a025b97d-55a2-4abf-a6f4-3b989e85f7bc
34390-5
HUMAN OTC DRUG LABEL
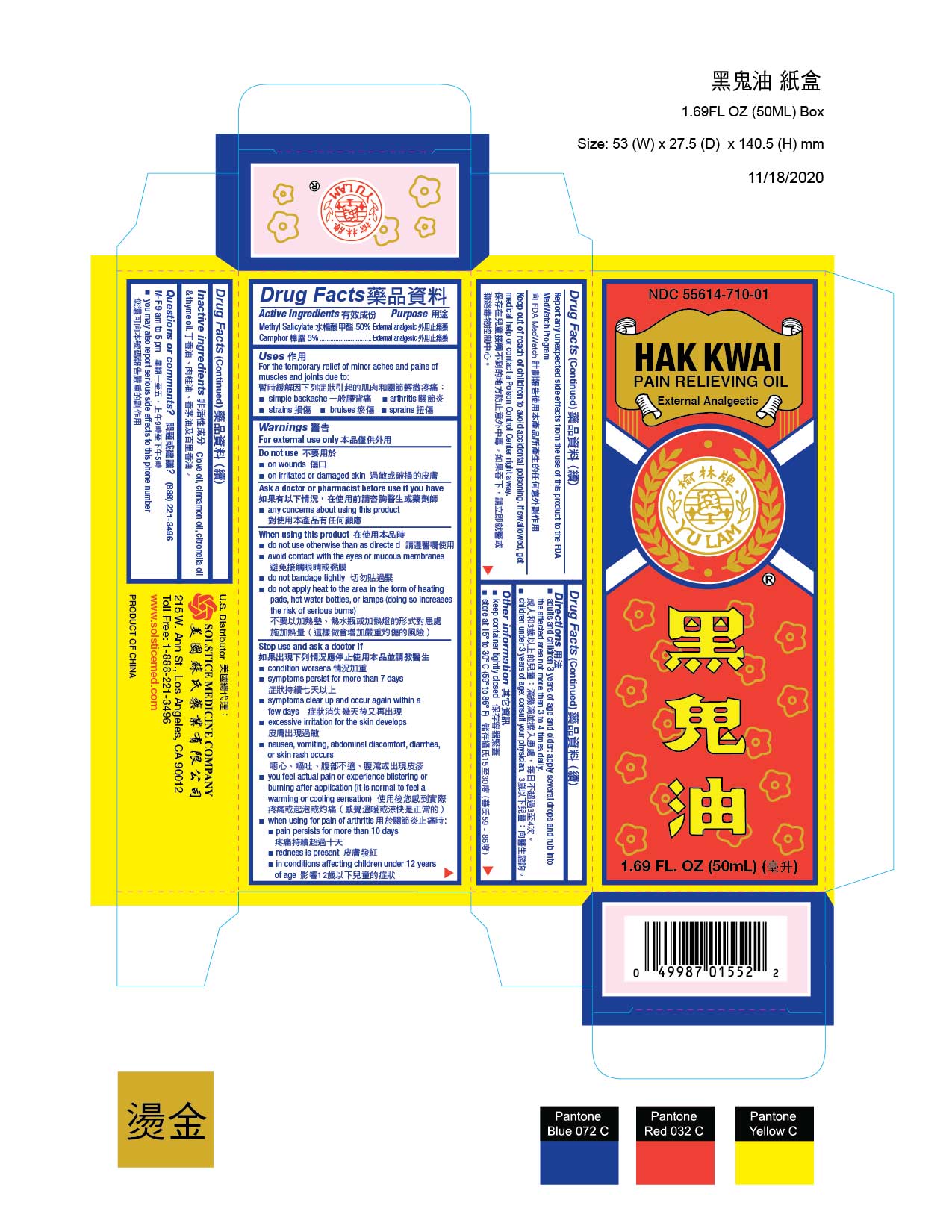
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredient Camphor 5% Methyl salicylate 50%
Medication Information
Warnings and Precautions
Warnings
For external use only
Indications and Usage
Uses
For the temporary relief of minor aches and pains of muscles and joints due to:
■ simple backache
■ arthritis
■ strains
■ bruises
■ sprains
Dosage and Administration
Directions
■ adults and children 3 years of age and older: apply several drops and rub Into the affected area not more than 3 to 4 times daily
■ children under 3 years of age: consult your physician
Description
Active ingredient Camphor 5% Methyl salicylate 50%
Section 34084-4
Report any unexpected side effects from the use of this product to the FDA MedWatch Program
Section 44425-7
Other information
■ keep container tightly closed
■ store protected from light at 15 to 30 C (59 to 86 F)
Section 50565-1
Keep out of reach of children to avoid accidental poisoning. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if
■ condition worsens
■ symptoms persist for more than 7 days
■ symptoms clear up and occur again within a few days
■ excessive irritation of the skin develops
■ nausea, vomiting, abdominal discomfort, diarrhea, or skin rash occurs
■ you feel actual pain or experience blistering or burning after application (it is normal to feel a warming or cooling sensation)
■ when using for pain of arthritis:
■ pain persists for more than 10 days
■ redness is present
■ in conditions affecting children under 12 years of age
Section 50567-7
When using this product
■ do not use otherwise than as directed
■ avoid contact with the eyes or mucous membranes
■ do not bandage tightly
■ do not apply heat to the area in the form of heating pads, hot water bottles, or lamps (doing so increases the risk of serious burns)
Section 50568-5
Ask a doctor or pharmacist before use if you have
■ any concerns about using this product
Section 50570-1
Do not use
■ on wounds
■ on irritated or damaged skin
Section 51727-6
Inactive ingredients Clove oil, cinnamon oil, citronella oil, and thyme oil.
Section 51945-4
HAK KWAI PAIN RELIEVING OIL
Methyl Salicylate and Camphor
NDC 55614-710-01
EXTERNAL ANALGESIC
1.69 FL OZ (50 ML)
Section 53413-1
Questions or comments? (888) 221-3496 M-F 9 am to 5 pm
■ you may also report serious side effects to this phone number
Section 55105-1
Purpose
External analgesic
External analgesic
Section 55106-9
Active ingredient Camphor 5% Methyl salicylate 50%
Structured Label Content
Indications and Usage (34067-9)
Uses
For the temporary relief of minor aches and pains of muscles and joints due to:
■ simple backache
■ arthritis
■ strains
■ bruises
■ sprains
Dosage and Administration (34068-7)
Directions
■ adults and children 3 years of age and older: apply several drops and rub Into the affected area not more than 3 to 4 times daily
■ children under 3 years of age: consult your physician
Warnings and Precautions (34071-1)
Warnings
For external use only
Section 34084-4 (34084-4)
Report any unexpected side effects from the use of this product to the FDA MedWatch Program
Section 44425-7 (44425-7)
Other information
■ keep container tightly closed
■ store protected from light at 15 to 30 C (59 to 86 F)
Section 50565-1 (50565-1)
Keep out of reach of children to avoid accidental poisoning. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if
■ condition worsens
■ symptoms persist for more than 7 days
■ symptoms clear up and occur again within a few days
■ excessive irritation of the skin develops
■ nausea, vomiting, abdominal discomfort, diarrhea, or skin rash occurs
■ you feel actual pain or experience blistering or burning after application (it is normal to feel a warming or cooling sensation)
■ when using for pain of arthritis:
■ pain persists for more than 10 days
■ redness is present
■ in conditions affecting children under 12 years of age
Section 50567-7 (50567-7)
When using this product
■ do not use otherwise than as directed
■ avoid contact with the eyes or mucous membranes
■ do not bandage tightly
■ do not apply heat to the area in the form of heating pads, hot water bottles, or lamps (doing so increases the risk of serious burns)
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if you have
■ any concerns about using this product
Section 50570-1 (50570-1)
Do not use
■ on wounds
■ on irritated or damaged skin
Section 51727-6 (51727-6)
Inactive ingredients Clove oil, cinnamon oil, citronella oil, and thyme oil.
Section 51945-4 (51945-4)
HAK KWAI PAIN RELIEVING OIL
Methyl Salicylate and Camphor
NDC 55614-710-01
EXTERNAL ANALGESIC
1.69 FL OZ (50 ML)
Section 53413-1 (53413-1)
Questions or comments? (888) 221-3496 M-F 9 am to 5 pm
■ you may also report serious side effects to this phone number
Section 55105-1 (55105-1)
Purpose
External analgesic
External analgesic
Section 55106-9 (55106-9)
Active ingredient Camphor 5% Methyl salicylate 50%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:59:11.334238 · Updated: 2026-03-14T22:59:04.734496