Drug Facts
a115cb42-a286-46ab-abb9-526bb3c2498e
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
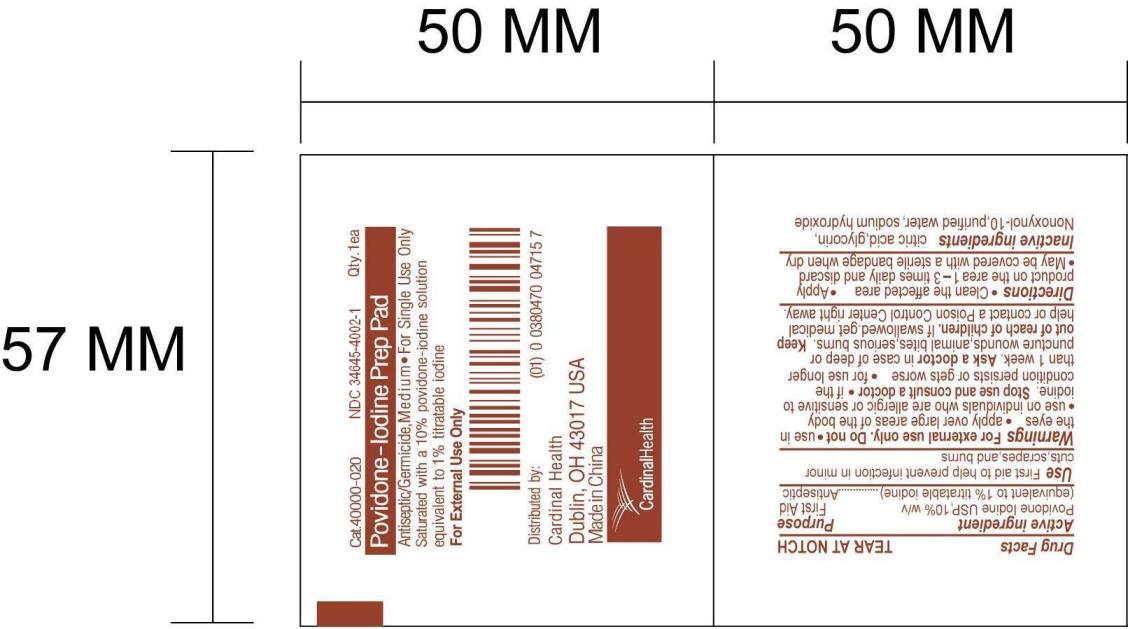
Cat.40000-020 NDC 34645-4002-1 Qty. 1ea Povidone-Iodine Prep Pad Antiseptic/Germicide, Medium*For Single Use Only Saturated with a 10% povidone-iodine solution equivalent to 1% titratable iodine For External Use Only Distributed by: Cardinal Health Dublin, OH 43017 USA Made in China
Medication Information
Warnings and Precautions
Warnings
For external use only.
Indications and Usage
Directions
Clean the affected area
Apply product on the area 1-3 times daily and discard
May be covered with a sterile bandage when dry
Description
Cat.40000-020 NDC 34645-4002-1 Qty. 1ea Povidone-Iodine Prep Pad Antiseptic/Germicide, Medium*For Single Use Only Saturated with a 10% povidone-iodine solution equivalent to 1% titratable iodine For External Use Only Distributed by: Cardinal Health Dublin, OH 43017 USA Made in China
Section 50565-1
Keep out of reach of children.
If swallowed, get medical help or contact Poison Control Center right away.
Section 50566-9
Stop use and consult a doctor
if the condition persists or gets worse
for use longer than 1 week
Section 50567-7
Use
First aid to help prevent infection in minor cuts, scrapes, and burns
Section 50569-3
Ask doctor
in case of deep or puncture wounds, animal bites, serious burns.
Section 50570-1
Do not * use in the eyes * apply over large areas of the body
* use on individuals who are allergic or sensitive to iodine
Section 51727-6
Inactive ingredients
ctric acid, glycerin, Nonoxynol-10, purified water, sodium hydroxide
Section 51945-4
Cat.40000-020 NDC 34645-4002-1 Qty. 1ea
Povidone-Iodine Prep Pad
Antiseptic/Germicide, Medium*For Single Use Only
Saturated with a 10% povidone-iodine solution
equivalent to 1% titratable iodine
For External Use Only
Distributed by:
Cardinal Health
Dublin, OH 43017 USA
Made in China
Section 55105-1
Purpose
First Aid
Antiseptic
Section 55106-9
Active ingredient
Povidone Iodine USP, 10% w/v
(equivalent to 1% titratable iodine)
Structured Label Content
Indications and Usage (34067-9)
Directions
Clean the affected area
Apply product on the area 1-3 times daily and discard
May be covered with a sterile bandage when dry
Warnings and Precautions (34071-1)
Warnings
For external use only.
Section 50565-1 (50565-1)
Keep out of reach of children.
If swallowed, get medical help or contact Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and consult a doctor
if the condition persists or gets worse
for use longer than 1 week
Section 50567-7 (50567-7)
Use
First aid to help prevent infection in minor cuts, scrapes, and burns
Section 50569-3 (50569-3)
Ask doctor
in case of deep or puncture wounds, animal bites, serious burns.
Section 50570-1 (50570-1)
Do not * use in the eyes * apply over large areas of the body
* use on individuals who are allergic or sensitive to iodine
Section 51727-6 (51727-6)
Inactive ingredients
ctric acid, glycerin, Nonoxynol-10, purified water, sodium hydroxide
Section 51945-4 (51945-4)
Cat.40000-020 NDC 34645-4002-1 Qty. 1ea
Povidone-Iodine Prep Pad
Antiseptic/Germicide, Medium*For Single Use Only
Saturated with a 10% povidone-iodine solution
equivalent to 1% titratable iodine
For External Use Only
Distributed by:
Cardinal Health
Dublin, OH 43017 USA
Made in China
Section 55105-1 (55105-1)
Purpose
First Aid
Antiseptic
Section 55106-9 (55106-9)
Active ingredient
Povidone Iodine USP, 10% w/v
(equivalent to 1% titratable iodine)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:01.657107 · Updated: 2026-03-14T22:52:20.145878