These Highlights Do Not Include All The Information Needed To Use Ztlido ®
a1b17507-4560-490d-a388-74e7fd7eaa5e
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dosage and Administration ( 2.1 ) 4/2021
Indications and Usage
ZTLIDO is indicated for relief of pain associated with post-herpetic neuralgia (PHN) in adults.
Dosage and Administration
Because of the difference in bioavailability of ZTLIDO compared to Lidoderm (lidocaine patch 5%), a different dosage strength is required to be administered to the patient. One ZTLIDO (lidocaine topical system) 1.8% provides equivalent lidocaine exposure to one Lidoderm (lidocaine patch 5%) [see Clinical Pharmacology (12.3) ]. When ZTLIDO is used concomitantly with other products containing local anesthetic agents, the total amount of drug absorbed from all formulations must be considered. Clearly instruct and advise patients: to wash hands immediately after handling ZTLIDO and to avoid contact with eyes [see Warnings and Precautions (5.6) ] . to store ZTLIDO inside the sealed envelope out of the reach of children, pets, and others and to apply immediately after removal from the envelope. to fold used ZTLIDO so that the adhesive side sticks to itself and to safely discard used ZTLIDO or pieces of cut ZTLIDO where children and pets cannot get to them [see Warnings and Precautions (5.1) ] . to never apply external heat sources, such as heating pads or electric blankets, directly to ZTLIDO, because plasma lidocaine levels are increased. ZTLIDO can be applied, however, to the administration site after moderate heat exposure, such as 15 minutes of heating pad exposure on a medium setting [see Warnings and Precautions (5.2) , Clinical Pharmacology (12.3) ] . that clothing may be worn over the area of application. that ZTLIDO may be used during moderate exercise, such as biking for 30 minutes. that ZTLIDO may be exposed to water, such as showering for 10 minutes or immersion for 15 minutes. to dry the topical system, after water exposure, by gently patting the skin, not by rubbing the skin or topical system. that ZTLIDO topical systems that have lifted at the edges may be reattached by pressing firmly on the edges and that fully detached topical systems may be reapplied as originally directed. that if a ZTLIDO topical system comes off completely and will not stick to patient's skin, to remove and properly dispose of the used ZTLIDO and to apply a new ZTLIDO topical system for a total duration of 12 hours of used and new topical system together. that if irritation or a burning sensation occurs during application, to remove ZTLIDO and to not reapply until the irritation subsides [see Warnings and Precautions (5.4) ] .
Warnings and Precautions
Accidental Exposure : Even a used ZTLIDO topical system contains residual lidocaine after use. It is important for patients to store and dispose of ZTLIDO properly and keep out of the reach of children, pets, and others. ( 5.1 ) Excessive Dosing/Overexposure : Applying ZTLIDO to larger surface areas or for a longer duration than recommended could lead to increased absorption and high blood concentrations of lidocaine, leading to adverse effects. ( 5.2 ) Increased Absorption on Non-Intact Skin : May result in higher blood concentrations of lidocaine. ( 5.2 ) Risk of Overexposure with External Heat Sources : Applying external heat sources to ZTLIDO may result in increased drug exposure. ( 5.2 ) Methemoglobinemia : Cases of methemoglobinemia have been reported in association with local anesthetic use. ( 5.3 ) Application Site Reactions : During or immediately after treatment with ZTLIDO, application site reactions may develop. ( 5.4 ) Hypersensitivity Reactions : Cross sensitivity to ZTLIDO in patients with a history of drug sensitivity to para-aminobenzoic acid (PABA) derivatives is possible. ( 5.5 ) Eye Exposure : Immediately wash out the eye with water or saline and protect the eye until sensation returns. ( 5.6 ).
Contraindications
ZTLIDO is contraindicated in patients with a known history of sensitivity to local anesthetics of the amide type, or to any other component of the product.
Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling: Excessive Dosing/Overexposure to Lidocaine [see Warnings and Precautions (5.2) ] Methemoglobinemia [see Warnings and Precautions (5.3) ] Application Site Reactions [see Warnings and Precautions (5.4) ] Hypersensitivity Reactions [see Warnings and Precautions (5.5) ] Eye Irritation [see Warnings and Precautions (5.6) ] The following adverse reactions from voluntary reports or clinical studies have been reported with lidocaine. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Skin and subcutaneous tissues : blisters, bruising, burning sensation, depigmentation, dermatitis, discoloration, edema, erosions, erythema, exfoliation, flushing, irritation, papules, petechia, pruritus, vesicles, and abnormal sensation. Immune system : angioedema, bronchospasm, dermatitis, dyspnea, hypersensitivity, laryngospasm, pruritus, shock, and urticaria. Central Nervous System : lightheadedness, nervousness, apprehension, euphoria, confusion, dizziness, drowsiness, tinnitus, blurred or double vision, sensations of heat, cold or numbness, twitching, tremors, convulsions, unconsciousness, somnolence, respiratory depression and arrest. Cardiovascular : bradycardia, hypotension, and cardiovascular collapse leading to arrest. Other : asthenia, disorientation, headache, hyperesthesia, hypoesthesia, metallic taste, nausea, pain exacerbated, paresthesia, taste alteration, and vomiting.
Drug Interactions
Class I Antiarrhythmic Drugs: When ZTLIDO is used in patients receiving Class I antiarrhythmic drugs (such as tocainide and mexiletine) the toxic effects are additive and potentially synergistic. Consider risk/benefit before concomitant use. ( 7.2 ) Local Anesthetic Agents: When ZTLIDO is used concomitantly with other products containing local anesthetic agents, the effects are additive. Consider the amount of drug absorbed from all formulations when local anesthetics are administered concomitantly. ( 7.3 )
Storage and Handling
ZTLIDO (lidocaine topical system) 1.8% is available as the following: Carton of 30 topical systems, packaged into individual child-resistant envelopes. NDC 69557-111-30
How Supplied
ZTLIDO (lidocaine topical system) 1.8% is available as the following: Carton of 30 topical systems, packaged into individual child-resistant envelopes. NDC 69557-111-30
Medication Information
Warnings and Precautions
Accidental Exposure : Even a used ZTLIDO topical system contains residual lidocaine after use. It is important for patients to store and dispose of ZTLIDO properly and keep out of the reach of children, pets, and others. ( 5.1 ) Excessive Dosing/Overexposure : Applying ZTLIDO to larger surface areas or for a longer duration than recommended could lead to increased absorption and high blood concentrations of lidocaine, leading to adverse effects. ( 5.2 ) Increased Absorption on Non-Intact Skin : May result in higher blood concentrations of lidocaine. ( 5.2 ) Risk of Overexposure with External Heat Sources : Applying external heat sources to ZTLIDO may result in increased drug exposure. ( 5.2 ) Methemoglobinemia : Cases of methemoglobinemia have been reported in association with local anesthetic use. ( 5.3 ) Application Site Reactions : During or immediately after treatment with ZTLIDO, application site reactions may develop. ( 5.4 ) Hypersensitivity Reactions : Cross sensitivity to ZTLIDO in patients with a history of drug sensitivity to para-aminobenzoic acid (PABA) derivatives is possible. ( 5.5 ) Eye Exposure : Immediately wash out the eye with water or saline and protect the eye until sensation returns. ( 5.6 ).
Indications and Usage
ZTLIDO is indicated for relief of pain associated with post-herpetic neuralgia (PHN) in adults.
Dosage and Administration
Because of the difference in bioavailability of ZTLIDO compared to Lidoderm (lidocaine patch 5%), a different dosage strength is required to be administered to the patient. One ZTLIDO (lidocaine topical system) 1.8% provides equivalent lidocaine exposure to one Lidoderm (lidocaine patch 5%) [see Clinical Pharmacology (12.3) ]. When ZTLIDO is used concomitantly with other products containing local anesthetic agents, the total amount of drug absorbed from all formulations must be considered. Clearly instruct and advise patients: to wash hands immediately after handling ZTLIDO and to avoid contact with eyes [see Warnings and Precautions (5.6) ] . to store ZTLIDO inside the sealed envelope out of the reach of children, pets, and others and to apply immediately after removal from the envelope. to fold used ZTLIDO so that the adhesive side sticks to itself and to safely discard used ZTLIDO or pieces of cut ZTLIDO where children and pets cannot get to them [see Warnings and Precautions (5.1) ] . to never apply external heat sources, such as heating pads or electric blankets, directly to ZTLIDO, because plasma lidocaine levels are increased. ZTLIDO can be applied, however, to the administration site after moderate heat exposure, such as 15 minutes of heating pad exposure on a medium setting [see Warnings and Precautions (5.2) , Clinical Pharmacology (12.3) ] . that clothing may be worn over the area of application. that ZTLIDO may be used during moderate exercise, such as biking for 30 minutes. that ZTLIDO may be exposed to water, such as showering for 10 minutes or immersion for 15 minutes. to dry the topical system, after water exposure, by gently patting the skin, not by rubbing the skin or topical system. that ZTLIDO topical systems that have lifted at the edges may be reattached by pressing firmly on the edges and that fully detached topical systems may be reapplied as originally directed. that if a ZTLIDO topical system comes off completely and will not stick to patient's skin, to remove and properly dispose of the used ZTLIDO and to apply a new ZTLIDO topical system for a total duration of 12 hours of used and new topical system together. that if irritation or a burning sensation occurs during application, to remove ZTLIDO and to not reapply until the irritation subsides [see Warnings and Precautions (5.4) ] .
Contraindications
ZTLIDO is contraindicated in patients with a known history of sensitivity to local anesthetics of the amide type, or to any other component of the product.
Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling: Excessive Dosing/Overexposure to Lidocaine [see Warnings and Precautions (5.2) ] Methemoglobinemia [see Warnings and Precautions (5.3) ] Application Site Reactions [see Warnings and Precautions (5.4) ] Hypersensitivity Reactions [see Warnings and Precautions (5.5) ] Eye Irritation [see Warnings and Precautions (5.6) ] The following adverse reactions from voluntary reports or clinical studies have been reported with lidocaine. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Skin and subcutaneous tissues : blisters, bruising, burning sensation, depigmentation, dermatitis, discoloration, edema, erosions, erythema, exfoliation, flushing, irritation, papules, petechia, pruritus, vesicles, and abnormal sensation. Immune system : angioedema, bronchospasm, dermatitis, dyspnea, hypersensitivity, laryngospasm, pruritus, shock, and urticaria. Central Nervous System : lightheadedness, nervousness, apprehension, euphoria, confusion, dizziness, drowsiness, tinnitus, blurred or double vision, sensations of heat, cold or numbness, twitching, tremors, convulsions, unconsciousness, somnolence, respiratory depression and arrest. Cardiovascular : bradycardia, hypotension, and cardiovascular collapse leading to arrest. Other : asthenia, disorientation, headache, hyperesthesia, hypoesthesia, metallic taste, nausea, pain exacerbated, paresthesia, taste alteration, and vomiting.
Drug Interactions
Class I Antiarrhythmic Drugs: When ZTLIDO is used in patients receiving Class I antiarrhythmic drugs (such as tocainide and mexiletine) the toxic effects are additive and potentially synergistic. Consider risk/benefit before concomitant use. ( 7.2 ) Local Anesthetic Agents: When ZTLIDO is used concomitantly with other products containing local anesthetic agents, the effects are additive. Consider the amount of drug absorbed from all formulations when local anesthetics are administered concomitantly. ( 7.3 )
Storage and Handling
ZTLIDO (lidocaine topical system) 1.8% is available as the following: Carton of 30 topical systems, packaged into individual child-resistant envelopes. NDC 69557-111-30
How Supplied
ZTLIDO (lidocaine topical system) 1.8% is available as the following: Carton of 30 topical systems, packaged into individual child-resistant envelopes. NDC 69557-111-30
Description
Dosage and Administration ( 2.1 ) 4/2021
Section 42229-5
Improper Application and Duration of Use: Application of more than the recommended number of ZTLIDO or applying ZTLIDO for longer than the recommended wearing time (12 hours of every 24 hours) could result in increased absorption and high blood concentrations of lidocaine, leading to adverse reactions. Advise patients on proper application and duration.
Section 42230-3
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Issued: 4/2021 | ||
|
PATIENT INFORMATION
ZTLIDO ®(ZEE-TEE-LIE-DOH) (lidocaine topical system) |
|||
| What is ZTLIDO? | |||
| ZTLIDO is a prescription medicine used for relief of pain from damaged nerves (neuropathic pain) that follows healing of shingles. It is not known if ZTLIDO is safe and effective in children. | |||
| Do not use ZTLIDO if you: | |||
|
|||
| Before using ZTLIDO, tell your healthcare provider about all your medical conditions, including if you: | |||
|
|||
| Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, vitamins, and herbal supplements. Especially, tell your healthcare provider if you are using other lidocaine containing products or anesthetic medicines. | |||
| How should I use ZTLIDO? | |||
| Read the Instructions for Use at the end of this Patient Information leaflet for information about how to apply the ZTLIDO topical system. | |||
|
|||
| What should I avoid while using ZTLIDO? | |||
|
|||
| What are the possible side effects of ZTLIDO? | |||
| ZTLIDO may cause serious side effects, including: | |||
|
|||
|
|
|
|
|
|||
|
|
||
| If you develop a skin reaction while wearing ZTLIDO, remove it. Do notreapply ZTLIDO until the site reaction goes away. | |||
|
|||
| These are not all the possible side effects of ZTLIDO. | |||
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | |||
| How should I store ZTLIDO? | |||
|
|||
| Keep ZTLIDO and all medicines out of the reach of children, pets, and others. | |||
| General information about the safe and effective use of ZTLIDO. | |||
| Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use ZTLIDO for a condition for which it was not prescribed. Do not give ZTLIDO to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about ZTLIDO that is written for health professionals. | |||
| What are the ingredients in ZTLIDO? | |||
| Active ingredient:lidocaine | |||
| Inactive ingredients:butylated hydroxytoluene, dipropylene glycol, isostearic acid, mineral oil, polyisobutylene, silicone dioxide, styrene/isoprene/styrene block copolymer, and terpene resin. | |||
| Manufactured for:Scilex Pharmaceuticals Inc., Palo Alto, CA 94303 | |||
| ZTLIDO ®is a trademark owned by Scilex Pharmaceuticals Inc. © 2021 Scilex Pharmaceuticals Inc. All rights reserved. | |||
| SCILEX
®
PHARMACEUTICALS |
|||
| For more information call [1-844-SCILEX1] or visit www.ZTLIDO.com. |
Section 43683-2
| Dosage and Administration ( 2.1) | 4/2021 |
Section 44425-7
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Wash hands thoroughly immediately after handling the topical system. Upon removal, fold the topical system in half so that the adhesive side sticks to itself. Discard used ZTLIDO and pieces of cut ZTLIDO in a manner that prevents accidental contact or ingestion by children, pets or others [see Dosage and Administration (2.2), Warnings and Precautions (5.1)] .
Section 51945-4
PRINCIPAL DISPLAY PANEL - lidocaine patch 1.8% CARTON LABEL
NDC 69557-111-30
ZTlido™
(lidocaine topical system) 1.8%*
For topical use only.
*One ZTlido™ (lidocaine topical system) 1.8%provides equivalent lidocaine exposure to one Lidoderm® (lidocaine patch 5%).
Inactive ingredients: butylated hydroxytoluene, dipropylene glycol, isostearic acid, mineral oil, polyisobutylene, silicone dioxide, styrene/isoprene/styrene block copolymer, and terpene resin.
DOSAGE:For dosage and full prescribing information, please read accompanying product information.
Store at 68-77°F (20-25°C). [See USP Controlled Room Temperature].
WARNING:Store and dispose of ZTlido™ out of the reach of children, pets, and others.
30 TOPICAL SYSTEMS
30 Envelopes Containing 1 Topical System Each
SCILEX ™ Rx Only
PHARMACEUTICALS
Manufactured by: Oishi Koseido Co., Ltd., Tosu, Saga, Japan
Manufactured for: SCILEX Pharmaceuticals Inc., San Diego, CA 92121
Active Ingredient Made in Spain
Section 59845-8
Instructions for Use
ZTLIDO
®(ZEE-TEE-LIE-DOH)
(lidocaine topical system)
Read this Instructions for Use before you start using ZTLIDO and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical treatment or condition.
Important information:
- One ZTLIDO (lidocaine topical system) 1.8% provides equivalent lidocaine exposure to one Lidoderm® (lidocaine patch 5%).
- ZTLIDO is for use on intact skin only.
- ZTLIDO comes in a child-resistant envelope. Do not open the ZTLIDO envelope until you are ready to use it.
- Apply the prescribed number of ZTLIDO topical systems at one time. Clothing may be worn over ZTLIDO.
- You should only use a maximum of 3 ZTLIDO topical systems at a time.
- Keep your prescribed number of ZTLIDO topical systems on for up to 12 hours within a 24-hour period (12 hours on and 12 hours off).
- ZTLIDO may be used during moderate exercise, such as biking for 30 minutes.
- ZTLIDO may be worn in water such as showering for 10 minutes or bathing for 15 minutes.
- After ZTLIDO is exposed to water, pat the topical system dry. Do not rub the topical system.
- ZTLIDO topical systems that have come off completely or are lifting at the edges may be re-attached to the affected area by firmly pressing down on the topical system or on the lifted areas.
- If ZTLIDO lifts off the skin or detaches more than 1 time, do nottry to re-attach it. Throw it away (See Step 6 ).
- If the ZTLIDO you are wearing comes off completely, and will not stick to your skin, throw it away (see Step 6 ).
Wash your hands well right away after handling ZTLIDO (when you apply ZTLIDO, when you re-attach it, or when you remove it). Avoid eye contact with your hands and fingers while handling ZTLIDO.
Figure A: ZTLIDO Topical System
Applying your ZTLIDO topical system:
Step 1:Select the application site.
|
|
Step 2:Using scissors, carefully cut the envelope along the dotted line and open it to remove ZTLIDO.
|
|
| Step 3a:Remove the transparent release liner before applying ZTLIDO to the skin. Apply ZTLIDO right away after removing the transparent release liner. Apply ZTLIDO only to intact skin. |
|
| Step 3b:Place the adhesive side of ZTLIDO to skin, while not touching the sticky side. Smooth the ZTLIDO using your hands and firmly press to make sure it sticks well to skin. | |
Step 4:Wash your hands well right away after applying ZTLIDO.
|
|
Removing your ZTLIDO topical system:
Step 5:Remove ZTLIDO from your skin after you have worn it for up to 12 hours.
|
|
Step 6:Throw away the used whole or cut pieces of ZTLIDO where children and pets cannot get to them.
|
|
How to reattach ZTLIDO or apply a replacement ZTLIDO:
- If the ZTLIDO you are wearing comes off completely or lifts at the edges, reattach to the affected area by pressing firmly on the edges of the topical system or lifted areas. If ZTLIDO lifts off the skin or comes off completely more than 1 time, do nottry to re-attach it. Throw away the used ZTLIDO as instructed above in Step 6.
- If the ZTLIDO you are wearing comes off completely and will not stick to your skin, throw away the used ZTLIDO as instructed above in Step 6.
- Pat dry the area of skin if ZTLIDO came off while swimming or showering.
- Apply a replacement ZTLIDO the same way you would apply a new ZTLIDO as described above in Steps 1 through 6.
- Remove the replacement ZTLIDO at your usual removal time as described above in Steps 5 and 6.
- The total timeyou may wear the used and replacement ZTLIDO is 12 hours.
Manufactured for:
Scilex Pharmaceuticals Inc.
Palo Alto, CA 94303
USA
ZTLIDO
®is a trademark owned by Scilex Pharmaceuticals Inc.
© 2021 Scilex Pharmaceuticals Inc. All rights reserved.
SCILEX
®
PHARMACEUTICALS
Revised: 4/2021
10 Overdosage
Lidocaine overdose from cutaneous absorption is rare, but could occur. If there is any suspicion of lidocaine overdose, check drug blood concentration. The management of overdose includes close monitoring, supportive care, and symptomatic treatment. Dialysis is of negligible value in the treatment of acute overdose with lidocaine.
In the absence of massive topical overdose or oral ingestion, evaluation of symptoms of toxicity should include consideration of other etiologies for the clinical effects, or overdosage from other sources of lidocaine or other local anesthetics.
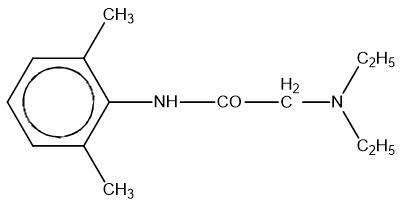
11 Description
ZTLIDO (lidocaine topical system) 1.8% is a single-layer, drug-in-adhesive topical delivery system comprised of an adhesive material containing 36 mg lidocaine, which is applied to a pliable nonwoven cloth backing and covered with a polyethylene terephthalate film release liner. The release liner is removed prior to application to the skin. The size of ZTLIDO is 10 cm × 14 cm × 0.08 cm.
Lidocaine, an amide local anesthetic, is chemically designated as acetamide, 2-(diethylamino)- N-(2,6-dimethylphenyl), has an octanol:water partition ratio of 43 at pH 7.4, and has the following structure:
Each ZTLIDO contains 36 mg of lidocaine (18 mg per gram adhesive) in a non-aqueous base and also contains the following inactive ingredients: butylated hydroxytoluene, dipropylene glycol, isostearic acid, mineral oil, polyisobutylene, silicone dioxide, styrene/isoprene/styrene block copolymer, and terpene resin.
5.6 Eye Exposure
The contact of ZTLIDO with eyes, although not studied, should be avoided based on findings of severe eye irritation with the application of similar products in animals. If eye contact occurs, immediately wash out the eye with water or saline and protect the eye (such as, eye glasses/eye wear) until sensation returns.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of ZTLIDO did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be done with caution, usually starting at the low end of the dosing range (e.g., a single topical system), reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
14 Clinical Studies
Single-dose treatment with lidocaine patch (currently preferred dosage form term for a patch is topical system) was compared to treatment with vehicle patch (without lidocaine), and to no treatment (observation only) in a double-blind, crossover clinical trial with 35 post-herpetic neuralgia patients. Pain intensity and pain relief scores were evaluated periodically for 12 hours. Lidocaine patch performed statistically better than vehicle patch in terms of pain intensity from 4 to 12 hours.
Multiple-dose, two-week treatment with lidocaine patch was compared to vehicle patch (without lidocaine) in a double-blind, crossover clinical trial of withdrawal-type design conducted in 32 patients, who were considered as responders to the open-label use of lidocaine patch prior to the study. The constant type of pain was evaluated but not the pain induced by sensory stimuli (dysesthesia). Statistically significant differences favoring lidocaine patch were observed in terms of time to exit from the trial (14 versus 3.8 days at p-value <0.001), daily average pain relief, and patient's preference of treatment. About half of the patients also took oral medication commonly used in the treatment of post-herpetic neuralgia. The extent of use of concomitant medication was similar in the two treatment groups.
Based on a clinical study in 54 subjects with ZTLIDO, 47 subjects (87%) had adhesion scores of 0 (≥ 90% adhered) for all evaluations performed every 3 hours during the 12 hours of administration, 7 subjects (13%) had adhesion scores of 1 (≥ 75% to < 90% adhered) for at least one evaluation, and no subjects had scores of 2 or greater (< 75% adhered).
4 Contraindications
ZTLIDO is contraindicated in patients with a known history of sensitivity to local anesthetics of the amide type, or to any other component of the product.
6 Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling:
- Excessive Dosing/Overexposure to Lidocaine [see Warnings and Precautions (5.2)]
- Methemoglobinemia [see Warnings and Precautions (5.3)]
- Application Site Reactions [see Warnings and Precautions (5.4)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.5)]
- Eye Irritation [see Warnings and Precautions (5.6)]
The following adverse reactions from voluntary reports or clinical studies have been reported with lidocaine. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Skin and subcutaneous tissues: blisters, bruising, burning sensation, depigmentation, dermatitis, discoloration, edema, erosions, erythema, exfoliation, flushing, irritation, papules, petechia, pruritus, vesicles, and abnormal sensation.
Immune system: angioedema, bronchospasm, dermatitis, dyspnea, hypersensitivity, laryngospasm, pruritus, shock, and urticaria.
Central Nervous System: lightheadedness, nervousness, apprehension, euphoria, confusion, dizziness, drowsiness, tinnitus, blurred or double vision, sensations of heat, cold or numbness, twitching, tremors, convulsions, unconsciousness, somnolence, respiratory depression and arrest.
Cardiovascular: bradycardia, hypotension, and cardiovascular collapse leading to arrest.
Other: asthenia, disorientation, headache, hyperesthesia, hypoesthesia, metallic taste, nausea, pain exacerbated, paresthesia, taste alteration, and vomiting.
7 Drug Interactions
- Class I Antiarrhythmic Drugs:When ZTLIDO is used in patients receiving Class I antiarrhythmic drugs (such as tocainide and mexiletine) the toxic effects are additive and potentially synergistic. Consider risk/benefit before concomitant use. ( 7.2)
- Local Anesthetic Agents:When ZTLIDO is used concomitantly with other products containing local anesthetic agents, the effects are additive. Consider the amount of drug absorbed from all formulations when local anesthetics are administered concomitantly. ( 7.3)
12.2 Pharmacodynamics
The penetration of lidocaine into intact skin after application of ZTLIDO is sufficient to produce an analgesic effect, but less than the amount necessary to produce a complete sensory block.
12.3 Pharmacokinetics
ZTLIDO has different bioavailability compared to Lidoderm. In a single-dose, crossover study conducted in 53 healthy volunteers, ZTLIDO (lidocaine topical system) 1.8% demonstrated equivalent exposure (AUC) and peak concentration (C max) of lidocaine to Lidoderm (lidocaine patch 5%).
5.3 Methemoglobinemia
Cases of methemoglobinemia have been reported in association with local anesthetic use. Although all patients are at risk for methemoglobinemia, patients with glucose-6-phosphate dehydrogenase deficiency, congenital or idiopathic methemoglobinemia, cardiac or pulmonary compromise, infants under 6 months of age, and concurrent exposure to oxidizing agents or their metabolites are more susceptible to developing clinical manifestations of the condition. If local anesthetics must be used in these patients, close monitoring for symptoms and signs of methemoglobinemia is recommended.
Signs of methemoglobinemia may occur immediately or may be delayed some hours after exposure, and are characterized by a cyanotic skin discoloration and/or abnormal coloration of the blood. Methemoglobin levels may continue to rise; therefore, immediate treatment is required to avert more serious central nervous system and cardiovascular adverse effects, including seizures, coma, arrhythmias, and death. Discontinue ZTLIDO and any other oxidizing agents. Depending on the severity of the signs and symptoms, patients may respond to supportive care, i.e., oxygen therapy, hydration. A more severe clinical presentation may require treatment with methylene blue, exchange transfusion, or hyperbaric oxygen.
7.3 Local Anesthetics
When ZTLIDO is used concomitantly with other products containing local anesthetic agents, the effects are additive. Consider the amount of drug absorbed from all formulations when local anesthetic agents are administered concomitantly.
1 Indications and Usage
ZTLIDO is indicated for relief of pain associated with post-herpetic neuralgia (PHN) in adults.
5.1 Accidental Exposure
A used ZTLIDO topical system contains residual lidocaine after use. The potential exists for a small child or a pet to suffer serious adverse effects from chewing or ingesting a new or used ZTLIDO. It is important for patients to store and dispose of ZTLIDO properly, and keep out of the reach of children, pets, and others [see Dosage and Administration (2.1)].
12.1 Mechanism of Action
Lidocaine is an amide local anesthetic. Lidocaine blocks sodium ion channels required for the initiation and conduction of neuronal impulses.
7.2 Antiarrhythmic Drugs
When ZTLIDO is used in patients receiving Class I antiarrhythmic drugs (such as tocainide and mexiletine), the toxic effects are additive and potentially synergistic. Consider risk/benefit during concomitant use.
5 Warnings and Precautions
- Accidental Exposure: Even a used ZTLIDO topical system contains residual lidocaine after use. It is important for patients to store and dispose of ZTLIDO properly and keep out of the reach of children, pets, and others. ( 5.1)
- Excessive Dosing/Overexposure: Applying ZTLIDO to larger surface areas or for a longer duration than recommended could lead to increased absorption and high blood concentrations of lidocaine, leading to adverse effects. ( 5.2)
- Increased Absorption on Non-Intact Skin: May result in higher blood concentrations of lidocaine. ( 5.2)
- Risk of Overexposure with External Heat Sources: Applying external heat sources to ZTLIDO may result in increased drug exposure. ( 5.2)
- Methemoglobinemia: Cases of methemoglobinemia have been reported in association with local anesthetic use. ( 5.3)
- Application Site Reactions: During or immediately after treatment with ZTLIDO, application site reactions may develop. ( 5.4)
- Hypersensitivity Reactions: Cross sensitivity to ZTLIDO in patients with a history of drug sensitivity to para-aminobenzoic acid (PABA) derivatives is possible. ( 5.5)
- Eye Exposure: Immediately wash out the eye with water or saline and protect the eye until sensation returns. ( 5.6).
2 Dosage and Administration
- Because of the difference in bioavailability of ZTLIDO compared to Lidoderm®, a different dosage strength is required to be administered to the patient. One ZTLIDO (lidocaine topical system) 1.8% provides equivalent lidocaine exposure to one Lidoderm (lidocaine patch 5%). ( 2.1)
- Apply ZTLIDO to intact skin to cover the most painful area. Apply the prescribed number of topical systems (maximum of 3) only once for up to 12 hours in a 24-hour period. ( 2.2)
- ZTLIDO may be cut into smaller sizes prior to removal of the release liner. ( 2.2)
- In patients who are debilitated or have impaired elimination, smaller areas of treatment are recommended. Achieve this by cutting ZTLIDO with scissors into a smaller size prior to removing the release liner. ( 2.2)
3 Dosage Forms and Strengths
Topical system: 1.8% packaged in an individual envelope.
8 Use in Specific Populations
Lactation: Lidocaine is excreted into human milk. Caution should be exercised when ZTLIDO is administered to a nursing mother, especially when administered with other local anesthetics. ( 8.2)
5.4 Application Site Reactions
During or immediately after treatment with ZTLIDO, the skin at the site of application may develop blisters, bruising, burning sensation, depigmentation, dermatitis, discoloration, edema, erythema, exfoliation, irritation, papules, petechia, pruritus, vesicles, or may be the locus of abnormal sensation. These reactions are generally mild and transient, resolving spontaneously within a few minutes to hours. If application site reactions occur while the topical system is being worn, advise the patient to remove ZTLIDO and not to reapply until skin reactions subside.
5.5 Hypersensitivity Reactions
Patients allergic to para-aminobenzoic acid (PABA) derivatives (procaine, tetracaine, benzocaine, etc.) have not shown cross-sensitivity to lidocaine. However, be aware of the potential for cross-sensitivity in patients allergic to PABA derivatives, especially if the etiologic agent is uncertain. Manage hypersensitivity reactions by conventional means. The detection of sensitivity by skin testing is of doubtful value.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
16 How Supplied/storage and Handling
ZTLIDO (lidocaine topical system) 1.8% is available as the following:
Carton of 30 topical systems, packaged into individual child-resistant envelopes.
NDC 69557-111-30
2.2 Treatment of Post Herpetic Neuralgia
Advise patients to apply ZTLIDO to intact skin to cover the most painful area and to apply the prescribed number of topical systems (maximum of 3), only once for up to 12 hours within a 24-hour period (12 hours on and 12 hours off) [see Warnings and Precautions (5.2)] .
In patients who are debilitated or have impaired elimination, smaller areas of treatment are recommended. Achieve this by cutting ZTLIDO with scissors into a smaller size prior to removing the release liner.
5.2 Excessive Dosing/overexposure to Lidocaine
Lidocaine toxicity can be expected at lidocaine blood concentrations above 5 mcg/mL. The blood concentration of lidocaine is determined by the rate and extent of lidocaine absorption and elimination. Longer duration of application, application of more than the recommended number of ZTLIDO, smaller patients, or impaired elimination may all contribute to increasing the blood concentration of lidocaine.
If lidocaine overdose is suspected, check drug blood concentration. Management of overdose includes close monitoring, supportive care, and symptomatic treatment [see Overdosage (10)] .
2.1 Important Dosage and Administration Instructions
Because of the difference in bioavailability of ZTLIDO compared to Lidoderm (lidocaine patch 5%), a different dosage strength is required to be administered to the patient. One ZTLIDO (lidocaine topical system) 1.8% provides equivalent lidocaine exposure to one Lidoderm (lidocaine patch 5%) [see Clinical Pharmacology (12.3)].
When ZTLIDO is used concomitantly with other products containing local anesthetic agents, the total amount of drug absorbed from all formulations must be considered.
Clearly instruct and advise patients:
- to wash hands immediately after handling ZTLIDO and to avoid contact with eyes [see Warnings and Precautions (5.6)] .
- to store ZTLIDO inside the sealed envelope out of the reach of children, pets, and others and to apply immediately after removal from the envelope.
- to fold used ZTLIDO so that the adhesive side sticks to itself and to safely discard used ZTLIDO or pieces of cut ZTLIDO where children and pets cannot get to them [see Warnings and Precautions (5.1)] .
- to never apply external heat sources, such as heating pads or electric blankets, directly to ZTLIDO, because plasma lidocaine levels are increased. ZTLIDO can be applied, however, to the administration site after moderate heat exposure, such as 15 minutes of heating pad exposure on a medium setting [see Warnings and Precautions (5.2) , Clinical Pharmacology (12.3)] .
- that clothing may be worn over the area of application.
- that ZTLIDO may be used during moderate exercise, such as biking for 30 minutes.
- that ZTLIDO may be exposed to water, such as showering for 10 minutes or immersion for 15 minutes.
- to dry the topical system, after water exposure, by gently patting the skin, not by rubbing the skin or topical system.
- that ZTLIDO topical systems that have lifted at the edges may be reattached by pressing firmly on the edges and that fully detached topical systems may be reapplied as originally directed.
- that if a ZTLIDO topical system comes off completely and will not stick to patient's skin, to remove and properly dispose of the used ZTLIDO and to apply a new ZTLIDO topical system for a total duration of 12 hours of used and new topical system together.
- that if irritation or a burning sensation occurs during application, to remove ZTLIDO and to not reapply until the irritation subsides [see Warnings and Precautions (5.4)] .
7.1 Drugs That May Cause Methemoglobinemia When Used With Ztlido
Patients who are administered local anesthetics may be at increased risk of developing methemoglobinemia when concurrently exposed to the following drugs, which could include other local anesthetics:
Examples of Drugs Associated with Methemoglobinemia:
| Class | Examples |
|---|---|
| Nitrates/Nitrites | nitric oxide, nitroglycerin, nitroprusside, nitrous oxide |
| Local anesthetics | articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine |
| Antineoplastic agents | cyclophosphamide, flutamide, hydroxyurea, ifosfamide, rasburicase |
| Antibiotics | dapsone, nitrofurantoin, para-aminosalicyclic acid, sulfonamides |
| Antimalarials | chloroquine, primaquine |
| Anticonvulsants | phenobarbital, phenytoin, sodium valproate |
| Other drugs | acetaminophen, metoclopramide, quinine, sulfasalazine |
Structured Label Content
Section 42229-5 (42229-5)
Improper Application and Duration of Use: Application of more than the recommended number of ZTLIDO or applying ZTLIDO for longer than the recommended wearing time (12 hours of every 24 hours) could result in increased absorption and high blood concentrations of lidocaine, leading to adverse reactions. Advise patients on proper application and duration.
Section 42230-3 (42230-3)
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Issued: 4/2021 | ||
|
PATIENT INFORMATION
ZTLIDO ®(ZEE-TEE-LIE-DOH) (lidocaine topical system) |
|||
| What is ZTLIDO? | |||
| ZTLIDO is a prescription medicine used for relief of pain from damaged nerves (neuropathic pain) that follows healing of shingles. It is not known if ZTLIDO is safe and effective in children. | |||
| Do not use ZTLIDO if you: | |||
|
|||
| Before using ZTLIDO, tell your healthcare provider about all your medical conditions, including if you: | |||
|
|||
| Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, vitamins, and herbal supplements. Especially, tell your healthcare provider if you are using other lidocaine containing products or anesthetic medicines. | |||
| How should I use ZTLIDO? | |||
| Read the Instructions for Use at the end of this Patient Information leaflet for information about how to apply the ZTLIDO topical system. | |||
|
|||
| What should I avoid while using ZTLIDO? | |||
|
|||
| What are the possible side effects of ZTLIDO? | |||
| ZTLIDO may cause serious side effects, including: | |||
|
|||
|
|
|
|
|
|||
|
|
||
| If you develop a skin reaction while wearing ZTLIDO, remove it. Do notreapply ZTLIDO until the site reaction goes away. | |||
|
|||
| These are not all the possible side effects of ZTLIDO. | |||
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | |||
| How should I store ZTLIDO? | |||
|
|||
| Keep ZTLIDO and all medicines out of the reach of children, pets, and others. | |||
| General information about the safe and effective use of ZTLIDO. | |||
| Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use ZTLIDO for a condition for which it was not prescribed. Do not give ZTLIDO to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about ZTLIDO that is written for health professionals. | |||
| What are the ingredients in ZTLIDO? | |||
| Active ingredient:lidocaine | |||
| Inactive ingredients:butylated hydroxytoluene, dipropylene glycol, isostearic acid, mineral oil, polyisobutylene, silicone dioxide, styrene/isoprene/styrene block copolymer, and terpene resin. | |||
| Manufactured for:Scilex Pharmaceuticals Inc., Palo Alto, CA 94303 | |||
| ZTLIDO ®is a trademark owned by Scilex Pharmaceuticals Inc. © 2021 Scilex Pharmaceuticals Inc. All rights reserved. | |||
| SCILEX
®
PHARMACEUTICALS |
|||
| For more information call [1-844-SCILEX1] or visit www.ZTLIDO.com. |
Section 43683-2 (43683-2)
| Dosage and Administration ( 2.1) | 4/2021 |
Section 44425-7 (44425-7)
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Wash hands thoroughly immediately after handling the topical system. Upon removal, fold the topical system in half so that the adhesive side sticks to itself. Discard used ZTLIDO and pieces of cut ZTLIDO in a manner that prevents accidental contact or ingestion by children, pets or others [see Dosage and Administration (2.2), Warnings and Precautions (5.1)] .
Section 51945-4 (51945-4)
PRINCIPAL DISPLAY PANEL - lidocaine patch 1.8% CARTON LABEL
NDC 69557-111-30
ZTlido™
(lidocaine topical system) 1.8%*
For topical use only.
*One ZTlido™ (lidocaine topical system) 1.8%provides equivalent lidocaine exposure to one Lidoderm® (lidocaine patch 5%).
Inactive ingredients: butylated hydroxytoluene, dipropylene glycol, isostearic acid, mineral oil, polyisobutylene, silicone dioxide, styrene/isoprene/styrene block copolymer, and terpene resin.
DOSAGE:For dosage and full prescribing information, please read accompanying product information.
Store at 68-77°F (20-25°C). [See USP Controlled Room Temperature].
WARNING:Store and dispose of ZTlido™ out of the reach of children, pets, and others.
30 TOPICAL SYSTEMS
30 Envelopes Containing 1 Topical System Each
SCILEX ™ Rx Only
PHARMACEUTICALS
Manufactured by: Oishi Koseido Co., Ltd., Tosu, Saga, Japan
Manufactured for: SCILEX Pharmaceuticals Inc., San Diego, CA 92121
Active Ingredient Made in Spain
Section 59845-8 (59845-8)
Instructions for Use
ZTLIDO
®(ZEE-TEE-LIE-DOH)
(lidocaine topical system)
Read this Instructions for Use before you start using ZTLIDO and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical treatment or condition.
Important information:
- One ZTLIDO (lidocaine topical system) 1.8% provides equivalent lidocaine exposure to one Lidoderm® (lidocaine patch 5%).
- ZTLIDO is for use on intact skin only.
- ZTLIDO comes in a child-resistant envelope. Do not open the ZTLIDO envelope until you are ready to use it.
- Apply the prescribed number of ZTLIDO topical systems at one time. Clothing may be worn over ZTLIDO.
- You should only use a maximum of 3 ZTLIDO topical systems at a time.
- Keep your prescribed number of ZTLIDO topical systems on for up to 12 hours within a 24-hour period (12 hours on and 12 hours off).
- ZTLIDO may be used during moderate exercise, such as biking for 30 minutes.
- ZTLIDO may be worn in water such as showering for 10 minutes or bathing for 15 minutes.
- After ZTLIDO is exposed to water, pat the topical system dry. Do not rub the topical system.
- ZTLIDO topical systems that have come off completely or are lifting at the edges may be re-attached to the affected area by firmly pressing down on the topical system or on the lifted areas.
- If ZTLIDO lifts off the skin or detaches more than 1 time, do nottry to re-attach it. Throw it away (See Step 6 ).
- If the ZTLIDO you are wearing comes off completely, and will not stick to your skin, throw it away (see Step 6 ).
Wash your hands well right away after handling ZTLIDO (when you apply ZTLIDO, when you re-attach it, or when you remove it). Avoid eye contact with your hands and fingers while handling ZTLIDO.
Figure A: ZTLIDO Topical System
Applying your ZTLIDO topical system:
Step 1:Select the application site.
|
|
Step 2:Using scissors, carefully cut the envelope along the dotted line and open it to remove ZTLIDO.
|
|
| Step 3a:Remove the transparent release liner before applying ZTLIDO to the skin. Apply ZTLIDO right away after removing the transparent release liner. Apply ZTLIDO only to intact skin. |
|
| Step 3b:Place the adhesive side of ZTLIDO to skin, while not touching the sticky side. Smooth the ZTLIDO using your hands and firmly press to make sure it sticks well to skin. | |
Step 4:Wash your hands well right away after applying ZTLIDO.
|
|
Removing your ZTLIDO topical system:
Step 5:Remove ZTLIDO from your skin after you have worn it for up to 12 hours.
|
|
Step 6:Throw away the used whole or cut pieces of ZTLIDO where children and pets cannot get to them.
|
|
How to reattach ZTLIDO or apply a replacement ZTLIDO:
- If the ZTLIDO you are wearing comes off completely or lifts at the edges, reattach to the affected area by pressing firmly on the edges of the topical system or lifted areas. If ZTLIDO lifts off the skin or comes off completely more than 1 time, do nottry to re-attach it. Throw away the used ZTLIDO as instructed above in Step 6.
- If the ZTLIDO you are wearing comes off completely and will not stick to your skin, throw away the used ZTLIDO as instructed above in Step 6.
- Pat dry the area of skin if ZTLIDO came off while swimming or showering.
- Apply a replacement ZTLIDO the same way you would apply a new ZTLIDO as described above in Steps 1 through 6.
- Remove the replacement ZTLIDO at your usual removal time as described above in Steps 5 and 6.
- The total timeyou may wear the used and replacement ZTLIDO is 12 hours.
Manufactured for:
Scilex Pharmaceuticals Inc.
Palo Alto, CA 94303
USA
ZTLIDO
®is a trademark owned by Scilex Pharmaceuticals Inc.
© 2021 Scilex Pharmaceuticals Inc. All rights reserved.
SCILEX
®
PHARMACEUTICALS
Revised: 4/2021
10 Overdosage (10 OVERDOSAGE)
Lidocaine overdose from cutaneous absorption is rare, but could occur. If there is any suspicion of lidocaine overdose, check drug blood concentration. The management of overdose includes close monitoring, supportive care, and symptomatic treatment. Dialysis is of negligible value in the treatment of acute overdose with lidocaine.
In the absence of massive topical overdose or oral ingestion, evaluation of symptoms of toxicity should include consideration of other etiologies for the clinical effects, or overdosage from other sources of lidocaine or other local anesthetics.
11 Description (11 DESCRIPTION)
ZTLIDO (lidocaine topical system) 1.8% is a single-layer, drug-in-adhesive topical delivery system comprised of an adhesive material containing 36 mg lidocaine, which is applied to a pliable nonwoven cloth backing and covered with a polyethylene terephthalate film release liner. The release liner is removed prior to application to the skin. The size of ZTLIDO is 10 cm × 14 cm × 0.08 cm.
Lidocaine, an amide local anesthetic, is chemically designated as acetamide, 2-(diethylamino)- N-(2,6-dimethylphenyl), has an octanol:water partition ratio of 43 at pH 7.4, and has the following structure:
Each ZTLIDO contains 36 mg of lidocaine (18 mg per gram adhesive) in a non-aqueous base and also contains the following inactive ingredients: butylated hydroxytoluene, dipropylene glycol, isostearic acid, mineral oil, polyisobutylene, silicone dioxide, styrene/isoprene/styrene block copolymer, and terpene resin.
5.6 Eye Exposure
The contact of ZTLIDO with eyes, although not studied, should be avoided based on findings of severe eye irritation with the application of similar products in animals. If eye contact occurs, immediately wash out the eye with water or saline and protect the eye (such as, eye glasses/eye wear) until sensation returns.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of ZTLIDO did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be done with caution, usually starting at the low end of the dosing range (e.g., a single topical system), reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
14 Clinical Studies (14 CLINICAL STUDIES)
Single-dose treatment with lidocaine patch (currently preferred dosage form term for a patch is topical system) was compared to treatment with vehicle patch (without lidocaine), and to no treatment (observation only) in a double-blind, crossover clinical trial with 35 post-herpetic neuralgia patients. Pain intensity and pain relief scores were evaluated periodically for 12 hours. Lidocaine patch performed statistically better than vehicle patch in terms of pain intensity from 4 to 12 hours.
Multiple-dose, two-week treatment with lidocaine patch was compared to vehicle patch (without lidocaine) in a double-blind, crossover clinical trial of withdrawal-type design conducted in 32 patients, who were considered as responders to the open-label use of lidocaine patch prior to the study. The constant type of pain was evaluated but not the pain induced by sensory stimuli (dysesthesia). Statistically significant differences favoring lidocaine patch were observed in terms of time to exit from the trial (14 versus 3.8 days at p-value <0.001), daily average pain relief, and patient's preference of treatment. About half of the patients also took oral medication commonly used in the treatment of post-herpetic neuralgia. The extent of use of concomitant medication was similar in the two treatment groups.
Based on a clinical study in 54 subjects with ZTLIDO, 47 subjects (87%) had adhesion scores of 0 (≥ 90% adhered) for all evaluations performed every 3 hours during the 12 hours of administration, 7 subjects (13%) had adhesion scores of 1 (≥ 75% to < 90% adhered) for at least one evaluation, and no subjects had scores of 2 or greater (< 75% adhered).
4 Contraindications (4 CONTRAINDICATIONS)
ZTLIDO is contraindicated in patients with a known history of sensitivity to local anesthetics of the amide type, or to any other component of the product.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described elsewhere in the labeling:
- Excessive Dosing/Overexposure to Lidocaine [see Warnings and Precautions (5.2)]
- Methemoglobinemia [see Warnings and Precautions (5.3)]
- Application Site Reactions [see Warnings and Precautions (5.4)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.5)]
- Eye Irritation [see Warnings and Precautions (5.6)]
The following adverse reactions from voluntary reports or clinical studies have been reported with lidocaine. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Skin and subcutaneous tissues: blisters, bruising, burning sensation, depigmentation, dermatitis, discoloration, edema, erosions, erythema, exfoliation, flushing, irritation, papules, petechia, pruritus, vesicles, and abnormal sensation.
Immune system: angioedema, bronchospasm, dermatitis, dyspnea, hypersensitivity, laryngospasm, pruritus, shock, and urticaria.
Central Nervous System: lightheadedness, nervousness, apprehension, euphoria, confusion, dizziness, drowsiness, tinnitus, blurred or double vision, sensations of heat, cold or numbness, twitching, tremors, convulsions, unconsciousness, somnolence, respiratory depression and arrest.
Cardiovascular: bradycardia, hypotension, and cardiovascular collapse leading to arrest.
Other: asthenia, disorientation, headache, hyperesthesia, hypoesthesia, metallic taste, nausea, pain exacerbated, paresthesia, taste alteration, and vomiting.
7 Drug Interactions (7 DRUG INTERACTIONS)
- Class I Antiarrhythmic Drugs:When ZTLIDO is used in patients receiving Class I antiarrhythmic drugs (such as tocainide and mexiletine) the toxic effects are additive and potentially synergistic. Consider risk/benefit before concomitant use. ( 7.2)
- Local Anesthetic Agents:When ZTLIDO is used concomitantly with other products containing local anesthetic agents, the effects are additive. Consider the amount of drug absorbed from all formulations when local anesthetics are administered concomitantly. ( 7.3)
12.2 Pharmacodynamics
The penetration of lidocaine into intact skin after application of ZTLIDO is sufficient to produce an analgesic effect, but less than the amount necessary to produce a complete sensory block.
12.3 Pharmacokinetics
ZTLIDO has different bioavailability compared to Lidoderm. In a single-dose, crossover study conducted in 53 healthy volunteers, ZTLIDO (lidocaine topical system) 1.8% demonstrated equivalent exposure (AUC) and peak concentration (C max) of lidocaine to Lidoderm (lidocaine patch 5%).
5.3 Methemoglobinemia
Cases of methemoglobinemia have been reported in association with local anesthetic use. Although all patients are at risk for methemoglobinemia, patients with glucose-6-phosphate dehydrogenase deficiency, congenital or idiopathic methemoglobinemia, cardiac or pulmonary compromise, infants under 6 months of age, and concurrent exposure to oxidizing agents or their metabolites are more susceptible to developing clinical manifestations of the condition. If local anesthetics must be used in these patients, close monitoring for symptoms and signs of methemoglobinemia is recommended.
Signs of methemoglobinemia may occur immediately or may be delayed some hours after exposure, and are characterized by a cyanotic skin discoloration and/or abnormal coloration of the blood. Methemoglobin levels may continue to rise; therefore, immediate treatment is required to avert more serious central nervous system and cardiovascular adverse effects, including seizures, coma, arrhythmias, and death. Discontinue ZTLIDO and any other oxidizing agents. Depending on the severity of the signs and symptoms, patients may respond to supportive care, i.e., oxygen therapy, hydration. A more severe clinical presentation may require treatment with methylene blue, exchange transfusion, or hyperbaric oxygen.
7.3 Local Anesthetics
When ZTLIDO is used concomitantly with other products containing local anesthetic agents, the effects are additive. Consider the amount of drug absorbed from all formulations when local anesthetic agents are administered concomitantly.
1 Indications and Usage (1 INDICATIONS AND USAGE)
ZTLIDO is indicated for relief of pain associated with post-herpetic neuralgia (PHN) in adults.
5.1 Accidental Exposure
A used ZTLIDO topical system contains residual lidocaine after use. The potential exists for a small child or a pet to suffer serious adverse effects from chewing or ingesting a new or used ZTLIDO. It is important for patients to store and dispose of ZTLIDO properly, and keep out of the reach of children, pets, and others [see Dosage and Administration (2.1)].
12.1 Mechanism of Action
Lidocaine is an amide local anesthetic. Lidocaine blocks sodium ion channels required for the initiation and conduction of neuronal impulses.
7.2 Antiarrhythmic Drugs
When ZTLIDO is used in patients receiving Class I antiarrhythmic drugs (such as tocainide and mexiletine), the toxic effects are additive and potentially synergistic. Consider risk/benefit during concomitant use.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Accidental Exposure: Even a used ZTLIDO topical system contains residual lidocaine after use. It is important for patients to store and dispose of ZTLIDO properly and keep out of the reach of children, pets, and others. ( 5.1)
- Excessive Dosing/Overexposure: Applying ZTLIDO to larger surface areas or for a longer duration than recommended could lead to increased absorption and high blood concentrations of lidocaine, leading to adverse effects. ( 5.2)
- Increased Absorption on Non-Intact Skin: May result in higher blood concentrations of lidocaine. ( 5.2)
- Risk of Overexposure with External Heat Sources: Applying external heat sources to ZTLIDO may result in increased drug exposure. ( 5.2)
- Methemoglobinemia: Cases of methemoglobinemia have been reported in association with local anesthetic use. ( 5.3)
- Application Site Reactions: During or immediately after treatment with ZTLIDO, application site reactions may develop. ( 5.4)
- Hypersensitivity Reactions: Cross sensitivity to ZTLIDO in patients with a history of drug sensitivity to para-aminobenzoic acid (PABA) derivatives is possible. ( 5.5)
- Eye Exposure: Immediately wash out the eye with water or saline and protect the eye until sensation returns. ( 5.6).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Because of the difference in bioavailability of ZTLIDO compared to Lidoderm®, a different dosage strength is required to be administered to the patient. One ZTLIDO (lidocaine topical system) 1.8% provides equivalent lidocaine exposure to one Lidoderm (lidocaine patch 5%). ( 2.1)
- Apply ZTLIDO to intact skin to cover the most painful area. Apply the prescribed number of topical systems (maximum of 3) only once for up to 12 hours in a 24-hour period. ( 2.2)
- ZTLIDO may be cut into smaller sizes prior to removal of the release liner. ( 2.2)
- In patients who are debilitated or have impaired elimination, smaller areas of treatment are recommended. Achieve this by cutting ZTLIDO with scissors into a smaller size prior to removing the release liner. ( 2.2)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Topical system: 1.8% packaged in an individual envelope.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: Lidocaine is excreted into human milk. Caution should be exercised when ZTLIDO is administered to a nursing mother, especially when administered with other local anesthetics. ( 8.2)
5.4 Application Site Reactions
During or immediately after treatment with ZTLIDO, the skin at the site of application may develop blisters, bruising, burning sensation, depigmentation, dermatitis, discoloration, edema, erythema, exfoliation, irritation, papules, petechia, pruritus, vesicles, or may be the locus of abnormal sensation. These reactions are generally mild and transient, resolving spontaneously within a few minutes to hours. If application site reactions occur while the topical system is being worn, advise the patient to remove ZTLIDO and not to reapply until skin reactions subside.
5.5 Hypersensitivity Reactions
Patients allergic to para-aminobenzoic acid (PABA) derivatives (procaine, tetracaine, benzocaine, etc.) have not shown cross-sensitivity to lidocaine. However, be aware of the potential for cross-sensitivity in patients allergic to PABA derivatives, especially if the etiologic agent is uncertain. Manage hypersensitivity reactions by conventional means. The detection of sensitivity by skin testing is of doubtful value.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
ZTLIDO (lidocaine topical system) 1.8% is available as the following:
Carton of 30 topical systems, packaged into individual child-resistant envelopes.
NDC 69557-111-30
2.2 Treatment of Post Herpetic Neuralgia (2.2 Treatment of Post-Herpetic Neuralgia)
Advise patients to apply ZTLIDO to intact skin to cover the most painful area and to apply the prescribed number of topical systems (maximum of 3), only once for up to 12 hours within a 24-hour period (12 hours on and 12 hours off) [see Warnings and Precautions (5.2)] .
In patients who are debilitated or have impaired elimination, smaller areas of treatment are recommended. Achieve this by cutting ZTLIDO with scissors into a smaller size prior to removing the release liner.
5.2 Excessive Dosing/overexposure to Lidocaine (5.2 Excessive Dosing/Overexposure to Lidocaine)
Lidocaine toxicity can be expected at lidocaine blood concentrations above 5 mcg/mL. The blood concentration of lidocaine is determined by the rate and extent of lidocaine absorption and elimination. Longer duration of application, application of more than the recommended number of ZTLIDO, smaller patients, or impaired elimination may all contribute to increasing the blood concentration of lidocaine.
If lidocaine overdose is suspected, check drug blood concentration. Management of overdose includes close monitoring, supportive care, and symptomatic treatment [see Overdosage (10)] .
2.1 Important Dosage and Administration Instructions
Because of the difference in bioavailability of ZTLIDO compared to Lidoderm (lidocaine patch 5%), a different dosage strength is required to be administered to the patient. One ZTLIDO (lidocaine topical system) 1.8% provides equivalent lidocaine exposure to one Lidoderm (lidocaine patch 5%) [see Clinical Pharmacology (12.3)].
When ZTLIDO is used concomitantly with other products containing local anesthetic agents, the total amount of drug absorbed from all formulations must be considered.
Clearly instruct and advise patients:
- to wash hands immediately after handling ZTLIDO and to avoid contact with eyes [see Warnings and Precautions (5.6)] .
- to store ZTLIDO inside the sealed envelope out of the reach of children, pets, and others and to apply immediately after removal from the envelope.
- to fold used ZTLIDO so that the adhesive side sticks to itself and to safely discard used ZTLIDO or pieces of cut ZTLIDO where children and pets cannot get to them [see Warnings and Precautions (5.1)] .
- to never apply external heat sources, such as heating pads or electric blankets, directly to ZTLIDO, because plasma lidocaine levels are increased. ZTLIDO can be applied, however, to the administration site after moderate heat exposure, such as 15 minutes of heating pad exposure on a medium setting [see Warnings and Precautions (5.2) , Clinical Pharmacology (12.3)] .
- that clothing may be worn over the area of application.
- that ZTLIDO may be used during moderate exercise, such as biking for 30 minutes.
- that ZTLIDO may be exposed to water, such as showering for 10 minutes or immersion for 15 minutes.
- to dry the topical system, after water exposure, by gently patting the skin, not by rubbing the skin or topical system.
- that ZTLIDO topical systems that have lifted at the edges may be reattached by pressing firmly on the edges and that fully detached topical systems may be reapplied as originally directed.
- that if a ZTLIDO topical system comes off completely and will not stick to patient's skin, to remove and properly dispose of the used ZTLIDO and to apply a new ZTLIDO topical system for a total duration of 12 hours of used and new topical system together.
- that if irritation or a burning sensation occurs during application, to remove ZTLIDO and to not reapply until the irritation subsides [see Warnings and Precautions (5.4)] .
7.1 Drugs That May Cause Methemoglobinemia When Used With Ztlido (7.1 Drugs That May Cause Methemoglobinemia When Used with ZTLIDO)
Patients who are administered local anesthetics may be at increased risk of developing methemoglobinemia when concurrently exposed to the following drugs, which could include other local anesthetics:
Examples of Drugs Associated with Methemoglobinemia:
| Class | Examples |
|---|---|
| Nitrates/Nitrites | nitric oxide, nitroglycerin, nitroprusside, nitrous oxide |
| Local anesthetics | articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine |
| Antineoplastic agents | cyclophosphamide, flutamide, hydroxyurea, ifosfamide, rasburicase |
| Antibiotics | dapsone, nitrofurantoin, para-aminosalicyclic acid, sulfonamides |
| Antimalarials | chloroquine, primaquine |
| Anticonvulsants | phenobarbital, phenytoin, sodium valproate |
| Other drugs | acetaminophen, metoclopramide, quinine, sulfasalazine |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:36.693915 · Updated: 2026-03-14T22:16:35.361266