These Highlights Do Not Include All The Information Needed To Use Carbidopa, Levodopa And Entacapone Safely And Effectively. See Full Prescribing Information For Carbidopa, Levodopa And Entacapone.
9c16c0e0-3b97-486b-b7d5-579f5853e2c5
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Carbidopa, levodopa and entacapone tablets are indicated for the treatment of Parkinson's disease. Carbidopa, levodopa and entacapone tablets can be used: • To substitute (with equivalent strengths of each of the three components) carbidopa/levodopa and entacapone previously administered as individual products. • To replace carbidopa/levodopa therapy (without entacapone) when patients experience the signs and symptoms of end-of-dose “wearing-off” and when they have been taking a total daily dose of levodopa of 600 mg or less and have not been experiencing dyskinesias.
Indications and Usage
Carbidopa, levodopa and entacapone tablets are indicated for the treatment of Parkinson's disease. Carbidopa, levodopa and entacapone tablets can be used: • To substitute (with equivalent strengths of each of the three components) carbidopa/levodopa and entacapone previously administered as individual products. • To replace carbidopa/levodopa therapy (without entacapone) when patients experience the signs and symptoms of end-of-dose “wearing-off” and when they have been taking a total daily dose of levodopa of 600 mg or less and have not been experiencing dyskinesias.
Dosage and Administration
Carbidopa, levodopa and entacapone tablets should be used as a substitute for patients already stabilized on equivalent doses of carbidopa/levodopa and entacapone. However, some patients who have been stabilized on a given dose of carbidopa/levodopa may be treated with carbidopa, levodopa and entacapone tablets if a decision has been made to add entacapone (see below). Therapy should be individualized and adjusted according to the desired therapeutic response.
Warnings and Precautions
The following adverse reactions described in this section are related to at least one of the components of carbidopa, levodopa and entacapone tablets (i.e., levodopa, carbidopa, and/or entacapone) based upon the safety experience in clinical trials (especially pivotal trials) or in postmarketing reports.
Contraindications
Carbidopa, levodopa and entacapone tablets are contraindicated in patients: • Taking nonselective monoamine oxidase (MAO) inhibitors (e.g., phenelzine and tranylcypromine). These nonselective MAO inhibitors must be discontinued at least two weeks prior to initiating therapy with carbidopa, levodopa and entacapone tablets. • With narrow-angle glaucoma.
Adverse Reactions
The following adverse reactions are discussed in more detail in the Warnings and Precautions sections of labeling: • Falling Asleep During Activities of Daily Living and Somnolence [see Warnings and Precautions (5.1) ] • Hypotension/Orthostatic Hypotension and Syncope [see Warnings and Precautions (5.2) ] • Dyskinesia [see Warnings and Precautions (5.3) ] • Depression and suicidality [see Warnings and Precautions (5.4) ] • Hallucinations/Psychotic-Like Behavior [see Warnings and Precautions (5.5) ] • Impulse Control and/or Compulsive Behaviors [see Warnings and Precautions (5.6) ] • Withdrawal-Emergent Hyperpyrexia and Confusion [see Warnings and Precautions (5.7) ] • Diarrhea and Colitis [see Warnings and Precautions (5.8) ] • Rhabdomyolysis [see Warnings and Precautions (5.9) ] • Peptic Ulcer Disease [see Warnings and Precautions (5.13) ]
Drug Interactions
• Drugs metabolized by COMT: use with caution ( 5.11 , 7.2 ) • Anti-hypertensive agents: dose adjustment may be required ( 7.3 ) • Tricyclic antidepressants: risk of hypertension and dyskinesia reported during concomitant use with carbidopa/levodopa ( 7.4 ) • Dopamine D2 receptor antagonists, isoniazid, phenytoin, papaverine and iron salts: may reduce efficacy of carbidopa, levodopa and entacapone tablets ( 7.5 , 7.6 , 7.7 , 7.8 , 7.9 ) • Drugs that interfere with biliary excretion, glucuronidation and intestinal beta-glucuronidase: dose adjustment of carbidopa, levodopa and entacapone tablets may be required ( 7.10 ) • Drugs metabolized by CYP2C9 (e.g., coumadin): dose adjustment of carbidopa, levodopa and entacapone tablets may be required; monitor INR when initiating carbidopa, levodopa and entacapone tablets in patients on coumadin ( 7.11 )
Storage and Handling
Carbidopa, levodopa and entacapone tablets are supplied as film-coated tablets for oral administration in the following six strengths: Carbidopa, levodopa and entacapone film-coated tablets containing 12.5 mg of carbidopa, 50 mg of levodopa, and 200 mg of entacapone. The round, bi-convex shaped tablets are brownish- or greyish-red, unscored, and embossed “LCE 50” on one side. NDC 0781-5613-01 HDPE bottle of 100 tablets Carbidopa, levodopa and entacapone film-coated tablets containing 18.75 mg of carbidopa, 75 mg of levodopa, and 200 mg of entacapone. The oval-shaped tablets are light brownish red, unscored and embossed with code “LCE 75” on one side. NDC 0781-5625-01 HDPE bottle of 100 tablets Carbidopa, levodopa and entacapone film-coated tablets containing 25 mg of carbidopa, 100 mg of levodopa, and 200 mg of entacapone. The oval-shaped tablets are brownish- or greyish-red, unscored, and embossed “LCE 100” on one side. NDC 0781-5637-01 HDPE bottle of 100 tablets Carbidopa, levodopa and entacapone film-coated tablets containing 31.25 mg of carbidopa, 125 mg of levodopa, and 200 mg of entacapone. The oval-shaped tablets are light brownish red, unscored and embossed with code “LCE 125” on one side. NDC 0781-5641-01 HDPE bottle of 100 tablets Carbidopa, levodopa and entacapone film-coated tablets containing 37.5 mg of carbidopa, 150 mg of levodopa, and 200 mg of entacapone. The elongated-ellipse shaped tablets are brownish- or greyish-red, unscored, and embossed “LCE 150” on one side. NDC 0781-5654-01 HDPE bottle of 100 tablets Carbidopa, levodopa and entacapone film-coated tablets containing 50 mg of carbidopa, 200 mg of levodopa, and 200 mg of entacapone. The oval shaped tablets are dark brownish red, unscored, and embossed “LCE 200” on one side. NDC 0781-5669-01 HDPE bottle of 100 tablets
How Supplied
Carbidopa, levodopa and entacapone tablets are supplied as film-coated tablets for oral administration in the following six strengths: Carbidopa, levodopa and entacapone film-coated tablets containing 12.5 mg of carbidopa, 50 mg of levodopa, and 200 mg of entacapone. The round, bi-convex shaped tablets are brownish- or greyish-red, unscored, and embossed “LCE 50” on one side. NDC 0781-5613-01 HDPE bottle of 100 tablets Carbidopa, levodopa and entacapone film-coated tablets containing 18.75 mg of carbidopa, 75 mg of levodopa, and 200 mg of entacapone. The oval-shaped tablets are light brownish red, unscored and embossed with code “LCE 75” on one side. NDC 0781-5625-01 HDPE bottle of 100 tablets Carbidopa, levodopa and entacapone film-coated tablets containing 25 mg of carbidopa, 100 mg of levodopa, and 200 mg of entacapone. The oval-shaped tablets are brownish- or greyish-red, unscored, and embossed “LCE 100” on one side. NDC 0781-5637-01 HDPE bottle of 100 tablets Carbidopa, levodopa and entacapone film-coated tablets containing 31.25 mg of carbidopa, 125 mg of levodopa, and 200 mg of entacapone. The oval-shaped tablets are light brownish red, unscored and embossed with code “LCE 125” on one side. NDC 0781-5641-01 HDPE bottle of 100 tablets Carbidopa, levodopa and entacapone film-coated tablets containing 37.5 mg of carbidopa, 150 mg of levodopa, and 200 mg of entacapone. The elongated-ellipse shaped tablets are brownish- or greyish-red, unscored, and embossed “LCE 150” on one side. NDC 0781-5654-01 HDPE bottle of 100 tablets Carbidopa, levodopa and entacapone film-coated tablets containing 50 mg of carbidopa, 200 mg of levodopa, and 200 mg of entacapone. The oval shaped tablets are dark brownish red, unscored, and embossed “LCE 200” on one side. NDC 0781-5669-01 HDPE bottle of 100 tablets
Medication Information
Warnings and Precautions
The following adverse reactions described in this section are related to at least one of the components of carbidopa, levodopa and entacapone tablets (i.e., levodopa, carbidopa, and/or entacapone) based upon the safety experience in clinical trials (especially pivotal trials) or in postmarketing reports.
Indications and Usage
Carbidopa, levodopa and entacapone tablets are indicated for the treatment of Parkinson's disease. Carbidopa, levodopa and entacapone tablets can be used: • To substitute (with equivalent strengths of each of the three components) carbidopa/levodopa and entacapone previously administered as individual products. • To replace carbidopa/levodopa therapy (without entacapone) when patients experience the signs and symptoms of end-of-dose “wearing-off” and when they have been taking a total daily dose of levodopa of 600 mg or less and have not been experiencing dyskinesias.
Dosage and Administration
Carbidopa, levodopa and entacapone tablets should be used as a substitute for patients already stabilized on equivalent doses of carbidopa/levodopa and entacapone. However, some patients who have been stabilized on a given dose of carbidopa/levodopa may be treated with carbidopa, levodopa and entacapone tablets if a decision has been made to add entacapone (see below). Therapy should be individualized and adjusted according to the desired therapeutic response.
Contraindications
Carbidopa, levodopa and entacapone tablets are contraindicated in patients: • Taking nonselective monoamine oxidase (MAO) inhibitors (e.g., phenelzine and tranylcypromine). These nonselective MAO inhibitors must be discontinued at least two weeks prior to initiating therapy with carbidopa, levodopa and entacapone tablets. • With narrow-angle glaucoma.
Adverse Reactions
The following adverse reactions are discussed in more detail in the Warnings and Precautions sections of labeling: • Falling Asleep During Activities of Daily Living and Somnolence [see Warnings and Precautions (5.1) ] • Hypotension/Orthostatic Hypotension and Syncope [see Warnings and Precautions (5.2) ] • Dyskinesia [see Warnings and Precautions (5.3) ] • Depression and suicidality [see Warnings and Precautions (5.4) ] • Hallucinations/Psychotic-Like Behavior [see Warnings and Precautions (5.5) ] • Impulse Control and/or Compulsive Behaviors [see Warnings and Precautions (5.6) ] • Withdrawal-Emergent Hyperpyrexia and Confusion [see Warnings and Precautions (5.7) ] • Diarrhea and Colitis [see Warnings and Precautions (5.8) ] • Rhabdomyolysis [see Warnings and Precautions (5.9) ] • Peptic Ulcer Disease [see Warnings and Precautions (5.13) ]
Drug Interactions
• Drugs metabolized by COMT: use with caution ( 5.11 , 7.2 ) • Anti-hypertensive agents: dose adjustment may be required ( 7.3 ) • Tricyclic antidepressants: risk of hypertension and dyskinesia reported during concomitant use with carbidopa/levodopa ( 7.4 ) • Dopamine D2 receptor antagonists, isoniazid, phenytoin, papaverine and iron salts: may reduce efficacy of carbidopa, levodopa and entacapone tablets ( 7.5 , 7.6 , 7.7 , 7.8 , 7.9 ) • Drugs that interfere with biliary excretion, glucuronidation and intestinal beta-glucuronidase: dose adjustment of carbidopa, levodopa and entacapone tablets may be required ( 7.10 ) • Drugs metabolized by CYP2C9 (e.g., coumadin): dose adjustment of carbidopa, levodopa and entacapone tablets may be required; monitor INR when initiating carbidopa, levodopa and entacapone tablets in patients on coumadin ( 7.11 )
Storage and Handling
Carbidopa, levodopa and entacapone tablets are supplied as film-coated tablets for oral administration in the following six strengths: Carbidopa, levodopa and entacapone film-coated tablets containing 12.5 mg of carbidopa, 50 mg of levodopa, and 200 mg of entacapone. The round, bi-convex shaped tablets are brownish- or greyish-red, unscored, and embossed “LCE 50” on one side. NDC 0781-5613-01 HDPE bottle of 100 tablets Carbidopa, levodopa and entacapone film-coated tablets containing 18.75 mg of carbidopa, 75 mg of levodopa, and 200 mg of entacapone. The oval-shaped tablets are light brownish red, unscored and embossed with code “LCE 75” on one side. NDC 0781-5625-01 HDPE bottle of 100 tablets Carbidopa, levodopa and entacapone film-coated tablets containing 25 mg of carbidopa, 100 mg of levodopa, and 200 mg of entacapone. The oval-shaped tablets are brownish- or greyish-red, unscored, and embossed “LCE 100” on one side. NDC 0781-5637-01 HDPE bottle of 100 tablets Carbidopa, levodopa and entacapone film-coated tablets containing 31.25 mg of carbidopa, 125 mg of levodopa, and 200 mg of entacapone. The oval-shaped tablets are light brownish red, unscored and embossed with code “LCE 125” on one side. NDC 0781-5641-01 HDPE bottle of 100 tablets Carbidopa, levodopa and entacapone film-coated tablets containing 37.5 mg of carbidopa, 150 mg of levodopa, and 200 mg of entacapone. The elongated-ellipse shaped tablets are brownish- or greyish-red, unscored, and embossed “LCE 150” on one side. NDC 0781-5654-01 HDPE bottle of 100 tablets Carbidopa, levodopa and entacapone film-coated tablets containing 50 mg of carbidopa, 200 mg of levodopa, and 200 mg of entacapone. The oval shaped tablets are dark brownish red, unscored, and embossed “LCE 200” on one side. NDC 0781-5669-01 HDPE bottle of 100 tablets
How Supplied
Carbidopa, levodopa and entacapone tablets are supplied as film-coated tablets for oral administration in the following six strengths: Carbidopa, levodopa and entacapone film-coated tablets containing 12.5 mg of carbidopa, 50 mg of levodopa, and 200 mg of entacapone. The round, bi-convex shaped tablets are brownish- or greyish-red, unscored, and embossed “LCE 50” on one side. NDC 0781-5613-01 HDPE bottle of 100 tablets Carbidopa, levodopa and entacapone film-coated tablets containing 18.75 mg of carbidopa, 75 mg of levodopa, and 200 mg of entacapone. The oval-shaped tablets are light brownish red, unscored and embossed with code “LCE 75” on one side. NDC 0781-5625-01 HDPE bottle of 100 tablets Carbidopa, levodopa and entacapone film-coated tablets containing 25 mg of carbidopa, 100 mg of levodopa, and 200 mg of entacapone. The oval-shaped tablets are brownish- or greyish-red, unscored, and embossed “LCE 100” on one side. NDC 0781-5637-01 HDPE bottle of 100 tablets Carbidopa, levodopa and entacapone film-coated tablets containing 31.25 mg of carbidopa, 125 mg of levodopa, and 200 mg of entacapone. The oval-shaped tablets are light brownish red, unscored and embossed with code “LCE 125” on one side. NDC 0781-5641-01 HDPE bottle of 100 tablets Carbidopa, levodopa and entacapone film-coated tablets containing 37.5 mg of carbidopa, 150 mg of levodopa, and 200 mg of entacapone. The elongated-ellipse shaped tablets are brownish- or greyish-red, unscored, and embossed “LCE 150” on one side. NDC 0781-5654-01 HDPE bottle of 100 tablets Carbidopa, levodopa and entacapone film-coated tablets containing 50 mg of carbidopa, 200 mg of levodopa, and 200 mg of entacapone. The oval shaped tablets are dark brownish red, unscored, and embossed “LCE 200” on one side. NDC 0781-5669-01 HDPE bottle of 100 tablets
Description
Carbidopa, levodopa and entacapone tablets are indicated for the treatment of Parkinson's disease. Carbidopa, levodopa and entacapone tablets can be used: • To substitute (with equivalent strengths of each of the three components) carbidopa/levodopa and entacapone previously administered as individual products. • To replace carbidopa/levodopa therapy (without entacapone) when patients experience the signs and symptoms of end-of-dose “wearing-off” and when they have been taking a total daily dose of levodopa of 600 mg or less and have not been experiencing dyskinesias.
Section 34077-8
Risk Summary
There are no adequate data on the developmental risk associated with the use of carbidopa, levodopa and entacapone in pregnant women. In animals, administration of carbidopa-levodopa or entacapone during pregnancy was associated with developmental toxicity, including increased incidences of fetal malformations (see Data). The estimated background risk of major birth defects and miscarriage in the indicated population is unknown. In the U.S. general population, the estimated background risks of major birth defects and miscarriage in clinically recognized pregnancies are 2 to 4% and 15 to 20%, respectively.
Data
Animal data
In nonclinical studies in which carbidopa-levodopa was administered to pregnant animals, increased incidences of visceral and skeletal malformations were observed in rabbits at all doses and ratios of carbidopa-levodopa tested, which ranged from 10 times (carbidopa)-5 times (levodopa) to 20 times (carbidopa)-10 times (levodopa) the maximum recommended human dose (MRHD) of 1,600 mg/day. In rats, there was a decrease in the number of live pups delivered by dams receiving approximately two times (carbidopa)-five times (levodopa) the MRHD throughout organogenesis. No effects on malformation frequencies were observed in mice receiving up to 20 times the MRHD of carbidopa-levodopa.
In embryo-fetal development studies of entacapone, pregnant animals received doses of up to 1,000 mg/kg/day (rats) or 300 mg/kg/day (rabbits) throughout organogenesis. Increased incidences of fetal variations were evident in litters from rats treated with the highest dose, in the absence of overt signs of maternal toxicity. The maternal plasma entacapone exposure (AUC) associated with this dose was approximately 34 times that in humans at the MRHD. Increased frequencies of abortions and late/total resorptions and decreased fetal weights were observed in the litters of rabbits treated with maternally toxic doses of 100 mg/kg/day (plasma AUCs less than that in humans at the MRHD) or greater. There were no increases in malformation rates in these studies.
When entacapone was administered to female rats prior to mating and during early gestation, an increased incidence of fetal eye anomalies (macrophthalmia, microphthalmia, anophthalmia) was observed in the litters of dams treated with doses of 160 mg/kg/day (plasma AUCs seven times that in humans at the MRHD) or greater, in the absence of maternal toxicity. Administration of up to 700 mg/kg/day (plasma AUCs 28 times that in humans at the MRHD) to rats during the latter part of gestation and throughout lactation produced no evidence of developmental impairment in the offspring.
Section 42229-5
Entacapone
The most commonly observed adverse reactions (incidence at least 3% greater than placebo incidence) in the double-blind, carbidopa-levodopa-placebo-controlled trials of entacapone (N=1,003 patients) associated with the use of carbidopa-levodopa-entacapone alone and not seen at an equivalent frequency among the placebo-treated patients were: dyskinesia, diarrhea, nausea, hyperkinesia, abdominal pain, vomiting, dry mouth, and urine discoloration.
The treatment difference incidence for premature study discontinuation for entacapone with levodopa and dopa decarboxylase inhibitor in the double-blind, placebo-controlled trials was 5%. The treatment difference incidence for the most frequent causes of study discontinuation was 2% for diarrhea, and 1% for other specific adverse reactions including psychiatric reasons, dyskinesia/ hyperkinesia, nausea, or abdominal pain.
Section 44425-7
Store at 25°C (77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Dispense contents in a tight, light-resistant container as defined in the USP with a child-resistant closure.
7.6 Isoniazid
Isoniazid may reduce the therapeutic effects of levodopa, a dose increase may be necessary.
7.7 Phenytoin
The beneficial effects of levodopa in Parkinson's disease have been reported to be reversed by phenytoin. Patients taking phenytoin with carbidopa/levodopa should be carefully observed for loss of therapeutic response. Carbidopa, levodopa and entacapone tablets dosage should be increased as clinically needed in patients receiving phenytoin.
8.2 Lactation
Risk Summary
Levodopa has been detected in human milk after administration of carbidopa-levodopa. There are no data on the presence of entacapone or carbidopa in human milk, the effects of levodopa, carbidopa, or entacapone on the breastfed infant, or the effects on milk production. However, inhibition of lactation may occur because levodopa decreases secretion of prolactin [see Clinical Pharmacology (12.6)]. Carbidopa and entacapone are excreted in rat milk. In lactating rat, oral administration of radiolabeled entacapone resulted in levels of entacapone and/or metabolites in milk up to 2 to 3 times that in plasma.
The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for carbidopa, levodopa and entacapone and any potential adverse effects on the breastfed infant from carbidopa, levodopa and entacapone or from the underlying maternal condition.
11 Description
Carbidopa, levodopa and entacapone tablets are a combination of carbidopa, levodopa, and entacapone for the treatment of Parkinson's disease.
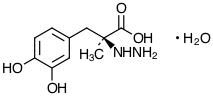
Carbidopa, an inhibitor of aromatic amino acid decarboxylation, is a white, crystalline compound, slightly soluble in water, with a molecular weight of 244.3. It is designated chemically as (-)-L-(α-hydrazino-(α-methyl-β-(3,4-dihydroxybenzene) propanoic acid monohydrate. Its empirical formula is C10H14N2O4•H2O, and its structural formula is:
Tablet content is expressed in terms of anhydrous carbidopa, which has a molecular weight of 226.3.
Levodopa, an aromatic amino acid, is a white, crystalline compound, slightly soluble in water, with a molecular weight of 197.2. It is designated chemically as (-)-L-α-amino-β-(3,4-dihydroxybenzene) propanoic acid. Its empirical formula is C9H11NO4, and its structural formula is:
Entacapone, a COMT inhibitor, is a nitro-catechol-structured compound with a molecular weight of 305.3. The chemical name of entacapone is (E)-2-cyano-3-(3,4-dihydroxy-5-nitrophenyl)-N,N-diethyl-2-propenamide. Its empirical formula is C14H15N3O5 and its structural formula is:
Carbidopa, Levodopa and Entacapone Tablets are supplied as tablets in 6 strengths:
Carbidopa, Levodopa and Entacapone Tablets: 12.5 mg of carbidopa, 50 mg of levodopa and 200 mg of entacapone
Carbidopa, Levodopa and Entacapone Tablets: 18.75 mg of carbidopa, 75 mg of levodopa and 200 mg of entacapone
Carbidopa, Levodopa and Entacapone Tablets: 25 mg of carbidopa, 100 mg of levodopa and 200 mg of entacapone
Carbidopa, Levodopa and Entacapone Tablets: 31.25 mg of carbidopa, 125 mg of levodopa and 200 mg of entacapone
Carbidopa, Levodopa and Entacapone Tablets: 37.5 mg of carbidopa, 150 mg of levodopa and 200 mg of entacapone
Carbidopa, Levodopa and Entacapone Tablets: 50 mg of carbidopa, 200 mg of levodopa and 200 mg of entacapone
Inactive Ingredients: corn starch, croscarmellose sodium, glycerol 85%, hypromellose, magnesium stearate, mannitol, polysorbate 80, povidone, sucrose, red iron oxide, and titanium dioxide. Tablets containing 12.5 mg of carbidopa, 50 mg of levodopa and 200 mg of entacapone, tablets containing 25 mg of carbidopa, 100 mg of levodopa and 200 mg of entacapone, and tablets containing 37.5 mg of carbidopa, 150 mg of levodopa and 200 mg of entacapone also contain yellow iron oxide.
5.3 Dyskinesia
Dyskinesia (involuntary movements) may occur or be exacerbated at lower dosages and sooner with carbidopa, levodopa and entacapone tablets than with preparations containing only carbidopa and levodopa. The occurrence of dyskinesias may require dosage reduction.
In pivotal trials, the treatment difference incidence of dyskinesia was 10% and for carbidopa-levodopa plus 200 mg entacapone. Although decreasing the dose of levodopa may ameliorate this side effect, many patients in controlled trials continued to experience frequent dyskinesias despite a reduction in their dose of levodopa. The treatment difference incidence of study withdrawal for dyskinesia was 1% for carbidopa-levodopa-entacapone.
7.8 Papaverine
The beneficial effects of levodopa in Parkinson's disease have been reported to be reversed by papaverine. Patients taking papaverine with carbidopa/levodopa should be carefully observed for loss of therapeutic response. Carbidopa, levodopa and entacapone tablets dosage should be increased as clinically needed in patients receiving papaverine.
7.9 Iron Salts
Iron salts or multi vitamins containing iron salts should be coadministered with caution. Iron salts can form chelates with levodopa, carbidopa and entacapone and consequently reduce bioavailability of levodopa, carbidopa and entacapone.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Of the total number of subjects in clinical studies of carbidopa, levodopa and entacapone tablets, 43.8% were 65 years old and over, while 7.2% were 75 years old and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients; however, greater sensitivity of some older individuals cannot be excluded.
Carbidopa, levodopa and entacapone tablets have not been studied in Parkinson's disease patients or in healthy volunteers older than 75 years [see Clinical Pharmacology (12.3)].
5.9 Rhabdomyolysis
Cases of severe rhabdomyolysis have been reported with entacapone when used in combination with carbidopa and levodopa. Severe prolonged motor activity including dyskinesia may possibly account for rhabdomyolysis. Most of the cases were manifested by myalgia and increased values of creatine phosphokinase (CPK) and myoglobin. Some of the reactions also included fever and/or alteration of consciousness. It is also possible that rhabdomyolysis may be a result of the syndrome described in Withdrawal-Emergent Hyperpyrexia and Confusion [see Warnings and Precautions (5.7)].
7.1 Mao Inhibitors
Patients receiving nonselective MAO inhibitors and carbidopa, levodopa and entacapone may be at risk of increased adrenergic tone. Therefore, the use of carbidopa, levodopa and entacapone tablets are contraindicated in patients receiving nonselective MAO inhibitors [see Contraindications (4)].
12.6 Hormone Levels
Of the ingredients in carbidopa, levodopa and entacapone tablets, levodopa is known to depress prolactin secretion and increase growth hormone levels.
14 Clinical Studies
The effectiveness of entacapone as an adjunct to levodopa in the treatment of Parkinson's disease was established in three 24-week multicenter, randomized, double-blind, placebo-controlled studies in patients with Parkinson's disease. In 2 of these studies (Studies 1 and 2), the patients' disease was “fluctuating”, i.e., was characterized by documented periods of “On” (periods of relatively good functioning) and “Off” (periods of relatively poor functioning), despite optimum levodopa therapy. There was also a withdrawal period following 6 months of treatment. In the third trial patients were not required to have been experiencing fluctuations. Prior to the controlled part of these studies, patients were stabilized on levodopa for 2 weeks to 4 weeks.
There is limited experience using entacapone in patients who do not experience fluctuations.
In Studies 1 and 2, patients were randomized to receive placebo or entacapone 200 mg administered concomitantly with each dose of carbidopa/levodopa (up to 10 times daily, but patients averaged 4 doses to 6 doses per day). The double‑blind portion of both studies was 6 months long. Patients periodically recorded the time spent in the “On” and “Off” states in home diaries throughout the duration of the trial. In one study, conducted in the Nordic countries, the primary outcome measure was the total mean time spent in the “On” state during an 18 hour diary recorded day (6 a.m. to midnight). In the other study, the primary outcome measure was the proportion of awake time spent over 24 hours in the “On” state.
In addition to the primary outcome measure, the amount of time spent in the “Off” state was evaluated, and patients were also evaluated by subparts of the Unified Parkinson's Disease Rating Scale (UPDRS), a frequently used multi-item rating scale intended to assess mentation (Part I), activities of daily living (Part II), motor function (Part III), complications of therapy (Part IV), and disease staging (Part V and VI); an investigator's and patient's global assessment of clinical condition, a 7-point subjective scale designed to assess global functioning in Parkinson's disease; and the change in daily carbidopa/levodopa dose.
In Study 1, 171 patients were randomized in 16 centers in Finland, Norway, Sweden, and Denmark (Study 1), all of whom received concomitant levodopa plus dopa decarboxylase inhibitor (either carbidopa/levodopa or benserazide/levodopa). In Study 2, 205 patients were randomized in 17 centers in North America (US and Canada); all patients received concomitant carbidopa/levodopa.
The following tables (Table 4 and Table 5) display the results of these two studies:
|
Primary Measure from Home Diary (from an 18-hour Diary Day) |
|||
|
Baseline |
Change from Baseline at Month 6 Mean; the month 6 values represent the average of weeks 8, 16, and 24, by protocol-defined outcome measure.
|
p-value vs. placebo |
|
|
Hours of Awake Time “On” |
|||
|
Placebo |
9.2 |
+0.1 |
– |
|
Entacapone |
9.3 |
+1.5 |
less than 0.001 |
|
Duration of “On” Time After First AM Dose (Hrs) |
|||
|
Placebo |
2.2 |
0.0 |
– |
|
Entacapone |
2.1 |
+0.2 |
less than 0.05 |
|
Secondary Measures from Home Diary (from an 18-hour Diary Day) P values for Secondary Measures are nominal P values without any adjustment for multiplicity.
|
|||
|
Hours of Awake Time “Off” |
|||
|
Placebo |
5.3 |
0.0 |
– |
|
Entacapone |
5.5 |
-1.3 |
less than 0.001 |
|
Proportion of Awake Time “On” Not an endpoint for this study but primary endpoint in the North American Study.
(%)
|
|||
|
Placebo |
63.8 |
+0.6 |
– |
|
Entacapone |
62.7 |
+9.3 |
less than 0.001 |
|
Levodopa Total Daily Dose (mg) |
|||
|
Placebo |
705 |
+14 |
– |
|
Entacapone |
701 |
-87 |
less than 0.001 |
|
Frequency of Levodopa Daily Intakes |
|||
|
Placebo |
6.1 |
+0.1 |
– |
|
Entacapone |
6.2 |
- 0.4 |
less than 0.001 |
|
Other Secondary Measures |
|||
|
Baseline |
Change from Baseline at Month 6 |
p-value vs. placebo |
|
|
Investigator's Global (overall) % Improved At least one category change at endpoint.
|
|||
|
Placebo |
– |
28 |
– |
|
Entacapone |
– |
56 |
less than 0.01 |
|
Patient's Global (overall) % Improved |
|||
|
Placebo |
– |
22 |
– |
|
Entacapone |
– |
39 |
N.S. Not significant.
|
|
UPDRS Total |
|||
|
Placebo |
37.4 |
-1.1 |
– |
|
Entacapone |
38.5 |
-4.8 |
less than 0.01 |
|
UPDRS Motor |
|||
|
Placebo |
24.6 |
-0.7 |
– |
|
Entacapone |
25.5 |
-3.3 |
less than 0.05 |
|
UPDRS ADL |
|||
|
Placebo |
11.0 |
-0.4 |
– |
|
Entacapone |
11.2 |
-1.8 |
less than 0.05 |
|
Primary Measure from Home Diary (for a 24-hour Diary Day) |
|||
|
Baseline |
Change from Baseline at Month 6 Mean; the month 6 values represent the average of weeks 8, 16, and 24, by protocol-defined outcome measure.
|
p-value vs. placebo |
|
|
Percent of Awake Time “On” |
|||
|
Placebo |
60.8 |
+2.0 |
– |
|
Entacapone |
60.0 |
+6.7 |
less than 0.05 |
|
Secondary Measures from Home Diary (for a 24-hour Diary Day) P values for Secondary Measures are nominal P values without any adjustment for multiplicity.
|
|||
|
Hours of Awake Time “Off” |
|||
|
Placebo |
6.6 |
-0.3 |
– |
|
Entacapone |
6.8 |
-1.2 |
less than 0.01 |
|
Hours of Awake Time “On” |
|||
|
Placebo |
10.3 |
+0.4 |
– |
|
Entacapone |
10.2 |
+1.0 |
N.S. Not significant.
|
|
Levodopa Total Daily Dose (mg) |
|||
|
Placebo |
758 |
+19 |
– |
|
Entacapone |
804 |
-93 |
less than 0.001 |
|
Frequency of Levodopa Daily Intakes |
|||
|
Placebo |
6.0 |
+0.2 |
– |
|
Entacapone |
6.2 |
0.0 |
N.S. |
|
Other Secondary Measures |
|||
|
Baseline |
Change from Baseline at Month 6 |
p-value vs. placebo |
|
|
Investigator's Global (overall) % Improved At least one category change at endpoint.
|
|||
|
Placebo |
– |
21 |
– |
|
Entacapone |
– |
34 |
less than 0.05 |
|
Patient's Global (overall) % Improved |
|||
|
Placebo |
– |
20 |
– |
|
Entacapone |
– |
31 |
less than 0.05 |
|
UPDRS Total Score change at endpoint similarly to the Nordic Study.
|
|||
|
Placebo |
35.6 |
+2.8 |
– |
|
Entacapone |
35.1 |
-0.6 |
less than 0.05 |
|
UPDRS Motor |
|||
|
Placebo |
22.6 |
+1.2 |
– |
|
Entacapone |
22.0 |
-0.9 |
less than 0.05 |
|
UPDRS ADL |
|||
|
Placebo |
11.7 |
+1.1 |
– |
|
Entacapone |
11.9 |
0.0 |
less than 0.05 |
Effects on “On” time did not differ by age, sex, weight, disease severity at baseline, levodopa dose and concurrent treatment with dopamine agonists or selegiline.
4 Contraindications
Carbidopa, levodopa and entacapone tablets are contraindicated in patients:
-
•Taking nonselective monoamine oxidase (MAO) inhibitors (e.g., phenelzine and tranylcypromine). These nonselective MAO inhibitors must be discontinued at least two weeks prior to initiating therapy with carbidopa, levodopa and entacapone tablets.
-
•With narrow-angle glaucoma.
6 Adverse Reactions
The following adverse reactions are discussed in more detail in the Warnings and Precautions sections of labeling:
-
•Falling Asleep During Activities of Daily Living and Somnolence [see Warnings and Precautions (5.1)]
-
•Hypotension/Orthostatic Hypotension and Syncope [see Warnings and Precautions (5.2)]
-
•Dyskinesia [see Warnings and Precautions (5.3)]
-
•Depression and suicidality [see Warnings and Precautions (5.4)]
-
•Hallucinations/Psychotic-Like Behavior [see Warnings and Precautions (5.5)]
-
•Impulse Control and/or Compulsive Behaviors [see Warnings and Precautions (5.6)]
-
•Withdrawal-Emergent Hyperpyrexia and Confusion [see Warnings and Precautions (5.7)]
-
•Diarrhea and Colitis [see Warnings and Precautions (5.8)]
-
•Rhabdomyolysis [see Warnings and Precautions (5.9)]
-
•Peptic Ulcer Disease [see Warnings and Precautions (5.13)]
7 Drug Interactions
-
•Drugs metabolized by COMT: use with caution (5.11, 7.2)
-
•Anti-hypertensive agents: dose adjustment may be required (7.3)
-
•Tricyclic antidepressants: risk of hypertension and dyskinesia reported during concomitant use with carbidopa/levodopa (7.4)
-
•Dopamine D2 receptor antagonists, isoniazid, phenytoin, papaverine and iron salts: may reduce efficacy of carbidopa, levodopa and entacapone tablets (7.5, 7.6, 7.7, 7.8, 7.9)
-
•Drugs that interfere with biliary excretion, glucuronidation and intestinal beta-glucuronidase: dose adjustment of carbidopa, levodopa and entacapone tablets may be required (7.10)
-
•Drugs metabolized by CYP2C9 (e.g., coumadin): dose adjustment of carbidopa, levodopa and entacapone tablets may be required; monitor INR when initiating carbidopa, levodopa and entacapone tablets in patients on coumadin (7.11)
8.6 Renal Impairment
Renal impairment does not affect pharmacokinetics of entacapone. There are no studies on the pharmacokinetics of levodopa and carbidopa in patients with renal impairment [see Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
The pharmacokinetics of carbidopa, levodopa and entacapone tablets has been studied in healthy subjects (age 45 years to 75 years). Overall, following administration of corresponding doses of levodopa, carbidopa and entacapone as carbidopa, levodopa and entacapone tablets or as carbidopa and levodopa product plus Comtan (entacapone) tablets, the mean plasma concentrations of levodopa, carbidopa, and entacapone are comparable.
5.14 Laboratory Tests
Abnormalities in laboratory tests may include elevations of liver function tests such as alkaline phosphatase, SGOT (AST), SGPT (ALT), lactic dehydrogenase, and bilirubin. Abnormalities in blood urea nitrogen and positive Coombs test have also been reported. Commonly, levels of blood urea nitrogen, creatinine, and uric acid are lower during administration of carbidopa, levodopa and entacapone tablets than with levodopa.
Carbidopa, levodopa and entacapone tablets may cause a false-positive reaction for urinary ketone bodies when a test tape is used for determination of ketonuria. This reaction will not be altered by boiling the urine specimen. False-negative tests may result with the use of glucose-oxidase methods of testing for glucosuria.
Cases of falsely diagnosed pheochromocytoma in patients on carbidopa/levodopa therapy have been reported very rarely. Caution should be exercised when interpreting the plasma and urine levels of catecholamines and their metabolites in patients on carbidopa/levodopa therapy.
2.1 Dosing Information
The optimum daily dosage of carbidopa, levodopa and entacapone tablets must be determined by careful titration in each patient.
Clinical experience with daily doses above 1,600 mg of entacapone is limited. The maximum recommended daily dose of carbidopa, levodopa and entacapone tablets depends on the strength used. The maximum number of tablets to be used in a 24-hour period is less with the highest strength of carbidopa, levodopa and entacapone tablets than with lower strengths (see Table 1). Studies show that peripheral dopa decarboxylase is saturated by carbidopa at approximately 70 mg per day to 100 mg per day. Patients receiving less than this amount of carbidopa are more likely to experience nausea and vomiting.
| Carbidopa, Levodopa and Entacapone Tablets Dosage Strength | Maximum Number of Tablets in a 24-hour Period |
|---|---|
|
12.5 mg per 50 mg per 200 mg |
8 |
|
18.75 mg per 75 mg per 200 mg |
|
|
25 mg per 100 mg per 200 mg |
|
|
31.25 mg per 125 mg per 200 mg |
|
|
37.5 mg per 150 mg per 200 mg |
|
|
50 mg per 200 mg per 200 mg |
6 |
1 Indications and Usage
Carbidopa, levodopa and entacapone tablets are indicated for the treatment of Parkinson's disease.
Carbidopa, levodopa and entacapone tablets can be used:
-
•To substitute (with equivalent strengths of each of the three components) carbidopa/levodopa and entacapone previously administered as individual products.
-
•To replace carbidopa/levodopa therapy (without entacapone) when patients experience the signs and symptoms of end-of-dose “wearing-off” and when they have been taking a total daily dose of levodopa of 600 mg or less and have not been experiencing dyskinesias.
5.13 Hepatic Impairment
Patients with hepatic impairment should be treated with caution [see Clinical Pharmacology (12.3)]. As with levodopa, periodic evaluation of hepatic function is recommended during extended therapy.
5.8 Diarrhea and Colitis
In clinical trials of entacapone, diarrhea developed in 60 of 603 (10.0%) and 16 of 400 (4.0%) of patients treated with 200 mg of entacapone or placebo in combination with levodopa and dopa decarboxylase inhibitor, respectively. In patients treated with entacapone, diarrhea was generally mild to moderate in severity (8.6%) but was regarded as severe in 1.3%. Diarrhea resulted in withdrawal in 10 of 603 (1.7%) patients, 7 (1.2%) with mild and moderate diarrhea and 3 (0.5%) with severe diarrhea. Diarrhea generally resolved after discontinuation of entacapone. Two patients with diarrhea were hospitalized. Typically, diarrhea presents within 4 to 12 weeks after entacapone is started, but it may appear as early as the first week and as late as many months after the initiation of treatment. Diarrhea may be associated with weight loss, dehydration, and hypokalemia.
Postmarketing experience has shown that diarrhea may be a sign of drug-induced microscopic colitis, primarily lymphocytic colitis. In these cases diarrhea has usually been moderate to severe, watery and non-bloody, at times associated with dehydration, abdominal pain, weight loss, and hypokalemia. In the majority of cases, diarrhea and other colitis-related symptoms resolved or significantly improved when entacapone treatment was stopped. In some patients with biopsy confirmed colitis, diarrhea had resolved or significantly improved after discontinuation of entacapone but recurred after retreatment with entacapone.
If prolonged diarrhea is suspected to be related to carbidopa, levodopa and entacapone tablets, the drug should be discontinued and appropriate medical therapy considered. If the cause of prolonged diarrhea remains unclear or continues after stopping entacapone, then further diagnostic investigations including colonoscopy and biopsies should be considered.
25 Mg/100 Mg/200 Mg Label
NDC 0781-5637-01 Rx Only
Carbidopa, Levodopa, and Entacapone Tablets
Each film-coated tablet contains:
Carbidopa USP 25 mg
Levodopa USP 100 mg
and Entacapone 200 mg
Do not combine tablets to achieve a
higher strength tablet due to the risk of
entacapone overdose.
100 Tablets
SANDOZ
5.12 Peptic Ulcer Disease
As with levodopa, treatment with carbidopa, levodopa and entacapone tablets may increase the possibility of upper gastrointestinal hemorrhage in patients with a history of peptic ulcer.
50 Mg/200 Mg/200 Mg Label
NDC 0781-5669-01 Rx Only
Carbidopa, Levodopa, and Entacapone Tablets
Each film-coated tablet contains:
Carbidopa USP 50 mg
Levodopa USP 200 mg
and Entacapone 200 mg
Do not combine tablets to achieve a
higher strength tablet due to the risk
of entacapone overdose.
100 Tablets
SANDOZ
12.5 Mg/50 Mg/200 Mg Label
NDC 0781-5613-01 Rx Only
Carbidopa, Levodopa, and Entacapone Tablets
Each film-coated tablet contains:
Carbidopa USP 12.5 mg
Levodopa USP 50 mg
and Entacapone 200 mg
Do not combine tablets to achieve a higher
strength tablet due to the risk of entacapone
overdose.
100 Tablets
SANDOZ
5 Warnings and Precautions
The following adverse reactions described in this section are related to at least one of the components of carbidopa, levodopa and entacapone tablets (i.e., levodopa, carbidopa, and/or entacapone) based upon the safety experience in clinical trials (especially pivotal trials) or in postmarketing reports.
18.75 Mg/75 Mg/200 Mg Label
NDC 0781-5625-01 Rx Only
Carbidopa, Levodopa, and Entacapone Tablets
Each film-coated tablet contains:
Carbidopa USP 18.75 mg
Levodopa USP 75 mg
and Entacapone 200 mg
Do not combine tablets to achieve a higher
strength tablet due to the risk of entacapone
overdose.
100 Tablets
SANDOZ
2 Dosage and Administration
Carbidopa, levodopa and entacapone tablets should be used as a substitute for patients already stabilized on equivalent doses of carbidopa/levodopa and entacapone. However, some patients who have been stabilized on a given dose of carbidopa/levodopa may be treated with carbidopa, levodopa and entacapone tablets if a decision has been made to add entacapone (see below). Therapy should be individualized and adjusted according to the desired therapeutic response.
37.5 Mg/150 Mg/200 Mg Label
NDC 0781-5654-01 Rx Only
Carbidopa, Levodopa, and Entacapone Tablets
Each film-coated tablet contains:
Carbidopa USP 37.5 mg
Levodopa USP 150 mg
and Entacapone 200 mg
Do not combine tablets to achieve a
higher strength tablet due to the risk
of entacapone overdose.
100 Tablets
SANDOZ
5.11 Fibrotic Complications
Cases of retroperitoneal fibrosis, pulmonary infiltrates, pleural effusion, and pleural thickening have been reported in some patients treated with ergot derived dopaminergic agents. These complications may resolve when the drug is discontinued, but complete resolution does not always occur. Although these adverse reactions may be related to the ergoline structure of these compounds, a possible causal role of nonergot derived drugs (e.g., entacapone, levodopa), which increase dopaminergic activity, has also been considered. The expected incidence of fibrotic complications is so low that even if entacapone caused these complications at rates similar to those attributable to other dopaminergic therapies, it is unlikely that it would have been detected in a cohort of the size exposed to entacapone during its clinical development. Four cases of pulmonary fibrosis have been reported during clinical development of entacapone; 3 of these patients were also treated with pergolide and 1 with bromocriptine. The duration of treatment with entacapone ranged from 7 months to 17 months.
7.3 Antihypertensive Agents
Symptomatic postural hypotension has occurred when carbidopa/levodopa was added to the treatment of patients receiving antihypertensive drugs. When starting therapy with carbidopa, levodopa and entacapone tablets, dosage adjustment of antihypertensive drug may be required.
3 Dosage Forms and Strengths
Each carbidopa, levodopa and entacapone tablet, provided in 6 single-dose strengths, contains carbidopa and levodopa in a 1:4 ratio and a 200 mg dose of entacapone. Carbidopa, levodopa and entacapone tablets are supplied as film-coated tablets for oral administration in the following 6 strengths:
Carbidopa, levodopa and entacapone film-coated tablets containing 12.5 mg of carbidopa, 50 mg of levodopa and 200 mg of entacapone. The round, bi-convex shaped tablets are brownish- or greyish-red, unscored, and embossed “LCE 50” on one side.
Carbidopa, levodopa and entacapone film-coated tablets containing 18.75 mg of carbidopa, 75 mg of levodopa and 200 mg of entacapone. The oval-shaped tablets are light brownish red, unscored and embossed with code “LCE 75” on one side.
Carbidopa, levodopa and entacapone film-coated tablets containing 25 mg of carbidopa, 100 mg of levodopa and 200 mg of entacapone. The oval-shaped tablets are brownish- or greyish-red, unscored, and embossed “LCE 100” on one side.
Carbidopa, levodopa and entacapone film-coated tablets containing 31.25 mg of carbidopa, 125 mg of levodopa and 200 mg of entacapone. The oval-shaped tablets are light brownish red, unscored and embossed with code “LCE 125” on one side.
Carbidopa, levodopa and entacapone film-coated tablets containing 37.5 mg of carbidopa, 150 mg of levodopa and 200 mg of entacapone. The elongated-ellipse shaped tablets are brownish- or greyish-red, unscored, and embossed “LCE 150” on one side.
Carbidopa, levodopa and entacapone film-coated tablets containing 50 mg of carbidopa, 200 mg of levodopa and 200 mg of entacapone. The oval shaped tablets are dark brownish red, unscored, and embossed “LCE 200” on one side.
31.25 Mg/125 Mg/200 Mg Label
NDC 0781-5641-01 Rx Only
Carbidopa, Levodopa, and Entacapone Tablets
Each film-coated tablet contains:
Carbidopa USP 31.25 mg
Levodopa USP 125 mg
and Entacapone 200 mg
Do not combine tablets to achieve a
higher strength tablet due to the risk
of entacapone overdose.
100 Tablets
SANDOZ
6.2 Postmarketing Experience
The following spontaneous reports of adverse events temporally associated with entacapone tablets or carbidopa, levodopa and entacapone tablets have been identified since market introduction and are not listed in Table 2. Because these reactions are reported voluntarily from a population of unknown size, it is not always possible to reliably estimate their frequency or establish causal relationship to entacapone or carbidopa, levodopa and entacapone tablets exposure.
Hepatitis with mainly cholestatic features has been reported.
10.2 Management of Overdosage
Hospitalization is advised, and general supportive measures should be employed, along with immediate gastric lavage and repeated doses of charcoal over time. This may hasten the elimination of entacapone in particular, by decreasing its absorption and reabsorption from the GI tract. Intravenous fluids should be administered judiciously and an adequate airway maintained.
Respiratory, circulatory and renal function should be monitored and appropriate supportive measures employed. Electrocardiographic monitoring should be instituted and the patient carefully observed for the development of arrhythmias; if required, appropriate antiarrhythmic therapy should be given. The possibility that the patient may have taken other drugs, increasing the risk of drug interactions (especially catechol-structured drugs) should be taken into consideration. To date, no experience has been reported with dialysis; hence, its value in overdosage is not known. Hemodialysis or hemoperfusion is unlikely to reduce entacapone levels due to its high binding to plasma proteins.
Pyridoxine is not effective in reversing the actions of carbidopa, levodopa and entacapone tablets.
7.4 Tricyclic Antidepressants
There have been reports of adverse reactions, including hypertension and dyskinesia, resulting from the concomitant use of tricyclic antidepressants and carbidopa/levodopa.
8 Use in Specific Populations
-
•Pregnancy: based on animal data, may cause fetal harm (8.1)
5.4 Depression and Suicidality
All patients should be observed carefully for the development of depression with concomitant suicidal tendencies. Patients with past or current psychoses should be treated with caution.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the incidence of adverse reactions (number of unique patients experiencing an adverse reaction associated with treatment/total number of patients treated) observed in the clinical trials of a drug cannot be directly compared to the incidence of adverse reactions in the clinical trials of another drug and may not reflect the incidence of adverse reactions observed in clinical practice.
16 How Supplied/storage and Handling
Carbidopa, levodopa and entacapone tablets are supplied as film-coated tablets for oral administration in the following six strengths:
Carbidopa, levodopa and entacapone film-coated tablets containing 12.5 mg of carbidopa, 50 mg of levodopa, and 200 mg of entacapone.
The round, bi-convex shaped tablets are brownish- or greyish-red, unscored, and embossed “LCE 50” on one side.
-
NDC 0781-5613-01 HDPE bottle of 100 tablets
Carbidopa, levodopa and entacapone film-coated tablets containing 18.75 mg of carbidopa, 75 mg of levodopa, and 200 mg of entacapone.
The oval-shaped tablets are light brownish red, unscored and embossed with code “LCE 75” on one side.
-
NDC 0781-5625-01 HDPE bottle of 100 tablets
Carbidopa, levodopa and entacapone film-coated tablets containing 25 mg of carbidopa, 100 mg of levodopa, and 200 mg of entacapone.
The oval-shaped tablets are brownish- or greyish-red, unscored, and embossed “LCE 100” on one side.
-
NDC 0781-5637-01 HDPE bottle of 100 tablets
Carbidopa, levodopa and entacapone film-coated tablets containing 31.25 mg of carbidopa, 125 mg of levodopa, and 200 mg of entacapone.
The oval-shaped tablets are light brownish red, unscored and embossed with code “LCE 125” on one side.
-
NDC 0781-5641-01 HDPE bottle of 100 tablets
Carbidopa, levodopa and entacapone film-coated tablets containing 37.5 mg of carbidopa, 150 mg of levodopa, and 200 mg of entacapone.
The elongated-ellipse shaped tablets are brownish- or greyish-red, unscored, and embossed “LCE 150” on one side.
-
NDC 0781-5654-01 HDPE bottle of 100 tablets
Carbidopa, levodopa and entacapone film-coated tablets containing 50 mg of carbidopa, 200 mg of levodopa, and 200 mg of entacapone.
The oval shaped tablets are dark brownish red, unscored, and embossed “LCE 200” on one side.
-
NDC 0781-5669-01 HDPE bottle of 100 tablets
7.5 Dopamine D2 Receptor Antagonists
Dopamine D2 receptor antagonists (e.g., metoclopramide, phenothiazines, butyrophenones, risperidone) may reduce the therapeutic effects of levodopa.
10.1 Signs and Symptoms of Overdosage
There are very few cases of overdose with levodopa reported in the published literature. Based on the available information, the acute symptoms of levodopa and dopa decarboxylase inhibitor overdose can be expected to arise from dopaminergic overstimulation. Doses of a few grams may result in CNS disturbances, with an increasing likelihood of cardiovascular disturbance (e.g., hypotension, tachycardia) and more severe psychiatric problems at higher doses. An isolated report of rhabdomyolysis and another of transient renal insufficiency suggest that levodopa overdose may give rise to systemic complications, secondary to dopaminergic overstimulation.
COMT inhibition by entacapone treatment is dose-dependent. A massive overdose of entacapone may theoretically produce a 100% inhibition of the COMT enzyme in people, thereby preventing the O-methylation of endogenous and exogenous catechols.
In clinical trials, the highest single dose of entacapone administered to humans was 800 mg, resulting in a plasma concentration of 14.1 mcg per mL. The highest daily dose given to humans was 2,400 mg, administered in one study as 400 mg six times daily with carbidopa/levodopa for 14 days in 15 Parkinson's disease patients, and in another study as 800 mg three times daily for 7 days in 8 healthy volunteers. At this daily dose, the peak plasma concentrations of entacapone averaged 2.0 mcg per mL (at 45 min, compared to 1.0 mcg per mL and 1.2 mcg per mL with 200 mg entacapone at 45 min.). Abdominal pain and loose stools were the most commonly observed adverse events during this study. Daily doses as high as 2,000 mg entacapone have been administered as 200 mg 10 times daily with carbidopa/levodopa or benserazide/levodopa for at least 1 year in 10 patients, for at least 2 years in 8 patients and for at least 3 years in 7 patients. Overall, however, clinical experience with daily doses above 1,600 mg is limited.
2.5 Decrease Or Interruption of Dosing
Avoid interruption of carbidopa, levodopa and entacapone tablets dosing because hyperpyrexia has been reported in patients who suddenly discontinue or reduce their use of levodopa [see Warnings and Precautions (5.7)].
2.6 Important Administration Instructions
Do not split, crush or chew carbidopa, levodopa and entacapone tablets. Administer only one tablet at each dosing interval. All strengths of carbidopa, levodopa and entacapone tablets contain 200 mg of entacapone. Combining multiple tablets or portions of tablets to achieve a higher levodopa dose may lead to an overdose of entacapone.
Administer carbidopa, levodopa and entacapone tablets with or without food. However, a high-fat, high-calorie meal may delay the absorption of levodopa by about 2 hours [see Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment Or Biliary Obstruction
There are no studies on the pharmacokinetics of carbidopa and levodopa in patients with hepatic impairment. Carbidopa, levodopa and entacapone tablets should be administered cautiously to patients with biliary obstruction or hepatic disease since biliary excretion appears to be the major route of excretion of entacapone and hepatic impairment had a significant effect on the pharmacokinetics of entacapone when 200 mg entacapone was administered alone [see Clinical Pharmacology (12.3)].
5.10 Interaction With Drugs Metabolized By Comt
Drugs known to be metabolized by COMT, such as isoproterenol, epinephrine, norepinephrine, dopamine, dobutamine, alpha-methyldopa, apomorphine, isoetherine, and bitolterol should be administered with caution in patients receiving entacapone regardless of the route of administration (including inhalation), as their interaction may result in increased heart rate, arrhythmia, and/or increased blood pressure.
5.6 Impulse Control And/or Compulsive Behaviors
Postmarketing reports suggest that patients treated with anti-Parkinson medications can experience intense urges to gamble, increased sexual urges, intense urges to spend money uncontrollably, and other intense urges. Patients may be unable to control these urges while taking one or more of the medications generally used for the treatment of Parkinson's disease and which increase central dopaminergic tone, including entacapone taken with levodopa and carbidopa. In some cases, although not all, these urges were reported to have stopped when the dose of anti-Parkinson medications was reduced or discontinued. Because patients may not recognize these behaviors as abnormal it is important for prescribers to specifically ask patients or their caregivers about the development of new or increased gambling urges, sexual urges, uncontrolled spending or other urges while being treated with entacapone. Physicians should consider dose reduction or stopping carbidopa, levodopa and entacapone tablets if a patient develops such urges while taking carbidopa, levodopa and entacapone tablets [see Dosage and Administration (2.5), Warnings and Precautions (5.7)].
5.5 Hallucinations And/or Psychotic Like Behavior
Dopaminergic therapy in patients with Parkinson's disease has been associated with hallucinations. Hallucinations led to drug discontinuation and premature withdrawal from clinical trials in 0.8% and 0% of patients treated with carbidopa, levodopa, entacapone and carbidopa, levodopa, respectively. Hallucinations led to hospitalization in 1.0% and 0.3% of patients in the carbidopa, levodopa, entacapone and carbidopa, levodopa, groups, respectively. Agitation occurred in 1% of patients treated with carbidopa, levodopa, entacapone and 0% treated with carbidopa, levodopa.
5.7 Withdrawal Emergent Hyperpyrexia and Confusion
Cases of hyperpyrexia and confusion resembling neuroleptic malignant syndrome (NMS) have been reported in association with dose reduction or withdrawal of therapy with carbidopa, levodopa and entacapone. However, in some cases, hyperpyrexia and confusion were reported after initiation of treatment with entacapone. Hyperpyrexia and confusion are uncommon but they may be life-threatening with a variety of features, including hyperpyrexia/fever/hyperthermia, muscle rigidity, involuntary movements, altered consciousness/mental status changes, delirium, autonomic dysfunction, tachycardia, tachypnea, sweating, hyper- or hypotension, and abnormal laboratory findings (e.g., creatine phosphokinase elevation, leukocytosis, myoglobinuria, and increased serum myoglobin).
If a patient needs to discontinue or reduce their daily dose of carbidopa, levodopa and entacapone tablets, the dose should be decreased slowly, with supervision from a health care provider [see Dosage and Administration (2.5)]. Specific methods for tapering entacapone have not been systematically evaluated.
7.11 Drugs Metabolized Via Cyp2c9 (e.g., Coumadin)
The dosage of carbidopa, levodopa and entacapone tablets should be adjusted as clinically needed in patients using other drugs metabolized via CYP2C9. An interaction study in healthy volunteers, entacapone increased the AUC of R-warfarin on average by 18%, and the INR values on average by 13%. Cases of increased INR in patients concomitantly using warfarin have been reported during the post-approval use of entacapone. Thus, monitoring of INR is recommended when carbidopa, levodopa and entacapone tablets treatment is initiated for patients receiving warfarin.
5.2 Hypotension, Orthostatic Hypotension and Syncope
Reports of syncope were generally more frequent in patients in both treatment groups who had had a prior episode of documented hypotension (although the episodes of syncope, obtained by history, were themselves not documented with vital sign measurement). Hypotension, orthostatic hypotension, and syncope are observed in patients treated with drugs that increase central dopaminergic tone including carbidopa, levodopa and entacapone tablets.
7.2 Drugs Metabolized By Catechol O Methyltransferase (comt)
Drugs known to be metabolized by COMT, such as isoproterenol, epinephrine, norepinephrine, dopamine, dobutamine, alpha-methyldopa, apomorphine, isoetherine, and bitolterol should be administered with caution in patients receiving entacapone regardless of the route of administration (including inhalation), as their interaction may result in increased heart rates, possibly arrhythmias, and excessive changes in blood pressure [see Warnings and Precautions (5.10) ].
2.4 Concomitant Use With Other Anti Parkinson's Disease Drugs
Anticholinergic agents, dopamine agonists, monoamine oxidase (MAO) - B inhibitors, amantadine, and other standard drugs for Parkinson's disease may be used concomitantly while carbidopa, levodopa and entacapone tablets are being administered; however, dosage adjustments of the concomitant medication or carbidopa, levodopa and entacapone tablets may be required.
5.1 Falling Asleep During Activities of Daily Living and Somnolence
Patients with Parkinson's disease treated with carbidopa, levodopa and entacapone tablets or other carbidopa/levodopa products have reported suddenly falling asleep without prior warning of sleepiness while engaged in activities of daily living (including the operation of motor vehicles). Some of these episodes resulted in accidents. Although many of these patients reported somnolence while taking entacapone, some did not perceive warning signs, such as excessive drowsiness, and believed that they were alert immediately prior to the event. Some of these events have been reported to occur up to one year after initiation of treatment.
Somnolence was reported in 2% of patients taking entacapone and 0% in placebo in controlled trials. It is reported that falling asleep while engaged in activities of daily living always occurs in a setting of pre-existing somnolence, although patients may not give such a history. For this reason, prescribers should reassess patients for drowsiness or sleepiness especially since some of the events occur well after the start of treatment. Prescribers should also be aware that patients may not acknowledge drowsiness or sleepiness until directly questioned about drowsiness or sleepiness during specific activities. Patients who have already experienced somnolence and/or an episode of sudden sleep onset should not participate in these activities during treatment with carbidopa, levodopa and entacapone tablets.
Before initiating treatment with carbidopa, levodopa and entacapone tablets, advise patients of the potential to develop drowsiness and specifically ask about factors that may increase this risk such as use of concomitant sedating medications and the presence of sleep disorders. If a patient develops daytime sleepiness or episodes of falling asleep during activities that require active participation (e.g., conversations, eating, etc.), carbidopa, levodopa and entacapone tablets should ordinarily be discontinued [see Dosage and Administration (2.5), and Warnings and Precautions (5.7)]. If the decision is made to continue carbidopa, levodopa and entacapone tablets, patients should be advised not to drive and to avoid other potentially dangerous activities. There is insufficient information to establish whether dose reduction will eliminate episodes of falling asleep while engaged in activities of daily living.
7.10 Drugs Known to Interfere With Biliary Excretion, Glucuronidation, and Intestinal Beta Glucuronidase
As most entacapone excretion is via the bile, caution should be exercised when drugs known to interfere with biliary excretion, glucuronidation, and intestinal beta-glucuronidase are given concurrently with entacapone. These include probenecid, cholestyramine, and some antibiotics (e.g., erythromycin, rifampicin, ampicillin and chloramphenicol).
2.3 Converting Patients From Carbidopa and Levodopa Products to Carbidopa, Levodopa and Entacapone Tablets
There is no experience in transferring patients currently treated with extended release formulations of carbidopa/levodopa, or carbidopa/levodopa products that are not combined in a 1:4 ratio of carbidopa to levodopa.
Patients with a history of moderate or severe dyskinesias or taking more than 600 mg of the levodopa component per day are likely to require a reduction in their daily levodopa dose when entacapone is added. Because dose adjustment of the individual carbidopa or levodopa component is not possible with fixed-dose products, initially titrate patients to a dose that is tolerated and that meets their individual therapeutic need using a separate carbidopa/levodopa tablet (1:4 ratio) plus an entacapone tablet. Once the patient's individual dose of carbidopa/levodopa plus entacapone dose has been established using two separate tablets; switch the patient to a corresponding single tablet of carbidopa, levodopa and entacapone tablets.
When less levodopa is required, reduce the total daily dosage of carbidopa/levodopa either by decreasing the strength of carbidopa, levodopa and entacapone tablets at each administration or by decreasing the frequency of administration by extending the time between doses.
2.2 Converting Patients From Carbidopa, Levodopa, and Entacapone Products to Carbidopa, Levodopa and Entacapone Tablets
Patients currently treated with entacapone 200 mg with each dose of non-extended release carbidopa/levodopa tablet, can switch to the corresponding strength of carbidopa, levodopa and entacapone tablets containing the same amounts of levodopa and carbidopa. For example, patients receiving one tablet of carbidopa/levodopa 25 mg/100 mg and one tablet of entacapone 200 mg at each administration can switch to a single carbidopa, levodopa and entacapone tablet containing 25 mg of carbidopa, 100 mg of levodopa and 200 mg of entacapone.
Structured Label Content
Section 34077-8 (34077-8)
Risk Summary
There are no adequate data on the developmental risk associated with the use of carbidopa, levodopa and entacapone in pregnant women. In animals, administration of carbidopa-levodopa or entacapone during pregnancy was associated with developmental toxicity, including increased incidences of fetal malformations (see Data). The estimated background risk of major birth defects and miscarriage in the indicated population is unknown. In the U.S. general population, the estimated background risks of major birth defects and miscarriage in clinically recognized pregnancies are 2 to 4% and 15 to 20%, respectively.
Data
Animal data
In nonclinical studies in which carbidopa-levodopa was administered to pregnant animals, increased incidences of visceral and skeletal malformations were observed in rabbits at all doses and ratios of carbidopa-levodopa tested, which ranged from 10 times (carbidopa)-5 times (levodopa) to 20 times (carbidopa)-10 times (levodopa) the maximum recommended human dose (MRHD) of 1,600 mg/day. In rats, there was a decrease in the number of live pups delivered by dams receiving approximately two times (carbidopa)-five times (levodopa) the MRHD throughout organogenesis. No effects on malformation frequencies were observed in mice receiving up to 20 times the MRHD of carbidopa-levodopa.
In embryo-fetal development studies of entacapone, pregnant animals received doses of up to 1,000 mg/kg/day (rats) or 300 mg/kg/day (rabbits) throughout organogenesis. Increased incidences of fetal variations were evident in litters from rats treated with the highest dose, in the absence of overt signs of maternal toxicity. The maternal plasma entacapone exposure (AUC) associated with this dose was approximately 34 times that in humans at the MRHD. Increased frequencies of abortions and late/total resorptions and decreased fetal weights were observed in the litters of rabbits treated with maternally toxic doses of 100 mg/kg/day (plasma AUCs less than that in humans at the MRHD) or greater. There were no increases in malformation rates in these studies.
When entacapone was administered to female rats prior to mating and during early gestation, an increased incidence of fetal eye anomalies (macrophthalmia, microphthalmia, anophthalmia) was observed in the litters of dams treated with doses of 160 mg/kg/day (plasma AUCs seven times that in humans at the MRHD) or greater, in the absence of maternal toxicity. Administration of up to 700 mg/kg/day (plasma AUCs 28 times that in humans at the MRHD) to rats during the latter part of gestation and throughout lactation produced no evidence of developmental impairment in the offspring.
Section 42229-5 (42229-5)
Entacapone
The most commonly observed adverse reactions (incidence at least 3% greater than placebo incidence) in the double-blind, carbidopa-levodopa-placebo-controlled trials of entacapone (N=1,003 patients) associated with the use of carbidopa-levodopa-entacapone alone and not seen at an equivalent frequency among the placebo-treated patients were: dyskinesia, diarrhea, nausea, hyperkinesia, abdominal pain, vomiting, dry mouth, and urine discoloration.
The treatment difference incidence for premature study discontinuation for entacapone with levodopa and dopa decarboxylase inhibitor in the double-blind, placebo-controlled trials was 5%. The treatment difference incidence for the most frequent causes of study discontinuation was 2% for diarrhea, and 1% for other specific adverse reactions including psychiatric reasons, dyskinesia/ hyperkinesia, nausea, or abdominal pain.
Section 44425-7 (44425-7)
Store at 25°C (77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Dispense contents in a tight, light-resistant container as defined in the USP with a child-resistant closure.
7.6 Isoniazid
Isoniazid may reduce the therapeutic effects of levodopa, a dose increase may be necessary.
7.7 Phenytoin
The beneficial effects of levodopa in Parkinson's disease have been reported to be reversed by phenytoin. Patients taking phenytoin with carbidopa/levodopa should be carefully observed for loss of therapeutic response. Carbidopa, levodopa and entacapone tablets dosage should be increased as clinically needed in patients receiving phenytoin.
8.2 Lactation
Risk Summary
Levodopa has been detected in human milk after administration of carbidopa-levodopa. There are no data on the presence of entacapone or carbidopa in human milk, the effects of levodopa, carbidopa, or entacapone on the breastfed infant, or the effects on milk production. However, inhibition of lactation may occur because levodopa decreases secretion of prolactin [see Clinical Pharmacology (12.6)]. Carbidopa and entacapone are excreted in rat milk. In lactating rat, oral administration of radiolabeled entacapone resulted in levels of entacapone and/or metabolites in milk up to 2 to 3 times that in plasma.
The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for carbidopa, levodopa and entacapone and any potential adverse effects on the breastfed infant from carbidopa, levodopa and entacapone or from the underlying maternal condition.
11 Description (11 DESCRIPTION)
Carbidopa, levodopa and entacapone tablets are a combination of carbidopa, levodopa, and entacapone for the treatment of Parkinson's disease.
Carbidopa, an inhibitor of aromatic amino acid decarboxylation, is a white, crystalline compound, slightly soluble in water, with a molecular weight of 244.3. It is designated chemically as (-)-L-(α-hydrazino-(α-methyl-β-(3,4-dihydroxybenzene) propanoic acid monohydrate. Its empirical formula is C10H14N2O4•H2O, and its structural formula is:
Tablet content is expressed in terms of anhydrous carbidopa, which has a molecular weight of 226.3.
Levodopa, an aromatic amino acid, is a white, crystalline compound, slightly soluble in water, with a molecular weight of 197.2. It is designated chemically as (-)-L-α-amino-β-(3,4-dihydroxybenzene) propanoic acid. Its empirical formula is C9H11NO4, and its structural formula is:
Entacapone, a COMT inhibitor, is a nitro-catechol-structured compound with a molecular weight of 305.3. The chemical name of entacapone is (E)-2-cyano-3-(3,4-dihydroxy-5-nitrophenyl)-N,N-diethyl-2-propenamide. Its empirical formula is C14H15N3O5 and its structural formula is:
Carbidopa, Levodopa and Entacapone Tablets are supplied as tablets in 6 strengths:
Carbidopa, Levodopa and Entacapone Tablets: 12.5 mg of carbidopa, 50 mg of levodopa and 200 mg of entacapone
Carbidopa, Levodopa and Entacapone Tablets: 18.75 mg of carbidopa, 75 mg of levodopa and 200 mg of entacapone
Carbidopa, Levodopa and Entacapone Tablets: 25 mg of carbidopa, 100 mg of levodopa and 200 mg of entacapone
Carbidopa, Levodopa and Entacapone Tablets: 31.25 mg of carbidopa, 125 mg of levodopa and 200 mg of entacapone
Carbidopa, Levodopa and Entacapone Tablets: 37.5 mg of carbidopa, 150 mg of levodopa and 200 mg of entacapone
Carbidopa, Levodopa and Entacapone Tablets: 50 mg of carbidopa, 200 mg of levodopa and 200 mg of entacapone
Inactive Ingredients: corn starch, croscarmellose sodium, glycerol 85%, hypromellose, magnesium stearate, mannitol, polysorbate 80, povidone, sucrose, red iron oxide, and titanium dioxide. Tablets containing 12.5 mg of carbidopa, 50 mg of levodopa and 200 mg of entacapone, tablets containing 25 mg of carbidopa, 100 mg of levodopa and 200 mg of entacapone, and tablets containing 37.5 mg of carbidopa, 150 mg of levodopa and 200 mg of entacapone also contain yellow iron oxide.
5.3 Dyskinesia
Dyskinesia (involuntary movements) may occur or be exacerbated at lower dosages and sooner with carbidopa, levodopa and entacapone tablets than with preparations containing only carbidopa and levodopa. The occurrence of dyskinesias may require dosage reduction.
In pivotal trials, the treatment difference incidence of dyskinesia was 10% and for carbidopa-levodopa plus 200 mg entacapone. Although decreasing the dose of levodopa may ameliorate this side effect, many patients in controlled trials continued to experience frequent dyskinesias despite a reduction in their dose of levodopa. The treatment difference incidence of study withdrawal for dyskinesia was 1% for carbidopa-levodopa-entacapone.
7.8 Papaverine
The beneficial effects of levodopa in Parkinson's disease have been reported to be reversed by papaverine. Patients taking papaverine with carbidopa/levodopa should be carefully observed for loss of therapeutic response. Carbidopa, levodopa and entacapone tablets dosage should be increased as clinically needed in patients receiving papaverine.
7.9 Iron Salts
Iron salts or multi vitamins containing iron salts should be coadministered with caution. Iron salts can form chelates with levodopa, carbidopa and entacapone and consequently reduce bioavailability of levodopa, carbidopa and entacapone.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Of the total number of subjects in clinical studies of carbidopa, levodopa and entacapone tablets, 43.8% were 65 years old and over, while 7.2% were 75 years old and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients; however, greater sensitivity of some older individuals cannot be excluded.
Carbidopa, levodopa and entacapone tablets have not been studied in Parkinson's disease patients or in healthy volunteers older than 75 years [see Clinical Pharmacology (12.3)].
5.9 Rhabdomyolysis
Cases of severe rhabdomyolysis have been reported with entacapone when used in combination with carbidopa and levodopa. Severe prolonged motor activity including dyskinesia may possibly account for rhabdomyolysis. Most of the cases were manifested by myalgia and increased values of creatine phosphokinase (CPK) and myoglobin. Some of the reactions also included fever and/or alteration of consciousness. It is also possible that rhabdomyolysis may be a result of the syndrome described in Withdrawal-Emergent Hyperpyrexia and Confusion [see Warnings and Precautions (5.7)].
7.1 Mao Inhibitors (7.1 MAO Inhibitors)
Patients receiving nonselective MAO inhibitors and carbidopa, levodopa and entacapone may be at risk of increased adrenergic tone. Therefore, the use of carbidopa, levodopa and entacapone tablets are contraindicated in patients receiving nonselective MAO inhibitors [see Contraindications (4)].
12.6 Hormone Levels
Of the ingredients in carbidopa, levodopa and entacapone tablets, levodopa is known to depress prolactin secretion and increase growth hormone levels.
14 Clinical Studies (14 CLINICAL STUDIES)
The effectiveness of entacapone as an adjunct to levodopa in the treatment of Parkinson's disease was established in three 24-week multicenter, randomized, double-blind, placebo-controlled studies in patients with Parkinson's disease. In 2 of these studies (Studies 1 and 2), the patients' disease was “fluctuating”, i.e., was characterized by documented periods of “On” (periods of relatively good functioning) and “Off” (periods of relatively poor functioning), despite optimum levodopa therapy. There was also a withdrawal period following 6 months of treatment. In the third trial patients were not required to have been experiencing fluctuations. Prior to the controlled part of these studies, patients were stabilized on levodopa for 2 weeks to 4 weeks.
There is limited experience using entacapone in patients who do not experience fluctuations.
In Studies 1 and 2, patients were randomized to receive placebo or entacapone 200 mg administered concomitantly with each dose of carbidopa/levodopa (up to 10 times daily, but patients averaged 4 doses to 6 doses per day). The double‑blind portion of both studies was 6 months long. Patients periodically recorded the time spent in the “On” and “Off” states in home diaries throughout the duration of the trial. In one study, conducted in the Nordic countries, the primary outcome measure was the total mean time spent in the “On” state during an 18 hour diary recorded day (6 a.m. to midnight). In the other study, the primary outcome measure was the proportion of awake time spent over 24 hours in the “On” state.
In addition to the primary outcome measure, the amount of time spent in the “Off” state was evaluated, and patients were also evaluated by subparts of the Unified Parkinson's Disease Rating Scale (UPDRS), a frequently used multi-item rating scale intended to assess mentation (Part I), activities of daily living (Part II), motor function (Part III), complications of therapy (Part IV), and disease staging (Part V and VI); an investigator's and patient's global assessment of clinical condition, a 7-point subjective scale designed to assess global functioning in Parkinson's disease; and the change in daily carbidopa/levodopa dose.
In Study 1, 171 patients were randomized in 16 centers in Finland, Norway, Sweden, and Denmark (Study 1), all of whom received concomitant levodopa plus dopa decarboxylase inhibitor (either carbidopa/levodopa or benserazide/levodopa). In Study 2, 205 patients were randomized in 17 centers in North America (US and Canada); all patients received concomitant carbidopa/levodopa.
The following tables (Table 4 and Table 5) display the results of these two studies:
|
Primary Measure from Home Diary (from an 18-hour Diary Day) |
|||
|
Baseline |
Change from Baseline at Month 6 Mean; the month 6 values represent the average of weeks 8, 16, and 24, by protocol-defined outcome measure.
|
p-value vs. placebo |
|
|
Hours of Awake Time “On” |
|||
|
Placebo |
9.2 |
+0.1 |
– |
|
Entacapone |
9.3 |
+1.5 |
less than 0.001 |
|
Duration of “On” Time After First AM Dose (Hrs) |
|||
|
Placebo |
2.2 |
0.0 |
– |
|
Entacapone |
2.1 |
+0.2 |
less than 0.05 |
|
Secondary Measures from Home Diary (from an 18-hour Diary Day) P values for Secondary Measures are nominal P values without any adjustment for multiplicity.
|
|||
|
Hours of Awake Time “Off” |
|||
|
Placebo |
5.3 |
0.0 |
– |
|
Entacapone |
5.5 |
-1.3 |
less than 0.001 |
|
Proportion of Awake Time “On” Not an endpoint for this study but primary endpoint in the North American Study.
(%)
|
|||
|
Placebo |
63.8 |
+0.6 |
– |
|
Entacapone |
62.7 |
+9.3 |
less than 0.001 |
|
Levodopa Total Daily Dose (mg) |
|||
|
Placebo |
705 |
+14 |
– |
|
Entacapone |
701 |
-87 |
less than 0.001 |
|
Frequency of Levodopa Daily Intakes |
|||
|
Placebo |
6.1 |
+0.1 |
– |
|
Entacapone |
6.2 |
- 0.4 |
less than 0.001 |
|
Other Secondary Measures |
|||
|
Baseline |
Change from Baseline at Month 6 |
p-value vs. placebo |
|
|
Investigator's Global (overall) % Improved At least one category change at endpoint.
|
|||
|
Placebo |
– |
28 |
– |
|
Entacapone |
– |
56 |
less than 0.01 |
|
Patient's Global (overall) % Improved |
|||
|
Placebo |
– |
22 |
– |
|
Entacapone |
– |
39 |
N.S. Not significant.
|
|
UPDRS Total |
|||
|
Placebo |
37.4 |
-1.1 |
– |
|
Entacapone |
38.5 |
-4.8 |
less than 0.01 |
|
UPDRS Motor |
|||
|
Placebo |
24.6 |
-0.7 |
– |
|
Entacapone |
25.5 |
-3.3 |
less than 0.05 |
|
UPDRS ADL |
|||
|
Placebo |
11.0 |
-0.4 |
– |
|
Entacapone |
11.2 |
-1.8 |
less than 0.05 |
|
Primary Measure from Home Diary (for a 24-hour Diary Day) |
|||
|
Baseline |
Change from Baseline at Month 6 Mean; the month 6 values represent the average of weeks 8, 16, and 24, by protocol-defined outcome measure.
|
p-value vs. placebo |
|
|
Percent of Awake Time “On” |
|||
|
Placebo |
60.8 |
+2.0 |
– |
|
Entacapone |
60.0 |
+6.7 |
less than 0.05 |
|
Secondary Measures from Home Diary (for a 24-hour Diary Day) P values for Secondary Measures are nominal P values without any adjustment for multiplicity.
|
|||
|
Hours of Awake Time “Off” |
|||
|
Placebo |
6.6 |
-0.3 |
– |
|
Entacapone |
6.8 |
-1.2 |
less than 0.01 |
|
Hours of Awake Time “On” |
|||
|
Placebo |
10.3 |
+0.4 |
– |
|
Entacapone |
10.2 |
+1.0 |
N.S. Not significant.
|
|
Levodopa Total Daily Dose (mg) |
|||
|
Placebo |
758 |
+19 |
– |
|
Entacapone |
804 |
-93 |
less than 0.001 |
|
Frequency of Levodopa Daily Intakes |
|||
|
Placebo |
6.0 |
+0.2 |
– |
|
Entacapone |
6.2 |
0.0 |
N.S. |
|
Other Secondary Measures |
|||
|
Baseline |
Change from Baseline at Month 6 |
p-value vs. placebo |
|
|
Investigator's Global (overall) % Improved At least one category change at endpoint.
|
|||
|
Placebo |
– |
21 |
– |
|
Entacapone |
– |
34 |
less than 0.05 |
|
Patient's Global (overall) % Improved |
|||
|
Placebo |
– |
20 |
– |
|
Entacapone |
– |
31 |
less than 0.05 |
|
UPDRS Total Score change at endpoint similarly to the Nordic Study.
|
|||
|
Placebo |
35.6 |
+2.8 |
– |
|
Entacapone |
35.1 |
-0.6 |
less than 0.05 |
|
UPDRS Motor |
|||
|
Placebo |
22.6 |
+1.2 |
– |
|
Entacapone |
22.0 |
-0.9 |
less than 0.05 |
|
UPDRS ADL |
|||
|
Placebo |
11.7 |
+1.1 |
– |
|
Entacapone |
11.9 |
0.0 |
less than 0.05 |
Effects on “On” time did not differ by age, sex, weight, disease severity at baseline, levodopa dose and concurrent treatment with dopamine agonists or selegiline.
4 Contraindications (4 CONTRAINDICATIONS)
Carbidopa, levodopa and entacapone tablets are contraindicated in patients:
-
•Taking nonselective monoamine oxidase (MAO) inhibitors (e.g., phenelzine and tranylcypromine). These nonselective MAO inhibitors must be discontinued at least two weeks prior to initiating therapy with carbidopa, levodopa and entacapone tablets.
-
•With narrow-angle glaucoma.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in more detail in the Warnings and Precautions sections of labeling:
-
•Falling Asleep During Activities of Daily Living and Somnolence [see Warnings and Precautions (5.1)]
-
•Hypotension/Orthostatic Hypotension and Syncope [see Warnings and Precautions (5.2)]
-
•Dyskinesia [see Warnings and Precautions (5.3)]
-
•Depression and suicidality [see Warnings and Precautions (5.4)]
-
•Hallucinations/Psychotic-Like Behavior [see Warnings and Precautions (5.5)]
-
•Impulse Control and/or Compulsive Behaviors [see Warnings and Precautions (5.6)]
-
•Withdrawal-Emergent Hyperpyrexia and Confusion [see Warnings and Precautions (5.7)]
-
•Diarrhea and Colitis [see Warnings and Precautions (5.8)]
-
•Rhabdomyolysis [see Warnings and Precautions (5.9)]
-
•Peptic Ulcer Disease [see Warnings and Precautions (5.13)]
7 Drug Interactions (7 DRUG INTERACTIONS)
-
•Drugs metabolized by COMT: use with caution (5.11, 7.2)
-
•Anti-hypertensive agents: dose adjustment may be required (7.3)
-
•Tricyclic antidepressants: risk of hypertension and dyskinesia reported during concomitant use with carbidopa/levodopa (7.4)
-
•Dopamine D2 receptor antagonists, isoniazid, phenytoin, papaverine and iron salts: may reduce efficacy of carbidopa, levodopa and entacapone tablets (7.5, 7.6, 7.7, 7.8, 7.9)
-
•Drugs that interfere with biliary excretion, glucuronidation and intestinal beta-glucuronidase: dose adjustment of carbidopa, levodopa and entacapone tablets may be required (7.10)
-
•Drugs metabolized by CYP2C9 (e.g., coumadin): dose adjustment of carbidopa, levodopa and entacapone tablets may be required; monitor INR when initiating carbidopa, levodopa and entacapone tablets in patients on coumadin (7.11)
8.6 Renal Impairment
Renal impairment does not affect pharmacokinetics of entacapone. There are no studies on the pharmacokinetics of levodopa and carbidopa in patients with renal impairment [see Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
The pharmacokinetics of carbidopa, levodopa and entacapone tablets has been studied in healthy subjects (age 45 years to 75 years). Overall, following administration of corresponding doses of levodopa, carbidopa and entacapone as carbidopa, levodopa and entacapone tablets or as carbidopa and levodopa product plus Comtan (entacapone) tablets, the mean plasma concentrations of levodopa, carbidopa, and entacapone are comparable.
5.14 Laboratory Tests
Abnormalities in laboratory tests may include elevations of liver function tests such as alkaline phosphatase, SGOT (AST), SGPT (ALT), lactic dehydrogenase, and bilirubin. Abnormalities in blood urea nitrogen and positive Coombs test have also been reported. Commonly, levels of blood urea nitrogen, creatinine, and uric acid are lower during administration of carbidopa, levodopa and entacapone tablets than with levodopa.
Carbidopa, levodopa and entacapone tablets may cause a false-positive reaction for urinary ketone bodies when a test tape is used for determination of ketonuria. This reaction will not be altered by boiling the urine specimen. False-negative tests may result with the use of glucose-oxidase methods of testing for glucosuria.
Cases of falsely diagnosed pheochromocytoma in patients on carbidopa/levodopa therapy have been reported very rarely. Caution should be exercised when interpreting the plasma and urine levels of catecholamines and their metabolites in patients on carbidopa/levodopa therapy.
2.1 Dosing Information
The optimum daily dosage of carbidopa, levodopa and entacapone tablets must be determined by careful titration in each patient.
Clinical experience with daily doses above 1,600 mg of entacapone is limited. The maximum recommended daily dose of carbidopa, levodopa and entacapone tablets depends on the strength used. The maximum number of tablets to be used in a 24-hour period is less with the highest strength of carbidopa, levodopa and entacapone tablets than with lower strengths (see Table 1). Studies show that peripheral dopa decarboxylase is saturated by carbidopa at approximately 70 mg per day to 100 mg per day. Patients receiving less than this amount of carbidopa are more likely to experience nausea and vomiting.
| Carbidopa, Levodopa and Entacapone Tablets Dosage Strength | Maximum Number of Tablets in a 24-hour Period |
|---|---|
|
12.5 mg per 50 mg per 200 mg |
8 |
|
18.75 mg per 75 mg per 200 mg |
|
|
25 mg per 100 mg per 200 mg |
|
|
31.25 mg per 125 mg per 200 mg |
|
|
37.5 mg per 150 mg per 200 mg |
|
|
50 mg per 200 mg per 200 mg |
6 |
1 Indications and Usage (1 INDICATIONS AND USAGE)
Carbidopa, levodopa and entacapone tablets are indicated for the treatment of Parkinson's disease.
Carbidopa, levodopa and entacapone tablets can be used:
-
•To substitute (with equivalent strengths of each of the three components) carbidopa/levodopa and entacapone previously administered as individual products.
-
•To replace carbidopa/levodopa therapy (without entacapone) when patients experience the signs and symptoms of end-of-dose “wearing-off” and when they have been taking a total daily dose of levodopa of 600 mg or less and have not been experiencing dyskinesias.
5.13 Hepatic Impairment
Patients with hepatic impairment should be treated with caution [see Clinical Pharmacology (12.3)]. As with levodopa, periodic evaluation of hepatic function is recommended during extended therapy.
5.8 Diarrhea and Colitis
In clinical trials of entacapone, diarrhea developed in 60 of 603 (10.0%) and 16 of 400 (4.0%) of patients treated with 200 mg of entacapone or placebo in combination with levodopa and dopa decarboxylase inhibitor, respectively. In patients treated with entacapone, diarrhea was generally mild to moderate in severity (8.6%) but was regarded as severe in 1.3%. Diarrhea resulted in withdrawal in 10 of 603 (1.7%) patients, 7 (1.2%) with mild and moderate diarrhea and 3 (0.5%) with severe diarrhea. Diarrhea generally resolved after discontinuation of entacapone. Two patients with diarrhea were hospitalized. Typically, diarrhea presents within 4 to 12 weeks after entacapone is started, but it may appear as early as the first week and as late as many months after the initiation of treatment. Diarrhea may be associated with weight loss, dehydration, and hypokalemia.
Postmarketing experience has shown that diarrhea may be a sign of drug-induced microscopic colitis, primarily lymphocytic colitis. In these cases diarrhea has usually been moderate to severe, watery and non-bloody, at times associated with dehydration, abdominal pain, weight loss, and hypokalemia. In the majority of cases, diarrhea and other colitis-related symptoms resolved or significantly improved when entacapone treatment was stopped. In some patients with biopsy confirmed colitis, diarrhea had resolved or significantly improved after discontinuation of entacapone but recurred after retreatment with entacapone.
If prolonged diarrhea is suspected to be related to carbidopa, levodopa and entacapone tablets, the drug should be discontinued and appropriate medical therapy considered. If the cause of prolonged diarrhea remains unclear or continues after stopping entacapone, then further diagnostic investigations including colonoscopy and biopsies should be considered.
25 Mg/100 Mg/200 Mg Label (25 mg/100 mg/200 mg Label)
NDC 0781-5637-01 Rx Only
Carbidopa, Levodopa, and Entacapone Tablets
Each film-coated tablet contains:
Carbidopa USP 25 mg
Levodopa USP 100 mg
and Entacapone 200 mg
Do not combine tablets to achieve a
higher strength tablet due to the risk of
entacapone overdose.
100 Tablets
SANDOZ
5.12 Peptic Ulcer Disease
As with levodopa, treatment with carbidopa, levodopa and entacapone tablets may increase the possibility of upper gastrointestinal hemorrhage in patients with a history of peptic ulcer.
50 Mg/200 Mg/200 Mg Label (50 mg/200 mg/200 mg Label)
NDC 0781-5669-01 Rx Only
Carbidopa, Levodopa, and Entacapone Tablets
Each film-coated tablet contains:
Carbidopa USP 50 mg
Levodopa USP 200 mg
and Entacapone 200 mg
Do not combine tablets to achieve a
higher strength tablet due to the risk
of entacapone overdose.
100 Tablets
SANDOZ
12.5 Mg/50 Mg/200 Mg Label (12.5 mg/50 mg/200 mg Label)
NDC 0781-5613-01 Rx Only
Carbidopa, Levodopa, and Entacapone Tablets
Each film-coated tablet contains:
Carbidopa USP 12.5 mg
Levodopa USP 50 mg
and Entacapone 200 mg
Do not combine tablets to achieve a higher
strength tablet due to the risk of entacapone
overdose.
100 Tablets
SANDOZ
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
The following adverse reactions described in this section are related to at least one of the components of carbidopa, levodopa and entacapone tablets (i.e., levodopa, carbidopa, and/or entacapone) based upon the safety experience in clinical trials (especially pivotal trials) or in postmarketing reports.
18.75 Mg/75 Mg/200 Mg Label (18.75 mg/75 mg/200 mg Label)
NDC 0781-5625-01 Rx Only
Carbidopa, Levodopa, and Entacapone Tablets
Each film-coated tablet contains:
Carbidopa USP 18.75 mg
Levodopa USP 75 mg
and Entacapone 200 mg
Do not combine tablets to achieve a higher
strength tablet due to the risk of entacapone
overdose.
100 Tablets
SANDOZ
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Carbidopa, levodopa and entacapone tablets should be used as a substitute for patients already stabilized on equivalent doses of carbidopa/levodopa and entacapone. However, some patients who have been stabilized on a given dose of carbidopa/levodopa may be treated with carbidopa, levodopa and entacapone tablets if a decision has been made to add entacapone (see below). Therapy should be individualized and adjusted according to the desired therapeutic response.
37.5 Mg/150 Mg/200 Mg Label (37.5 mg/150 mg/200 mg Label)
NDC 0781-5654-01 Rx Only
Carbidopa, Levodopa, and Entacapone Tablets
Each film-coated tablet contains:
Carbidopa USP 37.5 mg
Levodopa USP 150 mg
and Entacapone 200 mg
Do not combine tablets to achieve a
higher strength tablet due to the risk
of entacapone overdose.
100 Tablets
SANDOZ
5.11 Fibrotic Complications
Cases of retroperitoneal fibrosis, pulmonary infiltrates, pleural effusion, and pleural thickening have been reported in some patients treated with ergot derived dopaminergic agents. These complications may resolve when the drug is discontinued, but complete resolution does not always occur. Although these adverse reactions may be related to the ergoline structure of these compounds, a possible causal role of nonergot derived drugs (e.g., entacapone, levodopa), which increase dopaminergic activity, has also been considered. The expected incidence of fibrotic complications is so low that even if entacapone caused these complications at rates similar to those attributable to other dopaminergic therapies, it is unlikely that it would have been detected in a cohort of the size exposed to entacapone during its clinical development. Four cases of pulmonary fibrosis have been reported during clinical development of entacapone; 3 of these patients were also treated with pergolide and 1 with bromocriptine. The duration of treatment with entacapone ranged from 7 months to 17 months.
7.3 Antihypertensive Agents
Symptomatic postural hypotension has occurred when carbidopa/levodopa was added to the treatment of patients receiving antihypertensive drugs. When starting therapy with carbidopa, levodopa and entacapone tablets, dosage adjustment of antihypertensive drug may be required.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Each carbidopa, levodopa and entacapone tablet, provided in 6 single-dose strengths, contains carbidopa and levodopa in a 1:4 ratio and a 200 mg dose of entacapone. Carbidopa, levodopa and entacapone tablets are supplied as film-coated tablets for oral administration in the following 6 strengths:
Carbidopa, levodopa and entacapone film-coated tablets containing 12.5 mg of carbidopa, 50 mg of levodopa and 200 mg of entacapone. The round, bi-convex shaped tablets are brownish- or greyish-red, unscored, and embossed “LCE 50” on one side.
Carbidopa, levodopa and entacapone film-coated tablets containing 18.75 mg of carbidopa, 75 mg of levodopa and 200 mg of entacapone. The oval-shaped tablets are light brownish red, unscored and embossed with code “LCE 75” on one side.
Carbidopa, levodopa and entacapone film-coated tablets containing 25 mg of carbidopa, 100 mg of levodopa and 200 mg of entacapone. The oval-shaped tablets are brownish- or greyish-red, unscored, and embossed “LCE 100” on one side.
Carbidopa, levodopa and entacapone film-coated tablets containing 31.25 mg of carbidopa, 125 mg of levodopa and 200 mg of entacapone. The oval-shaped tablets are light brownish red, unscored and embossed with code “LCE 125” on one side.
Carbidopa, levodopa and entacapone film-coated tablets containing 37.5 mg of carbidopa, 150 mg of levodopa and 200 mg of entacapone. The elongated-ellipse shaped tablets are brownish- or greyish-red, unscored, and embossed “LCE 150” on one side.
Carbidopa, levodopa and entacapone film-coated tablets containing 50 mg of carbidopa, 200 mg of levodopa and 200 mg of entacapone. The oval shaped tablets are dark brownish red, unscored, and embossed “LCE 200” on one side.
31.25 Mg/125 Mg/200 Mg Label (31.25 mg/125 mg/200 mg Label)
NDC 0781-5641-01 Rx Only
Carbidopa, Levodopa, and Entacapone Tablets
Each film-coated tablet contains:
Carbidopa USP 31.25 mg
Levodopa USP 125 mg
and Entacapone 200 mg
Do not combine tablets to achieve a
higher strength tablet due to the risk
of entacapone overdose.
100 Tablets
SANDOZ
6.2 Postmarketing Experience
The following spontaneous reports of adverse events temporally associated with entacapone tablets or carbidopa, levodopa and entacapone tablets have been identified since market introduction and are not listed in Table 2. Because these reactions are reported voluntarily from a population of unknown size, it is not always possible to reliably estimate their frequency or establish causal relationship to entacapone or carbidopa, levodopa and entacapone tablets exposure.
Hepatitis with mainly cholestatic features has been reported.
10.2 Management of Overdosage
Hospitalization is advised, and general supportive measures should be employed, along with immediate gastric lavage and repeated doses of charcoal over time. This may hasten the elimination of entacapone in particular, by decreasing its absorption and reabsorption from the GI tract. Intravenous fluids should be administered judiciously and an adequate airway maintained.
Respiratory, circulatory and renal function should be monitored and appropriate supportive measures employed. Electrocardiographic monitoring should be instituted and the patient carefully observed for the development of arrhythmias; if required, appropriate antiarrhythmic therapy should be given. The possibility that the patient may have taken other drugs, increasing the risk of drug interactions (especially catechol-structured drugs) should be taken into consideration. To date, no experience has been reported with dialysis; hence, its value in overdosage is not known. Hemodialysis or hemoperfusion is unlikely to reduce entacapone levels due to its high binding to plasma proteins.
Pyridoxine is not effective in reversing the actions of carbidopa, levodopa and entacapone tablets.
7.4 Tricyclic Antidepressants
There have been reports of adverse reactions, including hypertension and dyskinesia, resulting from the concomitant use of tricyclic antidepressants and carbidopa/levodopa.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
-
•Pregnancy: based on animal data, may cause fetal harm (8.1)
5.4 Depression and Suicidality
All patients should be observed carefully for the development of depression with concomitant suicidal tendencies. Patients with past or current psychoses should be treated with caution.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the incidence of adverse reactions (number of unique patients experiencing an adverse reaction associated with treatment/total number of patients treated) observed in the clinical trials of a drug cannot be directly compared to the incidence of adverse reactions in the clinical trials of another drug and may not reflect the incidence of adverse reactions observed in clinical practice.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Carbidopa, levodopa and entacapone tablets are supplied as film-coated tablets for oral administration in the following six strengths:
Carbidopa, levodopa and entacapone film-coated tablets containing 12.5 mg of carbidopa, 50 mg of levodopa, and 200 mg of entacapone.
The round, bi-convex shaped tablets are brownish- or greyish-red, unscored, and embossed “LCE 50” on one side.
-
NDC 0781-5613-01 HDPE bottle of 100 tablets
Carbidopa, levodopa and entacapone film-coated tablets containing 18.75 mg of carbidopa, 75 mg of levodopa, and 200 mg of entacapone.
The oval-shaped tablets are light brownish red, unscored and embossed with code “LCE 75” on one side.
-
NDC 0781-5625-01 HDPE bottle of 100 tablets
Carbidopa, levodopa and entacapone film-coated tablets containing 25 mg of carbidopa, 100 mg of levodopa, and 200 mg of entacapone.
The oval-shaped tablets are brownish- or greyish-red, unscored, and embossed “LCE 100” on one side.
-
NDC 0781-5637-01 HDPE bottle of 100 tablets
Carbidopa, levodopa and entacapone film-coated tablets containing 31.25 mg of carbidopa, 125 mg of levodopa, and 200 mg of entacapone.
The oval-shaped tablets are light brownish red, unscored and embossed with code “LCE 125” on one side.
-
NDC 0781-5641-01 HDPE bottle of 100 tablets
Carbidopa, levodopa and entacapone film-coated tablets containing 37.5 mg of carbidopa, 150 mg of levodopa, and 200 mg of entacapone.
The elongated-ellipse shaped tablets are brownish- or greyish-red, unscored, and embossed “LCE 150” on one side.
-
NDC 0781-5654-01 HDPE bottle of 100 tablets
Carbidopa, levodopa and entacapone film-coated tablets containing 50 mg of carbidopa, 200 mg of levodopa, and 200 mg of entacapone.
The oval shaped tablets are dark brownish red, unscored, and embossed “LCE 200” on one side.
-
NDC 0781-5669-01 HDPE bottle of 100 tablets
7.5 Dopamine D2 Receptor Antagonists
Dopamine D2 receptor antagonists (e.g., metoclopramide, phenothiazines, butyrophenones, risperidone) may reduce the therapeutic effects of levodopa.
10.1 Signs and Symptoms of Overdosage
There are very few cases of overdose with levodopa reported in the published literature. Based on the available information, the acute symptoms of levodopa and dopa decarboxylase inhibitor overdose can be expected to arise from dopaminergic overstimulation. Doses of a few grams may result in CNS disturbances, with an increasing likelihood of cardiovascular disturbance (e.g., hypotension, tachycardia) and more severe psychiatric problems at higher doses. An isolated report of rhabdomyolysis and another of transient renal insufficiency suggest that levodopa overdose may give rise to systemic complications, secondary to dopaminergic overstimulation.
COMT inhibition by entacapone treatment is dose-dependent. A massive overdose of entacapone may theoretically produce a 100% inhibition of the COMT enzyme in people, thereby preventing the O-methylation of endogenous and exogenous catechols.
In clinical trials, the highest single dose of entacapone administered to humans was 800 mg, resulting in a plasma concentration of 14.1 mcg per mL. The highest daily dose given to humans was 2,400 mg, administered in one study as 400 mg six times daily with carbidopa/levodopa for 14 days in 15 Parkinson's disease patients, and in another study as 800 mg three times daily for 7 days in 8 healthy volunteers. At this daily dose, the peak plasma concentrations of entacapone averaged 2.0 mcg per mL (at 45 min, compared to 1.0 mcg per mL and 1.2 mcg per mL with 200 mg entacapone at 45 min.). Abdominal pain and loose stools were the most commonly observed adverse events during this study. Daily doses as high as 2,000 mg entacapone have been administered as 200 mg 10 times daily with carbidopa/levodopa or benserazide/levodopa for at least 1 year in 10 patients, for at least 2 years in 8 patients and for at least 3 years in 7 patients. Overall, however, clinical experience with daily doses above 1,600 mg is limited.
2.5 Decrease Or Interruption of Dosing (2.5 Decrease or Interruption of Dosing)
Avoid interruption of carbidopa, levodopa and entacapone tablets dosing because hyperpyrexia has been reported in patients who suddenly discontinue or reduce their use of levodopa [see Warnings and Precautions (5.7)].
2.6 Important Administration Instructions
Do not split, crush or chew carbidopa, levodopa and entacapone tablets. Administer only one tablet at each dosing interval. All strengths of carbidopa, levodopa and entacapone tablets contain 200 mg of entacapone. Combining multiple tablets or portions of tablets to achieve a higher levodopa dose may lead to an overdose of entacapone.
Administer carbidopa, levodopa and entacapone tablets with or without food. However, a high-fat, high-calorie meal may delay the absorption of levodopa by about 2 hours [see Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment Or Biliary Obstruction (8.7 Hepatic Impairment or Biliary Obstruction)
There are no studies on the pharmacokinetics of carbidopa and levodopa in patients with hepatic impairment. Carbidopa, levodopa and entacapone tablets should be administered cautiously to patients with biliary obstruction or hepatic disease since biliary excretion appears to be the major route of excretion of entacapone and hepatic impairment had a significant effect on the pharmacokinetics of entacapone when 200 mg entacapone was administered alone [see Clinical Pharmacology (12.3)].
5.10 Interaction With Drugs Metabolized By Comt (5.10 Interaction with Drugs Metabolized by COMT)
Drugs known to be metabolized by COMT, such as isoproterenol, epinephrine, norepinephrine, dopamine, dobutamine, alpha-methyldopa, apomorphine, isoetherine, and bitolterol should be administered with caution in patients receiving entacapone regardless of the route of administration (including inhalation), as their interaction may result in increased heart rate, arrhythmia, and/or increased blood pressure.
5.6 Impulse Control And/or Compulsive Behaviors (5.6 Impulse Control and/or Compulsive Behaviors)
Postmarketing reports suggest that patients treated with anti-Parkinson medications can experience intense urges to gamble, increased sexual urges, intense urges to spend money uncontrollably, and other intense urges. Patients may be unable to control these urges while taking one or more of the medications generally used for the treatment of Parkinson's disease and which increase central dopaminergic tone, including entacapone taken with levodopa and carbidopa. In some cases, although not all, these urges were reported to have stopped when the dose of anti-Parkinson medications was reduced or discontinued. Because patients may not recognize these behaviors as abnormal it is important for prescribers to specifically ask patients or their caregivers about the development of new or increased gambling urges, sexual urges, uncontrolled spending or other urges while being treated with entacapone. Physicians should consider dose reduction or stopping carbidopa, levodopa and entacapone tablets if a patient develops such urges while taking carbidopa, levodopa and entacapone tablets [see Dosage and Administration (2.5), Warnings and Precautions (5.7)].
5.5 Hallucinations And/or Psychotic Like Behavior (5.5 Hallucinations and/or Psychotic-Like Behavior)
Dopaminergic therapy in patients with Parkinson's disease has been associated with hallucinations. Hallucinations led to drug discontinuation and premature withdrawal from clinical trials in 0.8% and 0% of patients treated with carbidopa, levodopa, entacapone and carbidopa, levodopa, respectively. Hallucinations led to hospitalization in 1.0% and 0.3% of patients in the carbidopa, levodopa, entacapone and carbidopa, levodopa, groups, respectively. Agitation occurred in 1% of patients treated with carbidopa, levodopa, entacapone and 0% treated with carbidopa, levodopa.
5.7 Withdrawal Emergent Hyperpyrexia and Confusion (5.7 Withdrawal-Emergent Hyperpyrexia and Confusion)
Cases of hyperpyrexia and confusion resembling neuroleptic malignant syndrome (NMS) have been reported in association with dose reduction or withdrawal of therapy with carbidopa, levodopa and entacapone. However, in some cases, hyperpyrexia and confusion were reported after initiation of treatment with entacapone. Hyperpyrexia and confusion are uncommon but they may be life-threatening with a variety of features, including hyperpyrexia/fever/hyperthermia, muscle rigidity, involuntary movements, altered consciousness/mental status changes, delirium, autonomic dysfunction, tachycardia, tachypnea, sweating, hyper- or hypotension, and abnormal laboratory findings (e.g., creatine phosphokinase elevation, leukocytosis, myoglobinuria, and increased serum myoglobin).
If a patient needs to discontinue or reduce their daily dose of carbidopa, levodopa and entacapone tablets, the dose should be decreased slowly, with supervision from a health care provider [see Dosage and Administration (2.5)]. Specific methods for tapering entacapone have not been systematically evaluated.
7.11 Drugs Metabolized Via Cyp2c9 (e.g., Coumadin) (7.11 Drugs Metabolized via CYP2C9 (e.g., coumadin))
The dosage of carbidopa, levodopa and entacapone tablets should be adjusted as clinically needed in patients using other drugs metabolized via CYP2C9. An interaction study in healthy volunteers, entacapone increased the AUC of R-warfarin on average by 18%, and the INR values on average by 13%. Cases of increased INR in patients concomitantly using warfarin have been reported during the post-approval use of entacapone. Thus, monitoring of INR is recommended when carbidopa, levodopa and entacapone tablets treatment is initiated for patients receiving warfarin.
5.2 Hypotension, Orthostatic Hypotension and Syncope
Reports of syncope were generally more frequent in patients in both treatment groups who had had a prior episode of documented hypotension (although the episodes of syncope, obtained by history, were themselves not documented with vital sign measurement). Hypotension, orthostatic hypotension, and syncope are observed in patients treated with drugs that increase central dopaminergic tone including carbidopa, levodopa and entacapone tablets.
7.2 Drugs Metabolized By Catechol O Methyltransferase (comt) (7.2 Drugs Metabolized by Catechol-O-Methyltransferase (COMT))
Drugs known to be metabolized by COMT, such as isoproterenol, epinephrine, norepinephrine, dopamine, dobutamine, alpha-methyldopa, apomorphine, isoetherine, and bitolterol should be administered with caution in patients receiving entacapone regardless of the route of administration (including inhalation), as their interaction may result in increased heart rates, possibly arrhythmias, and excessive changes in blood pressure [see Warnings and Precautions (5.10) ].
2.4 Concomitant Use With Other Anti Parkinson's Disease Drugs (2.4 Concomitant Use with Other Anti-Parkinson's Disease Drugs)
Anticholinergic agents, dopamine agonists, monoamine oxidase (MAO) - B inhibitors, amantadine, and other standard drugs for Parkinson's disease may be used concomitantly while carbidopa, levodopa and entacapone tablets are being administered; however, dosage adjustments of the concomitant medication or carbidopa, levodopa and entacapone tablets may be required.
5.1 Falling Asleep During Activities of Daily Living and Somnolence
Patients with Parkinson's disease treated with carbidopa, levodopa and entacapone tablets or other carbidopa/levodopa products have reported suddenly falling asleep without prior warning of sleepiness while engaged in activities of daily living (including the operation of motor vehicles). Some of these episodes resulted in accidents. Although many of these patients reported somnolence while taking entacapone, some did not perceive warning signs, such as excessive drowsiness, and believed that they were alert immediately prior to the event. Some of these events have been reported to occur up to one year after initiation of treatment.
Somnolence was reported in 2% of patients taking entacapone and 0% in placebo in controlled trials. It is reported that falling asleep while engaged in activities of daily living always occurs in a setting of pre-existing somnolence, although patients may not give such a history. For this reason, prescribers should reassess patients for drowsiness or sleepiness especially since some of the events occur well after the start of treatment. Prescribers should also be aware that patients may not acknowledge drowsiness or sleepiness until directly questioned about drowsiness or sleepiness during specific activities. Patients who have already experienced somnolence and/or an episode of sudden sleep onset should not participate in these activities during treatment with carbidopa, levodopa and entacapone tablets.
Before initiating treatment with carbidopa, levodopa and entacapone tablets, advise patients of the potential to develop drowsiness and specifically ask about factors that may increase this risk such as use of concomitant sedating medications and the presence of sleep disorders. If a patient develops daytime sleepiness or episodes of falling asleep during activities that require active participation (e.g., conversations, eating, etc.), carbidopa, levodopa and entacapone tablets should ordinarily be discontinued [see Dosage and Administration (2.5), and Warnings and Precautions (5.7)]. If the decision is made to continue carbidopa, levodopa and entacapone tablets, patients should be advised not to drive and to avoid other potentially dangerous activities. There is insufficient information to establish whether dose reduction will eliminate episodes of falling asleep while engaged in activities of daily living.
7.10 Drugs Known to Interfere With Biliary Excretion, Glucuronidation, and Intestinal Beta Glucuronidase (7.10 Drugs Known to Interfere with Biliary Excretion, Glucuronidation, and Intestinal Beta-glucuronidase)
As most entacapone excretion is via the bile, caution should be exercised when drugs known to interfere with biliary excretion, glucuronidation, and intestinal beta-glucuronidase are given concurrently with entacapone. These include probenecid, cholestyramine, and some antibiotics (e.g., erythromycin, rifampicin, ampicillin and chloramphenicol).
2.3 Converting Patients From Carbidopa and Levodopa Products to Carbidopa, Levodopa and Entacapone Tablets (2.3 Converting Patients from Carbidopa and Levodopa Products to Carbidopa, Levodopa and Entacapone Tablets)
There is no experience in transferring patients currently treated with extended release formulations of carbidopa/levodopa, or carbidopa/levodopa products that are not combined in a 1:4 ratio of carbidopa to levodopa.
Patients with a history of moderate or severe dyskinesias or taking more than 600 mg of the levodopa component per day are likely to require a reduction in their daily levodopa dose when entacapone is added. Because dose adjustment of the individual carbidopa or levodopa component is not possible with fixed-dose products, initially titrate patients to a dose that is tolerated and that meets their individual therapeutic need using a separate carbidopa/levodopa tablet (1:4 ratio) plus an entacapone tablet. Once the patient's individual dose of carbidopa/levodopa plus entacapone dose has been established using two separate tablets; switch the patient to a corresponding single tablet of carbidopa, levodopa and entacapone tablets.
When less levodopa is required, reduce the total daily dosage of carbidopa/levodopa either by decreasing the strength of carbidopa, levodopa and entacapone tablets at each administration or by decreasing the frequency of administration by extending the time between doses.
2.2 Converting Patients From Carbidopa, Levodopa, and Entacapone Products to Carbidopa, Levodopa and Entacapone Tablets (2.2 Converting Patients from Carbidopa, Levodopa, and Entacapone Products to Carbidopa, Levodopa and Entacapone Tablets)
Patients currently treated with entacapone 200 mg with each dose of non-extended release carbidopa/levodopa tablet, can switch to the corresponding strength of carbidopa, levodopa and entacapone tablets containing the same amounts of levodopa and carbidopa. For example, patients receiving one tablet of carbidopa/levodopa 25 mg/100 mg and one tablet of entacapone 200 mg at each administration can switch to a single carbidopa, levodopa and entacapone tablet containing 25 mg of carbidopa, 100 mg of levodopa and 200 mg of entacapone.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:01.092967 · Updated: 2026-03-14T22:26:10.810615