These Highlights Do Not Include All The Information Needed To Use Prezcobix Safely And Effectively. See Full Prescribing Information For Prezcobix.
9c38fdb6-d0ba-4f16-a0e3-85d9ec334d9f
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage ( 1 ) 03/2025 Dosage and Administration ( 2.1 ) 03/2025
Indications and Usage
PREZCOBIX is indicated in combination with other antiretroviral agents for the treatment of human immunodeficiency virus (HIV-1) in treatment-naïve and treatment-experienced adults and pediatric patients weighing at least 25 kg with no darunavir resistance-associated substitutions (V11I, V32I, L33F, I47V, I50V, I54L, I54M, T74P, L76V, I84V, L89V).
Dosage and Administration
Recommended dosage: Adults and pediatric patients weighing at least 40 kg: One 800 mg/150 mg tablet taken once daily with food. ( 2.1 ) Pediatric patients weighing at least 25 kg to less than 40 kg: One 675 mg/150 mg tablet taken once daily with food. ( 2.1 ) Testing Prior to Initiation: HIV genotypic testing is recommended for antiretroviral treatment experienced patients. Assess estimated creatinine clearance in all patients prior to starting PREZCOBIX. When used with tenofovir DF: Assess urine glucose and urine protein at baseline and monitor creatinine clearance, urine glucose, and urine protein. Monitor serum phosphorus in patients with or at risk for renal impairment. ( 2.2 )
Warnings and Precautions
Drug-induced hepatitis (e.g., acute hepatitis, cytolytic hepatitis), liver injury, including some fatalities can occur with PREZCOBIX. Monitor liver function before and during therapy, especially in patients with underlying chronic hepatitis, cirrhosis, or in patients who have pre-treatment elevations of transaminases. ( 5.1 ) Skin reactions ranging from mild to severe, including Stevens-Johnson Syndrome, toxic epidermal necrolysis, drug rash with eosinophilia and systemic symptoms and acute generalized exanthematous pustulosis, can occur with PREZCOBIX. Discontinue treatment if severe reaction develops. ( 5.2 ) When PREZCOBIX is used in combination with a tenofovir disoproxil fumarate (tenofovir DF) containing regimen, cases of acute renal failure and Fanconi syndrome have been reported. ( 5.4 ) PREZCOBIX is not recommended in combination with other antiretroviral drugs that require pharmacokinetic boosting. ( 5.6 ) Monitor in patients with a known sulfonamide allergy. ( 5.7 ) Patients receiving PREZCOBIX may develop new onset or exacerbations of diabetes mellitus/hyperglycemia ( 5.8 ), redistribution/accumulation of body fat ( 5.9 ), and immune reconstitution syndrome. ( 5.10 ) Patients with hemophilia may develop increased bleeding events. ( 5.11 )
Contraindications
Darunavir and cobicistat are both inhibitors of the cytochrome P450 3A (CYP3A) isoform. PREZCOBIX should not be co-administered with medicinal products that are highly dependent on CYP3A for clearance and for which increased plasma concentrations are associated with serious and/or life-threatening events (narrow therapeutic index). Darunavir and cobicistat are both substrates of the cytochrome P450 3A (CYP3A) isoform. Co-administration of PREZCOBIX with CYP3A inducers may lead to lower exposures of darunavir and cobicistat and potential loss of efficacy of darunavir and possible resistance. Examples of drugs that are contraindicated for co-administration with PREZCOBIX [see Drug Interactions (7.3) and Clinical Pharmacology (12.3) ] are listed below. Alpha 1-adrenoreceptor antagonist: alfuzosin Anticonvulsants: carbamazepine, phenobarbital, phenytoin Anti-gout: colchicine, in patients with renal and/or hepatic impairment Antimycobacterial: rifampin Antipsychotics: lurasidone, pimozide Cardiac Disorders: dronedarone, ivabradine, ranolazine Ergot derivatives, e.g. dihydroergotamine, ergotamine, methylergonovine Herbal product: St. John's wort ( Hypericum perforatum ) Hepatitis C direct acting antiviral: elbasvir/grazoprevir Lipid modifying agents: lomitapide, lovastatin, simvastatin Opioid Antagonist: naloxegol PDE-5 inhibitor: sildenafil when used for treatment of pulmonary arterial hypertension Sedatives/hypnotics: orally administered midazolam, triazolam
Adverse Reactions
The following adverse reactions are discussed in other sections of the labeling: Hepatotoxicity [see Warnings and Precautions (5.1) ] Severe skin reactions [see Warnings and Precautions (5.2) ] Effects on serum creatinine [see Warnings and Precautions (5.3) ] New onset or worsening renal impairment when used with tenofovir DF [see Warnings and Precautions (5.4) ] Immune Reconstitution Syndrome [see Warnings and Precautions (5.10) ]
Drug Interactions
Table 2 provides dosing recommendations for expected clinically relevant interactions with PREZCOBIX (this table is not all inclusive). These recommendations are based on either drug interaction trials or predicted interactions due to the expected magnitude of interaction and potential for serious adverse events or loss of therapeutic effect. The table includes examples of potentially significant interactions but is not all inclusive , and therefore the label of each drug that is co-administered with PREZCOBIX should be consulted for information related to the route of metabolism, interaction pathways, potential risks, and specific actions to be taken with regard to co-administration. For the list of examples of contraindicated drugs, [see Contraindications (4) ] . Table 2: Established and Other Potentially Significant this table is not all inclusive Drug Interactions: Alterations in Dose or Regimen May Be Recommended Concomitant Drug Class: Drug Name Examples Effect on Concentration of Darunavir, Cobicistat, or Concomitant Drug Clinical Comment HIV-1 antiviral agents: Nucleoside Reverse Transcriptase Inhibitors (NRTIs) didanosine ↔ darunavir ↔ cobicistat ↔ didanosine Didanosine should be administered one hour before or two hours after PREZCOBIX (administered with food). HIV-1 antiviral agents: Non-Nucleoside Reverse Transcriptase Inhibitors (NNRTIs) efavirenz ↓ cobicistat ↓ darunavir Co-administration with efavirenz is not recommended because it may result in loss of therapeutic effect and development of resistance to darunavir. etravirine ↓ cobicistat darunavir: effect unknown Co-administration with etravirine is not recommended because it may result in loss of therapeutic effect and development of resistance to darunavir. nevirapine ↓ cobicistat darunavir: effect unknown Co-administration with nevirapine is not recommended because it may result in loss of therapeutic effect and development of resistance to darunavir. HIV-1 antiviral agents: CCR5 co-receptor antagonists maraviroc ↑ maraviroc Maraviroc is a substrate of CYP3A. When co-administered with PREZCOBIX, patients should receive maraviroc 150 mg twice daily. Other agents Alpha 1-adrenoreceptor antagonist: alfuzosin ↑ alfuzosin Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as hypotension. Antibacterials: clarithromycin, erythromycin, telithromycin ↑ darunavir ↑ cobicistat ↑ antibacterial Consider alternative antibiotics with concomitant use of PREZCOBIX. Anticancer agents: dasatinib, nilotinib ↑ anticancer agent A decrease in the dosage or an adjustment of the dosing interval of dasatinib or nilotinib may be necessary when co-administered with PREZCOBIX. Consult the dasatinib and nilotinib prescribing information for dosing instructions. vinblastine, vincristine For vincristine and vinblastine, consider temporarily withholding the cobicistat-containing antiretroviral regimen in patients who develop significant hematologic or gastrointestinal side effects when PREZCOBIX is administered concurrently with vincristine or vinblastine. If the antiretroviral regimen must be withheld for a prolonged period, consider initiating a revised regimen that does not include a CYP3A or P-gp inhibitor. Anticoagulants: Direct Oral Anticoagulants (DOACs) apixaban ↑ apixaban Due to potentially increased bleeding risk, dosing recommendations for co-administration of apixaban with PREZCOBIX depend on the apixaban dose. Refer to apixaban dosing instructions for co-administration with P-gp and strong CYP3A inhibitors in apixaban prescribing information. rivaroxaban ↑ rivaroxaban Co-administration of rivaroxaban with PREZCOBIX is not recommended because it may lead to an increased bleeding risk. dabigatran etexilate edoxaban ↑ dabigatran ↑ edoxaban Refer to the dabigatran etexilate or edoxaban prescribing information for recommendations regarding co-administration. The specific recommendations are based on indication, renal function, and effect of the co-administered P-gp inhibitors on the concentration of dabigatran or edoxaban. Clinical monitoring is recommended when a DOAC not affected by CYP3A4 but transported by P-gp, including dabigatran etexilate and edoxaban, is co-administered with PREZCOBIX. Other Anticoagulants: warfarin warfarin: effect unknown Monitor the international normalized ratio (INR) when co-administering with warfarin. Anticonvulsants: carbamazepine, phenobarbital, phenytoin ↓ darunavir ↓ cobicistat Co-administration is contraindicated due to potential for reduced plasma concentrations of darunavir, which may result in loss of therapeutic effect and development of resistance. Anticonvulsants with CYP3A induction effects that are NOT contraindicated: e.g. eslicarbazepine, oxcarbazepine ↓ cobicistat darunavir: effect unknown Consider alternative anticonvulsant or antiretroviral therapy to avoid potential changes in exposures. If co-administration is necessary, monitor for lack or loss of virologic response. Anticonvulsants that are metabolized by CYP3A: e.g. clonazepam ↑ clonazepam Clinical monitoring of anticonvulsants is recommended. Antidepressants: Selective Serotonin Reuptake Inhibitors (SSRIs): e.g. paroxetine, sertraline SSRIs: effects unknown When co-administering with SSRIs, TCAs, or trazodone, careful dose titration of the antidepressant to the desired effect, including using the lowest feasible initial or maintenance dose, and monitoring for antidepressant response are recommended. Tricyclic Antidepressants (TCAs): e.g. amitriptyline, desipramine, imipramine, nortriptyline ↑ TCAs Other antidepressants: trazodone ↑ trazodone Antifungals: itraconazole, isavuconazole, ketoconazole, posaconazole ↑ darunavir ↑ cobicistat Monitor for increased darunavir or cobicistat and/or antifungal adverse reactions. ↑ itraconazole ↑ ketoconazole ↑ isavuconazole ↔ posaconazole Specific dosing recommendations are not available for co-administration with these antifungals. Monitor for increased itraconazole or ketoconazole adverse reactions. voriconazole voriconazole: effects unknown Co-administration with voriconazole is not recommended unless benefit/risk assessment justifies the use of voriconazole. Anti-gout: colchicine ↑ colchicine Co-administration is contraindicated in patients with renal and/or hepatic impairment due to potential for serious and/or life-threatening reactions. For patients without renal or hepatic impairment: Treatment of gout flares – co-administration of colchicine : 0.6 mg (1 tablet) × 1 dose, followed by 0.3 mg (half tablet) 1 hour later. Treatment course to be repeated no earlier than 3 days. Prophylaxis of gout flares – co-administration of colchicine : If the original regimen was 0.6 mg twice a day, the regimen should be adjusted to 0.3 mg once a day. If the original regimen was 0.6 mg once a day, the regimen should be adjusted to 0.3 mg once every other day. Treatment of familial Mediterranean fever – co-administration of colchicine : Maximum daily dose of 0.6 mg (may be given as 0.3 mg twice a day). Antimalarial: artemether/lumefantrine artemether: effect unknown lumefantrine: effect unknown Monitor for a potential decrease of antimalarial efficacy or potential QT prolongation. Antimycobacterials: rifampin ↓ darunavir ↓ cobicistat Co-administration is contraindicated due to potential for loss of therapeutic effect and development of resistance. rifabutin ↑ rifabutin cobicistat: effects unknown darunavir: effects unknown When used in combination with PREZCOBIX, the recommended dose of rifabutin is 150 mg every other day. Monitor for rifabutin-associated adverse reactions including neutropenia and uveitis. rifapentine ↓ darunavir Co-administration with rifapentine is not recommended. Antipsychotics: lurasidone ↑ lurasidone Co-administration is contraindicated due to potential for serious and/or life-threatening reactions. pimozide ↑ pimozide Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as cardiac arrhythmias. e.g. perphenazine, risperidone, thioridazine ↑ antipsychotic A decrease in the dose of antipsychotics that are metabolized by CYP3A or CYP2D6 may be needed when co-administered with PREZCOBIX. quetiapine ↑ quetiapine Initiation of PREZCOBIX in patients taking quetiapine : Consider alternative antiretroviral therapy to avoid increases in quetiapine exposure. If co-administration is necessary, reduce the quetiapine dose to 1/6 of the current dose and monitor for quetiapine- associated adverse reactions. Refer to the quetiapine prescribing information for recommendations on adverse reaction monitoring. Initiation of quetiapine in patients taking PREZCOBIX : Refer to the quetiapine prescribing information for initial dosing and titration of quetiapine. β-Blockers: e.g. carvedilol, metoprolol, timolol ↑ beta-blockers Clinical monitoring is recommended for co-administration with beta-blockers that are metabolized by CYP2D6. Calcium channel blockers: e.g. amlodipine, diltiazem, felodipine, nifedipine, verapamil ↑ calcium channel blockers Clinical monitoring is recommended for co-administration with calcium channel blockers metabolized by CYP3A. Cardiac Disorders: ranolazine, ivabradine ↑ ranolazine ↑ ivabradine Co-administration is contraindicated due to potential for serious and/or life-threatening reactions. dronedarone ↑ dronedarone Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as cardiac arrhythmias. Other antiarrhythmics e.g. amiodarone, disopyramide, flecainide, lidocaine (systemic), mexiletine, propafenone, quinidine ↑ antiarrhythmics Clinical monitoring is recommended upon co-administration with antiarrhythmics. digoxin ↑ digoxin When co-administering with digoxin, titrate the digoxin dose and monitor digoxin concentrations. Corticosteroids: dexamethasone (systemic) Corticosteroids primarily metabolized by CYP3A: e.g. betamethasone budesonide ciclesonide fluticasone methylprednisolone mometasone triamcinolone ↓ darunavir ↓ cobicistat ↑ corticosteroids Co-administration with systemic dexamethasone or other systemic corticosteroids that induce CYP3A may result in loss of therapeutic effect and development of resistance to PREZCOBIX. Consider alternative corticosteroids. Co-administration with corticosteroids (all routes of administration) of which exposures are significantly increased by strong CYP3A inhibitors can increase the risk for Cushing's syndrome and adrenal suppression. Alternative corticosteroids including beclomethasone, prednisone and prednisolone (for which PK and/or PD are less affected by strong CYP3A inhibitors relative to other steroids) should be considered, particularly for long-term use. Endothelin receptor antagonists: bosentan ↓ darunavir ↓ cobicistat ↑ bosentan Initiation of bosentan in patients taking PREZCOBIX : In patients who have been receiving PREZCOBIX for at least 10 days, start bosentan at 62.5 mg once daily or every other day based upon individual tolerability. Initiation of PREZCOBIX in patients on bosentan : Discontinue use of bosentan at least 36 hours prior to initiation of PREZCOBIX. After at least 10 days following the initiation of PREZCOBIX, resume bosentan at 62.5 mg once daily or every other day based upon individual tolerability. Switching from darunavir co-administered with ritonavir to PREZCOBIX in patients on bosentan : Maintain bosentan dose. Ergot derivatives: e.g. dihydroergotamine, ergotamine, methylergonovine ↑ ergot derivatives Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as acute ergot toxicity characterized by peripheral vasospasm and ischemia of the extremities and other tissues. Hepatitis C virus (HCV): Direct-Acting Antivirals: elbasvir/grazoprevir ↑ elbasvir/grazoprevir Co-administration is contraindicated due to potential for the increased risk of alanine transaminase (ALT) elevations. glecaprevir/pibrentasvir ↑ glecaprevir ↑ pibrentasvir Co-administration of PREZCOBIX with glecaprevir/pibrentasvir is not recommended. Herbal product: St. John's wort ( Hypericum perforatum ) ↓ darunavir ↓ cobicistat Co-administration is contraindicated due to potential for reduced plasma concentrations of darunavir, which may result in loss of therapeutic effect and development of resistance. Hormonal contraceptives: Additional or alternative (non-hormonal) forms of contraception should be considered when estrogen-containing contraceptives are co-administered with PREZCOBIX [see Use in Specific Populations (8.3) ] . drospirenone/ethinylestradiol ↑ drospirenone ↓ ethinylestradiol For co-administration with drospirenone, clinical monitoring is recommended due to the potential for hyperkalemia. Other progestin/estrogen contraceptives progestin: effects unknown estrogen: effects unknown No data are available to make recommendations on co-administration with other hormonal contraceptives. Immunosuppressants: cyclosporine, sirolimus, tacrolimus ↑ immunosuppressants These immunosuppressant agents are metabolized by CYP3A. Therapeutic drug monitoring is recommended with concomitant use Immunosuppressant /neoplastic: everolimus ↑ immunosuppressants Co-administration of everolimus and PREZCOBIX is not recommended. irinotecan Discontinue PREZCOBIX at least 1 week prior to starting irinotecan therapy. Do not administer PREZCOBIX with irinotecan unless there are no therapeutic alternatives. Inhaled beta agonist: salmeterol ↑ salmeterol Co-administration with salmeterol is not recommended and may result in increased risk of cardiovascular adverse events associated with salmeterol, including QT prolongation, palpitations, and sinus tachycardia. Lipid Modifying Agents HMG-CoA reductase inhibitors: lovastatin, simvastatin ↑ lovastatin ↑ simvastatin Co-administration is contraindicated due to potential for serious reactions such as myopathy including rhabdomyolysis. atorvastatin, fluvastatin, pitavastatin, pravastatin, rosuvastatin ↑ atorvastatin ↑ fluvastatin ↑ pravastatin ↑ rosuvastatin pitavastatin: effect unknown For atorvastatin, fluvastatin, pitavastatin, pravastatin, and rosuvastatin, start with the lowest recommended dose and titrate while monitoring for safety (e.g. myopathy). Dosage recommendations with atorvastatin or rosuvastatin are as follows: atorvastatin dosage should not exceed 20 mg/day rosuvastatin dosage should not exceed 20 mg/day Other lipid modifying agents: lomitapide ↑ lomitapide Co-administration is contraindicated due to potential for markedly increased transaminases associated with increased plasma concentrations of lomitapide. Narcotic analgesics metabolized by CYP3A: e.g. fentanyl, oxycodone ↑ fentanyl ↑ oxycodone Careful monitoring of therapeutic effects and adverse reactions associated with CYP3A-metabolized narcotic analgesics (including potentially fatal respiratory depression) is recommended with co-administration. tramadol ↑ tramadol A dose decrease may be needed for tramadol with concomitant use. Narcotic analgesic for treatment of opioid dependence: buprenorphine, buprenorphine/naloxone, methadone buprenorphine or buprenorphine/ naloxone: effects unknown methadone: effects unknown Initiation of buprenorphine, buprenorphine/naloxone or methadone in patients taking PREZCOBIX : Carefully titrate the dose of buprenorphine, buprenorphine/naloxone or methadone to the desired effect; use the lowest feasible initial or maintenance dose. Initiation of PREZCOBIX in patients taking buprenorphine, buprenorphine/naloxone or methadone : A dose adjustment for buprenorphine, buprenorphine/naloxone or methadone may be needed. Monitor clinical signs and symptoms. Opioid Antagonist naloxegol ↑ naloxegol Co-administration of PREZCOBIX and naloxegol is contraindicated due to potential for precipitating opioid withdrawal symptoms. Phosphodiesterase PDE-5 inhibitors: e.g. avanafil, sildenafil, tadalafil, vardenafil ↑ PDE-5 inhibitors Co-administration with avanafil is not recommended because a safe and effective avanafil dosage regimen has not been established. Co-administration with PDE-5 inhibitors may result in an increase in PDE-5 inhibitor-associated adverse reactions including hypotension, syncope, visual disturbances and priapism. Use of PDE-5 inhibitors for pulmonary arterial hypertension (PAH): Co-administration with sildenafil used for PAH is contraindicated due to potential for sildenafil associated adverse reactions (which include visual disturbances, hypotension, prolonged erection, and syncope). The following dose adjustments are recommended for use of tadalafil with PREZCOBIX: Initiation of tadalafil in patients taking PREZCOBIX : In patients receiving PREZCOBIX for at least one week, start tadalafil at 20 mg once daily. Increase to 40 mg once daily based upon individual tolerability. Initiation of PREZCOBIX in patients taking tadalafil : Avoid use of tadalafil during the initiation of PREZCOBIX. Stop tadalafil at least 24 hours prior to starting PREZCOBIX. After at least one week following the initiation of PREZCOBIX, resume tadalafil at 20 mg once daily. Increase to 40 mg once daily based upon individual tolerability. Patients switching from darunavir co-administered with ritonavir to PREZCOBIX : Maintain tadalafil dose. Use of PDE-5 inhibitors for erectile dysfunction: Sildenafil at a single dose not exceeding 25 mg in 48 hours, vardenafil at a single dose not exceeding 2.5 mg dose in 72 hours, or tadalafil at a single dose not exceeding 10 mg dose in 72 hours can be used with increased monitoring for PDE-5 inhibitor-associated adverse reactions. Platelet aggregation inhibitor: ticagrelor ↑ ticagrelor Co-administration of PREZCOBIX and ticagrelor is not recommended. clopidogrel ↓ clopidogrel active metabolite Co-administration of PREZCOBIX with clopidogrel is not recommended due to the potential reduction of the antiplatelet activity of clopidogrel. prasugrel ↔ prasugrel active metabolite No dose adjustment is needed when prasugrel is co-administered with PREZCOBIX. Sedatives/hypnotics: orally administered midazolam, triazolam ↑ midazolam ↑ triazolam Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as prolonged or increased sedation or respiratory depression. Triazolam and orally administered midazolam are extensively metabolized by CYP3A. Co-administration of triazolam or orally administered midazolam with PREZCOBIX may cause large increases in the concentrations of these benzodiazepines. metabolized by CYP3A: e.g. buspirone, diazepam, estazolam, zolpidem ↑ sedatives/hypnotics With concomitant use, titration is recommended with sedatives/hypnotics metabolized by CYP3A and a lower dose of the sedatives/hypnotics should be considered with monitoring for increased and prolonged effects or adverse reactions. parenterally administered midazolam Co-administration of parenteral midazolam should be done in a setting that ensures close clinical monitoring and appropriate medical management in case of respiratory depression and/or prolonged sedation. Dose reduction for parenteral midazolam should be considered, especially if more than a single dose of midazolam is administered. Urinary antispasmodics fesoterodine ↑ fesoterodine When fesoterodine is co-administered with PREZCOBIX, do not exceed a fesoterodine dose of 4 mg once daily. solifenacin ↑ solifenacin When solifenacin is co-administered with PREZCOBIX, do not exceed a solifenacin dose of 5 mg once daily.
Storage and Handling
PREZCOBIX ® (darunavir and cobicistat) tablets, 800/150 mg, are supplied as pink, oval-shaped, film-coated tablets debossed with "800" on one side and "TG" on the other side. Bottle of 30 tablets (NDC 59676-575-30). PREZCOBIX ® (darunavir and cobicistat) tablets, 675/150 mg, are supplied as green to dark green, oval-shaped, scored film-coated tablet debossed with “675” on one side and “TG” on the other side. Bottle of 30 tablets (NDC 59676-578-30).
How Supplied
PREZCOBIX ® (darunavir and cobicistat) tablets, 800/150 mg, are supplied as pink, oval-shaped, film-coated tablets debossed with "800" on one side and "TG" on the other side. Bottle of 30 tablets (NDC 59676-575-30). PREZCOBIX ® (darunavir and cobicistat) tablets, 675/150 mg, are supplied as green to dark green, oval-shaped, scored film-coated tablet debossed with “675” on one side and “TG” on the other side. Bottle of 30 tablets (NDC 59676-578-30).
Medication Information
Warnings and Precautions
Drug-induced hepatitis (e.g., acute hepatitis, cytolytic hepatitis), liver injury, including some fatalities can occur with PREZCOBIX. Monitor liver function before and during therapy, especially in patients with underlying chronic hepatitis, cirrhosis, or in patients who have pre-treatment elevations of transaminases. ( 5.1 ) Skin reactions ranging from mild to severe, including Stevens-Johnson Syndrome, toxic epidermal necrolysis, drug rash with eosinophilia and systemic symptoms and acute generalized exanthematous pustulosis, can occur with PREZCOBIX. Discontinue treatment if severe reaction develops. ( 5.2 ) When PREZCOBIX is used in combination with a tenofovir disoproxil fumarate (tenofovir DF) containing regimen, cases of acute renal failure and Fanconi syndrome have been reported. ( 5.4 ) PREZCOBIX is not recommended in combination with other antiretroviral drugs that require pharmacokinetic boosting. ( 5.6 ) Monitor in patients with a known sulfonamide allergy. ( 5.7 ) Patients receiving PREZCOBIX may develop new onset or exacerbations of diabetes mellitus/hyperglycemia ( 5.8 ), redistribution/accumulation of body fat ( 5.9 ), and immune reconstitution syndrome. ( 5.10 ) Patients with hemophilia may develop increased bleeding events. ( 5.11 )
Indications and Usage
PREZCOBIX is indicated in combination with other antiretroviral agents for the treatment of human immunodeficiency virus (HIV-1) in treatment-naïve and treatment-experienced adults and pediatric patients weighing at least 25 kg with no darunavir resistance-associated substitutions (V11I, V32I, L33F, I47V, I50V, I54L, I54M, T74P, L76V, I84V, L89V).
Dosage and Administration
Recommended dosage: Adults and pediatric patients weighing at least 40 kg: One 800 mg/150 mg tablet taken once daily with food. ( 2.1 ) Pediatric patients weighing at least 25 kg to less than 40 kg: One 675 mg/150 mg tablet taken once daily with food. ( 2.1 ) Testing Prior to Initiation: HIV genotypic testing is recommended for antiretroviral treatment experienced patients. Assess estimated creatinine clearance in all patients prior to starting PREZCOBIX. When used with tenofovir DF: Assess urine glucose and urine protein at baseline and monitor creatinine clearance, urine glucose, and urine protein. Monitor serum phosphorus in patients with or at risk for renal impairment. ( 2.2 )
Contraindications
Darunavir and cobicistat are both inhibitors of the cytochrome P450 3A (CYP3A) isoform. PREZCOBIX should not be co-administered with medicinal products that are highly dependent on CYP3A for clearance and for which increased plasma concentrations are associated with serious and/or life-threatening events (narrow therapeutic index). Darunavir and cobicistat are both substrates of the cytochrome P450 3A (CYP3A) isoform. Co-administration of PREZCOBIX with CYP3A inducers may lead to lower exposures of darunavir and cobicistat and potential loss of efficacy of darunavir and possible resistance. Examples of drugs that are contraindicated for co-administration with PREZCOBIX [see Drug Interactions (7.3) and Clinical Pharmacology (12.3) ] are listed below. Alpha 1-adrenoreceptor antagonist: alfuzosin Anticonvulsants: carbamazepine, phenobarbital, phenytoin Anti-gout: colchicine, in patients with renal and/or hepatic impairment Antimycobacterial: rifampin Antipsychotics: lurasidone, pimozide Cardiac Disorders: dronedarone, ivabradine, ranolazine Ergot derivatives, e.g. dihydroergotamine, ergotamine, methylergonovine Herbal product: St. John's wort ( Hypericum perforatum ) Hepatitis C direct acting antiviral: elbasvir/grazoprevir Lipid modifying agents: lomitapide, lovastatin, simvastatin Opioid Antagonist: naloxegol PDE-5 inhibitor: sildenafil when used for treatment of pulmonary arterial hypertension Sedatives/hypnotics: orally administered midazolam, triazolam
Adverse Reactions
The following adverse reactions are discussed in other sections of the labeling: Hepatotoxicity [see Warnings and Precautions (5.1) ] Severe skin reactions [see Warnings and Precautions (5.2) ] Effects on serum creatinine [see Warnings and Precautions (5.3) ] New onset or worsening renal impairment when used with tenofovir DF [see Warnings and Precautions (5.4) ] Immune Reconstitution Syndrome [see Warnings and Precautions (5.10) ]
Drug Interactions
Table 2 provides dosing recommendations for expected clinically relevant interactions with PREZCOBIX (this table is not all inclusive). These recommendations are based on either drug interaction trials or predicted interactions due to the expected magnitude of interaction and potential for serious adverse events or loss of therapeutic effect. The table includes examples of potentially significant interactions but is not all inclusive , and therefore the label of each drug that is co-administered with PREZCOBIX should be consulted for information related to the route of metabolism, interaction pathways, potential risks, and specific actions to be taken with regard to co-administration. For the list of examples of contraindicated drugs, [see Contraindications (4) ] . Table 2: Established and Other Potentially Significant this table is not all inclusive Drug Interactions: Alterations in Dose or Regimen May Be Recommended Concomitant Drug Class: Drug Name Examples Effect on Concentration of Darunavir, Cobicistat, or Concomitant Drug Clinical Comment HIV-1 antiviral agents: Nucleoside Reverse Transcriptase Inhibitors (NRTIs) didanosine ↔ darunavir ↔ cobicistat ↔ didanosine Didanosine should be administered one hour before or two hours after PREZCOBIX (administered with food). HIV-1 antiviral agents: Non-Nucleoside Reverse Transcriptase Inhibitors (NNRTIs) efavirenz ↓ cobicistat ↓ darunavir Co-administration with efavirenz is not recommended because it may result in loss of therapeutic effect and development of resistance to darunavir. etravirine ↓ cobicistat darunavir: effect unknown Co-administration with etravirine is not recommended because it may result in loss of therapeutic effect and development of resistance to darunavir. nevirapine ↓ cobicistat darunavir: effect unknown Co-administration with nevirapine is not recommended because it may result in loss of therapeutic effect and development of resistance to darunavir. HIV-1 antiviral agents: CCR5 co-receptor antagonists maraviroc ↑ maraviroc Maraviroc is a substrate of CYP3A. When co-administered with PREZCOBIX, patients should receive maraviroc 150 mg twice daily. Other agents Alpha 1-adrenoreceptor antagonist: alfuzosin ↑ alfuzosin Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as hypotension. Antibacterials: clarithromycin, erythromycin, telithromycin ↑ darunavir ↑ cobicistat ↑ antibacterial Consider alternative antibiotics with concomitant use of PREZCOBIX. Anticancer agents: dasatinib, nilotinib ↑ anticancer agent A decrease in the dosage or an adjustment of the dosing interval of dasatinib or nilotinib may be necessary when co-administered with PREZCOBIX. Consult the dasatinib and nilotinib prescribing information for dosing instructions. vinblastine, vincristine For vincristine and vinblastine, consider temporarily withholding the cobicistat-containing antiretroviral regimen in patients who develop significant hematologic or gastrointestinal side effects when PREZCOBIX is administered concurrently with vincristine or vinblastine. If the antiretroviral regimen must be withheld for a prolonged period, consider initiating a revised regimen that does not include a CYP3A or P-gp inhibitor. Anticoagulants: Direct Oral Anticoagulants (DOACs) apixaban ↑ apixaban Due to potentially increased bleeding risk, dosing recommendations for co-administration of apixaban with PREZCOBIX depend on the apixaban dose. Refer to apixaban dosing instructions for co-administration with P-gp and strong CYP3A inhibitors in apixaban prescribing information. rivaroxaban ↑ rivaroxaban Co-administration of rivaroxaban with PREZCOBIX is not recommended because it may lead to an increased bleeding risk. dabigatran etexilate edoxaban ↑ dabigatran ↑ edoxaban Refer to the dabigatran etexilate or edoxaban prescribing information for recommendations regarding co-administration. The specific recommendations are based on indication, renal function, and effect of the co-administered P-gp inhibitors on the concentration of dabigatran or edoxaban. Clinical monitoring is recommended when a DOAC not affected by CYP3A4 but transported by P-gp, including dabigatran etexilate and edoxaban, is co-administered with PREZCOBIX. Other Anticoagulants: warfarin warfarin: effect unknown Monitor the international normalized ratio (INR) when co-administering with warfarin. Anticonvulsants: carbamazepine, phenobarbital, phenytoin ↓ darunavir ↓ cobicistat Co-administration is contraindicated due to potential for reduced plasma concentrations of darunavir, which may result in loss of therapeutic effect and development of resistance. Anticonvulsants with CYP3A induction effects that are NOT contraindicated: e.g. eslicarbazepine, oxcarbazepine ↓ cobicistat darunavir: effect unknown Consider alternative anticonvulsant or antiretroviral therapy to avoid potential changes in exposures. If co-administration is necessary, monitor for lack or loss of virologic response. Anticonvulsants that are metabolized by CYP3A: e.g. clonazepam ↑ clonazepam Clinical monitoring of anticonvulsants is recommended. Antidepressants: Selective Serotonin Reuptake Inhibitors (SSRIs): e.g. paroxetine, sertraline SSRIs: effects unknown When co-administering with SSRIs, TCAs, or trazodone, careful dose titration of the antidepressant to the desired effect, including using the lowest feasible initial or maintenance dose, and monitoring for antidepressant response are recommended. Tricyclic Antidepressants (TCAs): e.g. amitriptyline, desipramine, imipramine, nortriptyline ↑ TCAs Other antidepressants: trazodone ↑ trazodone Antifungals: itraconazole, isavuconazole, ketoconazole, posaconazole ↑ darunavir ↑ cobicistat Monitor for increased darunavir or cobicistat and/or antifungal adverse reactions. ↑ itraconazole ↑ ketoconazole ↑ isavuconazole ↔ posaconazole Specific dosing recommendations are not available for co-administration with these antifungals. Monitor for increased itraconazole or ketoconazole adverse reactions. voriconazole voriconazole: effects unknown Co-administration with voriconazole is not recommended unless benefit/risk assessment justifies the use of voriconazole. Anti-gout: colchicine ↑ colchicine Co-administration is contraindicated in patients with renal and/or hepatic impairment due to potential for serious and/or life-threatening reactions. For patients without renal or hepatic impairment: Treatment of gout flares – co-administration of colchicine : 0.6 mg (1 tablet) × 1 dose, followed by 0.3 mg (half tablet) 1 hour later. Treatment course to be repeated no earlier than 3 days. Prophylaxis of gout flares – co-administration of colchicine : If the original regimen was 0.6 mg twice a day, the regimen should be adjusted to 0.3 mg once a day. If the original regimen was 0.6 mg once a day, the regimen should be adjusted to 0.3 mg once every other day. Treatment of familial Mediterranean fever – co-administration of colchicine : Maximum daily dose of 0.6 mg (may be given as 0.3 mg twice a day). Antimalarial: artemether/lumefantrine artemether: effect unknown lumefantrine: effect unknown Monitor for a potential decrease of antimalarial efficacy or potential QT prolongation. Antimycobacterials: rifampin ↓ darunavir ↓ cobicistat Co-administration is contraindicated due to potential for loss of therapeutic effect and development of resistance. rifabutin ↑ rifabutin cobicistat: effects unknown darunavir: effects unknown When used in combination with PREZCOBIX, the recommended dose of rifabutin is 150 mg every other day. Monitor for rifabutin-associated adverse reactions including neutropenia and uveitis. rifapentine ↓ darunavir Co-administration with rifapentine is not recommended. Antipsychotics: lurasidone ↑ lurasidone Co-administration is contraindicated due to potential for serious and/or life-threatening reactions. pimozide ↑ pimozide Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as cardiac arrhythmias. e.g. perphenazine, risperidone, thioridazine ↑ antipsychotic A decrease in the dose of antipsychotics that are metabolized by CYP3A or CYP2D6 may be needed when co-administered with PREZCOBIX. quetiapine ↑ quetiapine Initiation of PREZCOBIX in patients taking quetiapine : Consider alternative antiretroviral therapy to avoid increases in quetiapine exposure. If co-administration is necessary, reduce the quetiapine dose to 1/6 of the current dose and monitor for quetiapine- associated adverse reactions. Refer to the quetiapine prescribing information for recommendations on adverse reaction monitoring. Initiation of quetiapine in patients taking PREZCOBIX : Refer to the quetiapine prescribing information for initial dosing and titration of quetiapine. β-Blockers: e.g. carvedilol, metoprolol, timolol ↑ beta-blockers Clinical monitoring is recommended for co-administration with beta-blockers that are metabolized by CYP2D6. Calcium channel blockers: e.g. amlodipine, diltiazem, felodipine, nifedipine, verapamil ↑ calcium channel blockers Clinical monitoring is recommended for co-administration with calcium channel blockers metabolized by CYP3A. Cardiac Disorders: ranolazine, ivabradine ↑ ranolazine ↑ ivabradine Co-administration is contraindicated due to potential for serious and/or life-threatening reactions. dronedarone ↑ dronedarone Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as cardiac arrhythmias. Other antiarrhythmics e.g. amiodarone, disopyramide, flecainide, lidocaine (systemic), mexiletine, propafenone, quinidine ↑ antiarrhythmics Clinical monitoring is recommended upon co-administration with antiarrhythmics. digoxin ↑ digoxin When co-administering with digoxin, titrate the digoxin dose and monitor digoxin concentrations. Corticosteroids: dexamethasone (systemic) Corticosteroids primarily metabolized by CYP3A: e.g. betamethasone budesonide ciclesonide fluticasone methylprednisolone mometasone triamcinolone ↓ darunavir ↓ cobicistat ↑ corticosteroids Co-administration with systemic dexamethasone or other systemic corticosteroids that induce CYP3A may result in loss of therapeutic effect and development of resistance to PREZCOBIX. Consider alternative corticosteroids. Co-administration with corticosteroids (all routes of administration) of which exposures are significantly increased by strong CYP3A inhibitors can increase the risk for Cushing's syndrome and adrenal suppression. Alternative corticosteroids including beclomethasone, prednisone and prednisolone (for which PK and/or PD are less affected by strong CYP3A inhibitors relative to other steroids) should be considered, particularly for long-term use. Endothelin receptor antagonists: bosentan ↓ darunavir ↓ cobicistat ↑ bosentan Initiation of bosentan in patients taking PREZCOBIX : In patients who have been receiving PREZCOBIX for at least 10 days, start bosentan at 62.5 mg once daily or every other day based upon individual tolerability. Initiation of PREZCOBIX in patients on bosentan : Discontinue use of bosentan at least 36 hours prior to initiation of PREZCOBIX. After at least 10 days following the initiation of PREZCOBIX, resume bosentan at 62.5 mg once daily or every other day based upon individual tolerability. Switching from darunavir co-administered with ritonavir to PREZCOBIX in patients on bosentan : Maintain bosentan dose. Ergot derivatives: e.g. dihydroergotamine, ergotamine, methylergonovine ↑ ergot derivatives Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as acute ergot toxicity characterized by peripheral vasospasm and ischemia of the extremities and other tissues. Hepatitis C virus (HCV): Direct-Acting Antivirals: elbasvir/grazoprevir ↑ elbasvir/grazoprevir Co-administration is contraindicated due to potential for the increased risk of alanine transaminase (ALT) elevations. glecaprevir/pibrentasvir ↑ glecaprevir ↑ pibrentasvir Co-administration of PREZCOBIX with glecaprevir/pibrentasvir is not recommended. Herbal product: St. John's wort ( Hypericum perforatum ) ↓ darunavir ↓ cobicistat Co-administration is contraindicated due to potential for reduced plasma concentrations of darunavir, which may result in loss of therapeutic effect and development of resistance. Hormonal contraceptives: Additional or alternative (non-hormonal) forms of contraception should be considered when estrogen-containing contraceptives are co-administered with PREZCOBIX [see Use in Specific Populations (8.3) ] . drospirenone/ethinylestradiol ↑ drospirenone ↓ ethinylestradiol For co-administration with drospirenone, clinical monitoring is recommended due to the potential for hyperkalemia. Other progestin/estrogen contraceptives progestin: effects unknown estrogen: effects unknown No data are available to make recommendations on co-administration with other hormonal contraceptives. Immunosuppressants: cyclosporine, sirolimus, tacrolimus ↑ immunosuppressants These immunosuppressant agents are metabolized by CYP3A. Therapeutic drug monitoring is recommended with concomitant use Immunosuppressant /neoplastic: everolimus ↑ immunosuppressants Co-administration of everolimus and PREZCOBIX is not recommended. irinotecan Discontinue PREZCOBIX at least 1 week prior to starting irinotecan therapy. Do not administer PREZCOBIX with irinotecan unless there are no therapeutic alternatives. Inhaled beta agonist: salmeterol ↑ salmeterol Co-administration with salmeterol is not recommended and may result in increased risk of cardiovascular adverse events associated with salmeterol, including QT prolongation, palpitations, and sinus tachycardia. Lipid Modifying Agents HMG-CoA reductase inhibitors: lovastatin, simvastatin ↑ lovastatin ↑ simvastatin Co-administration is contraindicated due to potential for serious reactions such as myopathy including rhabdomyolysis. atorvastatin, fluvastatin, pitavastatin, pravastatin, rosuvastatin ↑ atorvastatin ↑ fluvastatin ↑ pravastatin ↑ rosuvastatin pitavastatin: effect unknown For atorvastatin, fluvastatin, pitavastatin, pravastatin, and rosuvastatin, start with the lowest recommended dose and titrate while monitoring for safety (e.g. myopathy). Dosage recommendations with atorvastatin or rosuvastatin are as follows: atorvastatin dosage should not exceed 20 mg/day rosuvastatin dosage should not exceed 20 mg/day Other lipid modifying agents: lomitapide ↑ lomitapide Co-administration is contraindicated due to potential for markedly increased transaminases associated with increased plasma concentrations of lomitapide. Narcotic analgesics metabolized by CYP3A: e.g. fentanyl, oxycodone ↑ fentanyl ↑ oxycodone Careful monitoring of therapeutic effects and adverse reactions associated with CYP3A-metabolized narcotic analgesics (including potentially fatal respiratory depression) is recommended with co-administration. tramadol ↑ tramadol A dose decrease may be needed for tramadol with concomitant use. Narcotic analgesic for treatment of opioid dependence: buprenorphine, buprenorphine/naloxone, methadone buprenorphine or buprenorphine/ naloxone: effects unknown methadone: effects unknown Initiation of buprenorphine, buprenorphine/naloxone or methadone in patients taking PREZCOBIX : Carefully titrate the dose of buprenorphine, buprenorphine/naloxone or methadone to the desired effect; use the lowest feasible initial or maintenance dose. Initiation of PREZCOBIX in patients taking buprenorphine, buprenorphine/naloxone or methadone : A dose adjustment for buprenorphine, buprenorphine/naloxone or methadone may be needed. Monitor clinical signs and symptoms. Opioid Antagonist naloxegol ↑ naloxegol Co-administration of PREZCOBIX and naloxegol is contraindicated due to potential for precipitating opioid withdrawal symptoms. Phosphodiesterase PDE-5 inhibitors: e.g. avanafil, sildenafil, tadalafil, vardenafil ↑ PDE-5 inhibitors Co-administration with avanafil is not recommended because a safe and effective avanafil dosage regimen has not been established. Co-administration with PDE-5 inhibitors may result in an increase in PDE-5 inhibitor-associated adverse reactions including hypotension, syncope, visual disturbances and priapism. Use of PDE-5 inhibitors for pulmonary arterial hypertension (PAH): Co-administration with sildenafil used for PAH is contraindicated due to potential for sildenafil associated adverse reactions (which include visual disturbances, hypotension, prolonged erection, and syncope). The following dose adjustments are recommended for use of tadalafil with PREZCOBIX: Initiation of tadalafil in patients taking PREZCOBIX : In patients receiving PREZCOBIX for at least one week, start tadalafil at 20 mg once daily. Increase to 40 mg once daily based upon individual tolerability. Initiation of PREZCOBIX in patients taking tadalafil : Avoid use of tadalafil during the initiation of PREZCOBIX. Stop tadalafil at least 24 hours prior to starting PREZCOBIX. After at least one week following the initiation of PREZCOBIX, resume tadalafil at 20 mg once daily. Increase to 40 mg once daily based upon individual tolerability. Patients switching from darunavir co-administered with ritonavir to PREZCOBIX : Maintain tadalafil dose. Use of PDE-5 inhibitors for erectile dysfunction: Sildenafil at a single dose not exceeding 25 mg in 48 hours, vardenafil at a single dose not exceeding 2.5 mg dose in 72 hours, or tadalafil at a single dose not exceeding 10 mg dose in 72 hours can be used with increased monitoring for PDE-5 inhibitor-associated adverse reactions. Platelet aggregation inhibitor: ticagrelor ↑ ticagrelor Co-administration of PREZCOBIX and ticagrelor is not recommended. clopidogrel ↓ clopidogrel active metabolite Co-administration of PREZCOBIX with clopidogrel is not recommended due to the potential reduction of the antiplatelet activity of clopidogrel. prasugrel ↔ prasugrel active metabolite No dose adjustment is needed when prasugrel is co-administered with PREZCOBIX. Sedatives/hypnotics: orally administered midazolam, triazolam ↑ midazolam ↑ triazolam Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as prolonged or increased sedation or respiratory depression. Triazolam and orally administered midazolam are extensively metabolized by CYP3A. Co-administration of triazolam or orally administered midazolam with PREZCOBIX may cause large increases in the concentrations of these benzodiazepines. metabolized by CYP3A: e.g. buspirone, diazepam, estazolam, zolpidem ↑ sedatives/hypnotics With concomitant use, titration is recommended with sedatives/hypnotics metabolized by CYP3A and a lower dose of the sedatives/hypnotics should be considered with monitoring for increased and prolonged effects or adverse reactions. parenterally administered midazolam Co-administration of parenteral midazolam should be done in a setting that ensures close clinical monitoring and appropriate medical management in case of respiratory depression and/or prolonged sedation. Dose reduction for parenteral midazolam should be considered, especially if more than a single dose of midazolam is administered. Urinary antispasmodics fesoterodine ↑ fesoterodine When fesoterodine is co-administered with PREZCOBIX, do not exceed a fesoterodine dose of 4 mg once daily. solifenacin ↑ solifenacin When solifenacin is co-administered with PREZCOBIX, do not exceed a solifenacin dose of 5 mg once daily.
Storage and Handling
PREZCOBIX ® (darunavir and cobicistat) tablets, 800/150 mg, are supplied as pink, oval-shaped, film-coated tablets debossed with "800" on one side and "TG" on the other side. Bottle of 30 tablets (NDC 59676-575-30). PREZCOBIX ® (darunavir and cobicistat) tablets, 675/150 mg, are supplied as green to dark green, oval-shaped, scored film-coated tablet debossed with “675” on one side and “TG” on the other side. Bottle of 30 tablets (NDC 59676-578-30).
How Supplied
PREZCOBIX ® (darunavir and cobicistat) tablets, 800/150 mg, are supplied as pink, oval-shaped, film-coated tablets debossed with "800" on one side and "TG" on the other side. Bottle of 30 tablets (NDC 59676-575-30). PREZCOBIX ® (darunavir and cobicistat) tablets, 675/150 mg, are supplied as green to dark green, oval-shaped, scored film-coated tablet debossed with “675” on one side and “TG” on the other side. Bottle of 30 tablets (NDC 59676-578-30).
Description
Indications and Usage ( 1 ) 03/2025 Dosage and Administration ( 2.1 ) 03/2025
Section 42229-5
HIV Genotypic Testing
HIV genotypic testing is recommended for antiretroviral treatment-experienced patients. However, when HIV genotypic testing is not feasible, PREZCOBIX can be used in protease inhibitor-naïve patients, but is not recommended in protease inhibitor-experienced patients.
Section 42230-3
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 03/2025 | |
| PATIENT INFORMATION
PREZCOBIX ® (prez-koe-bix) (darunavir and cobicistat) tablets |
||
|
What is the most important information I should know about PREZCOBIX?
PREZCOBIX may cause serious side effects, including:
|
||
|
|
|
|
||
|
|
|
|
||
|
What is PREZCOBIX?
PREZCOBIX is a prescription medicine that is used with other human immunodeficiency virus (HIV-1) medicines to treat HIV-1 in adults and in children who weigh at least 55 pounds (25 kg). HIV-1 is the virus that causes Acquired Immune Deficiency Syndrome (AIDS). PREZCOBIX contains the prescription medicines darunavir and cobicistat. It is not known if PREZCOBIX is safe and effective in children weighing less than 55 pounds (25 kg). |
||
Do not take PREZCOBIX with any medicine that contains:
|
||
Before taking PREZCOBIX, tell your healthcare provider about all your medical conditions, including if you:
|
||
How should I take PREZCOBIX?
|
||
What are the possible side effects of PREZCOBIX?
PREZCOBIX may cause serious side effects, including:
|
||
|
|
|
| These are not all of the possible side effects of PREZCOBIX.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store PREZCOBIX?
|
||
|
General information about the safe and effective use of PREZCOBIX.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use PREZCOBIX for a condition for which it was not prescribed. Do not give PREZCOBIX to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about PREZCOBIX that is written for health professionals. |
||
|
What are the ingredients in PREZCOBIX?
Active ingredients: darunavir and cobicistat Inactive ingredients 800 mg/150 mg tablet: colloidal silicon dioxide, crospovidone, hypromellose, magnesium stearate, and silicified microcrystalline cellulose. The tablets are film-coated with a coating material containing iron oxide black, iron oxide red, polyethylene glycol, polyvinyl alcohol (partially hydrolyzed), talc, and titanium dioxide. Inactive ingredients 675 mg/150 mg tablet: colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, and microcrystalline cellulose. The tablets are film-coated with a coating material containing iron oxide black, iron oxide yellow, polyethylene glycol, polyvinyl alcohol (partially hydrolyzed), talc, and titanium dioxide. Manufactured for: Janssen Products, LP, Horsham, PA 19044, USA For patent information: www.janssenpatents.com © Johnson & Johnson and its affiliates 2015, 2025 For more information call 1-800-526-7736. |
Section 44425-7
Storage: Store at 20 °C–25 °C (between 68 °F–77 °F); with excursions permitted to 15 °C–30 °C (59 °F–86 °F) [see USP Controlled Room Temperature].
Keep PREZCOBIX and all medicines out of reach of children.
10 Overdosage
Human experience of acute overdose with PREZCOBIX is limited. No specific antidote is available for overdose with PREZCOBIX. Treatment of overdose with PREZCOBIX consists of general supportive measures including monitoring of vital signs and observation of the clinical status of the patient. Since both darunavir and cobicistat are highly protein bound, dialysis is unlikely to be beneficial in significant removal of the active substance.
11 Description
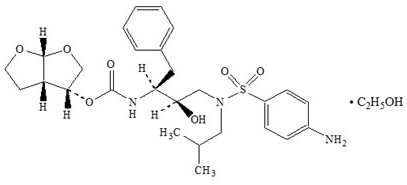
PREZCOBIX ® is a fixed-dose combination tablet containing darunavir and cobicistat. Darunavir is an inhibitor of the human immunodeficiency virus (HIV-1) protease. Cobicistat is a mechanism-based inhibitor of cytochrome P450 (CYP) enzymes of the CYP3A family.
PREZCOBIX ® 800 mg darunavir/150 mg cobicistat tablets are for oral administration. Each tablet contains darunavir ethanolate equivalent to 800 mg of darunavir and 150 mg of cobicistat. The tablets include the following inactive ingredients: colloidal silicon dioxide, crospovidone, hypromellose, magnesium stearate, and silicified microcrystalline cellulose. The tablets are film-coated with a coating material containing iron oxide black, iron oxide red, polyethylene glycol, polyvinyl alcohol (partially hydrolyzed), talc, and titanium dioxide.
PREZCOBIX ® 675 mg darunavir/150 mg cobicistat tablets are for oral administration. Each tablet contains darunavir ethanolate equivalent to 675 mg of darunavir and 150 mg of cobicistat. The tablets include the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, and microcrystalline cellulose. The tablets are film-coated with a coating material containing iron oxide black, iron oxide yellow, polyethylene glycol, polyvinyl alcohol (partially hydrolyzed), talc, and titanium dioxide.
5.11 Hemophilia
There have been reports of increased bleeding, including spontaneous skin hematomas and hemarthrosis in patients with hemophilia type A and B treated with HIV PIs. In some patients, additional factor VIII was given. In more than half of the reported cases, treatment with HIV PIs was continued or reintroduced if treatment had been discontinued. A causal relationship between PI therapy and these episodes has not been established.
12.4 Microbiology
5.7 Sulfa Allergy
Darunavir contains a sulfonamide moiety. Monitor patients with a known sulfonamide allergy after initiating PREZCOBIX. In clinical studies with darunavir co-administered with ritonavir, the incidence and severity of rash were similar in subjects with or without a history of sulfonamide allergy.
8.4 Pediatric Use
The safety and effectiveness of PREZCOBIX for the treatment of HIV-1 in pediatric patients weighing at least 25 kg was established through a trial with components of PREZCOBIX. Use of PREZCOBIX in this group is supported by evidence from adequate and well-controlled studies in adults with additional pharmacokinetic, safety, and virologic data from a study of components of PREZCOBIX (Trial GS-US-216-0128) in pediatric participants with HIV-1 aged 6 to less than 18 years [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.2)] .
The safety and effectiveness of PREZCOBIX have not been established in pediatric patients weighing less than 25 kg. Darunavir, a component of PREZCOBIX is not recommended in pediatric patients below 3 years of age because of toxicity and mortality observed in juvenile rats dosed with darunavir.
8.5 Geriatric Use
Clinical trials of PREZCOBIX did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently from younger patients. In general, caution should be exercised in the administration and monitoring of PREZCOBIX in elderly patients, reflecting the greater frequency of decreased hepatic function, and of concomitant disease or other drug therapy [see Clinical Pharmacology (12.3)] .
5.1 Hepatotoxicity
During the darunavir clinical development program (N=3063), where darunavir was co-administered with ritonavir 100 mg once or twice daily, drug-induced hepatitis (e.g., acute hepatitis, cytolytic hepatitis) was reported in 0.5% of participants. Patients with pre-existing liver dysfunction, including chronic active hepatitis B or C, have an increased risk for liver function abnormalities including severe hepatic adverse reactions.
Post-marketing cases of liver injury, including some fatalities, have also been reported with darunavir co-administered with ritonavir. These have generally occurred in patients with advanced HIV-1 disease taking multiple concomitant medications, having co-morbidities including hepatitis B or C co-infection, and/or developing immune reconstitution syndrome. A causal relationship with darunavir co-administered with ritonavir has not been established.
Appropriate laboratory testing should be conducted prior to initiating therapy with PREZCOBIX and patients should be monitored during treatment. Increased AST/ALT monitoring should be considered in patients with underlying chronic hepatitis, cirrhosis, or in patients who have pre-treatment elevations of transaminases, especially during the first several months of PREZCOBIX treatment.
Evidence of new or worsening liver dysfunction (including clinically significant elevation of liver enzymes and/or symptoms such as fatigue, anorexia, nausea, jaundice, dark urine, liver tenderness, hepatomegaly) in patients on PREZCOBIX should prompt consideration of interruption or discontinuation of treatment.
14 Clinical Studies
4 Contraindications
Darunavir and cobicistat are both inhibitors of the cytochrome P450 3A (CYP3A) isoform. PREZCOBIX should not be co-administered with medicinal products that are highly dependent on CYP3A for clearance and for which increased plasma concentrations are associated with serious and/or life-threatening events (narrow therapeutic index). Darunavir and cobicistat are both substrates of the cytochrome P450 3A (CYP3A) isoform. Co-administration of PREZCOBIX with CYP3A inducers may lead to lower exposures of darunavir and cobicistat and potential loss of efficacy of darunavir and possible resistance. Examples of drugs that are contraindicated for co-administration with PREZCOBIX [see Drug Interactions (7.3)and Clinical Pharmacology (12.3)] are listed below.
- Alpha 1-adrenoreceptor antagonist: alfuzosin
- Anticonvulsants: carbamazepine, phenobarbital, phenytoin
- Anti-gout: colchicine, in patients with renal and/or hepatic impairment
- Antimycobacterial: rifampin
- Antipsychotics: lurasidone, pimozide
- Cardiac Disorders: dronedarone, ivabradine, ranolazine
- Ergot derivatives, e.g. dihydroergotamine, ergotamine, methylergonovine
- Herbal product: St. John's wort ( Hypericum perforatum)
- Hepatitis C direct acting antiviral: elbasvir/grazoprevir
- Lipid modifying agents: lomitapide, lovastatin, simvastatin
- Opioid Antagonist: naloxegol
- PDE-5 inhibitor: sildenafil when used for treatment of pulmonary arterial hypertension
- Sedatives/hypnotics: orally administered midazolam, triazolam
6 Adverse Reactions
The following adverse reactions are discussed in other sections of the labeling:
- Hepatotoxicity [see Warnings and Precautions (5.1)]
- Severe skin reactions [see Warnings and Precautions (5.2)]
- Effects on serum creatinine [see Warnings and Precautions (5.3)]
- New onset or worsening renal impairment when used with tenofovir DF [see Warnings and Precautions (5.4)]
- Immune Reconstitution Syndrome [see Warnings and Precautions (5.10)]
7 Drug Interactions
8.7 Renal Impairment
A renal impairment trial was not conducted for darunavir co-administered with cobicistat [see Clinical Pharmacology (12.3)] . Cobicistat has been shown to decrease estimated creatinine clearance without affecting actual renal glomerular function. Dosing recommendations are not available for drugs that require dosage adjustment for renal impairment when used in combination with PREZCOBIX [see Warnings and Precautions (5.3)and Clinical Pharmacology (12.2)] .
12.2 Pharmacodynamics
12.3 Pharmacokinetics
The pharmacokinetics of darunavir co-administered with cobicistat (150 mg) have been evaluated in healthy adults and in adults with HIV-1.
Darunavir is primarily metabolized by CYP3A. Cobicistat inhibits CYP3A, thereby increasing the plasma concentrations of darunavir.
Under fed (535 total kcal, 171 kcal from fat, 268 kcal from carbohydrates, 96 kcal from protein) and fasted conditions in healthy participants, the 90% confidence intervals when comparing darunavir exposure between PREZCOBIX (800 mg/150 mg) and darunavir 800 mg co-administered with cobicistat 150 mg as single entities were within 80–125%. No clinically significant difference was observed between PREZCOBIX (675 mg/150 mg) and darunavir 675 mg co-administered with cobicistat 150 mg as single entities under fed conditions.
Darunavir exposure when comparing darunavir co-administered with cobicistat (as single entities) to darunavir co-administered with ritonavir was evaluated in a relative bioavailability trial [see cobicistat full prescribing information].Table 3 displays the pharmacokinetic estimates of darunavir after oral administration of darunavir 800 mg co-administered with ritonavir 100 mg once daily (based on sparse sampling in 335 participants in Trial TMC114-C211 and 280 participants in Trial TMC114-C229) and darunavir 800 mg co-administered with cobicistat 150 mg once daily administered as single entities (based on sparse sampling in 298 participants in Trial GS-US-216-0130) to HIV-1 participants.
| Trial TMC114-C211
(treatment-naïve) Darunavir 800 mg co-administered with ritonavir 100 mg once daily |
Trial TMC114-C229
(treatment-experienced) Darunavir 800 mg co-administered with ritonavir 100 mg once daily |
Trial GS-US-216-0130
(treatment-naïve and experienced) Darunavir 800 mg co-administered with cobicistat 150 mg once daily |
|
|---|---|---|---|
| Parameter | N=335 | N=280 | N=298 |
| AUC 0–24h=area under the concentration-time curve over a 24-hour dosing interval at steady state; C 0h=plasma concentration at the end of a 24-hour dosing interval at steady state; N=number of participants; SD=standard deviation | |||
| AUC 0–24h(ng∙h/mL) | |||
| Mean ± SD | 93026 ± 27050 | 93334 ± 28626 | 100152 ± 32042 |
| Median (Range) | 87854 (45000–219240) | 87788 (45456–236920) | 96900 (34500–224000) |
| C 0h(ng/mL) | |||
| Mean ± SD | 2282 ± 1168 | 2160 ± 1201 | 2043 ± 1257 |
| Median (Range) | 2041 (368–7242) | 1896 (184–7881) | 1875 (70–6890) |
5.9 Fat Redistribution
Redistribution/accumulation of body fat, including central obesity, dorsocervical fat enlargement (buffalo hump), peripheral wasting, facial wasting, breast enlargement, and "cushingoid appearance" have been observed in patients receiving antiretroviral therapy. The mechanism and long-term consequences of these events are currently unknown. A causal relationship has not been established.
8.6 Hepatic Impairment
No clinical trials were conducted with darunavir co-administered with cobicistat in hepatically impaired patients and the effect of hepatic impairment on darunavir exposure when co-administered with cobicistat has not been evaluated. Based on the recommendations for darunavir co-administered with ritonavir, a dose adjustment for patients with mild or moderate hepatic impairment is not necessary. No pharmacokinetic or safety data are available regarding the use of darunavir in patients with severe hepatic impairment. Therefore, PREZCOBIX is not recommended for use in patients with severe hepatic impairment [see Clinical Pharmacology (12.3)] .
1 Indications and Usage
PREZCOBIX is indicated in combination with other antiretroviral agents for the treatment of human immunodeficiency virus (HIV-1) in treatment-naïve and treatment-experienced adults and pediatric patients weighing at least 25 kg with no darunavir resistance-associated substitutions (V11I, V32I, L33F, I47V, I50V, I54L, I54M, T74P, L76V, I84V, L89V).
12 Clinical Pharmacology
12.1 Mechanism of Action
PREZCOBIX is a fixed-dose combination of an HIV-1 antiviral drug, darunavir and a CYP3A inhibitor, cobicistat [see Microbiology (12.4)].
5.2 Severe Skin Reactions
During the darunavir clinical development program (n=3063), where darunavir was co-administered with ritonavir 100 mg once or twice daily, severe skin reactions, accompanied by fever and/or elevations of transaminases in some cases, was reported in 0.4% of participants. Stevens-Johnson Syndrome was rarely (less than 0.1%) reported during the clinical development program. During post-marketing experience toxic epidermal necrolysis, drug rash with eosinophilia and systemic symptoms, and acute generalized exanthematous pustulosis have been reported. Discontinue PREZCOBIX immediately if signs or symptoms of severe skin reactions develop. These can include but are not limited to severe rash or rash accompanied with fever, general malaise, fatigue, muscle or joint aches, blisters, oral lesions, conjunctivitis, hepatitis and/or eosinophilia.
Mild-to-moderate rash was also reported and often occurred within the first four weeks of treatment and resolved with continued dosing.
5 Warnings and Precautions
2 Dosage and Administration
3 Dosage Forms and Strengths
PREZCOBIX 800 mg darunavir/150 mg cobicistat is a pink, oval-shaped, film-coated tablet debossed with “800” on one side and “TG” on the other side.
PREZCOBIX 675 mg darunavir/150 mg cobicistat is a green to dark green, oval-shaped, scored film-coated tablet debossed with “675” on one side and “TG” on the other side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of darunavir. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
5.3 Effects On Serum Creatinine
Cobicistat decreases estimated creatinine clearance due to inhibition of tubular secretion of creatinine without affecting actual renal glomerular function. This effect should be considered when interpreting changes in estimated creatinine clearance in patients initiating PREZCOBIX, particularly in patients with medical conditions or receiving drugs needing monitoring with estimated creatinine clearance.
Prior to initiating therapy with PREZCOBIX, assess estimated creatinine clearance [see Dosage and Administration (2.2)] . Dosage recommendations are not available for drugs that require dosage adjustments in PREZCOBIX-treated patients with renal impairment [see Drug Interactions (7.3) and Clinical Pharmacology (12.2)] . Consider alternative medications that do not require dosage adjustments in patients with renal impairment.
Although cobicistat may cause modest increases in serum creatinine and modest declines in estimated creatinine clearance without affecting renal glomerular function, patients who experience a confirmed increase in serum creatinine of greater than 0.4 mg/dL from baseline should be closely monitored for renal safety.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
5.10 Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including PREZCOBIX. During the initial phase of combination antiretroviral treatment, patients whose immune systems respond may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium avium infection, cytomegalovirus, Pneumocystis jirovecii pneumonia [PCP], or tuberculosis), which may necessitate further evaluation and treatment.
Autoimmune disorders (such as Graves' disease, polymyositis, Guillain-Barré syndrome and autoimmune hepatitis) have also been reported to occur in the setting of immune reconstitution; however, the time to onset is more variable, and can occur many months after initiation of antiretroviral treatment.
5.6 Antiretrovirals Not Recommended
PREZCOBIX is not recommended in combination with other antiretroviral drugs that require pharmacokinetic boosting (i.e., another protease inhibitor or elvitegravir) because dosing recommendations for such combinations have not been established and co-administration may result in decreased plasma concentrations of the antiretroviral agents, leading to loss of therapeutic effect and development of resistance.
PREZCOBIX is not recommended in combination with products containing the individual components of PREZCOBIX (darunavir and cobicistat) or with ritonavir. For additional recommendations on use of PREZCOBIX with other antiretroviral agents, [see Drug Interactions (7)] .
5.8 Diabetes Mellitus/hyperglycemia
New onset diabetes mellitus, exacerbation of pre-existing diabetes mellitus, and hyperglycemia have been reported during postmarketing surveillance in patients with HIV-1 receiving HIV protease inhibitor (PI) therapy. Some patients required either initiation or dose adjustments of insulin or oral hypoglycemic agents for treatment of these events. In some cases, diabetic ketoacidosis has occurred. In those patients who discontinued PI therapy, hyperglycemia persisted in some cases. Because these events have been reported voluntarily during clinical practice, estimates of frequency cannot be made and causal relationships between HIV PI therapy and these events have not been established.
16 How Supplied/storage and Handling
PREZCOBIX ® (darunavir and cobicistat) tablets, 800/150 mg, are supplied as pink, oval-shaped, film-coated tablets debossed with "800" on one side and "TG" on the other side. Bottle of 30 tablets (NDC 59676-575-30).
PREZCOBIX ®(darunavir and cobicistat) tablets, 675/150 mg, are supplied as green to dark green, oval-shaped, scored film-coated tablet debossed with “675” on one side and “TG” on the other side. Bottle of 30 tablets (NDC 59676-578-30).
2.5 Not Recommended During Pregnancy
PREZCOBIX is not recommended during pregnancy because of substantially lower exposures of darunavir and cobicistat during the second and third trimesters [see Use in Specific Populations (8.1)and Clinical Pharmacology (12.3)] .
PREZCOBIX should not be initiated in pregnant individuals. An alternative regimen is recommended for those who become pregnant during therapy with PREZCOBIX.
2.3 Not Recommended in Severe Renal Impairment
PREZCOBIX co-administered with tenofovir DF is not recommended in patients who have an estimated creatinine clearance below 70 mL per minute [see Warnings and Precautions (5.4) and Adverse Reactions (6.1)] .
14.1 Clinical Trial Results in Adults With Hiv 1
The efficacy of PREZCOBIX in adults with HIV-1 is based on efficacy demonstrated in clinical trials of darunavir co-administered with ritonavir [see darunavir full prescribing information].
2.4 Not Recommended in Severe Hepatic Impairment
PREZCOBIX is not recommended for use in patients with severe hepatic impairment [see Use in Specific Populations (8.6)and Clinical Pharmacology (12.3)] .
7.1 Potential for Prezcobix to Affect Other Drugs
Darunavir co-administered with cobicistat is an inhibitor of CYP3A and CYP2D6. Cobicistat inhibits the following transporters: P-glycoprotein (P-gp), BCRP, MATE1, OATP1B1 and OATP1B3. Therefore, co-administration of PREZCOBIX with drugs that are primarily metabolized by CYP3A and/or CYP2D6 or are substrates of P-gp, BCRP, MATE1, OATP1B1 or OATP1B3 may result in increased plasma concentrations of such drugs, which could increase or prolong their therapeutic effect and can be associated with adverse events. Co-administration of PREZCOBIX with drugs that have active metabolite(s) formed by CYP3A may result in reduced plasma concentrations of these active metabolite(s), potentially leading to loss of their therapeutic effect (see Table 2).
7.2 Potential for Other Drugs to Affect Prezcobix
Darunavir is metabolized by CYP3A. Cobicistat is metabolized by CYP3A, and to a minor extent, by CYP2D6. Co-administration of PREZCOBIX and drugs that induce CYP3A activity are expected to increase the clearance of darunavir and cobicistat, resulting in lowered plasma concentrations of darunavir and cobicistat which may lead to loss of therapeutic effect and development of resistance. Co-administration of PREZCOBIX and other drugs that inhibit CYP3A may result in increased plasma concentrations of darunavir and cobicistat (see Table 2).
14.2 Clinical Trial Results in Pediatrics With Hiv 1
Trial GS-US-216-0128 was a Phase 2/3 multicenter, open-label trial to evaluate the pharmacokinetics, safety, and efficacy of darunavir co-administered with cobicistat in adolescents aged 12 years and older (Cohort 1) and children aged 6 to less than 12 years old (Cohort 2) with HIV-1 who were virologically suppressed and had a baseline estimated creatinine clearance ≥90 mL/min/1.73 m 2. Participants were on a stable antiretroviral regimen including at least 2 NRTIs (for at least 3 months).
Principal Display Panel 800 Mg/150 Mg Tablet Bottle Label
NDC 59676-575-30
PREZCOBIX
®
(darunavir and cobicistat)
Tablets
800 mg/150 mg
ALERT: Find out about medicines
that should NOT be taken with PREZCOBIX ®
from your healthcare provider.
Rx only
30 Tablets
Johnson
&Johnson
Principal Display Panel 675 Mg/150 Mg Tablet Bottle Carton
NDC 59676-578-30
PREZCOBIX
®
(darunavir and
cobicistat)
Tablets
675 mg/150 mg
ALERT: Find out about medicines that
should NOT be taken with PREZCOBIX
®
from your healthcare provider.
Rx only
30 Tablets
Johnson
& Johnson
7.3 Established and Other Potentially Significant Drug Interactions
Table 2 provides dosing recommendations for expected clinically relevant interactions with PREZCOBIX (this table is not all inclusive). These recommendations are based on either drug interaction trials or predicted interactions due to the expected magnitude of interaction and potential for serious adverse events or loss of therapeutic effect. The table includes examples of potentially significant interactions but is not all inclusive , and therefore the label of each drug that is co-administered with PREZCOBIX should be consulted for information related to the route of metabolism, interaction pathways, potential risks, and specific actions to be taken with regard to co-administration. For the list of examples of contraindicated drugs, [see Contraindications (4)] .
| Concomitant Drug Class:
Drug Name Examples |
Effect on Concentration of Darunavir, Cobicistat, or Concomitant Drug | Clinical Comment |
|---|---|---|
| HIV-1 antiviral agents: Nucleoside Reverse Transcriptase Inhibitors (NRTIs) | ||
| didanosine | ↔ darunavir
↔ cobicistat ↔ didanosine |
Didanosine should be administered one hour before or two hours after PREZCOBIX (administered with food). |
| HIV-1 antiviral agents: Non-Nucleoside Reverse Transcriptase Inhibitors (NNRTIs) | ||
| efavirenz | ↓ cobicistat
↓ darunavir |
Co-administration with efavirenz is not recommended because it may result in loss of therapeutic effect and development of resistance to darunavir. |
| etravirine | ↓ cobicistat
darunavir: effect unknown |
Co-administration with etravirine is not recommended because it may result in loss of therapeutic effect and development of resistance to darunavir. |
| nevirapine | ↓ cobicistat
darunavir: effect unknown |
Co-administration with nevirapine is not recommended because it may result in loss of therapeutic effect and development of resistance to darunavir. |
| HIV-1 antiviral agents: CCR5 co-receptor antagonists | ||
| maraviroc | ↑ maraviroc | Maraviroc is a substrate of CYP3A. When co-administered with PREZCOBIX, patients should receive maraviroc 150 mg twice daily. |
| Other agents | ||
|
Alpha 1-adrenoreceptor antagonist:
alfuzosin |
↑ alfuzosin | Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as hypotension. |
|
Antibacterials:
clarithromycin, erythromycin, telithromycin |
↑ darunavir
↑ cobicistat ↑ antibacterial |
Consider alternative antibiotics with concomitant use of PREZCOBIX. |
|
Anticancer agents:
dasatinib, nilotinib |
↑ anticancer agent | A decrease in the dosage or an adjustment of the dosing interval of dasatinib or nilotinib may be necessary when co-administered with PREZCOBIX. Consult the dasatinib and nilotinib prescribing information for dosing instructions. |
| vinblastine, vincristine | For vincristine and vinblastine, consider temporarily withholding the cobicistat-containing antiretroviral regimen in patients who develop significant hematologic or gastrointestinal side effects when PREZCOBIX is administered concurrently with vincristine or vinblastine. If the antiretroviral regimen must be withheld for a prolonged period, consider initiating a revised regimen that does not include a CYP3A or P-gp inhibitor. | |
|
Anticoagulants:
Direct Oral Anticoagulants (DOACs) apixaban |
↑ apixaban | Due to potentially increased bleeding risk, dosing recommendations for co-administration of apixaban with PREZCOBIX depend on the apixaban dose. Refer to apixaban dosing instructions for co-administration with P-gp and strong CYP3A inhibitors in apixaban prescribing information. |
| rivaroxaban | ↑ rivaroxaban | Co-administration of rivaroxaban with PREZCOBIX is not recommended because it may lead to an increased bleeding risk. |
| dabigatran etexilate
edoxaban |
↑ dabigatran
↑ edoxaban |
Refer to the dabigatran etexilate or edoxaban prescribing information for recommendations regarding co-administration. The specific recommendations are based on indication, renal function, and effect of the co-administered P-gp inhibitors on the concentration of dabigatran or edoxaban. Clinical monitoring is recommended when a DOAC not affected by CYP3A4 but transported by P-gp, including dabigatran etexilate and edoxaban, is co-administered with PREZCOBIX. |
| Other Anticoagulants: | ||
| warfarin | warfarin: effect unknown | Monitor the international normalized ratio (INR) when co-administering with warfarin. |
|
Anticonvulsants:
carbamazepine, phenobarbital, phenytoin |
↓ darunavir
↓ cobicistat |
Co-administration is contraindicated due to potential for reduced plasma concentrations of darunavir, which may result in loss of therapeutic effect and development of resistance. |
|
Anticonvulsants with CYP3A induction effects that are NOT contraindicated:
e.g. eslicarbazepine, oxcarbazepine |
↓ cobicistat
darunavir: effect unknown |
Consider alternative anticonvulsant or antiretroviral therapy to avoid potential changes in exposures. If co-administration is necessary, monitor for lack or loss of virologic response. |
|
Anticonvulsants that are metabolized by CYP3A:
e.g. clonazepam |
↑ clonazepam | Clinical monitoring of anticonvulsants is recommended. |
|
Antidepressants:
Selective Serotonin Reuptake Inhibitors (SSRIs): e.g. paroxetine, sertraline |
SSRIs: effects unknown | When co-administering with SSRIs, TCAs, or trazodone, careful dose titration of the antidepressant to the desired effect, including using the lowest feasible initial or maintenance dose, and monitoring for antidepressant response are recommended. |
|
Tricyclic Antidepressants (TCAs):
e.g. amitriptyline, desipramine, imipramine, nortriptyline |
↑ TCAs | |
|
Other antidepressants:
trazodone |
↑ trazodone | |
|
Antifungals:
itraconazole, isavuconazole, ketoconazole, posaconazole |
↑ darunavir
↑ cobicistat |
Monitor for increased darunavir or cobicistat and/or antifungal adverse reactions. |
| ↑ itraconazole
↑ ketoconazole ↑ isavuconazole ↔ posaconazole |
Specific dosing recommendations are not available for co-administration with these antifungals. Monitor for increased itraconazole or ketoconazole adverse reactions. | |
| voriconazole | voriconazole: effects unknown | Co-administration with voriconazole is not recommended unless benefit/risk assessment justifies the use of voriconazole. |
|
Anti-gout:
colchicine |
↑ colchicine | Co-administration is contraindicated in patients with renal and/or hepatic impairment due to potential for serious and/or life-threatening reactions.
For patients without renal or hepatic impairment:
|
|
Antimalarial:
artemether/lumefantrine |
artemether: effect unknown
lumefantrine: effect unknown |
Monitor for a potential decrease of antimalarial efficacy or potential QT prolongation. |
| Antimycobacterials: | ||
| rifampin | ↓ darunavir
↓ cobicistat |
Co-administration is contraindicated due to potential for loss of therapeutic effect and development of resistance. |
| rifabutin | ↑ rifabutin
cobicistat: effects unknown darunavir: effects unknown |
When used in combination with PREZCOBIX, the recommended dose of rifabutin is 150 mg every other day. Monitor for rifabutin-associated adverse reactions including neutropenia and uveitis. |
| rifapentine | ↓ darunavir | Co-administration with rifapentine is not recommended. |
|
Antipsychotics:
lurasidone |
↑ lurasidone | Co-administration is contraindicated due to potential for serious and/or life-threatening reactions. |
| pimozide | ↑ pimozide | Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as cardiac arrhythmias. |
| e.g. perphenazine, risperidone, thioridazine | ↑ antipsychotic | A decrease in the dose of antipsychotics that are metabolized by CYP3A or CYP2D6 may be needed when co-administered with PREZCOBIX. |
| quetiapine | ↑ quetiapine |
Initiation of PREZCOBIX in patients taking quetiapine: Consider alternative antiretroviral therapy to avoid increases in quetiapine exposure. If co-administration is necessary, reduce the quetiapine dose to 1/6 of the current dose and monitor for quetiapine- associated adverse reactions. Refer to the quetiapine prescribing information for recommendations on adverse reaction monitoring.
Initiation of quetiapine in patients taking PREZCOBIX: Refer to the quetiapine prescribing information for initial dosing and titration of quetiapine. |
|
β-Blockers:
e.g. carvedilol, metoprolol, timolol |
↑ beta-blockers | Clinical monitoring is recommended for co-administration with beta-blockers that are metabolized by CYP2D6. |
|
Calcium channel blockers:
e.g. amlodipine, diltiazem, felodipine, nifedipine, verapamil |
↑ calcium channel blockers | Clinical monitoring is recommended for co-administration with calcium channel blockers metabolized by CYP3A. |
| Cardiac Disorders: | ||
| ranolazine, ivabradine | ↑ ranolazine
↑ ivabradine |
Co-administration is contraindicated due to potential for serious and/or life-threatening reactions. |
| dronedarone | ↑ dronedarone | Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as cardiac arrhythmias. |
| Other antiarrhythmics | ||
| e.g. amiodarone, disopyramide, flecainide, lidocaine (systemic), mexiletine, propafenone, quinidine | ↑ antiarrhythmics | Clinical monitoring is recommended upon co-administration with antiarrhythmics. |
| digoxin | ↑ digoxin | When co-administering with digoxin, titrate the digoxin dose and monitor digoxin concentrations. |
|
Corticosteroids:
dexamethasone (systemic) Corticosteroids primarily metabolized by CYP3A: e.g. betamethasone budesonide ciclesonide fluticasone methylprednisolone mometasone triamcinolone |
↓ darunavir
↓ cobicistat ↑ corticosteroids |
Co-administration with systemic dexamethasone or other systemic corticosteroids that induce CYP3A may result in loss of therapeutic effect and development of resistance to PREZCOBIX. Consider alternative corticosteroids.
Co-administration with corticosteroids (all routes of administration) of which exposures are significantly increased by strong CYP3A inhibitors can increase the risk for Cushing's syndrome and adrenal suppression. Alternative corticosteroids including beclomethasone, prednisone and prednisolone (for which PK and/or PD are less affected by strong CYP3A inhibitors relative to other steroids) should be considered, particularly for long-term use. |
|
Endothelin receptor antagonists:
bosentan |
↓ darunavir
↓ cobicistat ↑ bosentan |
Initiation of bosentan in patients taking PREZCOBIX: In patients who have been receiving PREZCOBIX for at least 10 days, start bosentan at 62.5 mg once daily or every other day based upon individual tolerability.
Initiation of PREZCOBIX in patients on bosentan: Discontinue use of bosentan at least 36 hours prior to initiation of PREZCOBIX. After at least 10 days following the initiation of PREZCOBIX, resume bosentan at 62.5 mg once daily or every other day based upon individual tolerability. Switching from darunavir co-administered with ritonavir to PREZCOBIX in patients on bosentan: Maintain bosentan dose. |
|
Ergot derivatives:
e.g. dihydroergotamine, ergotamine, methylergonovine |
↑ ergot derivatives | Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as acute ergot toxicity characterized by peripheral vasospasm and ischemia of the extremities and other tissues. |
|
Hepatitis C virus (HCV):
Direct-Acting Antivirals: elbasvir/grazoprevir |
↑ elbasvir/grazoprevir | Co-administration is contraindicated due to potential for the increased risk of alanine transaminase (ALT) elevations. |
| glecaprevir/pibrentasvir | ↑ glecaprevir
↑ pibrentasvir |
Co-administration of PREZCOBIX with glecaprevir/pibrentasvir is not recommended. |
|
Herbal product:
St. John's wort ( Hypericum perforatum) |
↓ darunavir
↓ cobicistat |
Co-administration is contraindicated due to potential for reduced plasma concentrations of darunavir, which may result in loss of therapeutic effect and development of resistance. |
| Hormonal contraceptives: | Additional or alternative (non-hormonal) forms of contraception should be considered when estrogen-containing contraceptives are co-administered with PREZCOBIX [see Use in Specific Populations (8.3)] . | |
| drospirenone/ethinylestradiol | ↑ drospirenone
↓ ethinylestradiol |
For co-administration with drospirenone, clinical monitoring is recommended due to the potential for hyperkalemia. |
| Other progestin/estrogen contraceptives | progestin: effects unknown
estrogen: effects unknown |
No data are available to make recommendations on co-administration with other hormonal contraceptives. |
|
Immunosuppressants:
cyclosporine, sirolimus, tacrolimus |
↑ immunosuppressants | These immunosuppressant agents are metabolized by CYP3A. Therapeutic drug monitoring is recommended with concomitant use |
|
Immunosuppressant /neoplastic:
everolimus |
↑ immunosuppressants |
Co-administration of everolimus and PREZCOBIX is not recommended. |
| irinotecan | Discontinue PREZCOBIX at least 1 week prior to starting irinotecan therapy. Do not administer PREZCOBIX with irinotecan unless there are no therapeutic alternatives. | |
|
Inhaled beta agonist:
salmeterol |
↑ salmeterol | Co-administration with salmeterol is not recommended and may result in increased risk of cardiovascular adverse events associated with salmeterol, including QT prolongation, palpitations, and sinus tachycardia. |
| Lipid Modifying Agents | ||
|
HMG-CoA reductase inhibitors:
lovastatin, simvastatin |
↑ lovastatin
↑ simvastatin |
Co-administration is contraindicated due to potential for serious reactions such as myopathy including rhabdomyolysis. |
| atorvastatin, fluvastatin, pitavastatin, pravastatin, rosuvastatin | ↑ atorvastatin
↑ fluvastatin ↑ pravastatin ↑ rosuvastatin pitavastatin: effect unknown |
For atorvastatin, fluvastatin, pitavastatin, pravastatin, and rosuvastatin, start with the lowest recommended dose and titrate while monitoring for safety (e.g. myopathy).
Dosage recommendations with atorvastatin or rosuvastatin are as follows:
|
|
Other lipid modifying agents:
lomitapide |
↑ lomitapide | Co-administration is contraindicated due to potential for markedly increased transaminases associated with increased plasma concentrations of lomitapide. |
|
Narcotic analgesics metabolized by CYP3A:
e.g. fentanyl, oxycodone |
↑ fentanyl
↑ oxycodone |
Careful monitoring of therapeutic effects and adverse reactions associated with CYP3A-metabolized narcotic analgesics (including potentially fatal respiratory depression) is recommended with co-administration. |
| tramadol | ↑ tramadol | A dose decrease may be needed for tramadol with concomitant use. |
|
Narcotic analgesic for treatment of opioid dependence:
buprenorphine, buprenorphine/naloxone, methadone |
buprenorphine or buprenorphine/ naloxone: effects unknown
methadone: effects unknown |
Initiation of buprenorphine, buprenorphine/naloxone or methadone in patients taking PREZCOBIX: Carefully titrate the dose of buprenorphine, buprenorphine/naloxone or methadone to the desired effect; use the lowest feasible initial or maintenance dose.
Initiation of PREZCOBIX in patients taking buprenorphine, buprenorphine/naloxone or methadone: A dose adjustment for buprenorphine, buprenorphine/naloxone or methadone may be needed. Monitor clinical signs and symptoms. |
| Opioid Antagonist | ||
| naloxegol | ↑ naloxegol | Co-administration of PREZCOBIX and naloxegol is contraindicated due to potential for precipitating opioid withdrawal symptoms. |
|
Phosphodiesterase PDE-5 inhibitors:
e.g. avanafil, sildenafil, tadalafil, vardenafil |
↑ PDE-5 inhibitors | Co-administration with avanafil is not recommended because a safe and effective avanafil dosage regimen has not been established.
Co-administration with PDE-5 inhibitors may result in an increase in PDE-5 inhibitor-associated adverse reactions including hypotension, syncope, visual disturbances and priapism. Use of PDE-5 inhibitors for pulmonary arterial hypertension (PAH): Co-administration with sildenafil used for PAH is contraindicated due to potential for sildenafil associated adverse reactions (which include visual disturbances, hypotension, prolonged erection, and syncope). The following dose adjustments are recommended for use of tadalafil with PREZCOBIX:
Sildenafil at a single dose not exceeding 25 mg in 48 hours, vardenafil at a single dose not exceeding 2.5 mg dose in 72 hours, or tadalafil at a single dose not exceeding 10 mg dose in 72 hours can be used with increased monitoring for PDE-5 inhibitor-associated adverse reactions. |
| Platelet aggregation inhibitor: | ||
| ticagrelor | ↑ ticagrelor | Co-administration of PREZCOBIX and ticagrelor is not recommended. |
| clopidogrel | ↓ clopidogrel active metabolite | Co-administration of PREZCOBIX with clopidogrel is not recommended due to the potential reduction of the antiplatelet activity of clopidogrel. |
| prasugrel | ↔ prasugrel active metabolite | No dose adjustment is needed when prasugrel is co-administered with PREZCOBIX. |
|
Sedatives/hypnotics:
orally administered midazolam, triazolam |
↑ midazolam
↑ triazolam |
Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as prolonged or increased sedation or respiratory depression. Triazolam and orally administered midazolam are extensively metabolized by CYP3A. Co-administration of triazolam or orally administered midazolam with PREZCOBIX may cause large increases in the concentrations of these benzodiazepines. |
|
metabolized by CYP3A:
e.g. buspirone, diazepam, estazolam, zolpidem |
↑ sedatives/hypnotics | With concomitant use, titration is recommended with sedatives/hypnotics metabolized by CYP3A and a lower dose of the sedatives/hypnotics should be considered with monitoring for increased and prolonged effects or adverse reactions. |
| parenterally administered midazolam | Co-administration of parenteral midazolam should be done in a setting that ensures close clinical monitoring and appropriate medical management in case of respiratory depression and/or prolonged sedation. Dose reduction for parenteral midazolam should be considered, especially if more than a single dose of midazolam is administered. | |
| Urinary antispasmodics | ||
| fesoterodine | ↑ fesoterodine | When fesoterodine is co-administered with PREZCOBIX, do not exceed a fesoterodine dose of 4 mg once daily. |
| solifenacin | ↑ solifenacin | When solifenacin is co-administered with PREZCOBIX, do not exceed a solifenacin dose of 5 mg once daily. |
7.4 Drugs Without Clinically Significant Interactions With Prezcobix
Clinically relevant drug-drug interactions have not been observed or are not anticipated with concomitant use of darunavir and cobicistat with rilpivirine, dolutegravir, raltegravir, abacavir, emtricitabine, emtricitabine/tenofovir alafenamide, tenofovir DF, lamivudine, stavudine, zidovudine, or acid modifying medications (antacids, H 2-receptor antagonists, proton pump inhibitors).
2.1 Recommended Dosage in Adults and Pediatric Patients Weighing At Least 25 Kg
The recommended dosages of PREZCOBIX for adults and pediatric patients weighing at least 25 kg are shown in Table 1. The pediatric dose is based on weight. Administer PREZCOBIX orally with food in conjunction with other antiretroviral agents.
| Patient Population | Dose (once daily with food) |
|---|---|
| Adult Patients | One 800 mg darunavir/150 mg cobicistat tablet |
| Pediatric Patients weighing at least 40 kg | |
| Pediatric Patients weighing at least 25 kg to less than 40 kg | One 675 mg darunavir/150 mg cobicistat tablet |
Before prescribing PREZCOBIX 675 mg/150 mg tablets, children should be assessed for the ability to swallow tablets. For patients unable to swallow the 675 mg/150 mg tablet whole, the scored tablet may be split by hand into two pieces. Each piece should be consumed immediately after splitting to ensure the entire dose is administered. The score line is only to facilitate breaking for ease of swallowing and not to divide into equal doses.
5.4 New Onset Or Worsening Renal Impairment When Used With Tenofovir Disoproxil Fumarate
Renal impairment, including cases of acute renal failure and Fanconi syndrome, has been reported when cobicistat, a component of PREZCOBIX, was used in an antiretroviral regimen that contained tenofovir DF. Co-administration of PREZCOBIX and tenofovir DF is not recommended in patients who have an estimated creatinine clearance below 70 mL/min [see Dosage and Administration (2.3)] .
- Document urine glucose and urine protein at baseline [see Dosage and Administration (2.2)] and perform routine monitoring of estimated creatinine clearance, urine glucose, and urine protein during treatment when PREZCOBIX is used with tenofovir DF. Measure serum phosphorus in patients with or at risk for renal impairment when used with tenofovir DF.
- Co-administration of PREZCOBIX and tenofovir DF in combination with concomitant or recent use of a nephrotoxic agent is not recommended.
See cobicistat full prescribing information for additional information regarding cobicistat.
5.5 Risk of Serious Adverse Reactions Or Loss of Virologic Response Due to Drug Interactions
Initiation of PREZCOBIX, which inhibits CYP3A, in patients receiving medications metabolized by CYP3A, or initiation of medications metabolized by CYP3A in patients already receiving PREZCOBIX may increase plasma concentrations of medications metabolized by CYP3A and reduce plasma concentrations of active metabolite(s) formed by CYP3A. Initiation of medications that inhibit or induce CYP3A may respectively increase or decrease concentrations of PREZCOBIX.
These interactions may lead to:
- clinically significant adverse reactions, potentially leading to severe, life threatening, or fatal events from higher exposures of concomitant medications.
- clinically significant adverse reactions from higher exposures of PREZCOBIX.
- loss of therapeutic effect of the concomitant medications from lower exposures of active metabolite(s).
- loss of therapeutic effect of PREZCOBIX and possible development of resistance from lower exposures of PREZCOBIX.
See Table 2 for steps to prevent or manage these possible and known significant drug interactions, including dosing recommendations. Consider the potential for drug interactions prior to and during PREZCOBIX therapy; review concomitant medications during PREZCOBIX therapy; and monitor for the adverse reactions associated with concomitant medications [see Contraindications (4) and Drug Interactions (7)] .
When used with concomitant medications, PREZCOBIX may result in different drug interactions than those observed or expected with darunavir co-administered with ritonavir. Complex or unknown mechanisms of drug interactions preclude extrapolation of drug interactions with darunavir co-administered with ritonavir to certain PREZCOBIX interactions [see Drug Interactions (7) and Clinical Pharmacology (12.3)] .
Structured Label Content
Section 42229-5 (42229-5)
HIV Genotypic Testing
HIV genotypic testing is recommended for antiretroviral treatment-experienced patients. However, when HIV genotypic testing is not feasible, PREZCOBIX can be used in protease inhibitor-naïve patients, but is not recommended in protease inhibitor-experienced patients.
Section 42230-3 (42230-3)
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 03/2025 | |
| PATIENT INFORMATION
PREZCOBIX ® (prez-koe-bix) (darunavir and cobicistat) tablets |
||
|
What is the most important information I should know about PREZCOBIX?
PREZCOBIX may cause serious side effects, including:
|
||
|
|
|
|
||
|
|
|
|
||
|
What is PREZCOBIX?
PREZCOBIX is a prescription medicine that is used with other human immunodeficiency virus (HIV-1) medicines to treat HIV-1 in adults and in children who weigh at least 55 pounds (25 kg). HIV-1 is the virus that causes Acquired Immune Deficiency Syndrome (AIDS). PREZCOBIX contains the prescription medicines darunavir and cobicistat. It is not known if PREZCOBIX is safe and effective in children weighing less than 55 pounds (25 kg). |
||
Do not take PREZCOBIX with any medicine that contains:
|
||
Before taking PREZCOBIX, tell your healthcare provider about all your medical conditions, including if you:
|
||
How should I take PREZCOBIX?
|
||
What are the possible side effects of PREZCOBIX?
PREZCOBIX may cause serious side effects, including:
|
||
|
|
|
| These are not all of the possible side effects of PREZCOBIX.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store PREZCOBIX?
|
||
|
General information about the safe and effective use of PREZCOBIX.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use PREZCOBIX for a condition for which it was not prescribed. Do not give PREZCOBIX to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about PREZCOBIX that is written for health professionals. |
||
|
What are the ingredients in PREZCOBIX?
Active ingredients: darunavir and cobicistat Inactive ingredients 800 mg/150 mg tablet: colloidal silicon dioxide, crospovidone, hypromellose, magnesium stearate, and silicified microcrystalline cellulose. The tablets are film-coated with a coating material containing iron oxide black, iron oxide red, polyethylene glycol, polyvinyl alcohol (partially hydrolyzed), talc, and titanium dioxide. Inactive ingredients 675 mg/150 mg tablet: colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, and microcrystalline cellulose. The tablets are film-coated with a coating material containing iron oxide black, iron oxide yellow, polyethylene glycol, polyvinyl alcohol (partially hydrolyzed), talc, and titanium dioxide. Manufactured for: Janssen Products, LP, Horsham, PA 19044, USA For patent information: www.janssenpatents.com © Johnson & Johnson and its affiliates 2015, 2025 For more information call 1-800-526-7736. |
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Storage: Store at 20 °C–25 °C (between 68 °F–77 °F); with excursions permitted to 15 °C–30 °C (59 °F–86 °F) [see USP Controlled Room Temperature].
Keep PREZCOBIX and all medicines out of reach of children.
10 Overdosage (10 OVERDOSAGE)
Human experience of acute overdose with PREZCOBIX is limited. No specific antidote is available for overdose with PREZCOBIX. Treatment of overdose with PREZCOBIX consists of general supportive measures including monitoring of vital signs and observation of the clinical status of the patient. Since both darunavir and cobicistat are highly protein bound, dialysis is unlikely to be beneficial in significant removal of the active substance.
11 Description (11 DESCRIPTION)
PREZCOBIX ® is a fixed-dose combination tablet containing darunavir and cobicistat. Darunavir is an inhibitor of the human immunodeficiency virus (HIV-1) protease. Cobicistat is a mechanism-based inhibitor of cytochrome P450 (CYP) enzymes of the CYP3A family.
PREZCOBIX ® 800 mg darunavir/150 mg cobicistat tablets are for oral administration. Each tablet contains darunavir ethanolate equivalent to 800 mg of darunavir and 150 mg of cobicistat. The tablets include the following inactive ingredients: colloidal silicon dioxide, crospovidone, hypromellose, magnesium stearate, and silicified microcrystalline cellulose. The tablets are film-coated with a coating material containing iron oxide black, iron oxide red, polyethylene glycol, polyvinyl alcohol (partially hydrolyzed), talc, and titanium dioxide.
PREZCOBIX ® 675 mg darunavir/150 mg cobicistat tablets are for oral administration. Each tablet contains darunavir ethanolate equivalent to 675 mg of darunavir and 150 mg of cobicistat. The tablets include the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, and microcrystalline cellulose. The tablets are film-coated with a coating material containing iron oxide black, iron oxide yellow, polyethylene glycol, polyvinyl alcohol (partially hydrolyzed), talc, and titanium dioxide.
5.11 Hemophilia
There have been reports of increased bleeding, including spontaneous skin hematomas and hemarthrosis in patients with hemophilia type A and B treated with HIV PIs. In some patients, additional factor VIII was given. In more than half of the reported cases, treatment with HIV PIs was continued or reintroduced if treatment had been discontinued. A causal relationship between PI therapy and these episodes has not been established.
12.4 Microbiology
5.7 Sulfa Allergy
Darunavir contains a sulfonamide moiety. Monitor patients with a known sulfonamide allergy after initiating PREZCOBIX. In clinical studies with darunavir co-administered with ritonavir, the incidence and severity of rash were similar in subjects with or without a history of sulfonamide allergy.
8.4 Pediatric Use
The safety and effectiveness of PREZCOBIX for the treatment of HIV-1 in pediatric patients weighing at least 25 kg was established through a trial with components of PREZCOBIX. Use of PREZCOBIX in this group is supported by evidence from adequate and well-controlled studies in adults with additional pharmacokinetic, safety, and virologic data from a study of components of PREZCOBIX (Trial GS-US-216-0128) in pediatric participants with HIV-1 aged 6 to less than 18 years [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.2)] .
The safety and effectiveness of PREZCOBIX have not been established in pediatric patients weighing less than 25 kg. Darunavir, a component of PREZCOBIX is not recommended in pediatric patients below 3 years of age because of toxicity and mortality observed in juvenile rats dosed with darunavir.
8.5 Geriatric Use
Clinical trials of PREZCOBIX did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently from younger patients. In general, caution should be exercised in the administration and monitoring of PREZCOBIX in elderly patients, reflecting the greater frequency of decreased hepatic function, and of concomitant disease or other drug therapy [see Clinical Pharmacology (12.3)] .
5.1 Hepatotoxicity
During the darunavir clinical development program (N=3063), where darunavir was co-administered with ritonavir 100 mg once or twice daily, drug-induced hepatitis (e.g., acute hepatitis, cytolytic hepatitis) was reported in 0.5% of participants. Patients with pre-existing liver dysfunction, including chronic active hepatitis B or C, have an increased risk for liver function abnormalities including severe hepatic adverse reactions.
Post-marketing cases of liver injury, including some fatalities, have also been reported with darunavir co-administered with ritonavir. These have generally occurred in patients with advanced HIV-1 disease taking multiple concomitant medications, having co-morbidities including hepatitis B or C co-infection, and/or developing immune reconstitution syndrome. A causal relationship with darunavir co-administered with ritonavir has not been established.
Appropriate laboratory testing should be conducted prior to initiating therapy with PREZCOBIX and patients should be monitored during treatment. Increased AST/ALT monitoring should be considered in patients with underlying chronic hepatitis, cirrhosis, or in patients who have pre-treatment elevations of transaminases, especially during the first several months of PREZCOBIX treatment.
Evidence of new or worsening liver dysfunction (including clinically significant elevation of liver enzymes and/or symptoms such as fatigue, anorexia, nausea, jaundice, dark urine, liver tenderness, hepatomegaly) in patients on PREZCOBIX should prompt consideration of interruption or discontinuation of treatment.
14 Clinical Studies (14 CLINICAL STUDIES)
4 Contraindications (4 CONTRAINDICATIONS)
Darunavir and cobicistat are both inhibitors of the cytochrome P450 3A (CYP3A) isoform. PREZCOBIX should not be co-administered with medicinal products that are highly dependent on CYP3A for clearance and for which increased plasma concentrations are associated with serious and/or life-threatening events (narrow therapeutic index). Darunavir and cobicistat are both substrates of the cytochrome P450 3A (CYP3A) isoform. Co-administration of PREZCOBIX with CYP3A inducers may lead to lower exposures of darunavir and cobicistat and potential loss of efficacy of darunavir and possible resistance. Examples of drugs that are contraindicated for co-administration with PREZCOBIX [see Drug Interactions (7.3)and Clinical Pharmacology (12.3)] are listed below.
- Alpha 1-adrenoreceptor antagonist: alfuzosin
- Anticonvulsants: carbamazepine, phenobarbital, phenytoin
- Anti-gout: colchicine, in patients with renal and/or hepatic impairment
- Antimycobacterial: rifampin
- Antipsychotics: lurasidone, pimozide
- Cardiac Disorders: dronedarone, ivabradine, ranolazine
- Ergot derivatives, e.g. dihydroergotamine, ergotamine, methylergonovine
- Herbal product: St. John's wort ( Hypericum perforatum)
- Hepatitis C direct acting antiviral: elbasvir/grazoprevir
- Lipid modifying agents: lomitapide, lovastatin, simvastatin
- Opioid Antagonist: naloxegol
- PDE-5 inhibitor: sildenafil when used for treatment of pulmonary arterial hypertension
- Sedatives/hypnotics: orally administered midazolam, triazolam
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in other sections of the labeling:
- Hepatotoxicity [see Warnings and Precautions (5.1)]
- Severe skin reactions [see Warnings and Precautions (5.2)]
- Effects on serum creatinine [see Warnings and Precautions (5.3)]
- New onset or worsening renal impairment when used with tenofovir DF [see Warnings and Precautions (5.4)]
- Immune Reconstitution Syndrome [see Warnings and Precautions (5.10)]
7 Drug Interactions (7 DRUG INTERACTIONS)
8.7 Renal Impairment
A renal impairment trial was not conducted for darunavir co-administered with cobicistat [see Clinical Pharmacology (12.3)] . Cobicistat has been shown to decrease estimated creatinine clearance without affecting actual renal glomerular function. Dosing recommendations are not available for drugs that require dosage adjustment for renal impairment when used in combination with PREZCOBIX [see Warnings and Precautions (5.3)and Clinical Pharmacology (12.2)] .
12.2 Pharmacodynamics
12.3 Pharmacokinetics
The pharmacokinetics of darunavir co-administered with cobicistat (150 mg) have been evaluated in healthy adults and in adults with HIV-1.
Darunavir is primarily metabolized by CYP3A. Cobicistat inhibits CYP3A, thereby increasing the plasma concentrations of darunavir.
Under fed (535 total kcal, 171 kcal from fat, 268 kcal from carbohydrates, 96 kcal from protein) and fasted conditions in healthy participants, the 90% confidence intervals when comparing darunavir exposure between PREZCOBIX (800 mg/150 mg) and darunavir 800 mg co-administered with cobicistat 150 mg as single entities were within 80–125%. No clinically significant difference was observed between PREZCOBIX (675 mg/150 mg) and darunavir 675 mg co-administered with cobicistat 150 mg as single entities under fed conditions.
Darunavir exposure when comparing darunavir co-administered with cobicistat (as single entities) to darunavir co-administered with ritonavir was evaluated in a relative bioavailability trial [see cobicistat full prescribing information].Table 3 displays the pharmacokinetic estimates of darunavir after oral administration of darunavir 800 mg co-administered with ritonavir 100 mg once daily (based on sparse sampling in 335 participants in Trial TMC114-C211 and 280 participants in Trial TMC114-C229) and darunavir 800 mg co-administered with cobicistat 150 mg once daily administered as single entities (based on sparse sampling in 298 participants in Trial GS-US-216-0130) to HIV-1 participants.
| Trial TMC114-C211
(treatment-naïve) Darunavir 800 mg co-administered with ritonavir 100 mg once daily |
Trial TMC114-C229
(treatment-experienced) Darunavir 800 mg co-administered with ritonavir 100 mg once daily |
Trial GS-US-216-0130
(treatment-naïve and experienced) Darunavir 800 mg co-administered with cobicistat 150 mg once daily |
|
|---|---|---|---|
| Parameter | N=335 | N=280 | N=298 |
| AUC 0–24h=area under the concentration-time curve over a 24-hour dosing interval at steady state; C 0h=plasma concentration at the end of a 24-hour dosing interval at steady state; N=number of participants; SD=standard deviation | |||
| AUC 0–24h(ng∙h/mL) | |||
| Mean ± SD | 93026 ± 27050 | 93334 ± 28626 | 100152 ± 32042 |
| Median (Range) | 87854 (45000–219240) | 87788 (45456–236920) | 96900 (34500–224000) |
| C 0h(ng/mL) | |||
| Mean ± SD | 2282 ± 1168 | 2160 ± 1201 | 2043 ± 1257 |
| Median (Range) | 2041 (368–7242) | 1896 (184–7881) | 1875 (70–6890) |
5.9 Fat Redistribution
Redistribution/accumulation of body fat, including central obesity, dorsocervical fat enlargement (buffalo hump), peripheral wasting, facial wasting, breast enlargement, and "cushingoid appearance" have been observed in patients receiving antiretroviral therapy. The mechanism and long-term consequences of these events are currently unknown. A causal relationship has not been established.
8.6 Hepatic Impairment
No clinical trials were conducted with darunavir co-administered with cobicistat in hepatically impaired patients and the effect of hepatic impairment on darunavir exposure when co-administered with cobicistat has not been evaluated. Based on the recommendations for darunavir co-administered with ritonavir, a dose adjustment for patients with mild or moderate hepatic impairment is not necessary. No pharmacokinetic or safety data are available regarding the use of darunavir in patients with severe hepatic impairment. Therefore, PREZCOBIX is not recommended for use in patients with severe hepatic impairment [see Clinical Pharmacology (12.3)] .
1 Indications and Usage (1 INDICATIONS AND USAGE)
PREZCOBIX is indicated in combination with other antiretroviral agents for the treatment of human immunodeficiency virus (HIV-1) in treatment-naïve and treatment-experienced adults and pediatric patients weighing at least 25 kg with no darunavir resistance-associated substitutions (V11I, V32I, L33F, I47V, I50V, I54L, I54M, T74P, L76V, I84V, L89V).
12 Clinical Pharmacology (12 CLINICAL PHARMACOLOGY)
12.1 Mechanism of Action
PREZCOBIX is a fixed-dose combination of an HIV-1 antiviral drug, darunavir and a CYP3A inhibitor, cobicistat [see Microbiology (12.4)].
5.2 Severe Skin Reactions
During the darunavir clinical development program (n=3063), where darunavir was co-administered with ritonavir 100 mg once or twice daily, severe skin reactions, accompanied by fever and/or elevations of transaminases in some cases, was reported in 0.4% of participants. Stevens-Johnson Syndrome was rarely (less than 0.1%) reported during the clinical development program. During post-marketing experience toxic epidermal necrolysis, drug rash with eosinophilia and systemic symptoms, and acute generalized exanthematous pustulosis have been reported. Discontinue PREZCOBIX immediately if signs or symptoms of severe skin reactions develop. These can include but are not limited to severe rash or rash accompanied with fever, general malaise, fatigue, muscle or joint aches, blisters, oral lesions, conjunctivitis, hepatitis and/or eosinophilia.
Mild-to-moderate rash was also reported and often occurred within the first four weeks of treatment and resolved with continued dosing.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
PREZCOBIX 800 mg darunavir/150 mg cobicistat is a pink, oval-shaped, film-coated tablet debossed with “800” on one side and “TG” on the other side.
PREZCOBIX 675 mg darunavir/150 mg cobicistat is a green to dark green, oval-shaped, scored film-coated tablet debossed with “675” on one side and “TG” on the other side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of darunavir. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
5.3 Effects On Serum Creatinine (5.3 Effects on Serum Creatinine)
Cobicistat decreases estimated creatinine clearance due to inhibition of tubular secretion of creatinine without affecting actual renal glomerular function. This effect should be considered when interpreting changes in estimated creatinine clearance in patients initiating PREZCOBIX, particularly in patients with medical conditions or receiving drugs needing monitoring with estimated creatinine clearance.
Prior to initiating therapy with PREZCOBIX, assess estimated creatinine clearance [see Dosage and Administration (2.2)] . Dosage recommendations are not available for drugs that require dosage adjustments in PREZCOBIX-treated patients with renal impairment [see Drug Interactions (7.3) and Clinical Pharmacology (12.2)] . Consider alternative medications that do not require dosage adjustments in patients with renal impairment.
Although cobicistat may cause modest increases in serum creatinine and modest declines in estimated creatinine clearance without affecting renal glomerular function, patients who experience a confirmed increase in serum creatinine of greater than 0.4 mg/dL from baseline should be closely monitored for renal safety.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
5.10 Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including PREZCOBIX. During the initial phase of combination antiretroviral treatment, patients whose immune systems respond may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium avium infection, cytomegalovirus, Pneumocystis jirovecii pneumonia [PCP], or tuberculosis), which may necessitate further evaluation and treatment.
Autoimmune disorders (such as Graves' disease, polymyositis, Guillain-Barré syndrome and autoimmune hepatitis) have also been reported to occur in the setting of immune reconstitution; however, the time to onset is more variable, and can occur many months after initiation of antiretroviral treatment.
5.6 Antiretrovirals Not Recommended
PREZCOBIX is not recommended in combination with other antiretroviral drugs that require pharmacokinetic boosting (i.e., another protease inhibitor or elvitegravir) because dosing recommendations for such combinations have not been established and co-administration may result in decreased plasma concentrations of the antiretroviral agents, leading to loss of therapeutic effect and development of resistance.
PREZCOBIX is not recommended in combination with products containing the individual components of PREZCOBIX (darunavir and cobicistat) or with ritonavir. For additional recommendations on use of PREZCOBIX with other antiretroviral agents, [see Drug Interactions (7)] .
5.8 Diabetes Mellitus/hyperglycemia (5.8 Diabetes Mellitus/Hyperglycemia)
New onset diabetes mellitus, exacerbation of pre-existing diabetes mellitus, and hyperglycemia have been reported during postmarketing surveillance in patients with HIV-1 receiving HIV protease inhibitor (PI) therapy. Some patients required either initiation or dose adjustments of insulin or oral hypoglycemic agents for treatment of these events. In some cases, diabetic ketoacidosis has occurred. In those patients who discontinued PI therapy, hyperglycemia persisted in some cases. Because these events have been reported voluntarily during clinical practice, estimates of frequency cannot be made and causal relationships between HIV PI therapy and these events have not been established.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
PREZCOBIX ® (darunavir and cobicistat) tablets, 800/150 mg, are supplied as pink, oval-shaped, film-coated tablets debossed with "800" on one side and "TG" on the other side. Bottle of 30 tablets (NDC 59676-575-30).
PREZCOBIX ®(darunavir and cobicistat) tablets, 675/150 mg, are supplied as green to dark green, oval-shaped, scored film-coated tablet debossed with “675” on one side and “TG” on the other side. Bottle of 30 tablets (NDC 59676-578-30).
2.5 Not Recommended During Pregnancy
PREZCOBIX is not recommended during pregnancy because of substantially lower exposures of darunavir and cobicistat during the second and third trimesters [see Use in Specific Populations (8.1)and Clinical Pharmacology (12.3)] .
PREZCOBIX should not be initiated in pregnant individuals. An alternative regimen is recommended for those who become pregnant during therapy with PREZCOBIX.
2.3 Not Recommended in Severe Renal Impairment
PREZCOBIX co-administered with tenofovir DF is not recommended in patients who have an estimated creatinine clearance below 70 mL per minute [see Warnings and Precautions (5.4) and Adverse Reactions (6.1)] .
14.1 Clinical Trial Results in Adults With Hiv 1 (14.1 Clinical Trial Results in Adults with HIV-1)
The efficacy of PREZCOBIX in adults with HIV-1 is based on efficacy demonstrated in clinical trials of darunavir co-administered with ritonavir [see darunavir full prescribing information].
2.4 Not Recommended in Severe Hepatic Impairment
PREZCOBIX is not recommended for use in patients with severe hepatic impairment [see Use in Specific Populations (8.6)and Clinical Pharmacology (12.3)] .
7.1 Potential for Prezcobix to Affect Other Drugs (7.1 Potential for PREZCOBIX to Affect Other Drugs)
Darunavir co-administered with cobicistat is an inhibitor of CYP3A and CYP2D6. Cobicistat inhibits the following transporters: P-glycoprotein (P-gp), BCRP, MATE1, OATP1B1 and OATP1B3. Therefore, co-administration of PREZCOBIX with drugs that are primarily metabolized by CYP3A and/or CYP2D6 or are substrates of P-gp, BCRP, MATE1, OATP1B1 or OATP1B3 may result in increased plasma concentrations of such drugs, which could increase or prolong their therapeutic effect and can be associated with adverse events. Co-administration of PREZCOBIX with drugs that have active metabolite(s) formed by CYP3A may result in reduced plasma concentrations of these active metabolite(s), potentially leading to loss of their therapeutic effect (see Table 2).
7.2 Potential for Other Drugs to Affect Prezcobix (7.2 Potential for Other Drugs to Affect PREZCOBIX)
Darunavir is metabolized by CYP3A. Cobicistat is metabolized by CYP3A, and to a minor extent, by CYP2D6. Co-administration of PREZCOBIX and drugs that induce CYP3A activity are expected to increase the clearance of darunavir and cobicistat, resulting in lowered plasma concentrations of darunavir and cobicistat which may lead to loss of therapeutic effect and development of resistance. Co-administration of PREZCOBIX and other drugs that inhibit CYP3A may result in increased plasma concentrations of darunavir and cobicistat (see Table 2).
14.2 Clinical Trial Results in Pediatrics With Hiv 1 (14.2 Clinical Trial Results in Pediatrics with HIV-1)
Trial GS-US-216-0128 was a Phase 2/3 multicenter, open-label trial to evaluate the pharmacokinetics, safety, and efficacy of darunavir co-administered with cobicistat in adolescents aged 12 years and older (Cohort 1) and children aged 6 to less than 12 years old (Cohort 2) with HIV-1 who were virologically suppressed and had a baseline estimated creatinine clearance ≥90 mL/min/1.73 m 2. Participants were on a stable antiretroviral regimen including at least 2 NRTIs (for at least 3 months).
Principal Display Panel 800 Mg/150 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 800 mg/150 mg Tablet Bottle Label)
NDC 59676-575-30
PREZCOBIX
®
(darunavir and cobicistat)
Tablets
800 mg/150 mg
ALERT: Find out about medicines
that should NOT be taken with PREZCOBIX ®
from your healthcare provider.
Rx only
30 Tablets
Johnson
&Johnson
Principal Display Panel 675 Mg/150 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 675 mg/150 mg Tablet Bottle Carton)
NDC 59676-578-30
PREZCOBIX
®
(darunavir and
cobicistat)
Tablets
675 mg/150 mg
ALERT: Find out about medicines that
should NOT be taken with PREZCOBIX
®
from your healthcare provider.
Rx only
30 Tablets
Johnson
& Johnson
7.3 Established and Other Potentially Significant Drug Interactions
Table 2 provides dosing recommendations for expected clinically relevant interactions with PREZCOBIX (this table is not all inclusive). These recommendations are based on either drug interaction trials or predicted interactions due to the expected magnitude of interaction and potential for serious adverse events or loss of therapeutic effect. The table includes examples of potentially significant interactions but is not all inclusive , and therefore the label of each drug that is co-administered with PREZCOBIX should be consulted for information related to the route of metabolism, interaction pathways, potential risks, and specific actions to be taken with regard to co-administration. For the list of examples of contraindicated drugs, [see Contraindications (4)] .
| Concomitant Drug Class:
Drug Name Examples |
Effect on Concentration of Darunavir, Cobicistat, or Concomitant Drug | Clinical Comment |
|---|---|---|
| HIV-1 antiviral agents: Nucleoside Reverse Transcriptase Inhibitors (NRTIs) | ||
| didanosine | ↔ darunavir
↔ cobicistat ↔ didanosine |
Didanosine should be administered one hour before or two hours after PREZCOBIX (administered with food). |
| HIV-1 antiviral agents: Non-Nucleoside Reverse Transcriptase Inhibitors (NNRTIs) | ||
| efavirenz | ↓ cobicistat
↓ darunavir |
Co-administration with efavirenz is not recommended because it may result in loss of therapeutic effect and development of resistance to darunavir. |
| etravirine | ↓ cobicistat
darunavir: effect unknown |
Co-administration with etravirine is not recommended because it may result in loss of therapeutic effect and development of resistance to darunavir. |
| nevirapine | ↓ cobicistat
darunavir: effect unknown |
Co-administration with nevirapine is not recommended because it may result in loss of therapeutic effect and development of resistance to darunavir. |
| HIV-1 antiviral agents: CCR5 co-receptor antagonists | ||
| maraviroc | ↑ maraviroc | Maraviroc is a substrate of CYP3A. When co-administered with PREZCOBIX, patients should receive maraviroc 150 mg twice daily. |
| Other agents | ||
|
Alpha 1-adrenoreceptor antagonist:
alfuzosin |
↑ alfuzosin | Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as hypotension. |
|
Antibacterials:
clarithromycin, erythromycin, telithromycin |
↑ darunavir
↑ cobicistat ↑ antibacterial |
Consider alternative antibiotics with concomitant use of PREZCOBIX. |
|
Anticancer agents:
dasatinib, nilotinib |
↑ anticancer agent | A decrease in the dosage or an adjustment of the dosing interval of dasatinib or nilotinib may be necessary when co-administered with PREZCOBIX. Consult the dasatinib and nilotinib prescribing information for dosing instructions. |
| vinblastine, vincristine | For vincristine and vinblastine, consider temporarily withholding the cobicistat-containing antiretroviral regimen in patients who develop significant hematologic or gastrointestinal side effects when PREZCOBIX is administered concurrently with vincristine or vinblastine. If the antiretroviral regimen must be withheld for a prolonged period, consider initiating a revised regimen that does not include a CYP3A or P-gp inhibitor. | |
|
Anticoagulants:
Direct Oral Anticoagulants (DOACs) apixaban |
↑ apixaban | Due to potentially increased bleeding risk, dosing recommendations for co-administration of apixaban with PREZCOBIX depend on the apixaban dose. Refer to apixaban dosing instructions for co-administration with P-gp and strong CYP3A inhibitors in apixaban prescribing information. |
| rivaroxaban | ↑ rivaroxaban | Co-administration of rivaroxaban with PREZCOBIX is not recommended because it may lead to an increased bleeding risk. |
| dabigatran etexilate
edoxaban |
↑ dabigatran
↑ edoxaban |
Refer to the dabigatran etexilate or edoxaban prescribing information for recommendations regarding co-administration. The specific recommendations are based on indication, renal function, and effect of the co-administered P-gp inhibitors on the concentration of dabigatran or edoxaban. Clinical monitoring is recommended when a DOAC not affected by CYP3A4 but transported by P-gp, including dabigatran etexilate and edoxaban, is co-administered with PREZCOBIX. |
| Other Anticoagulants: | ||
| warfarin | warfarin: effect unknown | Monitor the international normalized ratio (INR) when co-administering with warfarin. |
|
Anticonvulsants:
carbamazepine, phenobarbital, phenytoin |
↓ darunavir
↓ cobicistat |
Co-administration is contraindicated due to potential for reduced plasma concentrations of darunavir, which may result in loss of therapeutic effect and development of resistance. |
|
Anticonvulsants with CYP3A induction effects that are NOT contraindicated:
e.g. eslicarbazepine, oxcarbazepine |
↓ cobicistat
darunavir: effect unknown |
Consider alternative anticonvulsant or antiretroviral therapy to avoid potential changes in exposures. If co-administration is necessary, monitor for lack or loss of virologic response. |
|
Anticonvulsants that are metabolized by CYP3A:
e.g. clonazepam |
↑ clonazepam | Clinical monitoring of anticonvulsants is recommended. |
|
Antidepressants:
Selective Serotonin Reuptake Inhibitors (SSRIs): e.g. paroxetine, sertraline |
SSRIs: effects unknown | When co-administering with SSRIs, TCAs, or trazodone, careful dose titration of the antidepressant to the desired effect, including using the lowest feasible initial or maintenance dose, and monitoring for antidepressant response are recommended. |
|
Tricyclic Antidepressants (TCAs):
e.g. amitriptyline, desipramine, imipramine, nortriptyline |
↑ TCAs | |
|
Other antidepressants:
trazodone |
↑ trazodone | |
|
Antifungals:
itraconazole, isavuconazole, ketoconazole, posaconazole |
↑ darunavir
↑ cobicistat |
Monitor for increased darunavir or cobicistat and/or antifungal adverse reactions. |
| ↑ itraconazole
↑ ketoconazole ↑ isavuconazole ↔ posaconazole |
Specific dosing recommendations are not available for co-administration with these antifungals. Monitor for increased itraconazole or ketoconazole adverse reactions. | |
| voriconazole | voriconazole: effects unknown | Co-administration with voriconazole is not recommended unless benefit/risk assessment justifies the use of voriconazole. |
|
Anti-gout:
colchicine |
↑ colchicine | Co-administration is contraindicated in patients with renal and/or hepatic impairment due to potential for serious and/or life-threatening reactions.
For patients without renal or hepatic impairment:
|
|
Antimalarial:
artemether/lumefantrine |
artemether: effect unknown
lumefantrine: effect unknown |
Monitor for a potential decrease of antimalarial efficacy or potential QT prolongation. |
| Antimycobacterials: | ||
| rifampin | ↓ darunavir
↓ cobicistat |
Co-administration is contraindicated due to potential for loss of therapeutic effect and development of resistance. |
| rifabutin | ↑ rifabutin
cobicistat: effects unknown darunavir: effects unknown |
When used in combination with PREZCOBIX, the recommended dose of rifabutin is 150 mg every other day. Monitor for rifabutin-associated adverse reactions including neutropenia and uveitis. |
| rifapentine | ↓ darunavir | Co-administration with rifapentine is not recommended. |
|
Antipsychotics:
lurasidone |
↑ lurasidone | Co-administration is contraindicated due to potential for serious and/or life-threatening reactions. |
| pimozide | ↑ pimozide | Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as cardiac arrhythmias. |
| e.g. perphenazine, risperidone, thioridazine | ↑ antipsychotic | A decrease in the dose of antipsychotics that are metabolized by CYP3A or CYP2D6 may be needed when co-administered with PREZCOBIX. |
| quetiapine | ↑ quetiapine |
Initiation of PREZCOBIX in patients taking quetiapine: Consider alternative antiretroviral therapy to avoid increases in quetiapine exposure. If co-administration is necessary, reduce the quetiapine dose to 1/6 of the current dose and monitor for quetiapine- associated adverse reactions. Refer to the quetiapine prescribing information for recommendations on adverse reaction monitoring.
Initiation of quetiapine in patients taking PREZCOBIX: Refer to the quetiapine prescribing information for initial dosing and titration of quetiapine. |
|
β-Blockers:
e.g. carvedilol, metoprolol, timolol |
↑ beta-blockers | Clinical monitoring is recommended for co-administration with beta-blockers that are metabolized by CYP2D6. |
|
Calcium channel blockers:
e.g. amlodipine, diltiazem, felodipine, nifedipine, verapamil |
↑ calcium channel blockers | Clinical monitoring is recommended for co-administration with calcium channel blockers metabolized by CYP3A. |
| Cardiac Disorders: | ||
| ranolazine, ivabradine | ↑ ranolazine
↑ ivabradine |
Co-administration is contraindicated due to potential for serious and/or life-threatening reactions. |
| dronedarone | ↑ dronedarone | Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as cardiac arrhythmias. |
| Other antiarrhythmics | ||
| e.g. amiodarone, disopyramide, flecainide, lidocaine (systemic), mexiletine, propafenone, quinidine | ↑ antiarrhythmics | Clinical monitoring is recommended upon co-administration with antiarrhythmics. |
| digoxin | ↑ digoxin | When co-administering with digoxin, titrate the digoxin dose and monitor digoxin concentrations. |
|
Corticosteroids:
dexamethasone (systemic) Corticosteroids primarily metabolized by CYP3A: e.g. betamethasone budesonide ciclesonide fluticasone methylprednisolone mometasone triamcinolone |
↓ darunavir
↓ cobicistat ↑ corticosteroids |
Co-administration with systemic dexamethasone or other systemic corticosteroids that induce CYP3A may result in loss of therapeutic effect and development of resistance to PREZCOBIX. Consider alternative corticosteroids.
Co-administration with corticosteroids (all routes of administration) of which exposures are significantly increased by strong CYP3A inhibitors can increase the risk for Cushing's syndrome and adrenal suppression. Alternative corticosteroids including beclomethasone, prednisone and prednisolone (for which PK and/or PD are less affected by strong CYP3A inhibitors relative to other steroids) should be considered, particularly for long-term use. |
|
Endothelin receptor antagonists:
bosentan |
↓ darunavir
↓ cobicistat ↑ bosentan |
Initiation of bosentan in patients taking PREZCOBIX: In patients who have been receiving PREZCOBIX for at least 10 days, start bosentan at 62.5 mg once daily or every other day based upon individual tolerability.
Initiation of PREZCOBIX in patients on bosentan: Discontinue use of bosentan at least 36 hours prior to initiation of PREZCOBIX. After at least 10 days following the initiation of PREZCOBIX, resume bosentan at 62.5 mg once daily or every other day based upon individual tolerability. Switching from darunavir co-administered with ritonavir to PREZCOBIX in patients on bosentan: Maintain bosentan dose. |
|
Ergot derivatives:
e.g. dihydroergotamine, ergotamine, methylergonovine |
↑ ergot derivatives | Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as acute ergot toxicity characterized by peripheral vasospasm and ischemia of the extremities and other tissues. |
|
Hepatitis C virus (HCV):
Direct-Acting Antivirals: elbasvir/grazoprevir |
↑ elbasvir/grazoprevir | Co-administration is contraindicated due to potential for the increased risk of alanine transaminase (ALT) elevations. |
| glecaprevir/pibrentasvir | ↑ glecaprevir
↑ pibrentasvir |
Co-administration of PREZCOBIX with glecaprevir/pibrentasvir is not recommended. |
|
Herbal product:
St. John's wort ( Hypericum perforatum) |
↓ darunavir
↓ cobicistat |
Co-administration is contraindicated due to potential for reduced plasma concentrations of darunavir, which may result in loss of therapeutic effect and development of resistance. |
| Hormonal contraceptives: | Additional or alternative (non-hormonal) forms of contraception should be considered when estrogen-containing contraceptives are co-administered with PREZCOBIX [see Use in Specific Populations (8.3)] . | |
| drospirenone/ethinylestradiol | ↑ drospirenone
↓ ethinylestradiol |
For co-administration with drospirenone, clinical monitoring is recommended due to the potential for hyperkalemia. |
| Other progestin/estrogen contraceptives | progestin: effects unknown
estrogen: effects unknown |
No data are available to make recommendations on co-administration with other hormonal contraceptives. |
|
Immunosuppressants:
cyclosporine, sirolimus, tacrolimus |
↑ immunosuppressants | These immunosuppressant agents are metabolized by CYP3A. Therapeutic drug monitoring is recommended with concomitant use |
|
Immunosuppressant /neoplastic:
everolimus |
↑ immunosuppressants |
Co-administration of everolimus and PREZCOBIX is not recommended. |
| irinotecan | Discontinue PREZCOBIX at least 1 week prior to starting irinotecan therapy. Do not administer PREZCOBIX with irinotecan unless there are no therapeutic alternatives. | |
|
Inhaled beta agonist:
salmeterol |
↑ salmeterol | Co-administration with salmeterol is not recommended and may result in increased risk of cardiovascular adverse events associated with salmeterol, including QT prolongation, palpitations, and sinus tachycardia. |
| Lipid Modifying Agents | ||
|
HMG-CoA reductase inhibitors:
lovastatin, simvastatin |
↑ lovastatin
↑ simvastatin |
Co-administration is contraindicated due to potential for serious reactions such as myopathy including rhabdomyolysis. |
| atorvastatin, fluvastatin, pitavastatin, pravastatin, rosuvastatin | ↑ atorvastatin
↑ fluvastatin ↑ pravastatin ↑ rosuvastatin pitavastatin: effect unknown |
For atorvastatin, fluvastatin, pitavastatin, pravastatin, and rosuvastatin, start with the lowest recommended dose and titrate while monitoring for safety (e.g. myopathy).
Dosage recommendations with atorvastatin or rosuvastatin are as follows:
|
|
Other lipid modifying agents:
lomitapide |
↑ lomitapide | Co-administration is contraindicated due to potential for markedly increased transaminases associated with increased plasma concentrations of lomitapide. |
|
Narcotic analgesics metabolized by CYP3A:
e.g. fentanyl, oxycodone |
↑ fentanyl
↑ oxycodone |
Careful monitoring of therapeutic effects and adverse reactions associated with CYP3A-metabolized narcotic analgesics (including potentially fatal respiratory depression) is recommended with co-administration. |
| tramadol | ↑ tramadol | A dose decrease may be needed for tramadol with concomitant use. |
|
Narcotic analgesic for treatment of opioid dependence:
buprenorphine, buprenorphine/naloxone, methadone |
buprenorphine or buprenorphine/ naloxone: effects unknown
methadone: effects unknown |
Initiation of buprenorphine, buprenorphine/naloxone or methadone in patients taking PREZCOBIX: Carefully titrate the dose of buprenorphine, buprenorphine/naloxone or methadone to the desired effect; use the lowest feasible initial or maintenance dose.
Initiation of PREZCOBIX in patients taking buprenorphine, buprenorphine/naloxone or methadone: A dose adjustment for buprenorphine, buprenorphine/naloxone or methadone may be needed. Monitor clinical signs and symptoms. |
| Opioid Antagonist | ||
| naloxegol | ↑ naloxegol | Co-administration of PREZCOBIX and naloxegol is contraindicated due to potential for precipitating opioid withdrawal symptoms. |
|
Phosphodiesterase PDE-5 inhibitors:
e.g. avanafil, sildenafil, tadalafil, vardenafil |
↑ PDE-5 inhibitors | Co-administration with avanafil is not recommended because a safe and effective avanafil dosage regimen has not been established.
Co-administration with PDE-5 inhibitors may result in an increase in PDE-5 inhibitor-associated adverse reactions including hypotension, syncope, visual disturbances and priapism. Use of PDE-5 inhibitors for pulmonary arterial hypertension (PAH): Co-administration with sildenafil used for PAH is contraindicated due to potential for sildenafil associated adverse reactions (which include visual disturbances, hypotension, prolonged erection, and syncope). The following dose adjustments are recommended for use of tadalafil with PREZCOBIX:
Sildenafil at a single dose not exceeding 25 mg in 48 hours, vardenafil at a single dose not exceeding 2.5 mg dose in 72 hours, or tadalafil at a single dose not exceeding 10 mg dose in 72 hours can be used with increased monitoring for PDE-5 inhibitor-associated adverse reactions. |
| Platelet aggregation inhibitor: | ||
| ticagrelor | ↑ ticagrelor | Co-administration of PREZCOBIX and ticagrelor is not recommended. |
| clopidogrel | ↓ clopidogrel active metabolite | Co-administration of PREZCOBIX with clopidogrel is not recommended due to the potential reduction of the antiplatelet activity of clopidogrel. |
| prasugrel | ↔ prasugrel active metabolite | No dose adjustment is needed when prasugrel is co-administered with PREZCOBIX. |
|
Sedatives/hypnotics:
orally administered midazolam, triazolam |
↑ midazolam
↑ triazolam |
Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as prolonged or increased sedation or respiratory depression. Triazolam and orally administered midazolam are extensively metabolized by CYP3A. Co-administration of triazolam or orally administered midazolam with PREZCOBIX may cause large increases in the concentrations of these benzodiazepines. |
|
metabolized by CYP3A:
e.g. buspirone, diazepam, estazolam, zolpidem |
↑ sedatives/hypnotics | With concomitant use, titration is recommended with sedatives/hypnotics metabolized by CYP3A and a lower dose of the sedatives/hypnotics should be considered with monitoring for increased and prolonged effects or adverse reactions. |
| parenterally administered midazolam | Co-administration of parenteral midazolam should be done in a setting that ensures close clinical monitoring and appropriate medical management in case of respiratory depression and/or prolonged sedation. Dose reduction for parenteral midazolam should be considered, especially if more than a single dose of midazolam is administered. | |
| Urinary antispasmodics | ||
| fesoterodine | ↑ fesoterodine | When fesoterodine is co-administered with PREZCOBIX, do not exceed a fesoterodine dose of 4 mg once daily. |
| solifenacin | ↑ solifenacin | When solifenacin is co-administered with PREZCOBIX, do not exceed a solifenacin dose of 5 mg once daily. |
7.4 Drugs Without Clinically Significant Interactions With Prezcobix (7.4 Drugs without Clinically Significant Interactions with PREZCOBIX)
Clinically relevant drug-drug interactions have not been observed or are not anticipated with concomitant use of darunavir and cobicistat with rilpivirine, dolutegravir, raltegravir, abacavir, emtricitabine, emtricitabine/tenofovir alafenamide, tenofovir DF, lamivudine, stavudine, zidovudine, or acid modifying medications (antacids, H 2-receptor antagonists, proton pump inhibitors).
2.1 Recommended Dosage in Adults and Pediatric Patients Weighing At Least 25 Kg (2.1 Recommended Dosage in Adults and Pediatric Patients Weighing at Least 25 kg)
The recommended dosages of PREZCOBIX for adults and pediatric patients weighing at least 25 kg are shown in Table 1. The pediatric dose is based on weight. Administer PREZCOBIX orally with food in conjunction with other antiretroviral agents.
| Patient Population | Dose (once daily with food) |
|---|---|
| Adult Patients | One 800 mg darunavir/150 mg cobicistat tablet |
| Pediatric Patients weighing at least 40 kg | |
| Pediatric Patients weighing at least 25 kg to less than 40 kg | One 675 mg darunavir/150 mg cobicistat tablet |
Before prescribing PREZCOBIX 675 mg/150 mg tablets, children should be assessed for the ability to swallow tablets. For patients unable to swallow the 675 mg/150 mg tablet whole, the scored tablet may be split by hand into two pieces. Each piece should be consumed immediately after splitting to ensure the entire dose is administered. The score line is only to facilitate breaking for ease of swallowing and not to divide into equal doses.
5.4 New Onset Or Worsening Renal Impairment When Used With Tenofovir Disoproxil Fumarate (5.4 New Onset or Worsening Renal Impairment When Used With Tenofovir Disoproxil Fumarate)
Renal impairment, including cases of acute renal failure and Fanconi syndrome, has been reported when cobicistat, a component of PREZCOBIX, was used in an antiretroviral regimen that contained tenofovir DF. Co-administration of PREZCOBIX and tenofovir DF is not recommended in patients who have an estimated creatinine clearance below 70 mL/min [see Dosage and Administration (2.3)] .
- Document urine glucose and urine protein at baseline [see Dosage and Administration (2.2)] and perform routine monitoring of estimated creatinine clearance, urine glucose, and urine protein during treatment when PREZCOBIX is used with tenofovir DF. Measure serum phosphorus in patients with or at risk for renal impairment when used with tenofovir DF.
- Co-administration of PREZCOBIX and tenofovir DF in combination with concomitant or recent use of a nephrotoxic agent is not recommended.
See cobicistat full prescribing information for additional information regarding cobicistat.
5.5 Risk of Serious Adverse Reactions Or Loss of Virologic Response Due to Drug Interactions (5.5 Risk of Serious Adverse Reactions or Loss of Virologic Response Due to Drug Interactions)
Initiation of PREZCOBIX, which inhibits CYP3A, in patients receiving medications metabolized by CYP3A, or initiation of medications metabolized by CYP3A in patients already receiving PREZCOBIX may increase plasma concentrations of medications metabolized by CYP3A and reduce plasma concentrations of active metabolite(s) formed by CYP3A. Initiation of medications that inhibit or induce CYP3A may respectively increase or decrease concentrations of PREZCOBIX.
These interactions may lead to:
- clinically significant adverse reactions, potentially leading to severe, life threatening, or fatal events from higher exposures of concomitant medications.
- clinically significant adverse reactions from higher exposures of PREZCOBIX.
- loss of therapeutic effect of the concomitant medications from lower exposures of active metabolite(s).
- loss of therapeutic effect of PREZCOBIX and possible development of resistance from lower exposures of PREZCOBIX.
See Table 2 for steps to prevent or manage these possible and known significant drug interactions, including dosing recommendations. Consider the potential for drug interactions prior to and during PREZCOBIX therapy; review concomitant medications during PREZCOBIX therapy; and monitor for the adverse reactions associated with concomitant medications [see Contraindications (4) and Drug Interactions (7)] .
When used with concomitant medications, PREZCOBIX may result in different drug interactions than those observed or expected with darunavir co-administered with ritonavir. Complex or unknown mechanisms of drug interactions preclude extrapolation of drug interactions with darunavir co-administered with ritonavir to certain PREZCOBIX interactions [see Drug Interactions (7) and Clinical Pharmacology (12.3)] .
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:49:17.253366 · Updated: 2026-03-14T22:32:33.607555