Xermelo
a4ac9d62-887c-4c8f-b04d-a5f31cec7c86
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Xermelo is indicated for the treatment of carcinoid syndrome diarrhea in combination with somatostatin analog (SSA) therapy in adults inadequately controlled by SSA therapy.
Dosage and Administration
The recommended dosage of Xermelo in adult patients is 250 mg three times daily for patients whose diarrhea is inadequately controlled by SSA therapy. Administration Take Xermelo with food [see Clinical Pharmacology ( 12.3 ), Clinical Studies ( 14 )] . When short-acting octreotide is used in combination with Xermelo, administer short-acting octreotide at least 30 minutes after administering Xermelo [see Drug Interactions ( 7.3 )] . If a dose is missed, take the next dose at the regular time. Do not take 2 doses at the same time to make up for a missed dose. Discontinue Xermelo if severe constipation develops [see Warnings and Precautions ( 5.1 )].
Contraindications
Xermelo is contraindicated in patients with a history of a hypersensitivity reaction to telotristat. Reactions have included angioedema, rash and pruritis.
Warnings and Precautions
Constipation: Xermelo reduces bowel movement frequency; monitor patients for constipation, and/or severe persistent or worsening abdominal pain. Discontinue Xermelo if constipation or abdominal pain develops. ( 5.1 )
Adverse Reactions
Most common adverse reactions (≥5%) are nausea, headache, increased GGT, depression, flatulence, decreased appetite, peripheral edema, and pyrexia. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact TerSera Therapeutics LLC at 1-844-334-4035 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Drug Interactions
CYP3A4 Substrates (e.g., midazolam) and CYP2B6 Substrates (e.g., bupropion, efavirenz): Efficacy of concomitant drugs may be decreased; monitor patients’ response and consider increasing the dosage of the concomitant drug, if necessary. ( 7.1 )
Description
Contraindications ( 4 ) 9/2022 Warnings and Precautions, Constipation ( 5.1 ) 9/2022
Medication Information
Warnings and Precautions
Constipation: Xermelo reduces bowel movement frequency; monitor patients for constipation, and/or severe persistent or worsening abdominal pain. Discontinue Xermelo if constipation or abdominal pain develops. ( 5.1 )
Indications and Usage
Xermelo is indicated for the treatment of carcinoid syndrome diarrhea in combination with somatostatin analog (SSA) therapy in adults inadequately controlled by SSA therapy.
Dosage and Administration
The recommended dosage of Xermelo in adult patients is 250 mg three times daily for patients whose diarrhea is inadequately controlled by SSA therapy. Administration Take Xermelo with food [see Clinical Pharmacology ( 12.3 ), Clinical Studies ( 14 )] . When short-acting octreotide is used in combination with Xermelo, administer short-acting octreotide at least 30 minutes after administering Xermelo [see Drug Interactions ( 7.3 )] . If a dose is missed, take the next dose at the regular time. Do not take 2 doses at the same time to make up for a missed dose. Discontinue Xermelo if severe constipation develops [see Warnings and Precautions ( 5.1 )].
Contraindications
Xermelo is contraindicated in patients with a history of a hypersensitivity reaction to telotristat. Reactions have included angioedema, rash and pruritis.
Adverse Reactions
Most common adverse reactions (≥5%) are nausea, headache, increased GGT, depression, flatulence, decreased appetite, peripheral edema, and pyrexia. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact TerSera Therapeutics LLC at 1-844-334-4035 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Drug Interactions
CYP3A4 Substrates (e.g., midazolam) and CYP2B6 Substrates (e.g., bupropion, efavirenz): Efficacy of concomitant drugs may be decreased; monitor patients’ response and consider increasing the dosage of the concomitant drug, if necessary. ( 7.1 )
Description
Contraindications ( 4 ) 9/2022 Warnings and Precautions, Constipation ( 5.1 ) 9/2022
Section 42229-5
Less Common Adverse Reactions:
The following is a list of adverse reactions occurring in less than 5% of patients receiving Xermelo during the 12-week placebo-controlled period of the clinical trial:
Investigations: increased alkaline phosphatase, increased alanine aminotransferase, and increased aspartate aminotransferase.
Fecaloma was reported in one patient treated with Xermelo (at a higher than recommended dosage) during the 36-week open-label extension period following the 12-week double-blind period of the trial.
Section 44425-7
Storage
Store at 25°C (77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Section 51945-4
Principal Display Panel - 3x7 weekly pack
TO OPEN:
1 Press & hold button
2 Pull out card
WEEKLY PACK
21 TABLETS
Rx Only
XERMELO®
(telotristat ethyl) tablets
250 mg
per tablet
TerSera
therapeutics
For oral use only.
As with all medications, keep
out of reach of children.
NDC 70720-125-22
11 Description
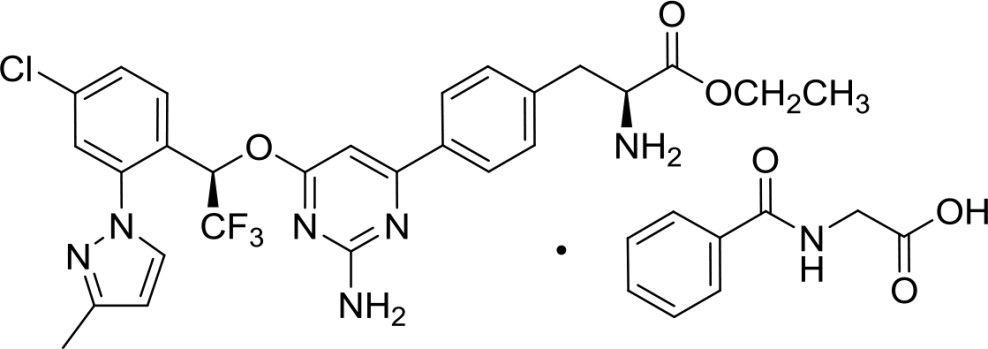
Xermelo (telotristat ethyl) tablets contain telotristat ethyl as telotristat etiprate, a tryptophan hydroxylase inhibitor. Telotristat etiprate is the hippurate salt of telotristat ethyl [(S)-ethyl 2-amino-3-(4-(2-amino-6-((R)-1-(4-chloro-2-(3-methyl-1H-pyrazol-1-yl)phenyl)-2,2,2-trifluoroethoxy)pyrimidin-4-yl)phenyl)propanoate], which undergoes hydrolysis to the active metabolite, (S)-2-amino-3-(4-(2-amino-6-((R)-1-(4-chloro-2-(3-methyl-1H-pyrazol-1-yl)phenyl)-2,2,2-trifluoroethoxy)pyrimidin-4-yl)phenyl)propanoic acid.
The molecular formula of telotristat etiprate is C27H26ClF3N6O3 • C9H9NO3 and its molecular weight is 754.2. The molecular weight of the free base (telotristat ethyl) is 575.0.
Chemical Structure:
Telotristat etiprate is a white to off-white solid. The solubility is a function of pH at 25°C; at pH 1 (0.1N HCl), the solubility is greater than 71 mg/mL, at pH 3 phosphate buffer, the solubility is 0.30 mg/mL, at a pH of 5 to 9, the solubility is negligible. In organic solvents, telotristat etiprate is freely soluble in methanol, soluble in acetone, and sparingly soluble in ethanol.
Each Xermelo tablet contains 250 mg of telotristat ethyl (free base) which is equivalent to 328 mg telotristat etiprate. The inactive ingredients of Xermelo tablets include: colloidal silicon dioxide, croscarmellose sodium, hydroxypropyl cellulose, lactose anhydrous, macrogol/PEG, magnesium stearate, polyvinyl alcohol [part hydrolyzed], talc and titanium dioxide.
5.1 Constipation
Xermelo reduces bowel movement frequency and may lead to constipation. Serious complications of constipation have been reported during clinical trials and postmarketing.
In a 12-week, placebo-controlled trial, in which patients had 4 or greater bowel movements per day, 2 out of 45 patients treated with a higher than recommended dosage of Xermelo reported constipation. In one patient the constipation was serious, resulting in hospitalization. During the 36-week extension period with higher than the recommended dosage of Xermelo, 10 of 115 patients reported constipation, with individual reports of intestinal perforation, obstruction, and fecaloma. In another 12-week, placebo-controlled trial in which patients had less than 4 bowel movements per day, 4 out of 25 patients treated with the recommended dosage of Xermelo reported constipation.
Serious complications of constipation in patients treated with Xermelo at the recommended dosage (e.g., intestinal obstruction) have also been reported in the postmarket setting. Most patients had additional risk factors, including underlying disease and concomitant constipating medications.
Given that patients with metastatic carcinoid tumors may have impaired integrity of the gastrointestinal tract wall, monitor for the development of constipation and/or severe, persistent, or worsening abdominal pain in patients taking Xermelo. Discontinue Xermelo if severe constipation or severe persistent or worsening abdominal pain develops [see Dosage and Administration (2), Adverse Reactions (6.1)].
8.4 Pediatric Use
The safety and effectiveness of Xermelo in pediatric patients have not been established.
8.5 Geriatric Use
Of 45 patients in a clinical trial of Xermelo, 19 (42%) patients were 65 years of age and over. No overall differences in safety or effectiveness were observed between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
14 Clinical Studies
A 12-week double-blind, placebo-controlled, randomized, multicenter trial of Xermelo was conducted in adult patients with a well-differentiated metastatic neuroendocrine tumor and carcinoid syndrome diarrhea who were having between 4 to 12 daily bowel movements despite the use of SSA therapy at a stable dose for at least 3 months. Patients were randomized to placebo or treatment with Xermelo 250 mg three times daily.
Study medication was administered within 15 minutes before or within 1 hour after a meal or snack [see Dosage and Administration (2)]. All patients were required to stay on their baseline SSA regimen and were allowed to use rescue medication (short-acting octreotide) and antidiarrheals (e.g., loperamide) for symptomatic relief. A total of 90 patients were evaluated for efficacy. The mean age of the population was 63 years of age (range 37 to 83 years), 50% were male, and 90% were White.
The primary efficacy endpoint was the change from baseline in the number of daily bowel movements averaged over the 12-week treatment period. The analysis results can be found in Table 2 below. The average was based on the number of days with valid, non-missing data. When a patient had more than 6 weeks of missing data, the change from baseline was considered equal to zero. A week of missing data was defined as a patient missing at least 4 days of diary data in that week.
| Parameter | Xermelo 250 mg three times daily | Placebo | |
|---|---|---|---|
|
CL=confidence limit; SD=standard deviation. |
|||
|
a Baseline Bowel Movements/Day was assessed over the 3-4 week screening/run-in period. |
|||
|
b Statistical tests used a blocked 2-sample Wilcoxon Rank Sum statistic (van Elteren test) stratified by the u5-HIAA stratification at randomization. CLs were based on the Hodges-Lehmann estimator of the median paired difference. |
|||
|
c p<0.001 |
|||
| Bowel Movements/Day At Baseline a | Number of Patients | 45 | 45 |
| Baseline Mean (SD) Median (Min, Max) |
6.1 (2.1) 5.5 (3.5, 13.0) |
5.2 (1.4) 5.1 (3.5, 9.0) |
|
| Change From Baseline In Bowel Movements/Day Averaged Over 12 Weeks | Change Averaged over 12 Weeks: Mean (SD) Median (Min, Max) |
˗1.4 (1.4) -1.3 (-6.1, 1.6) |
˗0.6 (0.8) -0.6 (-2.7, 0.8) |
| Estimate of Treatment Difference (97.5% CL)b | ˗0.8c
(˗1.3, ˗0.3) |
--- | |
In the 12-week study, a difference in average weekly reductions in bowel movement frequency between Xermelo and placebo was observed as early as 1 to 3 weeks, and persisted for the remaining 9 weeks of the study.
To aid in the interpretation of the bowel movement reduction results, the proportion of patients reporting any particular level of reduction in overall average bowel movement frequency is depicted in Figure 1 below. For example, 33% of patients randomized to Xermelo and 4% of patients randomized to placebo experienced a reduction in overall average bowel movements from baseline of at least 2 bowel movements per day.
Figure 1: Cumulative Proportion of Patients with Carcinoid Syndrome Diarrhea Reporting Change in Overall Average Bowel Movement Frequency
Other symptoms of carcinoid syndrome (abdominal pain or flushing) did not show improvement in the comparison of Xermelo to placebo.
The average number of daily short-acting octreotide injections used for rescue therapy over the 12-week double-blind treatment period was 0.3 and 0.7 in the Xermelo and placebo groups, respectively. In the subgroup of patients who received short-acting octreotide injections, observed reductions in the number of bowel movements per day and treatment differences were generally consistent with the reductions and differences observed in patients who did not receive rescue therapy, and were similar to the overall data presented in Table 2 above [see Dosage and Administration (2), Drug Interactions (7.2)].
A third randomized treatment arm of Xermelo 500 mg three times daily did not demonstrate additional treatment benefit on the primary endpoint and had a greater incidence of adverse reactions than Xermelo 250 mg three times daily. Therefore, Xermelo 500 mg three times daily is not recommended [see Dosage and Administration (2)].
4 Contraindications
Xermelo is contraindicated in patients with a history of a hypersensitivity reaction to telotristat. Reactions have included angioedema, rash and pruritis.
6 Adverse Reactions
Most common adverse reactions (≥5%) are nausea, headache, increased GGT, depression, flatulence, decreased appetite, peripheral edema, and pyrexia. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact TerSera Therapeutics LLC at 1-844-334-4035 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
7 Drug Interactions
CYP3A4 Substrates (e.g., midazolam) and CYP2B6 Substrates (e.g., bupropion, efavirenz): Efficacy of concomitant drugs may be decreased; monitor patients’ response and consider increasing the dosage of the concomitant drug, if necessary. (7.1)
8.6 Renal Impairment
No dosage adjustment of Xermelo is necessary in patients with mild, moderate or severe renal impairment who are not requiring dialysis.
There is no information on Xermelo in patients with end-stage renal disease who require dialysis (eGFR <15 mL/min/1.73 m2).
12.2 Pharmacodynamics
In healthy subjects, telotristat ethyl 500 mg three times daily (twice the recommended dosage) for 14 days decreased whole blood serotonin and 24-hour urinary 5-hydroxyindolacetic acid (u5-HIAA) from baseline. A decrease in 24-hour u5-HIAA was observed as early as after 5 days of treatment.
In patients with metastatic neuroendocrine tumors and carcinoid syndrome diarrhea, 24-hour u5-HIAA decreased from baseline following 6 and 12 weeks of treatment with Xermelo 250 mg three times a day, whereas placebo did not decrease u5-HIAA.
7.1 Cyp3a4 Substrates
Concomitant use of Xermelo may decrease the efficacy of drugs that are CYP3A4 substrates (e.g., midazolam) by decreasing their systemic exposure. Monitor patients’ response to CYP3A4 substrates when co-administered with Xermelo and consider increasing the dosage of the interacting drug, if necessary [see Clinical Pharmacology (12.3)].
7.2 Cyp2b6 Substrates
Concomitant use of Xermelo may decrease the efficacy of drugs that are CYP2B6 substrates (e.g., bupropion, efavirenz) by decreasing their systemic exposure. Monitor patients’ response to CYP2B6 substrates when co-administered with Xermelo and consider increasing the dosage of the interacting drug, if necessary [see Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
Systemic exposure of telotristat ethyl and its active metabolite, telotristat, were substantially increased in patients with moderate hepatic impairment (Child-Pugh Class B) and severe hepatic impairment (Child-Pugh C) (3.2- and 5.0-fold, respectively) compared to patients with normal hepatic function [see Clinical Pharmacology (12.3)]. Xermelo is not recommended in patients with moderate and severe hepatic impairment.
No dosage adjustment of Xermelo is necessary in patients with mild hepatic impairment (Child-Pugh A); however, additional monitoring of Xermelo-associated adverse reactions (e.g., constipation) is recommended in these patients [see Warnings and Precautions (5.1)].
1 Indications and Usage
Xermelo is indicated for the treatment of carcinoid syndrome diarrhea in combination with somatostatin analog (SSA) therapy in adults inadequately controlled by SSA therapy.
12.1 Mechanism of Action
Telotristat, the active metabolite of telotristat ethyl, is an inhibitor of tryptophan hydroxylase, which mediates the rate limiting step in serotonin biosynthesis. The in vitro inhibitory potency of telotristat towards tryptophan hydroxylase is 29 times higher than that of telotristat ethyl. Serotonin plays a role in mediating secretion, motility, inflammation, and sensation of the gastrointestinal tract, and is over-produced in patients with carcinoid syndrome. Through inhibition of tryptophan hydroxylase, telotristat and telotristat ethyl reduce the production of peripheral serotonin, and the frequency of carcinoid syndrome diarrhea.
5 Warnings and Precautions
Constipation: Xermelo reduces bowel movement frequency; monitor patients for constipation, and/or severe persistent or worsening abdominal pain. Discontinue Xermelo if constipation or abdominal pain develops. (5.1)
2 Dosage and Administration
The recommended dosage of Xermelo in adult patients is 250 mg three times daily for patients whose diarrhea is inadequately controlled by SSA therapy.
Administration
- Take Xermelo with food [see Clinical Pharmacology (12.3), Clinical Studies (14)].
- When short-acting octreotide is used in combination with Xermelo, administer short-acting octreotide at least 30 minutes after administering Xermelo [see Drug Interactions (7.3)].
- If a dose is missed, take the next dose at the regular time. Do not take 2 doses at the same time to make up for a missed dose.
- Discontinue Xermelo if severe constipation develops [see Warnings and Precautions (5.1)].
7.3 Short Acting Octreotide
Concurrent administration of short-acting octreotide with Xermelo significantly decreased the systemic exposure of telotristat ethyl and telotristat, the active metabolite. If treatment with short-acting octreotide is needed in combination with Xermelo, administer short-acting octreotide at least 30 minutes after administration of Xermelo [see Clinical Pharmacology (12.3)].
3 Dosage Forms and Strengths
Tablets: 250 mg telotristat ethyl; white to off-white, coated and oval with “T-E” debossed on one side and “250” debossed on the other side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of Xermelo. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal: intestinal obstruction [see Warnings and Precautions (5.1)]
Immune system disorders: angioedema
Skin and subcutaneous tissue disorders: pruritis, rash
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Xermelo was studied in a double-blind, placebo-controlled clinical trial of 90 patients with metastatic neuroendocrine tumors and carcinoid syndrome diarrhea. Patients reported between 4 to 12 bowel movements daily despite the use of SSA therapy at a stable dose for at least 3 months [see Clinical Studies (14)]. Placebo or Xermelo 250 mg was administered three times daily for 12 weeks. Concomitant anti-diarrheal medications (e.g., loperamide) were used by 43% (36% and 51% in the placebo and Xermelo group, respectively), pancreatic enzyme replacement medications by 39% (36% and 42% in the placebo and Xermelo group, respectively), and opioid analgesics by 29% (24% and 33% in the placebo and Xermelo group, respectively) of patients during the 12-week double-blind period of the trial.
Table 1 below lists adverse reactions occurring at an incidence of at least 5% in the Xermelo group (N=45) and at an incidence greater than placebo (N=45) during the 12-week placebo-controlled period of the trial.
|
a incidence of at least 5% in the Xermelo group and at an incidence greater than placebo |
||
|
b including depression, depressed mood and decreased interest |
||
| Adverse Reaction |
Xermelo
250 mg Three Times Daily, N=45 (%) |
Placebo,
N=45 (%) |
| Nausea | 13 | 11 |
| Headache | 11 | 4 |
| Increased gamma-glutamyl-transferase (GGT) | 9 | 0 |
| Depressionb | 9 | 7 |
| Peripheral edema | 7 | 2 |
| Flatulence | 7 | 2 |
| Decreased appetite | 7 | 4 |
| Pyrexia | 7 | 4 |
In another placebo-controlled clinical trial of patients with carcinoid syndrome diarrhea and less than 4 bowel movements per day, the following additional adverse reactions, not listed in Table 1, of abdominal pain (including upper and lower abdominal pain, abdominal distention and gastrointestinal pain) and constipation were reported in at least 5% of patients in the Xermelo treated group and at an incidence greater than placebo [see Warnings and Precautions (5.1)].
17 Patient Counseling Information
Advise patients:
- If they experience severe constipation or severe persistent or worsening abdominal pain, to discontinue Xermelo and contact their healthcare provider [see Warnings and Precautions (5.1)].
- To take Xermelo with food [see Clinical Pharmacology (12.3), Clinical Studies (14)].
- When short-acting octreotide is used in combination with Xermelo, administer short-acting octreotide at least 30 minutes after administering Xermelo [see Drug Interactions (7.3)].
- If a dose is missed, take the next dose at the regular time. Do not take 2 doses at the same time to make up for a missed dose.
Distributed by:
TerSera Therapeutics LLC, Deerfield, IL 60015
©2022 TerSera Therapeutics LLC
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 26-week study in transgenic (Tg.rasH2) mice, telotristat ethyl was not tumorigenic at oral doses up to 300 mg/kg/day (approximately 12 to 19 times the AUC for the active metabolite at the RHD).
In a 2-year carcinogenicity study in Sprague-Dawley rats, telotristat ethyl was not tumorigenic at oral doses up to 170 mg/kg/day (approximately 2 to 5 times the AUC for the active metabolite at the RHD).
Telotristat ethyl was negative in the in vitro Ames test, the in vitro chromosomal aberration test using Chinese hamster ovary cells, and the in vivo rat micronucleus test.
Telotristat ethyl at oral doses up to 500 mg/kg/day (approximately 5 times the AUC for the active metabolite at the RHD) was found to have no effect on fertility and reproductive performance of male or female rats.
Structured Label Content
Section 42229-5 (42229-5)
Less Common Adverse Reactions:
The following is a list of adverse reactions occurring in less than 5% of patients receiving Xermelo during the 12-week placebo-controlled period of the clinical trial:
Investigations: increased alkaline phosphatase, increased alanine aminotransferase, and increased aspartate aminotransferase.
Fecaloma was reported in one patient treated with Xermelo (at a higher than recommended dosage) during the 36-week open-label extension period following the 12-week double-blind period of the trial.
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Storage
Store at 25°C (77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Section 51945-4 (51945-4)
Principal Display Panel - 3x7 weekly pack
TO OPEN:
1 Press & hold button
2 Pull out card
WEEKLY PACK
21 TABLETS
Rx Only
XERMELO®
(telotristat ethyl) tablets
250 mg
per tablet
TerSera
therapeutics
For oral use only.
As with all medications, keep
out of reach of children.
NDC 70720-125-22
11 Description (11 DESCRIPTION)
Xermelo (telotristat ethyl) tablets contain telotristat ethyl as telotristat etiprate, a tryptophan hydroxylase inhibitor. Telotristat etiprate is the hippurate salt of telotristat ethyl [(S)-ethyl 2-amino-3-(4-(2-amino-6-((R)-1-(4-chloro-2-(3-methyl-1H-pyrazol-1-yl)phenyl)-2,2,2-trifluoroethoxy)pyrimidin-4-yl)phenyl)propanoate], which undergoes hydrolysis to the active metabolite, (S)-2-amino-3-(4-(2-amino-6-((R)-1-(4-chloro-2-(3-methyl-1H-pyrazol-1-yl)phenyl)-2,2,2-trifluoroethoxy)pyrimidin-4-yl)phenyl)propanoic acid.
The molecular formula of telotristat etiprate is C27H26ClF3N6O3 • C9H9NO3 and its molecular weight is 754.2. The molecular weight of the free base (telotristat ethyl) is 575.0.
Chemical Structure:
Telotristat etiprate is a white to off-white solid. The solubility is a function of pH at 25°C; at pH 1 (0.1N HCl), the solubility is greater than 71 mg/mL, at pH 3 phosphate buffer, the solubility is 0.30 mg/mL, at a pH of 5 to 9, the solubility is negligible. In organic solvents, telotristat etiprate is freely soluble in methanol, soluble in acetone, and sparingly soluble in ethanol.
Each Xermelo tablet contains 250 mg of telotristat ethyl (free base) which is equivalent to 328 mg telotristat etiprate. The inactive ingredients of Xermelo tablets include: colloidal silicon dioxide, croscarmellose sodium, hydroxypropyl cellulose, lactose anhydrous, macrogol/PEG, magnesium stearate, polyvinyl alcohol [part hydrolyzed], talc and titanium dioxide.
5.1 Constipation
Xermelo reduces bowel movement frequency and may lead to constipation. Serious complications of constipation have been reported during clinical trials and postmarketing.
In a 12-week, placebo-controlled trial, in which patients had 4 or greater bowel movements per day, 2 out of 45 patients treated with a higher than recommended dosage of Xermelo reported constipation. In one patient the constipation was serious, resulting in hospitalization. During the 36-week extension period with higher than the recommended dosage of Xermelo, 10 of 115 patients reported constipation, with individual reports of intestinal perforation, obstruction, and fecaloma. In another 12-week, placebo-controlled trial in which patients had less than 4 bowel movements per day, 4 out of 25 patients treated with the recommended dosage of Xermelo reported constipation.
Serious complications of constipation in patients treated with Xermelo at the recommended dosage (e.g., intestinal obstruction) have also been reported in the postmarket setting. Most patients had additional risk factors, including underlying disease and concomitant constipating medications.
Given that patients with metastatic carcinoid tumors may have impaired integrity of the gastrointestinal tract wall, monitor for the development of constipation and/or severe, persistent, or worsening abdominal pain in patients taking Xermelo. Discontinue Xermelo if severe constipation or severe persistent or worsening abdominal pain develops [see Dosage and Administration (2), Adverse Reactions (6.1)].
8.4 Pediatric Use
The safety and effectiveness of Xermelo in pediatric patients have not been established.
8.5 Geriatric Use
Of 45 patients in a clinical trial of Xermelo, 19 (42%) patients were 65 years of age and over. No overall differences in safety or effectiveness were observed between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
14 Clinical Studies (14 CLINICAL STUDIES)
A 12-week double-blind, placebo-controlled, randomized, multicenter trial of Xermelo was conducted in adult patients with a well-differentiated metastatic neuroendocrine tumor and carcinoid syndrome diarrhea who were having between 4 to 12 daily bowel movements despite the use of SSA therapy at a stable dose for at least 3 months. Patients were randomized to placebo or treatment with Xermelo 250 mg three times daily.
Study medication was administered within 15 minutes before or within 1 hour after a meal or snack [see Dosage and Administration (2)]. All patients were required to stay on their baseline SSA regimen and were allowed to use rescue medication (short-acting octreotide) and antidiarrheals (e.g., loperamide) for symptomatic relief. A total of 90 patients were evaluated for efficacy. The mean age of the population was 63 years of age (range 37 to 83 years), 50% were male, and 90% were White.
The primary efficacy endpoint was the change from baseline in the number of daily bowel movements averaged over the 12-week treatment period. The analysis results can be found in Table 2 below. The average was based on the number of days with valid, non-missing data. When a patient had more than 6 weeks of missing data, the change from baseline was considered equal to zero. A week of missing data was defined as a patient missing at least 4 days of diary data in that week.
| Parameter | Xermelo 250 mg three times daily | Placebo | |
|---|---|---|---|
|
CL=confidence limit; SD=standard deviation. |
|||
|
a Baseline Bowel Movements/Day was assessed over the 3-4 week screening/run-in period. |
|||
|
b Statistical tests used a blocked 2-sample Wilcoxon Rank Sum statistic (van Elteren test) stratified by the u5-HIAA stratification at randomization. CLs were based on the Hodges-Lehmann estimator of the median paired difference. |
|||
|
c p<0.001 |
|||
| Bowel Movements/Day At Baseline a | Number of Patients | 45 | 45 |
| Baseline Mean (SD) Median (Min, Max) |
6.1 (2.1) 5.5 (3.5, 13.0) |
5.2 (1.4) 5.1 (3.5, 9.0) |
|
| Change From Baseline In Bowel Movements/Day Averaged Over 12 Weeks | Change Averaged over 12 Weeks: Mean (SD) Median (Min, Max) |
˗1.4 (1.4) -1.3 (-6.1, 1.6) |
˗0.6 (0.8) -0.6 (-2.7, 0.8) |
| Estimate of Treatment Difference (97.5% CL)b | ˗0.8c
(˗1.3, ˗0.3) |
--- | |
In the 12-week study, a difference in average weekly reductions in bowel movement frequency between Xermelo and placebo was observed as early as 1 to 3 weeks, and persisted for the remaining 9 weeks of the study.
To aid in the interpretation of the bowel movement reduction results, the proportion of patients reporting any particular level of reduction in overall average bowel movement frequency is depicted in Figure 1 below. For example, 33% of patients randomized to Xermelo and 4% of patients randomized to placebo experienced a reduction in overall average bowel movements from baseline of at least 2 bowel movements per day.
Figure 1: Cumulative Proportion of Patients with Carcinoid Syndrome Diarrhea Reporting Change in Overall Average Bowel Movement Frequency
Other symptoms of carcinoid syndrome (abdominal pain or flushing) did not show improvement in the comparison of Xermelo to placebo.
The average number of daily short-acting octreotide injections used for rescue therapy over the 12-week double-blind treatment period was 0.3 and 0.7 in the Xermelo and placebo groups, respectively. In the subgroup of patients who received short-acting octreotide injections, observed reductions in the number of bowel movements per day and treatment differences were generally consistent with the reductions and differences observed in patients who did not receive rescue therapy, and were similar to the overall data presented in Table 2 above [see Dosage and Administration (2), Drug Interactions (7.2)].
A third randomized treatment arm of Xermelo 500 mg three times daily did not demonstrate additional treatment benefit on the primary endpoint and had a greater incidence of adverse reactions than Xermelo 250 mg three times daily. Therefore, Xermelo 500 mg three times daily is not recommended [see Dosage and Administration (2)].
4 Contraindications (4 CONTRAINDICATIONS)
Xermelo is contraindicated in patients with a history of a hypersensitivity reaction to telotristat. Reactions have included angioedema, rash and pruritis.
6 Adverse Reactions (6 ADVERSE REACTIONS)
Most common adverse reactions (≥5%) are nausea, headache, increased GGT, depression, flatulence, decreased appetite, peripheral edema, and pyrexia. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact TerSera Therapeutics LLC at 1-844-334-4035 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
7 Drug Interactions (7 DRUG INTERACTIONS)
CYP3A4 Substrates (e.g., midazolam) and CYP2B6 Substrates (e.g., bupropion, efavirenz): Efficacy of concomitant drugs may be decreased; monitor patients’ response and consider increasing the dosage of the concomitant drug, if necessary. (7.1)
8.6 Renal Impairment
No dosage adjustment of Xermelo is necessary in patients with mild, moderate or severe renal impairment who are not requiring dialysis.
There is no information on Xermelo in patients with end-stage renal disease who require dialysis (eGFR <15 mL/min/1.73 m2).
12.2 Pharmacodynamics
In healthy subjects, telotristat ethyl 500 mg three times daily (twice the recommended dosage) for 14 days decreased whole blood serotonin and 24-hour urinary 5-hydroxyindolacetic acid (u5-HIAA) from baseline. A decrease in 24-hour u5-HIAA was observed as early as after 5 days of treatment.
In patients with metastatic neuroendocrine tumors and carcinoid syndrome diarrhea, 24-hour u5-HIAA decreased from baseline following 6 and 12 weeks of treatment with Xermelo 250 mg three times a day, whereas placebo did not decrease u5-HIAA.
7.1 Cyp3a4 Substrates (7.1 CYP3A4 Substrates)
Concomitant use of Xermelo may decrease the efficacy of drugs that are CYP3A4 substrates (e.g., midazolam) by decreasing their systemic exposure. Monitor patients’ response to CYP3A4 substrates when co-administered with Xermelo and consider increasing the dosage of the interacting drug, if necessary [see Clinical Pharmacology (12.3)].
7.2 Cyp2b6 Substrates (7.2 CYP2B6 Substrates)
Concomitant use of Xermelo may decrease the efficacy of drugs that are CYP2B6 substrates (e.g., bupropion, efavirenz) by decreasing their systemic exposure. Monitor patients’ response to CYP2B6 substrates when co-administered with Xermelo and consider increasing the dosage of the interacting drug, if necessary [see Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
Systemic exposure of telotristat ethyl and its active metabolite, telotristat, were substantially increased in patients with moderate hepatic impairment (Child-Pugh Class B) and severe hepatic impairment (Child-Pugh C) (3.2- and 5.0-fold, respectively) compared to patients with normal hepatic function [see Clinical Pharmacology (12.3)]. Xermelo is not recommended in patients with moderate and severe hepatic impairment.
No dosage adjustment of Xermelo is necessary in patients with mild hepatic impairment (Child-Pugh A); however, additional monitoring of Xermelo-associated adverse reactions (e.g., constipation) is recommended in these patients [see Warnings and Precautions (5.1)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Xermelo is indicated for the treatment of carcinoid syndrome diarrhea in combination with somatostatin analog (SSA) therapy in adults inadequately controlled by SSA therapy.
12.1 Mechanism of Action
Telotristat, the active metabolite of telotristat ethyl, is an inhibitor of tryptophan hydroxylase, which mediates the rate limiting step in serotonin biosynthesis. The in vitro inhibitory potency of telotristat towards tryptophan hydroxylase is 29 times higher than that of telotristat ethyl. Serotonin plays a role in mediating secretion, motility, inflammation, and sensation of the gastrointestinal tract, and is over-produced in patients with carcinoid syndrome. Through inhibition of tryptophan hydroxylase, telotristat and telotristat ethyl reduce the production of peripheral serotonin, and the frequency of carcinoid syndrome diarrhea.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
Constipation: Xermelo reduces bowel movement frequency; monitor patients for constipation, and/or severe persistent or worsening abdominal pain. Discontinue Xermelo if constipation or abdominal pain develops. (5.1)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
The recommended dosage of Xermelo in adult patients is 250 mg three times daily for patients whose diarrhea is inadequately controlled by SSA therapy.
Administration
- Take Xermelo with food [see Clinical Pharmacology (12.3), Clinical Studies (14)].
- When short-acting octreotide is used in combination with Xermelo, administer short-acting octreotide at least 30 minutes after administering Xermelo [see Drug Interactions (7.3)].
- If a dose is missed, take the next dose at the regular time. Do not take 2 doses at the same time to make up for a missed dose.
- Discontinue Xermelo if severe constipation develops [see Warnings and Precautions (5.1)].
7.3 Short Acting Octreotide (7.3 Short-Acting Octreotide)
Concurrent administration of short-acting octreotide with Xermelo significantly decreased the systemic exposure of telotristat ethyl and telotristat, the active metabolite. If treatment with short-acting octreotide is needed in combination with Xermelo, administer short-acting octreotide at least 30 minutes after administration of Xermelo [see Clinical Pharmacology (12.3)].
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tablets: 250 mg telotristat ethyl; white to off-white, coated and oval with “T-E” debossed on one side and “250” debossed on the other side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of Xermelo. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal: intestinal obstruction [see Warnings and Precautions (5.1)]
Immune system disorders: angioedema
Skin and subcutaneous tissue disorders: pruritis, rash
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Xermelo was studied in a double-blind, placebo-controlled clinical trial of 90 patients with metastatic neuroendocrine tumors and carcinoid syndrome diarrhea. Patients reported between 4 to 12 bowel movements daily despite the use of SSA therapy at a stable dose for at least 3 months [see Clinical Studies (14)]. Placebo or Xermelo 250 mg was administered three times daily for 12 weeks. Concomitant anti-diarrheal medications (e.g., loperamide) were used by 43% (36% and 51% in the placebo and Xermelo group, respectively), pancreatic enzyme replacement medications by 39% (36% and 42% in the placebo and Xermelo group, respectively), and opioid analgesics by 29% (24% and 33% in the placebo and Xermelo group, respectively) of patients during the 12-week double-blind period of the trial.
Table 1 below lists adverse reactions occurring at an incidence of at least 5% in the Xermelo group (N=45) and at an incidence greater than placebo (N=45) during the 12-week placebo-controlled period of the trial.
|
a incidence of at least 5% in the Xermelo group and at an incidence greater than placebo |
||
|
b including depression, depressed mood and decreased interest |
||
| Adverse Reaction |
Xermelo
250 mg Three Times Daily, N=45 (%) |
Placebo,
N=45 (%) |
| Nausea | 13 | 11 |
| Headache | 11 | 4 |
| Increased gamma-glutamyl-transferase (GGT) | 9 | 0 |
| Depressionb | 9 | 7 |
| Peripheral edema | 7 | 2 |
| Flatulence | 7 | 2 |
| Decreased appetite | 7 | 4 |
| Pyrexia | 7 | 4 |
In another placebo-controlled clinical trial of patients with carcinoid syndrome diarrhea and less than 4 bowel movements per day, the following additional adverse reactions, not listed in Table 1, of abdominal pain (including upper and lower abdominal pain, abdominal distention and gastrointestinal pain) and constipation were reported in at least 5% of patients in the Xermelo treated group and at an incidence greater than placebo [see Warnings and Precautions (5.1)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise patients:
- If they experience severe constipation or severe persistent or worsening abdominal pain, to discontinue Xermelo and contact their healthcare provider [see Warnings and Precautions (5.1)].
- To take Xermelo with food [see Clinical Pharmacology (12.3), Clinical Studies (14)].
- When short-acting octreotide is used in combination with Xermelo, administer short-acting octreotide at least 30 minutes after administering Xermelo [see Drug Interactions (7.3)].
- If a dose is missed, take the next dose at the regular time. Do not take 2 doses at the same time to make up for a missed dose.
Distributed by:
TerSera Therapeutics LLC, Deerfield, IL 60015
©2022 TerSera Therapeutics LLC
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 26-week study in transgenic (Tg.rasH2) mice, telotristat ethyl was not tumorigenic at oral doses up to 300 mg/kg/day (approximately 12 to 19 times the AUC for the active metabolite at the RHD).
In a 2-year carcinogenicity study in Sprague-Dawley rats, telotristat ethyl was not tumorigenic at oral doses up to 170 mg/kg/day (approximately 2 to 5 times the AUC for the active metabolite at the RHD).
Telotristat ethyl was negative in the in vitro Ames test, the in vitro chromosomal aberration test using Chinese hamster ovary cells, and the in vivo rat micronucleus test.
Telotristat ethyl at oral doses up to 500 mg/kg/day (approximately 5 times the AUC for the active metabolite at the RHD) was found to have no effect on fertility and reproductive performance of male or female rats.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:37:16.738804 · Updated: 2026-03-14T21:49:36.542485