Instant Hand Antiseptic
9aca3f89-cdde-430c-8eb6-43a3ac3fc0df
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Antiseptic
Medication Information
Purpose
Antiseptic
Description
Drug Facts
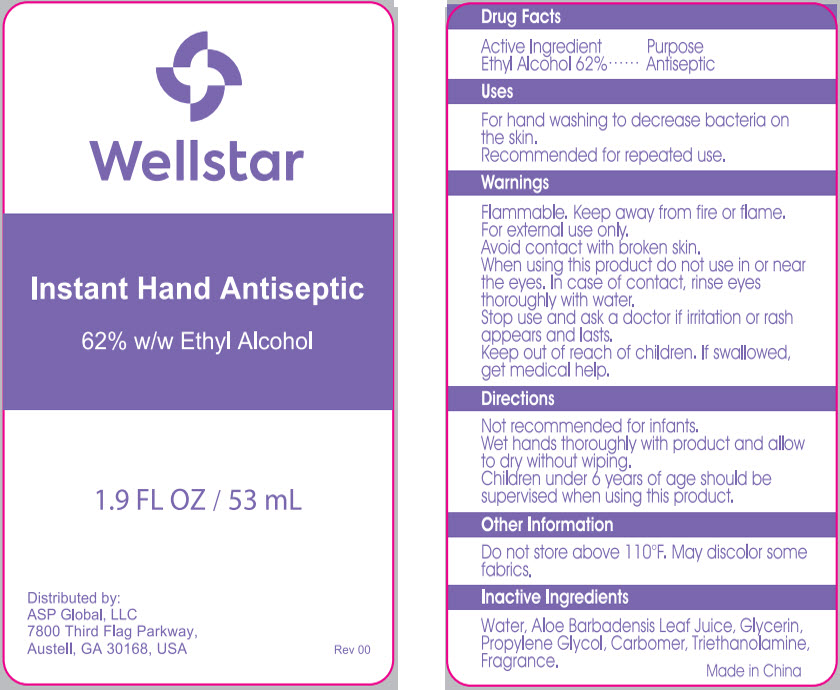
Uses
For hand washing to decrease bacteria on the skin.
Recommended for repeated use.
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. If swallowed, get medical help.
Section 50566-9
Stop use and ask a doctor if irritation or rash appears and lasts.
Section 50567-7
When using this product do not use in or near the eyes. In case of contact, rinse eyes thoroughly with water.
Warnings
Flammable. Keep away from fire or flame.
For external use only.
Avoid contact with broken skin.
Directions
Not recommended for infants.
Wet hands thoroughly with product and allow to dry without wiping.
Children under 6 years of age should be supervised when using this product.
Active Ingredient
Ethyl Alcohol 62%
Other Information
Do not store above 110°F. May discolor some fabrics.
Inactive Ingredients
Water, Aloe Barbadensis Leaf Juice, Glycerin, Propylene Glycol, Carbomer, Triethanolamine, Fragrance.
Principal Display Panel 53 Ml Bottle Label
Wellstar
Instant Hand Antiseptic
62% w/w Ethyl Alcohol
1.9 FL OZ / 53 mL
Distributed by:
ASP Global, LLC
7800 Third Flag Parkway,
Austell, GA 30168, USA
Rev 00
Structured Label Content
Uses
For hand washing to decrease bacteria on the skin.
Recommended for repeated use.
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help.
Section 50566-9 (50566-9)
Stop use and ask a doctor if irritation or rash appears and lasts.
Section 50567-7 (50567-7)
When using this product do not use in or near the eyes. In case of contact, rinse eyes thoroughly with water.
Purpose
Antiseptic
Warnings
Flammable. Keep away from fire or flame.
For external use only.
Avoid contact with broken skin.
Directions
Not recommended for infants.
Wet hands thoroughly with product and allow to dry without wiping.
Children under 6 years of age should be supervised when using this product.
Active Ingredient
Ethyl Alcohol 62%
Other Information
Do not store above 110°F. May discolor some fabrics.
Inactive Ingredients
Water, Aloe Barbadensis Leaf Juice, Glycerin, Propylene Glycol, Carbomer, Triethanolamine, Fragrance.
Principal Display Panel 53 Ml Bottle Label (PRINCIPAL DISPLAY PANEL - 53 mL Bottle Label)
Wellstar
Instant Hand Antiseptic
62% w/w Ethyl Alcohol
1.9 FL OZ / 53 mL
Distributed by:
ASP Global, LLC
7800 Third Flag Parkway,
Austell, GA 30168, USA
Rev 00
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:39.339152 · Updated: 2026-03-14T23:05:32.543899