Sulindac Tablets Usp
a47e4259-d44b-4b7b-b517-841cb6840982
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Cardiovascular Thrombotic Events Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use [see Warnings and Precautions ]. Sulindac tablets are contraindicated in the setting of coronary artery bypass graft (CABG) surgery [see Contraindications and Warnings]. Gastrointestinal Risk NSAIDs cause an increased risk of serious gastrointestinal adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients are at greater risk for serious gastrointestinal events. (see WARNINGS .)
Indications and Usage
Carefully consider the potential benefits and risks of sulindac tablets and other treatment options before deciding to use sulindac tablets. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS ). Sulindac tablets are indicated for acute or long-term use in the relief of signs and symptoms of the following: Osteoarthritis Rheumatoid arthritis* Ankylosing spondylitis Acute painful shoulder (Acute subacromial bursitis/supraspinatus tendinitis) Acute gouty arthritis
Dosage and Administration
Carefully consider the potential benefits and risks of sulindac tablets and other treatment options before deciding to use sulindac tablets. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS ). After observing the response to initial therapy with sulindac tablets, the dose and frequency should be adjusted to suit an individual patient's needs. Sulindac tablets should be administered orally twice a day with food. The maximum dosage is 400 mg per day. Dosages above 400 mg per day are not recommended. In osteoarthritis, rheumatoid arthritis, and ankylosing spondylitis, the recommended starting dosage is 150 mg twice a day. The dosage may be lowered or raised depending on the response. A prompt response (within one week) can be expected in about one-half of patients with osteoarthritis, ankylosing spondylitis, and rheumatoid arthritis. Others may require longer to respond. In acute painful shoulder (acute subacromial bursitis/supraspinatus tendinitis) and acute gouty arthritis, the recommended dosage is 200 mg twice a day. After a satisfactory response has been achieved, the dosage may be reduced according to the response. In acute painful shoulder, therapy for 7–14 days is usually adequate. In acute gouty arthritis, therapy for 7 days is usually adequate. **Incidence between 3% and 9%. Those reactions occurring in 1% to 3% of patients are not marked with an asterisk.
Contraindications
Sulindac tablets are contraindicated in patients with known hypersensitivity to sulindac or the excipients (see DESCRIPTION ). Sulindac tablets should not be given to patients who have experienced asthma, urticaria, or allergic-type reactions after taking aspirin or other NSAIDs. Severe, rarely fatal, anaphylactic/anaphylactoid reactions to NSAIDs have been reported in such patients (see WARNINGS – Anaphylactic/Anaphylactoid Reactions , and PRECAUTIONS – Preexisting Asthma ). In the setting of coronary artery bypass graft (CABG) surgery [see WARNINGS ].
Adverse Reactions
The following adverse reactions were reported in clinical trials or have been reported since the drug was marketed. The probability exists of a causal relationship between sulindac tablets and these adverse reactions. The adverse reactions which have been observed in clinical trials encompass observations in 1,865 patients, including 232 observed for at least 48 weeks.
How Supplied
Sulindac tablets USP are supplied as follows: Sulindac tablets, 150 mg, yellow, round, unscored, debossed MP 112 Bottles of 50 NDC 53489-478-02 Bottles of 60 NDC 53489-478-06 Bottles of 100 NDC 53489-478-01 Bottles of 250 NDC 53489-478-03 Bottles of 500 NDC 53489-478-05 Bottles of 1000 NDC 53489-478-10 Sulindac tablets, 200 mg, yellow, round, scored, debossed MP 116 Bottles of 50 NDC 53489-479-02 Bottles of 60 NDC 53489-479-06 Bottles of 100 NDC 53489-479-01 Bottles of 250 NDC 53489-479-03 Bottles of 500 NDC 53489-479-05 Bottles of 1000 NDC 53489-479-10
Medication Information
Recent Major Changes
Cardiovascular Thrombotic Events
- Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use [see Warnings and Precautions ].
- Sulindac tablets are contraindicated in the setting of coronary artery bypass graft (CABG) surgery [see Contraindications and Warnings].
Gastrointestinal Risk
- NSAIDs cause an increased risk of serious gastrointestinal adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients are at greater risk for serious gastrointestinal events. (see WARNINGS .)
Indications and Usage
Carefully consider the potential benefits and risks of sulindac tablets and other treatment options before deciding to use sulindac tablets. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS ).
Sulindac tablets are indicated for acute or long-term use in the relief of signs and symptoms of the following:
- Osteoarthritis
- Rheumatoid arthritis*
- Ankylosing spondylitis
- Acute painful shoulder (Acute subacromial bursitis/supraspinatus tendinitis)
- Acute gouty arthritis
Dosage and Administration
Carefully consider the potential benefits and risks of sulindac tablets and other treatment options before deciding to use sulindac tablets. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS ).
After observing the response to initial therapy with sulindac tablets, the dose and frequency should be adjusted to suit an individual patient's needs.
Sulindac tablets should be administered orally twice a day with food. The maximum dosage is 400 mg per day. Dosages above 400 mg per day are not recommended.
In osteoarthritis, rheumatoid arthritis, and ankylosing spondylitis, the recommended starting dosage is 150 mg twice a day. The dosage may be lowered or raised depending on the response.
A prompt response (within one week) can be expected in about one-half of patients with osteoarthritis, ankylosing spondylitis, and rheumatoid arthritis. Others may require longer to respond.
In acute painful shoulder (acute subacromial bursitis/supraspinatus tendinitis) and acute gouty arthritis, the recommended dosage is 200 mg twice a day. After a satisfactory response has been achieved, the dosage may be reduced according to the response. In acute painful shoulder, therapy for 7–14 days is usually adequate. In acute gouty arthritis, therapy for 7 days is usually adequate.
**Incidence between 3% and 9%. Those reactions occurring in 1% to 3% of patients are not marked with an asterisk.
Contraindications
Sulindac tablets are contraindicated in patients with known hypersensitivity to sulindac or the excipients (see DESCRIPTION ).
Sulindac tablets should not be given to patients who have experienced asthma, urticaria, or allergic-type reactions after taking aspirin or other NSAIDs. Severe, rarely fatal, anaphylactic/anaphylactoid reactions to NSAIDs have been reported in such patients (see WARNINGS – Anaphylactic/Anaphylactoid Reactions , and PRECAUTIONS – Preexisting Asthma ).
- In the setting of coronary artery bypass graft (CABG) surgery [see WARNINGS ].
Adverse Reactions
The following adverse reactions were reported in clinical trials or have been reported since the drug was marketed. The probability exists of a causal relationship between sulindac tablets and these adverse reactions. The adverse reactions which have been observed in clinical trials encompass observations in 1,865 patients, including 232 observed for at least 48 weeks.
How Supplied
Sulindac tablets USP are supplied as follows:
Sulindac tablets, 150 mg, yellow, round, unscored, debossed MP 112
|
Bottles of 50 |
NDC 53489-478-02 |
|
Bottles of 60 |
NDC 53489-478-06 |
|
Bottles of 100 |
NDC 53489-478-01 |
|
Bottles of 250 |
NDC 53489-478-03 |
|
Bottles of 500 |
NDC 53489-478-05 |
|
Bottles of 1000 |
NDC 53489-478-10 |
Sulindac tablets, 200 mg, yellow, round, scored, debossed MP 116
|
Bottles of 50 |
NDC 53489-479-02 |
|
Bottles of 60 |
NDC 53489-479-06 |
|
Bottles of 100 |
NDC 53489-479-01 |
|
Bottles of 250 |
NDC 53489-479-03 |
|
Bottles of 500 |
NDC 53489-479-05 |
|
Bottles of 1000 |
NDC 53489-479-10 |
Description
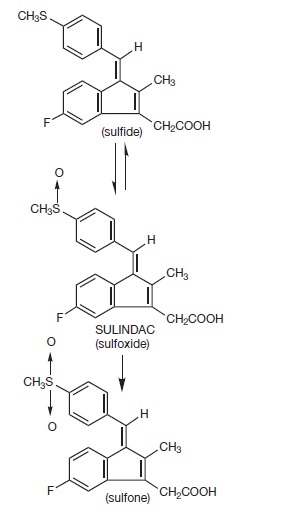
Sulindac is a non-steroidal, anti-inflammatory indene derivative designated chemically as (Z)-5-fluoro-2-methyl-1-[[ p-(methylsulfinyl)phenyl]methylene]-1 H-indene-3-acetic acid. It is not a salicylate, pyrazolone or propionic acid derivative. Its empirical formula is C 20H 17FO 3S, with a molecular weight of 356.42. Sulindac, a yellow crystalline compound, is a weak organic acid practically insoluble in water below pH 4.5, but very soluble as the sodium salt or in buffers of pH 6 or higher.
Sulindac tablets are available in 150 and 200 mg tablets for oral administration. Each tablet contains the following inactive ingredients: magnesium stearate, microcrystalline cellulose, and pregelatinized starch.
Following absorption, sulindac undergoes two major biotransformations — reversible reduction to the sulfide metabolite, and irreversible oxidation to the sulfone metabolite. Available evidence indicates that the biological activity resides with the sulfide metabolite.
The structural formulas of sulindac and its metabolites are:
Section 34072-9
General
Sulindac tablets cannot be expected to substitute for corticosteroids or to treat corticosteroid insufficiency. Abrupt discontinuation of corticosteroids may lead to disease exacerbation. Patients on prolonged corticosteroid therapy should have their therapy tapered slowly if a decision is made to discontinue corticosteroids.
The pharmacological activity of sulindac tablets in reducing fever and inflammation may diminish the utility of these diagnostic signs in detecting complications of presumed noninfectious, painful conditions.
Section 34073-7
Drug Interactions
Section 34075-2
Laboratory Tests
Because serious GI tract ulcerations and bleeding can occur without warning symptoms, physicians should monitor for signs or symptoms of GI bleeding. Patients on long-term treatment with NSAIDs should have their CBC and a chemistry profile checked periodically. If clinical signs and symptoms consistent with liver or renal disease develop, systemic manifestations occur (e.g., eosinophilia, rash, etc.) or if abnormal liver tests persist or worsen, sulindac tablets should be discontinued.
Section 34076-0
Information for Patients
Patients should be informed of the following information before initiating therapy with an NSAID and periodically during the course of ongoing therapy. Patients should also be encouraged to read the NSAID Medication Guide that accompanies each prescription dispensed.
- Cardiovascular Thrombotic Events
- Advise patients to be alert for the symptoms of cardiovascular thrombotic events, including chest pain, shortness of breath, weakness, or slurring of speech, and to report any of these symptoms to their healthcare provider immediately [see Warnings ].
- Sulindac tablets, like other NSAIDs, can cause GI discomfort and, rarely, serious GI side effects, such as ulcers and bleeding, which may result in hospitalization and even death. Although serious GI tract ulcerations and bleeding can occur without warning symptoms, patients should be alert for the signs and symptoms of ulcerations and bleeding, and should ask for medical advice when observing any indicative sign or symptoms including epigastric pain, dyspepsia, melena, and hematemesis. Patients should be apprised of the importance of this follow-up (see WARNINGS, Gastrointestinal Effects - Risk of Ulceration, Bleeding, and Perforation ).
- Serious Skin Reactions, including DRESS
- Advise patients to stop taking sulindac tablets immediately if they develop any type of rash or fever and to contact their healthcare provider as soon as possible (see WARNINGS).
- Heart Failure and Edema
- Advise patients to be alert for the symptoms of congestive heart failure including shortness of
- breath, unexplained weight gain, or edema and to contact their healthcare provider if such
- symptoms occur [see WARNINGS ].
- Patients should be informed of the warning signs and symptoms of hepatotoxicity (e.g., nausea, fatigue, lethargy, pruritus, jaundice, right upper quadrant tenderness, and "flu-like" symptoms). If these occur, patients should be instructed to stop therapy and seek immediate medical therapy.
- Patients should be informed of the signs of an anaphylactic/anaphylactoid reaction (e.g., difficulty breathing, swelling of the face or throat). If these occur, patients should be instructed to seek immediate emergency help (see WARNINGS ).
- Fetal Toxicity
- Inform pregnant women to avoid use of sulindac tablets and other NSAIDs starting at 30 weeks gestation because of the risk of the premature closing of the fetal ductus arteriosus. If treatment with sulindac tablets is needed for a pregnant woman between about 20 to 30 weeks gestation, advise her that she may need to be monitored for oligohydramnios, if treatment continues for longer than 48 hours (see WARNINGS; Fetal Toxicity, PRECAUTIONS; Pregnancy ).
Section 34077-8
Risk Summary
Use of NSAIDs, including sulindac tablets, can cause premature closure of the fetal ductus arteriosus and fetal renal dysfunction leading to oligohydramnios and, in some cases, neonatal renal impairment. Because of these risks, limit dose and duration of sulindac tablets use between about 20 and 30 weeks of gestation, and avoid sulindac tablets use at about 30 weeks of gestation and later in pregnancy (see WARNINGS; Fetal Toxicity ).
Premature Closure of Fetal Ductus Arteriosus
Use of NSAIDs, including sulindac tablets, at about 30 weeks gestation or later in pregnancy increases
the risk of premature closure of the fetal ductus arteriosus.
Oligohydramnios/Neonatal Renal Impairment
Use of NSAIDs at about 20 weeks gestation or later in pregnancy has been associated with cases of fetal
renal dysfunction leading to oligohydramnios, and in some cases, neonatal renal impairment.
Data from observational studies regarding other potential embryofetal risks of NSAID use in women in the first or second trimesters of pregnancy are inconclusive. In animal reproduction studies conducted in rats and rabbits have not demonstrated evidence of developmental abnormalities. In reproduction studies in the rat, a decrease in average fetal weight and an increase in numbers of dead pups were observed on the first day of the postpartum period at dosage levels of 20 and 40 mg/kg/day (2½ and 5 times the usual maximum daily dose in humans), although there was no adverse effect on the survival and growth during the remainder of the postpartum period. Sulindac prolongs the duration of gestation in rats, as do other compounds of this class. Visceral and skeletal malformations observed in low incidence among rabbits in some teratology studies did not occur at the same dosage levels in repeat studies, nor at a higher dosage level in the same species. However, animal reproduction studies are not always predictive of human response. Based on animal data, prostaglandins have been shown to have an important role in endometrial vascular permeability, blastocyst implantation, and decidualization. In animal studies, administration of prostaglandin synthesis inhibitors such as sulindac, resulted in increased pre- and post-implantation loss. Prostaglandins also have been shown to have an important role in fetal kidney development. In published animal studies, prostaglandin synthesis inhibitors have been reported to impair kidney development when administered at clinically relevant doses.
The estimated background risk of major birth defects and miscarriage for the indicated population(s) is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Section 34078-6
Clinical Considerations
Fetal/Neonatal Adverse Reactions
Premature Closure of Fetal Ductus Arteriosus:
Avoid use of NSAIDs in women at about 30 weeks gestation and later in pregnancy, because NSAIDs, including sulindac tablets, can cause premature closure of the fetal ductus arteriosus (see WARNINGS; Fetal Toxicity).
Oligohydramnios/Neonatal Renal Impairment
If an NSAID is necessary at about 20 weeks gestation or later in pregnancy, limit the use to the lowest effective dose and shortest duration possible. If sulindac tablets treatment extends beyond 48 hours, consider monitoring with ultrasound for oligohydramnios. If oligohydramnios occurs, discontinue sulindac tablets and follow up according to clinical practice (see WARNINGS; Fetal Toxicity).
Data
- Human Data
There are no adequate and well controlled studies in pregnant women. Sulindac should be used in pregnancy only if the potential benefit justifies the potential risk to the fetus. The known effects of drugs of this class on the human fetus during the third trimester of pregnancy include: constriction of the ductus arteriosus prenatally, tricuspid incompetence, and pulmonary hypertension; non-closure of the ductus arteriosus postnatally which may be resistant to medical management; myocardial degenerative changes, platelet dysfunction with resultant bleeding, intracranial bleeding, renal dysfunction or failure, renal injury/dysgenesis which may result in prolonged or permanent renal failure, oligohydramnios, gastrointestinal bleeding or perforation, and increased risk of necrotizing enterocolitis.
- Premature Closure of Fetal Ductus Arteriosus:
- Published literature reports that the use of NSAIDs at about 30 weeks of gestation and later in pregnancy may cause premature closure of the fetal ductus arteriosus.
- Oligohydramnios/Neonatal Renal Impairment:
- Published studies and postmarketing reports describe maternal NSAID use at about 20 weeks gestation or later in pregnancy associated with fetal renal dysfunction leading to oligohydramnios, and in some cases, neonatal renal impairment. These adverse outcomes are seen, on average, after days to weeks of treatment, although oligohydramnios has been infrequently reported as soon as 48 hours after NSAID initiation. In many cases, but not all, the decrease in amniotic fluid was transient and reversible with cessation of the drug. There have been a limited number of case reports of maternal NSAID use and neonatal renal dysfunction without oligohydramnios, some of which were irreversible. Some cases of neonatal renal dysfunction required treatment with invasive procedures, such as exchange transfusion or dialysis.
- Methodological limitations of these postmarketing studies and reports include lack of a control group; limited information regarding dose, duration, and timing of drug exposure; and concomitant use of other medications. These limitations preclude establishing a reliable estimate of the risk of adverse fetal and neonatal outcomes with maternal NSAID use. Because the published safety data on neonatal outcomes involved mostly preterm infants, the generalizability of certain reported risks to the full-term infant exposed to NSAIDs through maternal use is uncertain.
Section 34079-4
Labor and Delivery
In rat studies with NSAIDs, as with other drugs known to inhibit prostaglandin synthesis, an increased incidence of dystocia, delayed parturition, and decreased pup survival occurred. The effects of sulindac tablets on labor and delivery in pregnant women are unknown.
Section 34080-2
Nursing Mothers
It is not known whether this drug is excreted in human milk; however, it is secreted in the milk of lactating rats. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from sulindac tablets, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Section 34081-0
Pediatric
The pharmacokinetics of sulindac have not been investigated in pediatric patients.
Section 34082-8
Geriatric Use
As with any NSAID, caution should be exercised in treating the elderly (65 years and older) since advancing age appears to increase the possibility of adverse reactions. Elderly patients seem to tolerate ulceration or bleeding less well than other individuals and many spontaneous reports of fatal GI events are in this population (see WARNINGS, Gastrointestinal Effects - Risk of Ulceration, Bleeding, and Perforation ).
Sulindac tablets are known to be substantially excreted by the kidney and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection and it may be useful to monitor renal function (see WARNINGS, Renal Effects ).
Section 42228-7
Fetal Toxicity
Premature Closure of Fetal Ductus Arteriosus:
Avoid use of NSAIDs, including sulindac tablets, in pregnant women at about 30 weeks gestation and later. NSAIDs including sulindac tablets, increase the risk of premature closure of the fetal ductus arteriosus at approximately this gestational age.
Oligohydramnios/Neonatal Renal Impairment:
Use of NSAIDs, including sulindac tablets, at about 20 weeks gestation or later in pregnancy may cause fetal renal dysfunction leading to oligohydramnios and, in some cases, neonatal renal impairment. These adverse outcomes are seen, on average, after days to weeks of treatment, although oligohydramnios has been infrequently reported as soon as 48 hours after NSAID initiation.
Oligohydramnios is often, but not always, reversible with treatment discontinuation. Complications of prolonged oligohydramnios may, for example, include limb contractures and delayed lung maturation. In some postmarketing cases of impaired neonatal renal function, invasive procedures such as exchange transfusion or dialysis were required.
If NSAID treatment is necessary between about 20 weeks and 30 weeks gestation, limit sulindac tablets use to the lowest effective dose and shortest duration possible. Consider ultrasound monitoring of amniotic fluid if sulindac tablets treatment extends beyond 48 hours. Discontinue sulindac tablets if oligohydramnios occurs and follow up according to clinical practice (see PRECAUTIONS; Pregnancy).
Section 42229-5
Absorption
The extent of sulindac absorption from sulindac tablets is similar as compared to sulindac solution.
There is no information regarding food effect on sulindac absorption. Antacids containing magnesium hydroxide 200 mg and aluminum hydroxide 225 mg per 5 mL have been shown not to significantly decrease the extent of sulindac absorption.
|
PHARMACOKINETIC
PARAMETERS |
NORMAL | ELDERLY |
|---|---|---|
|
T max |
Age 19–41 (n=24) |
Age 65–87 (n=12) 400 mg qd |
|
|
|
|
|
|
(200 mg tablet) |
2.54 ± 1.52 S |
|
|
|
5.75 ± 2.81 SF |
|
|
3.38 ± 2.30 S |
6.83 ± 4.19 SP |
|
|
4.88 ± 2.57 SP |
|
|
|
4.96 ± 2.36 SF |
|
|
|
|
|
|
|
(150 mg tablet) |
|
|
|
|
|
|
|
3.90 ± 2.30 S |
|
|
|
5.85 ± 4.49 SP |
|
|
|
6.15 ± 3.07 SF |
|
|
Renal Clearance |
(200 mg tablet) |
|
|
|
|
|
|
|
68.12 ± 27.56 mL/min S |
|
|
|
36.58 ± 12.61 mL/min SP |
|
|
|
|
|
|
|
(150 mg tablet) |
|
|
|
|
|
|
|
74.39 ± 34.15 mL/min S |
|
|
|
41.75 ± 13.72 mL/min SP |
|
|
Mean effective Half life (h) |
7.8 S
|
|
|
S = Sulindac
|
Section 42231-1
Medication Guide
for
Nonsteroidal Anti-inflammatory Drugs (NSAIDs)
What is the most important information I should know about medicines called Nonsteroidal Anti-inflammatory Drugs (NSAIDs)?
NSAIDs can cause serious side effects, including:
- Increased risk of a heart attack or stroke that can lead to death.This risk may happen early in treatment and may increase:
- with increasing doses of NSAIDs
- with longer use of NSAIDs
- Do not take NSAIDs right before or after a heart surgery called a "coronary artery bypass graft (CABG)."
- Avoid taking NSAIDs after a recent heart attack, unless your healthcare provider tells you to. You may have an increased risk of another heart attack if you take NSAIDs after a recent heart attack.
- Increased risk of bleeding, ulcers, and tears (perforation) of the esophagus (tube leading from the mouth to the stomach), stomach and intestines:
- anytime during use
- without warning symptoms
- that may cause death
The risk of getting an ulcer or bleeding increases with:
- past history of stomach ulcers, or stomach or intestinal bleeding with use of NSAIDs
- taking medicines called "corticosteroids", "anti-coagulants", "SSRIs", or "SNRIs"
- increasing doses of NSAIDs
- longer use of NSAIDs
- smoking
- drinking alcohol
- older age
- poor health
- advanced liver disease
- bleeding problems
- NSAIDs should only be used:
- exactly as prescribed
- at the lowest dose possible for your treatment
- for the shortest time needed
What are NSAIDs?
NSAIDs are used to treat pain and redness, swelling, and heat (inflammation) from medical conditions such as different types of arthritis, menstrual cramps, and other types of short-term pain.
Who should not take NSAIDs?
Do not take NSAIDs:
- if you have had an asthma attack, hives, or other allergic reaction with aspirin or any other NSAIDs.
- right before or after heart bypass surgery.
Before taking NSAIDs, tell your healthcare provider about all of your medical conditions, including if you:
- have liver or kidney problems
- have high blood pressure
- have asthma
- are pregnant or plan to become pregnant. Taking NSAIDs at about 20 weeks of pregnancy or later may harm your unborn baby. If you need to take NSAIDs for more than 2 days when you are between 20 and 30 weeks of pregnancy, your healthcare provider may need to monitor the amount of fluid in your womb around your baby. You should not take NSAIDs after about 30 weeks of pregnancy.
- are breastfeeding or plan to breast feed.
Tell your healthcare provider about all of the medicines you take, including prescription or over-the-counter medicines, vitamins or herbal supplements.NSAIDs and some other medicines can interact with each other and cause serious side effects. Do not start taking any new medicine without talking to your healthcare provider first.
- What are the possible side effects of NSAIDs?
NSAIDs can cause serious side effects, including:
See “What is the most important information I should know about medicines called Nonsteroidal Anti-inflammatory Drugs (NSAIDs)?
- new or worse high blood pressure
- heart failure
- liver problems including liver failure
- kidney problems including kidney failure
- low red blood cells (anemia)
- life-threatening skin reactions
- life-threatening allergic reactions
- Other side effects of NSAIDs include: stomach pain, constipation, diarrhea, gas, heartburn, nausea, vomiting, and dizziness.
- Get emergency help right away if you get any of the following symptoms:
- shortness of breath or trouble breathing
- chest pain
- weakness in one part or side of your body
- slurred speech
- swelling of the face or throat
- Stop taking your NSAID and call your healthcare provider right away if you get any of the following symptoms:
- nausea
- more tired or weaker than usual
- diarrhea
- itching
- your skin or eyes look yellow
- indigestion or stomach pain
- flu-like symptoms
- vomit blood
- there is blood in your bowel movement or it is black and sticky like tar
- unusual weight gain
- skin rash or blisters with fever
- swelling of the arms, legs, hands and feet
- If you take too much of your NSAID, call your healthcare provider or get medical help right away.
- These are not all the possible side effects of NSAIDs. For more information, ask your healthcare provider or pharmacist about NSAIDs.
- Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
Other information about NSAIDs
- Aspirin is an NSAID but it does not increase the chance of a heart attack. Aspirin can cause bleeding in the brain, stomach, and intestines. Aspirin can also cause ulcers in the stomach and intestines.
- Some NSAIDs are sold in lower doses without a prescription (over-the-counter). Talk to your healthcare provider before using over-the-counter NSAIDs for more than 10 days.
General information about the safe and effective use of NSAIDs
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use NSAIDs for a condition for which it was not prescribed. Do not give NSAIDs to other people, even if they have the same symptoms that you have. It may harm them.
If you would like more information about NSAIDs, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about NSAIDs that is written for health professionals.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Distributed by: Sun Pharmaceutical Industries, Inc.
- Cranbury, NJ 08512
Rev 02, November 2020
Section 43681-6
Pharmacodynamics
Sulindac tablets are a non-steroidal anti-inflammatory drug (NSAID) that exhibits anti-inflammatory, analgesic and antipyretic activities in animal models. The mechanism of action, like that of other NSAIDs, is not completely understood but may be related to prostaglandin synthetase inhibition.
Section 43682-4
Pharmacokinetics
Section 44425-7
Store at 20° to 25°C (68° to 77°F).
[See USP Controlled Room Temperature]
DISPENSE IN TIGHT, LIGHT-RESISTANT CONTAINER.
Heart Failure and Edema
The Coxib and traditional NSAID Trialists’ Collaboration meta-analysis of randomized controlled trials demonstrated an approximately two-fold increase in hospitalizations for heart failure in COX-2 selective-treated patients and nonselective NSAID-treated patients compared to placebo-treated patients. In a Danish National Registry study of patients with heart failure, NSAID use increased the risk of MI, hospitalization for heart failure, and death.
Additionally, fluid retention and edema have been observed in some patients treated with NSAIDs. Use of sulindac may blunt the CV effects of several therapeutic agents used to treat these medical conditions [e.g., diuretics, ACE inhibitors, or angiotensin receptor blockers (ARBs)] [see Drug Interactions].
Avoid the use of sulindac tablets in patients with severe heart failure unless the benefits are expected to outweigh the risk of worsening heart failure. If sulindac tablets are used in patients with sever heart failure, monitor patients for signs of worsening heart failure.
Management of Overdosage
Cases of overdosage have been reported and rarely, deaths have occurred. The following signs and symptoms may be observed following overdosage: stupor, coma, diminished urine output and hypotension.
In the event of overdosage, the stomach should be emptied by inducing vomiting or by gastric lavage, and the patient carefully observed and given symptomatic and supportive treatment.
Animal studies show that absorption is decreased by the prompt administration of activated charcoal and excretion is enhanced by alkalinization of the urine.
Principal Display Panel 150 Mg Tablet Bottle Label
Principal Display Panel 200 Mg Tablet Bottle Label
Structured Label Content
Recent Major Changes (34066-1)
Cardiovascular Thrombotic Events
- Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use [see Warnings and Precautions ].
- Sulindac tablets are contraindicated in the setting of coronary artery bypass graft (CABG) surgery [see Contraindications and Warnings].
Gastrointestinal Risk
- NSAIDs cause an increased risk of serious gastrointestinal adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients are at greater risk for serious gastrointestinal events. (see WARNINGS .)
Section 34072-9 (34072-9)
General
Sulindac tablets cannot be expected to substitute for corticosteroids or to treat corticosteroid insufficiency. Abrupt discontinuation of corticosteroids may lead to disease exacerbation. Patients on prolonged corticosteroid therapy should have their therapy tapered slowly if a decision is made to discontinue corticosteroids.
The pharmacological activity of sulindac tablets in reducing fever and inflammation may diminish the utility of these diagnostic signs in detecting complications of presumed noninfectious, painful conditions.
Section 34073-7 (34073-7)
Drug Interactions
Section 34075-2 (34075-2)
Laboratory Tests
Because serious GI tract ulcerations and bleeding can occur without warning symptoms, physicians should monitor for signs or symptoms of GI bleeding. Patients on long-term treatment with NSAIDs should have their CBC and a chemistry profile checked periodically. If clinical signs and symptoms consistent with liver or renal disease develop, systemic manifestations occur (e.g., eosinophilia, rash, etc.) or if abnormal liver tests persist or worsen, sulindac tablets should be discontinued.
Section 34076-0 (34076-0)
Information for Patients
Patients should be informed of the following information before initiating therapy with an NSAID and periodically during the course of ongoing therapy. Patients should also be encouraged to read the NSAID Medication Guide that accompanies each prescription dispensed.
- Cardiovascular Thrombotic Events
- Advise patients to be alert for the symptoms of cardiovascular thrombotic events, including chest pain, shortness of breath, weakness, or slurring of speech, and to report any of these symptoms to their healthcare provider immediately [see Warnings ].
- Sulindac tablets, like other NSAIDs, can cause GI discomfort and, rarely, serious GI side effects, such as ulcers and bleeding, which may result in hospitalization and even death. Although serious GI tract ulcerations and bleeding can occur without warning symptoms, patients should be alert for the signs and symptoms of ulcerations and bleeding, and should ask for medical advice when observing any indicative sign or symptoms including epigastric pain, dyspepsia, melena, and hematemesis. Patients should be apprised of the importance of this follow-up (see WARNINGS, Gastrointestinal Effects - Risk of Ulceration, Bleeding, and Perforation ).
- Serious Skin Reactions, including DRESS
- Advise patients to stop taking sulindac tablets immediately if they develop any type of rash or fever and to contact their healthcare provider as soon as possible (see WARNINGS).
- Heart Failure and Edema
- Advise patients to be alert for the symptoms of congestive heart failure including shortness of
- breath, unexplained weight gain, or edema and to contact their healthcare provider if such
- symptoms occur [see WARNINGS ].
- Patients should be informed of the warning signs and symptoms of hepatotoxicity (e.g., nausea, fatigue, lethargy, pruritus, jaundice, right upper quadrant tenderness, and "flu-like" symptoms). If these occur, patients should be instructed to stop therapy and seek immediate medical therapy.
- Patients should be informed of the signs of an anaphylactic/anaphylactoid reaction (e.g., difficulty breathing, swelling of the face or throat). If these occur, patients should be instructed to seek immediate emergency help (see WARNINGS ).
- Fetal Toxicity
- Inform pregnant women to avoid use of sulindac tablets and other NSAIDs starting at 30 weeks gestation because of the risk of the premature closing of the fetal ductus arteriosus. If treatment with sulindac tablets is needed for a pregnant woman between about 20 to 30 weeks gestation, advise her that she may need to be monitored for oligohydramnios, if treatment continues for longer than 48 hours (see WARNINGS; Fetal Toxicity, PRECAUTIONS; Pregnancy ).
Section 34077-8 (34077-8)
Risk Summary
Use of NSAIDs, including sulindac tablets, can cause premature closure of the fetal ductus arteriosus and fetal renal dysfunction leading to oligohydramnios and, in some cases, neonatal renal impairment. Because of these risks, limit dose and duration of sulindac tablets use between about 20 and 30 weeks of gestation, and avoid sulindac tablets use at about 30 weeks of gestation and later in pregnancy (see WARNINGS; Fetal Toxicity ).
Premature Closure of Fetal Ductus Arteriosus
Use of NSAIDs, including sulindac tablets, at about 30 weeks gestation or later in pregnancy increases
the risk of premature closure of the fetal ductus arteriosus.
Oligohydramnios/Neonatal Renal Impairment
Use of NSAIDs at about 20 weeks gestation or later in pregnancy has been associated with cases of fetal
renal dysfunction leading to oligohydramnios, and in some cases, neonatal renal impairment.
Data from observational studies regarding other potential embryofetal risks of NSAID use in women in the first or second trimesters of pregnancy are inconclusive. In animal reproduction studies conducted in rats and rabbits have not demonstrated evidence of developmental abnormalities. In reproduction studies in the rat, a decrease in average fetal weight and an increase in numbers of dead pups were observed on the first day of the postpartum period at dosage levels of 20 and 40 mg/kg/day (2½ and 5 times the usual maximum daily dose in humans), although there was no adverse effect on the survival and growth during the remainder of the postpartum period. Sulindac prolongs the duration of gestation in rats, as do other compounds of this class. Visceral and skeletal malformations observed in low incidence among rabbits in some teratology studies did not occur at the same dosage levels in repeat studies, nor at a higher dosage level in the same species. However, animal reproduction studies are not always predictive of human response. Based on animal data, prostaglandins have been shown to have an important role in endometrial vascular permeability, blastocyst implantation, and decidualization. In animal studies, administration of prostaglandin synthesis inhibitors such as sulindac, resulted in increased pre- and post-implantation loss. Prostaglandins also have been shown to have an important role in fetal kidney development. In published animal studies, prostaglandin synthesis inhibitors have been reported to impair kidney development when administered at clinically relevant doses.
The estimated background risk of major birth defects and miscarriage for the indicated population(s) is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Section 34078-6 (34078-6)
Clinical Considerations
Fetal/Neonatal Adverse Reactions
Premature Closure of Fetal Ductus Arteriosus:
Avoid use of NSAIDs in women at about 30 weeks gestation and later in pregnancy, because NSAIDs, including sulindac tablets, can cause premature closure of the fetal ductus arteriosus (see WARNINGS; Fetal Toxicity).
Oligohydramnios/Neonatal Renal Impairment
If an NSAID is necessary at about 20 weeks gestation or later in pregnancy, limit the use to the lowest effective dose and shortest duration possible. If sulindac tablets treatment extends beyond 48 hours, consider monitoring with ultrasound for oligohydramnios. If oligohydramnios occurs, discontinue sulindac tablets and follow up according to clinical practice (see WARNINGS; Fetal Toxicity).
Data
- Human Data
There are no adequate and well controlled studies in pregnant women. Sulindac should be used in pregnancy only if the potential benefit justifies the potential risk to the fetus. The known effects of drugs of this class on the human fetus during the third trimester of pregnancy include: constriction of the ductus arteriosus prenatally, tricuspid incompetence, and pulmonary hypertension; non-closure of the ductus arteriosus postnatally which may be resistant to medical management; myocardial degenerative changes, platelet dysfunction with resultant bleeding, intracranial bleeding, renal dysfunction or failure, renal injury/dysgenesis which may result in prolonged or permanent renal failure, oligohydramnios, gastrointestinal bleeding or perforation, and increased risk of necrotizing enterocolitis.
- Premature Closure of Fetal Ductus Arteriosus:
- Published literature reports that the use of NSAIDs at about 30 weeks of gestation and later in pregnancy may cause premature closure of the fetal ductus arteriosus.
- Oligohydramnios/Neonatal Renal Impairment:
- Published studies and postmarketing reports describe maternal NSAID use at about 20 weeks gestation or later in pregnancy associated with fetal renal dysfunction leading to oligohydramnios, and in some cases, neonatal renal impairment. These adverse outcomes are seen, on average, after days to weeks of treatment, although oligohydramnios has been infrequently reported as soon as 48 hours after NSAID initiation. In many cases, but not all, the decrease in amniotic fluid was transient and reversible with cessation of the drug. There have been a limited number of case reports of maternal NSAID use and neonatal renal dysfunction without oligohydramnios, some of which were irreversible. Some cases of neonatal renal dysfunction required treatment with invasive procedures, such as exchange transfusion or dialysis.
- Methodological limitations of these postmarketing studies and reports include lack of a control group; limited information regarding dose, duration, and timing of drug exposure; and concomitant use of other medications. These limitations preclude establishing a reliable estimate of the risk of adverse fetal and neonatal outcomes with maternal NSAID use. Because the published safety data on neonatal outcomes involved mostly preterm infants, the generalizability of certain reported risks to the full-term infant exposed to NSAIDs through maternal use is uncertain.
Section 34079-4 (34079-4)
Labor and Delivery
In rat studies with NSAIDs, as with other drugs known to inhibit prostaglandin synthesis, an increased incidence of dystocia, delayed parturition, and decreased pup survival occurred. The effects of sulindac tablets on labor and delivery in pregnant women are unknown.
Section 34080-2 (34080-2)
Nursing Mothers
It is not known whether this drug is excreted in human milk; however, it is secreted in the milk of lactating rats. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from sulindac tablets, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Section 34081-0 (34081-0)
Pediatric
The pharmacokinetics of sulindac have not been investigated in pediatric patients.
Section 34082-8 (34082-8)
Geriatric Use
As with any NSAID, caution should be exercised in treating the elderly (65 years and older) since advancing age appears to increase the possibility of adverse reactions. Elderly patients seem to tolerate ulceration or bleeding less well than other individuals and many spontaneous reports of fatal GI events are in this population (see WARNINGS, Gastrointestinal Effects - Risk of Ulceration, Bleeding, and Perforation ).
Sulindac tablets are known to be substantially excreted by the kidney and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection and it may be useful to monitor renal function (see WARNINGS, Renal Effects ).
Section 42228-7 (42228-7)
Fetal Toxicity
Premature Closure of Fetal Ductus Arteriosus:
Avoid use of NSAIDs, including sulindac tablets, in pregnant women at about 30 weeks gestation and later. NSAIDs including sulindac tablets, increase the risk of premature closure of the fetal ductus arteriosus at approximately this gestational age.
Oligohydramnios/Neonatal Renal Impairment:
Use of NSAIDs, including sulindac tablets, at about 20 weeks gestation or later in pregnancy may cause fetal renal dysfunction leading to oligohydramnios and, in some cases, neonatal renal impairment. These adverse outcomes are seen, on average, after days to weeks of treatment, although oligohydramnios has been infrequently reported as soon as 48 hours after NSAID initiation.
Oligohydramnios is often, but not always, reversible with treatment discontinuation. Complications of prolonged oligohydramnios may, for example, include limb contractures and delayed lung maturation. In some postmarketing cases of impaired neonatal renal function, invasive procedures such as exchange transfusion or dialysis were required.
If NSAID treatment is necessary between about 20 weeks and 30 weeks gestation, limit sulindac tablets use to the lowest effective dose and shortest duration possible. Consider ultrasound monitoring of amniotic fluid if sulindac tablets treatment extends beyond 48 hours. Discontinue sulindac tablets if oligohydramnios occurs and follow up according to clinical practice (see PRECAUTIONS; Pregnancy).
Section 42229-5 (42229-5)
Absorption
The extent of sulindac absorption from sulindac tablets is similar as compared to sulindac solution.
There is no information regarding food effect on sulindac absorption. Antacids containing magnesium hydroxide 200 mg and aluminum hydroxide 225 mg per 5 mL have been shown not to significantly decrease the extent of sulindac absorption.
|
PHARMACOKINETIC
PARAMETERS |
NORMAL | ELDERLY |
|---|---|---|
|
T max |
Age 19–41 (n=24) |
Age 65–87 (n=12) 400 mg qd |
|
|
|
|
|
|
(200 mg tablet) |
2.54 ± 1.52 S |
|
|
|
5.75 ± 2.81 SF |
|
|
3.38 ± 2.30 S |
6.83 ± 4.19 SP |
|
|
4.88 ± 2.57 SP |
|
|
|
4.96 ± 2.36 SF |
|
|
|
|
|
|
|
(150 mg tablet) |
|
|
|
|
|
|
|
3.90 ± 2.30 S |
|
|
|
5.85 ± 4.49 SP |
|
|
|
6.15 ± 3.07 SF |
|
|
Renal Clearance |
(200 mg tablet) |
|
|
|
|
|
|
|
68.12 ± 27.56 mL/min S |
|
|
|
36.58 ± 12.61 mL/min SP |
|
|
|
|
|
|
|
(150 mg tablet) |
|
|
|
|
|
|
|
74.39 ± 34.15 mL/min S |
|
|
|
41.75 ± 13.72 mL/min SP |
|
|
Mean effective Half life (h) |
7.8 S
|
|
|
S = Sulindac
|
Section 42231-1 (42231-1)
Medication Guide
for
Nonsteroidal Anti-inflammatory Drugs (NSAIDs)
What is the most important information I should know about medicines called Nonsteroidal Anti-inflammatory Drugs (NSAIDs)?
NSAIDs can cause serious side effects, including:
- Increased risk of a heart attack or stroke that can lead to death.This risk may happen early in treatment and may increase:
- with increasing doses of NSAIDs
- with longer use of NSAIDs
- Do not take NSAIDs right before or after a heart surgery called a "coronary artery bypass graft (CABG)."
- Avoid taking NSAIDs after a recent heart attack, unless your healthcare provider tells you to. You may have an increased risk of another heart attack if you take NSAIDs after a recent heart attack.
- Increased risk of bleeding, ulcers, and tears (perforation) of the esophagus (tube leading from the mouth to the stomach), stomach and intestines:
- anytime during use
- without warning symptoms
- that may cause death
The risk of getting an ulcer or bleeding increases with:
- past history of stomach ulcers, or stomach or intestinal bleeding with use of NSAIDs
- taking medicines called "corticosteroids", "anti-coagulants", "SSRIs", or "SNRIs"
- increasing doses of NSAIDs
- longer use of NSAIDs
- smoking
- drinking alcohol
- older age
- poor health
- advanced liver disease
- bleeding problems
- NSAIDs should only be used:
- exactly as prescribed
- at the lowest dose possible for your treatment
- for the shortest time needed
What are NSAIDs?
NSAIDs are used to treat pain and redness, swelling, and heat (inflammation) from medical conditions such as different types of arthritis, menstrual cramps, and other types of short-term pain.
Who should not take NSAIDs?
Do not take NSAIDs:
- if you have had an asthma attack, hives, or other allergic reaction with aspirin or any other NSAIDs.
- right before or after heart bypass surgery.
Before taking NSAIDs, tell your healthcare provider about all of your medical conditions, including if you:
- have liver or kidney problems
- have high blood pressure
- have asthma
- are pregnant or plan to become pregnant. Taking NSAIDs at about 20 weeks of pregnancy or later may harm your unborn baby. If you need to take NSAIDs for more than 2 days when you are between 20 and 30 weeks of pregnancy, your healthcare provider may need to monitor the amount of fluid in your womb around your baby. You should not take NSAIDs after about 30 weeks of pregnancy.
- are breastfeeding or plan to breast feed.
Tell your healthcare provider about all of the medicines you take, including prescription or over-the-counter medicines, vitamins or herbal supplements.NSAIDs and some other medicines can interact with each other and cause serious side effects. Do not start taking any new medicine without talking to your healthcare provider first.
- What are the possible side effects of NSAIDs?
NSAIDs can cause serious side effects, including:
See “What is the most important information I should know about medicines called Nonsteroidal Anti-inflammatory Drugs (NSAIDs)?
- new or worse high blood pressure
- heart failure
- liver problems including liver failure
- kidney problems including kidney failure
- low red blood cells (anemia)
- life-threatening skin reactions
- life-threatening allergic reactions
- Other side effects of NSAIDs include: stomach pain, constipation, diarrhea, gas, heartburn, nausea, vomiting, and dizziness.
- Get emergency help right away if you get any of the following symptoms:
- shortness of breath or trouble breathing
- chest pain
- weakness in one part or side of your body
- slurred speech
- swelling of the face or throat
- Stop taking your NSAID and call your healthcare provider right away if you get any of the following symptoms:
- nausea
- more tired or weaker than usual
- diarrhea
- itching
- your skin or eyes look yellow
- indigestion or stomach pain
- flu-like symptoms
- vomit blood
- there is blood in your bowel movement or it is black and sticky like tar
- unusual weight gain
- skin rash or blisters with fever
- swelling of the arms, legs, hands and feet
- If you take too much of your NSAID, call your healthcare provider or get medical help right away.
- These are not all the possible side effects of NSAIDs. For more information, ask your healthcare provider or pharmacist about NSAIDs.
- Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
Other information about NSAIDs
- Aspirin is an NSAID but it does not increase the chance of a heart attack. Aspirin can cause bleeding in the brain, stomach, and intestines. Aspirin can also cause ulcers in the stomach and intestines.
- Some NSAIDs are sold in lower doses without a prescription (over-the-counter). Talk to your healthcare provider before using over-the-counter NSAIDs for more than 10 days.
General information about the safe and effective use of NSAIDs
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use NSAIDs for a condition for which it was not prescribed. Do not give NSAIDs to other people, even if they have the same symptoms that you have. It may harm them.
If you would like more information about NSAIDs, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about NSAIDs that is written for health professionals.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Distributed by: Sun Pharmaceutical Industries, Inc.
- Cranbury, NJ 08512
Rev 02, November 2020
Section 43681-6 (43681-6)
Pharmacodynamics
Sulindac tablets are a non-steroidal anti-inflammatory drug (NSAID) that exhibits anti-inflammatory, analgesic and antipyretic activities in animal models. The mechanism of action, like that of other NSAIDs, is not completely understood but may be related to prostaglandin synthetase inhibition.
Section 43682-4 (43682-4)
Pharmacokinetics
Section 44425-7 (44425-7)
Store at 20° to 25°C (68° to 77°F).
[See USP Controlled Room Temperature]
DISPENSE IN TIGHT, LIGHT-RESISTANT CONTAINER.
Description (DESCRIPTION)
Sulindac is a non-steroidal, anti-inflammatory indene derivative designated chemically as (Z)-5-fluoro-2-methyl-1-[[ p-(methylsulfinyl)phenyl]methylene]-1 H-indene-3-acetic acid. It is not a salicylate, pyrazolone or propionic acid derivative. Its empirical formula is C 20H 17FO 3S, with a molecular weight of 356.42. Sulindac, a yellow crystalline compound, is a weak organic acid practically insoluble in water below pH 4.5, but very soluble as the sodium salt or in buffers of pH 6 or higher.
Sulindac tablets are available in 150 and 200 mg tablets for oral administration. Each tablet contains the following inactive ingredients: magnesium stearate, microcrystalline cellulose, and pregelatinized starch.
Following absorption, sulindac undergoes two major biotransformations — reversible reduction to the sulfide metabolite, and irreversible oxidation to the sulfone metabolite. Available evidence indicates that the biological activity resides with the sulfide metabolite.
The structural formulas of sulindac and its metabolites are:
How Supplied (HOW SUPPLIED)
Sulindac tablets USP are supplied as follows:
Sulindac tablets, 150 mg, yellow, round, unscored, debossed MP 112
|
Bottles of 50 |
NDC 53489-478-02 |
|
Bottles of 60 |
NDC 53489-478-06 |
|
Bottles of 100 |
NDC 53489-478-01 |
|
Bottles of 250 |
NDC 53489-478-03 |
|
Bottles of 500 |
NDC 53489-478-05 |
|
Bottles of 1000 |
NDC 53489-478-10 |
Sulindac tablets, 200 mg, yellow, round, scored, debossed MP 116
|
Bottles of 50 |
NDC 53489-479-02 |
|
Bottles of 60 |
NDC 53489-479-06 |
|
Bottles of 100 |
NDC 53489-479-01 |
|
Bottles of 250 |
NDC 53489-479-03 |
|
Bottles of 500 |
NDC 53489-479-05 |
|
Bottles of 1000 |
NDC 53489-479-10 |
Adverse Reactions (ADVERSE REACTIONS)
The following adverse reactions were reported in clinical trials or have been reported since the drug was marketed. The probability exists of a causal relationship between sulindac tablets and these adverse reactions. The adverse reactions which have been observed in clinical trials encompass observations in 1,865 patients, including 232 observed for at least 48 weeks.
Contraindications (CONTRAINDICATIONS)
Sulindac tablets are contraindicated in patients with known hypersensitivity to sulindac or the excipients (see DESCRIPTION ).
Sulindac tablets should not be given to patients who have experienced asthma, urticaria, or allergic-type reactions after taking aspirin or other NSAIDs. Severe, rarely fatal, anaphylactic/anaphylactoid reactions to NSAIDs have been reported in such patients (see WARNINGS – Anaphylactic/Anaphylactoid Reactions , and PRECAUTIONS – Preexisting Asthma ).
- In the setting of coronary artery bypass graft (CABG) surgery [see WARNINGS ].
Indications and Usage (INDICATIONS AND USAGE)
Carefully consider the potential benefits and risks of sulindac tablets and other treatment options before deciding to use sulindac tablets. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS ).
Sulindac tablets are indicated for acute or long-term use in the relief of signs and symptoms of the following:
- Osteoarthritis
- Rheumatoid arthritis*
- Ankylosing spondylitis
- Acute painful shoulder (Acute subacromial bursitis/supraspinatus tendinitis)
- Acute gouty arthritis
Heart Failure and Edema
The Coxib and traditional NSAID Trialists’ Collaboration meta-analysis of randomized controlled trials demonstrated an approximately two-fold increase in hospitalizations for heart failure in COX-2 selective-treated patients and nonselective NSAID-treated patients compared to placebo-treated patients. In a Danish National Registry study of patients with heart failure, NSAID use increased the risk of MI, hospitalization for heart failure, and death.
Additionally, fluid retention and edema have been observed in some patients treated with NSAIDs. Use of sulindac may blunt the CV effects of several therapeutic agents used to treat these medical conditions [e.g., diuretics, ACE inhibitors, or angiotensin receptor blockers (ARBs)] [see Drug Interactions].
Avoid the use of sulindac tablets in patients with severe heart failure unless the benefits are expected to outweigh the risk of worsening heart failure. If sulindac tablets are used in patients with sever heart failure, monitor patients for signs of worsening heart failure.
Management of Overdosage (MANAGEMENT OF OVERDOSAGE)
Cases of overdosage have been reported and rarely, deaths have occurred. The following signs and symptoms may be observed following overdosage: stupor, coma, diminished urine output and hypotension.
In the event of overdosage, the stomach should be emptied by inducing vomiting or by gastric lavage, and the patient carefully observed and given symptomatic and supportive treatment.
Animal studies show that absorption is decreased by the prompt administration of activated charcoal and excretion is enhanced by alkalinization of the urine.
Dosage and Administration (DOSAGE AND ADMINISTRATION)
Carefully consider the potential benefits and risks of sulindac tablets and other treatment options before deciding to use sulindac tablets. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS ).
After observing the response to initial therapy with sulindac tablets, the dose and frequency should be adjusted to suit an individual patient's needs.
Sulindac tablets should be administered orally twice a day with food. The maximum dosage is 400 mg per day. Dosages above 400 mg per day are not recommended.
In osteoarthritis, rheumatoid arthritis, and ankylosing spondylitis, the recommended starting dosage is 150 mg twice a day. The dosage may be lowered or raised depending on the response.
A prompt response (within one week) can be expected in about one-half of patients with osteoarthritis, ankylosing spondylitis, and rheumatoid arthritis. Others may require longer to respond.
In acute painful shoulder (acute subacromial bursitis/supraspinatus tendinitis) and acute gouty arthritis, the recommended dosage is 200 mg twice a day. After a satisfactory response has been achieved, the dosage may be reduced according to the response. In acute painful shoulder, therapy for 7–14 days is usually adequate. In acute gouty arthritis, therapy for 7 days is usually adequate.
**Incidence between 3% and 9%. Those reactions occurring in 1% to 3% of patients are not marked with an asterisk.
Principal Display Panel 150 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 150 mg Tablet Bottle Label)
Principal Display Panel 200 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 200 mg Tablet Bottle Label)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:29.455426 · Updated: 2026-03-14T22:15:30.143580