These Highlights Do Not Include All The Information Needed To Use Alendronate Sodium Tablets Safely And Effectively. See Full Prescribing Information For Alendronate Sodium Tablets, Usp.
a24fd39c-34a7-4de2-9439-781e612189ca
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Alendronate sodium is a bisphosphonate indicated for: Treatment and prevention of osteoporosis in postmenopausal women ( 1.1 , 1.2 ) Treatment to increase bone mass in men with osteoporosis ( 1.3 ) Treatment of glucocorticoid-induced osteoporosis ( 1.4 ) Treatment of Paget's disease of bone ( 1.5 ) Limitations of use: Optimal duration of use has not been determined. For patients at low-risk for fracture, consider drug discontinuation after 3 to 5 years of use. ( 1.6 )
Indications and Usage
Alendronate sodium is a bisphosphonate indicated for: Treatment and prevention of osteoporosis in postmenopausal women ( 1.1 , 1.2 ) Treatment to increase bone mass in men with osteoporosis ( 1.3 ) Treatment of glucocorticoid-induced osteoporosis ( 1.4 ) Treatment of Paget's disease of bone ( 1.5 ) Limitations of use: Optimal duration of use has not been determined. For patients at low-risk for fracture, consider drug discontinuation after 3 to 5 years of use. ( 1.6 )
Dosage and Administration
Treatment of osteoporosis in postmenopausal women and in men: 10 mg daily or 70 mg tablet once weekly. ( 2.1 , 2.3 ) Prevention of osteoporosis in postmenopausal women: 5 mg daily or 35 mg once weekly. ( 2.2 ) Glucocorticoid-induced osteoporosis: 5 mg daily; or 10 mg daily in postmenopausal women not receiving estrogen. ( 2.4 ) Paget's disease: 40 mg daily for six months. ( 2.5 ) Instruct patients to: ( 2.6 ) Swallow tablets whole with 6-8 ounces plain water at least 30 minutes before the first food, drink, or medication of the day. Not lie down for at least 30 minutes after taking alendronate sodium tablet and until after food.
Warnings and Precautions
Upper Gastrointestinal Adverse Reactions can occur. Instruct patients to follow dosing instructions. Discontinue if new or worsening symptoms occur. ( 5.1 ) Hypocalcemia can worsen and must be corrected prior to use. ( 5.2 ) Severe Bone, Joint, Muscle Pain may occur. Discontinue use if severe symptoms develop. ( 5.3 ) Osteonecrosis of the Jaw has been reported. ( 5.4 ) Atypical Femur Fractures have been reported. Patients with new thigh or groin pain should be evaluated to rule out an incomplete femoral fracture. ( 5.5 )
Contraindications
Alendronate sodium is contraindicated in patients with the following conditions: Abnormalities of the esophagus which delay esophageal emptying such as stricture or achalasia [see Warnings and Precautions (5.1) ] Inability to stand or sit upright for at least 30 minutes [see Dosage and Administration (2.6) ; Warnings and Precautions (5.1) ] Hypocalcemia [see Warnings and Precautions (5.2) ] Hypersensitivity to any component of this product. Hypersensitivity reactions including urticaria and angioedema have been reported [see Adverse Reactions (6.2) ] .
Adverse Reactions
Most common adverse reactions (greater than or equal to 3%) are abdominal pain, acid regurgitation, constipation, diarrhea, dyspepsia, musculoskeletal pain, nausea. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Hangzhou Minsheng Binjiang Pharmaceutical Co., Ltd at 1-888-833-0728 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Calcium supplements, antacids, or oral medications containing multivalent cations interfere with absorption of alendronate. ( 2.6 , 7.1 ) Use caution when co-prescribing aspirin/nonsteroidal anti-inflammatory drugs that may worsen gastrointestinal irritation. ( 7.2 , 7.3 )
Medication Information
Warnings and Precautions
Upper Gastrointestinal Adverse Reactions can occur. Instruct patients to follow dosing instructions. Discontinue if new or worsening symptoms occur. ( 5.1 ) Hypocalcemia can worsen and must be corrected prior to use. ( 5.2 ) Severe Bone, Joint, Muscle Pain may occur. Discontinue use if severe symptoms develop. ( 5.3 ) Osteonecrosis of the Jaw has been reported. ( 5.4 ) Atypical Femur Fractures have been reported. Patients with new thigh or groin pain should be evaluated to rule out an incomplete femoral fracture. ( 5.5 )
Indications and Usage
Alendronate sodium is a bisphosphonate indicated for: Treatment and prevention of osteoporosis in postmenopausal women ( 1.1 , 1.2 ) Treatment to increase bone mass in men with osteoporosis ( 1.3 ) Treatment of glucocorticoid-induced osteoporosis ( 1.4 ) Treatment of Paget's disease of bone ( 1.5 ) Limitations of use: Optimal duration of use has not been determined. For patients at low-risk for fracture, consider drug discontinuation after 3 to 5 years of use. ( 1.6 )
Dosage and Administration
Treatment of osteoporosis in postmenopausal women and in men: 10 mg daily or 70 mg tablet once weekly. ( 2.1 , 2.3 ) Prevention of osteoporosis in postmenopausal women: 5 mg daily or 35 mg once weekly. ( 2.2 ) Glucocorticoid-induced osteoporosis: 5 mg daily; or 10 mg daily in postmenopausal women not receiving estrogen. ( 2.4 ) Paget's disease: 40 mg daily for six months. ( 2.5 ) Instruct patients to: ( 2.6 ) Swallow tablets whole with 6-8 ounces plain water at least 30 minutes before the first food, drink, or medication of the day. Not lie down for at least 30 minutes after taking alendronate sodium tablet and until after food.
Dosage Forms and Strengths
How Supplied
Alendronate sodium tablets USP, 35 mg (alendronate), are modified oval, white to off-white tablets; one side debossed 'AP207', the other side blank.
NDC 60723-207-31 blister packs of 4 with child resistant package.
NDC 60723-207-32 unit-of-use packages of 12 with child resistant package.
NDC 60723-207-33 unit dose packages of 20. This package not intended for household use. For institutional use only.
Alendronate sodium tablets USP, 70 mg (alendronate), are modified oval, white to off-white tablets; one side debossed 'AP205', the other side blank.
NDC 60723-205-31 blister packs of 4 with child resistant package.
NDC 60723-205-32 unit-of-use packages of 12 with child resistant package.
NDC 60723-205-33 unit dose packages of 20. This package not intended for household use. For institutional use only.
Contraindications
Alendronate sodium is contraindicated in patients with the following conditions: Abnormalities of the esophagus which delay esophageal emptying such as stricture or achalasia [see Warnings and Precautions (5.1) ] Inability to stand or sit upright for at least 30 minutes [see Dosage and Administration (2.6) ; Warnings and Precautions (5.1) ] Hypocalcemia [see Warnings and Precautions (5.2) ] Hypersensitivity to any component of this product. Hypersensitivity reactions including urticaria and angioedema have been reported [see Adverse Reactions (6.2) ] .
Adverse Reactions
Most common adverse reactions (greater than or equal to 3%) are abdominal pain, acid regurgitation, constipation, diarrhea, dyspepsia, musculoskeletal pain, nausea. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Hangzhou Minsheng Binjiang Pharmaceutical Co., Ltd at 1-888-833-0728 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Calcium supplements, antacids, or oral medications containing multivalent cations interfere with absorption of alendronate. ( 2.6 , 7.1 ) Use caution when co-prescribing aspirin/nonsteroidal anti-inflammatory drugs that may worsen gastrointestinal irritation. ( 7.2 , 7.3 )
Description
Alendronate sodium is a bisphosphonate indicated for: Treatment and prevention of osteoporosis in postmenopausal women ( 1.1 , 1.2 ) Treatment to increase bone mass in men with osteoporosis ( 1.3 ) Treatment of glucocorticoid-induced osteoporosis ( 1.4 ) Treatment of Paget's disease of bone ( 1.5 ) Limitations of use: Optimal duration of use has not been determined. For patients at low-risk for fracture, consider drug discontinuation after 3 to 5 years of use. ( 1.6 )
Section 34077-8
Risk Summary
Available data on the use of Alendronate sodium in pregnant women are insufficient to inform a drug-associated risk of adverse maternal or fetal outcomes. Discontinue Alendronate sodium when pregnancy is recognized.
In animal reproduction studies, daily oral administration of alendronate to rats from before mating through the end of gestation or lactation showed decreased postimplantation survival and decreased pup body weight gain starting at doses equivalent to less than half of the highest recommended 40 mg clinical daily dose (based on body surface area, mg/m 2). Oral administration of alendronate to rats during organogenesis resulted in reduced fetal ossification starting at doses 3 times the 40 mg clinical daily dose. No similar fetal effects were observed in pregnant rabbits dosed orally during organogenesis at doses equivalent to approximately 10 times the 40 mg clinical daily dose.
Delayed or failed delivery of offspring, protracted parturition, and late pregnancy maternal and fetal deaths due to maternal hypocalcemia occurred in rats at oral doses as low as one tenth the 40 mg clinical daily dose (see Data).
Bisphosphonates are incorporated into the bone matrix, from which they are gradually released over a period of years. The amount of bisphosphonate incorporated into adult bone and available for release into the systemic circulation is directly related to the dose and duration of bisphosphonate use. Consequently, based on the mechanism of action of bisphosphonates, there is a potential risk of fetal harm, predominantly skeletal, if a woman becomes pregnant after completing a course of bisphosphonate therapy. The impact of variables such as time between cessation of bisphosphonate therapy to conception, the particular bisphosphonate used, and the route of administration (intravenous versus oral) on the risk has not been studied.
The estimated background risk of major birth defects and miscarriage for the indicated population(s) is unknown. All pregnancies have a background risk of birth defects, loss, or other adverse outcomes. In the U.S. general population, the estimated background risks of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Data
Animal Data
Reproduction studies in rats dosed orally from before mating to the end of gestation or lactation showed decreased postimplantation survival starting at 2 mg/kg/day and decreased body weight gain starting at 1 mg/kg/day, doses equivalent to less than half the 40 mg clinical daily dose based on body surface area, mg/m 2. Incidence of incomplete fetal ossification in vertebral, skull, and sternebral bones were increased in rats dosed orally during organogenesis starting at 10 mg/kg/day (approximately 3 times the 40 mg clinical daily dose) . No similar fetal effects were observed in pregnant rabbits dosed orally during organogenesis at up to 35 mg/kg/day (equivalent to approximately 10 times the 40 mg clinical daily dose.
Both total and ionized calcium decreased in pregnant rats dosed orally with 15 mg/kg/day alendronate (approximately 4 times the 40 mg clinical daily dose) resulting in delays and failures of delivery. Protracted parturition due to maternal hypocalcemia was observed when rats were treated from before mating through gestation starting at 0.5 mg/kg/day (approximately one tenth the 40 mg clinical daily dose). Maternotoxicity (late pregnancy deaths) also occurred in the female rats treated orally with 15 mg/kg/day (approximately 4 times the 40 mg clinical daily dose) for varying gestational time periods. These maternal deaths were lessened but not eliminated by cessation of treatment. Calcium supplementation in the drinking water or by subcutaneous minipump to rats dosed orally with 15 mg/kg/day alendronate could not ameliorate the hypocalcemia or prevent the dystocia-related maternal and neonatal deaths. However, intravenous calcium supplementation prevented maternal, but not neonatal deaths.
Section 42229-5
Re-treatment of Paget's Disease
Re-treatment with alendronate sodium tablets may be considered, following a six-month post-treatment evaluation period in patients who have relapsed, based on increases in serum alkaline phosphatase, which should be measured periodically. Re-treatment may also be considered in those who failed to normalize their serum alkaline phosphatase.
Section 44425-7
Storage
Store in a well-closed container at 20° - 25°C (68° - 77°F); excursions permitted to 15° - 30°C (59° - 86°F) [see USP Controlled Room Temperature].
7.2 Aspirin
In clinical studies, the incidence of upper gastrointestinal adverse events was increased in patients receiving concomitant therapy with daily doses of alendronate greater than 10 mg and aspirin-containing products.
10 Overdosage
Significant lethality after single oral doses was seen in female rats and mice at 552 mg/kg (3256 mg/m 2) and 966 mg/kg (2898 mg/m 2), respectively. In males, these values were slightly higher, 626 and 1280 mg/kg, respectively. There was no lethality in dogs at oral doses up to 200 mg/kg (4000 mg/m 2).
No specific information is available on the treatment of overdosage with alendronate sodium. Hypocalcemia, hypophosphatemia, and upper gastrointestinal adverse events, such as upset stomach, heartburn, esophagitis, gastritis, or ulcer, may result from oral overdosage. Milk or antacids should be given to bind alendronate. Due to the risk of esophageal irritation, vomiting should not be induced and the patient should remain fully upright.
Dialysis would not be beneficial.
8.2 Lactation
Risk Summary
It is not known whether alendronate is present in human breast milk, affects human milk production, or has effects on the breastfed infant. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for alendronate sodium and any potential adverse effects on the breastfed child from alendronate sodium or from the underlying maternal condition.
11 Description
Alendronate sodium is a bisphosphonate that acts as a specific inhibitor of osteoclast-mediated bone resorption.
Bisphosphonates are synthetic analogs of pyrophosphate that bind to the hydroxyapatite found in bone.
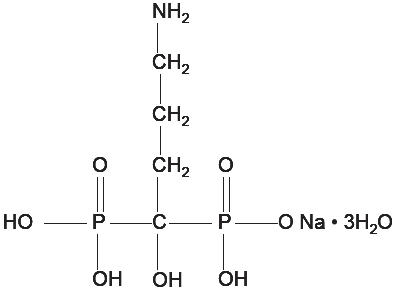
Alendronate sodium is chemically described as (4-amino-1-hydroxybutylidene) bisphosphonic acid monosodium salt trihydrate. The empirical formula of alendronate sodium is C 4H 12NNaO 7P 2∙3H 2O and its formula weight is 325.12. The structural formula is:
Alendronate sodium is a white, crystalline, nonhygroscopic powder. It is soluble in water, very slightly soluble in alcohol, and practically insoluble in chloroform.
Alendronate sodium tablets, USP for oral administration contain 6.53, 13.06, 45.69 or 91.37 mg of alendronate monosodium salt trihydrate, which is the molar equivalent of 5, 10, 35 and 70 mg, respectively, of free acid, and the following inactive ingredients: microcrystalline cellulose, croscarmellose sodium, and magnesium stearate.
Medication Guide
Read the Medication Guide that comes with Alendronate Sodium Tablets before you start taking it and each time you get a refill. There may be new information. This Medication Guide does not take the place of talking with your doctor about your medical condition or treatment. Talk to your doctor if you have any questions about Alendronate Sodium Tablets.
What is the most important information I should know about alendronate sodium tablets?
Alendronate sodium tablets can cause serious side effects including:
- Esophagus problems
- Low calcium levels in your blood (hypocalcemia)
- Bone, joint, or muscle pain
- Severe jaw bone problems (osteonecrosis)
- Unusual thigh bone fractures
- Esophagus problems.
Some people who take alendronate sodium tablets may develop problems in the esophagus (the tube that connects the mouth and the stomach).
These problems include irritation, inflammation, or ulcers of the esophagus which may sometimes bleed.
- It is important that you take alendronate sodium tablets exactly as prescribed to help lower your chance of getting esophagus problems. (See the section " How should I take alendronate sodium tablets?")
- Stop taking alendronate sodium tablets and call your doctor right away if you get chest pain, new or worsening heartburn, or have trouble or pain when you swallow.
- Low calcium levels in your blood (hypocalcemia).
Alendronate sodium tablets may lower the calcium levels in your blood. If you have low blood calcium before you start taking alendronate sodium tablets, it may get worse during treatment. Your low blood calcium must be treated before you take alendronate sodium tablets. Most people with low blood calcium levels do not have symptoms, but some people may have symptoms. Call your doctor right away if you have symptoms of low blood calcium such as:
- Spasms, twitches, or cramps in your muscles
- Numbness or tingling in your fingers, toes, or around your mouth
Your doctor may prescribe calcium and vitamin D to help prevent low calcium levels in your blood, while you take alendronate sodium tablets. Take calcium and vitamin D as your doctor tells you to.
- Bone, joint, or muscle pain.
Some people who take alendronate sodium tablets develop severe bone, joint, or muscle pain.
- Severe jaw bone problems (osteonecrosis).
Severe jaw bone problems may happen when you take alendronate sodium tablets. Your doctor should examine your mouth before you start alendronate sodium tablets. Your doctor may tell you to see your dentist before you start alendronate sodium tablets. It is important for you to practice good mouth care during treatment with alendronate sodium tablets.
- Unusual thigh bone fractures.
Some people have developed unusual fractures in their thigh bone. Symptoms of a fracture may include new or unusual pain in your hip, groin, or thigh.
Call your doctor right away if you have any of these side effects.
What is alendronate sodium tablet?
Alendronate sodium tablet is a prescription medicine used to:
- Treat or prevent osteoporosis in women after menopause. It helps reduce the chance of having a hip or spinal fracture (break).
- Increase bone mass in men with osteoporosis.
- Treat osteoporosis in either men or women who are taking corticosteroid medicines.
- Treat certain men and women who have Paget's disease of the bone.
It is not known how long alendronate sodium tablet works for the treatment and prevention of osteoporosis. You should see your doctor regularly to determine if alendronate sodium tablet is still right for you.
Alendronate sodium tablet is not for use in children.
Who should not take alendronate sodium tablets?
Do not take alendronate sodium tablets if you:
- Have certain problems with your esophagus, the tube that connects your mouth with your stomach
- Cannot stand or sit upright for at least 30 minutes
- Have low levels of calcium in your blood
- Are allergic to alendronate sodium tablet or any of its ingredients. A list of ingredients is at the end of this leaflet.
What should I tell my doctor before taking alendronate sodium tablets?
Before you start alendronate sodium tablets, be sure to talk to your doctor if you:
- Have problems with swallowing
- Have stomach or digestive problems
- Have low blood calcium
- Plan to have dental surgery or teeth removed
- Have kidney problems
- Have been told you have trouble absorbing minerals in your stomach or intestines (malabsorption syndrome)
- Are pregnant, trying to become pregnant or suspect that you are pregnant. If you become pregnant while taking alendronate sodium tablets, stop taking it and contact your doctor. It is not known if alendronate sodium tablets can harm your unborn baby.
- Are breast-feeding or plan to breast-feed. It is not known if alendronate sodium passes into your milk and may harm your baby.
Especially tell your doctor if you take:
- antacids
- aspirin
- Nonsteroidal Anti-Inflammatory (NSAID) medicines
Tell your doctor about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements. Certain medicines may affect how alendronate sodium tablet works.
Know the medicines you take. Keep a list of them and show it to your doctor and pharmacist each time you get a new medicine.
How should I take alendronate sodium tablets?
- Take alendronate sodium tablets exactly as your doctor tells you.
- Alendronate sodium tablet works only if taken on an empty stomach.
- Take alendronate sodium tablets, after you get up for the day and before taking your first food, drink, or other medicine.
- Take alendronate sodium tablets while you are sitting or standing.
- Do not chew or suck on a tablet of alendronate sodium.
- Swallow alendronate sodium tablet with a full glass (6-8 oz) of plain water only.
- Do not take alendronate sodium tablets with mineral water, coffee, tea, soda, or juice.
- If you take Alendronate Sodium Tablets Daily:
- Take 1 alendronate sodium tablet one time a day, every day after you get up for the day and before taking your first food, drink, or other medicine.
- If you take Once Weekly Alendronate Sodium Tablets:
- Choose the day of the week that best fits your schedule.
- Take 1 dose of alendronate sodium tablets every week on your chosen day after you get up for the day and before taking your first food, drink, or other medicine.
After swallowing alendronate sodium tablet, wait at least 30 minutes:
- Before you lie down. You may sit, stand or walk, and do normal activities like reading.
- Before you take your first food or drink except for plain water.
- Before you take other medicines, including antacids, calcium, and other supplements and vitamins.
Do not lie down for at least 30 minutes after you take alendronate sodium tablets and after you eat your first food of the day.
If you miss a dose of alendronate sodium tablets, do not take it later in the day. Take your missed dose on the next morning after you remember and then return to your normal schedule. Do not take 2 doses on the same day.
If you take too much alendronate sodium, call your doctor. Do not try to vomit. Do not lie down.
What are the possible side effects of alendronate sodium tablets?
Alendronate sodium tablets may cause serious side effects.
The most common side effects of alendronate sodium tablets are:
- Stomach area (abdominal) pain
- Heartburn
- Constipation
- Diarrhea
- Upset stomach
- Pain in your bones, joints, or muscles
- Nausea
You may get allergic reactions, such as hives or swelling of your face, lips, tongue, or throat. Worsening of asthma has been reported.
Tell your doctor if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of alendronate sodium tablets. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to Hangzhou Minsheng Binjiang Pharmaceutical Co., Ltd at 1-888-833-0728 or FDA at 1-800-FDA-1088.
How do I store alendronate sodium tablets?
- Store at 20° - 25°C (68° - 77°F); excursions permitted to 15° - 30°C (59° - 86°F).
- Keep alendronate sodium tablets in a tightly closed container.
Keep alendronate sodium tablets and all medicines out of the reach of children.
General information about the safe and effective use of alendronate sodium tablets.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use alendronate sodium tablets for a condition for which it was not prescribed.
Do not give alendronate sodium tablets to other people, even if they have the same symptoms you have. It may harm them.
This Medication Guide summarizes the most important information about alendronate sodium tablets. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about alendronate sodium tablets that is written for health professionals.
For more information, call 1-888-833-0728.
What are the ingredients in alendronate sodium tablets?
Active ingredient: alendronate sodium
Inactive ingredients: microcrystalline cellulose, croscarmellose sodium, and magnesium stearate.
Rx only
Manufactured by:
Hangzhou Minsheng Binjiang Pharmaceutical Co., Ltd.
Hangzhou, 310051, CHINA
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Revised: 02/2020
8.4 Pediatric Use
Alendronate is not indicated for use in pediatric patients.
The safety and efficacy of alendronate sodium were examined in a randomized, double-blind, placebo-controlled two-year study of 139 pediatric patients, aged 4-18 years, with severe osteogenesis imperfecta (OI). One-hundred-and-nine patients were randomized to 5 mg alendronate daily (weight less than 40 kg) or 10 mg alendronate daily (weight greater than or equal to 40 kg) and 30 patients to placebo. The mean baseline lumbar spine BMD Z-score of the patients was -4.5. The mean change in lumbar spine BMD Z-score from baseline to Month 24 was 1.3 in the alendronate sodium-treated patients and 0.1 in the placebo-treated patients. Treatment with alendronate sodium did not reduce the risk of fracture. Sixteen percent of the alendronate sodium patients who sustained a radiologically-confirmed fracture by Month 12 of the study had delayed fracture healing (callus remodeling) or fracture non-union when assessed radiographically at Month 24 compared with 9% of the placebo-treated patients. In alendronate sodium-treated patients, bone histomorphometry data obtained at Month 24 demonstrated decreased bone turnover and delayed mineralization time; however, there were no mineralization defects. There were no statistically significant differences between the alendronate sodium and placebo groups in reduction of bone pain. The oral bioavailability in children was similar to that observed in adults.
The overall safety profile of alendronate sodium in osteogenesis imperfecta patients treated for up to 24 months was generally similar to that of adults with osteoporosis treated with alendronate sodium. However, there was an increased occurrence of vomiting in osteogenesis imperfecta patients treated with alendronate sodium compared to placebo. During the 24-month treatment period, vomiting was observed in 32 of 109 (29.4%) patients treated with alendronate sodium and 3 of 30 (10%) patients treated with placebo.
In a pharmacokinetic study, 6 of 24 pediatric osteogenesis imperfecta patients who received a single oral dose of alendronate sodium 35 or 70 mg developed fever, flu-like symptoms, and/or mild lymphocytopenia within 24 to 48 hours after administration. These events, lasting no more than 2 to 3 days and responding to acetaminophen, are consistent with an acute-phase response that has been reported in patients receiving bisphosphonates, including alendronate sodium. [See Adverse Reactions (6.2).]
8.5 Geriatric Use
Of the patients receiving alendronate sodium in the Fracture Intervention Trial (FIT), 71% (n=2302) were greater than or equal to 65 years of age and 17% (n=550) were greater than or equal to 75 years of age. Of the patients receiving alendronate sodium in the United States and Multinational osteoporosis treatment studies in women, osteoporosis studies in men, glucocorticoid-induced osteoporosis studies, and Paget's disease studies [see Clinical Studies (14.1), (14.3), (14.4), (14.5)] , 45%, 54%, 37%, and 70%, respectively, were 65 years of age or over. No overall differences in efficacy or safety were observed between these patients and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
4 Contraindications
Alendronate sodium is contraindicated in patients with the following conditions:
- Abnormalities of the esophagus which delay esophageal emptying such as stricture or achalasia [see Warnings and Precautions (5.1)]
- Inability to stand or sit upright for at least 30 minutes [see Dosage and Administration (2.6); Warnings and Precautions (5.1)]
- Hypocalcemia [see Warnings and Precautions (5.2)]
- Hypersensitivity to any component of this product. Hypersensitivity reactions including urticaria and angioedema have been reported [see Adverse Reactions (6.2)] .
6 Adverse Reactions
Most common adverse reactions (greater than or equal to 3%) are abdominal pain, acid regurgitation, constipation, diarrhea, dyspepsia, musculoskeletal pain, nausea. (
6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Hangzhou Minsheng Binjiang Pharmaceutical Co., Ltd at 1-888-833-0728 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions
5.6 Renal Impairment
Alendronate sodium is not recommended for patients with creatinine clearance less than 35 mL/min.
8.6 Renal Impairment
Alendronate sodium is not recommended for patients with creatinine clearance less than 35 mL/min. No dosage adjustment is necessary in patients with creatinine clearance values between 35-60 mL/min [see Clinical Pharmacology (12.3)] .
12.2 Pharmacodynamics
Alendronate is a bisphosphonate that binds to bone hydroxyapatite and specifically inhibits the activity of osteoclasts, the bone-resorbing cells. Alendronate reduces bone resorption with no direct effect on bone formation, although the latter process is ultimately reduced because bone resorption and formation are coupled during bone turnover.
5.2 Mineral Metabolism
Hypocalcemia must be corrected before initiating therapy with alendronate sodium [see Contraindications (4)] . Other disorders affecting mineral metabolism (such as vitamin D deficiency) should also be effectively treated. In patients with these conditions, serum calcium and symptoms of hypocalcemia should be monitored during therapy with alendronate sodium.
Presumably due to the effects of alendronate sodium on increasing bone mineral, small, asymptomatic decreases in serum calcium and phosphate may occur, especially in patients with Paget's disease, in whom the pretreatment rate of bone turnover may be greatly elevated, and in patients receiving glucocorticoids, in whom calcium absorption may be decreased.
Ensuring adequate calcium and vitamin D intake is especially important in patients with Paget's disease of bone and in patients receiving glucocorticoids.
8.7 Hepatic Impairment
As there is evidence that alendronate is not metabolized or excreted in the bile, no studies were conducted in patients with hepatic impairment. No dosage adjustment is necessary [see Clinical Pharmacology (12.3)].
1 Indications and Usage
Alendronate sodium is a bisphosphonate indicated for:
- Treatment and prevention of osteoporosis in postmenopausal women ( 1.1, 1.2)
- Treatment to increase bone mass in men with osteoporosis ( 1.3)
- Treatment of glucocorticoid-induced osteoporosis ( 1.4)
- Treatment of Paget's disease of bone ( 1.5)
Limitations of use: Optimal duration of use has not been determined. For patients at low-risk for fracture, consider drug discontinuation after 3 to 5 years of use. ( 1.6)
12.1 Mechanism of Action
Animal studies have indicated the following mode of action. At the cellular level, alendronate shows preferential localization to sites of bone resorption, specifically under osteoclasts. The osteoclasts adhere normally to the bone surface but lack the ruffled border that is indicative of active resorption. Alendronate does not interfere with osteoclast recruitment or attachment, but it does inhibit osteoclast activity. Studies in mice on the localization of radioactive [ 3H]alendronate in bone showed about 10-fold higher uptake on osteoclast surfaces than on osteoblast surfaces. Bones examined 6 and 49 days after [ 3H]alendronate administration in rats and mice, respectively, showed that normal bone was formed on top of the alendronate, which was incorporated inside the matrix. While incorporated in bone matrix, alendronate is not pharmacologically active. Thus, alendronate must be continuously administered to suppress osteoclasts on newly formed resorption surfaces. Histomorphometry in baboons and rats showed that alendronate treatment reduces bone turnover (i.e., the number of sites at which bone is remodeled). In addition, bone formation exceeds bone resorption at these remodeling sites, leading to progressive gains in bone mass.
17.2 Dosing Instructions
Instruct patients that the expected benefits of alendronate sodium may only be obtained when it is taken with plain water the first thing upon arising for the day at least 30 minutes before the first food, beverage, or medication of the day.
Even dosing with orange juice or coffee has been shown to markedly reduce the absorption of alendronate sodium [see Clinical Pharmacology (12.3)].
Instruct patients not to chew or suck on the tablet because of a potential for oropharyngeal ulceration.
Instruct patients to swallow each tablet of alendronate sodium with a full glass of water (6-8 ounces) to facilitate delivery to the stomach and thus reduce the potential for esophageal irritation.
Instruct patients not to lie down for at least 30 minutes and until after their first food of the day.
Instruct patients not to take alendronate sodium at bedtime or before arising for the day. Patients should be informed that failure to follow these instructions may increase their risk of esophageal problems.
Instruct patients that if they develop symptoms of esophageal disease (such as difficulty or pain upon swallowing, retrosternal pain or new or worsening heartburn) they should stop taking alendronate sodium and consult their physician.
If patients miss a dose of once weekly alendronate sodium tablet, instruct patients to take one dose on the morning after they remember. They should not take two doses on the same day but should return to taking one dose once a week, as originally scheduled on their chosen day.
Manufactured by:
Hangzhou Minsheng Binjiang
Pharmaceutical Co., Ltd,
658 Bin’an Road Binjiang District,
Hangzhou, 310051, CHINA
Revised: 02/2020
5.3 Musculoskeletal Pain
In post-marketing experience, severe and occasionally incapacitating bone, joint, and/or muscle pain has been reported in patients taking bisphosphonates that are approved for the prevention and treatment of osteoporosis [see Adverse Reactions (6.2)] . This category of drugs includes alendronate. Most of the patients were postmenopausal women. The time to onset of symptoms varied from one day to several months after starting the drug. Discontinue use if severe symptoms develop. Most patients had relief of symptoms after stopping. A subset had recurrence of symptoms when rechallenged with the same drug or another bisphosphonate.
In placebo-controlled clinical studies of alendronate sodium, the percentages of patients with these symptoms were similar in the alendronate sodium and placebo groups.
5 Warnings and Precautions
- Upper Gastrointestinal Adverse Reactions can occur. Instruct patients to follow dosing instructions. Discontinue if new or worsening symptoms occur. ( 5.1)
- Hypocalcemia can worsen and must be corrected prior to use. ( 5.2)
- Severe Bone, Joint, Muscle Pain may occur. Discontinue use if severe symptoms develop. ( 5.3)
- Osteonecrosis of the Jaw has been reported. ( 5.4)
- Atypical Femur Fractures have been reported. Patients with new thigh or groin pain should be evaluated to rule out an incomplete femoral fracture. ( 5.5)
2 Dosage and Administration
- Treatment of osteoporosis in postmenopausal women and in men: 10 mg daily or 70 mg tablet once weekly. ( 2.1, 2.3)
- Prevention of osteoporosis in postmenopausal women: 5 mg daily or 35 mg once weekly. ( 2.2)
- Glucocorticoid-induced osteoporosis: 5 mg daily; or 10 mg daily in postmenopausal women not receiving estrogen. ( 2.4)
- Paget's disease: 40 mg daily for six months. ( 2.5)
- Instruct patients to: (
2.6)
- Swallow tablets whole with 6-8 ounces plain water at least 30 minutes before the first food, drink, or medication of the day.
- Not lie down for at least 30 minutes after taking alendronate sodium tablet and until after food.
3 Dosage Forms and Strengths
- 5 mg (alendronate) tablets are round, flat face beveled edged, white to off-white tablets; one side debossed "AP209", the other side blank.
- 10 mg (alendronate) tablets are modified oval, white to off-white tablets; one side debossed "AP208", the other side blank.
- 35 mg (alendronate) tablets are modified oval, white to off-white tablets; one side debossed "AP207", the other side blank.
- 70 mg (alendronate) tablets are modified oval, white to off-white tablets; one side debossed "AP205", the other side blank.
5.4 Osteonecrosis of the Jaw
Osteonecrosis of the jaw (ONJ), which can occur spontaneously, is generally associated with tooth extraction and/or local infection with delayed healing, and has been reported in patients taking bisphosphonates, including alendronate sodium. Known risk factors for osteonecrosis of the jaw include invasive dental procedures (e.g., tooth extraction, dental implants, boney surgery), diagnosis of cancer, concomitant therapies (e.g., chemotherapy, corticosteroids, angiogenesis inhibitors), poor oral hygiene, and co-morbid disorders (e.g., periodontal and/or other pre-existing dental disease, anemia, coagulopathy, infection, ill-fitting dentures). The risk of ONJ may increase with duration of exposure to bisphosphonates.
For patients requiring invasive dental procedures, discontinuation of bisphosphonate treatment may reduce the risk for ONJ. Clinical judgment of the treating physician and/or oral surgeon should guide the management plan of each patient based on individual benefit/risk assessment.
Patients who develop osteonecrosis of the jaw while on bisphosphonate therapy should receive care by an oral surgeon. In these patients, extensive dental surgery to treat ONJ may exacerbate the condition. Discontinuation of bisphosphonate therapy should be considered based on individual benefit/risk assessment.
6.2 Post Marketing Experience
The following adverse reactions have been identified during post-approval use of alendronate. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Body as a Whole: hypersensitivity reactions including urticaria and angioedema. Transient symptoms of myalgia, malaise, asthenia and fever have been reported with alendronate, typically in association with initiation of treatment. Symptomatic hypocalcemia has occurred, generally in association with predisposing conditions. Peripheral edema.
Gastrointestinal: esophagitis, esophageal erosions, esophageal ulcers, esophageal stricture or perforation, and oropharyngeal ulceration. Gastric or duodenal ulcers, some severe and with complications, have also been reported [see Dosage and Administration (2.6); Warnings and Precautions (5.1)].
Localized osteonecrosis of the jaw, generally associated with tooth extraction and/or local infection with delayed healing, has been reported [see Warnings and Precautions (5.4)].
Musculoskeletal: bone, joint, and/or muscle pain, occasionally severe, and incapacitating [see Warnings and Precautions (5.3)]; joint swelling; low-energy femoral shaft and subtrochanteric fractures [see Warnings and Precautions (5.5)].
Nervous System: dizziness and vertigo.
Pulmonary: acute asthma exacerbations.
Skin: rash (occasionally with photosensitivity), pruritus, alopecia, severe skin reactions, including Stevens-Johnson syndrome and toxic epidermal necrolysis.
Special Senses: uveitis, scleritis or episcleritis. Cholesteatoma of the external auditory canal (focal osteonecrosis)
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
1.6 Important Limitations of Use
The optimal duration of use has not been determined. The safety and effectiveness of alendronate sodium tablets, USP for the treatment of osteoporosis are based on clinical data of four years duration. All patients on bisphosphonate therapy should have the need for continued therapy re-evaluated on a periodic basis. Patients at low-risk for fracture should be considered for drug discontinuation after 3 to 5 years of use. Patients who discontinue therapy should have their risk for fracture re-evaluated periodically.
7.1 Calcium Supplements/antacids
Co-administration of alendronate sodium and calcium, antacids, or oral medications containing multivalent cations will interfere with absorption of alendronate sodium. Therefore, instruct patients to wait at least one-half hour after taking alendronate sodium before taking any other oral medications.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Instruct patients to read the Medication Guide before starting therapy with alendronate sodium and to reread it each time the prescription is renewed.
5.7 Glucocorticoid Induced Osteoporosis
The risk versus benefit of alendronate sodium for treatment at daily dosages of glucocorticoids less than 7.5 mg of prednisone or equivalent has not been established [see Indications and Usage (1.4)] . Before initiating treatment, the gonadal hormonal status of both men and women should be ascertained and appropriate replacement considered.
A bone mineral density measurement should be made at the initiation of therapy and repeated after 6 to 12 months of combined alendronate sodium and glucocorticoid treatment.
1.5 Treatment of Paget's Disease of Bone
Alendronate sodium tablets, USP are indicated for the treatment of Paget's disease of bone in men and women. Treatment is indicated in patients with Paget's disease of bone who have alkaline phosphatase at least two times the upper limit of normal, or those who are symptomatic, or those at risk for future complications from their disease. [See Clinical Studies (14.5).]
2.5 Treatment of Paget's Disease of Bone
The recommended treatment regimen is 40 mg once a day for six months.
7.3 Nonsteroidal Anti Inflammatory Drugs
Alendronate sodium may be administered to patients taking nonsteroidal anti-inflammatory drugs (NSAIDs). In a 3-year, controlled, clinical study (n=2027) during which a majority of patients received concomitant NSAIDs, the incidence of upper gastrointestinal adverse events was similar in patients taking alendronate 5 or 10 mg/day compared to those taking placebo. However, since NSAID use is associated with gastrointestinal irritation, caution should be used during concomitant use with alendronate sodium.
14.5 Treatment of Paget's Disease of Bone
The efficacy of alendronate 40 mg once daily for six months was demonstrated in two double-blind clinical studies of male and female patients with moderate to severe Paget's disease (alkaline phosphatase at least twice the upper limit of normal): a placebo-controlled, multinational study and a U.S. comparative study with etidronate disodium 400 mg/day. Figure 6 shows the mean percent changes from baseline in serum alkaline phosphatase for up to six months of randomized treatment.
|
Figure 6:
Studies in Paget's Disease of Bone
Effect on Serum Alkaline Phosphatase of Alendronate 40 mg/day Versus Placebo or Etidronate 400 mg/day |
At six months the suppression in alkaline phosphatase in patients treated with alendronate sodium was significantly greater than that achieved with etidronate and contrasted with the complete lack of response in placebo-treated patients. Response (defined as either normalization of serum alkaline phosphatase or decrease from baseline greater than or equal to 60%) occurred in approximately 85% of patients treated with alendronate sodium in the combined studies vs. 30% in the etidronate group and 0% in the placebo group. Alendronate sodium was similarly effective regardless of age, gender, race, prior use of other bisphosphonates, or baseline alkaline phosphatase within the range studied (at least twice the upper limit of normal). Bone histology was evaluated in 33 patients with Paget's disease treated with alendronate 40 mg/day for 6 months. As in patients treated for osteoporosis [see Clinical Studies (14.1)], alendronate sodium did not impair mineralization, and the expected decrease in the rate of bone turnover was observed. Normal lamellar bone was produced during treatment with alendronate sodium, even where preexisting bone was woven and disorganized. Overall, bone histology data support the conclusion that bone formed during treatment with alendronate sodium is of normal quality.
2.6 Important Administration Instructions
Instruct patients to do the following:
- Take alendronate sodium tablets at least one-half hour before the first food, beverage, or medication of the day with plain water only [see Patient Counseling Information (17.2)] . Other beverages (including mineral water), food, and some medications are likely to reduce the absorption of alendronate sodium tablets [see Drug Interactions (7.1)] . Waiting less than 30 minutes, or taking alendronate sodium tablets with food, beverages (other than plain water) or other medications will lessen the effect of alendronate sodium tablets by decreasing its absorption into the body.
- Take alendronate sodium tablets upon arising for the day. To facilitate delivery to the stomach and thus reduce the potential for esophageal irritation, an alendronate sodium tablet should be swallowed with a full glass of water (6-8 ounces). Patients should not lie down for at least 30 minutes and until after their first food of the day. Alendronate sodium tablets should not be taken at bedtime or before arising for the day. Failure to follow these instructions may increase the risk of esophageal adverse experiences [see Warnings and Precautions (5.1) and Patient Counseling Information (17.2)] .
13.2 Animal Toxicology And/or Pharmacology
The relative inhibitory activities on bone resorption and mineralization of alendronate and etidronate were compared in the Schenk assay, which is based on histological examination of the epiphyses of growing rats. In this assay, the lowest dose of alendronate that interfered with bone mineralization (leading to osteomalacia) was 6000-fold the antiresorptive dose. The corresponding ratio for etidronate was one to one. These data suggest that alendronate administered in therapeutic doses is highly unlikely to induce osteomalacia.
5.1 Upper Gastrointestinal Adverse Reactions
Alendronate sodium, like other bisphosphonates administered orally, may cause local irritation of the upper gastrointestinal mucosa. Because of these possible irritant effects and a potential for worsening of the underlying disease, caution should be used when alendronate sodium is given to patients with active upper gastrointestinal problems (such as known Barrett's esophagus, dysphagia, other esophageal diseases, gastritis, duodenitis, or ulcers).
Esophageal adverse experiences, such as esophagitis, esophageal ulcers and esophageal erosions, occasionally with bleeding and rarely followed by esophageal stricture or perforation, have been reported in patients receiving treatment with oral bisphosphonates including alendronate sodium. In some cases these have been severe and required hospitalization. Physicians should therefore be alert to any signs or symptoms signaling a possible esophageal reaction and patients should be instructed to discontinue alendronate sodium and seek medical attention if they develop dysphagia, odynophagia, retrosternal pain or new or worsening heartburn.
The risk of severe esophageal adverse experiences appears to be greater in patients who lie down after taking oral bisphosphonates including alendronate sodium and/or who fail to swallow oral bisphosphonates including alendronate sodium with the recommended full glass (6-8 ounces) of water, and/or who continue to take oral bisphosphonates including alendronate sodium after developing symptoms suggestive of esophageal irritation. Therefore, it is very important that the full dosing instructions are provided to, and understood by, the patient [see Dosage and Administration (2.6)] . In patients who cannot comply with dosing instructions due to mental disability, therapy with alendronate sodium should be used under appropriate supervision.
There have been post-marketing reports of gastric and duodenal ulcers with oral bisphosphonate use, some severe and with complications, although no increased risk was observed in controlled clinical trials [see Adverse Reactions (6.2)] .
2.8 Administration Instructions for Missed Doses
If a once-weekly dose of alendronate sodium tablet is missed, instruct patients to take one dose on the morning after they remember. They should not take two doses on the same day but should return to taking one dose once a week, as originally scheduled on their chosen day.
1.4 Treatment of Glucocorticoid Induced Osteoporosis
Alendronate sodium tablets, USP are indicated for the treatment of glucocorticoid-induced osteoporosis in men and women receiving glucocorticoids in a daily dosage equivalent to 7.5 mg or greater of prednisone and who have low bone mineral density [see Clinical Studies (14.4)] .
2.4 Treatment of Glucocorticoid Induced Osteoporosis
The recommended dosage is one 5 mg tablet once daily, except for postmenopausal women not receiving estrogen, for whom the recommended dosage is one 10 mg tablet once daily.
1.1 Treatment of Osteoporosis in Postmenopausal Women
Alendronate sodium tablets, USP are indicated for the treatment of osteoporosis in postmenopausal women. In postmenopausal women, alendronate sodium tablets, USP increase bone mass and reduce the incidence of fractures, including those of the hip and spine (vertebral compression fractures). [See Clinical Studies (14.1).]
14.4 Treatment of Glucocorticoid Induced Osteoporosis
The efficacy of alendronate 5 and 10 mg once daily in men and women receiving glucocorticoids (at least 7.5 mg/day of prednisone or equivalent) was demonstrated in two, one-year, double-blind, randomized, placebo-controlled, multicenter studies of virtually identical design, one performed in the United States and the other in 15 different countries (Multinational [which also included alendronate 2.5 mg/day]). These studies enrolled 232 and 328 patients, respectively, between the ages of 17 and 83 with a variety of glucocorticoid-requiring diseases. Patients received supplemental calcium and vitamin D. Figure 5 shows the mean increases relative to placebo in BMD of the lumbar spine, femoral neck, and trochanter in patients receiving alendronate 5 mg/day for each study.
|
Figure 5:
Studies in Glucocorticoid - Treated Patients
Increase in BMD Alendronate 5 mg/day at One Year |
After one year, significant increases relative to placebo in BMD were seen in the combined studies at each of these sites in patients who received alendronate 5 mg/day. In the placebo-treated patients, a significant decrease in BMD occurred at the femoral neck (-1.2%), and smaller decreases were seen at the lumbar spine and trochanter. Total body BMD was maintained with alendronate 5 mg/day. The increases in BMD with alendronate 10 mg/day were similar to those with alendronate 5 mg/day in all patients except for postmenopausal women not receiving estrogen therapy. In these women, the increases (relative to placebo) with alendronate 10 mg/day were greater than those with alendronate 5 mg/day at the lumbar spine (4.1% vs. 1.6%) and trochanter (2.8% vs. 1.7%), but not at other sites. Alendronate sodium was effective regardless of dose or duration of glucocorticoid use. In addition, alendronate sodium was similarly effective regardless of age (less than 65 vs. greater than or equal to 65 years), race (Caucasian vs. other races), gender, underlying disease, baseline BMD, baseline bone turnover, and use with a variety of common medications.
Bone histology was normal in the 49 patients biopsied at the end of one year who received alendronate at doses of up to 10 mg/day.
Of the original 560 patients in these studies, 208 patients who remained on at least 7.5 mg/day of prednisone or equivalent continued into a one-year double-blind extension. After two years of treatment, spine BMD increased by 3.7% and 5.0% relative to placebo with alendronate 5 and 10 mg/day, respectively. Significant increases in BMD (relative to placebo) were also observed at the femoral neck, trochanter, and total body.
After one year, 2.3% of patients treated with alendronate 5 or 10 mg/day (pooled) vs. 3.7% of those treated with placebo experienced a new vertebral fracture (not significant). However, in the population studied for two years, treatment with alendronate (pooled dosage groups: 5 or 10 mg for two years or 2.5 mg for one year followed by 10 mg for one year) significantly reduced the incidence of patients with a new vertebral fracture (alendronate sodium 0.7% vs. placebo 6.8%).
2.1 Treatment of Osteoporosis in Postmenopausal Women
The recommended dosage is:
| ● one 70 mg (alendronate) tablet once weekly |
| or |
| ● one 10 mg (alendronate) tablet once daily |
1.2 Prevention of Osteoporosis in Postmenopausal Women
Alendronate sodium tablets, USP are indicated for the prevention of postmenopausal osteoporosis [see Clinical Studies (14.2)] .
2.2 Prevention of Osteoporosis in Postmenopausal Women
The recommended dosage is:
| ● one 35 mg (alendronate) tablet once weekly |
| or |
| ● one 5 mg (alendronate) tablet once daily |
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Harderian gland (a retro-orbital gland not present in humans) adenomas were increased in high-dose female mice (p=0.003) in a 92-week oral carcinogenicity study at doses of alendronate of 1, 3, and 10 mg/kg/day (males) or 1, 2, and 5 mg/kg/day (females). These doses are equivalent to approximately 0.1 to 1 times the highest recommended clinical daily dose of 40 mg based on surface area, mg/m 2. The relevance of this finding to humans is unknown.
Parafollicular cell (thyroid) adenomas were increased in high-dose male rats (p=0.003) in a 2-year oral carcinogenicity study at doses of 1 and 3.75 mg/kg body weight. These doses are equivalent to approximately 0.3 and 1 times the 40 mg clinical daily dose based on surface area, mg/m 2. The relevance of this finding to humans is unknown.
Alendronate was not genotoxic in the in vitro microbial mutagenesis assay with and without metabolic activation, in an in vitro mammalian cell mutagenesis assay, in an in vitro alkaline elution assay in rat hepatocytes, and in an in vivo chromosomal aberration assay in mice. In an in vitro chromosomal aberration assay in Chinese hamster ovary cells, however, alendronate gave equivocal results.
Alendronate had no effect on fertility (male or female) in rats at oral doses up to 5 mg/kg/day (approximately 1 times the 40 mg clinical daily dose based on surface area, mg/m 2).
Principal Display Panel 35 Mg Tablet Blister Pack Carton
Hangzhou Minsheng Binjiang Pharma
4 Tablets
Rx only
NDC 60723-207-31
Once Weekly
Alendronate Sodium Tablets, USP 35 mg*
*
EACH TABLET CONTAINS 45.69 mg alendronate sodium (35 mg free acid equivalent).
USUAL ADULT DOSAGE: One 35 mg tablet once weekly. See accompanying package insert
for complete dosage information.
PHARMACIST: Dispense the enclosed Medication Guide to
each patient.
Apply Prescription Label Here
Principal Display Panel 70 Mg Tablet Blister Pack Carton
Hangzhou Minsheng Binjiang Pharma
4 Tablets
Rx only
NDC 60723-205-31
Once Weekly
Alendronate Sodium Tablets, USP 70 mg*
*
EACH TABLET CONTAINS 91.37 mg alendronate sodium (70 mg free acid equivalent).
USUAL ADULT DOSAGE: One 70 mg tablet once weekly. See accompanying package
insert for complete dosage information.
PHARMACIST: Dispense the enclosed Medication Guide to
each patient.
Apply Prescription Label Here
1.3 Treatment to Increase Bone Mass in Men With Osteoporosis
Alendronate sodium tablets, USP are indicated for treatment to increase bone mass in men with osteoporosis [see Clinical Studies (14.3)] .
2.3 Treatment to Increase Bone Mass in Men With Osteoporosis
The recommended dosage is:
| ● one 70 mg (alendronate) tablet once weekly |
| or |
| ● one 10 mg (alendronate) tablet once daily |
14.3 Treatment to Increase Bone Mass in Men With Osteoporosis
The efficacy of alendronate sodium in men with hypogonadal or idiopathic osteoporosis was demonstrated in two clinical studies.
2.7 Recommendations for Calcium and Vitamin D Supplementation
Instruct patients to take supplemental calcium if dietary intake is inadequate [see Warnings and Precautions (5.2)] . Patients at increased risk for vitamin D insufficiency (e.g., over the age of 70 years, nursing home-bound, or chronically ill) may need vitamin D supplementation. Patients with gastrointestinal malabsorption syndromes may require higher doses of vitamin D supplementation and measurement of 25-hydroxyvitamin D should be considered.
Patients treated with glucocorticoids should receive adequate amounts of calcium and vitamin D.
5.5 Atypical Subtrochanteric and Diaphyseal Femoral Fractures
Atypical, low-energy, or low trauma fractures of the femoral shaft have been reported in bisphosphonate-treated patients. These fractures can occur anywhere in the femoral shaft from just below the lesser trochanter to above the supracondylar flare and are transverse or short oblique in orientation without evidence of comminution. Causality has not been established as these fractures also occur in osteoporotic patients who have not been treated with bisphosphonates.
Atypical femur fractures most commonly occur with minimal or no trauma to the affected area. They may be bilateral and many patients report prodromal pain in the affected area, usually presenting as dull, aching thigh pain, weeks to months before a complete fracture occurs. A number of reports note that patients were also receiving treatment with glucocorticoids (e.g. prednisone) at the time of fracture.
Any patient with a history of bisphosphonate exposure who presents with thigh or groin pain should be suspected of having an atypical fracture and should be evaluated to rule out an incomplete femur fracture. Patients presenting with an atypical fracture should also be assessed for symptoms and signs of fracture in the contralateral limb. Interruption of bisphosphonate therapy should be considered, pending a risk/benefit assessment, on an individual basis.
17.1 Osteoporosis Recommendations, Including Calcium and Vitamin D Supplementation
Instruct patients to take supplemental calcium and vitamin D, if daily dietary intake is inadequate. Weight-bearing exercise should be considered along with the modification of certain behavioral factors, such as cigarette smoking and/or excessive alcohol consumption, if these factors exist.
Structured Label Content
Dosage Forms and Strengths (34069-5)
How Supplied
Alendronate sodium tablets USP, 35 mg (alendronate), are modified oval, white to off-white tablets; one side debossed 'AP207', the other side blank.
NDC 60723-207-31 blister packs of 4 with child resistant package.
NDC 60723-207-32 unit-of-use packages of 12 with child resistant package.
NDC 60723-207-33 unit dose packages of 20. This package not intended for household use. For institutional use only.
Alendronate sodium tablets USP, 70 mg (alendronate), are modified oval, white to off-white tablets; one side debossed 'AP205', the other side blank.
NDC 60723-205-31 blister packs of 4 with child resistant package.
NDC 60723-205-32 unit-of-use packages of 12 with child resistant package.
NDC 60723-205-33 unit dose packages of 20. This package not intended for household use. For institutional use only.
Section 34077-8 (34077-8)
Risk Summary
Available data on the use of Alendronate sodium in pregnant women are insufficient to inform a drug-associated risk of adverse maternal or fetal outcomes. Discontinue Alendronate sodium when pregnancy is recognized.
In animal reproduction studies, daily oral administration of alendronate to rats from before mating through the end of gestation or lactation showed decreased postimplantation survival and decreased pup body weight gain starting at doses equivalent to less than half of the highest recommended 40 mg clinical daily dose (based on body surface area, mg/m 2). Oral administration of alendronate to rats during organogenesis resulted in reduced fetal ossification starting at doses 3 times the 40 mg clinical daily dose. No similar fetal effects were observed in pregnant rabbits dosed orally during organogenesis at doses equivalent to approximately 10 times the 40 mg clinical daily dose.
Delayed or failed delivery of offspring, protracted parturition, and late pregnancy maternal and fetal deaths due to maternal hypocalcemia occurred in rats at oral doses as low as one tenth the 40 mg clinical daily dose (see Data).
Bisphosphonates are incorporated into the bone matrix, from which they are gradually released over a period of years. The amount of bisphosphonate incorporated into adult bone and available for release into the systemic circulation is directly related to the dose and duration of bisphosphonate use. Consequently, based on the mechanism of action of bisphosphonates, there is a potential risk of fetal harm, predominantly skeletal, if a woman becomes pregnant after completing a course of bisphosphonate therapy. The impact of variables such as time between cessation of bisphosphonate therapy to conception, the particular bisphosphonate used, and the route of administration (intravenous versus oral) on the risk has not been studied.
The estimated background risk of major birth defects and miscarriage for the indicated population(s) is unknown. All pregnancies have a background risk of birth defects, loss, or other adverse outcomes. In the U.S. general population, the estimated background risks of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Data
Animal Data
Reproduction studies in rats dosed orally from before mating to the end of gestation or lactation showed decreased postimplantation survival starting at 2 mg/kg/day and decreased body weight gain starting at 1 mg/kg/day, doses equivalent to less than half the 40 mg clinical daily dose based on body surface area, mg/m 2. Incidence of incomplete fetal ossification in vertebral, skull, and sternebral bones were increased in rats dosed orally during organogenesis starting at 10 mg/kg/day (approximately 3 times the 40 mg clinical daily dose) . No similar fetal effects were observed in pregnant rabbits dosed orally during organogenesis at up to 35 mg/kg/day (equivalent to approximately 10 times the 40 mg clinical daily dose.
Both total and ionized calcium decreased in pregnant rats dosed orally with 15 mg/kg/day alendronate (approximately 4 times the 40 mg clinical daily dose) resulting in delays and failures of delivery. Protracted parturition due to maternal hypocalcemia was observed when rats were treated from before mating through gestation starting at 0.5 mg/kg/day (approximately one tenth the 40 mg clinical daily dose). Maternotoxicity (late pregnancy deaths) also occurred in the female rats treated orally with 15 mg/kg/day (approximately 4 times the 40 mg clinical daily dose) for varying gestational time periods. These maternal deaths were lessened but not eliminated by cessation of treatment. Calcium supplementation in the drinking water or by subcutaneous minipump to rats dosed orally with 15 mg/kg/day alendronate could not ameliorate the hypocalcemia or prevent the dystocia-related maternal and neonatal deaths. However, intravenous calcium supplementation prevented maternal, but not neonatal deaths.
Section 42229-5 (42229-5)
Re-treatment of Paget's Disease
Re-treatment with alendronate sodium tablets may be considered, following a six-month post-treatment evaluation period in patients who have relapsed, based on increases in serum alkaline phosphatase, which should be measured periodically. Re-treatment may also be considered in those who failed to normalize their serum alkaline phosphatase.
Section 44425-7 (44425-7)
Storage
Store in a well-closed container at 20° - 25°C (68° - 77°F); excursions permitted to 15° - 30°C (59° - 86°F) [see USP Controlled Room Temperature].
7.2 Aspirin
In clinical studies, the incidence of upper gastrointestinal adverse events was increased in patients receiving concomitant therapy with daily doses of alendronate greater than 10 mg and aspirin-containing products.
10 Overdosage (10 OVERDOSAGE)
Significant lethality after single oral doses was seen in female rats and mice at 552 mg/kg (3256 mg/m 2) and 966 mg/kg (2898 mg/m 2), respectively. In males, these values were slightly higher, 626 and 1280 mg/kg, respectively. There was no lethality in dogs at oral doses up to 200 mg/kg (4000 mg/m 2).
No specific information is available on the treatment of overdosage with alendronate sodium. Hypocalcemia, hypophosphatemia, and upper gastrointestinal adverse events, such as upset stomach, heartburn, esophagitis, gastritis, or ulcer, may result from oral overdosage. Milk or antacids should be given to bind alendronate. Due to the risk of esophageal irritation, vomiting should not be induced and the patient should remain fully upright.
Dialysis would not be beneficial.
8.2 Lactation
Risk Summary
It is not known whether alendronate is present in human breast milk, affects human milk production, or has effects on the breastfed infant. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for alendronate sodium and any potential adverse effects on the breastfed child from alendronate sodium or from the underlying maternal condition.
11 Description (11 DESCRIPTION)
Alendronate sodium is a bisphosphonate that acts as a specific inhibitor of osteoclast-mediated bone resorption.
Bisphosphonates are synthetic analogs of pyrophosphate that bind to the hydroxyapatite found in bone.
Alendronate sodium is chemically described as (4-amino-1-hydroxybutylidene) bisphosphonic acid monosodium salt trihydrate. The empirical formula of alendronate sodium is C 4H 12NNaO 7P 2∙3H 2O and its formula weight is 325.12. The structural formula is:
Alendronate sodium is a white, crystalline, nonhygroscopic powder. It is soluble in water, very slightly soluble in alcohol, and practically insoluble in chloroform.
Alendronate sodium tablets, USP for oral administration contain 6.53, 13.06, 45.69 or 91.37 mg of alendronate monosodium salt trihydrate, which is the molar equivalent of 5, 10, 35 and 70 mg, respectively, of free acid, and the following inactive ingredients: microcrystalline cellulose, croscarmellose sodium, and magnesium stearate.
Medication Guide
Read the Medication Guide that comes with Alendronate Sodium Tablets before you start taking it and each time you get a refill. There may be new information. This Medication Guide does not take the place of talking with your doctor about your medical condition or treatment. Talk to your doctor if you have any questions about Alendronate Sodium Tablets.
What is the most important information I should know about alendronate sodium tablets?
Alendronate sodium tablets can cause serious side effects including:
- Esophagus problems
- Low calcium levels in your blood (hypocalcemia)
- Bone, joint, or muscle pain
- Severe jaw bone problems (osteonecrosis)
- Unusual thigh bone fractures
- Esophagus problems.
Some people who take alendronate sodium tablets may develop problems in the esophagus (the tube that connects the mouth and the stomach).
These problems include irritation, inflammation, or ulcers of the esophagus which may sometimes bleed.
- It is important that you take alendronate sodium tablets exactly as prescribed to help lower your chance of getting esophagus problems. (See the section " How should I take alendronate sodium tablets?")
- Stop taking alendronate sodium tablets and call your doctor right away if you get chest pain, new or worsening heartburn, or have trouble or pain when you swallow.
- Low calcium levels in your blood (hypocalcemia).
Alendronate sodium tablets may lower the calcium levels in your blood. If you have low blood calcium before you start taking alendronate sodium tablets, it may get worse during treatment. Your low blood calcium must be treated before you take alendronate sodium tablets. Most people with low blood calcium levels do not have symptoms, but some people may have symptoms. Call your doctor right away if you have symptoms of low blood calcium such as:
- Spasms, twitches, or cramps in your muscles
- Numbness or tingling in your fingers, toes, or around your mouth
Your doctor may prescribe calcium and vitamin D to help prevent low calcium levels in your blood, while you take alendronate sodium tablets. Take calcium and vitamin D as your doctor tells you to.
- Bone, joint, or muscle pain.
Some people who take alendronate sodium tablets develop severe bone, joint, or muscle pain.
- Severe jaw bone problems (osteonecrosis).
Severe jaw bone problems may happen when you take alendronate sodium tablets. Your doctor should examine your mouth before you start alendronate sodium tablets. Your doctor may tell you to see your dentist before you start alendronate sodium tablets. It is important for you to practice good mouth care during treatment with alendronate sodium tablets.
- Unusual thigh bone fractures.
Some people have developed unusual fractures in their thigh bone. Symptoms of a fracture may include new or unusual pain in your hip, groin, or thigh.
Call your doctor right away if you have any of these side effects.
What is alendronate sodium tablet?
Alendronate sodium tablet is a prescription medicine used to:
- Treat or prevent osteoporosis in women after menopause. It helps reduce the chance of having a hip or spinal fracture (break).
- Increase bone mass in men with osteoporosis.
- Treat osteoporosis in either men or women who are taking corticosteroid medicines.
- Treat certain men and women who have Paget's disease of the bone.
It is not known how long alendronate sodium tablet works for the treatment and prevention of osteoporosis. You should see your doctor regularly to determine if alendronate sodium tablet is still right for you.
Alendronate sodium tablet is not for use in children.
Who should not take alendronate sodium tablets?
Do not take alendronate sodium tablets if you:
- Have certain problems with your esophagus, the tube that connects your mouth with your stomach
- Cannot stand or sit upright for at least 30 minutes
- Have low levels of calcium in your blood
- Are allergic to alendronate sodium tablet or any of its ingredients. A list of ingredients is at the end of this leaflet.
What should I tell my doctor before taking alendronate sodium tablets?
Before you start alendronate sodium tablets, be sure to talk to your doctor if you:
- Have problems with swallowing
- Have stomach or digestive problems
- Have low blood calcium
- Plan to have dental surgery or teeth removed
- Have kidney problems
- Have been told you have trouble absorbing minerals in your stomach or intestines (malabsorption syndrome)
- Are pregnant, trying to become pregnant or suspect that you are pregnant. If you become pregnant while taking alendronate sodium tablets, stop taking it and contact your doctor. It is not known if alendronate sodium tablets can harm your unborn baby.
- Are breast-feeding or plan to breast-feed. It is not known if alendronate sodium passes into your milk and may harm your baby.
Especially tell your doctor if you take:
- antacids
- aspirin
- Nonsteroidal Anti-Inflammatory (NSAID) medicines
Tell your doctor about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements. Certain medicines may affect how alendronate sodium tablet works.
Know the medicines you take. Keep a list of them and show it to your doctor and pharmacist each time you get a new medicine.
How should I take alendronate sodium tablets?
- Take alendronate sodium tablets exactly as your doctor tells you.
- Alendronate sodium tablet works only if taken on an empty stomach.
- Take alendronate sodium tablets, after you get up for the day and before taking your first food, drink, or other medicine.
- Take alendronate sodium tablets while you are sitting or standing.
- Do not chew or suck on a tablet of alendronate sodium.
- Swallow alendronate sodium tablet with a full glass (6-8 oz) of plain water only.
- Do not take alendronate sodium tablets with mineral water, coffee, tea, soda, or juice.
- If you take Alendronate Sodium Tablets Daily:
- Take 1 alendronate sodium tablet one time a day, every day after you get up for the day and before taking your first food, drink, or other medicine.
- If you take Once Weekly Alendronate Sodium Tablets:
- Choose the day of the week that best fits your schedule.
- Take 1 dose of alendronate sodium tablets every week on your chosen day after you get up for the day and before taking your first food, drink, or other medicine.
After swallowing alendronate sodium tablet, wait at least 30 minutes:
- Before you lie down. You may sit, stand or walk, and do normal activities like reading.
- Before you take your first food or drink except for plain water.
- Before you take other medicines, including antacids, calcium, and other supplements and vitamins.
Do not lie down for at least 30 minutes after you take alendronate sodium tablets and after you eat your first food of the day.
If you miss a dose of alendronate sodium tablets, do not take it later in the day. Take your missed dose on the next morning after you remember and then return to your normal schedule. Do not take 2 doses on the same day.
If you take too much alendronate sodium, call your doctor. Do not try to vomit. Do not lie down.
What are the possible side effects of alendronate sodium tablets?
Alendronate sodium tablets may cause serious side effects.
The most common side effects of alendronate sodium tablets are:
- Stomach area (abdominal) pain
- Heartburn
- Constipation
- Diarrhea
- Upset stomach
- Pain in your bones, joints, or muscles
- Nausea
You may get allergic reactions, such as hives or swelling of your face, lips, tongue, or throat. Worsening of asthma has been reported.
Tell your doctor if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of alendronate sodium tablets. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to Hangzhou Minsheng Binjiang Pharmaceutical Co., Ltd at 1-888-833-0728 or FDA at 1-800-FDA-1088.
How do I store alendronate sodium tablets?
- Store at 20° - 25°C (68° - 77°F); excursions permitted to 15° - 30°C (59° - 86°F).
- Keep alendronate sodium tablets in a tightly closed container.
Keep alendronate sodium tablets and all medicines out of the reach of children.
General information about the safe and effective use of alendronate sodium tablets.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use alendronate sodium tablets for a condition for which it was not prescribed.
Do not give alendronate sodium tablets to other people, even if they have the same symptoms you have. It may harm them.
This Medication Guide summarizes the most important information about alendronate sodium tablets. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about alendronate sodium tablets that is written for health professionals.
For more information, call 1-888-833-0728.
What are the ingredients in alendronate sodium tablets?
Active ingredient: alendronate sodium
Inactive ingredients: microcrystalline cellulose, croscarmellose sodium, and magnesium stearate.
Rx only
Manufactured by:
Hangzhou Minsheng Binjiang Pharmaceutical Co., Ltd.
Hangzhou, 310051, CHINA
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Revised: 02/2020
8.4 Pediatric Use
Alendronate is not indicated for use in pediatric patients.
The safety and efficacy of alendronate sodium were examined in a randomized, double-blind, placebo-controlled two-year study of 139 pediatric patients, aged 4-18 years, with severe osteogenesis imperfecta (OI). One-hundred-and-nine patients were randomized to 5 mg alendronate daily (weight less than 40 kg) or 10 mg alendronate daily (weight greater than or equal to 40 kg) and 30 patients to placebo. The mean baseline lumbar spine BMD Z-score of the patients was -4.5. The mean change in lumbar spine BMD Z-score from baseline to Month 24 was 1.3 in the alendronate sodium-treated patients and 0.1 in the placebo-treated patients. Treatment with alendronate sodium did not reduce the risk of fracture. Sixteen percent of the alendronate sodium patients who sustained a radiologically-confirmed fracture by Month 12 of the study had delayed fracture healing (callus remodeling) or fracture non-union when assessed radiographically at Month 24 compared with 9% of the placebo-treated patients. In alendronate sodium-treated patients, bone histomorphometry data obtained at Month 24 demonstrated decreased bone turnover and delayed mineralization time; however, there were no mineralization defects. There were no statistically significant differences between the alendronate sodium and placebo groups in reduction of bone pain. The oral bioavailability in children was similar to that observed in adults.
The overall safety profile of alendronate sodium in osteogenesis imperfecta patients treated for up to 24 months was generally similar to that of adults with osteoporosis treated with alendronate sodium. However, there was an increased occurrence of vomiting in osteogenesis imperfecta patients treated with alendronate sodium compared to placebo. During the 24-month treatment period, vomiting was observed in 32 of 109 (29.4%) patients treated with alendronate sodium and 3 of 30 (10%) patients treated with placebo.
In a pharmacokinetic study, 6 of 24 pediatric osteogenesis imperfecta patients who received a single oral dose of alendronate sodium 35 or 70 mg developed fever, flu-like symptoms, and/or mild lymphocytopenia within 24 to 48 hours after administration. These events, lasting no more than 2 to 3 days and responding to acetaminophen, are consistent with an acute-phase response that has been reported in patients receiving bisphosphonates, including alendronate sodium. [See Adverse Reactions (6.2).]
8.5 Geriatric Use
Of the patients receiving alendronate sodium in the Fracture Intervention Trial (FIT), 71% (n=2302) were greater than or equal to 65 years of age and 17% (n=550) were greater than or equal to 75 years of age. Of the patients receiving alendronate sodium in the United States and Multinational osteoporosis treatment studies in women, osteoporosis studies in men, glucocorticoid-induced osteoporosis studies, and Paget's disease studies [see Clinical Studies (14.1), (14.3), (14.4), (14.5)] , 45%, 54%, 37%, and 70%, respectively, were 65 years of age or over. No overall differences in efficacy or safety were observed between these patients and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
4 Contraindications (4 CONTRAINDICATIONS)
Alendronate sodium is contraindicated in patients with the following conditions:
- Abnormalities of the esophagus which delay esophageal emptying such as stricture or achalasia [see Warnings and Precautions (5.1)]
- Inability to stand or sit upright for at least 30 minutes [see Dosage and Administration (2.6); Warnings and Precautions (5.1)]
- Hypocalcemia [see Warnings and Precautions (5.2)]
- Hypersensitivity to any component of this product. Hypersensitivity reactions including urticaria and angioedema have been reported [see Adverse Reactions (6.2)] .
6 Adverse Reactions (6 ADVERSE REACTIONS)
Most common adverse reactions (greater than or equal to 3%) are abdominal pain, acid regurgitation, constipation, diarrhea, dyspepsia, musculoskeletal pain, nausea. (
6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Hangzhou Minsheng Binjiang Pharmaceutical Co., Ltd at 1-888-833-0728 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions (7 DRUG INTERACTIONS)
5.6 Renal Impairment
Alendronate sodium is not recommended for patients with creatinine clearance less than 35 mL/min.
8.6 Renal Impairment
Alendronate sodium is not recommended for patients with creatinine clearance less than 35 mL/min. No dosage adjustment is necessary in patients with creatinine clearance values between 35-60 mL/min [see Clinical Pharmacology (12.3)] .
12.2 Pharmacodynamics
Alendronate is a bisphosphonate that binds to bone hydroxyapatite and specifically inhibits the activity of osteoclasts, the bone-resorbing cells. Alendronate reduces bone resorption with no direct effect on bone formation, although the latter process is ultimately reduced because bone resorption and formation are coupled during bone turnover.
5.2 Mineral Metabolism
Hypocalcemia must be corrected before initiating therapy with alendronate sodium [see Contraindications (4)] . Other disorders affecting mineral metabolism (such as vitamin D deficiency) should also be effectively treated. In patients with these conditions, serum calcium and symptoms of hypocalcemia should be monitored during therapy with alendronate sodium.
Presumably due to the effects of alendronate sodium on increasing bone mineral, small, asymptomatic decreases in serum calcium and phosphate may occur, especially in patients with Paget's disease, in whom the pretreatment rate of bone turnover may be greatly elevated, and in patients receiving glucocorticoids, in whom calcium absorption may be decreased.
Ensuring adequate calcium and vitamin D intake is especially important in patients with Paget's disease of bone and in patients receiving glucocorticoids.
8.7 Hepatic Impairment
As there is evidence that alendronate is not metabolized or excreted in the bile, no studies were conducted in patients with hepatic impairment. No dosage adjustment is necessary [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Alendronate sodium is a bisphosphonate indicated for:
- Treatment and prevention of osteoporosis in postmenopausal women ( 1.1, 1.2)
- Treatment to increase bone mass in men with osteoporosis ( 1.3)
- Treatment of glucocorticoid-induced osteoporosis ( 1.4)
- Treatment of Paget's disease of bone ( 1.5)
Limitations of use: Optimal duration of use has not been determined. For patients at low-risk for fracture, consider drug discontinuation after 3 to 5 years of use. ( 1.6)
12.1 Mechanism of Action
Animal studies have indicated the following mode of action. At the cellular level, alendronate shows preferential localization to sites of bone resorption, specifically under osteoclasts. The osteoclasts adhere normally to the bone surface but lack the ruffled border that is indicative of active resorption. Alendronate does not interfere with osteoclast recruitment or attachment, but it does inhibit osteoclast activity. Studies in mice on the localization of radioactive [ 3H]alendronate in bone showed about 10-fold higher uptake on osteoclast surfaces than on osteoblast surfaces. Bones examined 6 and 49 days after [ 3H]alendronate administration in rats and mice, respectively, showed that normal bone was formed on top of the alendronate, which was incorporated inside the matrix. While incorporated in bone matrix, alendronate is not pharmacologically active. Thus, alendronate must be continuously administered to suppress osteoclasts on newly formed resorption surfaces. Histomorphometry in baboons and rats showed that alendronate treatment reduces bone turnover (i.e., the number of sites at which bone is remodeled). In addition, bone formation exceeds bone resorption at these remodeling sites, leading to progressive gains in bone mass.
17.2 Dosing Instructions
Instruct patients that the expected benefits of alendronate sodium may only be obtained when it is taken with plain water the first thing upon arising for the day at least 30 minutes before the first food, beverage, or medication of the day.
Even dosing with orange juice or coffee has been shown to markedly reduce the absorption of alendronate sodium [see Clinical Pharmacology (12.3)].
Instruct patients not to chew or suck on the tablet because of a potential for oropharyngeal ulceration.
Instruct patients to swallow each tablet of alendronate sodium with a full glass of water (6-8 ounces) to facilitate delivery to the stomach and thus reduce the potential for esophageal irritation.
Instruct patients not to lie down for at least 30 minutes and until after their first food of the day.
Instruct patients not to take alendronate sodium at bedtime or before arising for the day. Patients should be informed that failure to follow these instructions may increase their risk of esophageal problems.
Instruct patients that if they develop symptoms of esophageal disease (such as difficulty or pain upon swallowing, retrosternal pain or new or worsening heartburn) they should stop taking alendronate sodium and consult their physician.
If patients miss a dose of once weekly alendronate sodium tablet, instruct patients to take one dose on the morning after they remember. They should not take two doses on the same day but should return to taking one dose once a week, as originally scheduled on their chosen day.
Manufactured by:
Hangzhou Minsheng Binjiang
Pharmaceutical Co., Ltd,
658 Bin’an Road Binjiang District,
Hangzhou, 310051, CHINA
Revised: 02/2020
5.3 Musculoskeletal Pain
In post-marketing experience, severe and occasionally incapacitating bone, joint, and/or muscle pain has been reported in patients taking bisphosphonates that are approved for the prevention and treatment of osteoporosis [see Adverse Reactions (6.2)] . This category of drugs includes alendronate. Most of the patients were postmenopausal women. The time to onset of symptoms varied from one day to several months after starting the drug. Discontinue use if severe symptoms develop. Most patients had relief of symptoms after stopping. A subset had recurrence of symptoms when rechallenged with the same drug or another bisphosphonate.
In placebo-controlled clinical studies of alendronate sodium, the percentages of patients with these symptoms were similar in the alendronate sodium and placebo groups.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Upper Gastrointestinal Adverse Reactions can occur. Instruct patients to follow dosing instructions. Discontinue if new or worsening symptoms occur. ( 5.1)
- Hypocalcemia can worsen and must be corrected prior to use. ( 5.2)
- Severe Bone, Joint, Muscle Pain may occur. Discontinue use if severe symptoms develop. ( 5.3)
- Osteonecrosis of the Jaw has been reported. ( 5.4)
- Atypical Femur Fractures have been reported. Patients with new thigh or groin pain should be evaluated to rule out an incomplete femoral fracture. ( 5.5)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Treatment of osteoporosis in postmenopausal women and in men: 10 mg daily or 70 mg tablet once weekly. ( 2.1, 2.3)
- Prevention of osteoporosis in postmenopausal women: 5 mg daily or 35 mg once weekly. ( 2.2)
- Glucocorticoid-induced osteoporosis: 5 mg daily; or 10 mg daily in postmenopausal women not receiving estrogen. ( 2.4)
- Paget's disease: 40 mg daily for six months. ( 2.5)
- Instruct patients to: (
2.6)
- Swallow tablets whole with 6-8 ounces plain water at least 30 minutes before the first food, drink, or medication of the day.
- Not lie down for at least 30 minutes after taking alendronate sodium tablet and until after food.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- 5 mg (alendronate) tablets are round, flat face beveled edged, white to off-white tablets; one side debossed "AP209", the other side blank.
- 10 mg (alendronate) tablets are modified oval, white to off-white tablets; one side debossed "AP208", the other side blank.
- 35 mg (alendronate) tablets are modified oval, white to off-white tablets; one side debossed "AP207", the other side blank.
- 70 mg (alendronate) tablets are modified oval, white to off-white tablets; one side debossed "AP205", the other side blank.
5.4 Osteonecrosis of the Jaw
Osteonecrosis of the jaw (ONJ), which can occur spontaneously, is generally associated with tooth extraction and/or local infection with delayed healing, and has been reported in patients taking bisphosphonates, including alendronate sodium. Known risk factors for osteonecrosis of the jaw include invasive dental procedures (e.g., tooth extraction, dental implants, boney surgery), diagnosis of cancer, concomitant therapies (e.g., chemotherapy, corticosteroids, angiogenesis inhibitors), poor oral hygiene, and co-morbid disorders (e.g., periodontal and/or other pre-existing dental disease, anemia, coagulopathy, infection, ill-fitting dentures). The risk of ONJ may increase with duration of exposure to bisphosphonates.
For patients requiring invasive dental procedures, discontinuation of bisphosphonate treatment may reduce the risk for ONJ. Clinical judgment of the treating physician and/or oral surgeon should guide the management plan of each patient based on individual benefit/risk assessment.
Patients who develop osteonecrosis of the jaw while on bisphosphonate therapy should receive care by an oral surgeon. In these patients, extensive dental surgery to treat ONJ may exacerbate the condition. Discontinuation of bisphosphonate therapy should be considered based on individual benefit/risk assessment.
6.2 Post Marketing Experience (6.2 Post-Marketing Experience)
The following adverse reactions have been identified during post-approval use of alendronate. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Body as a Whole: hypersensitivity reactions including urticaria and angioedema. Transient symptoms of myalgia, malaise, asthenia and fever have been reported with alendronate, typically in association with initiation of treatment. Symptomatic hypocalcemia has occurred, generally in association with predisposing conditions. Peripheral edema.
Gastrointestinal: esophagitis, esophageal erosions, esophageal ulcers, esophageal stricture or perforation, and oropharyngeal ulceration. Gastric or duodenal ulcers, some severe and with complications, have also been reported [see Dosage and Administration (2.6); Warnings and Precautions (5.1)].
Localized osteonecrosis of the jaw, generally associated with tooth extraction and/or local infection with delayed healing, has been reported [see Warnings and Precautions (5.4)].
Musculoskeletal: bone, joint, and/or muscle pain, occasionally severe, and incapacitating [see Warnings and Precautions (5.3)]; joint swelling; low-energy femoral shaft and subtrochanteric fractures [see Warnings and Precautions (5.5)].
Nervous System: dizziness and vertigo.
Pulmonary: acute asthma exacerbations.
Skin: rash (occasionally with photosensitivity), pruritus, alopecia, severe skin reactions, including Stevens-Johnson syndrome and toxic epidermal necrolysis.
Special Senses: uveitis, scleritis or episcleritis. Cholesteatoma of the external auditory canal (focal osteonecrosis)
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
1.6 Important Limitations of Use
The optimal duration of use has not been determined. The safety and effectiveness of alendronate sodium tablets, USP for the treatment of osteoporosis are based on clinical data of four years duration. All patients on bisphosphonate therapy should have the need for continued therapy re-evaluated on a periodic basis. Patients at low-risk for fracture should be considered for drug discontinuation after 3 to 5 years of use. Patients who discontinue therapy should have their risk for fracture re-evaluated periodically.
7.1 Calcium Supplements/antacids (7.1 Calcium Supplements/Antacids)
Co-administration of alendronate sodium and calcium, antacids, or oral medications containing multivalent cations will interfere with absorption of alendronate sodium. Therefore, instruct patients to wait at least one-half hour after taking alendronate sodium before taking any other oral medications.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Instruct patients to read the Medication Guide before starting therapy with alendronate sodium and to reread it each time the prescription is renewed.
5.7 Glucocorticoid Induced Osteoporosis (5.7 Glucocorticoid-Induced Osteoporosis)
The risk versus benefit of alendronate sodium for treatment at daily dosages of glucocorticoids less than 7.5 mg of prednisone or equivalent has not been established [see Indications and Usage (1.4)] . Before initiating treatment, the gonadal hormonal status of both men and women should be ascertained and appropriate replacement considered.
A bone mineral density measurement should be made at the initiation of therapy and repeated after 6 to 12 months of combined alendronate sodium and glucocorticoid treatment.
1.5 Treatment of Paget's Disease of Bone
Alendronate sodium tablets, USP are indicated for the treatment of Paget's disease of bone in men and women. Treatment is indicated in patients with Paget's disease of bone who have alkaline phosphatase at least two times the upper limit of normal, or those who are symptomatic, or those at risk for future complications from their disease. [See Clinical Studies (14.5).]
2.5 Treatment of Paget's Disease of Bone
The recommended treatment regimen is 40 mg once a day for six months.
7.3 Nonsteroidal Anti Inflammatory Drugs (7.3 Nonsteroidal Anti-Inflammatory Drugs)
Alendronate sodium may be administered to patients taking nonsteroidal anti-inflammatory drugs (NSAIDs). In a 3-year, controlled, clinical study (n=2027) during which a majority of patients received concomitant NSAIDs, the incidence of upper gastrointestinal adverse events was similar in patients taking alendronate 5 or 10 mg/day compared to those taking placebo. However, since NSAID use is associated with gastrointestinal irritation, caution should be used during concomitant use with alendronate sodium.
14.5 Treatment of Paget's Disease of Bone
The efficacy of alendronate 40 mg once daily for six months was demonstrated in two double-blind clinical studies of male and female patients with moderate to severe Paget's disease (alkaline phosphatase at least twice the upper limit of normal): a placebo-controlled, multinational study and a U.S. comparative study with etidronate disodium 400 mg/day. Figure 6 shows the mean percent changes from baseline in serum alkaline phosphatase for up to six months of randomized treatment.
|
Figure 6:
Studies in Paget's Disease of Bone
Effect on Serum Alkaline Phosphatase of Alendronate 40 mg/day Versus Placebo or Etidronate 400 mg/day |
At six months the suppression in alkaline phosphatase in patients treated with alendronate sodium was significantly greater than that achieved with etidronate and contrasted with the complete lack of response in placebo-treated patients. Response (defined as either normalization of serum alkaline phosphatase or decrease from baseline greater than or equal to 60%) occurred in approximately 85% of patients treated with alendronate sodium in the combined studies vs. 30% in the etidronate group and 0% in the placebo group. Alendronate sodium was similarly effective regardless of age, gender, race, prior use of other bisphosphonates, or baseline alkaline phosphatase within the range studied (at least twice the upper limit of normal). Bone histology was evaluated in 33 patients with Paget's disease treated with alendronate 40 mg/day for 6 months. As in patients treated for osteoporosis [see Clinical Studies (14.1)], alendronate sodium did not impair mineralization, and the expected decrease in the rate of bone turnover was observed. Normal lamellar bone was produced during treatment with alendronate sodium, even where preexisting bone was woven and disorganized. Overall, bone histology data support the conclusion that bone formed during treatment with alendronate sodium is of normal quality.
2.6 Important Administration Instructions
Instruct patients to do the following:
- Take alendronate sodium tablets at least one-half hour before the first food, beverage, or medication of the day with plain water only [see Patient Counseling Information (17.2)] . Other beverages (including mineral water), food, and some medications are likely to reduce the absorption of alendronate sodium tablets [see Drug Interactions (7.1)] . Waiting less than 30 minutes, or taking alendronate sodium tablets with food, beverages (other than plain water) or other medications will lessen the effect of alendronate sodium tablets by decreasing its absorption into the body.
- Take alendronate sodium tablets upon arising for the day. To facilitate delivery to the stomach and thus reduce the potential for esophageal irritation, an alendronate sodium tablet should be swallowed with a full glass of water (6-8 ounces). Patients should not lie down for at least 30 minutes and until after their first food of the day. Alendronate sodium tablets should not be taken at bedtime or before arising for the day. Failure to follow these instructions may increase the risk of esophageal adverse experiences [see Warnings and Precautions (5.1) and Patient Counseling Information (17.2)] .
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
The relative inhibitory activities on bone resorption and mineralization of alendronate and etidronate were compared in the Schenk assay, which is based on histological examination of the epiphyses of growing rats. In this assay, the lowest dose of alendronate that interfered with bone mineralization (leading to osteomalacia) was 6000-fold the antiresorptive dose. The corresponding ratio for etidronate was one to one. These data suggest that alendronate administered in therapeutic doses is highly unlikely to induce osteomalacia.
5.1 Upper Gastrointestinal Adverse Reactions
Alendronate sodium, like other bisphosphonates administered orally, may cause local irritation of the upper gastrointestinal mucosa. Because of these possible irritant effects and a potential for worsening of the underlying disease, caution should be used when alendronate sodium is given to patients with active upper gastrointestinal problems (such as known Barrett's esophagus, dysphagia, other esophageal diseases, gastritis, duodenitis, or ulcers).
Esophageal adverse experiences, such as esophagitis, esophageal ulcers and esophageal erosions, occasionally with bleeding and rarely followed by esophageal stricture or perforation, have been reported in patients receiving treatment with oral bisphosphonates including alendronate sodium. In some cases these have been severe and required hospitalization. Physicians should therefore be alert to any signs or symptoms signaling a possible esophageal reaction and patients should be instructed to discontinue alendronate sodium and seek medical attention if they develop dysphagia, odynophagia, retrosternal pain or new or worsening heartburn.
The risk of severe esophageal adverse experiences appears to be greater in patients who lie down after taking oral bisphosphonates including alendronate sodium and/or who fail to swallow oral bisphosphonates including alendronate sodium with the recommended full glass (6-8 ounces) of water, and/or who continue to take oral bisphosphonates including alendronate sodium after developing symptoms suggestive of esophageal irritation. Therefore, it is very important that the full dosing instructions are provided to, and understood by, the patient [see Dosage and Administration (2.6)] . In patients who cannot comply with dosing instructions due to mental disability, therapy with alendronate sodium should be used under appropriate supervision.
There have been post-marketing reports of gastric and duodenal ulcers with oral bisphosphonate use, some severe and with complications, although no increased risk was observed in controlled clinical trials [see Adverse Reactions (6.2)] .
2.8 Administration Instructions for Missed Doses
If a once-weekly dose of alendronate sodium tablet is missed, instruct patients to take one dose on the morning after they remember. They should not take two doses on the same day but should return to taking one dose once a week, as originally scheduled on their chosen day.
1.4 Treatment of Glucocorticoid Induced Osteoporosis (1.4 Treatment of Glucocorticoid-Induced Osteoporosis)
Alendronate sodium tablets, USP are indicated for the treatment of glucocorticoid-induced osteoporosis in men and women receiving glucocorticoids in a daily dosage equivalent to 7.5 mg or greater of prednisone and who have low bone mineral density [see Clinical Studies (14.4)] .
2.4 Treatment of Glucocorticoid Induced Osteoporosis (2.4 Treatment of Glucocorticoid-Induced Osteoporosis)
The recommended dosage is one 5 mg tablet once daily, except for postmenopausal women not receiving estrogen, for whom the recommended dosage is one 10 mg tablet once daily.
1.1 Treatment of Osteoporosis in Postmenopausal Women
Alendronate sodium tablets, USP are indicated for the treatment of osteoporosis in postmenopausal women. In postmenopausal women, alendronate sodium tablets, USP increase bone mass and reduce the incidence of fractures, including those of the hip and spine (vertebral compression fractures). [See Clinical Studies (14.1).]
14.4 Treatment of Glucocorticoid Induced Osteoporosis (14.4 Treatment of Glucocorticoid-Induced Osteoporosis)
The efficacy of alendronate 5 and 10 mg once daily in men and women receiving glucocorticoids (at least 7.5 mg/day of prednisone or equivalent) was demonstrated in two, one-year, double-blind, randomized, placebo-controlled, multicenter studies of virtually identical design, one performed in the United States and the other in 15 different countries (Multinational [which also included alendronate 2.5 mg/day]). These studies enrolled 232 and 328 patients, respectively, between the ages of 17 and 83 with a variety of glucocorticoid-requiring diseases. Patients received supplemental calcium and vitamin D. Figure 5 shows the mean increases relative to placebo in BMD of the lumbar spine, femoral neck, and trochanter in patients receiving alendronate 5 mg/day for each study.
|
Figure 5:
Studies in Glucocorticoid - Treated Patients
Increase in BMD Alendronate 5 mg/day at One Year |
After one year, significant increases relative to placebo in BMD were seen in the combined studies at each of these sites in patients who received alendronate 5 mg/day. In the placebo-treated patients, a significant decrease in BMD occurred at the femoral neck (-1.2%), and smaller decreases were seen at the lumbar spine and trochanter. Total body BMD was maintained with alendronate 5 mg/day. The increases in BMD with alendronate 10 mg/day were similar to those with alendronate 5 mg/day in all patients except for postmenopausal women not receiving estrogen therapy. In these women, the increases (relative to placebo) with alendronate 10 mg/day were greater than those with alendronate 5 mg/day at the lumbar spine (4.1% vs. 1.6%) and trochanter (2.8% vs. 1.7%), but not at other sites. Alendronate sodium was effective regardless of dose or duration of glucocorticoid use. In addition, alendronate sodium was similarly effective regardless of age (less than 65 vs. greater than or equal to 65 years), race (Caucasian vs. other races), gender, underlying disease, baseline BMD, baseline bone turnover, and use with a variety of common medications.
Bone histology was normal in the 49 patients biopsied at the end of one year who received alendronate at doses of up to 10 mg/day.
Of the original 560 patients in these studies, 208 patients who remained on at least 7.5 mg/day of prednisone or equivalent continued into a one-year double-blind extension. After two years of treatment, spine BMD increased by 3.7% and 5.0% relative to placebo with alendronate 5 and 10 mg/day, respectively. Significant increases in BMD (relative to placebo) were also observed at the femoral neck, trochanter, and total body.
After one year, 2.3% of patients treated with alendronate 5 or 10 mg/day (pooled) vs. 3.7% of those treated with placebo experienced a new vertebral fracture (not significant). However, in the population studied for two years, treatment with alendronate (pooled dosage groups: 5 or 10 mg for two years or 2.5 mg for one year followed by 10 mg for one year) significantly reduced the incidence of patients with a new vertebral fracture (alendronate sodium 0.7% vs. placebo 6.8%).
2.1 Treatment of Osteoporosis in Postmenopausal Women
The recommended dosage is:
| ● one 70 mg (alendronate) tablet once weekly |
| or |
| ● one 10 mg (alendronate) tablet once daily |
1.2 Prevention of Osteoporosis in Postmenopausal Women
Alendronate sodium tablets, USP are indicated for the prevention of postmenopausal osteoporosis [see Clinical Studies (14.2)] .
2.2 Prevention of Osteoporosis in Postmenopausal Women
The recommended dosage is:
| ● one 35 mg (alendronate) tablet once weekly |
| or |
| ● one 5 mg (alendronate) tablet once daily |
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Harderian gland (a retro-orbital gland not present in humans) adenomas were increased in high-dose female mice (p=0.003) in a 92-week oral carcinogenicity study at doses of alendronate of 1, 3, and 10 mg/kg/day (males) or 1, 2, and 5 mg/kg/day (females). These doses are equivalent to approximately 0.1 to 1 times the highest recommended clinical daily dose of 40 mg based on surface area, mg/m 2. The relevance of this finding to humans is unknown.
Parafollicular cell (thyroid) adenomas were increased in high-dose male rats (p=0.003) in a 2-year oral carcinogenicity study at doses of 1 and 3.75 mg/kg body weight. These doses are equivalent to approximately 0.3 and 1 times the 40 mg clinical daily dose based on surface area, mg/m 2. The relevance of this finding to humans is unknown.
Alendronate was not genotoxic in the in vitro microbial mutagenesis assay with and without metabolic activation, in an in vitro mammalian cell mutagenesis assay, in an in vitro alkaline elution assay in rat hepatocytes, and in an in vivo chromosomal aberration assay in mice. In an in vitro chromosomal aberration assay in Chinese hamster ovary cells, however, alendronate gave equivocal results.
Alendronate had no effect on fertility (male or female) in rats at oral doses up to 5 mg/kg/day (approximately 1 times the 40 mg clinical daily dose based on surface area, mg/m 2).
Principal Display Panel 35 Mg Tablet Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 35 mg Tablet Blister Pack Carton)
Hangzhou Minsheng Binjiang Pharma
4 Tablets
Rx only
NDC 60723-207-31
Once Weekly
Alendronate Sodium Tablets, USP 35 mg*
*
EACH TABLET CONTAINS 45.69 mg alendronate sodium (35 mg free acid equivalent).
USUAL ADULT DOSAGE: One 35 mg tablet once weekly. See accompanying package insert
for complete dosage information.
PHARMACIST: Dispense the enclosed Medication Guide to
each patient.
Apply Prescription Label Here
Principal Display Panel 70 Mg Tablet Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 70 mg Tablet Blister Pack Carton)
Hangzhou Minsheng Binjiang Pharma
4 Tablets
Rx only
NDC 60723-205-31
Once Weekly
Alendronate Sodium Tablets, USP 70 mg*
*
EACH TABLET CONTAINS 91.37 mg alendronate sodium (70 mg free acid equivalent).
USUAL ADULT DOSAGE: One 70 mg tablet once weekly. See accompanying package
insert for complete dosage information.
PHARMACIST: Dispense the enclosed Medication Guide to
each patient.
Apply Prescription Label Here
1.3 Treatment to Increase Bone Mass in Men With Osteoporosis (1.3 Treatment to Increase Bone Mass in Men with Osteoporosis)
Alendronate sodium tablets, USP are indicated for treatment to increase bone mass in men with osteoporosis [see Clinical Studies (14.3)] .
2.3 Treatment to Increase Bone Mass in Men With Osteoporosis (2.3 Treatment to Increase Bone Mass in Men with Osteoporosis)
The recommended dosage is:
| ● one 70 mg (alendronate) tablet once weekly |
| or |
| ● one 10 mg (alendronate) tablet once daily |
14.3 Treatment to Increase Bone Mass in Men With Osteoporosis (14.3 Treatment to Increase Bone Mass in Men with Osteoporosis)
The efficacy of alendronate sodium in men with hypogonadal or idiopathic osteoporosis was demonstrated in two clinical studies.
2.7 Recommendations for Calcium and Vitamin D Supplementation
Instruct patients to take supplemental calcium if dietary intake is inadequate [see Warnings and Precautions (5.2)] . Patients at increased risk for vitamin D insufficiency (e.g., over the age of 70 years, nursing home-bound, or chronically ill) may need vitamin D supplementation. Patients with gastrointestinal malabsorption syndromes may require higher doses of vitamin D supplementation and measurement of 25-hydroxyvitamin D should be considered.
Patients treated with glucocorticoids should receive adequate amounts of calcium and vitamin D.
5.5 Atypical Subtrochanteric and Diaphyseal Femoral Fractures
Atypical, low-energy, or low trauma fractures of the femoral shaft have been reported in bisphosphonate-treated patients. These fractures can occur anywhere in the femoral shaft from just below the lesser trochanter to above the supracondylar flare and are transverse or short oblique in orientation without evidence of comminution. Causality has not been established as these fractures also occur in osteoporotic patients who have not been treated with bisphosphonates.
Atypical femur fractures most commonly occur with minimal or no trauma to the affected area. They may be bilateral and many patients report prodromal pain in the affected area, usually presenting as dull, aching thigh pain, weeks to months before a complete fracture occurs. A number of reports note that patients were also receiving treatment with glucocorticoids (e.g. prednisone) at the time of fracture.
Any patient with a history of bisphosphonate exposure who presents with thigh or groin pain should be suspected of having an atypical fracture and should be evaluated to rule out an incomplete femur fracture. Patients presenting with an atypical fracture should also be assessed for symptoms and signs of fracture in the contralateral limb. Interruption of bisphosphonate therapy should be considered, pending a risk/benefit assessment, on an individual basis.
17.1 Osteoporosis Recommendations, Including Calcium and Vitamin D Supplementation
Instruct patients to take supplemental calcium and vitamin D, if daily dietary intake is inadequate. Weight-bearing exercise should be considered along with the modification of certain behavioral factors, such as cigarette smoking and/or excessive alcohol consumption, if these factors exist.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:14.817957 · Updated: 2026-03-14T22:39:42.922945