Diclofenac Sodium Delayed-release Tablets, Usp 25 Mg, 50 Mg And 75 Mg
9eeb2aef-3eb1-4583-8180-d480deb3651c
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Rx only
Medication Information
Recent Major Changes
Cardiovascular Thrombotic Events
-
•Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use [see Warnings and Precautions ].
-
•Diclofenac sodium delayed-release tablets is contraindicated in the setting of coronary artery bypass graft (CABG) surgery [see Error! Hyperlink reference not valid. and Warnings ].
Gastrointestinal Risk
-
•NSAIDs cause an increased risk of serious gastrointestinal adverse events including inflammation, bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients are at greater risk for serious gastrointestinal events. (See WARNINGS ).
Indications and Usage
INDICATIONS AND USAGE
Carefully consider the potential benefits and risks of Diclofenac Sodium Delayed-Release Tablets and other treatment options before deciding to use Diclofenac Sodium Delayed-Release Tablets. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS).
Diclofenac Sodium Delayed-Release Tablets, USP, are indicated:
-
•For relief of signs and symptoms of osteoarthritis
-
•For relief of signs and symptoms of rheumatoid arthritis
-
•For acute or long-term use in the relief of signs and symptoms of ankylosing spondylitis
Dosage and Administration
DOSAGE AND ADMINISTRATION
Carefully consider the potential benefits and risks of Diclofenac Sodium Delayed-Release Tablets and other treatment options before deciding to use Diclofenac Sodium Delayed-Release Tablets. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS).
After observing the response to initial therapy with Diclofenac Sodium Delayed-Release Tablets, the dose and frequency should be adjusted to suit an individual patient’s needs.
For the relief of osteoarthritis, the recommended dosage is 100-150 mg/day in divided doses (50 mg b.i.d. or t.i.d., or 75 mg b.i.d.).
For the relief of rheumatoid arthritis, the recommended dosage is 150-200 mg/day in divided doses (50 mg t.i.d. or q.i.d., or 75 mg b.i.d.).
For the relief of ankylosing spondylitis, the recommended dosage is 100-125 mg/day, administered as 25 mg q.i.d., with an extra 25-mg dose at bedtime if necessary.
Different formulations of Diclofenac (Diclofenac sodium enteric-coated tablets; Diclofenac sodium extended-release tablets, Diclofenac potassium immediate-Release tablets) are not necessarily bioequivalent even if the milligram strength is the same.
Dosage Forms and Strengths
HOW SUPPLIED
Diclofenac Sodium Delayed-Release Tablets, USP, for oral administration, are available as:
50 mg: round, Light brown, enteric-coated tablets P 50 imprinted on one side in black ink and plain on the reverse side are supplied as:
Bottles of 15.....................................NDC 63187-714-15
Bottles of 30.....................................NDC 63187-714-30
Bottles of 60.....................................NDC 63187-714-60
Bottles of 90.....................................NDC 63187-714-90
Store at 20°-25°C (68°-77°F)
(see USP Controlled Room Temperature). Protect from moisture.
Dispense in a tight, light-resistant container.
Manufactured by:
UNIQUE PHARMACEUTICAL LABORATORIES.
(A Div. of J. B. Chemicals & Pharmaceuticals Ltd.)
Mumbai 400 030, India.
Manufactured for:
Rising®
Rising Pharmaceuticals, Inc.
Allendale, NJ 07401
1-800-521-5340
Repackaged by:
Proficient Rx LP
Thousand Oaks, CA 91320
118063
July 2015
Contraindications
CONTRAINDICATIONS
Diclofenac Sodium Delayed-Release Tablets are contraindicated in patients with known hypersensitivity to Diclofenac.
Diclofenac Sodium Delayed-Release Tablets should not be given to patients who have experienced asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. Severe, rarely fatal, anaphylactic-like reactions to NSAIDs have been reported in such patients (see Error! Hyperlink reference not valid., and Error! Hyperlink reference not valid.).
Diclofenac Sodium Delayed-Release Tablets, USP are contraindicated in the setting of coronary artery bypass graft (CABG) surgery [see Warnings ].
Description
-
DESCRIPTION
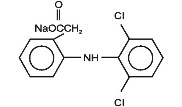
Diclofenac Sodium Delayed-Release Tablets, USP are a benzene-acetic acid derivative. Diclofenac Sodium Delayed-Release Tablets are available as delayed-release (delayed-release) tablets of 25 mg, 50 mg and 75mg for oral administration. The chemical name is 2-[(2,6-dichlorophenyl)amino] benzeneacetic acid, monosodium salt. The molecular weight is 318.14. Its molecular formula is C14H10Cl2NNaO2, and it has the following structural formula
-
The inactive ingredients in Diclofenac Sodium Delayed-Release Tablets include: lactose (monohydrate), microcrystalline cellulose, croscarmellose sodium, povidone, talc, magnesium stearate, methacrylic acid copolymer, polyethylene glycol, opadry brown (Titanium dioxide, hypromellose, polyethylene glycol, iron oxide red, iron oxide yellow) and purified water.
Section 34072-9
General
Diclofenac Sodium Delayed-Release Tablets cannot be expected to substitute for corticosteroids or to treat corticosteroid insufficiency. Abrupt discontinuation of corticosteroids may lead to disease exacerbation. Patients on prolonged corticosteroid therapy should have their therapy tapered slowly if a decision is made to discontinue corticosteroids.
The pharmacological activity of Diclofenac Sodium Delayed-Release Tablets, USP in reducing fever and inflammation may diminish the utility of these diagnostic signs in detecting complications of presumed noninfectious, painful conditions.
Section 34073-7
Drug Interactions
Aspirin :
When Diclofenac Sodium Delayed-Release Tablets are administered with aspirin, its protein binding is reduced. The clinical significance of this interaction is not known; however, as with other NSAIDs, concomitant administration of Diclofenac and aspirin is not generally recommended because of the potential of increased adverse effects.
Methotrexate : NSAIDs have been reported to competitively inhibit methotrexate accumulation in rabbit kidney slices. This may indicate that they could enhance the toxicity of methotrexate. Caution should be used when NSAIDs are administered concomitantly with methotrexate.
Cyclosporine: Diclofenac Sodium Delayed-Release Tablets, like other NSAIDs, may affect renal prostaglandins and increase the toxicity of certain drugs. Therefore, concomitant therapy with Diclofenac Sodium Delayed-Release Tablets may increase cyclosporine’s nephrotoxicity. Caution should be used when Diclofenac Sodium Delayed-Release Tablets are administered concomitantly with cyclosporine.
ACE-inhibitor: Reports suggest that NSAIDs may diminish the antihypertensive effect of ACE inhibitors. This interaction should be given consideration in patients taking NSAIDs concomitantly with ACE inhibitors.
Furosemide: Clinical studies, as well as post-marketing observations, have shown that Diclofenac Sodium Delayed-Release Tablets can reduce the natriuretic effect of furosemide and thiazides in some patients. This response has been attributed to inhibition of renal prostaglandin synthesis. During concomitant therapy with NSAIDs, the patient should be observed closely for signs of renal failure (see Error! Hyperlink reference not valid.), as well as to assure diuretic efficacy.
Lithium: NSAIDs have produced an elevation of plasma lithium levels and a reduction in renal lithium clearance. The mean minimum lithium concentration increased 15% and the renal clearance was decreased by approximately 20%. These effects have been attributed to inhibition of renal prostaglandin synthesis by the NSAID. Thus, when NSAIDs and lithium are administered concurrently, subjects should be observed carefully for signs of lithium toxicity.
Warfarin: The effects of warfarin and NSAIDs on GI bleeding are synergistic, such that users of both drugs together have a risk of serious GI bleeding higher than users of either drug alone.
CYP2C9 Inhibitors or Inducers: Diclofenac is metabolized by cytochrome P450 enzymes, predominantly by CYP2C9. Co-administration of Diclofenac with CYP2C9 inhibitors (e.g. voriconazole) may enhance the exposure and toxicity of Diclofenac whereas co-administration with CYP2C9 inducers (e.g. rifampin) may lead to compromised efficacy of Diclofenac. Use caution when dosing Diclofenac with CYP2C9 inhibitors or inducers; a dosage adjustment may be warranted (see Error! Hyperlink reference not valid.).
Section 34075-2
Laboratory Tests
Because serious GI tract ulcerations and bleeding can occur without warning symptoms, physicians should monitor for signs or symptoms of GI bleeding. In patients on long-term treatment with NSAIDs, including Diclofenac Sodium Delayed-Release Tablets, the CBC and a chemistry profile (including transaminase levels) should be checked periodically. If clinical signs and symptoms consistent with liver or renal disease develop, systemic manifestations occur (e.g., eosinophilia, rash, etc.) or if abnormal liver tests persist or worsen, Diclofenac Sodium Delayed-Release Tablets should be discontinued.
Section 34076-0
Information for Patients
Patients should be informed of the following information before initiating therapy with an NSAID and periodically during the course of ongoing therapy. Patients should also be encouraged to read the NSAID Medication Guide that accompanies each prescription dispensed.
-
1.Cardiovascular Thrombotic Events: Advise patients to be alert for the symptoms of cardiovascular thrombotic events, including chest pain, shortness of breath, weakness, or slurring of speech, and to report any of these symptoms to their health care provider immediately [see Warnings ].
-
2.Diclofenac Sodium Delayed-Release Tablets, like other NSAIDs, can cause GI discomfort and, rarely, more serious GI side effects, such as ulcers and bleeding, which may result in hospitalization and even death. Although serious GI tract ulcerations and bleeding can occur without warning symptoms, patients should be alert for the signs and symptoms of ulcerations and bleeding, and should ask for medical advice when observing any indicative sign or symptoms including epigastric pain, dyspepsia, melena, and hematemesis. Patients should be apprised of the importance of this follow-up (see Error! Hyperlink reference not valid.).
-
3.Diclofenac Sodium Delayed-Release Tablets, like other NSAIDs, can cause serious skin side effects such as exfoliative dermatitis, SJS, and TEN, which may result in hospitalizations and even death. Although serious skin reactions may occur without warning, patients should be alert for the signs and symptoms of skin rash and blisters, fever, or other signs of hypersensitivity such as itching, and should ask for medical advice when observing any indicative signs or symptoms. Patients should be advised to stop the drug immediately if they develop any type of rash and contact their physicians as soon as possible.
-
4.Heart Failure And Edema: Advise patients to be alert for the symptoms of congestive heart failure including shortness of breath, unexplained weight gain, or edema and to contact their healthcare provider if such symptoms occur [see Warnings].
-
5.Patients should be informed of the warning signs and symptoms of hepatotoxicity (e.g., nausea, fatigue, lethargy, pruritus, jaundice, right upper quadrant tenderness, and “flu-like” symptoms). If these occur, patients should be instructed to stop therapy and seek immediate medical therapy. (SeeError! Hyperlink reference not valid.)
-
6.Patients should be informed of the signs of an anaphylactic reaction (e.g., difficulty breathing, swelling of the face or throat). If these occur, patients should be instructed to seek immediate emergency help (see Error! Hyperlink reference not valid.).
-
7.In late pregnancy, as with other NSAIDs, Diclofenac Sodium Delayed-Release Tablets should be avoided because it may cause premature closure of the ductus arteriosus.
Section 34077-8
Pregnancy
Teratogenic Effects:
Pregnancy Category C
Reproductive studies conducted in rats and rabbits have not demonstrated evidence of developmental abnormalities. However, animal reproduction studies are not always predictive of human response. There are no adequate and well-controlled studies in pregnant women.
Section 34078-6
Nonteratogenic Effects: Because of the known effects of nonsteroidal anti-inflammatory drugs on the fetal cardiovascular system (closure of ductus arteriosus), use during pregnancy (particularly late pregnancy) should be avoided.
Section 34079-4
Labor and Delivery
In rat studies with NSAIDs, as with other drugs known to inhibit prostaglandin synthesis, an increased incidence of dystocia, delayed parturition, and decreased pup survival occurred. The effects of Diclofenac Sodium Delayed-Release Tablets on labor and delivery in pregnant women are unknown.
Section 34080-2
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from Diclofenac Sodium Delayed-Release Tablets, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Section 34081-0
Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
Section 34082-8
Geriatric Use
As with any NSAIDs, caution should be exercised in treating the elderly (65 years and older).
Section 34084-4
ADVERSE REACTIONS
In patients taking Diclofenac Sodium Delayed-Release Tablets, or other NSAIDs, the most frequently reported adverse experiences occurring in approximately 1%-10% of patients are:
Gastrointestinal experiences including: abdominal pain, constipation, diarrhea, dyspepsia, flatulence, gross bleeding/perforation, heartburn, nausea, GI ulcers (gastric/duodenal) and vomiting.
Abnormal renal function, anemia, dizziness, edema, elevated liver enzymes, headaches, increased bleeding time, pruritus, rashes and tinnitus.
Additional adverse experiences reported occasionally include:
-
Body as a Whole: fever, infection, sepsis
-
Cardiovascular System: congestive heart failure, hypertension, tachycardia, syncope
-
Digestive System: dry mouth, esophagitis, gastric/peptic ulcers, gastritis, gastrointestinal bleeding, glossitis, hematemesis, hepatitis, jaundice
-
Hemic and Lymphatic System: ecchymosis, eosinophilia, leukopenia, melena, purpura, rectal bleeding, stomatitis, thrombocytopenia
-
Metabolic and Nutritional: weight changes
-
Nervous System: anxiety, asthenia, confusion, depression, dream abnormalities, drowsiness, insomnia, malaise, nervousness, paresthesia, somnolence, tremors, vertigo
-
Respiratory System: asthma, dyspnea
-
Skin and Appendages: alopecia, photosensitivity, sweating increased
-
Special Senses: blurred vision
-
Urogenital System: cystitis, dysuria, hematuria, interstitial nephritis, oliguria/polyuria, proteinuria, renal failure
Other adverse reactions, which occur rarely are:
-
Body as a Whole: anaphylactic reactions, appetite changes, death
-
Cardiovascular System: arrhythmia, hypotension, myocardial infarction, palpitations, vasculitis
Digestive System: colitis, eructation, fulminant hepatitis with and without jaundice, liver failure, liver necrosis, pancreatitis
-
Hemic and Lymphatic System: agranulocytosis, hemolytic anemia, aplastic anemia, lymphadenopathy, pancytopenia
-
Metabolic and Nutritional: hyperglycemia
-
Nervous System: convulsions, coma, hallucinations, meningitis
-
Respiratory System: respiratory depression, pneumonia
-
Skin and Appendages: angioedema, toxic epidermal necrolysis, erythema multiforme, exfoliative dermatitis, Stevens-Johnson syndrome, urticaria
Special Senses: conjunctivitis, hearing impairment
To report SUSPECTED ADVERSE REACTIONS, contact Rising Pharmaceuticals, Inc. at 1-866-562-4597 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Section 34088-5
OVERDOSAGE
Symptoms following acute NSAID overdoses are usually limited to lethargy, drowsiness, nausea, vomiting, and epigastric pain, which are generally reversible with supportive care. Gastrointestinal bleeding can occur. Hypertension, acute renal failure, respiratory depression and coma may occur, but are rare. Anaphylactoid reactions have been reported with therapeutic ingestion of NSAIDs, and may occur following an overdose.
Patients should be managed by symptomatic and supportive care following a NSAID overdose. There are no specific antidotes. Emesis and/or activated charcoal (60 to 100 g in adults, 1 to 2 g/kg in children) and/or osmotic cathartic may be indicated in patients seen within 4 hours of ingestion with symptoms or following a large overdose (5 to 10 times the usual dose). Forced diuresis, alkalinization of urine, hemodialysis, or hemoperfusion may not be useful due to high protein binding.
Section 34090-1
CLINICAL PHARMACOLOGY
Section 42229-5
Rx only
Section 42231-1
Diclofenac Sodium Delayed-Release Tablets, USP
MEDICATION GUIDE for Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)
Section 51945-4
———PRINCIPAL DISPLAY PANEL - 50 mg———
NDC 63187-714-60
Diclofenac Sodium
Delayed-Release
Tablets, USP
50 mg
60 Tablets Rx only
Cardiovascular Thrombotic Events
Clinical trials of several COX-2 selective and nonselective NSAIDs of up to three years duration have shown an increased risk of serious cardiovascular (CV) thrombotic events, including myocardial infarction (MI) and stroke, which can be fatal. Based on available data, it is unclear that the risk for CV thrombotic events is similar for all NSAIDs. The relative increase in serious CV thrombotic events over baseline conferred by NSAID use appears to be similar in those with and without known CV disease or risk factors for CV disease. However, patients with known CV disease or risk factors had a higher absolute incidence of excess serious CV thrombotic events, due to their increased baseline rate. Some observational studies found that this increased risk of serious CV thrombotic events began as early as the first weeks of treatment. The increase in CV thrombotic risk has been observed most consistently at higher doses.
To minimize the potential risk for an adverse CV event in NSAID-treated patients, use the lowest effective dose for the shortest duration possible. Physicians and patients should remain alert for the development of such events, throughout the entire treatment course, even in the absence of previous CV symptoms. Patients should be informed about the symptoms of serious CV events and the steps to take if they occur.
There is no consistent evidence that concurrent use of aspirin mitigates the increased risk of serious CV thrombotic events associated with NSAID use. The concurrent use of aspirin and an NSAID, such as Diclofenac Sodium, increases the risk of serious gastrointestinal (GI) events [see Warnings ].
Status Post Coronary Artery Bypass Graft (CABG) Surgery
Two large, controlled clinical trials of a COX-2 selective NSAID for the treatment of pain in the first 10–14 days following CABG surgery found an increased incidence of myocardial infarction and stroke. NSAIDs are contraindicated in the setting of CABG [see
Error! Hyperlink reference not valid.
].
Post-MI Patients
Observational studies conducted in the Danish National Registry have demonstrated that patients treated with NSAIDs in the post-MI period were at increased risk of reinfarction, CV-related death, and all-cause mortality beginning in the first week of treatment. In this same cohort, the incidence of death in the first year post MI was 20 per 100 person years in NSAID-treated patients compared to 12 per 100 person years in non-NSAID exposed patients. Although the absolute rate of death declined somewhat after the first year post-MI, the increased relative risk of death in NSAID users persisted over at least the next four years of follow-up.
Avoid the use of Diclofenac Sodium Delayed-Release Tablets in patients with a recent MI unless the benefits are expected to outweigh the risk of recurrent CV thrombotic events. If Diclofenac Sodium Delayed-Release Tablets is used in patients with a recent MI, monitor patients for signs of cardiac ischemia.
Structured Label Content
Recent Major Changes (34066-1)
Cardiovascular Thrombotic Events
-
•Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use [see Warnings and Precautions ].
-
•Diclofenac sodium delayed-release tablets is contraindicated in the setting of coronary artery bypass graft (CABG) surgery [see Error! Hyperlink reference not valid. and Warnings ].
Gastrointestinal Risk
-
•NSAIDs cause an increased risk of serious gastrointestinal adverse events including inflammation, bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients are at greater risk for serious gastrointestinal events. (See WARNINGS ).
Indications and Usage (34067-9)
INDICATIONS AND USAGE
Carefully consider the potential benefits and risks of Diclofenac Sodium Delayed-Release Tablets and other treatment options before deciding to use Diclofenac Sodium Delayed-Release Tablets. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS).
Diclofenac Sodium Delayed-Release Tablets, USP, are indicated:
-
•For relief of signs and symptoms of osteoarthritis
-
•For relief of signs and symptoms of rheumatoid arthritis
-
•For acute or long-term use in the relief of signs and symptoms of ankylosing spondylitis
Dosage and Administration (34068-7)
DOSAGE AND ADMINISTRATION
Carefully consider the potential benefits and risks of Diclofenac Sodium Delayed-Release Tablets and other treatment options before deciding to use Diclofenac Sodium Delayed-Release Tablets. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS).
After observing the response to initial therapy with Diclofenac Sodium Delayed-Release Tablets, the dose and frequency should be adjusted to suit an individual patient’s needs.
For the relief of osteoarthritis, the recommended dosage is 100-150 mg/day in divided doses (50 mg b.i.d. or t.i.d., or 75 mg b.i.d.).
For the relief of rheumatoid arthritis, the recommended dosage is 150-200 mg/day in divided doses (50 mg t.i.d. or q.i.d., or 75 mg b.i.d.).
For the relief of ankylosing spondylitis, the recommended dosage is 100-125 mg/day, administered as 25 mg q.i.d., with an extra 25-mg dose at bedtime if necessary.
Different formulations of Diclofenac (Diclofenac sodium enteric-coated tablets; Diclofenac sodium extended-release tablets, Diclofenac potassium immediate-Release tablets) are not necessarily bioequivalent even if the milligram strength is the same.
Dosage Forms and Strengths (34069-5)
HOW SUPPLIED
Diclofenac Sodium Delayed-Release Tablets, USP, for oral administration, are available as:
50 mg: round, Light brown, enteric-coated tablets P 50 imprinted on one side in black ink and plain on the reverse side are supplied as:
Bottles of 15.....................................NDC 63187-714-15
Bottles of 30.....................................NDC 63187-714-30
Bottles of 60.....................................NDC 63187-714-60
Bottles of 90.....................................NDC 63187-714-90
Store at 20°-25°C (68°-77°F)
(see USP Controlled Room Temperature). Protect from moisture.
Dispense in a tight, light-resistant container.
Manufactured by:
UNIQUE PHARMACEUTICAL LABORATORIES.
(A Div. of J. B. Chemicals & Pharmaceuticals Ltd.)
Mumbai 400 030, India.
Manufactured for:
Rising®
Rising Pharmaceuticals, Inc.
Allendale, NJ 07401
1-800-521-5340
Repackaged by:
Proficient Rx LP
Thousand Oaks, CA 91320
118063
July 2015
Contraindications (34070-3)
CONTRAINDICATIONS
Diclofenac Sodium Delayed-Release Tablets are contraindicated in patients with known hypersensitivity to Diclofenac.
Diclofenac Sodium Delayed-Release Tablets should not be given to patients who have experienced asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. Severe, rarely fatal, anaphylactic-like reactions to NSAIDs have been reported in such patients (see Error! Hyperlink reference not valid., and Error! Hyperlink reference not valid.).
Diclofenac Sodium Delayed-Release Tablets, USP are contraindicated in the setting of coronary artery bypass graft (CABG) surgery [see Warnings ].
Section 34072-9 (34072-9)
General
Diclofenac Sodium Delayed-Release Tablets cannot be expected to substitute for corticosteroids or to treat corticosteroid insufficiency. Abrupt discontinuation of corticosteroids may lead to disease exacerbation. Patients on prolonged corticosteroid therapy should have their therapy tapered slowly if a decision is made to discontinue corticosteroids.
The pharmacological activity of Diclofenac Sodium Delayed-Release Tablets, USP in reducing fever and inflammation may diminish the utility of these diagnostic signs in detecting complications of presumed noninfectious, painful conditions.
Section 34073-7 (34073-7)
Drug Interactions
Aspirin :
When Diclofenac Sodium Delayed-Release Tablets are administered with aspirin, its protein binding is reduced. The clinical significance of this interaction is not known; however, as with other NSAIDs, concomitant administration of Diclofenac and aspirin is not generally recommended because of the potential of increased adverse effects.
Methotrexate : NSAIDs have been reported to competitively inhibit methotrexate accumulation in rabbit kidney slices. This may indicate that they could enhance the toxicity of methotrexate. Caution should be used when NSAIDs are administered concomitantly with methotrexate.
Cyclosporine: Diclofenac Sodium Delayed-Release Tablets, like other NSAIDs, may affect renal prostaglandins and increase the toxicity of certain drugs. Therefore, concomitant therapy with Diclofenac Sodium Delayed-Release Tablets may increase cyclosporine’s nephrotoxicity. Caution should be used when Diclofenac Sodium Delayed-Release Tablets are administered concomitantly with cyclosporine.
ACE-inhibitor: Reports suggest that NSAIDs may diminish the antihypertensive effect of ACE inhibitors. This interaction should be given consideration in patients taking NSAIDs concomitantly with ACE inhibitors.
Furosemide: Clinical studies, as well as post-marketing observations, have shown that Diclofenac Sodium Delayed-Release Tablets can reduce the natriuretic effect of furosemide and thiazides in some patients. This response has been attributed to inhibition of renal prostaglandin synthesis. During concomitant therapy with NSAIDs, the patient should be observed closely for signs of renal failure (see Error! Hyperlink reference not valid.), as well as to assure diuretic efficacy.
Lithium: NSAIDs have produced an elevation of plasma lithium levels and a reduction in renal lithium clearance. The mean minimum lithium concentration increased 15% and the renal clearance was decreased by approximately 20%. These effects have been attributed to inhibition of renal prostaglandin synthesis by the NSAID. Thus, when NSAIDs and lithium are administered concurrently, subjects should be observed carefully for signs of lithium toxicity.
Warfarin: The effects of warfarin and NSAIDs on GI bleeding are synergistic, such that users of both drugs together have a risk of serious GI bleeding higher than users of either drug alone.
CYP2C9 Inhibitors or Inducers: Diclofenac is metabolized by cytochrome P450 enzymes, predominantly by CYP2C9. Co-administration of Diclofenac with CYP2C9 inhibitors (e.g. voriconazole) may enhance the exposure and toxicity of Diclofenac whereas co-administration with CYP2C9 inducers (e.g. rifampin) may lead to compromised efficacy of Diclofenac. Use caution when dosing Diclofenac with CYP2C9 inhibitors or inducers; a dosage adjustment may be warranted (see Error! Hyperlink reference not valid.).
Section 34075-2 (34075-2)
Laboratory Tests
Because serious GI tract ulcerations and bleeding can occur without warning symptoms, physicians should monitor for signs or symptoms of GI bleeding. In patients on long-term treatment with NSAIDs, including Diclofenac Sodium Delayed-Release Tablets, the CBC and a chemistry profile (including transaminase levels) should be checked periodically. If clinical signs and symptoms consistent with liver or renal disease develop, systemic manifestations occur (e.g., eosinophilia, rash, etc.) or if abnormal liver tests persist or worsen, Diclofenac Sodium Delayed-Release Tablets should be discontinued.
Section 34076-0 (34076-0)
Information for Patients
Patients should be informed of the following information before initiating therapy with an NSAID and periodically during the course of ongoing therapy. Patients should also be encouraged to read the NSAID Medication Guide that accompanies each prescription dispensed.
-
1.Cardiovascular Thrombotic Events: Advise patients to be alert for the symptoms of cardiovascular thrombotic events, including chest pain, shortness of breath, weakness, or slurring of speech, and to report any of these symptoms to their health care provider immediately [see Warnings ].
-
2.Diclofenac Sodium Delayed-Release Tablets, like other NSAIDs, can cause GI discomfort and, rarely, more serious GI side effects, such as ulcers and bleeding, which may result in hospitalization and even death. Although serious GI tract ulcerations and bleeding can occur without warning symptoms, patients should be alert for the signs and symptoms of ulcerations and bleeding, and should ask for medical advice when observing any indicative sign or symptoms including epigastric pain, dyspepsia, melena, and hematemesis. Patients should be apprised of the importance of this follow-up (see Error! Hyperlink reference not valid.).
-
3.Diclofenac Sodium Delayed-Release Tablets, like other NSAIDs, can cause serious skin side effects such as exfoliative dermatitis, SJS, and TEN, which may result in hospitalizations and even death. Although serious skin reactions may occur without warning, patients should be alert for the signs and symptoms of skin rash and blisters, fever, or other signs of hypersensitivity such as itching, and should ask for medical advice when observing any indicative signs or symptoms. Patients should be advised to stop the drug immediately if they develop any type of rash and contact their physicians as soon as possible.
-
4.Heart Failure And Edema: Advise patients to be alert for the symptoms of congestive heart failure including shortness of breath, unexplained weight gain, or edema and to contact their healthcare provider if such symptoms occur [see Warnings].
-
5.Patients should be informed of the warning signs and symptoms of hepatotoxicity (e.g., nausea, fatigue, lethargy, pruritus, jaundice, right upper quadrant tenderness, and “flu-like” symptoms). If these occur, patients should be instructed to stop therapy and seek immediate medical therapy. (SeeError! Hyperlink reference not valid.)
-
6.Patients should be informed of the signs of an anaphylactic reaction (e.g., difficulty breathing, swelling of the face or throat). If these occur, patients should be instructed to seek immediate emergency help (see Error! Hyperlink reference not valid.).
-
7.In late pregnancy, as with other NSAIDs, Diclofenac Sodium Delayed-Release Tablets should be avoided because it may cause premature closure of the ductus arteriosus.
Section 34077-8 (34077-8)
Pregnancy
Teratogenic Effects:
Pregnancy Category C
Reproductive studies conducted in rats and rabbits have not demonstrated evidence of developmental abnormalities. However, animal reproduction studies are not always predictive of human response. There are no adequate and well-controlled studies in pregnant women.
Section 34078-6 (34078-6)
Nonteratogenic Effects: Because of the known effects of nonsteroidal anti-inflammatory drugs on the fetal cardiovascular system (closure of ductus arteriosus), use during pregnancy (particularly late pregnancy) should be avoided.
Section 34079-4 (34079-4)
Labor and Delivery
In rat studies with NSAIDs, as with other drugs known to inhibit prostaglandin synthesis, an increased incidence of dystocia, delayed parturition, and decreased pup survival occurred. The effects of Diclofenac Sodium Delayed-Release Tablets on labor and delivery in pregnant women are unknown.
Section 34080-2 (34080-2)
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from Diclofenac Sodium Delayed-Release Tablets, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Section 34081-0 (34081-0)
Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
Section 34082-8 (34082-8)
Geriatric Use
As with any NSAIDs, caution should be exercised in treating the elderly (65 years and older).
Section 34084-4 (34084-4)
ADVERSE REACTIONS
In patients taking Diclofenac Sodium Delayed-Release Tablets, or other NSAIDs, the most frequently reported adverse experiences occurring in approximately 1%-10% of patients are:
Gastrointestinal experiences including: abdominal pain, constipation, diarrhea, dyspepsia, flatulence, gross bleeding/perforation, heartburn, nausea, GI ulcers (gastric/duodenal) and vomiting.
Abnormal renal function, anemia, dizziness, edema, elevated liver enzymes, headaches, increased bleeding time, pruritus, rashes and tinnitus.
Additional adverse experiences reported occasionally include:
-
Body as a Whole: fever, infection, sepsis
-
Cardiovascular System: congestive heart failure, hypertension, tachycardia, syncope
-
Digestive System: dry mouth, esophagitis, gastric/peptic ulcers, gastritis, gastrointestinal bleeding, glossitis, hematemesis, hepatitis, jaundice
-
Hemic and Lymphatic System: ecchymosis, eosinophilia, leukopenia, melena, purpura, rectal bleeding, stomatitis, thrombocytopenia
-
Metabolic and Nutritional: weight changes
-
Nervous System: anxiety, asthenia, confusion, depression, dream abnormalities, drowsiness, insomnia, malaise, nervousness, paresthesia, somnolence, tremors, vertigo
-
Respiratory System: asthma, dyspnea
-
Skin and Appendages: alopecia, photosensitivity, sweating increased
-
Special Senses: blurred vision
-
Urogenital System: cystitis, dysuria, hematuria, interstitial nephritis, oliguria/polyuria, proteinuria, renal failure
Other adverse reactions, which occur rarely are:
-
Body as a Whole: anaphylactic reactions, appetite changes, death
-
Cardiovascular System: arrhythmia, hypotension, myocardial infarction, palpitations, vasculitis
Digestive System: colitis, eructation, fulminant hepatitis with and without jaundice, liver failure, liver necrosis, pancreatitis
-
Hemic and Lymphatic System: agranulocytosis, hemolytic anemia, aplastic anemia, lymphadenopathy, pancytopenia
-
Metabolic and Nutritional: hyperglycemia
-
Nervous System: convulsions, coma, hallucinations, meningitis
-
Respiratory System: respiratory depression, pneumonia
-
Skin and Appendages: angioedema, toxic epidermal necrolysis, erythema multiforme, exfoliative dermatitis, Stevens-Johnson syndrome, urticaria
Special Senses: conjunctivitis, hearing impairment
To report SUSPECTED ADVERSE REACTIONS, contact Rising Pharmaceuticals, Inc. at 1-866-562-4597 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Section 34088-5 (34088-5)
OVERDOSAGE
Symptoms following acute NSAID overdoses are usually limited to lethargy, drowsiness, nausea, vomiting, and epigastric pain, which are generally reversible with supportive care. Gastrointestinal bleeding can occur. Hypertension, acute renal failure, respiratory depression and coma may occur, but are rare. Anaphylactoid reactions have been reported with therapeutic ingestion of NSAIDs, and may occur following an overdose.
Patients should be managed by symptomatic and supportive care following a NSAID overdose. There are no specific antidotes. Emesis and/or activated charcoal (60 to 100 g in adults, 1 to 2 g/kg in children) and/or osmotic cathartic may be indicated in patients seen within 4 hours of ingestion with symptoms or following a large overdose (5 to 10 times the usual dose). Forced diuresis, alkalinization of urine, hemodialysis, or hemoperfusion may not be useful due to high protein binding.
Description (34089-3)
-
DESCRIPTION
Diclofenac Sodium Delayed-Release Tablets, USP are a benzene-acetic acid derivative. Diclofenac Sodium Delayed-Release Tablets are available as delayed-release (delayed-release) tablets of 25 mg, 50 mg and 75mg for oral administration. The chemical name is 2-[(2,6-dichlorophenyl)amino] benzeneacetic acid, monosodium salt. The molecular weight is 318.14. Its molecular formula is C14H10Cl2NNaO2, and it has the following structural formula
-
The inactive ingredients in Diclofenac Sodium Delayed-Release Tablets include: lactose (monohydrate), microcrystalline cellulose, croscarmellose sodium, povidone, talc, magnesium stearate, methacrylic acid copolymer, polyethylene glycol, opadry brown (Titanium dioxide, hypromellose, polyethylene glycol, iron oxide red, iron oxide yellow) and purified water.
Section 34090-1 (34090-1)
CLINICAL PHARMACOLOGY
Section 42229-5 (42229-5)
Rx only
Section 42231-1 (42231-1)
Diclofenac Sodium Delayed-Release Tablets, USP
MEDICATION GUIDE for Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)
Section 51945-4 (51945-4)
———PRINCIPAL DISPLAY PANEL - 50 mg———
NDC 63187-714-60
Diclofenac Sodium
Delayed-Release
Tablets, USP
50 mg
60 Tablets Rx only
Cardiovascular Thrombotic Events
Clinical trials of several COX-2 selective and nonselective NSAIDs of up to three years duration have shown an increased risk of serious cardiovascular (CV) thrombotic events, including myocardial infarction (MI) and stroke, which can be fatal. Based on available data, it is unclear that the risk for CV thrombotic events is similar for all NSAIDs. The relative increase in serious CV thrombotic events over baseline conferred by NSAID use appears to be similar in those with and without known CV disease or risk factors for CV disease. However, patients with known CV disease or risk factors had a higher absolute incidence of excess serious CV thrombotic events, due to their increased baseline rate. Some observational studies found that this increased risk of serious CV thrombotic events began as early as the first weeks of treatment. The increase in CV thrombotic risk has been observed most consistently at higher doses.
To minimize the potential risk for an adverse CV event in NSAID-treated patients, use the lowest effective dose for the shortest duration possible. Physicians and patients should remain alert for the development of such events, throughout the entire treatment course, even in the absence of previous CV symptoms. Patients should be informed about the symptoms of serious CV events and the steps to take if they occur.
There is no consistent evidence that concurrent use of aspirin mitigates the increased risk of serious CV thrombotic events associated with NSAID use. The concurrent use of aspirin and an NSAID, such as Diclofenac Sodium, increases the risk of serious gastrointestinal (GI) events [see Warnings ].
Status Post Coronary Artery Bypass Graft (CABG) Surgery
Two large, controlled clinical trials of a COX-2 selective NSAID for the treatment of pain in the first 10–14 days following CABG surgery found an increased incidence of myocardial infarction and stroke. NSAIDs are contraindicated in the setting of CABG [see
Error! Hyperlink reference not valid.
].
Post-MI Patients
Observational studies conducted in the Danish National Registry have demonstrated that patients treated with NSAIDs in the post-MI period were at increased risk of reinfarction, CV-related death, and all-cause mortality beginning in the first week of treatment. In this same cohort, the incidence of death in the first year post MI was 20 per 100 person years in NSAID-treated patients compared to 12 per 100 person years in non-NSAID exposed patients. Although the absolute rate of death declined somewhat after the first year post-MI, the increased relative risk of death in NSAID users persisted over at least the next four years of follow-up.
Avoid the use of Diclofenac Sodium Delayed-Release Tablets in patients with a recent MI unless the benefits are expected to outweigh the risk of recurrent CV thrombotic events. If Diclofenac Sodium Delayed-Release Tablets is used in patients with a recent MI, monitor patients for signs of cardiac ischemia.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:36:53.412382 · Updated: 2026-03-14T21:48:00.844863