goniotaire
9ebedd36-523a-4f92-8fe9-33beaf7288f9
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Altaire Goniotaire Hypromellose 2.5% Ophthalmic Demulcent Solution 15mL NDC 59390- 182-13 Drug Facts Each m L Contains:
Medication Information
Description
Altaire Goniotaire Hypromellose 2.5% Ophthalmic Demulcent Solution 15mL NDC 59390- 182-13 Drug Facts Each m L Contains:
Principal Display Panel
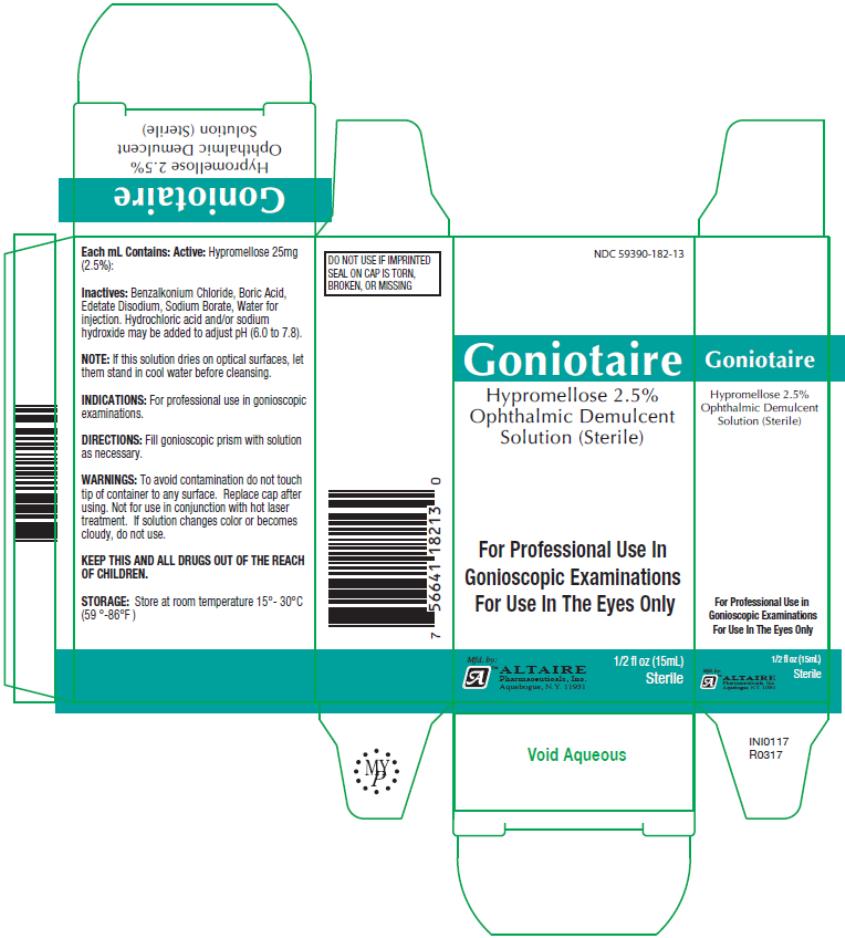
NDC 59390-182-13
Goniotaire
Hypromellose 2.5%
Ophthalmic Demulcent
Solution (Sterile)
½ fl oz (15mL)
sterile
Active
Hypromellose 25mg (2.5%):
Section 42229-5
Altaire
Goniotaire
Hypromellose 2.5% Ophthalmic
Demulcent Solution
15mL
NDC 59390- 182-13
Drug Facts
Each m L Contains:
Storage:
Store at room temperature 15°- 30°C (59°- 86°F).
Warnings:
To avoid contamination do not touch tip of container to any surface. Replace cap after using. Not for use in conjunction with hot laser treatment. If solution changes color or becomes cloudy, do not use.
Inactives:
Benzalkonium Chloride, Boric Acid, Edetate Disodium, Sodium Borate, Water for injection. Hydrochloric Acid and/or sodium hydroxide may be added to adjust pH (6.0 to 7.8).
NOTE: If this solution dries on optical surfaces, let them stand in cool water before cleansing.
Directions:
Fill gonioscopic prism with solution as necessary.
Indications:
For professional use in gonioscopic examinations.
Structured Label Content
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
NDC 59390-182-13
Goniotaire
Hypromellose 2.5%
Ophthalmic Demulcent
Solution (Sterile)
½ fl oz (15mL)
sterile
Active
Hypromellose 25mg (2.5%):
Section 42229-5 (42229-5)
Altaire
Goniotaire
Hypromellose 2.5% Ophthalmic
Demulcent Solution
15mL
NDC 59390- 182-13
Drug Facts
Each m L Contains:
Storage: (STORAGE:)
Store at room temperature 15°- 30°C (59°- 86°F).
Warnings: (WARNINGS:)
To avoid contamination do not touch tip of container to any surface. Replace cap after using. Not for use in conjunction with hot laser treatment. If solution changes color or becomes cloudy, do not use.
Inactives:
Benzalkonium Chloride, Boric Acid, Edetate Disodium, Sodium Borate, Water for injection. Hydrochloric Acid and/or sodium hydroxide may be added to adjust pH (6.0 to 7.8).
NOTE: If this solution dries on optical surfaces, let them stand in cool water before cleansing.
Directions: (DIRECTIONS:)
Fill gonioscopic prism with solution as necessary.
Indications: (INDICATIONS:)
For professional use in gonioscopic examinations.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:37:24.112855 · Updated: 2026-03-14T21:49:59.697322