moraxella catarrhalis | 11/12 | (92%) |
17 Patient Counseling Information
Advise patients to read the FDA-approved patient labeling (Medication Guide).
5.2 Tendinitis and Tendon Rupture
Fluoroquinolones, including moxifloxacin, have been associated with an increased risk of tendinitis and tendon rupture in all ages [see Warnings and Precautions (5.1) and Adverse Reactions (6.1)]. This adverse reaction most frequently involves the Achilles tendon, and has also been reported with the rotator cuff (the shoulder), the hand, the biceps, the thumb, and other tendons. Tendinitis or tendon rupture can occur within hours or days of starting moxifloxacin or as long as several months after completion of therapy. Tendinitis and tendon rupture can occur bilaterally.
The risk of developing fluoroquinolone-associated tendinitis and tendon rupture is increased in patients over 60 years of age, in patients taking corticosteroid drugs, and in patients with kidney, heart or lung transplants. Other factors that may independently increase the risk of tendon rupture include strenuous physical activity, renal failure, and previous tendon disorders such as rheumatoid arthritis. Tendinitis and tendon rupture have also occurred in patients taking fluoroquinolones who do not have the above risk factors. Discontinue moxifloxacin if the patient experiences pain, swelling, inflammation or rupture of a tendon. Patients should be advised to rest at the first sign of tendinitis or tendon rupture, and to contact their healthcare provider regarding changing to a non- quinolone antimicrobial drug [see Adverse Reactions (6.2) and Patient Counseling Information (17)]. Avoid fluoroquinolones, including moxifloxacin, in patients who have a history of tendon disorders or have experienced tendinitis or tendon rupture [see Adverse Reactions (6.1)].
5.14 Photosensitivity/phototoxicity
Moderate to severe photosensitivity/phototoxicity reactions, the latter of which may manifest as exaggerated sunburn reactions (for example, burning, erythema, exudation, vesicles, blistering, edema) involving areas exposed to light (typically the face, “V” area of the neck, extensor surfaces of the forearms, dorsa of the hands), can be associated with the use of quinolone antibiotics after sun or UV light exposure. Therefore, excessive exposure to these sources of light should be avoided. Drug therapy should be discontinued if phototoxicity occurs [see Adverse Reactions (6.2) and Clinical Pharmacology (12.3)].
5.12 Arthropathic Effects in Animals
The oral administration of moxifloxacin caused lameness in immature dogs. Histopathological examination of the weight-bearing joints of these dogs revealed permanent lesions of the cartilage. Related quinolone-class drugs also produce erosions of cartilage of weight-bearing joints and other signs of arthropathy in immature animals of various species [see Nonclinical Toxicology (13.2)].
5.5 Exacerbation of Myasthenia Gravis
Fluoroquinolones, including moxifloxacin, have neuromuscular blocking activity and may exacerbate muscle weakness in patients with myasthenia gravis. Postmarketing serious adverse reactions, including deaths and requirement for ventilatory support, have been associated with fluoroquinolone use in patients with myasthenia gravis. Avoid moxifloxacin in patients with known history of myasthenia gravis [see Patient Counseling Information (17)].
1.4 Complicated Intra Abdominal Infections
Moxifloxacin Injection is indicated in adults (18 years of age or older) for the treatment of Complicated Intra-Abdominal Infections including polymicrobial infections such as abscess caused by susceptible isolates of Escherichia coli, Bacteroides fragilis, Streptococcus anginosus, Streptococcus constellatus, Enterococcus faecalis, Proteus mirabilis, Clostridium perfringens, Bacteroides thetaiotaomicron, or Peptostreptococcus species [see Clinical Studies (14.7)].
13.2 Animal Toxicology And/or Pharmacology
Quinolones have been shown to cause arthropathy in immature animals. In studies in juvenile dogs oral doses of moxifloxacin ≥ 30 mg/kg/day (approximately 1.5 times the maximum recommended human dose based upon systemic exposure) for 28 days resulted in arthropathy. There was no evidence of arthropathy in mature monkeys and rats at oral doses up to 135 and 500 mg/kg/day, respectively.
Moxifloxacin at an oral dose of 300 mg/kg did not show an increase in acute toxicity or potential for CNS toxicity (for example, seizures) in mice when used in combination with NSAIDs such as diclofenac, ibuprofen, or fenbufen. Some quinolones have been reported to have proconvulsant activity that is exacerbated with concomitant use of non- steroidal anti-inflammatory drugs (NSAIDs).
A QT-prolonging effect of moxifloxacin was found in dog studies, at plasma concentrations about five times the human therapeutic level. The combined infusion of sotalol, a Class III antiarrhythmic agent, with moxifloxacin induced a higher degree of QTc prolongation in dogs than that induced by the same dose (30 mg/kg) of moxifloxacin alone. Electrophysiological in vitro studies suggested an inhibition of the rapid activating component of the delayed rectifier potassium current (IKr) as an underlying mechanism.
No signs of local intolerability were observed in dogs when moxifloxacin was administered intravenously. After intra-arterial injection, inflammatory changes involving the peri-arterial soft tissue were observed suggesting that intra-arterial administration of moxifloxacin should be avoided.
5.9 Risk of Aortic Aneurysm and Dissection
Epidemiologic studies report an increased rate of aortic aneurysm and dissection within two months following use of fluoroquinolones, particularly in elderly patients. The cause for the increased risk has not been identified. In patients with a known aortic aneurysm or patients who are at greater risk for aortic aneurysms, reserve Moxifloxacin Injection for use only when there are no alternative antibacterial treatments available.
14.7 Complicated Intra Abdominal Infections
Two randomized, active controlled trials of cIAI were performed. A double-blind trial was conducted primarily in North America to compare the efficacy of sequential IV/PO moxifloxacin 400 mg QD for 5 to 14 days to IV/piperacillin/tazobactam followed by PO amoxicillin/clavulanic acid in the treatment of patients with cIAI, including peritonitis, abscesses, appendicitis with perforation, and bowel perforation. This study enrolled 681 patients, 379 of which were considered clinically evaluable. A second open-label international study compared moxifloxacin 400 mg QD for 5 to 14 days to IV ceftriaxone plus IV metronidazole followed by PO amoxicillin/clavulanic acid in the treatment of patients with cIAI. This study enrolled 595 patients, 511 of which were considered clinically evaluable. The clinically evaluable population consisted of subjects with a surgically confirmed complicated infection, at least 5 days of treatment and a 25 to 50 day follow-up assessment for patients at the Test of Cure visit. The overall clinical success rates in the clinically evaluable patients are shown in Table 15.
|
a of difference in success rates between moxifloxacin and comparator (moxifloxacin – comparator) |
|||
|
b Excludes 2 patients who required additional surgery within the first 48 hours. |
|||
|
c NA – not applicable |
|||
|
Study |
Moxifloxacin
n/N (%) |
Comparator
n/N (%) |
95%
Confidence Interval a |
| North America (overall) | 146/183 (79.8%) | 153/196 (78.1%) | (-7.4%, 9.3%) |
| Abscess | 40/57 (70.2%) | 49/63 (77.8%)b | NAc |
| Non-abscess | 106/126 (84.1%) | 104/133 (78.2%) | NA |
| International (overall) | 199/246 (80.9%) | 218/265 (82.3%) | (-8.9%, 4.2%) |
| Abscess | 73/93 (78.5%) | 86/99 (86.9%) | NA |
| Non-abscess | 126/153 (82.4%) | 132/166 (79.5%) | NA |
5.15 Development of Drug Resistant Bacteria
Prescribing moxifloxacin in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria [see Patient Counseling Information (17)].
7.3 Nonsteroidal Anti Inflammatory Drugs (nsaids)
Although not observed with moxifloxacin in preclinical and clinical trials, the concomitant administration of a nonsteroidal anti-inflammatory drug with a quinolone may increase the risks of CNS stimulation and convulsions [see Warnings and Precautions (5.4), and Patient Counseling Information (17)].
1.3 Complicated Skin and Skin Structure Infections
Moxifloxacin Injection is indicated in adults (18 years of age or older) for the treatment of Complicated Skin and Skin Structure Infections caused by susceptible isolates of methicillin-susceptible Staphylococcus aureus, Escherichia coli, Klebsiella pneumoniae, or Enterobacter cloacae [see Clinical Studies (14.6)].
14.6 Complicated Skin and Skin Structure Infections
Two randomized, active controlled trials of cSSSI were performed. A double-blind trial was conducted primarily in North America to compare the efficacy of sequential IV/PO moxifloxacin 400 mg QD for 7 to 14 days to an IV/PO beta-lactam/beta-lactamase inhibitor control in the treatment of patients with cSSSI. This study enrolled 617 patients, 335 of which were valid for the efficacy analysis. A second open-label International study compared moxifloxacin 400 mg QD for 7 to 21 days to sequential IV/PO beta-lactam/beta-lactamase inhibitor control in the treatment of patients with cSSSI. This study enrolled 804 patients, 632 of which were valid for the efficacy analysis. Surgical incision and drainage or debridement was performed on 55% of the moxifloxacin-treated and 53% of the comparator treated patients in these studies and formed an integral part of therapy for this indication. Success rates varied with the type of diagnosis ranging from 61% in patients with infected ulcers to 90% in patients with complicated erysipelas. These rates were similar to those seen with comparator drugs. The overall success rates in the evaluable patients and the clinical success by pathogen are shown in Tables 13 and 14.
|
* of difference in success rates between moxifloxacin and comparator (moxifloxacin - comparator) |
|||
|
Study |
Moxifloxacin
n/N (%) |
Comparator
n/N (%) |
95%
Confidence Interval* |
| North America | 125/162 (77.2%) | 141/173 (81.5%) | (-14.4%, 2%) |
| International | 254/315 (80.6%) | 268/317 (84.5%) | (-9.4%, 2.2%) |
|
a methicillin susceptibility was only determined in the North American Study |
||
|
Pathogen |
Moxifloxacin
n/N (%) |
Comparator
n/N (%) |
|
Staphylococcus aureus
(methicillin-susceptible isolates)a |
106/129 (82.2%) | 120/137 (87.6%) |
| Escherichia coli | 31/38 (81.6%) | 28/33 (84.8%) |
| Klebsiella pneumoniae | 11/12 (91.7%) | 7/10 (70%) |
| Enterobacter cloacae | 9/11 (81.8%) | 4/7 (57.1%) |
1.2 Uncomplicated Skin and Skin Structure Infections
Moxifloxacin Injection is indicated in adults (18 years of age or older) for the treatment of Uncomplicated Skin and Skin Structure Infections caused by susceptible isolates of methicillin-susceptible Staphylococcus aureus or Streptococcus pyogenes [see Clinical Studies (14.5)].
14.5 Uncomplicated Skin and Skin Structure Infections
A randomized, double-blind, controlled clinical trial conducted in the US compared the efficacy of moxifloxacin 400 mg once daily for seven days with cephalexin HCl 500 mg three times daily for seven days. The percentage of patients treated for uncomplicated abscesses was 30%, furuncles 8%, cellulitis 16%, impetigo 20%, and other skin infections 26%. Adjunctive procedures (incision and drainage or debridement) were performed on 17% of the moxifloxacin-treated patients and 14% of the comparator treated patients. Clinical success rates in evaluable patients were 89% (108/122) for moxifloxacin and 91% (110/121) for cephalexin HCl.
1.6 Acute Bacterial Exacerbation of Chronic Bronchitis
Moxifloxacin Injection is indicated in adults (18 years of age or older) for the treatment of Acute Bacterial Exacerbation of Chronic Bronchitis (ABECB) caused by susceptible isolates of Streptococcus pneumoniae, Haemophilus influenzae, Haemophilus parainfluenzae, Klebsiella pneumoniae, methicillin-susceptible Staphylococcus aureus, or Moraxella catarrhalis [see Clinical Studies (14.1)].
Because fluoroquinolones, including Moxifloxacin Injection, have been associated with serious adverse reactions [see Warnings and Precautions (5.1 to 5.14)] and for some patients ABECB is self-limiting, reserve Moxifloxacin Injection for treatment of ABECB in patients who have no alternative treatment options.
14.1 Acute Bacterial Exacerbation of Chronic Bronchitis
Moxifloxacin tablets (400 mg once daily for five days) were evaluated for the treatment of acute bacterial exacerbation of chronic bronchitis in a randomized, double-blind, controlled clinical trial conducted in the US. This study compared moxifloxacin with clarithromycin (500 mg twice daily for 10 days) and enrolled 629 patients. Clinical success was assessed at 7 to 17 days post-therapy. The clinical success for moxifloxacin was 89% (222/250) compared to 89% (224/251) for clarithromycin.
| Pathogen | Moxifloxacin | Clarithromycin |
| Streptococcus pneumoniae | 16/16 (100%) | 20/23 (87%) |
| Haemophilus influenzae | 33/37 (89%) | 36/41 (88%) |
| Haemophilus parainfluenzae | 16/16 (100%) | 14/14 (100%) |
| Moraxella catarrhalis | 29/34 (85%) | 24/24 (100%) |
| Staphylococcus aureus | 15/16 (94%) | 6/8 (75%) |
| Klebsiella pneumoniae | 18/20 (90%) | 10/11 (91%) |
The microbiological eradication rates (eradication plus presumed eradication) in moxifloxacin-treated patients were Streptococcus pneumoniae 100%, Haemophilus influenzae 89%, Haemophilus parainfluenzae 100%, Moraxella catarrhalis 85%, Staphylococcus aureus 94%, and Klebsiella pneumoniae 85%.
5.8 Other Serious and Sometimes Fatal Adverse Reactions
Other serious and sometimes fatal adverse reactions, some due to hypersensitivity, and some due to uncertain etiology, have been reported rarely in patients receiving therapy with quinolones, including moxifloxacin. These events may be severe and generally occur following the administration of multiple doses. Clinical manifestations may include one or more of the following:
- Fever, rash, or severe dermatologic reactions (for example, toxic epidermal necrolysis, Stevens-Johnson syndrome)
- Vasculitis; arthralgia; myalgia; serum sickness
- Allergic pneumonitis
- Interstitial nephritis; acute renal insufficiency or failure
- Hepatitis; jaundice; acute hepatic necrosis or failure
- Anemia, including hemolytic and aplastic; thrombocytopenia, including thrombotic thrombocytopenic purpura; leukopenia; agranulocytosis; pancytopenia; and/or other hematologic abnormalities
Discontinue Moxifloxacin Injection immediately at the first appearance of a skin rash, jaundice, or any other sign of hypersensitivity and supportive measures instituted [see Patient Counseling Information (17) and Adverse Reactions (6.2)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long term studies in animals to determine the carcinogenic potential of moxifloxacin have not been performed.
Moxifloxacin was not mutagenic in 4 bacterial strains (TA 98, TA 100, TA 1535, TA 1537) used in the Ames Salmonella reversion assay. As with other quinolones, the positive response observed with moxifloxacin in strain TA 102 using the same assay may be due to the inhibition of DNA gyrase. Moxifloxacin was not mutagenic in the CHO/HGPRT mammalian cell gene mutation assay. An equivocal result was obtained in the same assay when v79 cells were used. Moxifloxacin was clastogenic in the v79 chromosome aberration assay, but it did not induce unscheduled DNA synthesis in cultured rat hepatocytes. There was no evidence of genotoxicity in vivo in a micronucleus test or a dominant lethal test in mice.
Moxifloxacin had no effect on fertility in male and female rats at oral doses as high as 500 mg/kg/day, (approximately 12 times the maximum recommended human dose based on body surface area), or at intravenous doses as high as 45 mg/kg/day, (approximately equal to the maximum recommended human dose based on body surface area). At 500 mg/kg orally there were slight effects on sperm morphology (head-tail separation) in male rats and on the estrous cycle in female rats.
2.3 Preparation for Administration of Moxifloxacin Injection
To prepare Moxifloxacin Injection premix in flexible bags:
- Close flow control clamp of administration set.
- Remove cover from port at bottom of container.
- Insert piercing pin from an appropriate transfer set (for example, one that does not require excessive force, such as ISO compatible administration set) into port with a gentle twisting motion until pin is firmly seated.
NOTE: Refer to complete directions that have been provided with the administration set.
Because the premix flexible bags are for single-dose only, any unused portion should be discarded.
14.3 Community Acquired Pneumonia Caused By Multi Drug Resistant Streptococcus Pneumoniae
Moxifloxacin was effective in the treatment of community acquired pneumonia (CAP) caused by multi-drug resistant MDRSP* isolates. Of 37 microbiologically evaluable patients with MDRSP isolates, 35 patients (95%) achieved clinical and bacteriological success post-therapy. The clinical and bacteriological success rates based on the number of patients treated are shown in Table 11.
* MDRSP, Multi-drug resistant Streptococcus pneumoniae includes isolates previously known as PRSP (Penicillin-resistant S. pneumoniae), and are isolates resistant to two or more of the following antibiotics: penicillin (MIC ≥ 2 mcg/mL), 2nd generation cephalosporins (for example, cefuroxime), macrolides, tetracyclines, and trimethoprim/sulfamethoxazole.
|
a n = number of patients successfully treated; N = number of patients with MDRSP (from a total of 37 patients) |
||||
|
b n = number of patients successfully treated (presumed eradication or eradication); N = number of patients with MDRSP (from a total of 37 patients) |
||||
|
c One patient had a respiratory isolate that was resistant to penicillin and cefuroxime but a blood isolate that was intermediate to penicillin and cefuroxime. The patient is included in the database based on the respiratory isolate. |
||||
|
d Azithromycin, clarithromycin, and erythromycin were the macrolide antimicrobials tested. |
||||
| Screening Susceptibility | Clinical Success | Bacteriological Success | ||
| n/Na | % | n/Nb | % | |
| Penicillin-resistant | 21/21 | 100%c | 21/21 | 100%c |
| 2nd generation cephalosporin-resistant | 25/26 | 96%c | 25/26 | 96%c |
| Macrolide-resistantd | 22/23 | 96% | 22/23 | 96% |
| Trimethoprim/sulfamethoxazole-resistant | 28/30 | 93% | 28/30 | 93% |
| Tetracycline-resistant | 17/18 | 94% | 17/18 | 94% |
Not all isolates were resistant to all antimicrobial classes tested. Success and eradication rates are summarized in Table 12.
|
a One patient had a respiratory isolate resistant to 5 antimicrobials and a blood isolate resistant to 3 antimicrobials. The patient was included in the category resistant to 5 antimicrobials. |
||
| S. pneumoniae with MDRSP | Clinical Success | Bacteriological Eradication Rate |
| Resistant to 2 antimicrobials | 12/13 (92.3%) | 12/13 (92.3%) |
| Resistant to 3 antimicrobials | 10/11 (90.9%)a | 10/11 (90.9%)a |
| Resistant to 4 antimicrobials | 6/6 (100%) | 6/6 (100%) |
| Resistant to 5 antimicrobials | 7/7 (100%)a | 7/7 (100%)a |
| Bacteremia with MDRSP | 9/9 (100%) | 9/9 (100%) |
Warning: Serious Adverse Reactions Including Tendinitis, Tendon Rupture, Peripheral Neuropathy, Central Nervous System Effects and Exacerbation of Myasthenia Gravis
-
Fluoroquinolones, including moxifloxacin, have been associated with disabling and potentially irreversible serious adverse reactions that have occurred together
[see Warnings and Precautions (5.1)]
, including:
- Tendinitis and tendon rupture [see Warnings and Precautions (5.2)]
- Peripheral neuropathy [see Warnings and Precautions (5.3)]
- Central nervous system effects [see Warnings and Precautions (5.4)]
Discontinue Moxifloxacin Injection immediately and avoid the use of fluoroquinolones, including Moxifloxacin Injection, in patients who experience any of these serious adverse reactions [see Warnings and Precautions (5.1)].
- Fluoroquinolones, including moxifloxacin, may exacerbate muscle weakness in patients with myasthenia gravis. Avoid Moxifloxacin Injection in patients with known history of myasthenia gravis [see Warnings and Precautions (5.5)].
- Because fluoroquinolones, including moxifloxacin, have been associated with serious adverse reactions [see Warnings and Precautions (5.1 to 5.14)] , reserve Moxifloxacin Injection for use in patients who have no alternative treatment options for the following indications:
5.1 Disabling and Potentially Irreversible Serious Adverse Reactions Including Tendinitis and Tendon Rupture, Peripheral Neuropathy, and Central Nervous System Effects
Fluoroquinolones, including Moxifloxacin Injection, have been associated with disabling and potentially irreversible serious adverse reactions from different body systems that can occur together in the same patient. Commonly seen adverse reactions include tendinitis, tendon rupture, arthralgia, myalgia, peripheral neuropathy, and central nervous system effects (hallucinations, anxiety, depression, insomnia, severe headaches, and confusion).
These reactions can occur within hours to weeks after starting moxifloxacin. Patients of any age or without pre-existing risk factors have experienced these adverse reactions [see Warnings and Precautions (5.2, 5.3, 5.4)].
Discontinue Moxifloxacin Injection immediately at the first signs or symptoms of any serious adverse reaction. In addition, avoid the use of fluoroquinolones, including moxifloxacin, in patients who have experienced any of these serious adverse reactions associated with fluoroquinolones.
Structured Label Content
Section 42229-5 (42229-5)
Moxifloxacin Injection Solution for Infusion
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Moxifloxacin Injection should be administered by intravenous infusion only. It is not intended for intra-arterial, intramuscular, intrathecal, intraperitoneal, or subcutaneous administration.
Moxifloxacin Injection should be administered by intravenous infusion over a period of 60 minutes by direct infusion or through a Y-type intravenous infusion set which may already be in place. Caution: rapid or bolus intravenous infusion must be avoided.
Because only limited data are available on the compatibility of moxifloxacin intravenous injection with other intravenous substances, additives or other medications should not be added to Moxifloxacin Injection or infused simultaneously through the same intravenous line. If the same intravenous line or a Y-type line is used for sequential infusion of other drugs, or if the “piggyback” method of administration is used, the line should be flushed before and after infusion of Moxifloxacin Injection with an infusion solution compatible with moxifloxacin injection as well as with other drug(s) administered via this common line.
Moxifloxacin Injection is compatible with the following intravenous solutions at ratios from 1:10 to 10:1
| 0.9% Sodium Chloride Injection, USP | Sterile Water for Injection, USP |
| 1 molar Sodium Chloride Injection | 10% Dextrose for Injection, USP |
| 5% Dextrose Injection, USP | Lactated Ringer's for Injection |
Section 42231-1 (42231-1)
|
This Medication Guide has been approved by the U.S. Food and Drug Administration |
Revised:10/2022 |
|
|
Medication Guide
MOXIFLOXACIN (mox i FLOX a sin) (in jek shun) injection, for intravenous use |
||
| Read the Medication Guide that comes with moxifloxacin injection before you start receiving it and each time you receive it. There may be new information. This Medication Guide does not take the place of talking to your healthcare provider about your medical condition or your treatment. | ||
|
What is the most important information I should know about moxifloxacin injection?
|
||
|
2. Changes in sensation and possible nerve damage (peripheral neuropathy).
Damage to the nerves in arms, hands, legs, or feet can happen in people who take fluoroquinolones, including moxifloxacin. Stop receiving moxifloxacin immediately and talk to your healthcare provider right away if you get any of the following symptoms of peripheral neuropathy in your arms, hands, legs, or feet: |
||
|
|
|
| Moxifloxacin may need to be stopped to prevent permanent nerve damage. 3. Central Nervous System (CNS) effects. Seizures have been reported in people who take fluoroquinolone antibiotic medicines, including moxifloxacin. Tell your healthcare provider if you have a history of seizures before you start taking moxifloxacin. CNS side effects may happen as soon as after taking the first dose of moxifloxacin. Stop taking moxifloxacin immediately and talk to your healthcare provider right away if you get any of these side effects, or other changes in mood or behavior: |
||
|
|
|
|
4. Worsening of myasthenia gravis (a disease which causes muscle weakness). Fluoroquinolones like moxifloxacin injection may cause worsening of myasthenia gravis symptoms, including muscle weakness and breathing problems. Tell your healthcare provider if you have a history of myasthenia gravis. Moxifloxacin should not be used in people who have a history of myasthenia gravis. Call your healthcare provider right away if you have any worsening muscle weakness or breathing problems. See the section “ What are the possible side effects of moxifloxacin injection? ” for more information about side effects. |
||
What is moxifloxacin injection?
|
||
| Call your healthcare provider if you think your condition is not getting better while you are receiving moxifloxacin injection. | ||
|
Who should not receive moxifloxacin injection? Do not receive moxifloxacin injection if you have ever had an allergic reaction to moxifloxacin, other fluoroquinolone antibiotics, or any of the ingredients in moxifloxacin injection. Ask your healthcare provider if you are not sure. See the end of this Medication Guide for a complete list of ingredients in moxifloxacin injection. |
||
What should I tell my healthcare provider before receiving moxifloxacin injection?
|
||
|
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, herbal and dietary supplements. Moxifloxacin injection and other medicines can affect each other causing side effects. Especially tell your healthcare provider if you take:
|
||
|
Ask your healthcare provider if you are not sure if any of your medicines are listed above. Know the medicines you take. Keep a list of your medicines and show it to your healthcare provider and pharmacist when you get a new medicine. |
||
How should I receive moxifloxacin injection?
|
||
|
What should I avoid while receiving moxifloxacin injection?
|
||
|
What are the possible side effects of moxifloxacin injection?
|
||
|
|
|
|
||
| The most common side effects of moxifloxacin injection include: | ||
|
|
|
| These are not all the possible side effects of moxifloxacin injection. Tell your healthcare provider about any side effect that bothers you or that does not go away. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA- 1088. | ||
|
General Information about the safe and effective use of moxifloxacin injection.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. This Medication Guide summarizes the most important information about moxifloxacin injection. If you would like more information about moxifloxacin injection, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about moxifloxacin injection that is written for health professionals |
||
|
What are the ingredients in moxifloxacin injection? Lake Zurich, IL 60047 www.fresenius-kabi.com/us For more information, call 1-800-551-7176. 451327G |
Section 43683-2 (43683-2)
Section 51945-4 (51945-4)
PACKAGE LABEL - PRINCIPAL DISPLAY - Moxifloxacin 250 mL Bag Label
NDC 63323-850-04
850174
Moxifloxacin Injection
400 mg per 250 mL (1.6 mg per mL)
For Intravenous Infusion Rx Only
USE IMMEDIATELY ONCE REMOVED FROM THE OVERWRAP.
INFUSE OVER 60 MINUTES.
1.7 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Moxifloxacin Injection and other antibacterial drugs, Moxifloxacin Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
7.1 Warfarin
Quinolones, including moxifloxacin, have been reported to enhance the anticoagulant effects of warfarin or its derivatives in the patient population. In addition, infectious disease and its accompanying inflammatory process, age, and general status of the patient are risk factors for increased anticoagulant activity. Therefore, the prothrombin time, International Normalized Ratio (INR), or other suitable anticoagulation tests should be closely monitored if a quinolone is administered concomitantly with warfarin or its derivatives [see Adverse Reactions (6, 6.1,), Clinical Pharmacology (12.3), and Patient Counseling Information (17)].
10 Overdosage (10 OVERDOSAGE)
Single oral overdoses up to 2.8 g were not associated with any serious adverse events.
In the event of acute overdose, the stomach should be emptied and adequate hydration maintained. ECG monitoring is recommended due to the possibility of QT interval prolongation. The patient should be carefully observed and given supportive treatment. The administration of activated charcoal as soon as possible after oral overdose may prevent excessive increase of systemic moxifloxacin exposure. About 3% and 9% of the dose of moxifloxacin, as well as about 2% and 4.5% of its glucuronide metabolite are removed by continuous ambulatory peritoneal dialysis and hemodialysis, respectively.
11 Description (11 DESCRIPTION)
Moxifloxacin is a synthetic broad spectrum antibacterial agent for intravenous administration. Moxifloxacin, a fluoroquinolone, is available as a buffered monohydrochloride salt of 1-cyclopropyl-7-[(S,S)-2,8-diazabicyclo[4.3.0]non-8-yl]-6- fluoro-8-methoxy-1,4-dihydro-4-oxo-3 quinoline carboxylic acid. It is a slightly yellow to yellow crystalline substance. Its chemical structure is as follows:
Moxifloxacin Injection is sterile solution for infusion in a ready-to-use, single-dose flexible bag.
8.4 Pediatric Use
Effectiveness in pediatric patients and adolescents less than 18 years of age has not been established. Moxifloxacin causes arthropathy in juvenile animals. Limited information on the safety of Moxifloxacin in 301 pediatric patients is available from the cIAI trial [see Boxed Warning, Warnings and Precautions (5.9) and Nonclinical Toxicology (13.2)].
8.5 Geriatric Use
Geriatric patients are at increased risk for developing severe tendon disorders including tendon rupture when being treated with a fluoroquinolone such as Moxifloxacin Injection. This risk is further increased in patients receiving concomitant corticosteroid therapy. Tendinitis or tendon rupture can involve the Achilles, hand, shoulder, or other tendon sites and can occur during or after completion of therapy; cases occurring up to several months after fluoroquinolone treatment have been reported. Caution should be used when prescribing Moxifloxacin Injection to elderly patients especially those on corticosteroids. Patients should be informed of this potential side effect and advised to discontinue Moxifloxacin Injection and contact their healthcare provider if any symptoms of tendinitis or tendon rupture occur [see Boxed Warning, Warnings and Precautions (5.1, 5.2), and Adverse Reactions (6.2)].
Epidemiologic studies report an increased rate of aortic aneurysm and dissection within two months following use of fluoroquinolones, particularly in elderly patients [see Warnings and Precautions (5.9)].
Moxifloxacin Injection contains 1,207 mg (52.5 mEq) of sodium per unit dose. The geriatric population may respond with a blunted natriuresis to salt loading. This may be clinically important with regard to such diseases as congestive heart failure [see Warnings and Precautions (5.11)].
In controlled multiple-dose clinical trials, 23% of patients receiving oral moxifloxacin were greater than or equal to 65 years of age and 9% were greater than or equal to 75 years of age. The clinical trial data demonstrate that there is no difference in the safety and efficacy of oral moxifloxacin in patients aged 65 or older compared to younger adults.
In trials of intravenous use, 42% of moxifloxacin patients were greater than or equal to 65 years of age, and 23% were greater than or equal to 75 years of age. The clinical trial data demonstrate that the safety of intravenous moxifloxacin in patients aged 65 or older was similar to that of comparator-treated patients. In general, elderly patients may be more susceptible to drug-associated effects of the QT interval. Therefore, Moxifloxacin Injection should be avoided in patients taking drugs that can result in prolongation of the QT interval (for example, Class IA or Class III antiarrhythmics) or in patients with risk factors for torsades de pointes (for example, known QT prolongation, uncorrected hypokalemia) [see Warnings and Precautions (5.6), Drug Interactions (7.4), and Clinical Pharmacology (12.3)].
4 Contraindications (4 CONTRAINDICATIONS)
Moxifloxacin is contraindicated in persons with a history of hypersensitivity to moxifloxacin or any member of the quinolone class of antimicrobial agents.
5.6 Qt Prolongation (5.6 QT Prolongation)
Moxifloxacin has been shown to prolong the QT interval of the electrocardiogram in some patients. Following oral dosing with 400 mg of moxifloxacin the mean (± SD) change in QTc from the pre-dose value at the time of maximum drug concentration was 6 msec (± 26) (n = 787). Following a course of daily intravenous dosing (400 mg; 1 hour infusion each day) the mean change in QTc from the Day 1 pre-dose value was 10 msec (± 22) on Day 1 (n = 667) and 7 msec (± 24) on Day 3 (n = 667).
The drug should be avoided in patients with known prolongation of the QT interval, patients with uncorrected hypokalemia and patients receiving Class IA (for example, quinidine, procainamide) or Class III (for example, amiodarone, sotalol) antiarrhythmic agents, due to the lack of clinical experience with the drug in these patient populations.
Pharmacokinetic studies between moxifloxacin and other drugs that prolong the QT interval such as cisapride, erythromycin, antipsychotics, and tricyclic antidepressants have not been performed. An additive effect of moxifloxacin and these drugs cannot be excluded; therefore caution should be exercised when moxifloxacin is given concurrently with these drugs. In premarketing clinical trials, the rate of cardiovascular adverse events was similar in 798 moxifloxacin and 702 comparator treated patients who received concomitant therapy with drugs known to prolong the QTc interval.
Moxifloxacin should be used with caution in patients with ongoing proarrhythmic conditions, such as clinically significant bradycardia, acute myocardial ischemia. The magnitude of QT prolongation may increase with increasing concentrations of the drug or increasing rates of infusion of the intravenous formulation. Therefore the recommended dose or infusion rate should not be exceeded. QT prolongation may lead to an increased risk for ventricular arrhythmias including torsades de pointes. No excess in cardiovascular morbidity or mortality attributable to QTc prolongation occurred with moxifloxacin treatment in over 15,500 patients in controlled clinical studies, including 759 patients who were hypokalemic at the start of treatment, and there was no increase in mortality in over 18,000 moxifloxacin tablet treated patients in a postmarketing observational study in which ECGs were not performed. Elderly patients using Moxifloxacin Injection may be more susceptible to drug-associated QT prolongation [see Use in Specific Populations (8.5)]. In addition, moxifloxacin should be used with caution in patients with mild, moderate, or severe liver cirrhosis [see Clinical Pharmacology (12.3) and Patient Counseling Information (17)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious and otherwise important adverse reactions are discussed in greater detail in the Warnings and Precautions section of the label:
- Disabling and Potentially Irreversible Serious Adverse Reactions Including Tendinitis and Tendon Rupture, Peripheral Neuropathy, and Central Nervous System Effects [see Warnings and Precautions (5.1)]
- Tendinitis and Tendon Rupture [see Warnings and Precautions (5.2)]
- Peripheral Neuropathy [see Warnings and Precautions (5.3)]
- Central Nervous System Effects [see Warnings and Precautions (5.4)]
- Exacerbation of Myasthenia Gravis [see Warnings and Precautions (5.5)]
- QT Prolongation [see Warnings and Precautions (5.6)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.7)]
- Other Serious and Sometimes Fatal Adverse Reactions [see Warnings and Precautions (5.8)]
- Clostridioides Difficile-Associated Diarrhea [see Warnings and Precautions (5.10)]
- Blood Glucose Disturbances [see Warnings and Precautions (5.13)]
- Photosensitivity/Phototoxicity [see Warnings and Precautions (5.14)]
- Development of Drug Resistant Bacteria [see Warnings and Precautions (5.15)]
7 Drug Interactions (7 DRUG INTERACTIONS)
| Interacting Drug | Interaction |
| Warfarin | Anticoagulant effect of warfarin may be enhanced. Monitor prothrombin time/INR, watch for bleeding. (6.2, 7.1, 12.3) |
| Class IA and Class III antiarrhythmics: | Proarrhythmic effect may be enhanced. Avoid concomitant use. (5.6, 7.4) |
| Antidiabetic agents | Carefully monitor blood glucose. (5.13, 7.2) |
8.6 Renal Impairment
The pharmacokinetic parameters of moxifloxacin are not significantly altered in mild, moderate, severe, or end-stage renal disease. No dosage adjustment is necessary in patients with renal impairment, including those patients requiring hemodialysis (HD) or continuous ambulatory peritoneal dialysis (CAPD) [see Dosage and Administration (2), and Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
The mean (± SD) pharmacokinetic parameters of moxifloxacin following single and multiple dose of 400 mg moxifloxacin given by 1 hour intravenous infusion are summarized in Table 7. The mean (± SD) elimination half-life from plasma is 12 ± 1.3 hours; steady-state is achieved after at least three days with a 400 mg once daily regimen. The absolute bioavailability of moxifloxacin is approximately 90 percent. When switching from intravenous to oral formulation, no dosage adjustment is necessary [see Dosage and Administration (2.1)].
|
a Range of means from different studies |
|||
|
b Expected Cmax (concentration obtained around the time of the end of the infusion) |
|||
|
Cmax
(mg/L) |
AUC
(mg•h/L) |
Half-life (hr) | |
| Single Dose IV | |||
| Healthy young male/female (n = 56) | 3.9 ± 0.9 | 39.3 ± 8.6 | 8.2 to 15.4a |
| Patients (n = 118) | |||
| Male (n = 64) | 4.4 ± 3.7 | ||
| Female (n = 54) | 4.5 ± 2 | ||
| < 65 years (n = 58) | 4.6 ± 4.2 | ||
| ≥ 65 years (n = 60) | 4.3 ± 1.3 | ||
| Multiple Dose IV | |||
| Healthy young male (n = 8) Healthy elderly (n =12; 8 male, 4 female) |
4.2 ± 0.8 6.1 ± 1.3 |
38 ± 4.7 48.2 ± 0.9 |
14.8 ± 2.2 10.1 ± 1.6 |
| Patientsb (n = 107) | |||
| Male (n = 58) | 4.2 ± 2.6 | ||
| Female (n = 49) | 4.6 ± 1.5 | ||
| < 65 years (n = 52) | 4.1 ± 1.4 | ||
| ≥ 65 years (n = 55) | 4.7 ± 2.7 |
5.11 High Sodium Load
8.7 Hepatic Impairment
No dosage adjustment is recommended for mild, moderate, or severe hepatic insufficiency (Child-Pugh Classes A, B, or C). However, due to metabolic disturbances associated with hepatic insufficiency, which may lead to QT prolongation, moxifloxacin should be used with caution in these patients [see Warnings and Precautions (5.6), and Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Moxifloxacin Injection is a fluoroquinolone antibacterial drug indicated for treating infections in adults ≥ 18 years of age caused by designated, susceptible bacteria. (1, 12.4)
- Community Acquired Pneumonia (1.1)
- Skin and Skin Structure Infections: Uncomplicated (1.2) and Complicated (1.3)
- Complicated Intra-Abdominal Infections (1.4)
- Acute Bacterial Sinusitis (1.5)
- Acute Bacterial Exacerbation of Chronic Bronchitis (1.6)
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Moxifloxacin Injection and other antibacterial drugs, Moxifloxacin Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. (1.7)
7.2 Antidiabetic Agents
Disturbances of blood glucose, including hyperglycemia and hypoglycemia, have been reported in patients treated concomitantly with fluoroquinolones and an antidiabetic agent. Therefore, careful monitoring of blood glucose is recommended when these agents are co-administered. If a hypoglycemic reaction occurs, moxifloxacin should be discontinued and appropriate therapy should be initiated immediately [see Warnings and Precautions (5.13), Adverse Reactions (6.1), and Patient Counseling Information (17)].
12.1 Mechanism of Action
Moxifloxacin is a member of the fluoroquinolone class of antibacterial agents [see Microbiology (12.4)].
5.3 Peripheral Neuropathy
Fluoroquinolones, including moxifloxacin, have been associated with an increased risk of peripheral neuropathy. Cases of sensory or sensorimotor axonal polyneuropathy affecting small and/or large axons resulting in paresthesias, hypoesthesias, dysesthesias and weakness have been reported in patients receiving fluoroquinolones including moxifloxacin. Symptoms may occur soon after initiation of moxifloxacin and may be irreversible in some patients [see Warnings and Precautions (5.1) and Adverse Reactions (6, 6.1, 6.2)].
Discontinue moxifloxacin immediately if the patient experiences symptoms of peripheral neuropathy including pain, burning, tingling, numbness, and/or weakness or other alterations of sensation including light touch, pain, temperature, position sense, and vibratory sensation. Avoid fluoroquinolones, including moxifloxacin, in patients who have previously experienced peripheral neuropathy [see Adverse Reactions (6.1, 6.2) and Patient Counseling Information (17)].
7.4 Drugs That Prolong Qt (7.4 Drugs that Prolong QT)
There is limited information available on the potential for a pharmacodynamic interaction in humans between moxifloxacin and other drugs that prolong the QTc interval of the electrocardiogram. Sotalol, a Class III antiarrhythmic, has been shown to further increase the QTc interval when combined with high doses of intravenous (IV) moxifloxacin in dogs. Therefore, moxifloxacin should be avoided with Class IA and Class III antiarrhythmics [see Warnings and Precautions (5.6), Nonclinical Toxicology (13.2), and Patient Counseling Information (17)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Prolongation of the QT interval and isolated cases of torsades de pointes has been reported. Avoid use in patients with known prolongation, hypokalemia, and with drugs that prolong the QT interval. (5.6, 7.4, 8.5). Use caution in patients with proarrhythmic conditions such as clinically significant bradycardia or acute myocardial ischemia. (5.6)
- Serious and sometimes fatal hypersensitivity reactions, including anaphylactic reactions, may occur after first or subsequent doses. Discontinue moxifloxacin at the first sign of skin rash, jaundice or any other sign of hypersensitivity. (5.7, 5.8)
- Clostridioides difficile-associated diarrhea: Evaluate if diarrhea occurs. (5.10)
- High sodium load: each unit dose contains 52.5 mEq (1,207 mg) of sodium. Avoid in patients with sodium restriction. (5.11)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
|
Type of Infection |
Dose Every 24 hours | Duration (days) |
| Community Acquired Pneumonia (1.1) | 400 mg | 7 to 14 |
| Uncomplicated Skin and Skin Structure Infections (SSSI) (1.2) |
400 mg |
7 |
| Complicated SSSI (1.3) | 400 mg | 7 to 21 |
| Complicated Intra-Abdominal Infections (1.4) | 400 mg | 5 to 14 |
| Acute Bacterial Sinusitis (1.5) | 400 mg | 10 |
| Acute Bacterial Exacerbation of Chronic Bronchitis (1.6) |
400 mg |
5 |
2.1 Dosage in Adult Patients
The dose of Moxifloxacin Injection is 400 mg intravenously once every 24 hours. The duration of therapy depends on the type of infection as described in Table 1.
|
a Due to the designated pathogens [see Indications and Usage (1), for IV use, see Use in Specific Populations (8.5)]. |
||
|
b Sequential therapy (intravenous to oral) may be instituted at the discretion of the physician. |
||
|
Type of Infection a |
Dose Every 24 hours | Duration b (days) |
| Community Acquired Pneumonia (1.1) | 400 mg | 7 to 14 |
| Uncomplicated Skin and Skin Structure Infections (SSSI) (1.2) | 400 mg | 7 |
| Complicated SSSI (1.3) | 400 mg | 7 to 21 |
| Complicated Intra-Abdominal Infections (1.4) | 400 mg | 5 to 14 |
| Acute Bacterial Sinusitis (1.5) | 400 mg | 10 |
| Acute Bacterial Exacerbation of Chronic Bronchitis (1.6) | 400 mg | 5 |
When switching from intravenous to oral formulation, no dosage adjustment is necessary [see Clinical Pharmacology (12.4)]. Patients whose therapy is started with Moxifloxacin Injection may be switched to moxifloxacin tablets when clinically indicated at the discretion of the physician.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: 400 mg moxifloxacin in 250 mL single-dose flexible bag. (3.1)
6.2 Postmarketing Experience
Table 4 lists adverse reactions that have been identified during post-approval use of moxifloxacin. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
| System/Organ Class | Adverse Reaction |
| Blood and Lymphatic System Disorders | Agranulocytosis Pancytopenia [see Warnings and Precautions (5.8)] |
| Cardiac Disorders | Ventricular tachyarrhythmias (including in very rare cases cardiac arrest and torsades de pointes, and usually in patients with concurrent severe underlying proarrhythmic conditions) |
| Ear and Labyrinth Disorders | Hearing impairment, including deafness (reversible in majority of cases) |
| Eye Disorders | Vision loss (especially in the course of CNS reactions, transient in majority of cases) |
| Hepatobiliary Disorders | Hepatitis (predominantly cholestatic) Hepatic failure (including fatal cases) Jaundice Acute hepatic necrosis [see Warnings and Precautions (5.8)] |
| Immune System Disorders | Anaphylactic reaction Anaphylactic shock Angioedema (including laryngeal edema) [see Warnings and Precautions (5.7, 5.8)] |
| Musculoskeletal and Connective Tissue Disorders | Tendon rupture [see Warnings and Precautions (5.2)] |
| Nervous System Disorders | Altered coordination Abnormal gait [see Warnings and Precautions (5.3)] Myasthenia gravis (exacerbation of) [see Warnings and Precautions (5.5)] Muscle weakness Peripheral neuropathy (that may be irreversible), polyneuropathy [see Warnings and Precautions (5.3)] |
| Psychiatric Disorders | Psychotic reaction (very rarely culminating in self- injurious behavior, such as suicidal ideation/thoughts or suicide attempts [see Warnings and Precautions (5.4)] |
| Renal and Urinary Disorders | Renal dysfunction Interstitial nephritis [see Warnings and Precautions (5.8)] |
| Respiratory, Thoracic and Mediastinal Disorders | Allergic pneumonitis [see Warnings and Precautions (5.8)] |
| Skin and Subcutaneous Tissue Disorders | Photosensitivity/phototoxicity reaction [see Warnings and Precautions (5.14)] Stevens-Johnson syndrome Toxic epidermal necrolysis [see Warnings and Precautions (5.8)] |
1.5 Acute Bacterial Sinusitis
Moxifloxacin Injection is indicated in adults (18 years of age or older) for the treatment of Acute Bacterial Sinusitis (ABS) caused by susceptible isolates of Streptococcus pneumoniae, Haemophilus influenzae, or Moraxella catarrhalis [see Clinical Studies (14.4)].
Because fluoroquinolones, including Moxifloxacin Injection, have been associated with serious adverse reactions [see Warnings and Precautions (5.1 to 5.14)] and for some patients ABS is self-limiting, reserve Moxifloxacin Injection for treatment of ABS in patients who have no alternative treatment options.
5.10 Clostridioides Difficile
Clostridioides difficile-associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including moxifloxacin, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibiotic use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated [see Adverse Reactions (6.1) and Patient Counseling Information (17)].
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
14.4 Acute Bacterial Sinusitis
In a controlled double-blind study conducted in the US, moxifloxacin tablets (400 mg once daily for ten days) were compared with cefuroxime axetil (250 mg twice daily for ten days) for the treatment of acute bacterial sinusitis. The trial included 457 patients valid for the efficacy analysis. Clinical success (cure plus improvement) at the 7 to 21 day post-therapy test of cure visit was 90% for moxifloxacin and 89% for cefuroxime.
An additional non-comparative study was conducted to gather bacteriological data and to evaluate microbiological eradication in adult patients treated with moxifloxacin 400 mg once daily for seven days. All patients (n = 336) underwent antral puncture in this study. Clinical success rates and eradication/presumed eradication rates at the 21 to 37 day follow-up visit were 97% (29 out of 30) for Streptococcus pneumoniae, 83% (15 out of 18) for Moraxella catarrhalis, and 80% (24 out of 30) for Haemophilus influenzae.
5.7 Hypersensitivity Reactions
Serious anaphylactic reactions, some following the first dose, have been reported in patients receiving fluoroquinolone therapy, including moxifloxacin. Some reactions were accompanied by cardiovascular collapse, loss of consciousness, tingling, pharyngeal or facial edema, dyspnea, urticaria, and itching. Discontinue Moxifloxacin Injection at the first appearance of a skin rash or any other sign of hypersensitivity [see Warnings and Precautions (5.7), Adverse Reactions (6) and Patient Counseling Information (17)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below reflect exposure to moxifloxacin in 14,981 patients in 71 active controlled Phase II - IV clinical trials in different indications [see Indications and Usage (1)]. The population studied had a mean age of 50 years (approximately 73% of the population was < 65 years of age), 50% were male, 63% were Caucasian, 12% were Asian and 9% were Black. Patients received moxifloxacin 400 mg once daily PO, IV, or sequentially (IV followed by PO). Treatment duration was usually 6 to 10 days, and the mean number of days on therapy was 9 days.
Discontinuation of moxifloxacin due to adverse events occurred in 5% of patients overall, 4.1% of patients treated with 400 mg PO, 3.9% with 400 mg IV and 8.2% with sequential therapy 400 mg PO/IV. The most common adverse events leading to discontinuation with the 400 mg PO doses were nausea (0.8%), diarrhea (0.5%), dizziness (0.5%), and vomiting (0.4%). The most common adverse event leading to discontinuation with the 400 mg IV dose was rash (0.5%). The most common adverse events leading to discontinuation with the 400 mg IV/PO sequential dose were diarrhea (0.5%) and pyrexia (0.4%).
Adverse reactions occurring in ≥ 1% of moxifloxacin-treated patients and less common adverse reactions, occurring in 0.1 to < 1% of moxifloxacin-treated patients, are shown in Table 2 and Table 3, respectively. The most common adverse drug reactions (≥ 3%) are nausea, diarrhea, headache, and dizziness.
|
a MedDRA Version 12.0 |
||
|
System Organ Class |
Adverse Reactions a |
% (N=14,981) |
| Blood and Lymphatic System Disorders | Anemia | 1.1 |
| Gastrointestinal Disorders | Nausea | 6.9 |
| Diarrhea | 6 | |
| Vomiting | 2.4 | |
| Constipation | 1.9 | |
| Abdominal pain | 1.5 | |
| Abdominal pain upper | 1.1 | |
| Dyspepsia | 1 | |
| General Disorders and Administration Site Conditions | Pyrexia | 1.1 |
| Investigations | Alanine aminotransferase increased | 1.1 |
| Metabolism and Nutritional Disorder | Hypokalemia | 1 |
| Nervous System Disorders | Headache | 4.2 |
| Dizziness | 3 | |
| Psychiatric Disorders | Insomnia | 1.9 |
|
a MedDRA Version 12.0 |
|
| System Organ Class | Adverse Reactions a |
| Blood and Lymphatic System Disorders | Thrombocythemia Eosinophilia Neutropenia Thrombocytopenia Leukopenia Leukocytosis |
| Cardiac Disorders | Atrial fibrillation Palpitations Tachycardia Cardiac failure congestive Angina pectoris Cardiac failure Cardiac arrest Bradycardia |
| Ear and Labyrinth Disorders | Vertigo Tinnitus |
| Eye Disorders | Vision blurred |
| Gastrointestinal Disorders | Dry mouth Abdominal discomfort Flatulence Abdominal distention Gastritis Gastroesophageal reflux disease |
| General Disorders and Administration Site Conditions | Fatigue Chest pain Asthenia Edema peripheral Pain Malaise |
| System Organ Class | Adverse Reactions a |
| Infusion site extravasation Edema Chills Chest discomfort Facial pain |
|
| Hepatobiliary Disorders | Hepatic function abnormal |
| Infections and Infestations | Vulvovaginal candidiasis Oral candidiasis Vulvovaginal mycotic infection Candidiasis Vaginal infection Oral fungal infection Fungal infection Gastroenteritis |
| Investigations | Aspartate aminotransferase increased Gamma-glutamyltransferase increased Blood alkaline phosphatase increased Hepatic enzyme increased Electrocardiogram QT prolonged Blood lactate dehydrogenase increased Platelet count increased Blood amylase increased Blood glucose increased Lipase increased Hemoglobin decreased Blood creatinine increased Transaminases increased White blood cell count increased Blood urea increased Liver function test abnormal Hematocrit decreased Prothrombin time prolonged Eosinophil count increased Activated partial thromboplastin time prolonged Blood bilirubin increased Blood triglycerides increased Blood uric acid increased Blood pressure increased |
| Metabolism and Nutrition Disorders | Hyperglycemia Anorexia Hypoglycemia Hyperlipidemia Decreased appetite Dehydration |
| Musculoskeletal and Connective Tissue Disorders | Back pain Pain in extremity Arthralgia Myalgia Muscle spasms Musculoskeletal chest pain Musculoskeletal pain |
| Nervous System Disorders | Dysgeusia Somnolence Tremor Lethargy |
| System Organ Class | Adverse Reactions a |
| Paresthesia Tension headache Hypoesthesia Syncope |
|
| Psychiatric Disorders | Anxiety Confusional state Agitation Depression Nervousness Restlessness Hallucination Disorientation |
| Renal and Urinary Disorders | Renal failure Dysuria Renal failure acute |
| Reproductive System and Breast Disorders | Vulvovaginal pruritus |
| Respiratory, Thoracic, and Mediastinal Disorders | Dyspnea Asthma Wheezing Bronchospasm |
| Skin and Subcutaneous Tissue Disorders | Rash Pruritus Hyperhidrosis Erythema Urticaria Dermatitis allergic Night sweats |
| Vascular Disorders | Hypertension Hypotension Phlebitis |
5.13 Blood Glucose Disturbances
As with all fluoroquinolones, disturbances in blood glucose, including both hypoglycemia and hyperglycemia have been reported with moxifloxacin. In moxifloxacin-treated patients, dysglycemia occurred predominantly in elderly diabetic patients receiving concomitant treatment with an oral hypoglycemic agent (for example, sulfonylurea) or with insulin. Severe cases of hypoglycemia resulting in coma or death have been reported. In diabetic patients, careful monitoring of blood glucose is recommended [see Adverse Reactions (6.1)]. If a hypoglycemic reaction occurs, discontinue moxifloxacin and initiate appropriate therapy immediately [see Adverse Reactions (6.1), Drug Interactions (7.2) and Patient Counseling Information (17)].
1.1 Community Acquired Pneumonia
Moxifloxacin Injection is indicated in adults (18 years of age or older) for the treatment of Community Acquired Pneumonia caused by susceptible isolates of Streptococcus pneumoniae (including multi-drug resistant isolates*), Haemophilus influenzae, Moraxella catarrhalis, methicillin-susceptible Staphylococcus aureus, Klebsiella pneumoniae, Mycoplasma pneumoniae, or Chlamydophila pneumoniae.
* MDRSP, Multi-drug resistant Streptococcus pneumoniae includes isolates previously known as PRSP (Penicillin-resistant S. pneumoniae), and are isolates resistant to two or more of the following antibiotics: penicillin (minimum inhibitory concentrations [MIC] ≥ 2 mcg/mL), 2nd generation cephalosporins (for example, cefuroxime), macrolides, tetracyclines, and trimethoprim/sulfamethoxazole [see Clinical Studies (14.2)].
14.2 Community Acquired Pneumonia
A randomized, double-blind, controlled clinical trial was conducted in the US to compare the efficacy of moxifloxacin tablets (400 mg once daily) to that of high-dose clarithromycin (500 mg twice daily) in the treatment of patients with clinically and radiologically documented community acquired pneumonia. This study enrolled 474 patients (382 of whom were valid for the efficacy analysis conducted at the 14 to 35 day follow-up visit). Clinical success for clinically evaluable patients was 95% (184/194) for moxifloxacin and 95% (178/188) for high dose clarithromycin.
A randomized, double-blind, controlled trial was conducted in the US and Canada to compare the efficacy of sequential IV/PO moxifloxacin 400 mg QD for 7 to 14 days to an IV/PO fluoroquinolone control (trovafloxacin or levofloxacin) in the treatment of patients with clinically and radiologically documented community acquired pneumonia. This study enrolled 516 patients, 362 of whom were valid for the efficacy analysis conducted at the 7 to 30 day post-therapy visit. The clinical success rate was 86% (157/182) for moxifloxacin therapy and 89% (161/180) for the fluoroquinolone comparators.
An open-label ex-US study that enrolled 628 patients compared moxifloxacin to sequential IV/PO amoxicillin/clavulanate (1.2 g IV q8h/625 mg PO q8h) with or without high-dose IV/PO clarithromycin (500 mg BID). The intravenous formulations of the comparators are not FDA approved. The clinical success rate at Day 5 to 7 for moxifloxacin therapy was 93% (241/258) and demonstrated superiority to amoxicillin/clavulanate ± clarithromycin (85%, 239/280) [95% C.I. of difference in success rates between moxifloxacin and comparator (2.9%, 13.2%)]. The clinical success rate at the 21 to 28 days post-therapy visit for moxifloxacin was 84% (216/258), which also demonstrated superiority to the comparators (74%, 208/280) [95% C.I. of difference in success rates between moxifloxacin and comparator (2.6%, 16.3%)].
The clinical success rates by pathogen across four CAP studies are presented in Table 10.
| Pathogen | Moxifloxacin | |
| Streptococcus pneumoniae | 80/85 | (94%) |
| Staphylococcus aureus | 17/20 | (85%) |
| Klebsiella pneumoniae | 11/12 | (92%) |
| Haemophilus influenzae | 56/61 | (92%) |
| Chlamydophila pneumoniae | 119/128 | (93%) |
| Mycoplasma pneumoniae | 73/76 | (96%) |
| Moraxella catarrhalis | 11/12 | (92%) |
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise patients to read the FDA-approved patient labeling (Medication Guide).
5.2 Tendinitis and Tendon Rupture
Fluoroquinolones, including moxifloxacin, have been associated with an increased risk of tendinitis and tendon rupture in all ages [see Warnings and Precautions (5.1) and Adverse Reactions (6.1)]. This adverse reaction most frequently involves the Achilles tendon, and has also been reported with the rotator cuff (the shoulder), the hand, the biceps, the thumb, and other tendons. Tendinitis or tendon rupture can occur within hours or days of starting moxifloxacin or as long as several months after completion of therapy. Tendinitis and tendon rupture can occur bilaterally.
The risk of developing fluoroquinolone-associated tendinitis and tendon rupture is increased in patients over 60 years of age, in patients taking corticosteroid drugs, and in patients with kidney, heart or lung transplants. Other factors that may independently increase the risk of tendon rupture include strenuous physical activity, renal failure, and previous tendon disorders such as rheumatoid arthritis. Tendinitis and tendon rupture have also occurred in patients taking fluoroquinolones who do not have the above risk factors. Discontinue moxifloxacin if the patient experiences pain, swelling, inflammation or rupture of a tendon. Patients should be advised to rest at the first sign of tendinitis or tendon rupture, and to contact their healthcare provider regarding changing to a non- quinolone antimicrobial drug [see Adverse Reactions (6.2) and Patient Counseling Information (17)]. Avoid fluoroquinolones, including moxifloxacin, in patients who have a history of tendon disorders or have experienced tendinitis or tendon rupture [see Adverse Reactions (6.1)].
5.14 Photosensitivity/phototoxicity (5.14 Photosensitivity/Phototoxicity)
Moderate to severe photosensitivity/phototoxicity reactions, the latter of which may manifest as exaggerated sunburn reactions (for example, burning, erythema, exudation, vesicles, blistering, edema) involving areas exposed to light (typically the face, “V” area of the neck, extensor surfaces of the forearms, dorsa of the hands), can be associated with the use of quinolone antibiotics after sun or UV light exposure. Therefore, excessive exposure to these sources of light should be avoided. Drug therapy should be discontinued if phototoxicity occurs [see Adverse Reactions (6.2) and Clinical Pharmacology (12.3)].
5.12 Arthropathic Effects in Animals
The oral administration of moxifloxacin caused lameness in immature dogs. Histopathological examination of the weight-bearing joints of these dogs revealed permanent lesions of the cartilage. Related quinolone-class drugs also produce erosions of cartilage of weight-bearing joints and other signs of arthropathy in immature animals of various species [see Nonclinical Toxicology (13.2)].
5.5 Exacerbation of Myasthenia Gravis
Fluoroquinolones, including moxifloxacin, have neuromuscular blocking activity and may exacerbate muscle weakness in patients with myasthenia gravis. Postmarketing serious adverse reactions, including deaths and requirement for ventilatory support, have been associated with fluoroquinolone use in patients with myasthenia gravis. Avoid moxifloxacin in patients with known history of myasthenia gravis [see Patient Counseling Information (17)].
1.4 Complicated Intra Abdominal Infections (1.4 Complicated Intra-Abdominal Infections)
Moxifloxacin Injection is indicated in adults (18 years of age or older) for the treatment of Complicated Intra-Abdominal Infections including polymicrobial infections such as abscess caused by susceptible isolates of Escherichia coli, Bacteroides fragilis, Streptococcus anginosus, Streptococcus constellatus, Enterococcus faecalis, Proteus mirabilis, Clostridium perfringens, Bacteroides thetaiotaomicron, or Peptostreptococcus species [see Clinical Studies (14.7)].
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Quinolones have been shown to cause arthropathy in immature animals. In studies in juvenile dogs oral doses of moxifloxacin ≥ 30 mg/kg/day (approximately 1.5 times the maximum recommended human dose based upon systemic exposure) for 28 days resulted in arthropathy. There was no evidence of arthropathy in mature monkeys and rats at oral doses up to 135 and 500 mg/kg/day, respectively.
Moxifloxacin at an oral dose of 300 mg/kg did not show an increase in acute toxicity or potential for CNS toxicity (for example, seizures) in mice when used in combination with NSAIDs such as diclofenac, ibuprofen, or fenbufen. Some quinolones have been reported to have proconvulsant activity that is exacerbated with concomitant use of non- steroidal anti-inflammatory drugs (NSAIDs).
A QT-prolonging effect of moxifloxacin was found in dog studies, at plasma concentrations about five times the human therapeutic level. The combined infusion of sotalol, a Class III antiarrhythmic agent, with moxifloxacin induced a higher degree of QTc prolongation in dogs than that induced by the same dose (30 mg/kg) of moxifloxacin alone. Electrophysiological in vitro studies suggested an inhibition of the rapid activating component of the delayed rectifier potassium current (IKr) as an underlying mechanism.
No signs of local intolerability were observed in dogs when moxifloxacin was administered intravenously. After intra-arterial injection, inflammatory changes involving the peri-arterial soft tissue were observed suggesting that intra-arterial administration of moxifloxacin should be avoided.
5.9 Risk of Aortic Aneurysm and Dissection
Epidemiologic studies report an increased rate of aortic aneurysm and dissection within two months following use of fluoroquinolones, particularly in elderly patients. The cause for the increased risk has not been identified. In patients with a known aortic aneurysm or patients who are at greater risk for aortic aneurysms, reserve Moxifloxacin Injection for use only when there are no alternative antibacterial treatments available.
14.7 Complicated Intra Abdominal Infections (14.7 Complicated Intra-Abdominal Infections)
Two randomized, active controlled trials of cIAI were performed. A double-blind trial was conducted primarily in North America to compare the efficacy of sequential IV/PO moxifloxacin 400 mg QD for 5 to 14 days to IV/piperacillin/tazobactam followed by PO amoxicillin/clavulanic acid in the treatment of patients with cIAI, including peritonitis, abscesses, appendicitis with perforation, and bowel perforation. This study enrolled 681 patients, 379 of which were considered clinically evaluable. A second open-label international study compared moxifloxacin 400 mg QD for 5 to 14 days to IV ceftriaxone plus IV metronidazole followed by PO amoxicillin/clavulanic acid in the treatment of patients with cIAI. This study enrolled 595 patients, 511 of which were considered clinically evaluable. The clinically evaluable population consisted of subjects with a surgically confirmed complicated infection, at least 5 days of treatment and a 25 to 50 day follow-up assessment for patients at the Test of Cure visit. The overall clinical success rates in the clinically evaluable patients are shown in Table 15.
|
a of difference in success rates between moxifloxacin and comparator (moxifloxacin – comparator) |
|||
|
b Excludes 2 patients who required additional surgery within the first 48 hours. |
|||
|
c NA – not applicable |
|||
|
Study |
Moxifloxacin
n/N (%) |
Comparator
n/N (%) |
95%
Confidence Interval a |
| North America (overall) | 146/183 (79.8%) | 153/196 (78.1%) | (-7.4%, 9.3%) |
| Abscess | 40/57 (70.2%) | 49/63 (77.8%)b | NAc |
| Non-abscess | 106/126 (84.1%) | 104/133 (78.2%) | NA |
| International (overall) | 199/246 (80.9%) | 218/265 (82.3%) | (-8.9%, 4.2%) |
| Abscess | 73/93 (78.5%) | 86/99 (86.9%) | NA |
| Non-abscess | 126/153 (82.4%) | 132/166 (79.5%) | NA |
5.15 Development of Drug Resistant Bacteria
Prescribing moxifloxacin in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria [see Patient Counseling Information (17)].
7.3 Nonsteroidal Anti Inflammatory Drugs (nsaids) (7.3 Nonsteroidal Anti-Inflammatory Drugs (NSAIDs))
Although not observed with moxifloxacin in preclinical and clinical trials, the concomitant administration of a nonsteroidal anti-inflammatory drug with a quinolone may increase the risks of CNS stimulation and convulsions [see Warnings and Precautions (5.4), and Patient Counseling Information (17)].
1.3 Complicated Skin and Skin Structure Infections
Moxifloxacin Injection is indicated in adults (18 years of age or older) for the treatment of Complicated Skin and Skin Structure Infections caused by susceptible isolates of methicillin-susceptible Staphylococcus aureus, Escherichia coli, Klebsiella pneumoniae, or Enterobacter cloacae [see Clinical Studies (14.6)].
14.6 Complicated Skin and Skin Structure Infections
Two randomized, active controlled trials of cSSSI were performed. A double-blind trial was conducted primarily in North America to compare the efficacy of sequential IV/PO moxifloxacin 400 mg QD for 7 to 14 days to an IV/PO beta-lactam/beta-lactamase inhibitor control in the treatment of patients with cSSSI. This study enrolled 617 patients, 335 of which were valid for the efficacy analysis. A second open-label International study compared moxifloxacin 400 mg QD for 7 to 21 days to sequential IV/PO beta-lactam/beta-lactamase inhibitor control in the treatment of patients with cSSSI. This study enrolled 804 patients, 632 of which were valid for the efficacy analysis. Surgical incision and drainage or debridement was performed on 55% of the moxifloxacin-treated and 53% of the comparator treated patients in these studies and formed an integral part of therapy for this indication. Success rates varied with the type of diagnosis ranging from 61% in patients with infected ulcers to 90% in patients with complicated erysipelas. These rates were similar to those seen with comparator drugs. The overall success rates in the evaluable patients and the clinical success by pathogen are shown in Tables 13 and 14.
|
* of difference in success rates between moxifloxacin and comparator (moxifloxacin - comparator) |
|||
|
Study |
Moxifloxacin
n/N (%) |
Comparator
n/N (%) |
95%
Confidence Interval* |
| North America | 125/162 (77.2%) | 141/173 (81.5%) | (-14.4%, 2%) |
| International | 254/315 (80.6%) | 268/317 (84.5%) | (-9.4%, 2.2%) |
|
a methicillin susceptibility was only determined in the North American Study |
||
|
Pathogen |
Moxifloxacin
n/N (%) |
Comparator
n/N (%) |
|
Staphylococcus aureus
(methicillin-susceptible isolates)a |
106/129 (82.2%) | 120/137 (87.6%) |
| Escherichia coli | 31/38 (81.6%) | 28/33 (84.8%) |
| Klebsiella pneumoniae | 11/12 (91.7%) | 7/10 (70%) |
| Enterobacter cloacae | 9/11 (81.8%) | 4/7 (57.1%) |
1.2 Uncomplicated Skin and Skin Structure Infections
Moxifloxacin Injection is indicated in adults (18 years of age or older) for the treatment of Uncomplicated Skin and Skin Structure Infections caused by susceptible isolates of methicillin-susceptible Staphylococcus aureus or Streptococcus pyogenes [see Clinical Studies (14.5)].
14.5 Uncomplicated Skin and Skin Structure Infections
A randomized, double-blind, controlled clinical trial conducted in the US compared the efficacy of moxifloxacin 400 mg once daily for seven days with cephalexin HCl 500 mg three times daily for seven days. The percentage of patients treated for uncomplicated abscesses was 30%, furuncles 8%, cellulitis 16%, impetigo 20%, and other skin infections 26%. Adjunctive procedures (incision and drainage or debridement) were performed on 17% of the moxifloxacin-treated patients and 14% of the comparator treated patients. Clinical success rates in evaluable patients were 89% (108/122) for moxifloxacin and 91% (110/121) for cephalexin HCl.
1.6 Acute Bacterial Exacerbation of Chronic Bronchitis
Moxifloxacin Injection is indicated in adults (18 years of age or older) for the treatment of Acute Bacterial Exacerbation of Chronic Bronchitis (ABECB) caused by susceptible isolates of Streptococcus pneumoniae, Haemophilus influenzae, Haemophilus parainfluenzae, Klebsiella pneumoniae, methicillin-susceptible Staphylococcus aureus, or Moraxella catarrhalis [see Clinical Studies (14.1)].
Because fluoroquinolones, including Moxifloxacin Injection, have been associated with serious adverse reactions [see Warnings and Precautions (5.1 to 5.14)] and for some patients ABECB is self-limiting, reserve Moxifloxacin Injection for treatment of ABECB in patients who have no alternative treatment options.
14.1 Acute Bacterial Exacerbation of Chronic Bronchitis
Moxifloxacin tablets (400 mg once daily for five days) were evaluated for the treatment of acute bacterial exacerbation of chronic bronchitis in a randomized, double-blind, controlled clinical trial conducted in the US. This study compared moxifloxacin with clarithromycin (500 mg twice daily for 10 days) and enrolled 629 patients. Clinical success was assessed at 7 to 17 days post-therapy. The clinical success for moxifloxacin was 89% (222/250) compared to 89% (224/251) for clarithromycin.
| Pathogen | Moxifloxacin | Clarithromycin |
| Streptococcus pneumoniae | 16/16 (100%) | 20/23 (87%) |
| Haemophilus influenzae | 33/37 (89%) | 36/41 (88%) |
| Haemophilus parainfluenzae | 16/16 (100%) | 14/14 (100%) |
| Moraxella catarrhalis | 29/34 (85%) | 24/24 (100%) |
| Staphylococcus aureus | 15/16 (94%) | 6/8 (75%) |
| Klebsiella pneumoniae | 18/20 (90%) | 10/11 (91%) |
The microbiological eradication rates (eradication plus presumed eradication) in moxifloxacin-treated patients were Streptococcus pneumoniae 100%, Haemophilus influenzae 89%, Haemophilus parainfluenzae 100%, Moraxella catarrhalis 85%, Staphylococcus aureus 94%, and Klebsiella pneumoniae 85%.
5.8 Other Serious and Sometimes Fatal Adverse Reactions
Other serious and sometimes fatal adverse reactions, some due to hypersensitivity, and some due to uncertain etiology, have been reported rarely in patients receiving therapy with quinolones, including moxifloxacin. These events may be severe and generally occur following the administration of multiple doses. Clinical manifestations may include one or more of the following:
- Fever, rash, or severe dermatologic reactions (for example, toxic epidermal necrolysis, Stevens-Johnson syndrome)
- Vasculitis; arthralgia; myalgia; serum sickness
- Allergic pneumonitis
- Interstitial nephritis; acute renal insufficiency or failure
- Hepatitis; jaundice; acute hepatic necrosis or failure
- Anemia, including hemolytic and aplastic; thrombocytopenia, including thrombotic thrombocytopenic purpura; leukopenia; agranulocytosis; pancytopenia; and/or other hematologic abnormalities
Discontinue Moxifloxacin Injection immediately at the first appearance of a skin rash, jaundice, or any other sign of hypersensitivity and supportive measures instituted [see Patient Counseling Information (17) and Adverse Reactions (6.2)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long term studies in animals to determine the carcinogenic potential of moxifloxacin have not been performed.
Moxifloxacin was not mutagenic in 4 bacterial strains (TA 98, TA 100, TA 1535, TA 1537) used in the Ames Salmonella reversion assay. As with other quinolones, the positive response observed with moxifloxacin in strain TA 102 using the same assay may be due to the inhibition of DNA gyrase. Moxifloxacin was not mutagenic in the CHO/HGPRT mammalian cell gene mutation assay. An equivocal result was obtained in the same assay when v79 cells were used. Moxifloxacin was clastogenic in the v79 chromosome aberration assay, but it did not induce unscheduled DNA synthesis in cultured rat hepatocytes. There was no evidence of genotoxicity in vivo in a micronucleus test or a dominant lethal test in mice.
Moxifloxacin had no effect on fertility in male and female rats at oral doses as high as 500 mg/kg/day, (approximately 12 times the maximum recommended human dose based on body surface area), or at intravenous doses as high as 45 mg/kg/day, (approximately equal to the maximum recommended human dose based on body surface area). At 500 mg/kg orally there were slight effects on sperm morphology (head-tail separation) in male rats and on the estrous cycle in female rats.
2.3 Preparation for Administration of Moxifloxacin Injection
To prepare Moxifloxacin Injection premix in flexible bags:
- Close flow control clamp of administration set.
- Remove cover from port at bottom of container.
- Insert piercing pin from an appropriate transfer set (for example, one that does not require excessive force, such as ISO compatible administration set) into port with a gentle twisting motion until pin is firmly seated.
NOTE: Refer to complete directions that have been provided with the administration set.
Because the premix flexible bags are for single-dose only, any unused portion should be discarded.
14.3 Community Acquired Pneumonia Caused By Multi Drug Resistant Streptococcus Pneumoniae (14.3 Community Acquired Pneumonia Caused by Multi-Drug Resistant Streptococcus pneumoniae)
Moxifloxacin was effective in the treatment of community acquired pneumonia (CAP) caused by multi-drug resistant MDRSP* isolates. Of 37 microbiologically evaluable patients with MDRSP isolates, 35 patients (95%) achieved clinical and bacteriological success post-therapy. The clinical and bacteriological success rates based on the number of patients treated are shown in Table 11.
* MDRSP, Multi-drug resistant Streptococcus pneumoniae includes isolates previously known as PRSP (Penicillin-resistant S. pneumoniae), and are isolates resistant to two or more of the following antibiotics: penicillin (MIC ≥ 2 mcg/mL), 2nd generation cephalosporins (for example, cefuroxime), macrolides, tetracyclines, and trimethoprim/sulfamethoxazole.
|
a n = number of patients successfully treated; N = number of patients with MDRSP (from a total of 37 patients) |
||||
|
b n = number of patients successfully treated (presumed eradication or eradication); N = number of patients with MDRSP (from a total of 37 patients) |
||||
|
c One patient had a respiratory isolate that was resistant to penicillin and cefuroxime but a blood isolate that was intermediate to penicillin and cefuroxime. The patient is included in the database based on the respiratory isolate. |
||||
|
d Azithromycin, clarithromycin, and erythromycin were the macrolide antimicrobials tested. |
||||
| Screening Susceptibility | Clinical Success | Bacteriological Success | ||
| n/Na | % | n/Nb | % | |
| Penicillin-resistant | 21/21 | 100%c | 21/21 | 100%c |
| 2nd generation cephalosporin-resistant | 25/26 | 96%c | 25/26 | 96%c |
| Macrolide-resistantd | 22/23 | 96% | 22/23 | 96% |
| Trimethoprim/sulfamethoxazole-resistant | 28/30 | 93% | 28/30 | 93% |
| Tetracycline-resistant | 17/18 | 94% | 17/18 | 94% |
Not all isolates were resistant to all antimicrobial classes tested. Success and eradication rates are summarized in Table 12.
|
a One patient had a respiratory isolate resistant to 5 antimicrobials and a blood isolate resistant to 3 antimicrobials. The patient was included in the category resistant to 5 antimicrobials. |
||
| S. pneumoniae with MDRSP | Clinical Success | Bacteriological Eradication Rate |
| Resistant to 2 antimicrobials | 12/13 (92.3%) | 12/13 (92.3%) |
| Resistant to 3 antimicrobials | 10/11 (90.9%)a | 10/11 (90.9%)a |
| Resistant to 4 antimicrobials | 6/6 (100%) | 6/6 (100%) |
| Resistant to 5 antimicrobials | 7/7 (100%)a | 7/7 (100%)a |
| Bacteremia with MDRSP | 9/9 (100%) | 9/9 (100%) |
Warning: Serious Adverse Reactions Including Tendinitis, Tendon Rupture, Peripheral Neuropathy, Central Nervous System Effects and Exacerbation of Myasthenia Gravis (WARNING: SERIOUS ADVERSE REACTIONS INCLUDING TENDINITIS, TENDON RUPTURE, PERIPHERAL NEUROPATHY, CENTRAL NERVOUS SYSTEM EFFECTS and EXACERBATION OF MYASTHENIA GRAVIS)
-
Fluoroquinolones, including moxifloxacin, have been associated with disabling and potentially irreversible serious adverse reactions that have occurred together
[see Warnings and Precautions (5.1)]
, including:
- Tendinitis and tendon rupture [see Warnings and Precautions (5.2)]
- Peripheral neuropathy [see Warnings and Precautions (5.3)]
- Central nervous system effects [see Warnings and Precautions (5.4)]
Discontinue Moxifloxacin Injection immediately and avoid the use of fluoroquinolones, including Moxifloxacin Injection, in patients who experience any of these serious adverse reactions [see Warnings and Precautions (5.1)].
- Fluoroquinolones, including moxifloxacin, may exacerbate muscle weakness in patients with myasthenia gravis. Avoid Moxifloxacin Injection in patients with known history of myasthenia gravis [see Warnings and Precautions (5.5)].
- Because fluoroquinolones, including moxifloxacin, have been associated with serious adverse reactions [see Warnings and Precautions (5.1 to 5.14)] , reserve Moxifloxacin Injection for use in patients who have no alternative treatment options for the following indications:
5.1 Disabling and Potentially Irreversible Serious Adverse Reactions Including Tendinitis and Tendon Rupture, Peripheral Neuropathy, and Central Nervous System Effects
Fluoroquinolones, including Moxifloxacin Injection, have been associated with disabling and potentially irreversible serious adverse reactions from different body systems that can occur together in the same patient. Commonly seen adverse reactions include tendinitis, tendon rupture, arthralgia, myalgia, peripheral neuropathy, and central nervous system effects (hallucinations, anxiety, depression, insomnia, severe headaches, and confusion).
These reactions can occur within hours to weeks after starting moxifloxacin. Patients of any age or without pre-existing risk factors have experienced these adverse reactions [see Warnings and Precautions (5.2, 5.3, 5.4)].
Discontinue Moxifloxacin Injection immediately at the first signs or symptoms of any serious adverse reaction. In addition, avoid the use of fluoroquinolones, including moxifloxacin, in patients who have experienced any of these serious adverse reactions associated with fluoroquinolones.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:40:59.530459 · Updated: 2026-03-14T22:04:41.081691