Devanov®
a353dc32-4a83-4c2e-8759-2be06c3d0eab
34390-5
HUMAN OTC DRUG LABEL
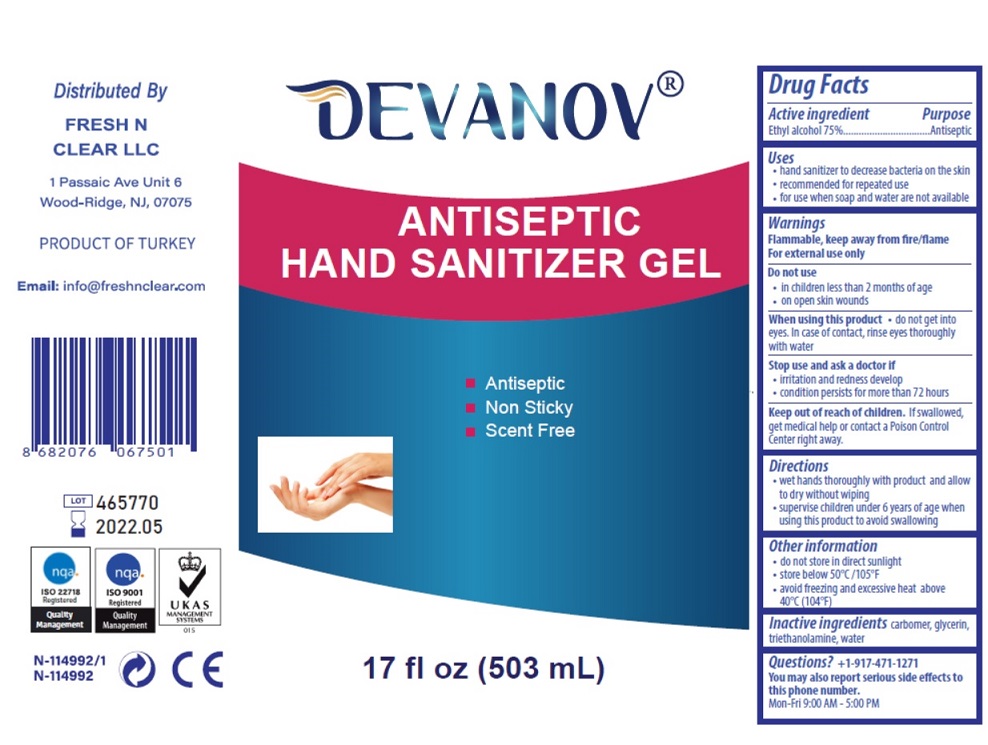
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Ethyl alcohol 75%
Purpose
Antiseptic
Medication Information
Purpose
Antiseptic
Description
Ethyl alcohol 75%
Uses
- hand sanitizer to decrease bacteria on the skin
- recommended for repeated use
- for use when soap and water are not available
Section 42229-5
■ Antiseptic
■ Non Sticky
■ Scent Free
Distributed By
FRESH N CLEAR LLC
1 Passaic Ave Unit 6
Wood-Ridge, NJ, 07075
PRODUCT OF TURKEY
Email: [email protected]
nqa ISO 22718 Registered Quality Management
nqa ISO 9001 Registered Quality Management
U K A S MANAGEMENT SYSTEMS
N-114992/1 CE
N-114992
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 51727-6
Inactive ingredients carbomer, glycerin, triethanolamine, water
Section 53413-1
Questions? +1-917-471-1271
You may also report serious side effects to this phone number.
Mon-Fri 9:00 AM - 5:00 PM
Warnings
Flammable, keep away from fire/flame
For external use only
Do not use
- in children less than 2 months of age
- on open skin wounds
When using this product • do not get into eyes. In case of contact, rinse eyes thoroughly with water
Stop use and ask a doctor if
- irritation and redness develop
- condition persists for more than 72 hours
Packaging
Directions
- wet hands thoroughly with product and allow to dry without wiping
- supervise children under 6 years of age when using this product to avoid swallowing
Drug Facts
Active Ingredient
Ethyl alcohol 75%
Other Information
- do not store in direct sunlight
- store below 50°C /105°F
- avoid freezing and excessive heat above 40°C (104°F)
Structured Label Content
Uses
- hand sanitizer to decrease bacteria on the skin
- recommended for repeated use
- for use when soap and water are not available
Section 42229-5 (42229-5)
■ Antiseptic
■ Non Sticky
■ Scent Free
Distributed By
FRESH N CLEAR LLC
1 Passaic Ave Unit 6
Wood-Ridge, NJ, 07075
PRODUCT OF TURKEY
Email: [email protected]
nqa ISO 22718 Registered Quality Management
nqa ISO 9001 Registered Quality Management
U K A S MANAGEMENT SYSTEMS
N-114992/1 CE
N-114992
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 51727-6 (51727-6)
Inactive ingredients carbomer, glycerin, triethanolamine, water
Section 53413-1 (53413-1)
Questions? +1-917-471-1271
You may also report serious side effects to this phone number.
Mon-Fri 9:00 AM - 5:00 PM
Purpose
Antiseptic
Warnings
Flammable, keep away from fire/flame
For external use only
Do not use
- in children less than 2 months of age
- on open skin wounds
When using this product • do not get into eyes. In case of contact, rinse eyes thoroughly with water
Stop use and ask a doctor if
- irritation and redness develop
- condition persists for more than 72 hours
Packaging
Directions
- wet hands thoroughly with product and allow to dry without wiping
- supervise children under 6 years of age when using this product to avoid swallowing
Drug Facts
Active Ingredient (Active ingredient)
Ethyl alcohol 75%
Other Information (Other information)
- do not store in direct sunlight
- store below 50°C /105°F
- avoid freezing and excessive heat above 40°C (104°F)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:57:47.236737 · Updated: 2026-03-14T22:57:04.097940