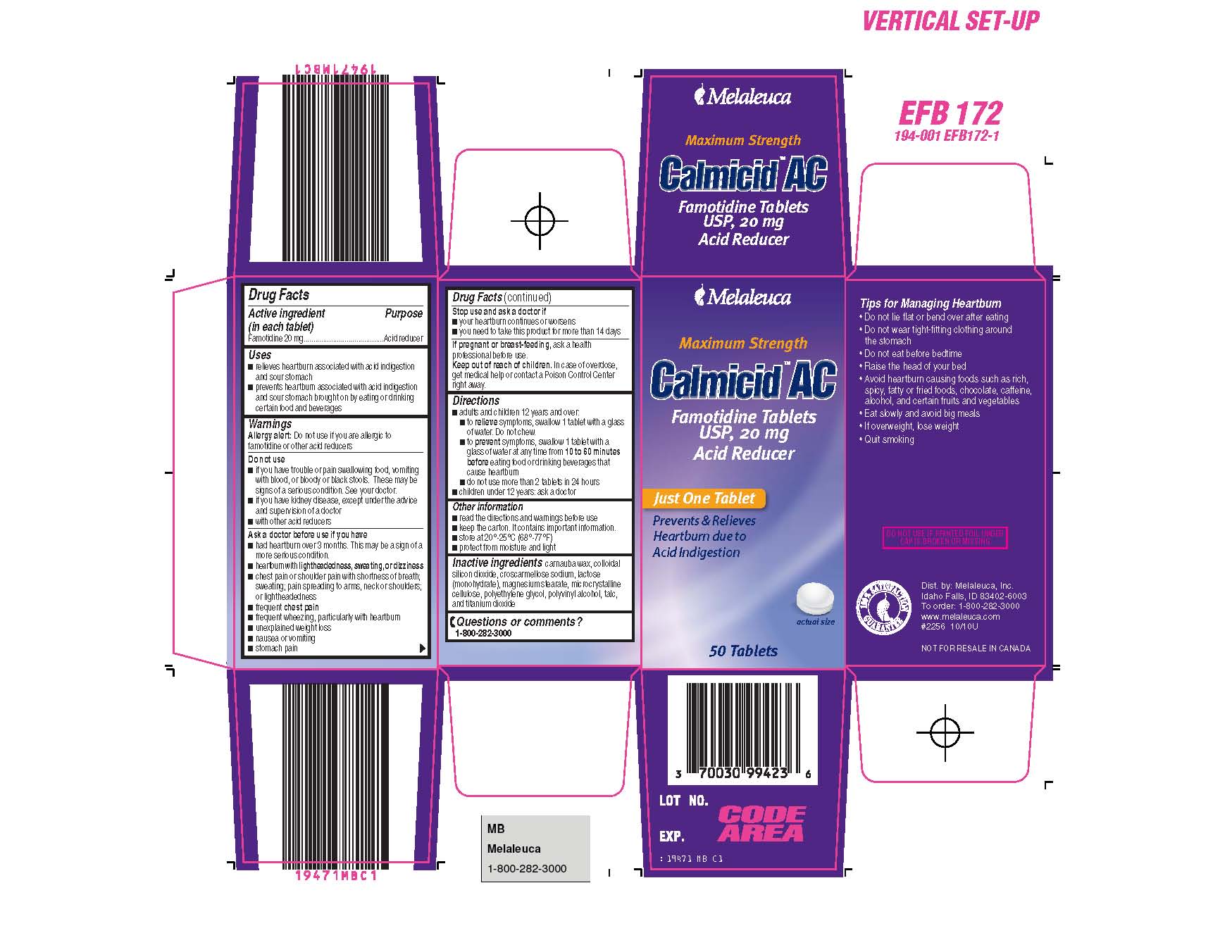
Calmicid Ac Content Of Labeling
9aed4009-307c-44ef-8dce-394a8d4c6597
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Purpose Acid reducer
Medication Information
Recent Major Changes
DO NOT USE IF PRINTED FOIL UNDER CAP IS BROKEN OR MISSING
Warnings and Precautions
Warnings
Allergy alert: Do not use if you are allergic to famotidine or other acid reducers
Indications and Usage
Use
- relieves heartburn associated with acid indigestion and sour stomach
- prevents heartburn associated with acid indigestion and sour stomach brought on by eating or drinking certain food and beverages
Dosage and Administration
Directions
- adults and children 12 years and over:
-
- to relieve symptoms, swallow 1 tablet with a glass of water. Do not chew.
- to prevent symptoms, swallow 1 tablet with a glass of water at any time from 10 to 60 minutes before eating food or drinking beverages that cause heartburn
- do not use more than 2 tablets in 24 hours
- children under 12 years: ask a doctor
Description
Purpose Acid reducer
Section 34072-9
Other information
- read the directions and warnings before use
- keep the carton, it contains important information.
Section 44425-7
Other information
- store at 20°-25°C (68°-77°F)
- protect from moisture and light
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if
- your heartburn continues or worsens
- you need to take this product for more than 14 days
Section 50569-3
Ask a doctor before use if you have
- had heartburn over three months. This may be a sign of a more serious condition.
- heartburn with lightheadedness, sweating, or dizziness
- chest pain or shoulder pain with shortness of breath; sweating; pain spreading to arms, neck or shoulders; or lightheadedness
- frequent chest pain
- frequent wheezing, particularly with heartburn
- unexplained weight loss
- nausea or vomiting
- stomach pain
Section 50570-1
Do not use
- if you have trouble or pain swallowing food, vomiting with blood, or bloody or black stools. These may be signs of a serious condition. See your doctor.
- if you have kidney disease, except under the advice and supervision of a doctor
- with other acid reducers
Section 51727-6
Inactive ingredients carnauba wax, colloidal silicon dioxide, croscarmellose sodium, lactose (monohydrrate), magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide
Section 51945-4
Section 53413-1
Questions or comments?
1-800-282-3000
Section 53414-9
If pregnant or breastfeeding, ask a health professional before use.
Section 55105-1
Purpose
Acid reducer
Section 55106-9
Active ingredient (in each tablet)
Famotidine 20 mg
Structured Label Content
Recent Major Changes (34066-1)
DO NOT USE IF PRINTED FOIL UNDER CAP IS BROKEN OR MISSING
Indications and Usage (34067-9)
Use
- relieves heartburn associated with acid indigestion and sour stomach
- prevents heartburn associated with acid indigestion and sour stomach brought on by eating or drinking certain food and beverages
Dosage and Administration (34068-7)
Directions
- adults and children 12 years and over:
-
- to relieve symptoms, swallow 1 tablet with a glass of water. Do not chew.
- to prevent symptoms, swallow 1 tablet with a glass of water at any time from 10 to 60 minutes before eating food or drinking beverages that cause heartburn
- do not use more than 2 tablets in 24 hours
- children under 12 years: ask a doctor
Warnings and Precautions (34071-1)
Warnings
Allergy alert: Do not use if you are allergic to famotidine or other acid reducers
Section 34072-9 (34072-9)
Other information
- read the directions and warnings before use
- keep the carton, it contains important information.
Section 44425-7 (44425-7)
Other information
- store at 20°-25°C (68°-77°F)
- protect from moisture and light
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if
- your heartburn continues or worsens
- you need to take this product for more than 14 days
Section 50569-3 (50569-3)
Ask a doctor before use if you have
- had heartburn over three months. This may be a sign of a more serious condition.
- heartburn with lightheadedness, sweating, or dizziness
- chest pain or shoulder pain with shortness of breath; sweating; pain spreading to arms, neck or shoulders; or lightheadedness
- frequent chest pain
- frequent wheezing, particularly with heartburn
- unexplained weight loss
- nausea or vomiting
- stomach pain
Section 50570-1 (50570-1)
Do not use
- if you have trouble or pain swallowing food, vomiting with blood, or bloody or black stools. These may be signs of a serious condition. See your doctor.
- if you have kidney disease, except under the advice and supervision of a doctor
- with other acid reducers
Section 51727-6 (51727-6)
Inactive ingredients carnauba wax, colloidal silicon dioxide, croscarmellose sodium, lactose (monohydrrate), magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide
Section 51945-4 (51945-4)
Section 53413-1 (53413-1)
Questions or comments?
1-800-282-3000
Section 53414-9 (53414-9)
If pregnant or breastfeeding, ask a health professional before use.
Section 55105-1 (55105-1)
Purpose
Acid reducer
Section 55106-9 (55106-9)
Active ingredient (in each tablet)
Famotidine 20 mg
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:00:27.904860 · Updated: 2026-03-14T23:00:54.830446