Fever 1527l
9d762268-8c30-8a86-e053-2a95a90a419c
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
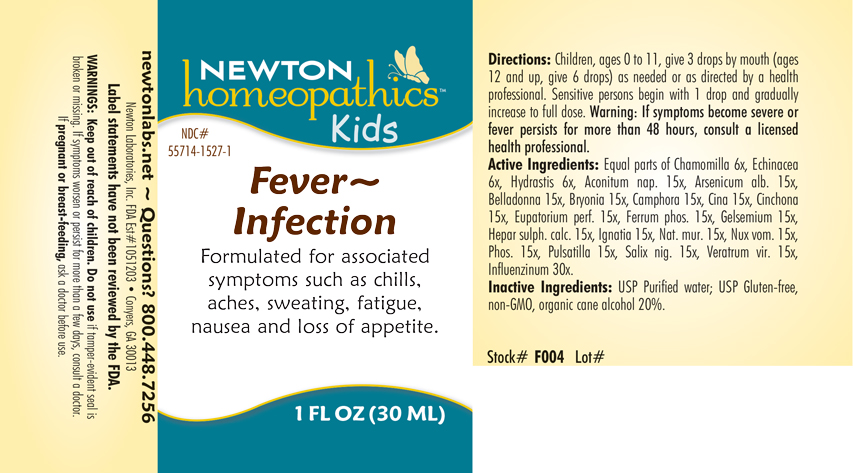
Formuated for associated symptoms such as chills, aches, sweating, fatigue, nausea and loss of appetite.
Indications and Usage
Formuated for associated symptoms such as chills, aches, sweating, fatigue, nausea and loss of appetite.
Purpose
Formulated for associated symptoms such as chills, aches, sweating, fatigue, nausea and loss of appetite.
Medication Information
Indications and Usage
Formuated for associated symptoms such as chills, aches, sweating, fatigue, nausea and loss of appetite.
Purpose
Formulated for associated symptoms such as chills, aches, sweating, fatigue, nausea and loss of appetite.
Description
Formuated for associated symptoms such as chills, aches, sweating, fatigue, nausea and loss of appetite.
Package Label
Warnings Section
WARNINGS: Keep out of reach of children. Do not use if tamper-evident seal is broken or missing. If symptoms worsen or persist for more than a few days, consult a doctor. If pregnant or breast-feeding, ask a doctor before use.
Questions Section
newtonlabs.net – Questions? 800.448.7256
Newton Laboratories, Inc. FDA Est # 1051203 - Conyers, GA 30013
Otc Purpose Section
Formulated for associated symptoms such as chills, aches, sweating, fatigue, nausea and loss of appetite.
Inactive Ingredient Section
USP Purified water; USP Gluten-free, non-GMO, organic cane alcohol 20%
Indications and Usage Section
Formuated for associated symptoms such as chills, aches, sweating, fatigue, nausea and loss of appetite.
Dosage & Administration Section
Directions: Children, ages 0 to 11, give 3 drops by mouth (ages 12 and up, give 6 drops) as needed or a directed by a health professional. Sensitive person begin with 1 drop and gradually increase to full dose.
Warning: If symptoms become severe or fever persists for more than 48 hous, consult a licensed health professional.
Otc Active Ingredient Section
Equal parts of Chamomilla 6x, Echinacea 6x, Hydrastis canadensis 6x, Aconitum napellus 15x, Arsenicum album 15x, Belladonna 15x, Bryonia 15x, Camphora 15x, Cina 15x, Cinchona officinalis 15x, Eupatorium perfoliatum 15x, Ferrum phosphoricum 15x, Gelsemium sempervirens 15x, Hepar sulphuris calcareum 15x, Ignatia amara 15x, Natrum muriaticum 15x, Nux vomica 15x, Phosphorus 15x, Pulsatilla 15x, Salix nigra 15x, Veratrum viride 15x, Influenzinum 30x.
Otc Pregnancy Or Breast Feeding Section
If pregnant or breast-feeding, ask a doctor before use.
Otc Keep Out of Reach of Children Section
WARNINGS: Keep out of reach of children. Do not use if tamper-evident seal is broken or missing. If symptoms worsen or persist for more than a few days, consult a doctor. If pregnant or breast-feeding, ask a doctor before use.
Structured Label Content
Package Label (PACKAGE LABEL)
Warnings Section (WARNINGS SECTION)
WARNINGS: Keep out of reach of children. Do not use if tamper-evident seal is broken or missing. If symptoms worsen or persist for more than a few days, consult a doctor. If pregnant or breast-feeding, ask a doctor before use.
Questions Section (QUESTIONS SECTION)
newtonlabs.net – Questions? 800.448.7256
Newton Laboratories, Inc. FDA Est # 1051203 - Conyers, GA 30013
Otc Purpose Section (OTC - PURPOSE SECTION)
Formulated for associated symptoms such as chills, aches, sweating, fatigue, nausea and loss of appetite.
Inactive Ingredient Section (INACTIVE INGREDIENT SECTION)
USP Purified water; USP Gluten-free, non-GMO, organic cane alcohol 20%
Indications and Usage Section (INDICATIONS AND USAGE SECTION)
Formuated for associated symptoms such as chills, aches, sweating, fatigue, nausea and loss of appetite.
Dosage & Administration Section (DOSAGE & ADMINISTRATION SECTION)
Directions: Children, ages 0 to 11, give 3 drops by mouth (ages 12 and up, give 6 drops) as needed or a directed by a health professional. Sensitive person begin with 1 drop and gradually increase to full dose.
Warning: If symptoms become severe or fever persists for more than 48 hous, consult a licensed health professional.
Otc Active Ingredient Section (OTC - ACTIVE INGREDIENT SECTION)
Equal parts of Chamomilla 6x, Echinacea 6x, Hydrastis canadensis 6x, Aconitum napellus 15x, Arsenicum album 15x, Belladonna 15x, Bryonia 15x, Camphora 15x, Cina 15x, Cinchona officinalis 15x, Eupatorium perfoliatum 15x, Ferrum phosphoricum 15x, Gelsemium sempervirens 15x, Hepar sulphuris calcareum 15x, Ignatia amara 15x, Natrum muriaticum 15x, Nux vomica 15x, Phosphorus 15x, Pulsatilla 15x, Salix nigra 15x, Veratrum viride 15x, Influenzinum 30x.
Otc Pregnancy Or Breast Feeding Section (OTC - PREGNANCY OR BREAST FEEDING SECTION)
If pregnant or breast-feeding, ask a doctor before use.
Otc Keep Out of Reach of Children Section (OTC - KEEP OUT OF REACH OF CHILDREN SECTION)
WARNINGS: Keep out of reach of children. Do not use if tamper-evident seal is broken or missing. If symptoms worsen or persist for more than a few days, consult a doctor. If pregnant or breast-feeding, ask a doctor before use.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:11:55.876768 · Updated: 2026-03-14T23:17:14.395389