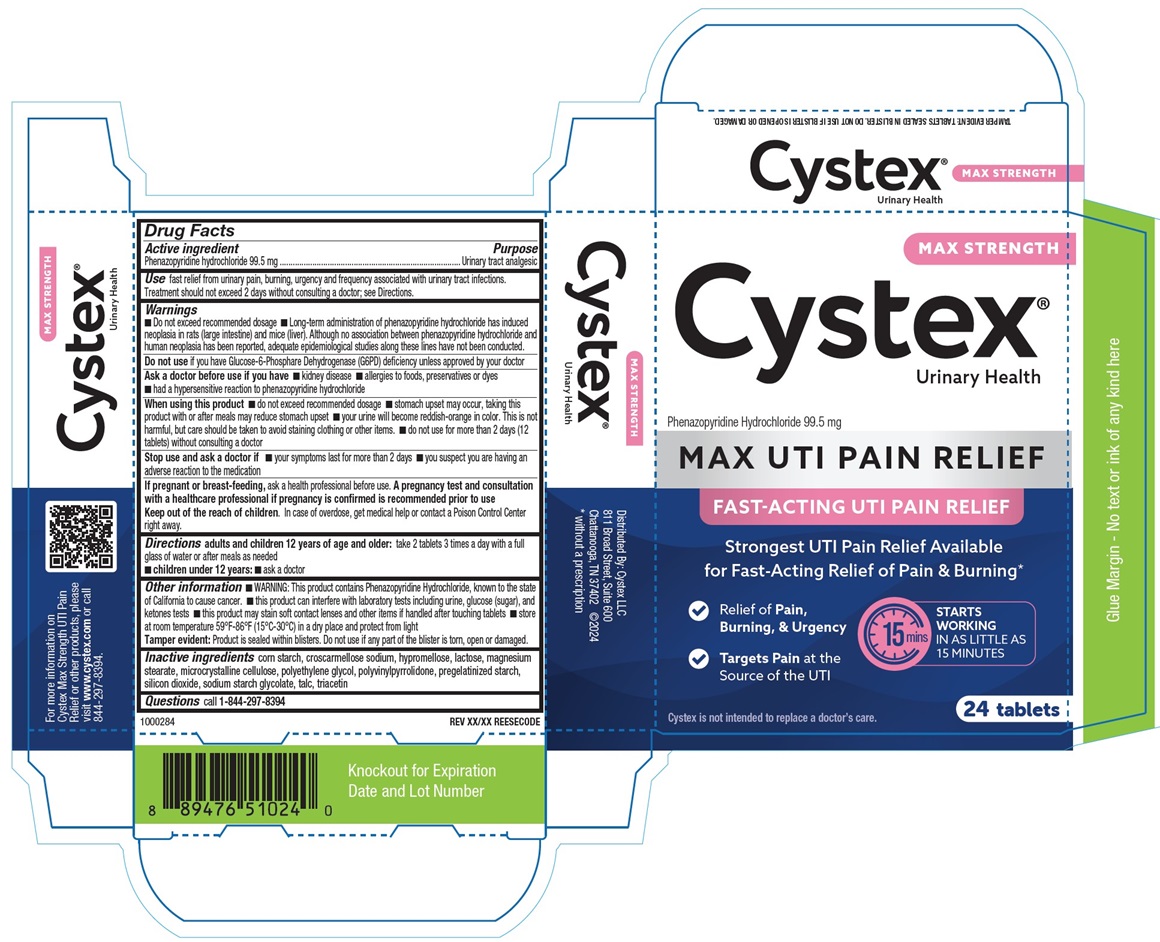
Drug Facts
a4d34c20-62af-4a3c-ad2c-046ba08d1ef6
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Phenazopyridine hydrochloride 99.5 mg
Purpose
Urinary tract analgesic
Medication Information
Indications and Usage
Use fast relief from urinary pain, burning, urgency and frequency associated with urinary tract infections. Treatment should not exceed 2 days without consulting a doctor; see Directions.
Purpose
Urinary tract analgesic
Description
Phenazopyridine hydrochloride 99.5 mg
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
A pregnancy test and consultation with a healthcare professional if pregnancy is confirmed is recommended prior to use
Keep out of the reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Warnings
- Do not exceed recommended dosage
- Long-term administration of phenazopyridine hydrochloride has induced neoplasia in rats (large intestine) and mice (liver). Although no association between phenazopyridine hydrochloride and human neoplasia has been reported, adequate epidemiological studies along these lines have not been conducted.
Questions
call 1-844-297-8394
Directions
Adults and children 12 years of age and older:take 2 tablets 3 times a day with a full glass of water or after meals as needed
- children under 12 years:
- ask a doctor
Do Not Use
if you have Glucose-6-Phosphare Dehydrogenase (G6PD) deficiency unless approved by your doctor
Active Ingredient
Phenazopyridine hydrochloride 99.5 mg
Other Information
- WARNING: This product contains Phenazopyridine Hydrochloride, known to the state of California to cause cancer.
- this product can interfere with laboratory tests including urine, glucose (sugar), and ketones tests
- this product may stain soft contact lenses and other items if handled after touching tablets
- store at room temperature 59°F-86°F (15°C-30°C) in a dry place and protect from light
Tamper evident:Product is sealed within blisters. Do not use if any part of the blister is torn, open or damaged.
Inactive Ingredients
corn starch, croscarmellose sodium, hypromellose, lactose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinylpyrrolidone, pregelatinized starch, silicon dioxide, sodium starch glycolate, talc, triacetin
Principal Display Panel
MAX STRENGTH
Cystex
®
Urinary Health
Phenazopyridine Hydrochloride 99.5 mg
MAX UTI PAIN RELIEF
FAST-ACTING UTI PAIN RELIEF
Strongest UTI Pain Relief Available
for Fast-Acting Relief of Pain & Burning
*
√Relief of
Pain, Burning, & Urgency
√
Targets Painat the Source of the UTI
STARTS
WORKING
IN AS LITTLE AS
15 MINUTES
Cystex is not intended to replace a doctor's care.
24 Tablets
TAMPER EVIDENT: TABLETS SEALED IN BLISTER. DO NOT USE IF BLISTER IS OPENED OR DAMAGED.
1000284
REV XX/XX REESECODE
For more information on
Cystex Max Strength UTI Pain
Relief or other products, please
visit
www.cystex.comor call
844-297-8394
Distributed by: Cystex LLC
811 Broad Street, Suite 600
Chattanooga, TN 37402 ©2024
*without a prescription
When Using This Product
- do not exceed recommended dosage
- stomach upset may occur, taking this product with or after meals may reduce stomach upset
- your urine will become reddish-orange in color. This is not harmful, but care should be taken to avoid staining clothing or other items.
- do not use for more than 2 days (12 tablets) without consulting a doctor
Stop Use and Ask A Doctor If
- your symptoms last for more than 2 days
- you suspect you are having an adverse reaction to the medication
Ask A Doctor Before Use If You Have
- kidney disease
- allergies to foods, preservatives or dyes
- had a hypersensitive reaction to phenazopyridine hydrochloride
Structured Label Content
Indications and Usage (34067-9)
Use fast relief from urinary pain, burning, urgency and frequency associated with urinary tract infections. Treatment should not exceed 2 days without consulting a doctor; see Directions.
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
A pregnancy test and consultation with a healthcare professional if pregnancy is confirmed is recommended prior to use
Keep out of the reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Purpose
Urinary tract analgesic
Warnings
- Do not exceed recommended dosage
- Long-term administration of phenazopyridine hydrochloride has induced neoplasia in rats (large intestine) and mice (liver). Although no association between phenazopyridine hydrochloride and human neoplasia has been reported, adequate epidemiological studies along these lines have not been conducted.
Questions
call 1-844-297-8394
Directions
Adults and children 12 years of age and older:take 2 tablets 3 times a day with a full glass of water or after meals as needed
- children under 12 years:
- ask a doctor
Do Not Use (Do not use)
if you have Glucose-6-Phosphare Dehydrogenase (G6PD) deficiency unless approved by your doctor
Active Ingredient (Active ingredient)
Phenazopyridine hydrochloride 99.5 mg
Other Information (Other information)
- WARNING: This product contains Phenazopyridine Hydrochloride, known to the state of California to cause cancer.
- this product can interfere with laboratory tests including urine, glucose (sugar), and ketones tests
- this product may stain soft contact lenses and other items if handled after touching tablets
- store at room temperature 59°F-86°F (15°C-30°C) in a dry place and protect from light
Tamper evident:Product is sealed within blisters. Do not use if any part of the blister is torn, open or damaged.
Inactive Ingredients (Inactive ingredients)
corn starch, croscarmellose sodium, hypromellose, lactose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinylpyrrolidone, pregelatinized starch, silicon dioxide, sodium starch glycolate, talc, triacetin
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
MAX STRENGTH
Cystex
®
Urinary Health
Phenazopyridine Hydrochloride 99.5 mg
MAX UTI PAIN RELIEF
FAST-ACTING UTI PAIN RELIEF
Strongest UTI Pain Relief Available
for Fast-Acting Relief of Pain & Burning
*
√Relief of
Pain, Burning, & Urgency
√
Targets Painat the Source of the UTI
STARTS
WORKING
IN AS LITTLE AS
15 MINUTES
Cystex is not intended to replace a doctor's care.
24 Tablets
TAMPER EVIDENT: TABLETS SEALED IN BLISTER. DO NOT USE IF BLISTER IS OPENED OR DAMAGED.
1000284
REV XX/XX REESECODE
For more information on
Cystex Max Strength UTI Pain
Relief or other products, please
visit
www.cystex.comor call
844-297-8394
Distributed by: Cystex LLC
811 Broad Street, Suite 600
Chattanooga, TN 37402 ©2024
*without a prescription
When Using This Product (When using this product)
- do not exceed recommended dosage
- stomach upset may occur, taking this product with or after meals may reduce stomach upset
- your urine will become reddish-orange in color. This is not harmful, but care should be taken to avoid staining clothing or other items.
- do not use for more than 2 days (12 tablets) without consulting a doctor
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- your symptoms last for more than 2 days
- you suspect you are having an adverse reaction to the medication
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- kidney disease
- allergies to foods, preservatives or dyes
- had a hypersensitive reaction to phenazopyridine hydrochloride
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:13.352907 · Updated: 2026-03-14T23:12:23.079612