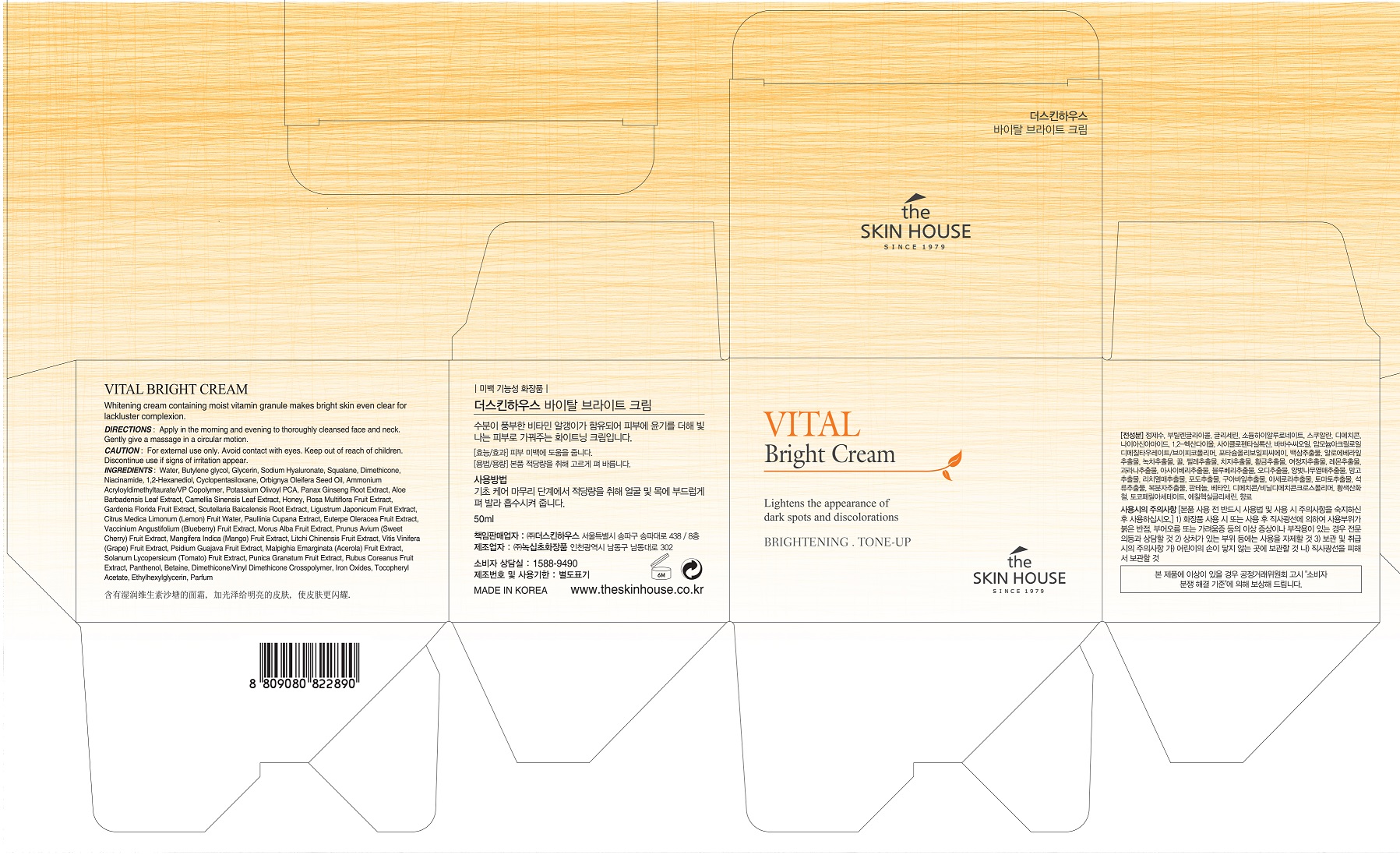
Drug Facts
a21cbf42-482c-2d95-e053-2a95a90a1a48
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Niacinamide
Medication Information
Warnings and Precautions
For external use only
When using this product
■ if the following symptoms occurs after use, stop use and consult with a skin specialist
red specks, swelling, itching
■ don’t use on the part where there is injury, eczema, or dermatitis
Keep out of reach of children
■ if swallowed, get medical help or contact a person control center immediately
Indications and Usage
apply proper amount to the skin
Description
Niacinamide
Section 50565-1
keep out of reach of the children
Section 51727-6
Water
Glycerin
Sodium Hyaluronate
Butylene glycol
Squalane
Dimethicone
1,2-Hexanediol
Cyclopentasiloxane
Orbignya Oleifera Seed Oil
Ammonium Acryloyldimethyltaurate/VP Copolymer
Potassium Olivoyl PCA
Panax Ginseng Root Extract
Aloe Barbadensis Leaf Extract
Camellia Sinensis Leaf Extract
Honey
Rosa Multiflora Fruit Extract
Gardenia Florida Fruit Extract
Scutellaria Baicalensis Root Extract
Ligustrum Japonicum Fruit Extract
Citrus Medica Limonum (Lemon) Fruit Water
Paullinia Cupana Extract
Euterpe Oleracea Fruit Extract
Vaccinium Angustifolium (Blueberry) Fruit Extract
Morus Alba Fruit Extract
Prunus Avium (Sweet Cherry) Fruit Extract
Mangifera Indica (Mango) Fruit Extract
Litchi Chinensis Fruit Extract
Vitis Vinifera (Grape) Fruit Extract
Psidium Guajava Fruit Extract
Malpighia Emarginata (Acerola) Fruit Extract
Solanum Lycopersicum (Tomato) Fruit Extract
Punica Granatum Fruit Extract
Rubus Coreanus Fruit Extract
Panthenol
Tocopheryl Acetate
Betaine
Dimethicone/Vinyl Dimethicone Crosspolymer
Iron Oxides
Ethylhexylglycerin
Parfum
Section 51945-4
Section 55105-1
skin brightening
Section 55106-9
Niacinamide
Structured Label Content
Indications and Usage (34067-9)
apply proper amount to the skin
Warnings and Precautions (34071-1)
For external use only
When using this product
■ if the following symptoms occurs after use, stop use and consult with a skin specialist
red specks, swelling, itching
■ don’t use on the part where there is injury, eczema, or dermatitis
Keep out of reach of children
■ if swallowed, get medical help or contact a person control center immediately
Section 50565-1 (50565-1)
keep out of reach of the children
Section 51727-6 (51727-6)
Water
Glycerin
Sodium Hyaluronate
Butylene glycol
Squalane
Dimethicone
1,2-Hexanediol
Cyclopentasiloxane
Orbignya Oleifera Seed Oil
Ammonium Acryloyldimethyltaurate/VP Copolymer
Potassium Olivoyl PCA
Panax Ginseng Root Extract
Aloe Barbadensis Leaf Extract
Camellia Sinensis Leaf Extract
Honey
Rosa Multiflora Fruit Extract
Gardenia Florida Fruit Extract
Scutellaria Baicalensis Root Extract
Ligustrum Japonicum Fruit Extract
Citrus Medica Limonum (Lemon) Fruit Water
Paullinia Cupana Extract
Euterpe Oleracea Fruit Extract
Vaccinium Angustifolium (Blueberry) Fruit Extract
Morus Alba Fruit Extract
Prunus Avium (Sweet Cherry) Fruit Extract
Mangifera Indica (Mango) Fruit Extract
Litchi Chinensis Fruit Extract
Vitis Vinifera (Grape) Fruit Extract
Psidium Guajava Fruit Extract
Malpighia Emarginata (Acerola) Fruit Extract
Solanum Lycopersicum (Tomato) Fruit Extract
Punica Granatum Fruit Extract
Rubus Coreanus Fruit Extract
Panthenol
Tocopheryl Acetate
Betaine
Dimethicone/Vinyl Dimethicone Crosspolymer
Iron Oxides
Ethylhexylglycerin
Parfum
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
skin brightening
Section 55106-9 (55106-9)
Niacinamide
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:57:05.978577 · Updated: 2026-03-14T22:56:02.443544