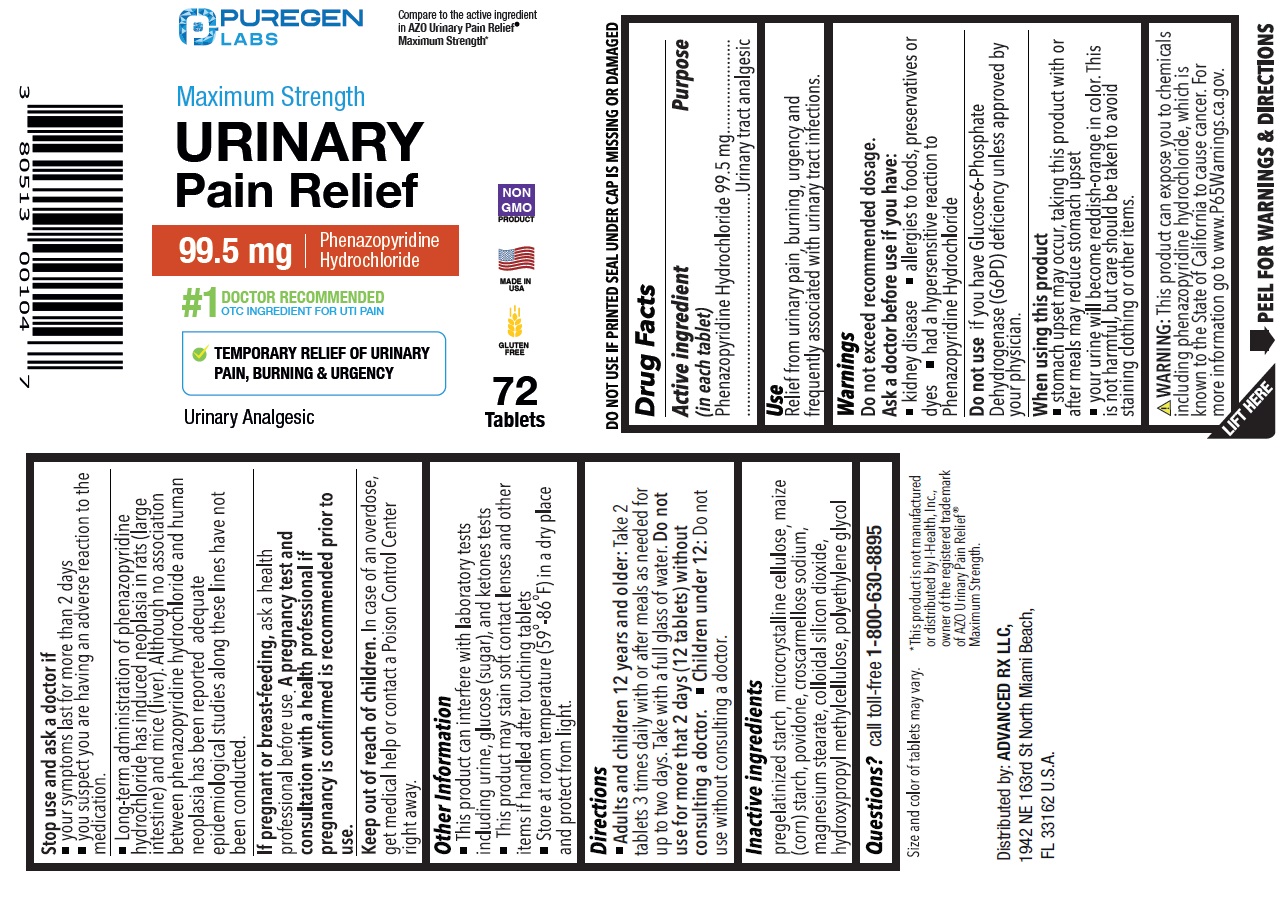
maximum strength urinary pain relief
a10464cf-36b2-460f-b31c-2e0edee3e4cf
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Urinary tract analgesic
Description
Phenazopyridine Hydrochloride 99.5 mg
Medication Information
Warnings
Do not exceed recommended dosage.
Ask a doctor before use if you have:
• kidney disease
• allergies to foods, preservatives or dyes
• had a hypersensitive reaction to Phenazopyridine Hydrochloride
Do not useif you have Glucose-6-Phosphate Dehydrogenase (G6PD) deficiency unless approved by your physician.
When using this product
• stomach upset may occur, taking this product with or after meals may reduce stomach upset
• your urine will become reddish-orange in color. This is not harmful, but care should be taken to avoid staining clothing or other items.
Stop use and ask a doctor if
• your symptoms last for more than 2 days
• you suspect you are having an adverse reaction to the medication.
• Long-term administration of phenazopyridine hydrochloride has induced neoplasia in rats (large intestine) and mice (liver). Although no association between phenazopyridine hydrochloride and human neoplasia has been reported, adequate epidemiological studies along these lines have not been conducted.
If pregnant or breastfeeding, ask a health professional before use. A pregnancy test and consultation with a health professional if pregnancy is confirmed is recommended prior to use.
Active Ingredient
Phenazopyridine Hydrochloride 99.5 mg
Purpose
Urinary tract analgesic
Directions
• Adults and children 12 years and older:Take 2 tablets 3 times daily with or after meals as needed for up to two days. Take with a full glass of water. Do not use for more than 2 days (12 tablets) without consulting a doctor.
• Children under 12 years:Do not use without consulting a doctor
Other Information
• This product can interfere with laboratory tests including urine, glucose (sugar), and ketones tests
• This product may stain contact lenses and other items if handled after touching tablets.
• Store at room temperature between (59ºF -86ºF) in a dry place and protect from light.
Inactive Ingredients
pregelatinized starch, microcrystalline cellulose, maize (corn) strach, povidone, croscarmellose sodium, magnesium stearate, colloidal silicone dioxide, hydroxypropyl methylcellulose, polyethylene glycol.
Description
Phenazopyridine Hydrochloride 99.5 mg
Questions?
call toll-free 1-800-630-8895
Use
Relief from urinary pain, burning, urgency and frequency associated with urinary tract infections.
Section 50565-1
Keep out of reach of children.In case of an overdose, get medical help or contact a Poison Control Center right away.
Section 51945-4
Compare to the active ingredient in AZO Urinary Pain Relief ®Maximum Strength*
NDC 80513-517-72
Maximum Strength Urinary Pain Relief
99.5 mg Phenazopyridine Hydrochloride
#1 DOCTOR RECOMMENDED OTC INGREDIENT FOR UTI PAIN
TEMPORARY RELIEF OF URINARY PAIN, BURNING & URGENCY
Urinary Analgesic
72 Tablets
DO NOT USE IF PRINTED SEAL UNDER CAP IS MISSING OR DAMAGED
Size and color of tablet may very.
WARNING: This product can expose you to chemicals including phenazopyridine hydrochloride, which is known to the State of California to cause cancer. For more information go to www.P65Warnings.ca.gov.
*This product is not manufactured or distributed by I-Health, Inc., owner of the registered trademark of AZO Urinary Pain Relief ®Maximum Strength
Distributed by:
ADVANCED RX LLC,
1942 NE 163rd St North Miami Beach,
FL 33162 U.S.A.
MADE IN USA
Drug Facts
Structured Label Content
Warnings
Do not exceed recommended dosage.
Ask a doctor before use if you have:
• kidney disease
• allergies to foods, preservatives or dyes
• had a hypersensitive reaction to Phenazopyridine Hydrochloride
Do not useif you have Glucose-6-Phosphate Dehydrogenase (G6PD) deficiency unless approved by your physician.
When using this product
• stomach upset may occur, taking this product with or after meals may reduce stomach upset
• your urine will become reddish-orange in color. This is not harmful, but care should be taken to avoid staining clothing or other items.
Stop use and ask a doctor if
• your symptoms last for more than 2 days
• you suspect you are having an adverse reaction to the medication.
• Long-term administration of phenazopyridine hydrochloride has induced neoplasia in rats (large intestine) and mice (liver). Although no association between phenazopyridine hydrochloride and human neoplasia has been reported, adequate epidemiological studies along these lines have not been conducted.
If pregnant or breastfeeding, ask a health professional before use. A pregnancy test and consultation with a health professional if pregnancy is confirmed is recommended prior to use.
Active Ingredient (Active ingredient (in each tablet))
Phenazopyridine Hydrochloride 99.5 mg
Purpose
Urinary tract analgesic
Directions
• Adults and children 12 years and older:Take 2 tablets 3 times daily with or after meals as needed for up to two days. Take with a full glass of water. Do not use for more than 2 days (12 tablets) without consulting a doctor.
• Children under 12 years:Do not use without consulting a doctor
Other Information (Other information)
• This product can interfere with laboratory tests including urine, glucose (sugar), and ketones tests
• This product may stain contact lenses and other items if handled after touching tablets.
• Store at room temperature between (59ºF -86ºF) in a dry place and protect from light.
Inactive Ingredients (Inactive ingredients)
pregelatinized starch, microcrystalline cellulose, maize (corn) strach, povidone, croscarmellose sodium, magnesium stearate, colloidal silicone dioxide, hydroxypropyl methylcellulose, polyethylene glycol.
Questions?
call toll-free 1-800-630-8895
Use
Relief from urinary pain, burning, urgency and frequency associated with urinary tract infections.
Section 50565-1 (50565-1)
Keep out of reach of children.In case of an overdose, get medical help or contact a Poison Control Center right away.
Section 51945-4 (51945-4)
Compare to the active ingredient in AZO Urinary Pain Relief ®Maximum Strength*
NDC 80513-517-72
Maximum Strength Urinary Pain Relief
99.5 mg Phenazopyridine Hydrochloride
#1 DOCTOR RECOMMENDED OTC INGREDIENT FOR UTI PAIN
TEMPORARY RELIEF OF URINARY PAIN, BURNING & URGENCY
Urinary Analgesic
72 Tablets
DO NOT USE IF PRINTED SEAL UNDER CAP IS MISSING OR DAMAGED
Size and color of tablet may very.
WARNING: This product can expose you to chemicals including phenazopyridine hydrochloride, which is known to the State of California to cause cancer. For more information go to www.P65Warnings.ca.gov.
*This product is not manufactured or distributed by I-Health, Inc., owner of the registered trademark of AZO Urinary Pain Relief ®Maximum Strength
Distributed by:
ADVANCED RX LLC,
1942 NE 163rd St North Miami Beach,
FL 33162 U.S.A.
MADE IN USA
Drug Facts
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:11.812757 · Updated: 2026-03-14T23:03:07.791514