These Highlights Do Not Include All The Information Needed To Use Colesevelam Hydrochloride Tablets Safely And Effectively. See Full Prescribing Information For Colesevelam Hydrochloride Tablets.
a2c5e881-2901-4f90-95cc-74930569e51e
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Colesevelam hydrochloride is a bile acid sequestrant indicated as an adjunct to diet and exercise to: reduce elevated low-density lipoprotein cholesterol (LDL-C) in adults with primary hyperlipidemia ( 1.1 ). reduce LDL-C levels in boys and post-menarchal girls, 10 years to 17 years of age, with heterozygous familial hypercholesterolemia (HeFH), unable to reach LDL-C target levels despite an adequate trial of diet and lifestyle modification ( 1.1 ). improve glycemic control in adults with type 2 diabetes mellitus ( 1.2 ). Limitations of Use ( 1.3 ): Do not use for treatment of type 1 diabetes or for diabetic ketoacidosis. Not studied in Fredrickson Type I, III, IV, and V dyslipidemias.
Indications and Usage
Colesevelam hydrochloride is a bile acid sequestrant indicated as an adjunct to diet and exercise to: reduce elevated low-density lipoprotein cholesterol (LDL-C) in adults with primary hyperlipidemia ( 1.1 ). reduce LDL-C levels in boys and post-menarchal girls, 10 years to 17 years of age, with heterozygous familial hypercholesterolemia (HeFH), unable to reach LDL-C target levels despite an adequate trial of diet and lifestyle modification ( 1.1 ). improve glycemic control in adults with type 2 diabetes mellitus ( 1.2 ). Limitations of Use ( 1.3 ): Do not use for treatment of type 1 diabetes or for diabetic ketoacidosis. Not studied in Fredrickson Type I, III, IV, and V dyslipidemias.
Dosage and Administration
Obtain lipid parameters, including serum triglyceride (TG) levels, before starting colesevelam hydrochloride tablets ( 2.1 ) The recommended dosage for adults and for boys and post-menarchal girls aged 10 years to 17 years with primary hyperlipidemia is 3.75 grams daily. The recommended dosage for adults with type 2 diabetes mellitus is 3.75 grams daily. Colesevelam hydrochloride tablets should be taken as follows ( 2.2 , 2.4 ): Take 6 tablets once daily or 3 tablets twice daily with a meal and liquid.
Warnings and Precautions
Hypertriglyceridemia and Pancreatitis: Colesevelam hydrochloride can increase TG. Hypertriglyceridemia can cause acute pancreatitis. Monitor lipids, including TG. Instruct patients to discontinue colesevelam hydrochloride and seek prompt medical attention if the symptoms of acute pancreatitis occur ( 5.1 ). Gastrointestinal Obstruction: Cases of bowel obstruction have occurred. Colesevelam hydrochloride is not recommended in patients with gastroparesis, other gastrointestinal motility disorders, and in those who have had major gastrointestinal tract surgery and who may be at risk for bowel obstruction ( 5.2 ). Vitamin K or Fat-Soluble Vitamin Deficiencies : Colesevelam hydrochloride may decrease absorption of fat-soluble vitamins. Patients with a susceptibility to deficiencies of vitamin K (e.g., patients on warfarin, patients with malabsorption syndromes) or other fat-soluble vitamins may be at increased risk. Patients on oral vitamin supplementation should take their vitamins at least 4 hours prior to colesevelam hydrochloride ( 5.3 ). Drug Interactions: Due to the potential for decreased absorption of other drugs that have not been tested for interaction, consider administering at least 4 hours prior to colesevelam hydrochloride ( 5.4 , 7 , 12.3 ).
Contraindications
Colesevelam hydrochloride is contraindicated in patients with: Serum TG concentrations > 500 mg/dL [see Warnings and Precautions (5.1) ] History of hypertriglyceridemia-induced pancreatitis [see Warnings and Precautions (5.1) ] A history of bowel obstruction [see Warnings and Precautions (5.2) ]
Adverse Reactions
The following important adverse reactions are described below and elsewhere in the labeling: Hypertriglyceridemia and Pancreatitis [see Warnings and Precautions (5.1) ] Gastrointestinal Obstruction [see Warnings and Precautions (5.2) ] Vitamin K or Fat-Soluble Vitamin Deficiencies [see Warnings and Precautions (5.3) ]
Drug Interactions
Colesevelam hydrochloride reduces gastrointestinal absorption of some drugs. Administer drugs with a known interaction at least 4 hours prior to colesevelam hydrochloride [see Drug Interactions (7) ] . Due to the potential for decreased absorption of other drugs that have not been tested for interaction, especially those with a narrow therapeutic index, consider administering at least 4 hours prior to colesevelam hydrochloride [see Clinical Pharmacology (12.3) ].
Storage and Handling
Colesevelam hydrochloride tablets, 625 mg are supplied as white to off-white, oval-shaped, film-coated tablets, printed with “COL” on one side with black ink and are available as follows: Bottles of 180 - NDC 69452-158-25
How Supplied
Colesevelam hydrochloride tablets, 625 mg are supplied as white to off-white, oval-shaped, film-coated tablets, printed with “COL” on one side with black ink and are available as follows: Bottles of 180 - NDC 69452-158-25
Medication Information
Warnings and Precautions
Hypertriglyceridemia and Pancreatitis: Colesevelam hydrochloride can increase TG. Hypertriglyceridemia can cause acute pancreatitis. Monitor lipids, including TG. Instruct patients to discontinue colesevelam hydrochloride and seek prompt medical attention if the symptoms of acute pancreatitis occur ( 5.1 ). Gastrointestinal Obstruction: Cases of bowel obstruction have occurred. Colesevelam hydrochloride is not recommended in patients with gastroparesis, other gastrointestinal motility disorders, and in those who have had major gastrointestinal tract surgery and who may be at risk for bowel obstruction ( 5.2 ). Vitamin K or Fat-Soluble Vitamin Deficiencies : Colesevelam hydrochloride may decrease absorption of fat-soluble vitamins. Patients with a susceptibility to deficiencies of vitamin K (e.g., patients on warfarin, patients with malabsorption syndromes) or other fat-soluble vitamins may be at increased risk. Patients on oral vitamin supplementation should take their vitamins at least 4 hours prior to colesevelam hydrochloride ( 5.3 ). Drug Interactions: Due to the potential for decreased absorption of other drugs that have not been tested for interaction, consider administering at least 4 hours prior to colesevelam hydrochloride ( 5.4 , 7 , 12.3 ).
Indications and Usage
Colesevelam hydrochloride is a bile acid sequestrant indicated as an adjunct to diet and exercise to: reduce elevated low-density lipoprotein cholesterol (LDL-C) in adults with primary hyperlipidemia ( 1.1 ). reduce LDL-C levels in boys and post-menarchal girls, 10 years to 17 years of age, with heterozygous familial hypercholesterolemia (HeFH), unable to reach LDL-C target levels despite an adequate trial of diet and lifestyle modification ( 1.1 ). improve glycemic control in adults with type 2 diabetes mellitus ( 1.2 ). Limitations of Use ( 1.3 ): Do not use for treatment of type 1 diabetes or for diabetic ketoacidosis. Not studied in Fredrickson Type I, III, IV, and V dyslipidemias.
Dosage and Administration
Obtain lipid parameters, including serum triglyceride (TG) levels, before starting colesevelam hydrochloride tablets ( 2.1 ) The recommended dosage for adults and for boys and post-menarchal girls aged 10 years to 17 years with primary hyperlipidemia is 3.75 grams daily. The recommended dosage for adults with type 2 diabetes mellitus is 3.75 grams daily. Colesevelam hydrochloride tablets should be taken as follows ( 2.2 , 2.4 ): Take 6 tablets once daily or 3 tablets twice daily with a meal and liquid.
Contraindications
Colesevelam hydrochloride is contraindicated in patients with: Serum TG concentrations > 500 mg/dL [see Warnings and Precautions (5.1) ] History of hypertriglyceridemia-induced pancreatitis [see Warnings and Precautions (5.1) ] A history of bowel obstruction [see Warnings and Precautions (5.2) ]
Adverse Reactions
The following important adverse reactions are described below and elsewhere in the labeling: Hypertriglyceridemia and Pancreatitis [see Warnings and Precautions (5.1) ] Gastrointestinal Obstruction [see Warnings and Precautions (5.2) ] Vitamin K or Fat-Soluble Vitamin Deficiencies [see Warnings and Precautions (5.3) ]
Drug Interactions
Colesevelam hydrochloride reduces gastrointestinal absorption of some drugs. Administer drugs with a known interaction at least 4 hours prior to colesevelam hydrochloride [see Drug Interactions (7) ] . Due to the potential for decreased absorption of other drugs that have not been tested for interaction, especially those with a narrow therapeutic index, consider administering at least 4 hours prior to colesevelam hydrochloride [see Clinical Pharmacology (12.3) ].
Storage and Handling
Colesevelam hydrochloride tablets, 625 mg are supplied as white to off-white, oval-shaped, film-coated tablets, printed with “COL” on one side with black ink and are available as follows: Bottles of 180 - NDC 69452-158-25
How Supplied
Colesevelam hydrochloride tablets, 625 mg are supplied as white to off-white, oval-shaped, film-coated tablets, printed with “COL” on one side with black ink and are available as follows: Bottles of 180 - NDC 69452-158-25
Description
Colesevelam hydrochloride is a bile acid sequestrant indicated as an adjunct to diet and exercise to: reduce elevated low-density lipoprotein cholesterol (LDL-C) in adults with primary hyperlipidemia ( 1.1 ). reduce LDL-C levels in boys and post-menarchal girls, 10 years to 17 years of age, with heterozygous familial hypercholesterolemia (HeFH), unable to reach LDL-C target levels despite an adequate trial of diet and lifestyle modification ( 1.1 ). improve glycemic control in adults with type 2 diabetes mellitus ( 1.2 ). Limitations of Use ( 1.3 ): Do not use for treatment of type 1 diabetes or for diabetic ketoacidosis. Not studied in Fredrickson Type I, III, IV, and V dyslipidemias.
Section 42229-5
Take 6 tablets once daily or 3 tablets twice daily. Due to tablet size, colesevelam hydrochloride for oral suspension is recommended for use in the pediatric population.
Section 44425-7
Store at 25°C (77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Protect from moisture. Brief exposure to 40°C (104°F) does not adversely affect colesevelam hydrochloride tablets.
10 Overdosage
Colesevelam hydrochloride is not absorbed and the risk of systemic toxicity is low. Excessive doses of colesevelam hydrochloride may cause more severe local gastrointestinal effects (e.g., constipation).
11 Description
Colesevelam hydrochloride is a non-absorbed, polymeric, lipid-lowering and glucose-lowering agent for oral administration. Colesevelam hydrochloride is a high-capacity bile acid-binding molecule.
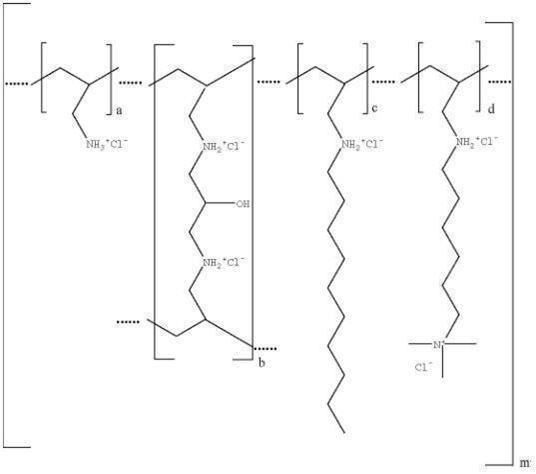
Colesevelam hydrochloride is poly(allylamine hydrochloride) cross-linked with epichlorohydrin and alkylated with 1-bromodecane and (6-bromohexyl)-trimethylammonium bromide. The chemical name (IUPAC) of colesevelam hydrochloride is allylamine polymer with 1-chloro-2,3-epoxypropane, [6 (allylamino)-hexyl]trimethylammonium chloride and N-allyldecylamine, hydrochloride. The chemical structure of colesevelam hydrochloride is represented by the following formula:
wherein (a) represents allyl amine monomer units that have not been alkylated by either of the 1‑bromodecane or (6-bromohexyl)-trimethylammonium bromide alkylating agents or cross-linked by epichlorohydrin; (b) represents allyl amine units that have undergone cross-linking with epichlorohydrin; (c) represents allyl amine units that have been alkylated with a decyl group; (d) represents allyl amine units that have been alkylated with a (6-trimethylammonium) hexyl group, and m represents a number ≥ 100 to indicate an extended polymer network. A small amount of the amines are dialkylated and are not depicted in the formula above. No regular order of the groups is implied by the structure; cross-linking and alkylation are expected to occur randomly along the polymer chains. A large amount of the amines are protonated. The polymer is depicted in the hydrochloride form; a small amount of the halides are bromide. Colesevelam hydrochloride is hydrophilic and insoluble in water.
Colesevelam hydrochloride tablets are white to off-white, oval-shaped, film-coated tablets, printed with “COL” on one side with black ink containing 625 mg colesevelam hydrochloride. In addition, each tablet contains the following inactive ingredients: magnesium stearate, microcrystalline cellulose, silicon dioxide. The tablets are imprinted using a water-soluble black ink (ferrosoferric oxide, hypromellose, and propylene glycol). The coating material contains polyethylene glycol, polysorbate 80 and talc (approximately 5 calories per 6 tablets).
8.4 Pediatric Use
Primary Hyperlipidemia
The safety and effectiveness of colesevelam hydrochloride to reduce LDL-C levels in boys and post-menarchal girls 10 years to 17 years of age with HeFH who are unable to reach LDL-C target levels despite an adequate trial of dietary therapy and lifestyle modification have been established. Use of colesevelam hydrochloride for this indication is supported by a study in 129 colesevelam hydrochloride-treated pediatric patients aged 10 years to 17 years with HeFH [see Clinical Studies (14.1)] . Adverse reactions commonly observed in pediatric patients compared to placebo, but not in adults, included headache (3.9%), creatine phosphokinase increase (2.3%), and vomiting (2.3%) [see Adverse Reactions (6.1)] . There were no significant effects on fat-soluble vitamin levels or clotting factors in the adolescent boys or girls relative to placebo. Due to colesevelam hydrochloride tablet size, colesevelam hydrochloride for oral suspension is recommended for use in the pediatric population [see Dosage and Administration (2.2, 2.4)]. The safety and effectiveness of colesevelam hydrochloride in pediatric patients with HeFH less than 10 years of age or in premenarchal females have not been established.
Type 2 Diabetes Mellitus
The safety and effectiveness of colesevelam hydrochloride to improve glycemic control in pediatric patients with type 2 diabetes mellitus have not been established. Effectiveness was not demonstrated in a 6-month, adequate and well-controlled study conducted in 141 colesevelam hydrochloride-treated pediatric patients aged 10 years to 17 years with type 2 diabetes mellitus.
4 Contraindications
Colesevelam hydrochloride is contraindicated in patients with:
- Serum TG concentrations > 500 mg/dL [see Warnings and Precautions (5.1)]
- History of hypertriglyceridemia-induced pancreatitis [see Warnings and Precautions (5.1)]
- A history of bowel obstruction [see Warnings and Precautions (5.2)]
6 Adverse Reactions
The following important adverse reactions are described below and elsewhere in the labeling:
- Hypertriglyceridemia and Pancreatitis [see Warnings and Precautions (5.1)]
- Gastrointestinal Obstruction [see Warnings and Precautions (5.2)]
- Vitamin K or Fat-Soluble Vitamin Deficiencies [see Warnings and Precautions (5.3)]
7 Drug Interactions
Concomitant use with colesevelam hydrochloride may decrease the exposure of the following drugs:Drugs with a narrow therapeutic index (e.g., cyclosporine), phenytoin, thyroid hormone replacement therapy, warfarin, oral contraceptives containing ethinyl estradiol and norethindrone, olmesartan medoxomil, and sulfonylureas (glimepiride, glipizide, glyburide). Administer these drugs 4 hours prior to colesevelam hydrochloride. For patients on warfarin, monitor International Normalized Ratio (INR) frequently during initiation then periodically ( 7.1).
Concomitant use with colesevelam hydrochloride may increase the exposure of the following drugs:Metformin extended release. Monitor patients' glycemic control ( 7.2).
8.6 Renal Impairment
Type 2 Diabetes Mellitus
Of the 2,048 patients enrolled in the six diabetes studies, 807 (39%) had mild renal insufficiency (creatinine clearance [CrCl] 50 mL/min to < 80 mL/min), 61 (3%) had moderate renal insufficiency (CrCl 30 mL/min to < 50 mL/min), and none had severe renal insufficiency (CrCl < 30 mL/min), as estimated from baseline serum creatinine using the Modification of Diet in Renal Disease (MDRD) equation. No overall differences in safety or effectiveness were observed between patients with CrCl < 50 mL/min (n = 53) and those with a CrCl ≥ 50 mL/min (n = 1,075) in the add-on to metformin, sulfonylureas, and insulin diabetes studies. In the monotherapy study and add-on to pioglitazone study, only 3 and 5 patients, respectively, had moderate renal insufficiency.
12.2 Pharmacodynamics
A maximum therapeutic response to the lipid-lowering effects of colesevelam hydrochloride was achieved within 2 weeks and was maintained during long-term therapy. In the diabetes clinical studies, a therapeutic response to colesevelam hydrochloride, as reflected by a reduction in HbA1c, was initially noted following 4 weeks to 6 weeks of treatment and reached maximal or near-maximal effect after 12 weeks to 18 weeks of treatment.
5.4 Drug Interactions
Colesevelam hydrochloride reduces gastrointestinal absorption of some drugs. Administer drugs with a known interaction at least 4 hours prior to colesevelam hydrochloride [see Drug Interactions (7)] .
Due to the potential for decreased absorption of other drugs that have not been tested for interaction, especially those with a narrow therapeutic index, consider administering at least 4 hours prior to colesevelam hydrochloride [see Clinical Pharmacology (12.3)].
1.3 Limitations of Use
- Colesevelam hydrochloride should not be used for the treatment of type 1 diabetes or for the treatment of diabetic ketoacidosis.
- Colesevelam hydrochloride has not been studied in Fredrickson Type I, III, IV, and V dyslipidemias.
1 Indications and Usage
Colesevelam hydrochloride is a bile acid sequestrant indicated as an adjunct to diet and exercise to:
- reduce elevated low-density lipoprotein cholesterol (LDL-C) in adults with primary hyperlipidemia ( 1.1).
- reduce LDL-C levels in boys and post-menarchal girls, 10 years to 17 years of age, with heterozygous familial hypercholesterolemia (HeFH), unable to reach LDL-C target levels despite an adequate trial of diet and lifestyle modification ( 1.1).
- improve glycemic control in adults with type 2 diabetes mellitus ( 1.2).
Limitations of Use ( 1.3):
- Do not use for treatment of type 1 diabetes or for diabetic ketoacidosis.
- Not studied in Fredrickson Type I, III, IV, and V dyslipidemias.
1.1 Primary Hyperlipidemia
Colesevelam hydrochloride tablets are indicated as an adjunct to diet and exercise to reduce elevated low-density lipoprotein cholesterol (LDL-C) in adults with primary hyperlipidemia.
Colesevelam hydrochloride tablets are indicated to reduce LDL-C levels in boys and post-menarchal girls, 10 years to 17 years of age, with heterozygous familial hypercholesterolemia (HeFH) who are unable to reach LDL-C target levels despite an adequate trial of dietary therapy and lifestyle modification.
5 Warnings and Precautions
- Hypertriglyceridemia and Pancreatitis:Colesevelam hydrochloride can increase TG. Hypertriglyceridemia can cause acute pancreatitis. Monitor lipids, including TG. Instruct patients to discontinue colesevelam hydrochloride and seek prompt medical attention if the symptoms of acute pancreatitis occur ( 5.1).
- Gastrointestinal Obstruction:Cases of bowel obstruction have occurred. Colesevelam hydrochloride is not recommended in patients with gastroparesis, other gastrointestinal motility disorders, and in those who have had major gastrointestinal tract surgery and who may be at risk for bowel obstruction ( 5.2).
- Vitamin K or Fat-Soluble Vitamin Deficiencies : Colesevelam hydrochloride may decrease absorption of fat-soluble vitamins. Patients with a susceptibility to deficiencies of vitamin K (e.g., patients on warfarin, patients with malabsorption syndromes) or other fat-soluble vitamins may be at increased risk. Patients on oral vitamin supplementation should take their vitamins at least 4 hours prior to colesevelam hydrochloride ( 5.3).
- Drug Interactions:Due to the potential for decreased absorption of other drugs that have not been tested for interaction, consider administering at least 4 hours prior to colesevelam hydrochloride ( 5.4, 7, 12.3).
14.1 Primary Hyperlipidemia
Colesevelam hydrochloride reduces total cholesterol (TC), LDL-C, apolipoprotein B (Apo B), and non-high-density lipoprotein cholesterol (non-HDL-C) when administered alone or in combination with a statin in patients with primary hyperlipidemia. Approximately 1,600 patients were studied in 9 clinical trials with treatment durations ranging from 4 weeks to 50 weeks. With the exception of one open-label, uncontrolled, long-term extension study, all studies were multicenter, randomized, double-blind, and placebo-controlled. A maximum therapeutic response to colesevelam hydrochloride was achieved within 2 weeks and was maintained during long-term therapy.
2 Dosage and Administration
- Obtain lipid parameters, including serum triglyceride (TG) levels, before starting colesevelam hydrochloride tablets ( 2.1)
- The recommended dosage for adults and for boys and post-menarchal girls aged 10 years to 17 years with primary hyperlipidemia is 3.75 grams daily. The recommended dosage for adults with type 2 diabetes mellitus is 3.75 grams daily. Colesevelam hydrochloride tablets should be taken as follows ( 2.2, 2.4):
Take 6 tablets once daily or 3 tablets twice daily with a meal and liquid.
1.2 Type 2 Diabetes Mellitus
Colesevelam hydrochloride tablets are indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
3 Dosage Forms and Strengths
- Tablets: 625 mg tablets are white to off-white, oval-shaped, film-coated tablets and printed with “COL” on one side with black ink.
14.2 Type 2 Diabetes Mellitus
Colesevelam hydrochloride has been studied as monotherapy and in combination with metformin, pioglitazone, sulfonylureas, and insulin. In these studies, colesevelam hydrochloride and placebo were administered either as 3 tablets twice daily with lunch and dinner or as 6 tablets with dinner alone.
Monotherapy
The efficacy of colesevelam hydrochloride 3.8 g/day as anti-diabetes monotherapy was evaluated in a randomized double-blind, placebo-controlled trial involving 357 patients (176 colesevelam hydrochloride and 181 placebo) with type 2 diabetes mellitus who were treatment-naïve or had not received antihyperglycemic medication within 3 months prior to the start of the study. Statin use at baseline was reported in 13% of the colesevelam hydrochloride-treated patients and 16% of the placebo‑treated patients.
Colesevelam hydrochloride resulted in a statistically significant reduction in HbA1c of 0.27% compared to placebo (Table 10).
The mean baseline LDL-C was 121 mg/dL in the monotherapy trial. Colesevelam hydrochloride treatment resulted in a placebo-corrected 11% reduction in LDL-C. Colesevelam hydrochloride treatment also reduced serum TC, ApoB, and non-HDL-C (Table 11). The mean change in body weight was -0.6 kg for colesevelam hydrochloride and ‑0.7 kg for placebo treatment groups.
|
Colesevelam Hydrochloride
3.8 g/day |
Placebo | |
| HbA1c (%), Mean | ||
| N | 175 | 169 |
| Baseline | 8.25 | 8.17 |
| Change from baseline
Least-squares mean change calculated from an Analysis of Covariance model
|
-0.26 | 0.01 |
| Treatment difference (p-value) | -0.27 (p = 0.013) | |
| FPG (mg/dL), Mean | ||
| N | 172 | 166 |
| Baseline | 172 | 168 |
| Change from baseline * | -4.6 | 5.7 |
| Treatment difference (p-value) | -10.3 (p = 0.037
Nominal p = value, not controlled for multiplicity testing )
|
FPG = fasting plasma glucose
| Dose/Day |
N
The number of patients with analyzable data, i.e., a baseline and post-treatment value (last observation carried forward), varied slightly among different parameters. The N given represents the smallest number of patients included in the analysis for any parameter.
|
TC | LDL-C | Apo B | HDL-C | Non-HDL-C |
TG
Median % change from baseline
|
| Colesevelam Hydrochloride 3.8 g | 162 | -3.3
p < 0.001 for lipid parameters compared to placebo (This more stringent criterion for statistical significance accounts for multiplicity testing of the lipid parameters, which were secondary endpoints in the diabetes trials.)
|
-10.0 ‡ | -5.6 ‡ | 1.7 | -4.4 ‡ | 15.5 |
| Placebo | 160 | 1.8 | 1.2 | 0.9 | -0.1 | 3.0 | 5.8 |
Add-on Combination Therapy
The efficacy of colesevelam hydrochloride 3.8 g/day in patients with type 2 diabetes mellitus was evaluated in 5 double-blind, placebo‑controlled add-on therapy trials involving a total of 1,691 patients with baseline HbA1c 7.5% to 9.5%. Patients were enrolled and maintained on their pre-existing, stable, background anti‑diabetic regimen. Statin use at baseline was reported in 41% of the colesevelam hydrochloride-treated patients and 48% of the placebo-treated patients.
In 3 add-on combination therapy trials (metformin, sulfonylurea and insulin), treatment with colesevelam hydrochloride resulted in a statistically significant reduction in HbA1c of 0.5% compared to placebo. Similar placebo-corrected reductions in HbA1c occurred in patients who received colesevelam hydrochloride in combination with metformin, sulfonylurea, or insulin monotherapy or combinations of these therapies with other anti-diabetic agents. In the pioglitazone trial, treatment with colesevelam hydrochloride resulted in a statistically significant reduction in HbA1c of 0.32% compared to placebo. In the metformin, pioglitazone, and sulfonylurea trials, treatment with colesevelam hydrochloride also resulted in statistically significant reductions in FPG of at least 14 mg/dL compared to placebo.
Colesevelam hydrochloride had consistent effects on HbA1c across subgroups of age, gender, race, body mass index, and baseline HbA1c. Colesevelam hydrochloride's effects on HbA1c were also similar for the two dosing regimens (3 tablets with lunch and with dinner or 6 tablets with dinner alone).
The mean baseline LDL-C was 104 mg/dL in the metformin study (range 32 mg/dL to 214 mg/dL), 107 mg/dL in the pioglitazone study (range 48 mg/dL to 263 mg/dL), 106 mg/dL in the sulfonylurea study (range 41 mg/dL to 264 mg/dL), 102 mg/dL in the insulin study (range 35 mg/dL to 204 mg/dL). In these trials, colesevelam hydrochloride treatment was associated with a 12% to 16% reduction in LDL-C levels. The percentage decreases in LDL-C were of similar magnitude to those observed in patients with primary hyperlipidemia. Colesevelam hydrochloride treatment was associated with statistically significant increases in TG levels in the studies of patients on insulin, patients on a sulfonylurea, and patients on pioglitazone but not in the study of patients on metformin. The clinical significance of these increases is unknown. Colesevelam hydrochloride is contraindicated in patients with TG levels > 500 mg/dL [see Contraindications (4)] , and periodic monitoring of lipid parameters including TG is recommended [see Warnings and Precautions (5.1) and Adverse Reactions (6.1)] .
Body weight did not significantly increase from baseline with colesevelam hydrochloride therapy, compared with placebo, in any of the add-on combination diabetes studies.
Add-on Combination Therapy with Metformin
Colesevelam hydrochloride 3.8 g/day or placebo was added to background anti-diabetic therapy in a 26-week trial of 316 patients already receiving treatment with metformin alone (N = 159) or metformin in combination with other oral agents (N = 157). A total of 60% of these patients were receiving ≥ 1,500 mg/day of metformin. In combination with metformin, colesevelam hydrochloride resulted in statistically significant placebo-corrected reductions in HbA1c and FPG (Table 12). Colesevelam hydrochloride also reduced TC, LDL-C, Apo B, and non-HDL-C (Table 13). The mean percent change in serum LDL-C levels with colesevelam hydrochloride compared to placebo was -16% among statin users and statin non-users; the median percent change in serum TG levels with colesevelam hydrochloride compared to placebo was -2% among statin users and 10% among statin non-users. The mean change in body weight was -0.5 kg for colesevelam hydrochloride and -0.3 kg for placebo.
| Total Patient Population | Metformin Alone | Metformin in Combination with Other Oral Anti-diabetic Agents | ||||
| Colesevelam Hydrochloride 3.8 g/day | Placebo | Colesevelam Hydrochloride 3.8 g/day | Placebo | Colesevelam Hydrochloride 3.8 g/day | Placebo | |
| HbA1c (%), Mean | ||||||
| N | 148 | 152 | 79 | 76 | 69 | 76 |
| Baseline | 8.1 | 8.1 | 8.2 | 8.2 | 8.1 | 8.0 |
| Change from baseline
Least-squares mean change calculated from an Analysis of Covariance model
|
-0.4 | 0.2 | -0.4 | 0.0 | -0.4 | 0.3 |
| Treatment difference (p-value) | -0.5 (p < 0.001) | -0.5 (p = 0.002) | -0.6 (p < 0.001) | |||
| FPG (mg/dL), Mean | ||||||
| N | 149 | 152 | 79 | 76 | 70 | 76 |
| Baseline | 178 | 174 | 184 | 180 | 171 | 168 |
| Change from baseline* | -3 | 11 | -7 | 8 | 0 | 13 |
| Treatment difference (p-value) | -14 (p = 0.01) | -14 (p = 0.07) | -14 (p = 0.10) |
| Dose/Day |
N
The number of patients with analyzable data, i.e., a baseline and post-treatment value (last observation carried forward), varied slightly among different parameters. The N given represents the smallest number of patients included in the analysis for any parameter.
|
TC | LDL-C | Apo B | HDL-C | Non-HDL- C |
TG
Median % change from baseline
|
| Total Patient Population | |||||||
| Colesevelam Hydrochloride 3.8 g | 125 | -4
p < 0.001 for lipid parameters compared to placebo (This more stringent criterion for statistical significance accounts for multiplicity testing of the lipid parameters, which were secondary endpoints in the diabetes trials.)
|
-12 ‡ | -4 ‡ | 1 | -6 ‡ | 12 |
| Placebo | 126 | 3 | 4 | 4 | 0 | 5 | 7 |
| Metformin Alone | |||||||
| Colesevelam Hydrochloride 3.8 g | 66 | -3 | -9 | -2 | 1 | -4 | 15 |
| Placebo | 61 | 2 | 0 | 1 | -2 | 4 | 8 |
| Metformin in Combination with Other Oral Anti-diabetic Agents | |||||||
| Colesevelam Hydrochloride 3.8 g | 59 | -6 ‡ | -15 ‡ | -6 ‡ | 1 | -7 ‡ | 8 |
| Placebo | 65 | 4 | 7 | 7 | 2 | 6 | 5 |
Add-on Combination Therapy with Pioglitazone
Colesevelam hydrochloride 3.8 g/day or placebo was added to background anti-diabetic therapy in a 24-week trial of 562 patients already receiving treatment with pioglitazone alone (N = 51) or pioglitazone in combination with other oral agents (N = 511). Of these, most were on dual therapy with metformin (N = 298) or triple therapy with metformin and a sulfonylurea (N = 139). In combination with pioglitazone-based therapy, colesevelam hydrochloride resulted in statistically significant reductions in HbA1c and FPG compared to placebo (Table 14). Colesevelam hydrochloride also reduced TC, LDL-C, Apo B, and non-HDL-C but increased serum TG (Table 15). The mean change in body weight was 0.8 kg for colesevelam hydrochloride and 0.4 kg for placebo.
|
Colesevelam Hydrochloride
3.8 g/day |
Placebo | |
| HbA1c (%), Mean | ||
| N | 271 | 276 |
| Baseline | 8.2 | 8.1 |
| Change from baseline
Least-squares mean change calculated from an Analysis of Covariance model
|
-0.34 | -0.02 |
| Treatment difference (p-value) | -0.32 (0.0001) | |
| FPG (mg/dL), Mean | ||
| N | 268 | 270 |
| Baseline | 155 | 157 |
| Change from baseline * | -4.8 | +9.9 |
| Treatment difference (p-value) | -14.7 (< 0.0001) |
| Dose/Day |
N
The N given represents the smallest number of patients included in the analysis for any parameter.
|
TC | LDL-C | Apo B | HDL-C | Non-HDL- C |
TG
Median % change from baseline
|
| Total Patient Cohort | |||||||
| Colesevelam Hydrochloride 3.8 g | 262 | -3
p < 0.001 for lipid parameters compared to placebo
|
-9 ‡ | -5 ‡ | +3 | -5 ‡ | +14 ‡ |
| Placebo | 262 | +3 | +7 | +4 | +1 | +5 | +2 |
Add-on Combination Therapy with Sulfonylurea
Colesevelam hydrochloride 3.8 g/day or placebo was added to background anti-diabetic therapy in a 26-week trial of 460 patients already treated with sulfonylurea alone (N = 156) or sulfonylurea in combination with other oral agents (N = 304). A total of 72% of these patients were receiving at least half-maximal doses of sulfonylurea therapy. In combination with a sulfonylurea, colesevelam hydrochloride resulted in statistically significant placebo-corrected reductions in HbA1c and FPG (Table 16). Colesevelam hydrochloride also reduced TC, LDL-C, Apo B, and non-HDL-C, but increased serum TG (Table 17). The mean percent change in serum LDL-C levels with colesevelam hydrochloride compared to placebo was -18% among statin users and -15% among statin non-users; the median percent increase in serum TG with colesevelam hydrochloride compared to placebo was 29% among statin users and 9% among statin non-users. The mean change in body weight was 0.0 kg for colesevelam hydrochloride and -0.4 kg for placebo.
| Total Patient Population | Sulfonylurea Alone | Sulfonylurea in Combination with Other Oral Anti-diabetic Agents | ||||
| Colesevelam Hydrochloride 3.8 g/day | Placebo | Colesevelam Hydrochloride 3.8 g/day | Placebo | Colesevelam Hydrochloride 3.8 g/day | Placebo | |
| HbA1c (%), Mean | ||||||
| n | 218 | 218 | 69 | 80 | 149 | 138 |
| Baseline | 8.2 | 8.3 | 8.2 | 8.4 | 8.2 | 8.3 |
| Change from baseline
Least-squares mean change calculated from an Analysis of Covariance model
|
-0.3 | 0.2 | -0.3 | 0.5 | -0.4 | 0.0 |
| Treatment difference (p-value) | -0.5 (p < 0.001) | -0.8 (p < 0.001) | -0.4 (p < 0.001) | |||
| FPG (mg/dL), Mean | ||||||
| n | 218 | 217 | 70 | 80 | 148 | 137 |
| Baseline | 177 | 181 | 181 | 186 | 175 | 178 |
| Change from baseline * | -4 | 10 | 3 | 15 | -11 | 4 |
| Treatment difference (p-value) | -14 (p = 0.009) | -12 (p = 0.18) | -14 (p = 0.03) |
| Dose/Day |
N
The number of patients with analyzable data, i.e., a baseline and post-treatment value (last observation carried forward), varied slightly among different parameters. The N given represents the smallest number of patients included in the analysis for any parameter.
|
TC | LDL-C | Apo B | HDL-C | Non-HDL- C |
TG
Median % change from baseline
|
| Total Patient Population | |||||||
| Colesevelam Hydrochloride 3.8 g | 186 | -5
p < 0.001 for lipid parameters compared to placebo (This more stringent criterion for statistical significance accounts for multiplicity testing of the lipid parameters, which were secondary endpoints in the diabetes trials.)
|
-16 ‡ | -6 ‡ | 1 | -6 ‡ | 20 ‡ |
| Placebo | 193 | 0 | 1 | 1 | 0 | 1 | 1 |
| Sulfonylurea Alone | |||||||
| Colesevelam Hydrochloride 3.8 g | 57 | -5 | -14 ‡ | -5 | -1 | -6 | 17 |
| Placebo | 68 | 0 | 1 | 1 | 1 | 0 | -1 |
| Sulfonylurea in Combination with Other Oral Anti-diabetic Agents | |||||||
| Colesevelam Hydrochloride 3.8 g | 129 | -5 | -18 ‡ | -7 ‡ | 1 | -6 | 21 ‡ |
| Placebo | 125 | 0 | 0 | 1 | 0 | 1 | 2 |
Add-on Combination Therapy with Insulin
Colesevelam hydrochloride 3.8 g/day or placebo was added to background anti-diabetic therapy in a 16-week trial of 287 patients already treated with insulin alone (N = 116) or insulin in combination with oral agents (N = 171). At baseline, the median daily insulin dose was 70 units in the colesevelam hydrochloride group and 65 units in the placebo group. In combination with insulin, colesevelam hydrochloride resulted in a statistically significant placebo‑corrected reduction in HbA1c (Table 18). Colesevelam hydrochloride also reduced LDL-C and Apo B, but increased serum TG (Table 19). The mean percent change in serum LDL-C levels with colesevelam hydrochloride compared to placebo was -13% among statin users and statin non-users; the median percent increase in serum TG levels with colesevelam hydrochloride compared to placebo was 24% among statin users and 17% among statin non-users. The mean change in body weight was 0.6 kg for colesevelam hydrochloride and 0.2 kg for placebo.
| Total Patient Population | Insulin Alone | Insulin in Combination with Oral Anti-diabetic Agents | ||||
| Colesevelam Hydrochloride 3.8 g/day | Placebo | Colesevelam Hydrochloride 3.8 g/day | Placebo | Colesevelam Hydrochloride 3.8 g/day | Placebo | |
| HbA1c (%), Mean | ||||||
| n | 144 | 136 | 54 | 55 | 90 | 81 |
| Baseline | 8.3 | 8.2 | 8.2 | 8.3 | 8.3 | 8.2 |
| Change from baseline
Least-squares mean change calculated from an Analysis of Covariance model
|
-0.4 | 0.1 | -0.4 | 0.2 | -0.4 | 0.0 |
| Treatment difference (p-value) | -0.5 (p < 0.001) | -0.6 (p < 0.001) | -0.4 (p < 0.001) | |||
| FPG (mg/dL), Mean | ||||||
| n | 144 | 136 | 54 | 55 | 90 | 81 |
| Baseline | 165 | 151 | 165 | 163 | 165 | 143 |
| Change from baseline * | 2 | 16 | 8 | 17 | -4 | 14 |
| Treatment difference (p-value) | -15 (p = 0.08) | -9 (p = 0.51) | -18 (p = 0.09) |
| Dose/Day |
N
The number of patients with analyzable data, i.e., a baseline and post-treatment value (last observation carried forward), varied slightly among different parameters. The N given represents the smallest number of patients included in the analysis for any parameter.
|
TC | LDL-C | Apo B | HDL-C | Non-HDL- C |
TG
Median % change from baseline
|
| Total Patient Cohort | |||||||
| Colesevelam Hydrochloride 3.8 g | 129 | -3 | -12
p < 0.001 for lipid parameters compared to placebo (This more stringent criterion for statistical significance accounts for multiplicity testing of the lipid parameters, which were secondary endpoints in the diabetes trials.)
|
-4 | -1 | -3 | 23 ‡ |
| Placebo | 121 | 1 | 1 | 1 | 0 | 1 | 0 |
| Insulin Alone | |||||||
| Colesevelam Hydrochloride 3.8 g | 46 | -3 | -12 | -5 | 0 | -3 | 19 |
| Placebo | 48 | 2 | 4 | 2 | 3 | 2 | -2 |
| Insulin in Combination with Oral Anti-diabetic Agents | |||||||
| Colesevelam Hydrochloride 3.8 g | 83 | -4 | -13 | -4 | -1 | -3 | 25 ‡ |
| Placebo | 73 | -1 | -3 | 0 | -1 | -1 | 2 |
6.2 Post Marketing Experience
The following additional adverse reactions have been identified during post-approval use of colesevelam hydrochloride. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
6.1 Clinical Studies Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in clinical studies of another drug and may not reflect the rates observed in practice.
5.2 Gastrointestinal Obstruction
Postmarketing cases of bowel obstruction have occurred with colesevelam hydrochloride [see Adverse Reactions (6.2)] . Because of its constipating effects, colesevelam hydrochloride is not recommended in patients with gastroparesis, other gastrointestinal motility disorders, and in those who have had major gastrointestinal tract surgery and who may be at risk for bowel obstruction. Colesevelam hydrochloride is contraindicated in patients with a history of bowel obstruction [see Contraindications (4)] . Instruct patients to promptly discontinue colesevelam hydrochloride and seek medical attention if severe abdominal pain or severe constipation occurs.
Because of the tablet size, colesevelam hydrochloride tablets can cause dysphagia or esophageal obstruction. For patients with difficulty swallowing tablets, use colesevelam hydrochloride for oral suspension.
16 How Supplied/storage and Handling
Colesevelam hydrochloride tablets, 625 mg are supplied as white to off-white, oval-shaped, film-coated tablets, printed with “COL” on one side with black ink and are available as follows:
- Bottles of 180 - NDC 69452-158-25
5.1 Hypertriglyceridemia and Pancreatitis
Colesevelam hydrochloride, like other bile acid sequestrants, can increase serum TG concentrations. Hypertriglyceridemia can cause acute pancreatitis.
Colesevelam hydrochloride had effects on serum TG (median increase 5% compared to placebo) in trials of patients with primary hyperlipidemia.
In trials in patients with type 2 diabetes, greater increases in TG levels occurred when colesevelam hydrochloride was used as monotherapy (median increase 9.7% compared to placebo) and when colesevelam hydrochloride was used in combination with pioglitazone (median increase 11% compared to placebo in combination with pioglitazone), sulfonylureas (median increase 18% compared to placebo in combination with sulfonylureas), and insulin (median increase 22% compared to placebo in combination with insulin) [see Adverse Reactions (6.1)].
Obtain lipid parameters, including TG levels, before starting colesevelam hydrochloride and periodically thereafter. Colesevelam hydrochloride is contraindicated in patients with TG levels > 500 mg/dL or patients with a history of hypertriglyceridemia-induced pancreatitis [see Contraindications (4)] . Patients with TG levels greater than 300 mg/dL could have greater increases in serum TG levels with colesevelam hydrochloride and may require additional TG monitoring. Instruct patients to discontinue colesevelam hydrochloride and seek prompt medical attention if the symptoms of acute pancreatitis occur (e.g., severe abdominal pain with or without nausea and vomiting). Discontinue colesevelam hydrochloride if TG levels exceed 500 mg/dL [see Adverse Reactions (6.1)] .
5.3 Vitamin K Or Fat Soluble Vitamin Deficiencies
Colesevelam hydrochloride may decrease the absorption of fat-soluble vitamins A, D, E, and K. Patients with a susceptibility to deficiencies of vitamin K (e.g., patients on warfarin, patients with malabsorption syndromes) or other fat-soluble vitamins may be at increased risk when taking colesevelam hydrochloride.
Patients on oral vitamin supplementation should take their vitamins at least 4 hours prior to colesevelam hydrochloride [see Drug Interactions (7.1)].
2.3 Important Dosing Information for Primary Hyperlipidemia
Colesevelam hydrochloride can be dosed at the same time as a statin, or colesevelam hydrochloride and the statin can be dosed apart .Monitor lipid levels within 4 weeks to 6 weeks after initiation of colesevelam hydrochloride.
Principal Display Panel 625 Mg, 180 Tablets (india Mfg Site)
BIONPHARMA
NDC 69452-158-25
Colesevelam Hydrochloride Tablets
625 mg
Rx only
180 Tablets
2.1 Testing Prior to Initiation Of colesevelam Hydrochloride Tablets
Obtain lipid parameters, including triglyceride (TG) levels, before starting colesevelam hydrochloride tablets. Colesevelam hydrochloride is contraindicated in patients with TG levels > 500 mg/dL [see Contraindications (4) and Warnings and Precautions (5.1)] .
2.2 Recommended Dosage in Primary Hyperlipidemia and Type 2 Diabetes Mellitus
The recommended dosage of colesevelam hydrochloride for adults and for boys and post-menarchal girls aged 10 years to 17 years with primary hyperlipidemia is 3.75 grams daily. The recommended dosage of colesevelam hydrochloride for adults with type 2 diabetes mellitus is 3.75 grams daily. Colesevelam hydrochloride tablets should be taken as follows:
7.1 colesevelam Hydrochloride Drug Interactions That Decrease the Exposure of the Concomitant Medication
Table 4 includes a list of drugs that decrease exposure of the concomitant medication when administered concomitantly with colesevelam hydrochloride and instructions for preventing or managing them.
| Drugs with a Narrow Therapeutic Index | |
|---|---|
| Clinical Impact: | Concomitant use with colesevelam hydrochloride may decrease the exposure of the narrow therapeutic index drug. In vivodrug interactions studies showed a decrease in exposure of cyclosporine when co-administered with colesevelam hydrochloride [see Clinical Pharmacology (12.3)]. |
| Intervention: | Administer the narrow therapeutic index drug at least 4 hours prior to colesevelam hydrochloride. Monitor drug levels when appropriate. |
| Examples: | Cyclosporine |
| Phenytoin | |
| Clinical Impact: | There have been postmarketing reports of increased seizure activity or decreased phenytoin levels in patients receiving phenytoin [see Adverse Reactions (6.2)]. |
| Intervention: | Administer phenytoin 4 hours prior to colesevelam hydrochloride. |
| Thyroid Hormone Replacement Therapy | |
| Clinical Impact: | In vivodrug interactions studies showed a decrease in exposure of levothyroxine when co-administered with colesevelam hydrochloride [see Clinical Pharmacology (12.3)] .There have been postmarketing reports of elevated thyroid-stimulating hormone (TSH) in patients receiving thyroid hormone replacement therapy [see Adverse Reactions (6.2)] . |
| Intervention: | Administer thyroid hormone replacement therapy 4 hours prior to colesevelam hydrochloride. |
| Warfarin | |
| Clinical Impact: | There have been postmarketing reports of reduced INR in patients receiving warfarin therapy [see Adverse Reactions (6.2)]. |
| Intervention: | Monitor INR frequently during colesevelam hydrochloride initiation then periodically thereafter. |
| Oral Contraceptives Containing Ethinyl Estradiol and Norethindrone | |
| Clinical Impact: | In vivodrug interactions studies showed a decrease in exposure of ethinyl estradiol and norethindrone when co-administered with colesevelam hydrochloride [see Clinical Pharmacology (12.3)]. |
| Intervention: | Administer oral contraceptives containing ethinyl estradiol and norethindrone 4 hours prior to colesevelam hydrochloride. |
| Olmesartan Medoxomil | |
| Clinical Impact: | In vivodrug interactions studies showed a decrease in olmesartan medoxomil when co-administered with colesevelam hydrochloride [see Clinical Pharmacology (12.3)]. |
| Intervention: | Administer olmesartan medoxomil 4 hours prior to colesevelam hydrochloride. |
| Sulfonylureas | |
| Clinical Impact: | In vivodrug interactions studies showed a decrease in sulfonylureas when co-administered with colesevelam hydrochloride [see Clinical Pharmacology (12.3)]. |
| Intervention: | Administer sulfonylureas 4 hours prior to colesevelam hydrochloride. |
| Examples: | Glimepiride, glipizide, and glyburide |
| Oral Vitamin Supplements | |
| Clinical Impact: | Colesevelam hydrochloride may decrease the absorption of fat-soluble vitamins A, D, E, and K [see Warnings and Precautions (5.3)]. |
| Intervention: | Patients on oral vitamin supplementation should take their vitamins at least 4 hours prior to colesevelam hydrochloride. |
7.2 colesevelam Hydrochloride Drug Interactions That Increase the Exposure of the Concomitant Medication
| Metformin Extended-Release (ER) | |
|---|---|
| Clinical Impact: | In vivodrug interactions studies showed an increase in metformin extended release (ER) when co-administered with colesevelam hydrochloride [see Clinical Pharmacology (12.3)]. |
| Intervention: | Monitor patients' glycemic control. |
Structured Label Content
Section 42229-5 (42229-5)
Take 6 tablets once daily or 3 tablets twice daily. Due to tablet size, colesevelam hydrochloride for oral suspension is recommended for use in the pediatric population.
Section 44425-7 (44425-7)
Store at 25°C (77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Protect from moisture. Brief exposure to 40°C (104°F) does not adversely affect colesevelam hydrochloride tablets.
10 Overdosage (10 OVERDOSAGE)
Colesevelam hydrochloride is not absorbed and the risk of systemic toxicity is low. Excessive doses of colesevelam hydrochloride may cause more severe local gastrointestinal effects (e.g., constipation).
11 Description (11 DESCRIPTION)
Colesevelam hydrochloride is a non-absorbed, polymeric, lipid-lowering and glucose-lowering agent for oral administration. Colesevelam hydrochloride is a high-capacity bile acid-binding molecule.
Colesevelam hydrochloride is poly(allylamine hydrochloride) cross-linked with epichlorohydrin and alkylated with 1-bromodecane and (6-bromohexyl)-trimethylammonium bromide. The chemical name (IUPAC) of colesevelam hydrochloride is allylamine polymer with 1-chloro-2,3-epoxypropane, [6 (allylamino)-hexyl]trimethylammonium chloride and N-allyldecylamine, hydrochloride. The chemical structure of colesevelam hydrochloride is represented by the following formula:
wherein (a) represents allyl amine monomer units that have not been alkylated by either of the 1‑bromodecane or (6-bromohexyl)-trimethylammonium bromide alkylating agents or cross-linked by epichlorohydrin; (b) represents allyl amine units that have undergone cross-linking with epichlorohydrin; (c) represents allyl amine units that have been alkylated with a decyl group; (d) represents allyl amine units that have been alkylated with a (6-trimethylammonium) hexyl group, and m represents a number ≥ 100 to indicate an extended polymer network. A small amount of the amines are dialkylated and are not depicted in the formula above. No regular order of the groups is implied by the structure; cross-linking and alkylation are expected to occur randomly along the polymer chains. A large amount of the amines are protonated. The polymer is depicted in the hydrochloride form; a small amount of the halides are bromide. Colesevelam hydrochloride is hydrophilic and insoluble in water.
Colesevelam hydrochloride tablets are white to off-white, oval-shaped, film-coated tablets, printed with “COL” on one side with black ink containing 625 mg colesevelam hydrochloride. In addition, each tablet contains the following inactive ingredients: magnesium stearate, microcrystalline cellulose, silicon dioxide. The tablets are imprinted using a water-soluble black ink (ferrosoferric oxide, hypromellose, and propylene glycol). The coating material contains polyethylene glycol, polysorbate 80 and talc (approximately 5 calories per 6 tablets).
8.4 Pediatric Use
Primary Hyperlipidemia
The safety and effectiveness of colesevelam hydrochloride to reduce LDL-C levels in boys and post-menarchal girls 10 years to 17 years of age with HeFH who are unable to reach LDL-C target levels despite an adequate trial of dietary therapy and lifestyle modification have been established. Use of colesevelam hydrochloride for this indication is supported by a study in 129 colesevelam hydrochloride-treated pediatric patients aged 10 years to 17 years with HeFH [see Clinical Studies (14.1)] . Adverse reactions commonly observed in pediatric patients compared to placebo, but not in adults, included headache (3.9%), creatine phosphokinase increase (2.3%), and vomiting (2.3%) [see Adverse Reactions (6.1)] . There were no significant effects on fat-soluble vitamin levels or clotting factors in the adolescent boys or girls relative to placebo. Due to colesevelam hydrochloride tablet size, colesevelam hydrochloride for oral suspension is recommended for use in the pediatric population [see Dosage and Administration (2.2, 2.4)]. The safety and effectiveness of colesevelam hydrochloride in pediatric patients with HeFH less than 10 years of age or in premenarchal females have not been established.
Type 2 Diabetes Mellitus
The safety and effectiveness of colesevelam hydrochloride to improve glycemic control in pediatric patients with type 2 diabetes mellitus have not been established. Effectiveness was not demonstrated in a 6-month, adequate and well-controlled study conducted in 141 colesevelam hydrochloride-treated pediatric patients aged 10 years to 17 years with type 2 diabetes mellitus.
4 Contraindications (4 CONTRAINDICATIONS)
Colesevelam hydrochloride is contraindicated in patients with:
- Serum TG concentrations > 500 mg/dL [see Warnings and Precautions (5.1)]
- History of hypertriglyceridemia-induced pancreatitis [see Warnings and Precautions (5.1)]
- A history of bowel obstruction [see Warnings and Precautions (5.2)]
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following important adverse reactions are described below and elsewhere in the labeling:
- Hypertriglyceridemia and Pancreatitis [see Warnings and Precautions (5.1)]
- Gastrointestinal Obstruction [see Warnings and Precautions (5.2)]
- Vitamin K or Fat-Soluble Vitamin Deficiencies [see Warnings and Precautions (5.3)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Concomitant use with colesevelam hydrochloride may decrease the exposure of the following drugs:Drugs with a narrow therapeutic index (e.g., cyclosporine), phenytoin, thyroid hormone replacement therapy, warfarin, oral contraceptives containing ethinyl estradiol and norethindrone, olmesartan medoxomil, and sulfonylureas (glimepiride, glipizide, glyburide). Administer these drugs 4 hours prior to colesevelam hydrochloride. For patients on warfarin, monitor International Normalized Ratio (INR) frequently during initiation then periodically ( 7.1).
Concomitant use with colesevelam hydrochloride may increase the exposure of the following drugs:Metformin extended release. Monitor patients' glycemic control ( 7.2).
8.6 Renal Impairment
Type 2 Diabetes Mellitus
Of the 2,048 patients enrolled in the six diabetes studies, 807 (39%) had mild renal insufficiency (creatinine clearance [CrCl] 50 mL/min to < 80 mL/min), 61 (3%) had moderate renal insufficiency (CrCl 30 mL/min to < 50 mL/min), and none had severe renal insufficiency (CrCl < 30 mL/min), as estimated from baseline serum creatinine using the Modification of Diet in Renal Disease (MDRD) equation. No overall differences in safety or effectiveness were observed between patients with CrCl < 50 mL/min (n = 53) and those with a CrCl ≥ 50 mL/min (n = 1,075) in the add-on to metformin, sulfonylureas, and insulin diabetes studies. In the monotherapy study and add-on to pioglitazone study, only 3 and 5 patients, respectively, had moderate renal insufficiency.
12.2 Pharmacodynamics
A maximum therapeutic response to the lipid-lowering effects of colesevelam hydrochloride was achieved within 2 weeks and was maintained during long-term therapy. In the diabetes clinical studies, a therapeutic response to colesevelam hydrochloride, as reflected by a reduction in HbA1c, was initially noted following 4 weeks to 6 weeks of treatment and reached maximal or near-maximal effect after 12 weeks to 18 weeks of treatment.
5.4 Drug Interactions
Colesevelam hydrochloride reduces gastrointestinal absorption of some drugs. Administer drugs with a known interaction at least 4 hours prior to colesevelam hydrochloride [see Drug Interactions (7)] .
Due to the potential for decreased absorption of other drugs that have not been tested for interaction, especially those with a narrow therapeutic index, consider administering at least 4 hours prior to colesevelam hydrochloride [see Clinical Pharmacology (12.3)].
1.3 Limitations of Use
- Colesevelam hydrochloride should not be used for the treatment of type 1 diabetes or for the treatment of diabetic ketoacidosis.
- Colesevelam hydrochloride has not been studied in Fredrickson Type I, III, IV, and V dyslipidemias.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Colesevelam hydrochloride is a bile acid sequestrant indicated as an adjunct to diet and exercise to:
- reduce elevated low-density lipoprotein cholesterol (LDL-C) in adults with primary hyperlipidemia ( 1.1).
- reduce LDL-C levels in boys and post-menarchal girls, 10 years to 17 years of age, with heterozygous familial hypercholesterolemia (HeFH), unable to reach LDL-C target levels despite an adequate trial of diet and lifestyle modification ( 1.1).
- improve glycemic control in adults with type 2 diabetes mellitus ( 1.2).
Limitations of Use ( 1.3):
- Do not use for treatment of type 1 diabetes or for diabetic ketoacidosis.
- Not studied in Fredrickson Type I, III, IV, and V dyslipidemias.
1.1 Primary Hyperlipidemia
Colesevelam hydrochloride tablets are indicated as an adjunct to diet and exercise to reduce elevated low-density lipoprotein cholesterol (LDL-C) in adults with primary hyperlipidemia.
Colesevelam hydrochloride tablets are indicated to reduce LDL-C levels in boys and post-menarchal girls, 10 years to 17 years of age, with heterozygous familial hypercholesterolemia (HeFH) who are unable to reach LDL-C target levels despite an adequate trial of dietary therapy and lifestyle modification.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypertriglyceridemia and Pancreatitis:Colesevelam hydrochloride can increase TG. Hypertriglyceridemia can cause acute pancreatitis. Monitor lipids, including TG. Instruct patients to discontinue colesevelam hydrochloride and seek prompt medical attention if the symptoms of acute pancreatitis occur ( 5.1).
- Gastrointestinal Obstruction:Cases of bowel obstruction have occurred. Colesevelam hydrochloride is not recommended in patients with gastroparesis, other gastrointestinal motility disorders, and in those who have had major gastrointestinal tract surgery and who may be at risk for bowel obstruction ( 5.2).
- Vitamin K or Fat-Soluble Vitamin Deficiencies : Colesevelam hydrochloride may decrease absorption of fat-soluble vitamins. Patients with a susceptibility to deficiencies of vitamin K (e.g., patients on warfarin, patients with malabsorption syndromes) or other fat-soluble vitamins may be at increased risk. Patients on oral vitamin supplementation should take their vitamins at least 4 hours prior to colesevelam hydrochloride ( 5.3).
- Drug Interactions:Due to the potential for decreased absorption of other drugs that have not been tested for interaction, consider administering at least 4 hours prior to colesevelam hydrochloride ( 5.4, 7, 12.3).
14.1 Primary Hyperlipidemia
Colesevelam hydrochloride reduces total cholesterol (TC), LDL-C, apolipoprotein B (Apo B), and non-high-density lipoprotein cholesterol (non-HDL-C) when administered alone or in combination with a statin in patients with primary hyperlipidemia. Approximately 1,600 patients were studied in 9 clinical trials with treatment durations ranging from 4 weeks to 50 weeks. With the exception of one open-label, uncontrolled, long-term extension study, all studies were multicenter, randomized, double-blind, and placebo-controlled. A maximum therapeutic response to colesevelam hydrochloride was achieved within 2 weeks and was maintained during long-term therapy.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Obtain lipid parameters, including serum triglyceride (TG) levels, before starting colesevelam hydrochloride tablets ( 2.1)
- The recommended dosage for adults and for boys and post-menarchal girls aged 10 years to 17 years with primary hyperlipidemia is 3.75 grams daily. The recommended dosage for adults with type 2 diabetes mellitus is 3.75 grams daily. Colesevelam hydrochloride tablets should be taken as follows ( 2.2, 2.4):
Take 6 tablets once daily or 3 tablets twice daily with a meal and liquid.
1.2 Type 2 Diabetes Mellitus
Colesevelam hydrochloride tablets are indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- Tablets: 625 mg tablets are white to off-white, oval-shaped, film-coated tablets and printed with “COL” on one side with black ink.
14.2 Type 2 Diabetes Mellitus
Colesevelam hydrochloride has been studied as monotherapy and in combination with metformin, pioglitazone, sulfonylureas, and insulin. In these studies, colesevelam hydrochloride and placebo were administered either as 3 tablets twice daily with lunch and dinner or as 6 tablets with dinner alone.
Monotherapy
The efficacy of colesevelam hydrochloride 3.8 g/day as anti-diabetes monotherapy was evaluated in a randomized double-blind, placebo-controlled trial involving 357 patients (176 colesevelam hydrochloride and 181 placebo) with type 2 diabetes mellitus who were treatment-naïve or had not received antihyperglycemic medication within 3 months prior to the start of the study. Statin use at baseline was reported in 13% of the colesevelam hydrochloride-treated patients and 16% of the placebo‑treated patients.
Colesevelam hydrochloride resulted in a statistically significant reduction in HbA1c of 0.27% compared to placebo (Table 10).
The mean baseline LDL-C was 121 mg/dL in the monotherapy trial. Colesevelam hydrochloride treatment resulted in a placebo-corrected 11% reduction in LDL-C. Colesevelam hydrochloride treatment also reduced serum TC, ApoB, and non-HDL-C (Table 11). The mean change in body weight was -0.6 kg for colesevelam hydrochloride and ‑0.7 kg for placebo treatment groups.
|
Colesevelam Hydrochloride
3.8 g/day |
Placebo | |
| HbA1c (%), Mean | ||
| N | 175 | 169 |
| Baseline | 8.25 | 8.17 |
| Change from baseline
Least-squares mean change calculated from an Analysis of Covariance model
|
-0.26 | 0.01 |
| Treatment difference (p-value) | -0.27 (p = 0.013) | |
| FPG (mg/dL), Mean | ||
| N | 172 | 166 |
| Baseline | 172 | 168 |
| Change from baseline * | -4.6 | 5.7 |
| Treatment difference (p-value) | -10.3 (p = 0.037
Nominal p = value, not controlled for multiplicity testing )
|
FPG = fasting plasma glucose
| Dose/Day |
N
The number of patients with analyzable data, i.e., a baseline and post-treatment value (last observation carried forward), varied slightly among different parameters. The N given represents the smallest number of patients included in the analysis for any parameter.
|
TC | LDL-C | Apo B | HDL-C | Non-HDL-C |
TG
Median % change from baseline
|
| Colesevelam Hydrochloride 3.8 g | 162 | -3.3
p < 0.001 for lipid parameters compared to placebo (This more stringent criterion for statistical significance accounts for multiplicity testing of the lipid parameters, which were secondary endpoints in the diabetes trials.)
|
-10.0 ‡ | -5.6 ‡ | 1.7 | -4.4 ‡ | 15.5 |
| Placebo | 160 | 1.8 | 1.2 | 0.9 | -0.1 | 3.0 | 5.8 |
Add-on Combination Therapy
The efficacy of colesevelam hydrochloride 3.8 g/day in patients with type 2 diabetes mellitus was evaluated in 5 double-blind, placebo‑controlled add-on therapy trials involving a total of 1,691 patients with baseline HbA1c 7.5% to 9.5%. Patients were enrolled and maintained on their pre-existing, stable, background anti‑diabetic regimen. Statin use at baseline was reported in 41% of the colesevelam hydrochloride-treated patients and 48% of the placebo-treated patients.
In 3 add-on combination therapy trials (metformin, sulfonylurea and insulin), treatment with colesevelam hydrochloride resulted in a statistically significant reduction in HbA1c of 0.5% compared to placebo. Similar placebo-corrected reductions in HbA1c occurred in patients who received colesevelam hydrochloride in combination with metformin, sulfonylurea, or insulin monotherapy or combinations of these therapies with other anti-diabetic agents. In the pioglitazone trial, treatment with colesevelam hydrochloride resulted in a statistically significant reduction in HbA1c of 0.32% compared to placebo. In the metformin, pioglitazone, and sulfonylurea trials, treatment with colesevelam hydrochloride also resulted in statistically significant reductions in FPG of at least 14 mg/dL compared to placebo.
Colesevelam hydrochloride had consistent effects on HbA1c across subgroups of age, gender, race, body mass index, and baseline HbA1c. Colesevelam hydrochloride's effects on HbA1c were also similar for the two dosing regimens (3 tablets with lunch and with dinner or 6 tablets with dinner alone).
The mean baseline LDL-C was 104 mg/dL in the metformin study (range 32 mg/dL to 214 mg/dL), 107 mg/dL in the pioglitazone study (range 48 mg/dL to 263 mg/dL), 106 mg/dL in the sulfonylurea study (range 41 mg/dL to 264 mg/dL), 102 mg/dL in the insulin study (range 35 mg/dL to 204 mg/dL). In these trials, colesevelam hydrochloride treatment was associated with a 12% to 16% reduction in LDL-C levels. The percentage decreases in LDL-C were of similar magnitude to those observed in patients with primary hyperlipidemia. Colesevelam hydrochloride treatment was associated with statistically significant increases in TG levels in the studies of patients on insulin, patients on a sulfonylurea, and patients on pioglitazone but not in the study of patients on metformin. The clinical significance of these increases is unknown. Colesevelam hydrochloride is contraindicated in patients with TG levels > 500 mg/dL [see Contraindications (4)] , and periodic monitoring of lipid parameters including TG is recommended [see Warnings and Precautions (5.1) and Adverse Reactions (6.1)] .
Body weight did not significantly increase from baseline with colesevelam hydrochloride therapy, compared with placebo, in any of the add-on combination diabetes studies.
Add-on Combination Therapy with Metformin
Colesevelam hydrochloride 3.8 g/day or placebo was added to background anti-diabetic therapy in a 26-week trial of 316 patients already receiving treatment with metformin alone (N = 159) or metformin in combination with other oral agents (N = 157). A total of 60% of these patients were receiving ≥ 1,500 mg/day of metformin. In combination with metformin, colesevelam hydrochloride resulted in statistically significant placebo-corrected reductions in HbA1c and FPG (Table 12). Colesevelam hydrochloride also reduced TC, LDL-C, Apo B, and non-HDL-C (Table 13). The mean percent change in serum LDL-C levels with colesevelam hydrochloride compared to placebo was -16% among statin users and statin non-users; the median percent change in serum TG levels with colesevelam hydrochloride compared to placebo was -2% among statin users and 10% among statin non-users. The mean change in body weight was -0.5 kg for colesevelam hydrochloride and -0.3 kg for placebo.
| Total Patient Population | Metformin Alone | Metformin in Combination with Other Oral Anti-diabetic Agents | ||||
| Colesevelam Hydrochloride 3.8 g/day | Placebo | Colesevelam Hydrochloride 3.8 g/day | Placebo | Colesevelam Hydrochloride 3.8 g/day | Placebo | |
| HbA1c (%), Mean | ||||||
| N | 148 | 152 | 79 | 76 | 69 | 76 |
| Baseline | 8.1 | 8.1 | 8.2 | 8.2 | 8.1 | 8.0 |
| Change from baseline
Least-squares mean change calculated from an Analysis of Covariance model
|
-0.4 | 0.2 | -0.4 | 0.0 | -0.4 | 0.3 |
| Treatment difference (p-value) | -0.5 (p < 0.001) | -0.5 (p = 0.002) | -0.6 (p < 0.001) | |||
| FPG (mg/dL), Mean | ||||||
| N | 149 | 152 | 79 | 76 | 70 | 76 |
| Baseline | 178 | 174 | 184 | 180 | 171 | 168 |
| Change from baseline* | -3 | 11 | -7 | 8 | 0 | 13 |
| Treatment difference (p-value) | -14 (p = 0.01) | -14 (p = 0.07) | -14 (p = 0.10) |
| Dose/Day |
N
The number of patients with analyzable data, i.e., a baseline and post-treatment value (last observation carried forward), varied slightly among different parameters. The N given represents the smallest number of patients included in the analysis for any parameter.
|
TC | LDL-C | Apo B | HDL-C | Non-HDL- C |
TG
Median % change from baseline
|
| Total Patient Population | |||||||
| Colesevelam Hydrochloride 3.8 g | 125 | -4
p < 0.001 for lipid parameters compared to placebo (This more stringent criterion for statistical significance accounts for multiplicity testing of the lipid parameters, which were secondary endpoints in the diabetes trials.)
|
-12 ‡ | -4 ‡ | 1 | -6 ‡ | 12 |
| Placebo | 126 | 3 | 4 | 4 | 0 | 5 | 7 |
| Metformin Alone | |||||||
| Colesevelam Hydrochloride 3.8 g | 66 | -3 | -9 | -2 | 1 | -4 | 15 |
| Placebo | 61 | 2 | 0 | 1 | -2 | 4 | 8 |
| Metformin in Combination with Other Oral Anti-diabetic Agents | |||||||
| Colesevelam Hydrochloride 3.8 g | 59 | -6 ‡ | -15 ‡ | -6 ‡ | 1 | -7 ‡ | 8 |
| Placebo | 65 | 4 | 7 | 7 | 2 | 6 | 5 |
Add-on Combination Therapy with Pioglitazone
Colesevelam hydrochloride 3.8 g/day or placebo was added to background anti-diabetic therapy in a 24-week trial of 562 patients already receiving treatment with pioglitazone alone (N = 51) or pioglitazone in combination with other oral agents (N = 511). Of these, most were on dual therapy with metformin (N = 298) or triple therapy with metformin and a sulfonylurea (N = 139). In combination with pioglitazone-based therapy, colesevelam hydrochloride resulted in statistically significant reductions in HbA1c and FPG compared to placebo (Table 14). Colesevelam hydrochloride also reduced TC, LDL-C, Apo B, and non-HDL-C but increased serum TG (Table 15). The mean change in body weight was 0.8 kg for colesevelam hydrochloride and 0.4 kg for placebo.
|
Colesevelam Hydrochloride
3.8 g/day |
Placebo | |
| HbA1c (%), Mean | ||
| N | 271 | 276 |
| Baseline | 8.2 | 8.1 |
| Change from baseline
Least-squares mean change calculated from an Analysis of Covariance model
|
-0.34 | -0.02 |
| Treatment difference (p-value) | -0.32 (0.0001) | |
| FPG (mg/dL), Mean | ||
| N | 268 | 270 |
| Baseline | 155 | 157 |
| Change from baseline * | -4.8 | +9.9 |
| Treatment difference (p-value) | -14.7 (< 0.0001) |
| Dose/Day |
N
The N given represents the smallest number of patients included in the analysis for any parameter.
|
TC | LDL-C | Apo B | HDL-C | Non-HDL- C |
TG
Median % change from baseline
|
| Total Patient Cohort | |||||||
| Colesevelam Hydrochloride 3.8 g | 262 | -3
p < 0.001 for lipid parameters compared to placebo
|
-9 ‡ | -5 ‡ | +3 | -5 ‡ | +14 ‡ |
| Placebo | 262 | +3 | +7 | +4 | +1 | +5 | +2 |
Add-on Combination Therapy with Sulfonylurea
Colesevelam hydrochloride 3.8 g/day or placebo was added to background anti-diabetic therapy in a 26-week trial of 460 patients already treated with sulfonylurea alone (N = 156) or sulfonylurea in combination with other oral agents (N = 304). A total of 72% of these patients were receiving at least half-maximal doses of sulfonylurea therapy. In combination with a sulfonylurea, colesevelam hydrochloride resulted in statistically significant placebo-corrected reductions in HbA1c and FPG (Table 16). Colesevelam hydrochloride also reduced TC, LDL-C, Apo B, and non-HDL-C, but increased serum TG (Table 17). The mean percent change in serum LDL-C levels with colesevelam hydrochloride compared to placebo was -18% among statin users and -15% among statin non-users; the median percent increase in serum TG with colesevelam hydrochloride compared to placebo was 29% among statin users and 9% among statin non-users. The mean change in body weight was 0.0 kg for colesevelam hydrochloride and -0.4 kg for placebo.
| Total Patient Population | Sulfonylurea Alone | Sulfonylurea in Combination with Other Oral Anti-diabetic Agents | ||||
| Colesevelam Hydrochloride 3.8 g/day | Placebo | Colesevelam Hydrochloride 3.8 g/day | Placebo | Colesevelam Hydrochloride 3.8 g/day | Placebo | |
| HbA1c (%), Mean | ||||||
| n | 218 | 218 | 69 | 80 | 149 | 138 |
| Baseline | 8.2 | 8.3 | 8.2 | 8.4 | 8.2 | 8.3 |
| Change from baseline
Least-squares mean change calculated from an Analysis of Covariance model
|
-0.3 | 0.2 | -0.3 | 0.5 | -0.4 | 0.0 |
| Treatment difference (p-value) | -0.5 (p < 0.001) | -0.8 (p < 0.001) | -0.4 (p < 0.001) | |||
| FPG (mg/dL), Mean | ||||||
| n | 218 | 217 | 70 | 80 | 148 | 137 |
| Baseline | 177 | 181 | 181 | 186 | 175 | 178 |
| Change from baseline * | -4 | 10 | 3 | 15 | -11 | 4 |
| Treatment difference (p-value) | -14 (p = 0.009) | -12 (p = 0.18) | -14 (p = 0.03) |
| Dose/Day |
N
The number of patients with analyzable data, i.e., a baseline and post-treatment value (last observation carried forward), varied slightly among different parameters. The N given represents the smallest number of patients included in the analysis for any parameter.
|
TC | LDL-C | Apo B | HDL-C | Non-HDL- C |
TG
Median % change from baseline
|
| Total Patient Population | |||||||
| Colesevelam Hydrochloride 3.8 g | 186 | -5
p < 0.001 for lipid parameters compared to placebo (This more stringent criterion for statistical significance accounts for multiplicity testing of the lipid parameters, which were secondary endpoints in the diabetes trials.)
|
-16 ‡ | -6 ‡ | 1 | -6 ‡ | 20 ‡ |
| Placebo | 193 | 0 | 1 | 1 | 0 | 1 | 1 |
| Sulfonylurea Alone | |||||||
| Colesevelam Hydrochloride 3.8 g | 57 | -5 | -14 ‡ | -5 | -1 | -6 | 17 |
| Placebo | 68 | 0 | 1 | 1 | 1 | 0 | -1 |
| Sulfonylurea in Combination with Other Oral Anti-diabetic Agents | |||||||
| Colesevelam Hydrochloride 3.8 g | 129 | -5 | -18 ‡ | -7 ‡ | 1 | -6 | 21 ‡ |
| Placebo | 125 | 0 | 0 | 1 | 0 | 1 | 2 |
Add-on Combination Therapy with Insulin
Colesevelam hydrochloride 3.8 g/day or placebo was added to background anti-diabetic therapy in a 16-week trial of 287 patients already treated with insulin alone (N = 116) or insulin in combination with oral agents (N = 171). At baseline, the median daily insulin dose was 70 units in the colesevelam hydrochloride group and 65 units in the placebo group. In combination with insulin, colesevelam hydrochloride resulted in a statistically significant placebo‑corrected reduction in HbA1c (Table 18). Colesevelam hydrochloride also reduced LDL-C and Apo B, but increased serum TG (Table 19). The mean percent change in serum LDL-C levels with colesevelam hydrochloride compared to placebo was -13% among statin users and statin non-users; the median percent increase in serum TG levels with colesevelam hydrochloride compared to placebo was 24% among statin users and 17% among statin non-users. The mean change in body weight was 0.6 kg for colesevelam hydrochloride and 0.2 kg for placebo.
| Total Patient Population | Insulin Alone | Insulin in Combination with Oral Anti-diabetic Agents | ||||
| Colesevelam Hydrochloride 3.8 g/day | Placebo | Colesevelam Hydrochloride 3.8 g/day | Placebo | Colesevelam Hydrochloride 3.8 g/day | Placebo | |
| HbA1c (%), Mean | ||||||
| n | 144 | 136 | 54 | 55 | 90 | 81 |
| Baseline | 8.3 | 8.2 | 8.2 | 8.3 | 8.3 | 8.2 |
| Change from baseline
Least-squares mean change calculated from an Analysis of Covariance model
|
-0.4 | 0.1 | -0.4 | 0.2 | -0.4 | 0.0 |
| Treatment difference (p-value) | -0.5 (p < 0.001) | -0.6 (p < 0.001) | -0.4 (p < 0.001) | |||
| FPG (mg/dL), Mean | ||||||
| n | 144 | 136 | 54 | 55 | 90 | 81 |
| Baseline | 165 | 151 | 165 | 163 | 165 | 143 |
| Change from baseline * | 2 | 16 | 8 | 17 | -4 | 14 |
| Treatment difference (p-value) | -15 (p = 0.08) | -9 (p = 0.51) | -18 (p = 0.09) |
| Dose/Day |
N
The number of patients with analyzable data, i.e., a baseline and post-treatment value (last observation carried forward), varied slightly among different parameters. The N given represents the smallest number of patients included in the analysis for any parameter.
|
TC | LDL-C | Apo B | HDL-C | Non-HDL- C |
TG
Median % change from baseline
|
| Total Patient Cohort | |||||||
| Colesevelam Hydrochloride 3.8 g | 129 | -3 | -12
p < 0.001 for lipid parameters compared to placebo (This more stringent criterion for statistical significance accounts for multiplicity testing of the lipid parameters, which were secondary endpoints in the diabetes trials.)
|
-4 | -1 | -3 | 23 ‡ |
| Placebo | 121 | 1 | 1 | 1 | 0 | 1 | 0 |
| Insulin Alone | |||||||
| Colesevelam Hydrochloride 3.8 g | 46 | -3 | -12 | -5 | 0 | -3 | 19 |
| Placebo | 48 | 2 | 4 | 2 | 3 | 2 | -2 |
| Insulin in Combination with Oral Anti-diabetic Agents | |||||||
| Colesevelam Hydrochloride 3.8 g | 83 | -4 | -13 | -4 | -1 | -3 | 25 ‡ |
| Placebo | 73 | -1 | -3 | 0 | -1 | -1 | 2 |
6.2 Post Marketing Experience (6.2 Post-marketing Experience)
The following additional adverse reactions have been identified during post-approval use of colesevelam hydrochloride. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
6.1 Clinical Studies Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in clinical studies of another drug and may not reflect the rates observed in practice.
5.2 Gastrointestinal Obstruction
Postmarketing cases of bowel obstruction have occurred with colesevelam hydrochloride [see Adverse Reactions (6.2)] . Because of its constipating effects, colesevelam hydrochloride is not recommended in patients with gastroparesis, other gastrointestinal motility disorders, and in those who have had major gastrointestinal tract surgery and who may be at risk for bowel obstruction. Colesevelam hydrochloride is contraindicated in patients with a history of bowel obstruction [see Contraindications (4)] . Instruct patients to promptly discontinue colesevelam hydrochloride and seek medical attention if severe abdominal pain or severe constipation occurs.
Because of the tablet size, colesevelam hydrochloride tablets can cause dysphagia or esophageal obstruction. For patients with difficulty swallowing tablets, use colesevelam hydrochloride for oral suspension.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Colesevelam hydrochloride tablets, 625 mg are supplied as white to off-white, oval-shaped, film-coated tablets, printed with “COL” on one side with black ink and are available as follows:
- Bottles of 180 - NDC 69452-158-25
5.1 Hypertriglyceridemia and Pancreatitis
Colesevelam hydrochloride, like other bile acid sequestrants, can increase serum TG concentrations. Hypertriglyceridemia can cause acute pancreatitis.
Colesevelam hydrochloride had effects on serum TG (median increase 5% compared to placebo) in trials of patients with primary hyperlipidemia.
In trials in patients with type 2 diabetes, greater increases in TG levels occurred when colesevelam hydrochloride was used as monotherapy (median increase 9.7% compared to placebo) and when colesevelam hydrochloride was used in combination with pioglitazone (median increase 11% compared to placebo in combination with pioglitazone), sulfonylureas (median increase 18% compared to placebo in combination with sulfonylureas), and insulin (median increase 22% compared to placebo in combination with insulin) [see Adverse Reactions (6.1)].
Obtain lipid parameters, including TG levels, before starting colesevelam hydrochloride and periodically thereafter. Colesevelam hydrochloride is contraindicated in patients with TG levels > 500 mg/dL or patients with a history of hypertriglyceridemia-induced pancreatitis [see Contraindications (4)] . Patients with TG levels greater than 300 mg/dL could have greater increases in serum TG levels with colesevelam hydrochloride and may require additional TG monitoring. Instruct patients to discontinue colesevelam hydrochloride and seek prompt medical attention if the symptoms of acute pancreatitis occur (e.g., severe abdominal pain with or without nausea and vomiting). Discontinue colesevelam hydrochloride if TG levels exceed 500 mg/dL [see Adverse Reactions (6.1)] .
5.3 Vitamin K Or Fat Soluble Vitamin Deficiencies (5.3 Vitamin K or Fat-Soluble Vitamin Deficiencies)
Colesevelam hydrochloride may decrease the absorption of fat-soluble vitamins A, D, E, and K. Patients with a susceptibility to deficiencies of vitamin K (e.g., patients on warfarin, patients with malabsorption syndromes) or other fat-soluble vitamins may be at increased risk when taking colesevelam hydrochloride.
Patients on oral vitamin supplementation should take their vitamins at least 4 hours prior to colesevelam hydrochloride [see Drug Interactions (7.1)].
2.3 Important Dosing Information for Primary Hyperlipidemia
Colesevelam hydrochloride can be dosed at the same time as a statin, or colesevelam hydrochloride and the statin can be dosed apart .Monitor lipid levels within 4 weeks to 6 weeks after initiation of colesevelam hydrochloride.
Principal Display Panel 625 Mg, 180 Tablets (india Mfg Site) (PRINCIPAL DISPLAY PANEL - 625 mg, 180 Tablets (India Mfg Site))
BIONPHARMA
NDC 69452-158-25
Colesevelam Hydrochloride Tablets
625 mg
Rx only
180 Tablets
2.1 Testing Prior to Initiation Of colesevelam Hydrochloride Tablets (2.1 Testing Prior to Initiation of Colesevelam Hydrochloride Tablets)
Obtain lipid parameters, including triglyceride (TG) levels, before starting colesevelam hydrochloride tablets. Colesevelam hydrochloride is contraindicated in patients with TG levels > 500 mg/dL [see Contraindications (4) and Warnings and Precautions (5.1)] .
2.2 Recommended Dosage in Primary Hyperlipidemia and Type 2 Diabetes Mellitus
The recommended dosage of colesevelam hydrochloride for adults and for boys and post-menarchal girls aged 10 years to 17 years with primary hyperlipidemia is 3.75 grams daily. The recommended dosage of colesevelam hydrochloride for adults with type 2 diabetes mellitus is 3.75 grams daily. Colesevelam hydrochloride tablets should be taken as follows:
7.1 colesevelam Hydrochloride Drug Interactions That Decrease the Exposure of the Concomitant Medication (7.1 Colesevelam Hydrochloride Drug Interactions that Decrease the Exposure of the Concomitant Medication)
Table 4 includes a list of drugs that decrease exposure of the concomitant medication when administered concomitantly with colesevelam hydrochloride and instructions for preventing or managing them.
| Drugs with a Narrow Therapeutic Index | |
|---|---|
| Clinical Impact: | Concomitant use with colesevelam hydrochloride may decrease the exposure of the narrow therapeutic index drug. In vivodrug interactions studies showed a decrease in exposure of cyclosporine when co-administered with colesevelam hydrochloride [see Clinical Pharmacology (12.3)]. |
| Intervention: | Administer the narrow therapeutic index drug at least 4 hours prior to colesevelam hydrochloride. Monitor drug levels when appropriate. |
| Examples: | Cyclosporine |
| Phenytoin | |
| Clinical Impact: | There have been postmarketing reports of increased seizure activity or decreased phenytoin levels in patients receiving phenytoin [see Adverse Reactions (6.2)]. |
| Intervention: | Administer phenytoin 4 hours prior to colesevelam hydrochloride. |
| Thyroid Hormone Replacement Therapy | |
| Clinical Impact: | In vivodrug interactions studies showed a decrease in exposure of levothyroxine when co-administered with colesevelam hydrochloride [see Clinical Pharmacology (12.3)] .There have been postmarketing reports of elevated thyroid-stimulating hormone (TSH) in patients receiving thyroid hormone replacement therapy [see Adverse Reactions (6.2)] . |
| Intervention: | Administer thyroid hormone replacement therapy 4 hours prior to colesevelam hydrochloride. |
| Warfarin | |
| Clinical Impact: | There have been postmarketing reports of reduced INR in patients receiving warfarin therapy [see Adverse Reactions (6.2)]. |
| Intervention: | Monitor INR frequently during colesevelam hydrochloride initiation then periodically thereafter. |
| Oral Contraceptives Containing Ethinyl Estradiol and Norethindrone | |
| Clinical Impact: | In vivodrug interactions studies showed a decrease in exposure of ethinyl estradiol and norethindrone when co-administered with colesevelam hydrochloride [see Clinical Pharmacology (12.3)]. |
| Intervention: | Administer oral contraceptives containing ethinyl estradiol and norethindrone 4 hours prior to colesevelam hydrochloride. |
| Olmesartan Medoxomil | |
| Clinical Impact: | In vivodrug interactions studies showed a decrease in olmesartan medoxomil when co-administered with colesevelam hydrochloride [see Clinical Pharmacology (12.3)]. |
| Intervention: | Administer olmesartan medoxomil 4 hours prior to colesevelam hydrochloride. |
| Sulfonylureas | |
| Clinical Impact: | In vivodrug interactions studies showed a decrease in sulfonylureas when co-administered with colesevelam hydrochloride [see Clinical Pharmacology (12.3)]. |
| Intervention: | Administer sulfonylureas 4 hours prior to colesevelam hydrochloride. |
| Examples: | Glimepiride, glipizide, and glyburide |
| Oral Vitamin Supplements | |
| Clinical Impact: | Colesevelam hydrochloride may decrease the absorption of fat-soluble vitamins A, D, E, and K [see Warnings and Precautions (5.3)]. |
| Intervention: | Patients on oral vitamin supplementation should take their vitamins at least 4 hours prior to colesevelam hydrochloride. |
7.2 colesevelam Hydrochloride Drug Interactions That Increase the Exposure of the Concomitant Medication (7.2 Colesevelam Hydrochloride Drug Interactions that Increase the Exposure of the Concomitant Medication)
| Metformin Extended-Release (ER) | |
|---|---|
| Clinical Impact: | In vivodrug interactions studies showed an increase in metformin extended release (ER) when co-administered with colesevelam hydrochloride [see Clinical Pharmacology (12.3)]. |
| Intervention: | Monitor patients' glycemic control. |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:14.416309 · Updated: 2026-03-14T22:24:19.550175