Blink ®
a2b34eb0-001b-4f42-a8cc-c2a0150fafcc
34390-5
HUMAN OTC DRUG LABEL
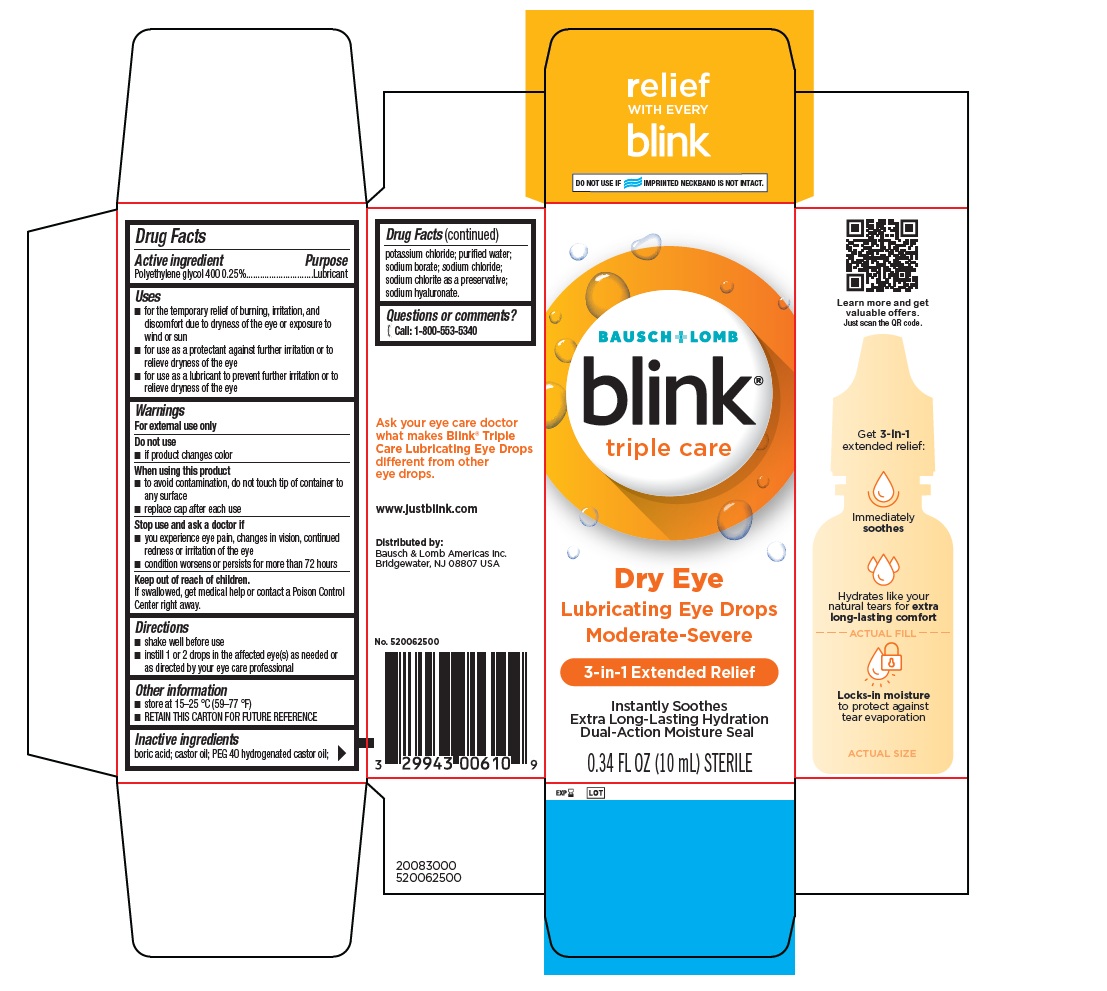
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Lubricant
Medication Information
Purpose
Lubricant
Description
Drug Facts
Uses
- for the temporary relief of burning, irritation, and discomfort due to dryness of the eye or exposure to wind or sun
- for use as a protectant against further irritation or to relieve dryness of the eye
- for use as a lubricant to prevent further irritation or to relieve dryness of the eye.
Section 42229-5
Drug Facts
Section 50565-1
Keep out of the reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if
- you experience eye pain, changes in vision, continued redness or irritation of the eye
- condition worsens or persists for more than 72 hours
Warnings
For external use only
Do not use
- if product changes color
When using this product
- to avoid contamination, do not touch tip of container to any surface
- replace cap after each use
Directions
- shake well before use.
- instill 1 or 2 drops in the affected eye(s) as needed or as directed by your eye care professional.
Active Ingredient
Polyethylene glycol 400 0.25%
Other Information
- Store at 15-25 °C (59-77 °F)
- RETAIN THIS CARTON FOR FUTURE REFERENCE
Inactive Ingredients
boric acid; castor oil; PEG 40 hydrogenated castor oil; potassium chloride; purified water; sodium borate; sodium chloride; sodium chlorite as a preservative; sodium hyaluronate.
Questions Or Comments?
[phone icon] Call: 1-800-553-5340
Principal Display Panel
BAUSCH + LOMB
blink
®
triple care
Dry Eye
Lubricating Eye Drops
Moderate-Severe
3-in-1 Extended Relief
Instantly Soothes
Extra Long-Lasting Hydration
Dual-Action Moisture Seal
0.34 FL OZ (10 mL) STERILE
Structured Label Content
Uses
- for the temporary relief of burning, irritation, and discomfort due to dryness of the eye or exposure to wind or sun
- for use as a protectant against further irritation or to relieve dryness of the eye
- for use as a lubricant to prevent further irritation or to relieve dryness of the eye.
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of the reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if
- you experience eye pain, changes in vision, continued redness or irritation of the eye
- condition worsens or persists for more than 72 hours
Purpose
Lubricant
Warnings
For external use only
Do not use
- if product changes color
When using this product
- to avoid contamination, do not touch tip of container to any surface
- replace cap after each use
Directions
- shake well before use.
- instill 1 or 2 drops in the affected eye(s) as needed or as directed by your eye care professional.
Active Ingredient
Polyethylene glycol 400 0.25%
Other Information
- Store at 15-25 °C (59-77 °F)
- RETAIN THIS CARTON FOR FUTURE REFERENCE
Inactive Ingredients
boric acid; castor oil; PEG 40 hydrogenated castor oil; potassium chloride; purified water; sodium borate; sodium chloride; sodium chlorite as a preservative; sodium hyaluronate.
Questions Or Comments? (Questions or comments?)
[phone icon] Call: 1-800-553-5340
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
BAUSCH + LOMB
blink
®
triple care
Dry Eye
Lubricating Eye Drops
Moderate-Severe
3-in-1 Extended Relief
Instantly Soothes
Extra Long-Lasting Hydration
Dual-Action Moisture Seal
0.34 FL OZ (10 mL) STERILE
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:20.387823 · Updated: 2026-03-14T23:12:32.924893