walgreens multi-symptom relief eye drops (pld)
a03789ce-66bd-7b12-e053-2995a90af85a
34390-5
HUMAN OTC DRUG LABEL
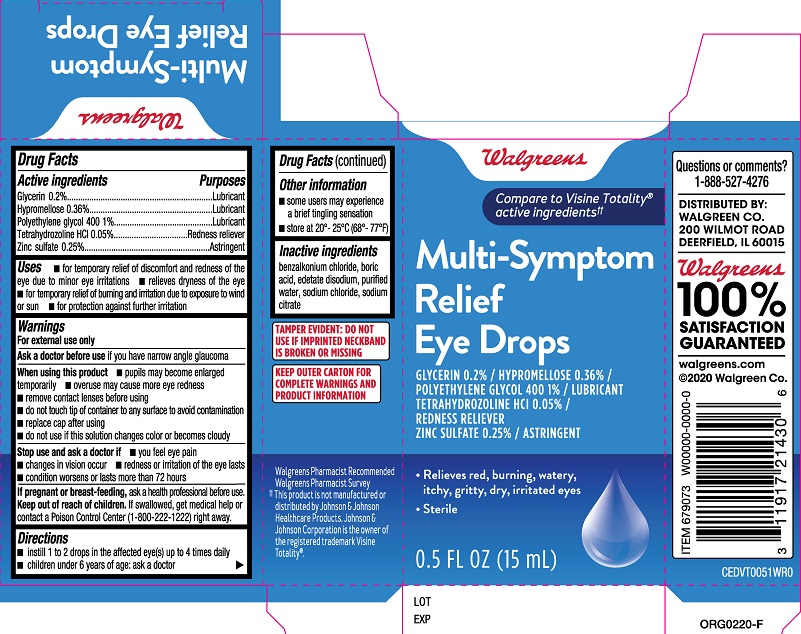
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredients Glycerin ....0.2% Hypromellose.....0.36% Polyethylene glycol 400.....1% Tetrahydrozoline HCl.....0.05% Zinc sulfate 0.25%
Medication Information
Warnings and Precautions
Warnings
For external use only
Active Ingredient
Active ingredients
Glycerin ....0.2%
Hypromellose.....0.36%
Polyethylene glycol 400.....1%
Tetrahydrozoline HCl.....0.05%
Zinc sulfate 0.25%
Indications and Usage
Uses
- for temporary relief of discomfort and redness of the eye due to minor eye irritations
- relieves dryness of the eye
- for temporary relief of burning and irritation due to exposure to wind or sun
- for protection against further irritation
Dosage and Administration
Directions
- instill 1 to 2 drops in the affected eye(s) up to 4 times daily
- children under 6 years of age: ask a doctor
Description
Active ingredients Glycerin ....0.2% Hypromellose.....0.36% Polyethylene glycol 400.....1% Tetrahydrozoline HCl.....0.05% Zinc sulfate 0.25%
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Section 50566-9
Stop use and ask a doctor if
- you feel eye pain
- changes in vision occur
- redness or irritation of the eye lasts
- condition worsens or lasts more than 72 hours
Section 50567-7
When using this product
- pupils may become enlarged temporarily
- overuse may cause more eye redness
- remove contact lenses before using
- do not touch tip of container to any surface to avoid contamination
- replace cap after using
- do not use if this solution changes color or becomes cloudy
Section 50569-3
Ask a doctor before use if you have narrow angle glaucoma
Section 51727-6
Inactive ingredients
- benzalkonium chloride, boric acid, edetate disodium, purified water, sodium chloride, sodium citrate
Section 51945-4
Section 53413-1
Questions or comments?
1-888-527-4276
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1
Purposes
Glycerin ....Lubricant
Hypromellose.....Lubricant
Polyethylene glycol 400.....Lubricant
Tetrahydrozoline HCl.....Redness reliever
Zinc sulfate.....Astringent
Section 60561-8
Other information
- some users may experience a brief tingling sensation
- store at 20º-25ºC (68º-77ºF)
Structured Label Content
Warnings and Precautions (34071-1)
Warnings
For external use only
Active Ingredient (55106-9)
Active ingredients
Glycerin ....0.2%
Hypromellose.....0.36%
Polyethylene glycol 400.....1%
Tetrahydrozoline HCl.....0.05%
Zinc sulfate 0.25%
Indications and Usage (34067-9)
Uses
- for temporary relief of discomfort and redness of the eye due to minor eye irritations
- relieves dryness of the eye
- for temporary relief of burning and irritation due to exposure to wind or sun
- for protection against further irritation
Dosage and Administration (34068-7)
Directions
- instill 1 to 2 drops in the affected eye(s) up to 4 times daily
- children under 6 years of age: ask a doctor
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if
- you feel eye pain
- changes in vision occur
- redness or irritation of the eye lasts
- condition worsens or lasts more than 72 hours
Section 50567-7 (50567-7)
When using this product
- pupils may become enlarged temporarily
- overuse may cause more eye redness
- remove contact lenses before using
- do not touch tip of container to any surface to avoid contamination
- replace cap after using
- do not use if this solution changes color or becomes cloudy
Section 50569-3 (50569-3)
Ask a doctor before use if you have narrow angle glaucoma
Section 51727-6 (51727-6)
Inactive ingredients
- benzalkonium chloride, boric acid, edetate disodium, purified water, sodium chloride, sodium citrate
Section 51945-4 (51945-4)
Section 53413-1 (53413-1)
Questions or comments?
1-888-527-4276
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1 (55105-1)
Purposes
Glycerin ....Lubricant
Hypromellose.....Lubricant
Polyethylene glycol 400.....Lubricant
Tetrahydrozoline HCl.....Redness reliever
Zinc sulfate.....Astringent
Section 60561-8 (60561-8)
Other information
- some users may experience a brief tingling sensation
- store at 20º-25ºC (68º-77ºF)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:33.521500 · Updated: 2026-03-14T23:02:10.811423