These Highlights Do Not Include All The Information Needed To Use
9d502961-e9c0-4611-92bf-ce742a7b962c
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Warnings and Precautions ( 5.5 , 5.10 , 5.16 ) 11/2022
Indications and Usage
Tacrolimus is a is a calcineurin-inhibitor immunosuppressant indicated for the prophylaxis of organ rejection in adult patients receiving allogeneic liver, kidney or heart transplants, and pediatric patients receiving allogeneic liver transplants in combination with other immunosuppressants. ( 1.1 )
Dosage and Administration
Intravenous (IV) use recommended for patients who cannot tolerate oral formulations (capsules or suspension). ( 2.1 , 2.2 ) Therapeutic drug monitoring is recommended. ( 2.1 , 2.6 ) Avoid eating grapefruit or drinking grapefruit juice. ( 2.1 ) See dosage adjustments for African-American patients ( 2.2 ), hepatic and renal impaired. ( 2.4 , 2.5 ) For complete dosing information, see Full Prescribing Information.
Warnings and Precautions
New Onset Diabetes After Transplant: Monitor blood glucose. ( 5.4 ) Nephrotoxicity (acute and/or chronic): Reduce the dose; use caution with other nephrotoxic drugs. ( 5.5 ) Neurotoxicity: Including risk of Posterior Reversible Encephalopathy Syndrome (PRES); monitor for neurologic abnormalities; reduce or discontinue tacrolimus. ( 5.6 ) Hyperkalemia: Monitor serum potassium levels. Consider carefully before using with other agents also associated with hyperkalemia. ( 5.7 ) Hypertension: May require antihypertensive therapy. Monitor relevant drug-drug interactions. ( 5.8 ) Anaphylactic Reactions with intravenous formulation: Observe patients receiving tacrolimus injection for signs and symptoms of anaphylaxis. ( 5.9 ) Not recommended for use with sirolimus: Not recommended in liver and heart transplant due to increased risk of serious adverse reactions. ( 5.10 ) Myocardial Hypertrophy: Consider dose reduction/discontinuation. ( 5.13 ) Immunizations: Avoid live vaccines. ( 5.14 ) Pure Red Cell Aplasia: Consider discontinuation of tacrolimus injection. ( 5.15 ) Thrombotic Microangiopathy, Including Hemolytic Uremic Syndrome and Thrombotic Thrombocytopenic Purpura: May occur, especially in patients with infections and certain concomitant medications. ( 5.16 )
Contraindications
Tacrolimus injection is contraindicated in patients with a hypersensitivity to tacrolimus. Tacrolimus injection is contraindicated in patients with a hypersensitivity to HCO-60 (polyoxyl 60 hydrogenated castor oil). Hypersensitivity symptoms reported include dyspnea, rash, pruritus, and acute respiratory distress syndrome [see Adverse Reactions ( 6 )] .
Adverse Reactions
The following serious and otherwise important adverse drug reactions are discussed in greater detail in other sections of labeling: Lymphoma and Other Malignancies [see Warnings and Precautions ( 5.1 )] Serious Infections [see Warnings and Precautions ( 5.2 )] New Onset Diabetes After Transplant [see Warnings and Precautions ( 5.4 )] Nephrotoxicity [see Warnings and Precautions ( 5.5 )] Neurotoxicity [see Warnings and Precautions ( 5.6 )] Hyperkalemia [see Warnings and Precautions ( 5.7 )] Hypertension [see Warnings and Precautions ( 5.8 )] Anaphylactic Reactions with Tacrolimus Injection [see Warnings and Precautions ( 5.9 )] Myocardial Hypertrophy [see Warnings and Precautions ( 5.13 )] Pure Red Cell Aplasia [see Warnings and Precautions ( 5.15 )] Thrombotic Microangiopathy, Including Hemolytic Uremic Syndrome and Thrombotic Thrombocytopenic Purpura [see Warnings and Precautions ( 5.16 )]
Drug Interactions
Instruct patients to tell their healthcare providers when they start or stop taking any medicines, including prescription medicines and nonprescription medicines, natural or herbal remedies, nutritional supplements, and vitamins. Advise patients to avoid grapefruit and grapefruit juice [see Drug Interactions ( 7 )].
Medication Information
Warnings and Precautions
New Onset Diabetes After Transplant: Monitor blood glucose. ( 5.4 ) Nephrotoxicity (acute and/or chronic): Reduce the dose; use caution with other nephrotoxic drugs. ( 5.5 ) Neurotoxicity: Including risk of Posterior Reversible Encephalopathy Syndrome (PRES); monitor for neurologic abnormalities; reduce or discontinue tacrolimus. ( 5.6 ) Hyperkalemia: Monitor serum potassium levels. Consider carefully before using with other agents also associated with hyperkalemia. ( 5.7 ) Hypertension: May require antihypertensive therapy. Monitor relevant drug-drug interactions. ( 5.8 ) Anaphylactic Reactions with intravenous formulation: Observe patients receiving tacrolimus injection for signs and symptoms of anaphylaxis. ( 5.9 ) Not recommended for use with sirolimus: Not recommended in liver and heart transplant due to increased risk of serious adverse reactions. ( 5.10 ) Myocardial Hypertrophy: Consider dose reduction/discontinuation. ( 5.13 ) Immunizations: Avoid live vaccines. ( 5.14 ) Pure Red Cell Aplasia: Consider discontinuation of tacrolimus injection. ( 5.15 ) Thrombotic Microangiopathy, Including Hemolytic Uremic Syndrome and Thrombotic Thrombocytopenic Purpura: May occur, especially in patients with infections and certain concomitant medications. ( 5.16 )
Indications and Usage
Tacrolimus is a is a calcineurin-inhibitor immunosuppressant indicated for the prophylaxis of organ rejection in adult patients receiving allogeneic liver, kidney or heart transplants, and pediatric patients receiving allogeneic liver transplants in combination with other immunosuppressants. ( 1.1 )
Dosage and Administration
Intravenous (IV) use recommended for patients who cannot tolerate oral formulations (capsules or suspension). ( 2.1 , 2.2 ) Therapeutic drug monitoring is recommended. ( 2.1 , 2.6 ) Avoid eating grapefruit or drinking grapefruit juice. ( 2.1 ) See dosage adjustments for African-American patients ( 2.2 ), hepatic and renal impaired. ( 2.4 , 2.5 ) For complete dosing information, see Full Prescribing Information.
Contraindications
Tacrolimus injection is contraindicated in patients with a hypersensitivity to tacrolimus. Tacrolimus injection is contraindicated in patients with a hypersensitivity to HCO-60 (polyoxyl 60 hydrogenated castor oil). Hypersensitivity symptoms reported include dyspnea, rash, pruritus, and acute respiratory distress syndrome [see Adverse Reactions ( 6 )] .
Adverse Reactions
The following serious and otherwise important adverse drug reactions are discussed in greater detail in other sections of labeling: Lymphoma and Other Malignancies [see Warnings and Precautions ( 5.1 )] Serious Infections [see Warnings and Precautions ( 5.2 )] New Onset Diabetes After Transplant [see Warnings and Precautions ( 5.4 )] Nephrotoxicity [see Warnings and Precautions ( 5.5 )] Neurotoxicity [see Warnings and Precautions ( 5.6 )] Hyperkalemia [see Warnings and Precautions ( 5.7 )] Hypertension [see Warnings and Precautions ( 5.8 )] Anaphylactic Reactions with Tacrolimus Injection [see Warnings and Precautions ( 5.9 )] Myocardial Hypertrophy [see Warnings and Precautions ( 5.13 )] Pure Red Cell Aplasia [see Warnings and Precautions ( 5.15 )] Thrombotic Microangiopathy, Including Hemolytic Uremic Syndrome and Thrombotic Thrombocytopenic Purpura [see Warnings and Precautions ( 5.16 )]
Drug Interactions
Instruct patients to tell their healthcare providers when they start or stop taking any medicines, including prescription medicines and nonprescription medicines, natural or herbal remedies, nutritional supplements, and vitamins. Advise patients to avoid grapefruit and grapefruit juice [see Drug Interactions ( 7 )].
Description
Warnings and Precautions ( 5.5 , 5.10 , 5.16 ) 11/2022
Section 42229-5
Intravenous Formulation - Administration Precautions due to Risk of Anaphylaxis
Intravenous use is recommended for patients who cannot tolerate oral formulations, and conversion from intravenous to oral tacrolimus is recommended as soon as oral therapy can be tolerated to minimize the risk of anaphylactic reactions that occurred with injectables containing castor oil derivatives [see Warnings and Precautions ( 5.9)] .
Patients receiving tacrolimus injection should be under continuous observation for at least the first 30 minutes following the start of the infusion and at frequent intervals thereafter. If signs or symptoms of anaphylaxis occur, the infusion should be stopped. An aqueous solution of epinephrine should be available at the bedside as well as a source of oxygen.
Section 44425-7
Store and Dispense
Store between 5°C and 25°C (41°F and 77°F).
Section 51945-4
Principal Display Panel - 1 mL Carton Label
NDC 14789-135-05
Rx Only
Tacrolimus
Injection
5 mg/1 mL
(For intravenous use)
For Intravenous Infusion Only After Dilution
10 x 1 mL Single-dose Vials
NEXUS
PHARMACEUTICALS
10 Overdosage
Limited overdosage experience is available. Acute overdosages of up to 30 times the intended dose have been reported. Almost all cases have been asymptomatic and all patients recovered with no sequelae. Acute overdosage was sometimes followed by adverse reactions consistent with those reported with the use of Tacrolimus [see Adverse Reactions ( 6.1, 6.2)] , including tremors, abnormal renal function, hypertension, and peripheral edema; in one case of acute overdosage, transient urticaria and lethargy were observed. Based on the poor aqueous solubility and extensive erythrocyte and plasma protein binding, it is anticipated that tacrolimus is not dialyzable to any significant extent; there is no experience with charcoal hemoperfusion. The oral use of activated charcoal has been reported in treating acute overdoses, but experience has not been sufficient to warrant recommending its use. General supportive measures and treatment of specific symptoms should be followed in all cases of overdosage.
15 References
1. “OSHA Hazardous Drugs.” OSHA. http://www.osha.gov/SLTC/hazardousdrugs/index.html
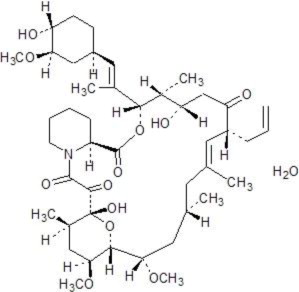
11 Description
Tacrolimus, previously known as FK506, is the active ingredient in Tacrolimus. Tacrolimus is a calcineurin-inhibitor immunosuppressant produced by Streptomyces tsukubaensis. Chemically, tacrolimus is designated as [3 S- [3 R*[ E(1 S*,3 S*,4 S*)], 4 S*,5 R*,8 S*,9 E,12 R*,14 R*,15 S*,16 R*,18 S*,19 S*,26a R*]] -
5,6,8,11,12,13,14,15,16,17,18,19,24,25,26,26a-hexadecahydro-5,19-dihydroxy-3-[2-(4-hydroxy-3-methoxycyclohexyl)-1- methylethenyl]-14,16-dimethoxy-4,10,12,18-tetramethyl-8-(2-propenyl)-15,19-epoxy-3H-pyrido[2,1- c][1,4] oxaazacyclotricosine-1,7,20,21(4H,23H)-tetrone, monohydrate.
The chemical structure of tacrolimus is:
Tacrolimus has an empirical formula of C 44H 69NO 12•H 2O and a formula weight of 822.03. Tacrolimus appears as white crystals or crystalline powder. It is practically insoluble in water, freely soluble in ethanol, and very soluble in methanol and chloroform.
Tacrolimus injection is a sterile solution containing the equivalent of 5 mg anhydrous tacrolimus USP in 1 mL for administration by intravenous infusion only. Each mL contains the following inactive ingredients: dehydrated alcohol USP, 80.0% v/v and polyoxyl 60 hydrogenated castor oil (HCO-60), 200 mg. Tacrolimus injection must be diluted with 0.9% Sodium Chloride Injection or 5% Dextrose Injection before use.
5.7 Hyperkalemia
Hyperkalemia has been reported with tacrolimus use. Serum potassium levels should be monitored. Careful consideration should be given prior to use of other agents also associated with hyperkalemia (e.g., potassium-sparing diuretics, ACE inhibitors, angiotensin receptor blockers) during Tacrolimus therapy [see Adverse Reactions ( 6.1)] . Monitor serum potassium levels periodically during treatment.
5.8 Hypertension
Hypertension is a common adverse effect of tacrolimus therapy and may require antihypertensive therapy [see Adverse Reactions ( 6.1)]. The control of blood pressure can be accomplished with any of the common antihypertensive agents, though careful consideration should be given prior to use of antihypertensive agents associated with hyperkalemia (e.g., potassium-sparing diuretics, ACE inhibitors, angiotensin receptor blockers) [see Warnings and Precautions ( 5.7)] .Calcium-channel blocking agents may increase tacrolimus blood concentrations and therefore require dosage reduction of Tacrolimus [see Drug Interactions ( 7.2)].
17.7 Hyperkalemia
Inform patients that tacrolimus injection can cause hyperkalemia. Monitoring of potassium levels may be necessary, especially with concomitant use of other drugs known to cause hyperkalemia [see Warnings and Precautions ( 5.7)] .
17.8 Hypertension
Inform patients that tacrolimus can cause high blood pressure which may require treatment with antihypertensive therapy. Advise patients to monitor their blood pressure [see Warnings and Precautions ( 5.8)] .
5.6 Neurotoxicity
Tacrolimus may cause a spectrum of neurotoxicities. The most severe neurotoxicities include posterior reversible encephalopathy syndrome (PRES), delirium, seizure and coma; others include tremors, paresthesias, headache, mental status changes, and changes in motor and sensory functions [see Adverse Reactions ( 6.1, 6.2)] . As symptoms may be associated with tacrolimus whole blood trough concentrations at or above the recommended range, monitor for neurologic symptoms and consider dosage reduction or discontinuation of Tacrolimus Injection if neurotoxicity occurs.
8.4 Pediatric Use
Safety and effectiveness have been established in pediatric liver transplant patients.
8.5 Geriatric Use
Clinical trials of tacrolimus did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
17.6 Neurotoxicity
Inform patients that they are at risk of developing adverse neurologic reactions including seizure, altered mental status, and tremor. Advise patients to contact their physician should they develop vision changes, delirium, or tremors [see Warnings and Precautions ( 5.6)] .
5.14 Immunizations
Whenever possible, administer the complete complement of vaccines before transplantation and treatment with tacrolimus.
The use of live vaccines should be avoided during treatment with tacrolimus; examples include (not limited to) the following: intranasal influenza, measles, mumps, rubella, oral polio, BCG, yellow fever, varicella, and TY21a typhoid vaccines.
Inactivated vaccines noted to be safe for administration after transplantation may not be sufficiently immunogenic during treatment with tacrolimus.
17.1 Administration
Advise the patient or caregiver to:
- Not to eat grapefruit or drink grapefruit juice in combination with tacrolimus [see Drug Interactions ( 7.2)].
17.13 Immunizations
Inform patients that Tacrolimus can interfere with the usual response to immunizations and that they should avoid live vaccines . [see Warnings and Precautions ( 5.14)] .
Manufactured in the USA for:
Nexus Pharmaceuticals, LLC
Lincolnshire, IL 60069 USA
NEXUS
PHARMACEUTICALS
TACPI01USR01
Revised: 2/2025
17.5 Nephrotoxicity
Inform patients that tacrolimus injection can have toxic effects on the kidney that should be monitored. Advise patients to attend all visits and complete all blood tests ordered by their medical team [see Warnings and Precautions ( 5.5)] .
4 Contraindications
Tacrolimus injection is contraindicated in patients with a hypersensitivity to tacrolimus. Tacrolimus injection is contraindicated in patients with a hypersensitivity to HCO-60 (polyoxyl 60 hydrogenated castor oil). Hypersensitivity symptoms reported include dyspnea, rash, pruritus, and acute respiratory distress syndrome [see Adverse Reactions ( 6)] .
6 Adverse Reactions
The following serious and otherwise important adverse drug reactions are discussed in greater detail in other sections of labeling:
- Lymphoma and Other Malignancies [see Warnings and Precautions ( 5.1)]
- Serious Infections [see Warnings and Precautions ( 5.2)]
- New Onset Diabetes After Transplant [see Warnings and Precautions ( 5.4)]
- Nephrotoxicity [see Warnings and Precautions ( 5.5)]
- Neurotoxicity [see Warnings and Precautions ( 5.6)]
- Hyperkalemia [see Warnings and Precautions ( 5.7)]
- Hypertension [see Warnings and Precautions ( 5.8)]
- Anaphylactic Reactions with Tacrolimus Injection [see Warnings and Precautions ( 5.9)]
- Myocardial Hypertrophy [see Warnings and Precautions ( 5.13)]
- Pure Red Cell Aplasia [see Warnings and Precautions ( 5.15)]
- Thrombotic Microangiopathy, Including Hemolytic Uremic Syndrome and Thrombotic Thrombocytopenic Purpura [see Warnings and Precautions ( 5.16)]
7 Drug Interactions
- Mycophenolic Acid Products: Can increase MPA exposure after crossover from cyclosporine to tacrolimus; monitor for MPA-related adverse reactions and adjust MMF or MPA dose as needed. ( 7.1)
- Nelfinavir and Grapefruit Juice: Increased tacrolimus concentrations via CYP3A inhibition; avoid concomitant use. ( 7.2)
- CYP3A Inhibitors: Increased tacrolimus concentrations; monitor concentrations and adjust tacrolimus dose as needed. ( 5.11, 7.2)
- CYP3A4 Inducers: Decreased tacrolimus concentrations; monitor concentrations and adjust tacrolimus dose as needed. ( 5.11, 7.2)
5.12 Qt Prolongation
Tacrolimus injection may prolong the QT/QTc interval and may cause Torsade de pointes. Avoid Tacrolimus Injection in patients with congenital long QT syndrome. In patients with congestive heart failure, bradyarrhythmias, those taking certain antiarrhythmic medications or other medicinal products that lead to QT prolongation, and those with electrolyte disturbances such as hypokalemia, hypocalcemia, or hypomagnesemia, consider obtaining electrocardiograms and monitoring electrolytes (magnesium, potassium, calcium) periodically during treatment.
When co-administering tacrolimus with other substrates and/or inhibitors of CYP3A4 that also have the potential to prolong the QT interval, a reduction in tacrolimus dose, frequent monitoring of tacrolimus whole blood concentrations, and monitoring for QT prolongation is recommended. Use of tacrolimus with amiodarone has been reported to result in increased tacrolimus whole blood concentrations with or without concurrent QT prolongation [see Drug Interactions ( 7.2)] .
8.6 Renal Impairment
The pharmacokinetics of tacrolimus in patients with renal impairment was similar to that in healthy volunteers with normal renal function. However, consideration should be given to dosing tacrolimus at the lower end of the therapeutic dosing range in patients who have received a liver or heart transplant and have pre-existing renal impairment. Further reductions in dose below the targeted range may be required [see Dosage and Administration ( 2.4) and Clinical Pharmacology ( 12.3)] .
12.3 Pharmacokinetics
Tacrolimus activity is primarily due to the parent drug. The pharmacokinetic parameters (mean ± S.D.) of tacrolimus have been determined following intravenous (IV) and/or oral (PO) administration in healthy volunteers, and in kidney transplant, liver transplant, and heart transplant patients ( Table 17).
|
1. Not applicable |
||||||||
|
2. AUC 0-inf |
||||||||
|
3. Not available |
||||||||
|
4. AUC 0-t |
||||||||
|
5. Determined after the first dose |
||||||||
|
6. Median [range] |
||||||||
|
7.AUC 0-12 |
||||||||
| Population |
N
|
Route (Dose) | Parameters | |||||
| C max(nanogram/mL) | T max(hr) | AUC (nanogram•hr/mL) | t 1/2(hr) | CL (L/hr/kg) | V (L/kg) | |||
| Healthy Volunteers | 8 | Intravenous (0.025 mg/kg/4 hr) | 1 | 1 | 652 2± 156 | 34.2 ± 7.7 | 0.040 ± 0.009 | 1.91 ± 0.31 |
| 16 | PO (5 mg) (capsules) | 28.8 ± 8.9 | 1.5 ± 0.7 | 266 2± 95 | 32.3 ± 8.8 | 3 | 3 | |
| Kidney Transplant Patients | 26 | Intravenous (0.02 mg/kg/12 hr) | 1 | 1 | 294 2± 262 | 18.8 ± 16.7 | 0.083 ± 0.050 | 1.41 ± 0.66 |
| PO (0.2 mg/kg/day) | 19.2 ± 10.3 | 3.0 | 203 2± 42 | 3 | 3 | 3 | ||
| PO (0.3 mg/kg/day) | 24.2 ± 15.8 | 1.5 | 288 2± 93 | 3 | 3 | 3 | ||
| Liver Transplant Patients | 17 | Intravenous (0.05 mg/kg/12 hr) | 1 | 1 | 3300 2±2130 | 11.7 ±3.9 | 0.053 ±0.017 | 0.85 ±0.30 |
| PO (0.3 mg/kg/day) | 68.5 ± 30.0 | 2.3 ± 1.5 | 519 2± 179 | 3 | 3 | 3 | ||
|
Heart Transplant Patients |
11 | Intravenous (0.01 mg/kg/day as a continuous infusion) | 1 | 1 | 954 4± 334 | 23.6 ± 9.22 | 0.051 ± 0.015 | 3 |
| 11 | PO (0.075 mg/kg/day) 5 | 14.7 ± 7.79 | 2.1
[0.5-6.0] 6 |
82.7 7± 63.2 | 1 | 3 | 3 | |
| 14 | PO (0.15 mg/kg/day) 5 | 24.5 ± 13.7 | 1.5
[0.4-4.0] 6 |
142 7± 116 | 1 | 3 | 3 |
Due to intersubject variability in tacrolimus pharmacokinetics, individualization of the dosing regimen is necessary for optimal therapy [see Dosage and Administration ( 2.6)] . Pharmacokinetic data indicate that whole blood concentrations rather than plasma concentrations serve as the more appropriate sampling compartment to describe tacrolimus pharmacokinetics.
7.1 Mycophenolic Acid
When tacrolimus is prescribed with a given dose of a mycophenolic acid (MPA) product, exposure to MPA is higher with tacrolimus co-administration than with cyclosporine co-administration with MPA, because cyclosporine interrupts the enterohepatic recirculation of MPA while tacrolimus does not. Monitor for MPA-associated adverse reactions and reduce the dose of concomitantly administered mycophenolic acid products as needed.
8.8 Race Or Ethnicity
African-American patients may need to be titrated to higher dosages to attain comparable trough concentrations compared to Caucasian patients [see Dosage and Administration ( 2.2) and Clinical Pharmacology ( 12.3)].
African-American and Hispanic patients are at increased risk for new onset diabetes after transplant. Monitor blood glucose concentrations and treat appropriately [see Warnings and Precautions ( 5.4)].
5.2 Serious Infections
Patients receiving immunosuppressants, including tacrolimus, are at increased risk of developing bacterial, viral, fungal, and protozoal infections, including opportunistic infections. These infections may lead to serious, including fatal, outcomes. Serious viral infections reported include:
- Polyomavirus-associated nephropathy (PVAN), mostly due to BK virus infection
- JC virus-associated progressive multifocal leukoencephalopathy (PML)
- Cytomegalovirus infections: CMV seronegative transplant patients who receive an organ from a CMV seropositive donor disease are at higher risk of developing CMV viremia and CMV disease.
Monitor for the development of infection and adjust the immunosuppressive regimen to balance the risk of rejection with the risk of infection [see Adverse Reactions ( 6.1, 6.2)].
8.7 Hepatic Impairment
The mean clearance of tacrolimus was substantially lower in patients with severe hepatic impairment (mean Child-Pugh score: > 10) compared to healthy volunteers with normal hepatic function. Close monitoring of tacrolimus trough concentrations is warranted in patients with hepatic impairment [see Clinical Pharmacology ( 12.3)] .
The use of Tacrolimus in liver transplant recipients experiencing post-transplant hepatic impairment may be associated with increased risk of developing renal insufficiency related to high whole blood trough concentrations of tacrolimus. These patients should be monitored closely and dosage adjustments should be considered. Some evidence suggests that lower doses should be used in these patients [see Dosage and Administration ( 2.5) and Clinical Pharmacology ( 12.3)].
1 Indications and Usage
Tacrolimus is a is a calcineurin-inhibitor immunosuppressant indicated for the prophylaxis of organ rejection in adult patients receiving allogeneic liver, kidney or heart transplants, and pediatric patients receiving allogeneic liver transplants in combination with other immunosuppressants. ( 1.1)
17.10 Drug Interactions
Instruct patients to tell their healthcare providers when they start or stop taking any medicines, including prescription medicines and nonprescription medicines, natural or herbal remedies, nutritional supplements, and vitamins. Advise patients to avoid grapefruit and grapefruit juice [see Drug Interactions ( 7)].
12.1 Mechanism of Action
Tacrolimus binds to an intracellular protein, FKBP-12. A complex of tacrolimus-FKBP-12, calcium, calmodulin, and calcineurin (a ubiquitous mammalian intracellular enzyme) is then formed, after which the phosphatase activity of calcineurin is inhibited. Such inhibition prevents the dephosphorylation and translocation of various factors such as the nuclear factor of activated T-cells (NF-AT), and nuclear factor kappa-light-chain enhancer of activated B-cells (NF-ϰB).
Tacrolimus inhibits the expression and/or production of several cytokines that include interleukin (IL)-1 beta, IL-2, IL-3, IL-4, IL-5, IL-6, IL-8, IL-10, gamma interferon, tumor necrosis factor-alpha, and granulocyte macrophage colony-stimulating factor. Tacrolimus also inhibits IL-2 receptor expression and nitric oxide release, induces apoptosis and production of transforming growth factor beta that can lead to immunosuppressive activity. The net result is the inhibition of T-lymphocyte activation and proliferation, as well as T-helper-cell-dependent B-cell response (i.e., immunosuppression).
16.2 Tacrolimus Injection
(for Intravenous infusion only)
| Unit of Sale | Concentration | Each |
| NDC 14789-135-05
Carton containing 10 vials |
5 mg/mL | NDC 14789-135-07
1 mL Single use vial |
5 mg/mL (equivalent of 5 mg of anhydrous tacrolimus USP per mL) supplied as a sterile solution in a 1 mL vial, in a carton of 10 vials.
14.2 Liver Transplantation
The safety and efficacy of tacrolimus-based immunosuppression following orthotopic liver transplantation were assessed in two prospective, randomized, non-blinded multicenter trials. The active control groups were treated with a cyclosporine-based immunosuppressive regimen (CsA/AZA). Both trials used concomitant adrenal corticosteroids as part of the immunosuppressive regimens. These trials compared patient and graft survival rates at 12 months following transplantation.
In one trial, 529 patients were enrolled at 12 clinical sites in the United States; prior to surgery, 263 were randomized to the tacrolimus-based immunosuppressive regimen and 266 to the CsA/AZA. In 10 of the 12 sites, the same CsA/AZA protocol was used, while 2 sites used different control protocols. This trial excluded patients with renal dysfunction, fulminant hepatic failure with Stage IV encephalopathy, and cancers; pediatric patients (≤ 12 years old) were allowed.
In the second trial, 545 patients were enrolled at 8 clinical sites in Europe; prior to surgery, 270 were randomized to the tacrolimus-based immunosuppressive regimen and 275 to CsA/AZA. In this trial, each center used its local standard CsA/AZA protocol in the active-control arm. This trial excluded pediatric patients, but did allow enrollment of subjects with renal dysfunction, fulminant hepatic failure in Stage IV encephalopathy, and cancers other than primary hepatic with metastases.
One-year patient survival and graft survival in the Tacrolimus-based treatment groups were similar to those in the CsA/AZA treatment groups in both trials. The overall 1-year patient survival (CsA/AZA and tacrolimus-based treatment groups combined) was 88% in the U.S. trial and 78% in the European trial. The overall 1-year graft survival (CsA/AZA and tacrolimus-based treatment groups combined) was 81% in the U.S. trial and 73% in the European trial. In both trials, the median time to convert from intravenous to oral tacrolimus dosing was 2 days.
Although there is a lack of direct correlation between tacrolimus concentrations and drug efficacy, data from clinical trials of liver transplant patients have shown an increasing incidence of adverse reactions with increasing trough blood concentrations. Most patients are stable when trough whole blood concentrations are maintained between 5 to 20 nanogram/mL. Long-term post-transplant patients are often maintained at the low end of this target range.
Data from the U.S. clinical trial show that the median trough blood concentrations, measured at intervals from the second week to one year post-transplantation, ranged from 9.8 ng/mL to 19.4 ng/mL.
14.3 Heart Transplantation
Two open-label, randomized, comparative trials evaluated the safety and efficacy of tacrolimus-based and cyclosporine-based immunosuppression in primary orthotopic heart transplantation. In a trial conducted in Europe, 314 patients received a regimen of antibody induction, corticosteroids, and azathioprine in combination with tacrolimus or cyclosporine modified for 18 months. In a 3-arm trial conducted in the U.S., 331 patients received corticosteroids and tacrolimus plus sirolimus, tacrolimus plus mycophenolate mofetil (MMF) or cyclosporine modified plus MMF for 1 year.
In the European trial, patient/graft survival at 18 months post-transplant was similar between treatment arms, 92% in the tacrolimus group and 90% in the cyclosporine group. In the U.S. trial, patient and graft survival at 12 months was similar with 93% survival in the tacrolimus plus MMF group and 86% survival in the cyclosporine modified plus MMF group. In the European trial, the cyclosporine trough concentrations were above the pre-defined target range (i.e., 100 to 200 nanogram/mL) at Day 122 and beyond in 32% to 68% of the patients in the cyclosporine treatment arm, whereas the tacrolimus trough concentrations were within the pre-defined target range (i.e., 5 to 15 nanogram/mL) in 74% to 86% of the patients in the tacrolimus treatment arm. Data from this European trial indicate that from 1 week to 3 months post-transplant, approximately 80% of patients maintained trough concentrations between 8 to 20 nanogram/mL and, from 3 months through 18 months post-transplant, approximately 80% of patients maintained trough concentrations between 6 to 18 nanogram/mL.
The U.S. trial contained a third arm of a combination regimen of sirolimus, 2 mg per day, and full-dose tacrolimus; however, this regimen was associated with increased risk of wound-healing complications, renal function impairment, and insulin-dependent post-transplant diabetes mellitus, and is not recommended [see Warnings and Precautions ( 5.10)].
16.4 Handling and Disposal
Tacrolimus can cause fetal harm. Wearing disposable gloves is recommended during dilution of the injection in the hospital and when wiping any spills. In case a spill occurs, wipe the surface with a wet paper towel. Follow applicable special handling and disposal procedures 1.
5 Warnings and Precautions
- New Onset Diabetes After Transplant: Monitor blood glucose. ( 5.4)
- Nephrotoxicity (acute and/or chronic): Reduce the dose; use caution with other nephrotoxic drugs. ( 5.5)
- Neurotoxicity: Including risk of Posterior Reversible Encephalopathy Syndrome (PRES); monitor for neurologic abnormalities; reduce or discontinue tacrolimus. ( 5.6)
- Hyperkalemia: Monitor serum potassium levels. Consider carefully before using with other agents also associated with hyperkalemia. ( 5.7)
- Hypertension: May require antihypertensive therapy. Monitor relevant drug-drug interactions. ( 5.8)
- Anaphylactic Reactions with intravenous formulation: Observe patients receiving tacrolimus injection for signs and symptoms of anaphylaxis. ( 5.9)
- Not recommended for use with sirolimus: Not recommended in liver and heart transplant due to increased risk of serious adverse reactions. ( 5.10)
- Myocardial Hypertrophy: Consider dose reduction/discontinuation. ( 5.13)
- Immunizations: Avoid live vaccines. ( 5.14)
- Pure Red Cell Aplasia: Consider discontinuation of tacrolimus injection. ( 5.15)
- Thrombotic Microangiopathy, Including Hemolytic Uremic Syndrome and Thrombotic Thrombocytopenic Purpura: May occur, especially in patients with infections and certain concomitant medications. ( 5.16)
5.15 Pure Red Cell Aplasia
Cases of pure red cell aplasia (PRCA) have been reported in patients treated with tacrolimus. A mechanism for tacrolimus-induced PRCA has not been elucidated. All patients reported risk factors for PRCA such as parvovirus B19 infection, underlying disease, or concomitant medications associated with PRCA. If PRCA is diagnosed, discontinuation of tacrolimus should be considered [see Adverse Reactions ( 6.2)] .
2 Dosage and Administration
- Intravenous (IV) use recommended for patients who cannot tolerate oral formulations (capsules or suspension). ( 2.1, 2.2)
- Therapeutic drug monitoring is recommended. ( 2.1, 2.6)
- Avoid eating grapefruit or drinking grapefruit juice. ( 2.1)
- See dosage adjustments for African-American patients ( 2.2), hepatic and renal impaired. ( 2.4, 2.5)
- For complete dosing information, see Full Prescribing Information.
5.13 Myocardial Hypertrophy
Myocardial hypertrophy has been reported in infants, children, and adults, particularly those with high tacrolimus trough concentrations, and is generally manifested by echocardiographically demonstrated concentric increases in left ventricular posterior wall and interventricular septum thickness. This condition appears reversible in most cases following dose reduction or discontinuance of therapy. In patients who develop renal failure or clinical manifestations of ventricular dysfunction while receiving tacrolimus injection therapy, echocardiographic evaluation should be considered. If myocardial hypertrophy is diagnosed, dosage reduction or discontinuation of tacrolimus injection should be considered [see Adverse Reactions ( 6.2)] .
17.12 Myocardial Hypertrophy
Inform patients to report symptoms of tiredness, swelling, and/or shortness of breath (heart failure).
3 Dosage Forms and Strengths
The vial contains 1 mL anhydrous tacrolimus USP for intravenous infusion as follows:
- 5 mg/mL, sterile solution
6.2 Postmarketing Experience
The following adverse reactions have been reported from worldwide marketing experience with tacrolimus. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Decisions to include these reactions in labeling are typically based on one or more of the following factors: (1) seriousness of the reaction, (2) frequency of the reporting, or (3) strength of causal connection to the drug.
Other reactions include:
- Cardiovascular: Atrial fibrillation, atrial flutter, cardiac arrhythmia, cardiac arrest, electrocardiogram T wave abnormal, flushing, myocardial infarction, myocardial ischemia, pericardial effusion, QT prolongation, Torsade de Pointes, venous thrombosis deep limb, ventricular extrasystoles, ventricular fibrillation, myocardial hypertrophy
- Gastrointestinal: Bile duct stenosis, colitis, enterocolitis, gastroenteritis, gastroesophageal reflux disease, hepatic cytolysis, hepatic necrosis, hepatotoxicity, impaired gastric emptying, liver fatty, mouth ulceration, pancreatitis hemorrhagic, pancreatitis necrotizing, stomach ulcer, veno-occlusive liver disease
- Hemic/Lymphatic: Agranulocytosis, disseminated intravascular coagulation, hemolytic anemia, neutropenia, febrile neutropenia, pancytopenia, thrombocytopenic purpura, thrombotic thrombocytopenic purpura, pure red cell aplasia, thrombotic microangiopathy
- Infections: Cases of progressive multifocal leukoencephalopathy (PML), sometimes fatal; polyoma virus- associated nephropathy (PVAN) including graft loss
- Metabolic/Nutritional: Glycosuria, increased amylase including pancreatitis, weight decreased
- Miscellaneous: Feeling hot and cold, feeling jittery, hot flushes, multi-organ failure, primary graft dysfunction
- Musculoskeletal and Connective Tissue Disorders: Pain in extremity including Calcineurin-Inhibitor Induced Pain Syndrome (CIPS)
- Nervous System: Carpal tunnel syndrome, cerebral infarction, hemiparesis, leukoencephalopathy, mental disorder, mutism, posterior reversible encephalopathy syndrome (PRES), progressive multifocal leukoencephalopathy (PML), quadriplegia, speech disorder, syncope
- Respiratory: Acute respiratory distress syndrome, interstitial lung disease, lung infiltration, respiratory distress, respiratory failure
- Skin: Stevens-Johnson syndrome, toxic epidermal necrolysis
- Special Senses: Blindness, optic neuropathy, blindness cortical, hearing loss including deafness, photophobia
- Urogenital: Acute renal failure, cystitis hemorrhagic, hemolytic-uremic syndrome
8 Use in Specific Populations
- Pregnancy: Can cause fetal harm. Advise pregnant women of the potential risk to the fetus. ( 8.1, 8.3)
Additional pediatric use information is approved for Astellas Pharma US, Inc.'s Prograf ® (tacrolimus) products. However, due to Astellas Pharma US, Inc.'s marketing exclusivity rights, this drug product is not labeled with that information.
17.9 Thrombotic Microangiopathy
Inform patients that tacrolimus can cause blood clotting problems. The risk of this occurring increases when patients take tacrolimus and sirolimus or everolimus concomitantly, or when patients develop certain infections. Advise them to seek medical attention promptly if they develop fever, petequiae or bruises, fatigue, confusion, jaundice, oliguria. [see Warnings and Precautions ( 5.16)]
2.6 Therapeutic Drug Monitoring
Monitoring of tacrolimus blood concentrations in conjunction with other laboratory and clinical parameters is considered an essential aid to patient management for the evaluation of rejection, toxicity, dose adjustments, and compliance.
Factors influencing frequency of monitoring include but are not limited to hepatic or renal dysfunction, the addition or discontinuation of potentially interacting drugs and the post-transplant time. Blood concentration monitoring is not a replacement for renal and liver function monitoring and tissue biopsies. Data from clinical trials show that tacrolimus whole blood concentrations were most variable during the first week post-transplantation.
The relative risks of toxicity and efficacy failure are related to tacrolimus whole blood trough concentrations. Therefore, monitoring of whole blood trough concentrations is recommended to assist in the clinical evaluation of toxicity and efficacy failure.
Methods commonly used for the assay of tacrolimus include high-performance liquid chromatography with tandem mass spectrometric detection (HPLC/MS/MS) and immunoassays. Immunoassays may react with metabolites as well as parent compound. Therefore, assay results obtained with immunoassays may have a positive bias relative to results of HPLC/MS. The bias may depend upon the specific assay and laboratory. Comparison of the concentrations in published literature to patient concentrations using the current assays must be made with detailed knowledge of the assay methods and biological matrices employed. Whole blood is the matrix of choice and specimens should be collected into tubes containing ethylene diamine tetraacetic acid (EDTA) anticoagulant. Heparin anticoagulation is not recommended because of the tendency to form clots on storage. Samples which are not analyzed immediately should be stored at room temperature or in a refrigerator and assayed within 7 days; see assay instructions for specifics. If samples are to be kept longer, they should be deep frozen at -20°C. One study showed drug recovery > 90% for samples stored at -20°C for 6 months, with reduced recovery observed after 6 months.
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. In addition, the clinical trials were not designed to establish comparative differences across study arms with regards to the adverse reactions discussed below.
17.3 Increased Risk of Infection
Inform patients they are at increased risk of developing a variety of infections, including opportunistic infections, due to immunosuppression and to contact their physician if they develop any symptoms of infection such as fever, sweats or chills, cough or flu-like symptoms, muscle aches, or warm, red, painful areas on the skin [see Warnings and Precautions ( 5.2)] .
5.1 Lymphoma and Other Malignancies
Patients receiving immunosuppressants, including Tacrolimus Injection, are at increased risk of developing lymphomas and other malignancies, particularly of the skin. The risk appears to be related to the intensity and duration of immunosuppression rather than to the use of any specific agent.
As usual for patients with increased risk for skin cancer, examine patients for skin changes; exposure to sunlight and UV light should be limited by wearing protective clothing and using a broad-spectrum sunscreen with a high protection factor.
Post-transplant lymphoproliferative disorder (PTLD) has been reported in immunosuppressed organ transplant recipients. The majority of PTLD events appear related to Epstein-Barr Virus (EBV) infection. The risk of PTLD appears greatest in those individuals who are EBV seronegative, a population which includes many young children. Monitor EBV serology during treatment.
5.4 New Onset Diabetes After Transplant
Tacrolimus was shown to cause new onset diabetes mellitus in clinical trials of kidney, liver, and heart transplantation. New onset diabetes after transplantation may be reversible in some patients. African-American and Hispanic kidney transplant patients are at an increased risk. Blood glucose concentrations should be monitored closely in patients using tacrolimus [see Adverse Reactions ( 6.1)] .
17.4 New Onset Diabetes After Transplant
Inform patients that tacrolimus can cause diabetes mellitus and should be advised to contact their physician if they develop frequent urination, increased thirst, or hunger [see Warnings and Precautions ( 5.4)] .
7.2 Effects of Other Drugs On Tacrolimus
Table 15displays the effects of other drugs on Tacrolimus.
|
1. Tacrolimus dosage adjustment recommendation based on observed effect of coadministered drug on tacrolimus exposures [see Clinical Pharmacology ( 12.3)] , literature reports of altered tacrolimus exposures, or the other drug's known CYP3A inhibitor/inducer status. |
||
|
2. High dose or double strength grapefruit juice is a strongCYP3A inhibitor; low dose or single strength grapefruit juice is a moderate |
||
|
3. CYP3A inhibitor.Strong CYP3A inhibitor/inducer, based on reported effect on exposures to tacrolimus along with supporting in vitroCYP3A inhibitor/inducer data, or based on drug-drug interaction studies with midazolam (sensitive CYP3A probe substrate). |
||
| Drug/Substance Class or Name | Drug Interaction Effect | Recommendations |
| Grapefruit or grapefruit juice 2 | May increase tacrolimus whole blood trough concentrations and increase the risk of serious adverse reactions (e.g., neurotoxicity, QT prolongation)
[see Warnings and Precautions (
5.6,
5.11,
5.12)] . |
Avoid grapefruit or grapefruit juice. |
| Strong CYP3A Inducers
3:
Antimycobacterials (e.g., rifampin, rifabutin), anticonvulsants (e.g., phenytoin, carbamazepine and phenobarbital), St John's Wort |
May decrease tacrolimus whole blood trough concentrations and increase the risk of rejection [see Warnings and Precautions ( 5.11)] . | Increase tacrolimus dose and monitor tacrolimus whole blood trough concentrations
[see Dosage and Administration (
2.2,
2.6) and Clinical Pharmacology
( 12.3)] . |
| Strong CYP3A Inhibitors
3:
Protease inhibitors (e.g, nelfinavir, telaprevir, boceprevir, ritonavir), azole antifungals (e.g., voriconazole, posaconazole, itraconazole, ketoconazole), antibiotics (e.g., clarithromycin, troleandomycin, chloramphenicol), nefazodone, letermovir, Schisandra sphenantheraextracts |
May increase tacrolimus whole blood trough concentrations and increase the risk of serious adverse reactions (e.g., neurotoxicity, QT prolongation). A rapid, sharp rise in tacrolimus levels may occur early, despite an immediate reduction of tacrolimus dose
[see Warnings and Precautions (
5.6,
5.11,
5.12)] . |
Reduce tacrolimus dose (for voriconazole and posaconazole, give one-third of the original dose) and adjust dose based on tacrolimus whole blood trough concentrations
[see Dosage and Administration (
2.2,
2.6) and
Clinical Pharmacology ( 12.3)] . Early and frequent monitoring of tacrolimus whole blood trough levels should start within 1-3 days and continue monitoring as necessary [see Warnings and Precautions ( 5.11)]. |
| Mild or Moderate CYP3A Inhibitors: Clotrimazole, antibiotics (e.g., erythromycin, fluconazole), calcium channel blockers (e.g., verapamil, diltiazem, nifedipine, nicardipine), amiodarone, danazol, ethinyl estradiol, cimetidine, lansoprazole and
omeprazole |
May increase tacrolimus whole blood trough concentrations and increase the risk of serious adverse reactions (e.g., neurotoxicity, QT prolongation)
[see Warnings and Precautions (
5.6,
5.11,
5.12)] . |
Monitor tacrolimus whole blood trough concentrations and reduce Tacrolimus dose if needed [see Dosage and Administration ( 2.2, 2.6) and Clinical Pharmacology ( 12.3)] . |
| Other drugs, such as:
Magnesium and aluminum hydroxide antacids Metoclopramide |
May increase tacrolimus whole blood trough concentrations and increase the risk of serious adverse reactions (e.g., neurotoxicity, QT prolongation)
[see Warnings and Precautions (
5.6,
5.11,
5.12)] . |
Monitor tacrolimus whole blood trough concentrations and reduce Tacrolimus dose if needed [see Dosage and Administration ( 2.2, 2.6) and Clinical Pharmacology ( 12.3)] . |
| Mild or Moderate CYP3A Inducers Methylprednisolone, prednisone | May decrease tacrolimus whole blood trough concentrations. | Monitor tacrolimus whole blood trough concentrations and adjust Tacrolimus dose if needed [see Dosage and Administration ( 2.2, 2.6)] . |
| Caspofungin | May decrease tacrolimus whole blood trough concentrations. | Monitor tacrolimus whole blood trough concentrations and adjust tacrolimus dose if needed [see Dosage and Administration ( 2.2, 2.6)] . |
2.1 Important Administration Instructions
Tacrolimus should not be used without supervision by a physician with experience in immunosuppressive therapy.
17.11 Pregnancy, Lactation and Infertility
Inform women of childbearing potential that tacrolimus can harm the fetus. Instruct male and female patients to discuss with their healthcare provider family planning options including appropriate contraception. Also, discuss with pregnant patients the risks and benefits of breastfeeding their infant [see Use in Specific Populations ( 8.1, 8.2, 8.3)].
Encourage female transplant patients who become pregnant and male patients who have fathered a pregnancy, exposed to immunosuppressants including tacrolimus, to enroll in the voluntary Transplantation Pregnancy Registry International. To enroll or register, patients can call the toll free number 1-877-955-6877 or https://www.transplantpregnancyregistry.org/ [see Use in Specific Populations ( 8.1)] .
Based on animal studies, tacrolimus may affect fertility in males and females [see Nonclinical Toxicology ( 13.1)].
5.10 Not Recommended for Use With Sirolimus
Tacrolimus is not recommended for use with sirolimus:
- The use of sirolimus with Tacrolimus in studies of de novo liver transplant patients was associated with an excess mortality, graft loss, and hepatic artery thrombosis (HAT), and is not recommended.
- The use of sirolimus (2 mg per day) with Tacrolimus in heart transplant patients in a U.S. trial was associated with increased risk of renal function impairment, wound healing complications, and insulin-dependent post-transplant diabetes mellitus, and is not recommended [see Clinical Studies ( 14.3)].
- The use of sirolimus with Tacrolimus may increase the risk of thrombotic microangiopathy [see Warnings and Precautions ( 5.16)].
Warning: Malignancies and Serious Infections
17.2 Development of Lymphoma and Other Malignancies
Inform patients they are at increased risk of developing lymphomas and other malignancies, particularly of the skin, due to immunosuppression. Advise patients to limit exposure to sunlight and ultraviolet (UV) light by wearing protective clothing and using a broad spectrum sunscreen with a high protection factor [see Warnings and Precautions ( 5.1)] .
5.9 Anaphylactic Reactions With Tacrolimus Injection
Anaphylactic reactions have occurred with injectables containing castor oil derivatives, including tacrolimus injection, in a small percentage of patients (0.6%). The exact cause of these reactions is not known. Tacrolimus injection should be reserved for patients who are unable to take tacrolimus orally. Monitor patients for anaphylaxis when using the intravenous route of administration [see Dosage and Administration ( 2.1)].
5.11 Interactions With Cyp3a4 Inhibitors and Inducers
When co-administering tacrolimus with strong CYP3A4 inhibitors (e.g., telaprevir, boceprevir, ritonavir, ketoconazole, itraconazole, voriconazole, clarithromycin) and strong inducers (e.g., rifampin, rifabutin), adjustments in the dosing regimen of tacrolimus and subsequent frequent monitoring of tacrolimus whole blood trough concentrations and tacrolimus-associated adverse reactions are recommended. A rapid, sharp rise in tacrolimus levels has been reported after co-administration with a strong CYP3A4 inhibitor, clarithromycin, despite an initial reduction of tacrolimus dose. Early and frequent monitoring of tacrolimus whole blood trough levels is recommended [see Drug Interactions ( 7.2)] .
2.4 Dosage Modification for Patients With Renal Impairment
Due to its potential for nephrotoxicity, consider dosing tacrolimus injection at the lower end of the therapeutic dosing range in patients who have received a liver or heart transplant and have pre-existing renal impairment. Further reductions in dose below the targeted range may be required.
In kidney transplant patients with post-operative oliguria, the initial dose of tacrolimus should be administered no sooner than 6 hours and within 24 hours of transplantation, but may be delayed until renal function shows evidence of recovery [see Dosage and Administration ( 2.2), Warnings and Precautions ( 5.5), Use in Specific Populations ( 8.6), and Clinical Pharmacology ( 12.3)] .
5.5 Nephrotoxicity Due to Tacrolimus and Drug Interactions
Tacrolimus, like other calcineurin inhibitors, can cause acute or chronic nephrotoxicity in transplant patients due to its vasoconstrictive effect on renal vasculature, toxic tubulopathy and tubular-interstitial effects. Nephrotoxicity was reported in clinical trials [see Adverse Reactions ( 6.1)].
Acute renal impairment associated with tacrolimus toxicity can result in high serum creatinine, hyperkalemia, decreased secretion of urea and hyperuricemia, and is usually reversible. In patients with elevated serum creatinine and tacrolimus whole blood trough concentrations greater than the recommended range, consider dosage reduction or temporary interruption of tacrolimus administration.
The risk for nephrotoxicity may increase when Tacrolimus Injection is concomitantly administered with CYP3A inhibitors (by increasing tacrolimus whole blood concentrations) or drugs associated with nephrotoxicity (e.g., aminoglycosides, ganciclovir, amphotericin B, cisplatin, nucleotide reverse transcriptase inhibitors, protease inhibitors) When tacrolimus is used concurrently with other known nephrotoxic drugs, monitor renal function and tacrolimus blood concentrations, and adjust doses of both tacrolimus and/or concomitant medications during concurrent use [see Drug Interactions ( 7.2)] .
2.5 Dosage Modification for Patients With Hepatic Impairment
Due to the reduced clearance and prolonged half-life, patients with severe hepatic impairment (Child-Pugh ≥ 10) may require lower doses of tacrolimus. Close monitoring of blood concentrations is warranted. The use of tacrolimus in liver transplant recipients experiencing post-transplant hepatic impairment may be associated with increased risk of developing renal insufficiency related to high whole blood concentrations of tacrolimus. These patients should be monitored closely, and dosage adjustments should be considered. Some evidence suggests that lower doses should be used in these patients [see Dosage and Administration ( 2.2), Warnings and Precautions ( 5.5), Use in Specific Populations ( 8.7), and Clinical Pharmacology ( 12.3)].
1.1 Prophylaxis of Organ Rejection in Kidney, Liver Or Heart Transplant
Tacrolimus is indicated for the prophylaxis of organ rejection, in adult patients receiving allogeneic kidney transplant [see Clinical Studies ( 14.1)], liver transplants [see Clinical Studies ( 14.2)] and heart transplant [see Clinical Studies ( 14.3)], and pediatric patients receiving allogeneic liver transplants [see Clinical Studies ( 14.2)] in combination with other immunosuppressants.
Additional pediatric use information is approved for Astellas Pharma US, Inc.'s Prograf ® (tacrolimus) products. However, due to Astellas Pharma US, Inc.'s marketing exclusivity rights, this drug product is not labeled with that information.
2.7 Preparation and Administration Instructions of Tacrolimus Injection for Pharmacists
Tacrolimus can cause fetal harm. Follow applicable special handling and disposal procedures 1 [see How Supplied/ Storage and Handling ( 16.4)] .
Tacrolimus injection must be diluted with 0.9% Sodium Chloride Injection or 5% Dextrose Injection to a concentration between 0.004 mg/mL and 0.02 mg/mL prior to use. Diluted infusion solution should be stored in glass or polyethylene containers and should be discarded after 24 hours. The diluted infusion solution should not be stored in a polyvinyl chloride (PVC) container due to decreased stability and the potential for extraction of phthalates. In situations where more dilute solutions are utilized (e.g., pediatric dosing, etc.), PVC-free tubing should likewise be used to minimize the potential for significant drug adsorption onto the tubing.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Due to the chemical instability of tacrolimus in alkaline media, Tacrolimus injection should not be mixed or co-infused with solutions of pH 9 or greater (e.g., ganciclovir or acyclovir).
5.16 Thrombotic Microangiopathy (including Hemolytic Uremic Syndrome and Thrombotic Thrombocytopenic Purpura)
Cases of thrombotic microangiopathy (TMA), including hemolytic uremic syndrome (HUS) and thrombotic thrombocytopenic purpura (TTP), have been reported in patients treated with tacrolimus. TMA may have a multifactorial etiology. Risk factors for TMA that can occur in transplant patients include, for example, severe infections, graft-versus-host disease (GVHD), Human Leukocyte Antigen (HLA) mismatch, the use of calcineurin inhibitors and mammalian target of rapamycin (mTOR) inhibitors. These risk factors may, either alone or combined, contribute to the risk of TMA.
In patients with signs and symptoms of TMA, consider tacrolimus as a risk factor. Concurrent use of tacrolimus and mTOR inhibitors may contribute to the risk of TMA.
Structured Label Content
Section 42229-5 (42229-5)
Intravenous Formulation - Administration Precautions due to Risk of Anaphylaxis
Intravenous use is recommended for patients who cannot tolerate oral formulations, and conversion from intravenous to oral tacrolimus is recommended as soon as oral therapy can be tolerated to minimize the risk of anaphylactic reactions that occurred with injectables containing castor oil derivatives [see Warnings and Precautions ( 5.9)] .
Patients receiving tacrolimus injection should be under continuous observation for at least the first 30 minutes following the start of the infusion and at frequent intervals thereafter. If signs or symptoms of anaphylaxis occur, the infusion should be stopped. An aqueous solution of epinephrine should be available at the bedside as well as a source of oxygen.
Section 44425-7 (44425-7)
Store and Dispense
Store between 5°C and 25°C (41°F and 77°F).
Section 51945-4 (51945-4)
Principal Display Panel - 1 mL Carton Label
NDC 14789-135-05
Rx Only
Tacrolimus
Injection
5 mg/1 mL
(For intravenous use)
For Intravenous Infusion Only After Dilution
10 x 1 mL Single-dose Vials
NEXUS
PHARMACEUTICALS
10 Overdosage (10 OVERDOSAGE)
Limited overdosage experience is available. Acute overdosages of up to 30 times the intended dose have been reported. Almost all cases have been asymptomatic and all patients recovered with no sequelae. Acute overdosage was sometimes followed by adverse reactions consistent with those reported with the use of Tacrolimus [see Adverse Reactions ( 6.1, 6.2)] , including tremors, abnormal renal function, hypertension, and peripheral edema; in one case of acute overdosage, transient urticaria and lethargy were observed. Based on the poor aqueous solubility and extensive erythrocyte and plasma protein binding, it is anticipated that tacrolimus is not dialyzable to any significant extent; there is no experience with charcoal hemoperfusion. The oral use of activated charcoal has been reported in treating acute overdoses, but experience has not been sufficient to warrant recommending its use. General supportive measures and treatment of specific symptoms should be followed in all cases of overdosage.
15 References (15 REFERENCES)
1. “OSHA Hazardous Drugs.” OSHA. http://www.osha.gov/SLTC/hazardousdrugs/index.html
11 Description (11 DESCRIPTION)
Tacrolimus, previously known as FK506, is the active ingredient in Tacrolimus. Tacrolimus is a calcineurin-inhibitor immunosuppressant produced by Streptomyces tsukubaensis. Chemically, tacrolimus is designated as [3 S- [3 R*[ E(1 S*,3 S*,4 S*)], 4 S*,5 R*,8 S*,9 E,12 R*,14 R*,15 S*,16 R*,18 S*,19 S*,26a R*]] -
5,6,8,11,12,13,14,15,16,17,18,19,24,25,26,26a-hexadecahydro-5,19-dihydroxy-3-[2-(4-hydroxy-3-methoxycyclohexyl)-1- methylethenyl]-14,16-dimethoxy-4,10,12,18-tetramethyl-8-(2-propenyl)-15,19-epoxy-3H-pyrido[2,1- c][1,4] oxaazacyclotricosine-1,7,20,21(4H,23H)-tetrone, monohydrate.
The chemical structure of tacrolimus is:
Tacrolimus has an empirical formula of C 44H 69NO 12•H 2O and a formula weight of 822.03. Tacrolimus appears as white crystals or crystalline powder. It is practically insoluble in water, freely soluble in ethanol, and very soluble in methanol and chloroform.
Tacrolimus injection is a sterile solution containing the equivalent of 5 mg anhydrous tacrolimus USP in 1 mL for administration by intravenous infusion only. Each mL contains the following inactive ingredients: dehydrated alcohol USP, 80.0% v/v and polyoxyl 60 hydrogenated castor oil (HCO-60), 200 mg. Tacrolimus injection must be diluted with 0.9% Sodium Chloride Injection or 5% Dextrose Injection before use.
5.7 Hyperkalemia
Hyperkalemia has been reported with tacrolimus use. Serum potassium levels should be monitored. Careful consideration should be given prior to use of other agents also associated with hyperkalemia (e.g., potassium-sparing diuretics, ACE inhibitors, angiotensin receptor blockers) during Tacrolimus therapy [see Adverse Reactions ( 6.1)] . Monitor serum potassium levels periodically during treatment.
5.8 Hypertension
Hypertension is a common adverse effect of tacrolimus therapy and may require antihypertensive therapy [see Adverse Reactions ( 6.1)]. The control of blood pressure can be accomplished with any of the common antihypertensive agents, though careful consideration should be given prior to use of antihypertensive agents associated with hyperkalemia (e.g., potassium-sparing diuretics, ACE inhibitors, angiotensin receptor blockers) [see Warnings and Precautions ( 5.7)] .Calcium-channel blocking agents may increase tacrolimus blood concentrations and therefore require dosage reduction of Tacrolimus [see Drug Interactions ( 7.2)].
17.7 Hyperkalemia
Inform patients that tacrolimus injection can cause hyperkalemia. Monitoring of potassium levels may be necessary, especially with concomitant use of other drugs known to cause hyperkalemia [see Warnings and Precautions ( 5.7)] .
17.8 Hypertension
Inform patients that tacrolimus can cause high blood pressure which may require treatment with antihypertensive therapy. Advise patients to monitor their blood pressure [see Warnings and Precautions ( 5.8)] .
5.6 Neurotoxicity
Tacrolimus may cause a spectrum of neurotoxicities. The most severe neurotoxicities include posterior reversible encephalopathy syndrome (PRES), delirium, seizure and coma; others include tremors, paresthesias, headache, mental status changes, and changes in motor and sensory functions [see Adverse Reactions ( 6.1, 6.2)] . As symptoms may be associated with tacrolimus whole blood trough concentrations at or above the recommended range, monitor for neurologic symptoms and consider dosage reduction or discontinuation of Tacrolimus Injection if neurotoxicity occurs.
8.4 Pediatric Use
Safety and effectiveness have been established in pediatric liver transplant patients.
8.5 Geriatric Use
Clinical trials of tacrolimus did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
17.6 Neurotoxicity
Inform patients that they are at risk of developing adverse neurologic reactions including seizure, altered mental status, and tremor. Advise patients to contact their physician should they develop vision changes, delirium, or tremors [see Warnings and Precautions ( 5.6)] .
5.14 Immunizations
Whenever possible, administer the complete complement of vaccines before transplantation and treatment with tacrolimus.
The use of live vaccines should be avoided during treatment with tacrolimus; examples include (not limited to) the following: intranasal influenza, measles, mumps, rubella, oral polio, BCG, yellow fever, varicella, and TY21a typhoid vaccines.
Inactivated vaccines noted to be safe for administration after transplantation may not be sufficiently immunogenic during treatment with tacrolimus.
17.1 Administration
Advise the patient or caregiver to:
- Not to eat grapefruit or drink grapefruit juice in combination with tacrolimus [see Drug Interactions ( 7.2)].
17.13 Immunizations
Inform patients that Tacrolimus can interfere with the usual response to immunizations and that they should avoid live vaccines . [see Warnings and Precautions ( 5.14)] .
Manufactured in the USA for:
Nexus Pharmaceuticals, LLC
Lincolnshire, IL 60069 USA
NEXUS
PHARMACEUTICALS
TACPI01USR01
Revised: 2/2025
17.5 Nephrotoxicity
Inform patients that tacrolimus injection can have toxic effects on the kidney that should be monitored. Advise patients to attend all visits and complete all blood tests ordered by their medical team [see Warnings and Precautions ( 5.5)] .
4 Contraindications (4 CONTRAINDICATIONS)
Tacrolimus injection is contraindicated in patients with a hypersensitivity to tacrolimus. Tacrolimus injection is contraindicated in patients with a hypersensitivity to HCO-60 (polyoxyl 60 hydrogenated castor oil). Hypersensitivity symptoms reported include dyspnea, rash, pruritus, and acute respiratory distress syndrome [see Adverse Reactions ( 6)] .
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious and otherwise important adverse drug reactions are discussed in greater detail in other sections of labeling:
- Lymphoma and Other Malignancies [see Warnings and Precautions ( 5.1)]
- Serious Infections [see Warnings and Precautions ( 5.2)]
- New Onset Diabetes After Transplant [see Warnings and Precautions ( 5.4)]
- Nephrotoxicity [see Warnings and Precautions ( 5.5)]
- Neurotoxicity [see Warnings and Precautions ( 5.6)]
- Hyperkalemia [see Warnings and Precautions ( 5.7)]
- Hypertension [see Warnings and Precautions ( 5.8)]
- Anaphylactic Reactions with Tacrolimus Injection [see Warnings and Precautions ( 5.9)]
- Myocardial Hypertrophy [see Warnings and Precautions ( 5.13)]
- Pure Red Cell Aplasia [see Warnings and Precautions ( 5.15)]
- Thrombotic Microangiopathy, Including Hemolytic Uremic Syndrome and Thrombotic Thrombocytopenic Purpura [see Warnings and Precautions ( 5.16)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- Mycophenolic Acid Products: Can increase MPA exposure after crossover from cyclosporine to tacrolimus; monitor for MPA-related adverse reactions and adjust MMF or MPA dose as needed. ( 7.1)
- Nelfinavir and Grapefruit Juice: Increased tacrolimus concentrations via CYP3A inhibition; avoid concomitant use. ( 7.2)
- CYP3A Inhibitors: Increased tacrolimus concentrations; monitor concentrations and adjust tacrolimus dose as needed. ( 5.11, 7.2)
- CYP3A4 Inducers: Decreased tacrolimus concentrations; monitor concentrations and adjust tacrolimus dose as needed. ( 5.11, 7.2)
5.12 Qt Prolongation (5.12 QT Prolongation)
Tacrolimus injection may prolong the QT/QTc interval and may cause Torsade de pointes. Avoid Tacrolimus Injection in patients with congenital long QT syndrome. In patients with congestive heart failure, bradyarrhythmias, those taking certain antiarrhythmic medications or other medicinal products that lead to QT prolongation, and those with electrolyte disturbances such as hypokalemia, hypocalcemia, or hypomagnesemia, consider obtaining electrocardiograms and monitoring electrolytes (magnesium, potassium, calcium) periodically during treatment.
When co-administering tacrolimus with other substrates and/or inhibitors of CYP3A4 that also have the potential to prolong the QT interval, a reduction in tacrolimus dose, frequent monitoring of tacrolimus whole blood concentrations, and monitoring for QT prolongation is recommended. Use of tacrolimus with amiodarone has been reported to result in increased tacrolimus whole blood concentrations with or without concurrent QT prolongation [see Drug Interactions ( 7.2)] .
8.6 Renal Impairment
The pharmacokinetics of tacrolimus in patients with renal impairment was similar to that in healthy volunteers with normal renal function. However, consideration should be given to dosing tacrolimus at the lower end of the therapeutic dosing range in patients who have received a liver or heart transplant and have pre-existing renal impairment. Further reductions in dose below the targeted range may be required [see Dosage and Administration ( 2.4) and Clinical Pharmacology ( 12.3)] .
12.3 Pharmacokinetics
Tacrolimus activity is primarily due to the parent drug. The pharmacokinetic parameters (mean ± S.D.) of tacrolimus have been determined following intravenous (IV) and/or oral (PO) administration in healthy volunteers, and in kidney transplant, liver transplant, and heart transplant patients ( Table 17).
|
1. Not applicable |
||||||||
|
2. AUC 0-inf |
||||||||
|
3. Not available |
||||||||
|
4. AUC 0-t |
||||||||
|
5. Determined after the first dose |
||||||||
|
6. Median [range] |
||||||||
|
7.AUC 0-12 |
||||||||
| Population |
N
|
Route (Dose) | Parameters | |||||
| C max(nanogram/mL) | T max(hr) | AUC (nanogram•hr/mL) | t 1/2(hr) | CL (L/hr/kg) | V (L/kg) | |||
| Healthy Volunteers | 8 | Intravenous (0.025 mg/kg/4 hr) | 1 | 1 | 652 2± 156 | 34.2 ± 7.7 | 0.040 ± 0.009 | 1.91 ± 0.31 |
| 16 | PO (5 mg) (capsules) | 28.8 ± 8.9 | 1.5 ± 0.7 | 266 2± 95 | 32.3 ± 8.8 | 3 | 3 | |
| Kidney Transplant Patients | 26 | Intravenous (0.02 mg/kg/12 hr) | 1 | 1 | 294 2± 262 | 18.8 ± 16.7 | 0.083 ± 0.050 | 1.41 ± 0.66 |
| PO (0.2 mg/kg/day) | 19.2 ± 10.3 | 3.0 | 203 2± 42 | 3 | 3 | 3 | ||
| PO (0.3 mg/kg/day) | 24.2 ± 15.8 | 1.5 | 288 2± 93 | 3 | 3 | 3 | ||
| Liver Transplant Patients | 17 | Intravenous (0.05 mg/kg/12 hr) | 1 | 1 | 3300 2±2130 | 11.7 ±3.9 | 0.053 ±0.017 | 0.85 ±0.30 |
| PO (0.3 mg/kg/day) | 68.5 ± 30.0 | 2.3 ± 1.5 | 519 2± 179 | 3 | 3 | 3 | ||
|
Heart Transplant Patients |
11 | Intravenous (0.01 mg/kg/day as a continuous infusion) | 1 | 1 | 954 4± 334 | 23.6 ± 9.22 | 0.051 ± 0.015 | 3 |
| 11 | PO (0.075 mg/kg/day) 5 | 14.7 ± 7.79 | 2.1
[0.5-6.0] 6 |
82.7 7± 63.2 | 1 | 3 | 3 | |
| 14 | PO (0.15 mg/kg/day) 5 | 24.5 ± 13.7 | 1.5
[0.4-4.0] 6 |
142 7± 116 | 1 | 3 | 3 |
Due to intersubject variability in tacrolimus pharmacokinetics, individualization of the dosing regimen is necessary for optimal therapy [see Dosage and Administration ( 2.6)] . Pharmacokinetic data indicate that whole blood concentrations rather than plasma concentrations serve as the more appropriate sampling compartment to describe tacrolimus pharmacokinetics.
7.1 Mycophenolic Acid
When tacrolimus is prescribed with a given dose of a mycophenolic acid (MPA) product, exposure to MPA is higher with tacrolimus co-administration than with cyclosporine co-administration with MPA, because cyclosporine interrupts the enterohepatic recirculation of MPA while tacrolimus does not. Monitor for MPA-associated adverse reactions and reduce the dose of concomitantly administered mycophenolic acid products as needed.
8.8 Race Or Ethnicity (8.8 Race or Ethnicity)
African-American patients may need to be titrated to higher dosages to attain comparable trough concentrations compared to Caucasian patients [see Dosage and Administration ( 2.2) and Clinical Pharmacology ( 12.3)].
African-American and Hispanic patients are at increased risk for new onset diabetes after transplant. Monitor blood glucose concentrations and treat appropriately [see Warnings and Precautions ( 5.4)].
5.2 Serious Infections
Patients receiving immunosuppressants, including tacrolimus, are at increased risk of developing bacterial, viral, fungal, and protozoal infections, including opportunistic infections. These infections may lead to serious, including fatal, outcomes. Serious viral infections reported include:
- Polyomavirus-associated nephropathy (PVAN), mostly due to BK virus infection
- JC virus-associated progressive multifocal leukoencephalopathy (PML)
- Cytomegalovirus infections: CMV seronegative transplant patients who receive an organ from a CMV seropositive donor disease are at higher risk of developing CMV viremia and CMV disease.
Monitor for the development of infection and adjust the immunosuppressive regimen to balance the risk of rejection with the risk of infection [see Adverse Reactions ( 6.1, 6.2)].
8.7 Hepatic Impairment
The mean clearance of tacrolimus was substantially lower in patients with severe hepatic impairment (mean Child-Pugh score: > 10) compared to healthy volunteers with normal hepatic function. Close monitoring of tacrolimus trough concentrations is warranted in patients with hepatic impairment [see Clinical Pharmacology ( 12.3)] .
The use of Tacrolimus in liver transplant recipients experiencing post-transplant hepatic impairment may be associated with increased risk of developing renal insufficiency related to high whole blood trough concentrations of tacrolimus. These patients should be monitored closely and dosage adjustments should be considered. Some evidence suggests that lower doses should be used in these patients [see Dosage and Administration ( 2.5) and Clinical Pharmacology ( 12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Tacrolimus is a is a calcineurin-inhibitor immunosuppressant indicated for the prophylaxis of organ rejection in adult patients receiving allogeneic liver, kidney or heart transplants, and pediatric patients receiving allogeneic liver transplants in combination with other immunosuppressants. ( 1.1)
17.10 Drug Interactions
Instruct patients to tell their healthcare providers when they start or stop taking any medicines, including prescription medicines and nonprescription medicines, natural or herbal remedies, nutritional supplements, and vitamins. Advise patients to avoid grapefruit and grapefruit juice [see Drug Interactions ( 7)].
12.1 Mechanism of Action
Tacrolimus binds to an intracellular protein, FKBP-12. A complex of tacrolimus-FKBP-12, calcium, calmodulin, and calcineurin (a ubiquitous mammalian intracellular enzyme) is then formed, after which the phosphatase activity of calcineurin is inhibited. Such inhibition prevents the dephosphorylation and translocation of various factors such as the nuclear factor of activated T-cells (NF-AT), and nuclear factor kappa-light-chain enhancer of activated B-cells (NF-ϰB).
Tacrolimus inhibits the expression and/or production of several cytokines that include interleukin (IL)-1 beta, IL-2, IL-3, IL-4, IL-5, IL-6, IL-8, IL-10, gamma interferon, tumor necrosis factor-alpha, and granulocyte macrophage colony-stimulating factor. Tacrolimus also inhibits IL-2 receptor expression and nitric oxide release, induces apoptosis and production of transforming growth factor beta that can lead to immunosuppressive activity. The net result is the inhibition of T-lymphocyte activation and proliferation, as well as T-helper-cell-dependent B-cell response (i.e., immunosuppression).
16.2 Tacrolimus Injection
(for Intravenous infusion only)
| Unit of Sale | Concentration | Each |
| NDC 14789-135-05
Carton containing 10 vials |
5 mg/mL | NDC 14789-135-07
1 mL Single use vial |
5 mg/mL (equivalent of 5 mg of anhydrous tacrolimus USP per mL) supplied as a sterile solution in a 1 mL vial, in a carton of 10 vials.
14.2 Liver Transplantation
The safety and efficacy of tacrolimus-based immunosuppression following orthotopic liver transplantation were assessed in two prospective, randomized, non-blinded multicenter trials. The active control groups were treated with a cyclosporine-based immunosuppressive regimen (CsA/AZA). Both trials used concomitant adrenal corticosteroids as part of the immunosuppressive regimens. These trials compared patient and graft survival rates at 12 months following transplantation.
In one trial, 529 patients were enrolled at 12 clinical sites in the United States; prior to surgery, 263 were randomized to the tacrolimus-based immunosuppressive regimen and 266 to the CsA/AZA. In 10 of the 12 sites, the same CsA/AZA protocol was used, while 2 sites used different control protocols. This trial excluded patients with renal dysfunction, fulminant hepatic failure with Stage IV encephalopathy, and cancers; pediatric patients (≤ 12 years old) were allowed.
In the second trial, 545 patients were enrolled at 8 clinical sites in Europe; prior to surgery, 270 were randomized to the tacrolimus-based immunosuppressive regimen and 275 to CsA/AZA. In this trial, each center used its local standard CsA/AZA protocol in the active-control arm. This trial excluded pediatric patients, but did allow enrollment of subjects with renal dysfunction, fulminant hepatic failure in Stage IV encephalopathy, and cancers other than primary hepatic with metastases.
One-year patient survival and graft survival in the Tacrolimus-based treatment groups were similar to those in the CsA/AZA treatment groups in both trials. The overall 1-year patient survival (CsA/AZA and tacrolimus-based treatment groups combined) was 88% in the U.S. trial and 78% in the European trial. The overall 1-year graft survival (CsA/AZA and tacrolimus-based treatment groups combined) was 81% in the U.S. trial and 73% in the European trial. In both trials, the median time to convert from intravenous to oral tacrolimus dosing was 2 days.
Although there is a lack of direct correlation between tacrolimus concentrations and drug efficacy, data from clinical trials of liver transplant patients have shown an increasing incidence of adverse reactions with increasing trough blood concentrations. Most patients are stable when trough whole blood concentrations are maintained between 5 to 20 nanogram/mL. Long-term post-transplant patients are often maintained at the low end of this target range.
Data from the U.S. clinical trial show that the median trough blood concentrations, measured at intervals from the second week to one year post-transplantation, ranged from 9.8 ng/mL to 19.4 ng/mL.
14.3 Heart Transplantation
Two open-label, randomized, comparative trials evaluated the safety and efficacy of tacrolimus-based and cyclosporine-based immunosuppression in primary orthotopic heart transplantation. In a trial conducted in Europe, 314 patients received a regimen of antibody induction, corticosteroids, and azathioprine in combination with tacrolimus or cyclosporine modified for 18 months. In a 3-arm trial conducted in the U.S., 331 patients received corticosteroids and tacrolimus plus sirolimus, tacrolimus plus mycophenolate mofetil (MMF) or cyclosporine modified plus MMF for 1 year.
In the European trial, patient/graft survival at 18 months post-transplant was similar between treatment arms, 92% in the tacrolimus group and 90% in the cyclosporine group. In the U.S. trial, patient and graft survival at 12 months was similar with 93% survival in the tacrolimus plus MMF group and 86% survival in the cyclosporine modified plus MMF group. In the European trial, the cyclosporine trough concentrations were above the pre-defined target range (i.e., 100 to 200 nanogram/mL) at Day 122 and beyond in 32% to 68% of the patients in the cyclosporine treatment arm, whereas the tacrolimus trough concentrations were within the pre-defined target range (i.e., 5 to 15 nanogram/mL) in 74% to 86% of the patients in the tacrolimus treatment arm. Data from this European trial indicate that from 1 week to 3 months post-transplant, approximately 80% of patients maintained trough concentrations between 8 to 20 nanogram/mL and, from 3 months through 18 months post-transplant, approximately 80% of patients maintained trough concentrations between 6 to 18 nanogram/mL.
The U.S. trial contained a third arm of a combination regimen of sirolimus, 2 mg per day, and full-dose tacrolimus; however, this regimen was associated with increased risk of wound-healing complications, renal function impairment, and insulin-dependent post-transplant diabetes mellitus, and is not recommended [see Warnings and Precautions ( 5.10)].
16.4 Handling and Disposal
Tacrolimus can cause fetal harm. Wearing disposable gloves is recommended during dilution of the injection in the hospital and when wiping any spills. In case a spill occurs, wipe the surface with a wet paper towel. Follow applicable special handling and disposal procedures 1.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- New Onset Diabetes After Transplant: Monitor blood glucose. ( 5.4)
- Nephrotoxicity (acute and/or chronic): Reduce the dose; use caution with other nephrotoxic drugs. ( 5.5)
- Neurotoxicity: Including risk of Posterior Reversible Encephalopathy Syndrome (PRES); monitor for neurologic abnormalities; reduce or discontinue tacrolimus. ( 5.6)
- Hyperkalemia: Monitor serum potassium levels. Consider carefully before using with other agents also associated with hyperkalemia. ( 5.7)
- Hypertension: May require antihypertensive therapy. Monitor relevant drug-drug interactions. ( 5.8)
- Anaphylactic Reactions with intravenous formulation: Observe patients receiving tacrolimus injection for signs and symptoms of anaphylaxis. ( 5.9)
- Not recommended for use with sirolimus: Not recommended in liver and heart transplant due to increased risk of serious adverse reactions. ( 5.10)
- Myocardial Hypertrophy: Consider dose reduction/discontinuation. ( 5.13)
- Immunizations: Avoid live vaccines. ( 5.14)
- Pure Red Cell Aplasia: Consider discontinuation of tacrolimus injection. ( 5.15)
- Thrombotic Microangiopathy, Including Hemolytic Uremic Syndrome and Thrombotic Thrombocytopenic Purpura: May occur, especially in patients with infections and certain concomitant medications. ( 5.16)
5.15 Pure Red Cell Aplasia
Cases of pure red cell aplasia (PRCA) have been reported in patients treated with tacrolimus. A mechanism for tacrolimus-induced PRCA has not been elucidated. All patients reported risk factors for PRCA such as parvovirus B19 infection, underlying disease, or concomitant medications associated with PRCA. If PRCA is diagnosed, discontinuation of tacrolimus should be considered [see Adverse Reactions ( 6.2)] .
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Intravenous (IV) use recommended for patients who cannot tolerate oral formulations (capsules or suspension). ( 2.1, 2.2)
- Therapeutic drug monitoring is recommended. ( 2.1, 2.6)
- Avoid eating grapefruit or drinking grapefruit juice. ( 2.1)
- See dosage adjustments for African-American patients ( 2.2), hepatic and renal impaired. ( 2.4, 2.5)
- For complete dosing information, see Full Prescribing Information.
5.13 Myocardial Hypertrophy
Myocardial hypertrophy has been reported in infants, children, and adults, particularly those with high tacrolimus trough concentrations, and is generally manifested by echocardiographically demonstrated concentric increases in left ventricular posterior wall and interventricular septum thickness. This condition appears reversible in most cases following dose reduction or discontinuance of therapy. In patients who develop renal failure or clinical manifestations of ventricular dysfunction while receiving tacrolimus injection therapy, echocardiographic evaluation should be considered. If myocardial hypertrophy is diagnosed, dosage reduction or discontinuation of tacrolimus injection should be considered [see Adverse Reactions ( 6.2)] .
17.12 Myocardial Hypertrophy
Inform patients to report symptoms of tiredness, swelling, and/or shortness of breath (heart failure).
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
The vial contains 1 mL anhydrous tacrolimus USP for intravenous infusion as follows:
- 5 mg/mL, sterile solution
6.2 Postmarketing Experience
The following adverse reactions have been reported from worldwide marketing experience with tacrolimus. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Decisions to include these reactions in labeling are typically based on one or more of the following factors: (1) seriousness of the reaction, (2) frequency of the reporting, or (3) strength of causal connection to the drug.
Other reactions include:
- Cardiovascular: Atrial fibrillation, atrial flutter, cardiac arrhythmia, cardiac arrest, electrocardiogram T wave abnormal, flushing, myocardial infarction, myocardial ischemia, pericardial effusion, QT prolongation, Torsade de Pointes, venous thrombosis deep limb, ventricular extrasystoles, ventricular fibrillation, myocardial hypertrophy
- Gastrointestinal: Bile duct stenosis, colitis, enterocolitis, gastroenteritis, gastroesophageal reflux disease, hepatic cytolysis, hepatic necrosis, hepatotoxicity, impaired gastric emptying, liver fatty, mouth ulceration, pancreatitis hemorrhagic, pancreatitis necrotizing, stomach ulcer, veno-occlusive liver disease
- Hemic/Lymphatic: Agranulocytosis, disseminated intravascular coagulation, hemolytic anemia, neutropenia, febrile neutropenia, pancytopenia, thrombocytopenic purpura, thrombotic thrombocytopenic purpura, pure red cell aplasia, thrombotic microangiopathy
- Infections: Cases of progressive multifocal leukoencephalopathy (PML), sometimes fatal; polyoma virus- associated nephropathy (PVAN) including graft loss
- Metabolic/Nutritional: Glycosuria, increased amylase including pancreatitis, weight decreased
- Miscellaneous: Feeling hot and cold, feeling jittery, hot flushes, multi-organ failure, primary graft dysfunction
- Musculoskeletal and Connective Tissue Disorders: Pain in extremity including Calcineurin-Inhibitor Induced Pain Syndrome (CIPS)
- Nervous System: Carpal tunnel syndrome, cerebral infarction, hemiparesis, leukoencephalopathy, mental disorder, mutism, posterior reversible encephalopathy syndrome (PRES), progressive multifocal leukoencephalopathy (PML), quadriplegia, speech disorder, syncope
- Respiratory: Acute respiratory distress syndrome, interstitial lung disease, lung infiltration, respiratory distress, respiratory failure
- Skin: Stevens-Johnson syndrome, toxic epidermal necrolysis
- Special Senses: Blindness, optic neuropathy, blindness cortical, hearing loss including deafness, photophobia
- Urogenital: Acute renal failure, cystitis hemorrhagic, hemolytic-uremic syndrome
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Pregnancy: Can cause fetal harm. Advise pregnant women of the potential risk to the fetus. ( 8.1, 8.3)
Additional pediatric use information is approved for Astellas Pharma US, Inc.'s Prograf ® (tacrolimus) products. However, due to Astellas Pharma US, Inc.'s marketing exclusivity rights, this drug product is not labeled with that information.
17.9 Thrombotic Microangiopathy
Inform patients that tacrolimus can cause blood clotting problems. The risk of this occurring increases when patients take tacrolimus and sirolimus or everolimus concomitantly, or when patients develop certain infections. Advise them to seek medical attention promptly if they develop fever, petequiae or bruises, fatigue, confusion, jaundice, oliguria. [see Warnings and Precautions ( 5.16)]
2.6 Therapeutic Drug Monitoring
Monitoring of tacrolimus blood concentrations in conjunction with other laboratory and clinical parameters is considered an essential aid to patient management for the evaluation of rejection, toxicity, dose adjustments, and compliance.
Factors influencing frequency of monitoring include but are not limited to hepatic or renal dysfunction, the addition or discontinuation of potentially interacting drugs and the post-transplant time. Blood concentration monitoring is not a replacement for renal and liver function monitoring and tissue biopsies. Data from clinical trials show that tacrolimus whole blood concentrations were most variable during the first week post-transplantation.
The relative risks of toxicity and efficacy failure are related to tacrolimus whole blood trough concentrations. Therefore, monitoring of whole blood trough concentrations is recommended to assist in the clinical evaluation of toxicity and efficacy failure.
Methods commonly used for the assay of tacrolimus include high-performance liquid chromatography with tandem mass spectrometric detection (HPLC/MS/MS) and immunoassays. Immunoassays may react with metabolites as well as parent compound. Therefore, assay results obtained with immunoassays may have a positive bias relative to results of HPLC/MS. The bias may depend upon the specific assay and laboratory. Comparison of the concentrations in published literature to patient concentrations using the current assays must be made with detailed knowledge of the assay methods and biological matrices employed. Whole blood is the matrix of choice and specimens should be collected into tubes containing ethylene diamine tetraacetic acid (EDTA) anticoagulant. Heparin anticoagulation is not recommended because of the tendency to form clots on storage. Samples which are not analyzed immediately should be stored at room temperature or in a refrigerator and assayed within 7 days; see assay instructions for specifics. If samples are to be kept longer, they should be deep frozen at -20°C. One study showed drug recovery > 90% for samples stored at -20°C for 6 months, with reduced recovery observed after 6 months.
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. In addition, the clinical trials were not designed to establish comparative differences across study arms with regards to the adverse reactions discussed below.
17.3 Increased Risk of Infection
Inform patients they are at increased risk of developing a variety of infections, including opportunistic infections, due to immunosuppression and to contact their physician if they develop any symptoms of infection such as fever, sweats or chills, cough or flu-like symptoms, muscle aches, or warm, red, painful areas on the skin [see Warnings and Precautions ( 5.2)] .
5.1 Lymphoma and Other Malignancies
Patients receiving immunosuppressants, including Tacrolimus Injection, are at increased risk of developing lymphomas and other malignancies, particularly of the skin. The risk appears to be related to the intensity and duration of immunosuppression rather than to the use of any specific agent.
As usual for patients with increased risk for skin cancer, examine patients for skin changes; exposure to sunlight and UV light should be limited by wearing protective clothing and using a broad-spectrum sunscreen with a high protection factor.
Post-transplant lymphoproliferative disorder (PTLD) has been reported in immunosuppressed organ transplant recipients. The majority of PTLD events appear related to Epstein-Barr Virus (EBV) infection. The risk of PTLD appears greatest in those individuals who are EBV seronegative, a population which includes many young children. Monitor EBV serology during treatment.
5.4 New Onset Diabetes After Transplant
Tacrolimus was shown to cause new onset diabetes mellitus in clinical trials of kidney, liver, and heart transplantation. New onset diabetes after transplantation may be reversible in some patients. African-American and Hispanic kidney transplant patients are at an increased risk. Blood glucose concentrations should be monitored closely in patients using tacrolimus [see Adverse Reactions ( 6.1)] .
17.4 New Onset Diabetes After Transplant
Inform patients that tacrolimus can cause diabetes mellitus and should be advised to contact their physician if they develop frequent urination, increased thirst, or hunger [see Warnings and Precautions ( 5.4)] .
7.2 Effects of Other Drugs On Tacrolimus (7.2 Effects of Other Drugs on Tacrolimus)
Table 15displays the effects of other drugs on Tacrolimus.
|
1. Tacrolimus dosage adjustment recommendation based on observed effect of coadministered drug on tacrolimus exposures [see Clinical Pharmacology ( 12.3)] , literature reports of altered tacrolimus exposures, or the other drug's known CYP3A inhibitor/inducer status. |
||
|
2. High dose or double strength grapefruit juice is a strongCYP3A inhibitor; low dose or single strength grapefruit juice is a moderate |
||
|
3. CYP3A inhibitor.Strong CYP3A inhibitor/inducer, based on reported effect on exposures to tacrolimus along with supporting in vitroCYP3A inhibitor/inducer data, or based on drug-drug interaction studies with midazolam (sensitive CYP3A probe substrate). |
||
| Drug/Substance Class or Name | Drug Interaction Effect | Recommendations |
| Grapefruit or grapefruit juice 2 | May increase tacrolimus whole blood trough concentrations and increase the risk of serious adverse reactions (e.g., neurotoxicity, QT prolongation)
[see Warnings and Precautions (
5.6,
5.11,
5.12)] . |
Avoid grapefruit or grapefruit juice. |
| Strong CYP3A Inducers
3:
Antimycobacterials (e.g., rifampin, rifabutin), anticonvulsants (e.g., phenytoin, carbamazepine and phenobarbital), St John's Wort |
May decrease tacrolimus whole blood trough concentrations and increase the risk of rejection [see Warnings and Precautions ( 5.11)] . | Increase tacrolimus dose and monitor tacrolimus whole blood trough concentrations
[see Dosage and Administration (
2.2,
2.6) and Clinical Pharmacology
( 12.3)] . |
| Strong CYP3A Inhibitors
3:
Protease inhibitors (e.g, nelfinavir, telaprevir, boceprevir, ritonavir), azole antifungals (e.g., voriconazole, posaconazole, itraconazole, ketoconazole), antibiotics (e.g., clarithromycin, troleandomycin, chloramphenicol), nefazodone, letermovir, Schisandra sphenantheraextracts |
May increase tacrolimus whole blood trough concentrations and increase the risk of serious adverse reactions (e.g., neurotoxicity, QT prolongation). A rapid, sharp rise in tacrolimus levels may occur early, despite an immediate reduction of tacrolimus dose
[see Warnings and Precautions (
5.6,
5.11,
5.12)] . |
Reduce tacrolimus dose (for voriconazole and posaconazole, give one-third of the original dose) and adjust dose based on tacrolimus whole blood trough concentrations
[see Dosage and Administration (
2.2,
2.6) and
Clinical Pharmacology ( 12.3)] . Early and frequent monitoring of tacrolimus whole blood trough levels should start within 1-3 days and continue monitoring as necessary [see Warnings and Precautions ( 5.11)]. |
| Mild or Moderate CYP3A Inhibitors: Clotrimazole, antibiotics (e.g., erythromycin, fluconazole), calcium channel blockers (e.g., verapamil, diltiazem, nifedipine, nicardipine), amiodarone, danazol, ethinyl estradiol, cimetidine, lansoprazole and
omeprazole |
May increase tacrolimus whole blood trough concentrations and increase the risk of serious adverse reactions (e.g., neurotoxicity, QT prolongation)
[see Warnings and Precautions (
5.6,
5.11,
5.12)] . |
Monitor tacrolimus whole blood trough concentrations and reduce Tacrolimus dose if needed [see Dosage and Administration ( 2.2, 2.6) and Clinical Pharmacology ( 12.3)] . |
| Other drugs, such as:
Magnesium and aluminum hydroxide antacids Metoclopramide |
May increase tacrolimus whole blood trough concentrations and increase the risk of serious adverse reactions (e.g., neurotoxicity, QT prolongation)
[see Warnings and Precautions (
5.6,
5.11,
5.12)] . |
Monitor tacrolimus whole blood trough concentrations and reduce Tacrolimus dose if needed [see Dosage and Administration ( 2.2, 2.6) and Clinical Pharmacology ( 12.3)] . |
| Mild or Moderate CYP3A Inducers Methylprednisolone, prednisone | May decrease tacrolimus whole blood trough concentrations. | Monitor tacrolimus whole blood trough concentrations and adjust Tacrolimus dose if needed [see Dosage and Administration ( 2.2, 2.6)] . |
| Caspofungin | May decrease tacrolimus whole blood trough concentrations. | Monitor tacrolimus whole blood trough concentrations and adjust tacrolimus dose if needed [see Dosage and Administration ( 2.2, 2.6)] . |
2.1 Important Administration Instructions
Tacrolimus should not be used without supervision by a physician with experience in immunosuppressive therapy.
17.11 Pregnancy, Lactation and Infertility
Inform women of childbearing potential that tacrolimus can harm the fetus. Instruct male and female patients to discuss with their healthcare provider family planning options including appropriate contraception. Also, discuss with pregnant patients the risks and benefits of breastfeeding their infant [see Use in Specific Populations ( 8.1, 8.2, 8.3)].
Encourage female transplant patients who become pregnant and male patients who have fathered a pregnancy, exposed to immunosuppressants including tacrolimus, to enroll in the voluntary Transplantation Pregnancy Registry International. To enroll or register, patients can call the toll free number 1-877-955-6877 or https://www.transplantpregnancyregistry.org/ [see Use in Specific Populations ( 8.1)] .
Based on animal studies, tacrolimus may affect fertility in males and females [see Nonclinical Toxicology ( 13.1)].
5.10 Not Recommended for Use With Sirolimus (5.10 Not Recommended for Use with Sirolimus)
Tacrolimus is not recommended for use with sirolimus:
- The use of sirolimus with Tacrolimus in studies of de novo liver transplant patients was associated with an excess mortality, graft loss, and hepatic artery thrombosis (HAT), and is not recommended.
- The use of sirolimus (2 mg per day) with Tacrolimus in heart transplant patients in a U.S. trial was associated with increased risk of renal function impairment, wound healing complications, and insulin-dependent post-transplant diabetes mellitus, and is not recommended [see Clinical Studies ( 14.3)].
- The use of sirolimus with Tacrolimus may increase the risk of thrombotic microangiopathy [see Warnings and Precautions ( 5.16)].
Warning: Malignancies and Serious Infections (WARNING: MALIGNANCIES and SERIOUS INFECTIONS)
17.2 Development of Lymphoma and Other Malignancies
Inform patients they are at increased risk of developing lymphomas and other malignancies, particularly of the skin, due to immunosuppression. Advise patients to limit exposure to sunlight and ultraviolet (UV) light by wearing protective clothing and using a broad spectrum sunscreen with a high protection factor [see Warnings and Precautions ( 5.1)] .
5.9 Anaphylactic Reactions With Tacrolimus Injection (5.9 Anaphylactic Reactions with Tacrolimus Injection)
Anaphylactic reactions have occurred with injectables containing castor oil derivatives, including tacrolimus injection, in a small percentage of patients (0.6%). The exact cause of these reactions is not known. Tacrolimus injection should be reserved for patients who are unable to take tacrolimus orally. Monitor patients for anaphylaxis when using the intravenous route of administration [see Dosage and Administration ( 2.1)].
5.11 Interactions With Cyp3a4 Inhibitors and Inducers (5.11 Interactions with CYP3A4 Inhibitors and Inducers)
When co-administering tacrolimus with strong CYP3A4 inhibitors (e.g., telaprevir, boceprevir, ritonavir, ketoconazole, itraconazole, voriconazole, clarithromycin) and strong inducers (e.g., rifampin, rifabutin), adjustments in the dosing regimen of tacrolimus and subsequent frequent monitoring of tacrolimus whole blood trough concentrations and tacrolimus-associated adverse reactions are recommended. A rapid, sharp rise in tacrolimus levels has been reported after co-administration with a strong CYP3A4 inhibitor, clarithromycin, despite an initial reduction of tacrolimus dose. Early and frequent monitoring of tacrolimus whole blood trough levels is recommended [see Drug Interactions ( 7.2)] .
2.4 Dosage Modification for Patients With Renal Impairment (2.4 Dosage Modification for Patients with Renal Impairment)
Due to its potential for nephrotoxicity, consider dosing tacrolimus injection at the lower end of the therapeutic dosing range in patients who have received a liver or heart transplant and have pre-existing renal impairment. Further reductions in dose below the targeted range may be required.
In kidney transplant patients with post-operative oliguria, the initial dose of tacrolimus should be administered no sooner than 6 hours and within 24 hours of transplantation, but may be delayed until renal function shows evidence of recovery [see Dosage and Administration ( 2.2), Warnings and Precautions ( 5.5), Use in Specific Populations ( 8.6), and Clinical Pharmacology ( 12.3)] .
5.5 Nephrotoxicity Due to Tacrolimus and Drug Interactions (5.5 Nephrotoxicity due to Tacrolimus and Drug Interactions)
Tacrolimus, like other calcineurin inhibitors, can cause acute or chronic nephrotoxicity in transplant patients due to its vasoconstrictive effect on renal vasculature, toxic tubulopathy and tubular-interstitial effects. Nephrotoxicity was reported in clinical trials [see Adverse Reactions ( 6.1)].
Acute renal impairment associated with tacrolimus toxicity can result in high serum creatinine, hyperkalemia, decreased secretion of urea and hyperuricemia, and is usually reversible. In patients with elevated serum creatinine and tacrolimus whole blood trough concentrations greater than the recommended range, consider dosage reduction or temporary interruption of tacrolimus administration.
The risk for nephrotoxicity may increase when Tacrolimus Injection is concomitantly administered with CYP3A inhibitors (by increasing tacrolimus whole blood concentrations) or drugs associated with nephrotoxicity (e.g., aminoglycosides, ganciclovir, amphotericin B, cisplatin, nucleotide reverse transcriptase inhibitors, protease inhibitors) When tacrolimus is used concurrently with other known nephrotoxic drugs, monitor renal function and tacrolimus blood concentrations, and adjust doses of both tacrolimus and/or concomitant medications during concurrent use [see Drug Interactions ( 7.2)] .
2.5 Dosage Modification for Patients With Hepatic Impairment (2.5 Dosage Modification for Patients with Hepatic Impairment)
Due to the reduced clearance and prolonged half-life, patients with severe hepatic impairment (Child-Pugh ≥ 10) may require lower doses of tacrolimus. Close monitoring of blood concentrations is warranted. The use of tacrolimus in liver transplant recipients experiencing post-transplant hepatic impairment may be associated with increased risk of developing renal insufficiency related to high whole blood concentrations of tacrolimus. These patients should be monitored closely, and dosage adjustments should be considered. Some evidence suggests that lower doses should be used in these patients [see Dosage and Administration ( 2.2), Warnings and Precautions ( 5.5), Use in Specific Populations ( 8.7), and Clinical Pharmacology ( 12.3)].
1.1 Prophylaxis of Organ Rejection in Kidney, Liver Or Heart Transplant (1.1 Prophylaxis of Organ Rejection in Kidney, Liver or Heart Transplant)
Tacrolimus is indicated for the prophylaxis of organ rejection, in adult patients receiving allogeneic kidney transplant [see Clinical Studies ( 14.1)], liver transplants [see Clinical Studies ( 14.2)] and heart transplant [see Clinical Studies ( 14.3)], and pediatric patients receiving allogeneic liver transplants [see Clinical Studies ( 14.2)] in combination with other immunosuppressants.
Additional pediatric use information is approved for Astellas Pharma US, Inc.'s Prograf ® (tacrolimus) products. However, due to Astellas Pharma US, Inc.'s marketing exclusivity rights, this drug product is not labeled with that information.
2.7 Preparation and Administration Instructions of Tacrolimus Injection for Pharmacists
Tacrolimus can cause fetal harm. Follow applicable special handling and disposal procedures 1 [see How Supplied/ Storage and Handling ( 16.4)] .
Tacrolimus injection must be diluted with 0.9% Sodium Chloride Injection or 5% Dextrose Injection to a concentration between 0.004 mg/mL and 0.02 mg/mL prior to use. Diluted infusion solution should be stored in glass or polyethylene containers and should be discarded after 24 hours. The diluted infusion solution should not be stored in a polyvinyl chloride (PVC) container due to decreased stability and the potential for extraction of phthalates. In situations where more dilute solutions are utilized (e.g., pediatric dosing, etc.), PVC-free tubing should likewise be used to minimize the potential for significant drug adsorption onto the tubing.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Due to the chemical instability of tacrolimus in alkaline media, Tacrolimus injection should not be mixed or co-infused with solutions of pH 9 or greater (e.g., ganciclovir or acyclovir).
5.16 Thrombotic Microangiopathy (including Hemolytic Uremic Syndrome and Thrombotic Thrombocytopenic Purpura) (5.16 Thrombotic Microangiopathy (Including Hemolytic Uremic Syndrome and Thrombotic Thrombocytopenic Purpura))
Cases of thrombotic microangiopathy (TMA), including hemolytic uremic syndrome (HUS) and thrombotic thrombocytopenic purpura (TTP), have been reported in patients treated with tacrolimus. TMA may have a multifactorial etiology. Risk factors for TMA that can occur in transplant patients include, for example, severe infections, graft-versus-host disease (GVHD), Human Leukocyte Antigen (HLA) mismatch, the use of calcineurin inhibitors and mammalian target of rapamycin (mTOR) inhibitors. These risk factors may, either alone or combined, contribute to the risk of TMA.
In patients with signs and symptoms of TMA, consider tacrolimus as a risk factor. Concurrent use of tacrolimus and mTOR inhibitors may contribute to the risk of TMA.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:44.392007 · Updated: 2026-03-14T22:38:30.428423