These Highlights Do Not Include All The Information Needed To Use Miudella ®
9f5c5008-8166-40fa-9627-e9aa620b547e
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Improper insertion of intrauterine systems, including Miudella, increases the risk of complications [see Warnings and Precautions (5.1) ] . Proper training prior to first use of Miudella can minimize the risk of improper Miudella insertion [see Warnings and Precautions (5.1) ]. Miudella is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the Miudella REMS program to ensure healthcare providers are trained on the proper insertion of Miudella prior to first use [see Warnings and Precautions (5.2) ].
Indications and Usage
Miudella is indicated for prevention of pregnancy in females of reproductive potential for up to 3 years.
Dosage and Administration
Miudella is supplied sterile in a sealed package with a sterile single use inserter for placement. Do not open the package until required for insertion. Do not use if the seal of the sterile package is broken or appears compromised. Before considering use of Miudella, make sure that the patient is an appropriate candidate for Miudella. Exclude pregnancy (consider the possibility of ovulation and conception) prior to use [see Contraindications (4) and Warnings and Precautions (5) ]. Only a healthcare provider should insert Miudella. Healthcare providers should be thoroughly familiar with the product, product educational materials, product insertion instructions, and prescribing information before inserting Miudella. The IUS is provided preloaded in the inserter (see Figure A ). Remove Miudella on or before 3 years from the date of insertion. Replace Miudella at the time of removal with a new Miudella if continued contraceptive protection is desired. Figure A : Miudella Intrauterine System (IUS) Preloaded Inserter Figure B: Detailed diagram of Miudella
Warnings and Precautions
Risk of Ectopic Pregnancy : Promptly evaluate females who become pregnant for ectopic pregnancy while using Miudella. ( 5.3 ) Risks with Intrauterine Pregnancy : Increased risk of spontaneous abortion, septic abortion, premature delivery, sepsis, septic shock, and death if pregnancy occurs. Remove Miudella if pregnancy occurs with Miudella in place. ( 5.4 ) Sepsis : Group A streptococcal infection has been reported; strict aseptic technique is essential during insertion. ( 5.5 ) Pelvic Inflammatory Disease (PID) : Promptly evaluate patients with complaints of fever or abdominal pain after insertion of Miudella. ( 5.6 ) Perforation resulting in embedment or translocation : May reduce contraceptive effectiveness and require surgery. Risk is increased if inserted in postpartum and lactating females and may be increased if inserted in females with fixed, retroverted uteri or noninvoluted uteri. ( 5.7 ) Expulsion : Partial or complete expulsion may occur. Remove a partially expelled Miudella. ( 5.8 ) Bleeding patterns : May be altered and result in heavier and longer bleeding with spotting. ( 5.10 ) MRI Safety Information : Patients using Miudella can be safely scanned with MRI only under certain conditions. ( 5.11 )
Contraindications
The use of Miudella is contraindicated when one or more of the following conditions exist: Pregnancy or suspicion of pregnancy [see Warnings and Precautions (5.4) and Use in Specific Populations (8.1) ] Congenital or acquired abnormalities of the uterus, including leiomyomas, resulting in distortion of the uterine cavity Acute pelvic inflammatory disease (PID) [see Warnings and Precautions (5.6) ] Postpartum endometritis or postabortal endometritis in the past 3 months. [see Warnings and Precautions (5.6) ] Known or suspected uterine or cervical malignancy For use as post-coital contraception (emergency contraception) Uterine bleeding of unknown etiology Untreated acute cervicitis or vaginitis or other lower genital tract infection Conditions associated with increased susceptibility to pelvic infections [see Warnings and Precautions (5.6) ] Wilson's disease [see Warnings and Precautions (5.9) ] A previously placed IUS that has not been removed Hypersensitivity to any component of Miudella including to polypropylene, copper, nitinol, an alloy of nickel and titanium, or any of the trace elements present in the copper component of Miudella [see Adverse Reactions (6) and Description (11) ] . Persons with allergic reactions to these components may suffer an allergic reaction to this intrauterine system. Prior to placement, patients should be counseled on the materials contained in the IUS, as well as potential for allergy/hypersensitivity to these materials.
Adverse Reactions
The following serious adverse reactions are discussed elsewhere in the labeling: Ectopic pregnancy [see Warnings and Precautions (5.3) ] Intrauterine pregnancy [see Warnings and Precautions (5.4) ] Sepsis [see Warnings and Precautions (5.5) ] Pelvic Infection [see Warnings and Precautions (5.6) ] Perforation [see Warnings and Precautions (5.7) ] Expulsion [see Warnings and Precautions (5.8) ] Wilson's Diseas e [see Warnings and Precautions (5.9) ] Bleeding Pattern Alteration [see Warnings and Precautions (5.10) ]
Storage and Handling
Miudella (copper intrauterine system) is a copper-containing sterile IUS preloaded in a single use inserter. Each single-use unit is packaged in a sealed tray with lid (NDC-82686-300-01). The IUS is comprised of a nitinol frame with 10 copper sleeves attached to provide a total copper surface area of 175mm 2 , and a monofilament polymer retrieval thread tied to the vertical arm.
How Supplied
Miudella (copper intrauterine system) is a copper-containing sterile IUS preloaded in a single use inserter. Each single-use unit is packaged in a sealed tray with lid (NDC-82686-300-01). The IUS is comprised of a nitinol frame with 10 copper sleeves attached to provide a total copper surface area of 175mm 2 , and a monofilament polymer retrieval thread tied to the vertical arm.
Medication Information
Warnings and Precautions
Risk of Ectopic Pregnancy : Promptly evaluate females who become pregnant for ectopic pregnancy while using Miudella. ( 5.3 ) Risks with Intrauterine Pregnancy : Increased risk of spontaneous abortion, septic abortion, premature delivery, sepsis, septic shock, and death if pregnancy occurs. Remove Miudella if pregnancy occurs with Miudella in place. ( 5.4 ) Sepsis : Group A streptococcal infection has been reported; strict aseptic technique is essential during insertion. ( 5.5 ) Pelvic Inflammatory Disease (PID) : Promptly evaluate patients with complaints of fever or abdominal pain after insertion of Miudella. ( 5.6 ) Perforation resulting in embedment or translocation : May reduce contraceptive effectiveness and require surgery. Risk is increased if inserted in postpartum and lactating females and may be increased if inserted in females with fixed, retroverted uteri or noninvoluted uteri. ( 5.7 ) Expulsion : Partial or complete expulsion may occur. Remove a partially expelled Miudella. ( 5.8 ) Bleeding patterns : May be altered and result in heavier and longer bleeding with spotting. ( 5.10 ) MRI Safety Information : Patients using Miudella can be safely scanned with MRI only under certain conditions. ( 5.11 )
Indications and Usage
Miudella is indicated for prevention of pregnancy in females of reproductive potential for up to 3 years.
Dosage and Administration
Miudella is supplied sterile in a sealed package with a sterile single use inserter for placement. Do not open the package until required for insertion. Do not use if the seal of the sterile package is broken or appears compromised. Before considering use of Miudella, make sure that the patient is an appropriate candidate for Miudella. Exclude pregnancy (consider the possibility of ovulation and conception) prior to use [see Contraindications (4) and Warnings and Precautions (5) ]. Only a healthcare provider should insert Miudella. Healthcare providers should be thoroughly familiar with the product, product educational materials, product insertion instructions, and prescribing information before inserting Miudella. The IUS is provided preloaded in the inserter (see Figure A ). Remove Miudella on or before 3 years from the date of insertion. Replace Miudella at the time of removal with a new Miudella if continued contraceptive protection is desired. Figure A : Miudella Intrauterine System (IUS) Preloaded Inserter Figure B: Detailed diagram of Miudella
Contraindications
The use of Miudella is contraindicated when one or more of the following conditions exist: Pregnancy or suspicion of pregnancy [see Warnings and Precautions (5.4) and Use in Specific Populations (8.1) ] Congenital or acquired abnormalities of the uterus, including leiomyomas, resulting in distortion of the uterine cavity Acute pelvic inflammatory disease (PID) [see Warnings and Precautions (5.6) ] Postpartum endometritis or postabortal endometritis in the past 3 months. [see Warnings and Precautions (5.6) ] Known or suspected uterine or cervical malignancy For use as post-coital contraception (emergency contraception) Uterine bleeding of unknown etiology Untreated acute cervicitis or vaginitis or other lower genital tract infection Conditions associated with increased susceptibility to pelvic infections [see Warnings and Precautions (5.6) ] Wilson's disease [see Warnings and Precautions (5.9) ] A previously placed IUS that has not been removed Hypersensitivity to any component of Miudella including to polypropylene, copper, nitinol, an alloy of nickel and titanium, or any of the trace elements present in the copper component of Miudella [see Adverse Reactions (6) and Description (11) ] . Persons with allergic reactions to these components may suffer an allergic reaction to this intrauterine system. Prior to placement, patients should be counseled on the materials contained in the IUS, as well as potential for allergy/hypersensitivity to these materials.
Adverse Reactions
The following serious adverse reactions are discussed elsewhere in the labeling: Ectopic pregnancy [see Warnings and Precautions (5.3) ] Intrauterine pregnancy [see Warnings and Precautions (5.4) ] Sepsis [see Warnings and Precautions (5.5) ] Pelvic Infection [see Warnings and Precautions (5.6) ] Perforation [see Warnings and Precautions (5.7) ] Expulsion [see Warnings and Precautions (5.8) ] Wilson's Diseas e [see Warnings and Precautions (5.9) ] Bleeding Pattern Alteration [see Warnings and Precautions (5.10) ]
Storage and Handling
Miudella (copper intrauterine system) is a copper-containing sterile IUS preloaded in a single use inserter. Each single-use unit is packaged in a sealed tray with lid (NDC-82686-300-01). The IUS is comprised of a nitinol frame with 10 copper sleeves attached to provide a total copper surface area of 175mm 2 , and a monofilament polymer retrieval thread tied to the vertical arm.
How Supplied
Miudella (copper intrauterine system) is a copper-containing sterile IUS preloaded in a single use inserter. Each single-use unit is packaged in a sealed tray with lid (NDC-82686-300-01). The IUS is comprised of a nitinol frame with 10 copper sleeves attached to provide a total copper surface area of 175mm 2 , and a monofilament polymer retrieval thread tied to the vertical arm.
Description
Improper insertion of intrauterine systems, including Miudella, increases the risk of complications [see Warnings and Precautions (5.1) ] . Proper training prior to first use of Miudella can minimize the risk of improper Miudella insertion [see Warnings and Precautions (5.1) ]. Miudella is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the Miudella REMS program to ensure healthcare providers are trained on the proper insertion of Miudella prior to first use [see Warnings and Precautions (5.2) ].
Section 42229-5
Following placement:
- Examine the female after her first menses to confirm that Miudella is still in place. You should be able to visualize or feel only the threads. The length of the visible threads may change with time. However, no action is needed unless you suspect partial expulsion, perforation, pregnancy, or breakage.
- If you cannot find the threads in the vagina, check that Miudella is still in the uterus. The threads can retract into the uterus or break, or Miudella can break, perforate the uterus, or be expelled. Gentle probing of the cavity, x-ray, or sonography may be required to locate Miudella.
- Remove Miudella if it has been partially expelled or perforated the uterus [see Warnings and Precautions (5.7, 5.8)].
Section 44425-7
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
5.5 Sepsis
Severe infection or sepsis, including Group A streptococcal sepsis (GAS), have been reported following insertion of IUSs. In some cases, severe pain occurred within hours of insertion followed by sepsis within days. Because death from GAS is more likely if treatment is delayed, it is important to be aware of these rare but serious infections. Aseptic technique during insertion of Miudella is essential to minimize serious infections such as GAS.
11.1 Miudella
Miudella (copper intrauterine system) contains 99.99% pure copper for use as a contraceptive. Copper (Cu) has a molecular weight of 63.546 g/mol. Miudella consists of ten copper sleeves attached to a flexible nitinol (nickel-titanium alloy) frame measuring 32 mm horizontally and 30 mm vertically, and a polypropylene monofilament retrieval thread tied to the vertical stem. Miudella has a total exposed copper surface area of 175 mm 2and is provided sterile and preloaded in an inserter.
- Figure C: Diagram of Miudella
11.2 Inserter
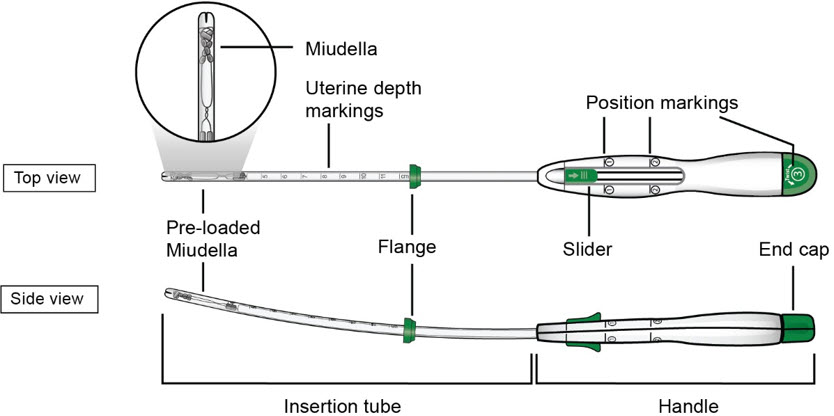
The inserter device provided with Miudella is a single use, disposable, sterile insertion system (insertion tube with a tapered rounded tip, depth markings and flange, and handle with slider and endcap); preloaded with the IUS for intrauterine administration. Once Miudella has been inserted, the inserter is discarded.
Figure D: Diagram of Inserter
5.8 Expulsion
Partial or complete expulsion of Miudella has been reported, resulting in the loss of contraceptive protection. The incidence of expulsion in the clinical trials with Miudella was 3.6% through 3 years of use. Consider further diagnostic imaging, such as x-ray, to confirm expulsion if the IUS is not found in the uterus on ultrasound.
Miudella should be placed no earlier than 4 weeks post-pregnancy to mitigate the risk of expulsion that may be increased when the uterus is not completely involuted at the time of insertion. Remove a partially expelled Miudella and do not attempt to push a partially expelled Miudella into the uterus. If expulsion has occurred, a new Miudella may be inserted when there is reasonable certainty the patient is not pregnant.
5.7 Perforation
Partial or total perforation of the uterine wall or cervix may occur during insertions, although the perforation may not be detected until sometime later. Perforation may also occur at any time during IUS use. Perforation that results in embedment or translocation may reduce contraceptive efficacy and result in pregnancy. The incidence of perforation during or following Miudella insertion in clinical trials was 0.1% (2 out of 1904).
A post-marketing safety study conducted in Europe (EURAS IUS) with IUSs, including copper IUSs, demonstrated an increased risk of perforation in postpartum and lactating women. The risk of perforation may be increased if an IUS, such as Miudella, is inserted when the uterus is fixed, retroverted or not completely involuted during the postpartum period.
If perforation is suspected or if known perforation occurs during placement, the IUS should be removed as soon as possible. Surgery may be required. Preoperative imaging followed by laparoscopy or laparotomy may be required to remove the IUS from the peritoneal cavity. Delayed detection or removal of Miudella in cases of perforation may result in migration outside the uterine cavity, adhesions, peritonitis, intestinal penetration, intestinal obstruction, abscesses and/or damage to adjacent organs.
5.2 Miudella Rems
Miudella is only available through a restricted program under a REMS called Miudella REMS Programto ensure healthcare providers are trained prior to first use [see Warnings and Precautions (5.1)]. Notable requirements include the following:
- Healthcare providers must be certified with the program by enrolling and completing training on the proper insertion of Miudella prior to first use [see Warnings and Precautions (5.2)].
- Pharmacies and healthcare settings that dispense Miudella must be certified by enrolling in the REMS and must only dispense Miudella to certified healthcare providers.
Further information is available at miudellarems.com and 1-855-337-0772.
8.4 Pediatric Use
The safety and effectiveness of Miudella have been established in females of reproductive potential. Efficacy is expected to be the same for postmenarcheal females regardless of age. Miudella is not indicated in females before menarche.
8.5 Geriatric Use
Miudella has not been studied in women over 65 years of age and is not indicated in this population.
14 Clinical Studies
The efficacy of Miudella for the prevention of pregnancy in women of reproductive potential for up to 3 years was demonstrated in Study 1 (NCT03633799), a multi-center, single-arm, open-label study conducted in the U.S. Women were excluded if they were less than six weeks postpartum, had a history of previous IUS complications, were known to be HIV positive, or were otherwise at high risk for sexually transmitted infections. There were no exclusions based on BMI.
Study 1 included 1601 generally healthy women aged 17 to 45 years who had Miudella successfully placed. Of these 60% (960/1601) were nulliparous. Successful placement of Miudella with first attempt occurred in 96% of the subjects and 99% with two insertion attempts by healthcare providers who received training on proper insertion prior to first use. The median age was 27 years, the mean weight was 75.3 kg (range 40.6 kg to 170.1 kg); mean BMI was 27.9 kg/m 2(range 15.8 kg/m 2to 63.5 kg/m 2); 74% were White, 15% were Black, 5% were Asian, and 6% were Other; 19% were Hispanic.
The primary endpoint for Study 1 was the contraceptive efficacy of Miudella through 3 years of use as measured by the Pearl Index (PI) in women 17 to 35 years of age. The PI was calculated based on 28-day equivalent exposure cycles; evaluable cycles excluded those in which no intercourse occurred, or back-up contraception was used unless a pregnancy occurred in that cycle.
Women enrolled in the study provided 12,493 evaluable 28-day cycle equivalents in the first year and 27,115 evaluable cycles over the three-year treatment period. The PI for Year 1 was based on 9 pregnancies and the cumulative 3-year pregnancy rate was based on 22 pregnancies that occurred after the onset of treatment and within 7 days after Miudella removal or expulsion.
Table 4 shows the calculated annual pregnancy rates and cumulative 3-year pregnancy rate estimated by Pearl Index.
| Miudella Clinical Trial | Pearl Index | Cumulative 3-Year
Pearl Index |
||
|---|---|---|---|---|
| Year 1 | Year 2 | Year 3 | ||
| Number of Evaluable 28- day Cycles of Exposure | 12,493 | 8,150 | 6,472 | 27,115 |
| Pregnancy Rate
(95% Confidence Interval) |
0.94
(0.43,1.78) |
1.60
(0.76, 2.93) |
0.60
(0.12, 1.76) |
1.05
(0.66, 1.60) |
Of 54 subjects who desired pregnancy after study discontinuation and provided follow-up information, approximately 74.1% conceived within 12 months after removal of Miudella. The majority of these subjects conceived within 4 months of removal and as early as the first week post-removal, demonstrating the reversibility of Miudella.
4 Contraindications
The use of Miudella is contraindicated when one or more of the following conditions exist:
- Pregnancy or suspicion of pregnancy [see Warnings and Precautions (5.4)and Use in Specific Populations (8.1)]
- Congenital or acquired abnormalities of the uterus, including leiomyomas, resulting in distortion of the uterine cavity
- Acute pelvic inflammatory disease (PID) [see Warnings and Precautions (5.6)]
- Postpartum endometritis or postabortal endometritis in the past 3 months. [see Warnings and Precautions (5.6)]
- Known or suspected uterine or cervical malignancy
- For use as post-coital contraception (emergency contraception)
- Uterine bleeding of unknown etiology
- Untreated acute cervicitis or vaginitis or other lower genital tract infection
- Conditions associated with increased susceptibility to pelvic infections [see Warnings and Precautions (5.6)]
- Wilson's disease [see Warnings and Precautions (5.9) ]
- A previously placed IUS that has not been removed
- Hypersensitivity to any component of Miudella including to polypropylene, copper, nitinol, an alloy of nickel and titanium, or any of the trace elements present in the copper component of Miudella [see Adverse Reactions (6)and Description (11)] . Persons with allergic reactions to these components may suffer an allergic reaction to this intrauterine system. Prior to placement, patients should be counseled on the materials contained in the IUS, as well as potential for allergy/hypersensitivity to these materials.
6 Adverse Reactions
The following serious adverse reactions are discussed elsewhere in the labeling:
- Ectopic pregnancy [see Warnings and Precautions (5.3)]
- Intrauterine pregnancy [see Warnings and Precautions (5.4)]
- Sepsis [see Warnings and Precautions (5.5)]
- Pelvic Infection [see Warnings and Precautions (5.6)]
- Perforation [see Warnings and Precautions (5.7)]
- Expulsion [see Warnings and Precautions (5.8)]
- Wilson's Diseas e [see Warnings and Precautions (5.9)]
- Bleeding Pattern Alteration [see Warnings and Precautions (5.10)]
5.6 Pelvic Infection
Promptly examine users with complaints of lower abdominal or pelvic pain, odorous discharge, unexplained bleeding, fever, genital lesions or sores. Remove Miudella in cases of recurrent pelvic inflammatory disease or endometritis, or if an acute pelvic infection is severe or does not respond to treatment.
5.9 Wilson's Disease
Miudella may exacerbate Wilson's disease, a rare genetic disease affecting copper excretion; therefore, the use of Miudella is contraindicated in females with Wilson's disease [see Contraindications (4)] .
12.2 Pharmacodynamics
The exposure-response relationship and time course of pharmacodynamic response for the safety and effectiveness of Miudella have not been fully characterized.
12.3 Pharmacokinetics
The pharmacokinetics (PK) of copper following insertion of Miudella were investigated in 20 healthy premenopausal female adult subjects. Table 3 shows PK parameters of serum copper with or without baseline-adjustment following 57 days after insertion, and serum copper concentrations during 3 years after insertion.
| Parameters | Mean (standard deviation) | ||
|---|---|---|---|
| No adjustment | Baseline-adjusted | ||
| C max: peak concentration | |||
| C: concentration | |||
| AUC: area under the serum concentration-time curve | |||
| PK parameters | C max, Day1-57(ng/mL) | 1210 (202) | 63.5 (84.1) |
| AUC Day1-57(day×ng/mL) | 59700 (8310) | 992 (1680) | |
| C Year1(ng/mL) | 965 (197) | 7.5 (19.4) | |
| C Year2(ng/mL) | 1020 (168) | 25.7 (48.7) | |
| C Year3(ng/mL) | 986 (148) | 17.2 (43.0) |
5.12 Medical Diathermy
Medical equipment that contains high levels of Radiofrequency (RF) energy such as diathermy may cause health effects (by heating tissue) in females with a metal-containing IUS including Miudella. Avoid using high medical RF transmitter devices in females with Miudella.
1 Indications and Usage
Miudella is indicated for prevention of pregnancy in females of reproductive potential for up to 3 years.
2.2 Timing of Insertion
- Miudella can be inserted at any time during the menstrual cycle. If switching to Miudella from another contraceptive, discontinue the previous method (see Table 1).
| Clinical Scenario | Recommended Timing of Miudella Insertion |
|---|---|
|
At any time during the menstrual cycle. |
|
At any time during the menstrual cycle; discontinue the previous method. |
|
Same day the implant or IUS is removed (insert at any time during the menstrual cycle). |
2.4 Insertion Procedure
| Step 1 – Open the Sterile Package | |||
|
|||
|
c. |
||
| • Step 2 – Setting the Flange | |||
|
a. |
||
|
c. |
||
| • Step 3 – Inserter Placement | |||
|
a. |
||
| Step 4 -Miudella Placement | |||
|
a. |
||
|
b. |
||
|
c. |
||
|
d. |
||
|
|||
|
|||
|
iii. |
||
|
iv. |
||
|
-
Step 5 – Confirm the Retrieval Thread Ends
- Confirm that the blue retrieval thread ends are visible past the external os. The retrieval thread ends are pre-cut to typically allow 3-4 cm of exposed thread in the vaginal cavity without the need for trimming, and hence cutting of excess thread length is generally not required.
- Should additional trimming of the thread ends be desired, cut the thread ends perpendicular to the thread length, for example, with sterile curved scissors, leaving the desired amount of visible length outside the cervix.
- Remove the tenaculum and achieve adequate hemostasis.
- Remove speculum from the vaginal cavity.
- Miudella placement is now complete.
-
Important information to consider during and after placement:
- If you suspect that Miudella is not in the correct position, check placement (for example, with transvaginal ultrasound). Remove Miudella if it is not positioned completely within the uterus. A removed Miudella must not be reused.
- If there is clinical concern and/or exceptional pain or bleeding during or after placement, conduct appropriate and timely measures and assessments. For example, ultrasound should be performed to exclude perforation, embedment, or translocation.
12.1 Mechanism of Action
Copper continuously released into the uterine cavity contributes to the contraceptive effectiveness of Miudella. Mechanism(s) by which copper enhances contraceptive efficacy include interference with sperm transport and fertilization of an egg.
5 Warnings and Precautions
- Risk of Ectopic Pregnancy: Promptly evaluate females who become pregnant for ectopic pregnancy while using Miudella. ( 5.3)
- Risks with Intrauterine Pregnancy: Increased risk of spontaneous abortion, septic abortion, premature delivery, sepsis, septic shock, and death if pregnancy occurs. Remove Miudella if pregnancy occurs with Miudella in place. ( 5.4)
- Sepsis: Group A streptococcal infection has been reported; strict aseptic technique is essential during insertion. ( 5.5)
- Pelvic Inflammatory Disease (PID): Promptly evaluate patients with complaints of fever or abdominal pain after insertion of Miudella. ( 5.6)
- Perforation resulting in embedment or translocation: May reduce contraceptive effectiveness and require surgery. Risk is increased if inserted in postpartum and lactating females and may be increased if inserted in females with fixed, retroverted uteri or noninvoluted uteri. ( 5.7)
- Expulsion: Partial or complete expulsion may occur. Remove a partially expelled Miudella. ( 5.8)
- Bleeding patterns: May be altered and result in heavier and longer bleeding with spotting. ( 5.10)
- MRI Safety Information: Patients using Miudella can be safely scanned with MRI only under certain conditions. ( 5.11)
2 Dosage and Administration
- Insert a single Miudella at the fundus of the uterine cavity. Miudella must be removed or replaced after 3 years. ( 2.1)
- Insert Miudella only if you are a trained healthcare provider using clean technique. Follow insertion instructions exactly as described. ( 2.1)
- See the Full Prescribing Information for recommended timing of insertion, preparation instructions, insertion procedures, postplacement management, and instructions on removing Miudella. ( 2.2, 2.3, 2.4, 2.5, 2.6)
- Patient can be re-examined as clinically indicated.
3 Dosage Forms and Strengths
2.3 Preparation for Insertion
- Ensure use of clean technique during the procedure.
- Perform a bimanual exam to establish the size, shape, and position of the uterus and to rule out abnormalities that would preclude safe placement of Miudella.
- Gently insert a speculum to visualize the cervix.
- Thoroughly cleanse the cervix with chlorhexidine or povidone-iodine.
- Apply a sterile tenaculum to the cervix. Apply traction with the tenaculum. Gently advance a sterile uterine sound to the fundus. Note any uterine anomaly and the depth in centimeters.
5.3 Risk of Ectopic Pregnancy
Evaluate for possible ectopic pregnancy in any female who becomes pregnant while using Miudella because a pregnancy that occurs with Miudella in place is more likely to be ectopic than a pregnancy in the general population. However, because Miudella prevents most pregnancies, females who use Miudella have a lower risk of an ectopic pregnancy than sexually active females who do not use any contraception.
The incidence of ectopic pregnancy in the clinical trials with Miudella was approximately 0.3%. Ectopic pregnancy may require surgery and may result in loss of fertility.
Patients who use Miudella should be informed about recognizing the signs and symptoms of ectopic pregnancy and promptly reporting them to their healthcare professional, and about the associated risks of ectopic pregnancy (e.g., loss of fertility).
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The data described below reflect exposure of 1,904 healthy 17- to 45-year-old women (mean age 27.5 ± 5.72 years) to Miudella. These data come from two multi-center contraceptive trials with a 3-year duration and a single, multi-center comparative bioavailability study with a 3 year duration, all conducted in the United States, enrolling generally healthy, postmenarcheal females to 45 years old. In total 1,904 subjects were exposed to Miudella for one year and 916 completed three years. The data in these trials cover approximately 51,692 cycles of exposure.
The following adverse reactions have been observed in ≥5% of users in clinical trials with Miudella: heavy menstrual bleeding, dysmenorrhea, intermenstrual bleeding, pelvic discomfort, procedural pain, pelvic pain, post procedural hemorrhage, and dyspareunia.
Table 2 shows discontinuation rates from the 3 multi-center clinical studies by adverse reaction and year.
| Year | |||
|---|---|---|---|
| 1 | 2 | 3 | |
| Number of Women at Start of Year | 1,904 | 1,463 | 1,118 |
|
Bleeding/Pain
Bleeding/Pain includes the preferred terms (PTs) Abdominal pain, Abdominal pain lower, Post procedural discomfort, Procedural pain, Coital bleeding, Dysmenorrhea, Dyspareunia, Heavy menstrual bleeding, Intermenstrual bleeding, Menometrorrhagia, Pelvic discomfort, Pelvic pain, Polymenorrhea, Uterine hemorrhage, Uterine spasm
|
8.5 | 5.1 | 3.2 |
| Expulsion | 1.9 | 1.0 | 0.9 |
|
Other Medical Event
Includes PTs embedded device (n=6) and uterine perforation (n=1)
|
0.2 | 0.1 | 0.3 |
According to the safety analyses of these three studies, it was determined the incidence of expulsion of Miudella did not vary significantly by body mass index (BMI). There was no statistically significant difference in the incidence of expulsion of Miudella in normal weight versus obese females.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Before inserting Miudella, counsel patients on the following:
5.10 Bleeding Pattern Alterations
Miudella can alter the bleeding pattern and result in heavier and longer menstrual cycles with intermenstrual spotting.
In three clinical trials with Miudella [see Adverse Reactions (6.1)] , menstrual changes were the most common medical reason for discontinuation. Discontinuation rates for pain and bleeding combined were highest in the first year of use and diminished thereafter. The percentage of females who discontinued Miudella because of bleeding problems or pain during this study ranged from 8.5% in the first year to 3.2% in Year 3. Females complaining of heavy vaginal bleeding should be evaluated and treated, and may need to discontinue Miudella [see Adverse Reactions (6.1)] .
16 How Supplied/storage and Handling
Miudella (copper intrauterine system) is a copper-containing sterile IUS preloaded in a single use inserter. Each single-use unit is packaged in a sealed tray with lid (NDC-82686-300-01). The IUS is comprised of a nitinol frame with 10 copper sleeves attached to provide a total copper surface area of 175mm 2, and a monofilament polymer retrieval thread tied to the vertical arm.
5.4 Risks With Intrauterine Pregnancy
If intrauterine pregnancy occurs with Miudella in place and the thread ends are visible or can be retrieved from the cervical canal, remove Miudella. Leaving it in place may increase the risk of spontaneous abortion and preterm labor. Removal of Miudella may also result in spontaneous abortion. In the event of an intrauterine pregnancy with Miudella, consider the following:
Principal Display Panel 175 Mm Unit Tray Box
NDC 82686-300-01
Miudella
intrauterine copper contraceptive
Important: To be inserted in the uterus by a trained heathcare
provider by carefully following the insertion instructions.
Rx Only
ONE (1) STERILE UNIT: INTRAUTERINE USE
Contents: One (1) sterile packaged
system containing one intrauterine
copper contraceptive with approximately
175 mm
2of copper surface area,
preloaded in a single-use disposable
inserter. A Patient Information Booklet
is provided with each unit.
Ensure the Patient Information Booklet is
provided to the patient and review
contents prior to placement.
Insert MIUDELLA before the expiration
date printed on this box.
5.1 Risk of Complications Due to Improper Insertion
Improper insertion of intrauterine systems, including Miudella, increases the risk of perforation, infection, undiagnosed abnormal bleeding, pregnancy loss (if pregnancy occurs with IUS in situ), and expulsion.
Proper training prior to first use of Miudella can minimize the risk of improper insertion. Miudella is available only through a restricted program under a REMS [see Warnings and Precautions (5.2)].
2.1 Important Dosage and Administration Instructions
- Miudella is supplied sterile in a sealed package with a sterile single use inserter for placement. Do not open the package until required for insertion. Do not use if the seal of the sterile package is broken or appears compromised.
- Before considering use of Miudella, make sure that the patient is an appropriate candidate for Miudella. Exclude pregnancy (consider the possibility of ovulation and conception) prior to use [see Contraindications (4)and Warnings and Precautions (5)].
- Only a healthcare provider should insert Miudella. Healthcare providers should be thoroughly familiar with the product, product educational materials, product insertion instructions, and prescribing information before inserting Miudella.
- The IUS is provided preloaded in the inserter (see Figure A).
- Remove Miudella on or before 3 years from the date of insertion.
- Replace Miudella at the time of removal with a new Miudella if continued contraceptive protection is desired.
FigureA : Miudella Intrauterine System (IUS) Preloaded Inserter
- Figure B: Detailed diagram of Miudella
5.11 Magnetic Resonance Imaging (mri) Safety Information
| MRI Safety Information MR Conditional | The
Miudella Intrauterine Device (IUD)is MR Conditional. A patient with
Miudellamay be safely scanned under the following conditions.
Failure to follow these conditions may result in injury to the patient. |
| Nominal Values of Static Magnetic Field (T) | 1.5-Tesla or 3.0-Tesla |
| Maximum Spatial Field Gradient | 40 T/m (4,000 gauss/cm) |
| Type of RF Excitation | Circularly Polarized (CP) (i.e., Quadrature-Transmission) |
| Transmit RF Coil Information | There are no transmit RF coil restrictions. Accordingly, the following may be used: body transmit RF coil and all other RF coil combinations (i.e., body RF coil combined with any receive-only RF coil, transmit/receive head RF coil, transmit/receive knee RF coil, etc.) |
| Operating Mode | Normal Operating Mode |
| Maximum Whole-Body Averaged SAR | 2 W/kg (Normal Operating Mode) |
| Limits on Scan Duration | 2 W/kg whole body average SAR for 60 minutes of continuous RF exposure (a sequence or back to back sequences/series without breaks) |
| MR Image Artifact | The presence of this implant produces an imaging artifact. In testing with gradient-echo sequencing, the shape of the image artifact follows the approximate contour of the device and extends radially up to 0.7 cm from the device. |
Warning: Risk of Complications Due to Improper Insertion
- Improper insertion of intrauterine systems, including Miudella, increases the risk of complications [see Warnings and Precautions (5.1)] .
- Proper training prior to first use of Miudella can minimize the risk of improper Miudella insertion [see Warnings and Precautions (5.1)].
- Miudella is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the Miudella REMS program to ensure healthcare providers are trained on the proper insertion of Miudella prior to first use [see Warnings and Precautions (5.2)].
13.1 Carcinogenicity, Mutagenicity, Impairment of Fertility
Adequate long-term studies in animals to assess the carcinogenic potential of a copper-containing IUS have not been performed.
A chemical characterization and toxicological risk assessment of the extractables derived from the copper-containing IUS were utilized to address carcinogenicity, mutagenicity and reproductive/developmental toxicity risks. The toxicological risk assessment indicates that the risks of carcinogenicity, mutagenicity and reproductive/developmental toxicity caused by the copper-containing IUS are negligible.
Structured Label Content
Section 42229-5 (42229-5)
Following placement:
- Examine the female after her first menses to confirm that Miudella is still in place. You should be able to visualize or feel only the threads. The length of the visible threads may change with time. However, no action is needed unless you suspect partial expulsion, perforation, pregnancy, or breakage.
- If you cannot find the threads in the vagina, check that Miudella is still in the uterus. The threads can retract into the uterus or break, or Miudella can break, perforate the uterus, or be expelled. Gentle probing of the cavity, x-ray, or sonography may be required to locate Miudella.
- Remove Miudella if it has been partially expelled or perforated the uterus [see Warnings and Precautions (5.7, 5.8)].
Section 44425-7 (44425-7)
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
5.5 Sepsis
Severe infection or sepsis, including Group A streptococcal sepsis (GAS), have been reported following insertion of IUSs. In some cases, severe pain occurred within hours of insertion followed by sepsis within days. Because death from GAS is more likely if treatment is delayed, it is important to be aware of these rare but serious infections. Aseptic technique during insertion of Miudella is essential to minimize serious infections such as GAS.
11.1 Miudella
Miudella (copper intrauterine system) contains 99.99% pure copper for use as a contraceptive. Copper (Cu) has a molecular weight of 63.546 g/mol. Miudella consists of ten copper sleeves attached to a flexible nitinol (nickel-titanium alloy) frame measuring 32 mm horizontally and 30 mm vertically, and a polypropylene monofilament retrieval thread tied to the vertical stem. Miudella has a total exposed copper surface area of 175 mm 2and is provided sterile and preloaded in an inserter.
- Figure C: Diagram of Miudella
11.2 Inserter
The inserter device provided with Miudella is a single use, disposable, sterile insertion system (insertion tube with a tapered rounded tip, depth markings and flange, and handle with slider and endcap); preloaded with the IUS for intrauterine administration. Once Miudella has been inserted, the inserter is discarded.
Figure D: Diagram of Inserter
5.8 Expulsion
Partial or complete expulsion of Miudella has been reported, resulting in the loss of contraceptive protection. The incidence of expulsion in the clinical trials with Miudella was 3.6% through 3 years of use. Consider further diagnostic imaging, such as x-ray, to confirm expulsion if the IUS is not found in the uterus on ultrasound.
Miudella should be placed no earlier than 4 weeks post-pregnancy to mitigate the risk of expulsion that may be increased when the uterus is not completely involuted at the time of insertion. Remove a partially expelled Miudella and do not attempt to push a partially expelled Miudella into the uterus. If expulsion has occurred, a new Miudella may be inserted when there is reasonable certainty the patient is not pregnant.
5.7 Perforation
Partial or total perforation of the uterine wall or cervix may occur during insertions, although the perforation may not be detected until sometime later. Perforation may also occur at any time during IUS use. Perforation that results in embedment or translocation may reduce contraceptive efficacy and result in pregnancy. The incidence of perforation during or following Miudella insertion in clinical trials was 0.1% (2 out of 1904).
A post-marketing safety study conducted in Europe (EURAS IUS) with IUSs, including copper IUSs, demonstrated an increased risk of perforation in postpartum and lactating women. The risk of perforation may be increased if an IUS, such as Miudella, is inserted when the uterus is fixed, retroverted or not completely involuted during the postpartum period.
If perforation is suspected or if known perforation occurs during placement, the IUS should be removed as soon as possible. Surgery may be required. Preoperative imaging followed by laparoscopy or laparotomy may be required to remove the IUS from the peritoneal cavity. Delayed detection or removal of Miudella in cases of perforation may result in migration outside the uterine cavity, adhesions, peritonitis, intestinal penetration, intestinal obstruction, abscesses and/or damage to adjacent organs.
5.2 Miudella Rems (5.2 Miudella REMS)
Miudella is only available through a restricted program under a REMS called Miudella REMS Programto ensure healthcare providers are trained prior to first use [see Warnings and Precautions (5.1)]. Notable requirements include the following:
- Healthcare providers must be certified with the program by enrolling and completing training on the proper insertion of Miudella prior to first use [see Warnings and Precautions (5.2)].
- Pharmacies and healthcare settings that dispense Miudella must be certified by enrolling in the REMS and must only dispense Miudella to certified healthcare providers.
Further information is available at miudellarems.com and 1-855-337-0772.
8.4 Pediatric Use
The safety and effectiveness of Miudella have been established in females of reproductive potential. Efficacy is expected to be the same for postmenarcheal females regardless of age. Miudella is not indicated in females before menarche.
8.5 Geriatric Use
Miudella has not been studied in women over 65 years of age and is not indicated in this population.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of Miudella for the prevention of pregnancy in women of reproductive potential for up to 3 years was demonstrated in Study 1 (NCT03633799), a multi-center, single-arm, open-label study conducted in the U.S. Women were excluded if they were less than six weeks postpartum, had a history of previous IUS complications, were known to be HIV positive, or were otherwise at high risk for sexually transmitted infections. There were no exclusions based on BMI.
Study 1 included 1601 generally healthy women aged 17 to 45 years who had Miudella successfully placed. Of these 60% (960/1601) were nulliparous. Successful placement of Miudella with first attempt occurred in 96% of the subjects and 99% with two insertion attempts by healthcare providers who received training on proper insertion prior to first use. The median age was 27 years, the mean weight was 75.3 kg (range 40.6 kg to 170.1 kg); mean BMI was 27.9 kg/m 2(range 15.8 kg/m 2to 63.5 kg/m 2); 74% were White, 15% were Black, 5% were Asian, and 6% were Other; 19% were Hispanic.
The primary endpoint for Study 1 was the contraceptive efficacy of Miudella through 3 years of use as measured by the Pearl Index (PI) in women 17 to 35 years of age. The PI was calculated based on 28-day equivalent exposure cycles; evaluable cycles excluded those in which no intercourse occurred, or back-up contraception was used unless a pregnancy occurred in that cycle.
Women enrolled in the study provided 12,493 evaluable 28-day cycle equivalents in the first year and 27,115 evaluable cycles over the three-year treatment period. The PI for Year 1 was based on 9 pregnancies and the cumulative 3-year pregnancy rate was based on 22 pregnancies that occurred after the onset of treatment and within 7 days after Miudella removal or expulsion.
Table 4 shows the calculated annual pregnancy rates and cumulative 3-year pregnancy rate estimated by Pearl Index.
| Miudella Clinical Trial | Pearl Index | Cumulative 3-Year
Pearl Index |
||
|---|---|---|---|---|
| Year 1 | Year 2 | Year 3 | ||
| Number of Evaluable 28- day Cycles of Exposure | 12,493 | 8,150 | 6,472 | 27,115 |
| Pregnancy Rate
(95% Confidence Interval) |
0.94
(0.43,1.78) |
1.60
(0.76, 2.93) |
0.60
(0.12, 1.76) |
1.05
(0.66, 1.60) |
Of 54 subjects who desired pregnancy after study discontinuation and provided follow-up information, approximately 74.1% conceived within 12 months after removal of Miudella. The majority of these subjects conceived within 4 months of removal and as early as the first week post-removal, demonstrating the reversibility of Miudella.
4 Contraindications (4 CONTRAINDICATIONS)
The use of Miudella is contraindicated when one or more of the following conditions exist:
- Pregnancy or suspicion of pregnancy [see Warnings and Precautions (5.4)and Use in Specific Populations (8.1)]
- Congenital or acquired abnormalities of the uterus, including leiomyomas, resulting in distortion of the uterine cavity
- Acute pelvic inflammatory disease (PID) [see Warnings and Precautions (5.6)]
- Postpartum endometritis or postabortal endometritis in the past 3 months. [see Warnings and Precautions (5.6)]
- Known or suspected uterine or cervical malignancy
- For use as post-coital contraception (emergency contraception)
- Uterine bleeding of unknown etiology
- Untreated acute cervicitis or vaginitis or other lower genital tract infection
- Conditions associated with increased susceptibility to pelvic infections [see Warnings and Precautions (5.6)]
- Wilson's disease [see Warnings and Precautions (5.9) ]
- A previously placed IUS that has not been removed
- Hypersensitivity to any component of Miudella including to polypropylene, copper, nitinol, an alloy of nickel and titanium, or any of the trace elements present in the copper component of Miudella [see Adverse Reactions (6)and Description (11)] . Persons with allergic reactions to these components may suffer an allergic reaction to this intrauterine system. Prior to placement, patients should be counseled on the materials contained in the IUS, as well as potential for allergy/hypersensitivity to these materials.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed elsewhere in the labeling:
- Ectopic pregnancy [see Warnings and Precautions (5.3)]
- Intrauterine pregnancy [see Warnings and Precautions (5.4)]
- Sepsis [see Warnings and Precautions (5.5)]
- Pelvic Infection [see Warnings and Precautions (5.6)]
- Perforation [see Warnings and Precautions (5.7)]
- Expulsion [see Warnings and Precautions (5.8)]
- Wilson's Diseas e [see Warnings and Precautions (5.9)]
- Bleeding Pattern Alteration [see Warnings and Precautions (5.10)]
5.6 Pelvic Infection
Promptly examine users with complaints of lower abdominal or pelvic pain, odorous discharge, unexplained bleeding, fever, genital lesions or sores. Remove Miudella in cases of recurrent pelvic inflammatory disease or endometritis, or if an acute pelvic infection is severe or does not respond to treatment.
5.9 Wilson's Disease
Miudella may exacerbate Wilson's disease, a rare genetic disease affecting copper excretion; therefore, the use of Miudella is contraindicated in females with Wilson's disease [see Contraindications (4)] .
12.2 Pharmacodynamics
The exposure-response relationship and time course of pharmacodynamic response for the safety and effectiveness of Miudella have not been fully characterized.
12.3 Pharmacokinetics
The pharmacokinetics (PK) of copper following insertion of Miudella were investigated in 20 healthy premenopausal female adult subjects. Table 3 shows PK parameters of serum copper with or without baseline-adjustment following 57 days after insertion, and serum copper concentrations during 3 years after insertion.
| Parameters | Mean (standard deviation) | ||
|---|---|---|---|
| No adjustment | Baseline-adjusted | ||
| C max: peak concentration | |||
| C: concentration | |||
| AUC: area under the serum concentration-time curve | |||
| PK parameters | C max, Day1-57(ng/mL) | 1210 (202) | 63.5 (84.1) |
| AUC Day1-57(day×ng/mL) | 59700 (8310) | 992 (1680) | |
| C Year1(ng/mL) | 965 (197) | 7.5 (19.4) | |
| C Year2(ng/mL) | 1020 (168) | 25.7 (48.7) | |
| C Year3(ng/mL) | 986 (148) | 17.2 (43.0) |
5.12 Medical Diathermy
Medical equipment that contains high levels of Radiofrequency (RF) energy such as diathermy may cause health effects (by heating tissue) in females with a metal-containing IUS including Miudella. Avoid using high medical RF transmitter devices in females with Miudella.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Miudella is indicated for prevention of pregnancy in females of reproductive potential for up to 3 years.
2.2 Timing of Insertion
- Miudella can be inserted at any time during the menstrual cycle. If switching to Miudella from another contraceptive, discontinue the previous method (see Table 1).
| Clinical Scenario | Recommended Timing of Miudella Insertion |
|---|---|
|
At any time during the menstrual cycle. |
|
At any time during the menstrual cycle; discontinue the previous method. |
|
Same day the implant or IUS is removed (insert at any time during the menstrual cycle). |
2.4 Insertion Procedure
| Step 1 – Open the Sterile Package | |||
|
|||
|
c. |
||
| • Step 2 – Setting the Flange | |||
|
a. |
||
|
c. |
||
| • Step 3 – Inserter Placement | |||
|
a. |
||
| Step 4 -Miudella Placement | |||
|
a. |
||
|
b. |
||
|
c. |
||
|
d. |
||
|
|||
|
|||
|
iii. |
||
|
iv. |
||
|
-
Step 5 – Confirm the Retrieval Thread Ends
- Confirm that the blue retrieval thread ends are visible past the external os. The retrieval thread ends are pre-cut to typically allow 3-4 cm of exposed thread in the vaginal cavity without the need for trimming, and hence cutting of excess thread length is generally not required.
- Should additional trimming of the thread ends be desired, cut the thread ends perpendicular to the thread length, for example, with sterile curved scissors, leaving the desired amount of visible length outside the cervix.
- Remove the tenaculum and achieve adequate hemostasis.
- Remove speculum from the vaginal cavity.
- Miudella placement is now complete.
-
Important information to consider during and after placement:
- If you suspect that Miudella is not in the correct position, check placement (for example, with transvaginal ultrasound). Remove Miudella if it is not positioned completely within the uterus. A removed Miudella must not be reused.
- If there is clinical concern and/or exceptional pain or bleeding during or after placement, conduct appropriate and timely measures and assessments. For example, ultrasound should be performed to exclude perforation, embedment, or translocation.
12.1 Mechanism of Action
Copper continuously released into the uterine cavity contributes to the contraceptive effectiveness of Miudella. Mechanism(s) by which copper enhances contraceptive efficacy include interference with sperm transport and fertilization of an egg.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Risk of Ectopic Pregnancy: Promptly evaluate females who become pregnant for ectopic pregnancy while using Miudella. ( 5.3)
- Risks with Intrauterine Pregnancy: Increased risk of spontaneous abortion, septic abortion, premature delivery, sepsis, septic shock, and death if pregnancy occurs. Remove Miudella if pregnancy occurs with Miudella in place. ( 5.4)
- Sepsis: Group A streptococcal infection has been reported; strict aseptic technique is essential during insertion. ( 5.5)
- Pelvic Inflammatory Disease (PID): Promptly evaluate patients with complaints of fever or abdominal pain after insertion of Miudella. ( 5.6)
- Perforation resulting in embedment or translocation: May reduce contraceptive effectiveness and require surgery. Risk is increased if inserted in postpartum and lactating females and may be increased if inserted in females with fixed, retroverted uteri or noninvoluted uteri. ( 5.7)
- Expulsion: Partial or complete expulsion may occur. Remove a partially expelled Miudella. ( 5.8)
- Bleeding patterns: May be altered and result in heavier and longer bleeding with spotting. ( 5.10)
- MRI Safety Information: Patients using Miudella can be safely scanned with MRI only under certain conditions. ( 5.11)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Insert a single Miudella at the fundus of the uterine cavity. Miudella must be removed or replaced after 3 years. ( 2.1)
- Insert Miudella only if you are a trained healthcare provider using clean technique. Follow insertion instructions exactly as described. ( 2.1)
- See the Full Prescribing Information for recommended timing of insertion, preparation instructions, insertion procedures, postplacement management, and instructions on removing Miudella. ( 2.2, 2.3, 2.4, 2.5, 2.6)
- Patient can be re-examined as clinically indicated.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
2.3 Preparation for Insertion
- Ensure use of clean technique during the procedure.
- Perform a bimanual exam to establish the size, shape, and position of the uterus and to rule out abnormalities that would preclude safe placement of Miudella.
- Gently insert a speculum to visualize the cervix.
- Thoroughly cleanse the cervix with chlorhexidine or povidone-iodine.
- Apply a sterile tenaculum to the cervix. Apply traction with the tenaculum. Gently advance a sterile uterine sound to the fundus. Note any uterine anomaly and the depth in centimeters.
5.3 Risk of Ectopic Pregnancy
Evaluate for possible ectopic pregnancy in any female who becomes pregnant while using Miudella because a pregnancy that occurs with Miudella in place is more likely to be ectopic than a pregnancy in the general population. However, because Miudella prevents most pregnancies, females who use Miudella have a lower risk of an ectopic pregnancy than sexually active females who do not use any contraception.
The incidence of ectopic pregnancy in the clinical trials with Miudella was approximately 0.3%. Ectopic pregnancy may require surgery and may result in loss of fertility.
Patients who use Miudella should be informed about recognizing the signs and symptoms of ectopic pregnancy and promptly reporting them to their healthcare professional, and about the associated risks of ectopic pregnancy (e.g., loss of fertility).
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The data described below reflect exposure of 1,904 healthy 17- to 45-year-old women (mean age 27.5 ± 5.72 years) to Miudella. These data come from two multi-center contraceptive trials with a 3-year duration and a single, multi-center comparative bioavailability study with a 3 year duration, all conducted in the United States, enrolling generally healthy, postmenarcheal females to 45 years old. In total 1,904 subjects were exposed to Miudella for one year and 916 completed three years. The data in these trials cover approximately 51,692 cycles of exposure.
The following adverse reactions have been observed in ≥5% of users in clinical trials with Miudella: heavy menstrual bleeding, dysmenorrhea, intermenstrual bleeding, pelvic discomfort, procedural pain, pelvic pain, post procedural hemorrhage, and dyspareunia.
Table 2 shows discontinuation rates from the 3 multi-center clinical studies by adverse reaction and year.
| Year | |||
|---|---|---|---|
| 1 | 2 | 3 | |
| Number of Women at Start of Year | 1,904 | 1,463 | 1,118 |
|
Bleeding/Pain
Bleeding/Pain includes the preferred terms (PTs) Abdominal pain, Abdominal pain lower, Post procedural discomfort, Procedural pain, Coital bleeding, Dysmenorrhea, Dyspareunia, Heavy menstrual bleeding, Intermenstrual bleeding, Menometrorrhagia, Pelvic discomfort, Pelvic pain, Polymenorrhea, Uterine hemorrhage, Uterine spasm
|
8.5 | 5.1 | 3.2 |
| Expulsion | 1.9 | 1.0 | 0.9 |
|
Other Medical Event
Includes PTs embedded device (n=6) and uterine perforation (n=1)
|
0.2 | 0.1 | 0.3 |
According to the safety analyses of these three studies, it was determined the incidence of expulsion of Miudella did not vary significantly by body mass index (BMI). There was no statistically significant difference in the incidence of expulsion of Miudella in normal weight versus obese females.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Before inserting Miudella, counsel patients on the following:
5.10 Bleeding Pattern Alterations
Miudella can alter the bleeding pattern and result in heavier and longer menstrual cycles with intermenstrual spotting.
In three clinical trials with Miudella [see Adverse Reactions (6.1)] , menstrual changes were the most common medical reason for discontinuation. Discontinuation rates for pain and bleeding combined were highest in the first year of use and diminished thereafter. The percentage of females who discontinued Miudella because of bleeding problems or pain during this study ranged from 8.5% in the first year to 3.2% in Year 3. Females complaining of heavy vaginal bleeding should be evaluated and treated, and may need to discontinue Miudella [see Adverse Reactions (6.1)] .
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Miudella (copper intrauterine system) is a copper-containing sterile IUS preloaded in a single use inserter. Each single-use unit is packaged in a sealed tray with lid (NDC-82686-300-01). The IUS is comprised of a nitinol frame with 10 copper sleeves attached to provide a total copper surface area of 175mm 2, and a monofilament polymer retrieval thread tied to the vertical arm.
5.4 Risks With Intrauterine Pregnancy (5.4 Risks with Intrauterine Pregnancy)
If intrauterine pregnancy occurs with Miudella in place and the thread ends are visible or can be retrieved from the cervical canal, remove Miudella. Leaving it in place may increase the risk of spontaneous abortion and preterm labor. Removal of Miudella may also result in spontaneous abortion. In the event of an intrauterine pregnancy with Miudella, consider the following:
Principal Display Panel 175 Mm Unit Tray Box (PRINCIPAL DISPLAY PANEL - 175 mm Unit Tray Box)
NDC 82686-300-01
Miudella
intrauterine copper contraceptive
Important: To be inserted in the uterus by a trained heathcare
provider by carefully following the insertion instructions.
Rx Only
ONE (1) STERILE UNIT: INTRAUTERINE USE
Contents: One (1) sterile packaged
system containing one intrauterine
copper contraceptive with approximately
175 mm
2of copper surface area,
preloaded in a single-use disposable
inserter. A Patient Information Booklet
is provided with each unit.
Ensure the Patient Information Booklet is
provided to the patient and review
contents prior to placement.
Insert MIUDELLA before the expiration
date printed on this box.
5.1 Risk of Complications Due to Improper Insertion
Improper insertion of intrauterine systems, including Miudella, increases the risk of perforation, infection, undiagnosed abnormal bleeding, pregnancy loss (if pregnancy occurs with IUS in situ), and expulsion.
Proper training prior to first use of Miudella can minimize the risk of improper insertion. Miudella is available only through a restricted program under a REMS [see Warnings and Precautions (5.2)].
2.1 Important Dosage and Administration Instructions
- Miudella is supplied sterile in a sealed package with a sterile single use inserter for placement. Do not open the package until required for insertion. Do not use if the seal of the sterile package is broken or appears compromised.
- Before considering use of Miudella, make sure that the patient is an appropriate candidate for Miudella. Exclude pregnancy (consider the possibility of ovulation and conception) prior to use [see Contraindications (4)and Warnings and Precautions (5)].
- Only a healthcare provider should insert Miudella. Healthcare providers should be thoroughly familiar with the product, product educational materials, product insertion instructions, and prescribing information before inserting Miudella.
- The IUS is provided preloaded in the inserter (see Figure A).
- Remove Miudella on or before 3 years from the date of insertion.
- Replace Miudella at the time of removal with a new Miudella if continued contraceptive protection is desired.
FigureA : Miudella Intrauterine System (IUS) Preloaded Inserter
- Figure B: Detailed diagram of Miudella
5.11 Magnetic Resonance Imaging (mri) Safety Information (5.11 Magnetic Resonance Imaging (MRI) Safety Information)
| MRI Safety Information MR Conditional | The
Miudella Intrauterine Device (IUD)is MR Conditional. A patient with
Miudellamay be safely scanned under the following conditions.
Failure to follow these conditions may result in injury to the patient. |
| Nominal Values of Static Magnetic Field (T) | 1.5-Tesla or 3.0-Tesla |
| Maximum Spatial Field Gradient | 40 T/m (4,000 gauss/cm) |
| Type of RF Excitation | Circularly Polarized (CP) (i.e., Quadrature-Transmission) |
| Transmit RF Coil Information | There are no transmit RF coil restrictions. Accordingly, the following may be used: body transmit RF coil and all other RF coil combinations (i.e., body RF coil combined with any receive-only RF coil, transmit/receive head RF coil, transmit/receive knee RF coil, etc.) |
| Operating Mode | Normal Operating Mode |
| Maximum Whole-Body Averaged SAR | 2 W/kg (Normal Operating Mode) |
| Limits on Scan Duration | 2 W/kg whole body average SAR for 60 minutes of continuous RF exposure (a sequence or back to back sequences/series without breaks) |
| MR Image Artifact | The presence of this implant produces an imaging artifact. In testing with gradient-echo sequencing, the shape of the image artifact follows the approximate contour of the device and extends radially up to 0.7 cm from the device. |
Warning: Risk of Complications Due to Improper Insertion (WARNING: RISK OF COMPLICATIONS DUE TO IMPROPER INSERTION)
- Improper insertion of intrauterine systems, including Miudella, increases the risk of complications [see Warnings and Precautions (5.1)] .
- Proper training prior to first use of Miudella can minimize the risk of improper Miudella insertion [see Warnings and Precautions (5.1)].
- Miudella is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the Miudella REMS program to ensure healthcare providers are trained on the proper insertion of Miudella prior to first use [see Warnings and Precautions (5.2)].
13.1 Carcinogenicity, Mutagenicity, Impairment of Fertility
Adequate long-term studies in animals to assess the carcinogenic potential of a copper-containing IUS have not been performed.
A chemical characterization and toxicological risk assessment of the extractables derived from the copper-containing IUS were utilized to address carcinogenicity, mutagenicity and reproductive/developmental toxicity risks. The toxicological risk assessment indicates that the risks of carcinogenicity, mutagenicity and reproductive/developmental toxicity caused by the copper-containing IUS are negligible.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:49:59.673317 · Updated: 2026-03-14T22:35:35.043059