These Highlights Do Not Include All The Information Needed To Use Rocuronium Bromide Injection Safely And Effectively. See Full Prescribing Information For Rocuronium Bromide Injection.
9a622308-7bda-4ca0-9de5-d6b3b4be3384
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Rocuronium Bromide Injection is indicated as an adjunct to general anesthesia to facilitate both rapid sequence and routine tracheal intubation, and to provide skeletal muscle relaxation during surgery or mechanical ventilation.
Indications and Usage
Rocuronium Bromide Injection is indicated as an adjunct to general anesthesia to facilitate both rapid sequence and routine tracheal intubation, and to provide skeletal muscle relaxation during surgery or mechanical ventilation.
Dosage and Administration
Rocuronium Bromide Injection should only be administered by experienced clinicians or trained individuals supervised by an experienced clinician familiar with the use, actions, characteristics, and complications of neuromuscular blocking agents. ( 2.1 ) Individualize the dose for each patient. ( 2.1 ) Peripheral nerve stimulator recommended for determination of drug response and need for additional doses, and to evaluate recovery. ( 2.1 ) Store Rocuronium Bromide Injection with cap and ferrule intact and in a manner that minimizes the possibility of selecting the wrong product. ( 2.1 ) Tracheal intubation : Recommended initial dose is 0.6 mg/kg. ( 2.2 ) Rapid sequence intubation : 0.6 to 1.2 mg/kg. ( 2.3 ) Maintenance doses : Guided by response to prior dose, not administered until recovery is evident. ( 2.4 ) Continuous infusion : Initial rate of 10 to 12 mcg/kg/min. Start only after early evidence of spontaneous recovery from an intubating dose. ( 2.5 )
Warnings and Precautions
Appropriate Administration and Monitorin g: Use only if facilities for intubation, mechanical ventilation, oxygen therapy, and an antagonist are immediately available. ( 5.1 ) Anaphylaxis : Severe anaphylaxis has been reported. Consider cross-reactivity among neuromuscular blocking agents. ( 5.2 ) Risk of Death due to Medication Errors : Accidental administration can cause death. ( 5.3 ) Need for Adequate Anesthesia : Must be accompanied by adequate anesthesia or sedation. ( 5.4 ) Residual Paralysis : Consider using a reversal agent in cases where residual paralysis is more likely to occur. ( 5.5 )
Contraindications
Rocuronium Bromide Injection is contraindicated in patients known to have hypersensitivity (e.g., anaphylaxis) to rocuronium bromide or other neuromuscular blocking agents [see Warnings and Precautions ( 5.2 )] .
Adverse Reactions
In clinical trials, the most common adverse reactions (2%) are transient hypotension and hypertension. The following adverse reactions are described, or described in greater detail, in other sections: Anaphylaxis [see Warnings and Precautions ( 5.2 )] Residual paralysis [see Warnings and Precautions ( 5.5 )] Myopathy [see Warnings and Precautions ( 5.6 )] Increased pulmonary vascular resistance [see Warnings and Precautions ( 5.12 )]
Drug Interactions
Succinylcholine : Use before succinylcholine has not been studied. ( 7.11 ) Nondepolarizing muscle relaxants : Interactions have been observed. ( 7.7 ) Enhanced Rocuronium Bromide Injection activity possible : Inhalation anesthetics ( 7.3 ), certain antibiotics ( 7.1 ), quinidine ( 7.10 ), magnesium ( 7.6 ), lithium ( 7.4 ), local anesthetics ( 7.5 ), procainamide ( 7.8 ) Reduced Rocuronium Bromide Injection activity possible : Anticonvulsants. ( 7.2 )
Storage and Handling
Rocuronium Bromide Injection is a clear, colorless to yellow/orange solution, free from visible particulate matter and available in single-dose vials in the following packaging configurations: Product Code Unit of Sale Strength Each 442105 NDC 65219-442-05 Unit of 10 50 mg per 5 mL (10 mg per mL) NDC 65219-442-02 Single-Dose Vial RF442105 NDC 65219-695-05 Unit of 10 50 mg per 5 mL (10 mg per mL) NDC 65219-695-01 Single-Dose Vial This product is RFID-enabled. 442110 NDC 65219-444-10 Unit of 10 100 mg per 10 mL (10 mg per mL) NDC 65219-444-04 Single-Dose Vial RF442110 NDC 65219-697-10 Unit of 10 100 mg per 10 mL (10 mg per mL) NDC 65219-697-01 Single-Dose Vial This product is RFID-enabled. The container closure is not made with natural rubber latex. Rocuronium Bromide Injection should be stored at 20°C to 25°C (68°F to 77°F); Excursion permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature]. DO NOT FREEZE.
How Supplied
Rocuronium Bromide Injection is a clear, colorless to yellow/orange solution, free from visible particulate matter and available in single-dose vials in the following packaging configurations: Product Code Unit of Sale Strength Each 442105 NDC 65219-442-05 Unit of 10 50 mg per 5 mL (10 mg per mL) NDC 65219-442-02 Single-Dose Vial RF442105 NDC 65219-695-05 Unit of 10 50 mg per 5 mL (10 mg per mL) NDC 65219-695-01 Single-Dose Vial This product is RFID-enabled. 442110 NDC 65219-444-10 Unit of 10 100 mg per 10 mL (10 mg per mL) NDC 65219-444-04 Single-Dose Vial RF442110 NDC 65219-697-10 Unit of 10 100 mg per 10 mL (10 mg per mL) NDC 65219-697-01 Single-Dose Vial This product is RFID-enabled. The container closure is not made with natural rubber latex. Rocuronium Bromide Injection should be stored at 20°C to 25°C (68°F to 77°F); Excursion permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature]. DO NOT FREEZE.
Medication Information
Warnings and Precautions
Appropriate Administration and Monitorin g: Use only if facilities for intubation, mechanical ventilation, oxygen therapy, and an antagonist are immediately available. ( 5.1 ) Anaphylaxis : Severe anaphylaxis has been reported. Consider cross-reactivity among neuromuscular blocking agents. ( 5.2 ) Risk of Death due to Medication Errors : Accidental administration can cause death. ( 5.3 ) Need for Adequate Anesthesia : Must be accompanied by adequate anesthesia or sedation. ( 5.4 ) Residual Paralysis : Consider using a reversal agent in cases where residual paralysis is more likely to occur. ( 5.5 )
Indications and Usage
Rocuronium Bromide Injection is indicated as an adjunct to general anesthesia to facilitate both rapid sequence and routine tracheal intubation, and to provide skeletal muscle relaxation during surgery or mechanical ventilation.
Dosage and Administration
Rocuronium Bromide Injection should only be administered by experienced clinicians or trained individuals supervised by an experienced clinician familiar with the use, actions, characteristics, and complications of neuromuscular blocking agents. ( 2.1 ) Individualize the dose for each patient. ( 2.1 ) Peripheral nerve stimulator recommended for determination of drug response and need for additional doses, and to evaluate recovery. ( 2.1 ) Store Rocuronium Bromide Injection with cap and ferrule intact and in a manner that minimizes the possibility of selecting the wrong product. ( 2.1 ) Tracheal intubation : Recommended initial dose is 0.6 mg/kg. ( 2.2 ) Rapid sequence intubation : 0.6 to 1.2 mg/kg. ( 2.3 ) Maintenance doses : Guided by response to prior dose, not administered until recovery is evident. ( 2.4 ) Continuous infusion : Initial rate of 10 to 12 mcg/kg/min. Start only after early evidence of spontaneous recovery from an intubating dose. ( 2.5 )
Contraindications
Rocuronium Bromide Injection is contraindicated in patients known to have hypersensitivity (e.g., anaphylaxis) to rocuronium bromide or other neuromuscular blocking agents [see Warnings and Precautions ( 5.2 )] .
Adverse Reactions
In clinical trials, the most common adverse reactions (2%) are transient hypotension and hypertension. The following adverse reactions are described, or described in greater detail, in other sections: Anaphylaxis [see Warnings and Precautions ( 5.2 )] Residual paralysis [see Warnings and Precautions ( 5.5 )] Myopathy [see Warnings and Precautions ( 5.6 )] Increased pulmonary vascular resistance [see Warnings and Precautions ( 5.12 )]
Drug Interactions
Succinylcholine : Use before succinylcholine has not been studied. ( 7.11 ) Nondepolarizing muscle relaxants : Interactions have been observed. ( 7.7 ) Enhanced Rocuronium Bromide Injection activity possible : Inhalation anesthetics ( 7.3 ), certain antibiotics ( 7.1 ), quinidine ( 7.10 ), magnesium ( 7.6 ), lithium ( 7.4 ), local anesthetics ( 7.5 ), procainamide ( 7.8 ) Reduced Rocuronium Bromide Injection activity possible : Anticonvulsants. ( 7.2 )
Storage and Handling
Rocuronium Bromide Injection is a clear, colorless to yellow/orange solution, free from visible particulate matter and available in single-dose vials in the following packaging configurations: Product Code Unit of Sale Strength Each 442105 NDC 65219-442-05 Unit of 10 50 mg per 5 mL (10 mg per mL) NDC 65219-442-02 Single-Dose Vial RF442105 NDC 65219-695-05 Unit of 10 50 mg per 5 mL (10 mg per mL) NDC 65219-695-01 Single-Dose Vial This product is RFID-enabled. 442110 NDC 65219-444-10 Unit of 10 100 mg per 10 mL (10 mg per mL) NDC 65219-444-04 Single-Dose Vial RF442110 NDC 65219-697-10 Unit of 10 100 mg per 10 mL (10 mg per mL) NDC 65219-697-01 Single-Dose Vial This product is RFID-enabled. The container closure is not made with natural rubber latex. Rocuronium Bromide Injection should be stored at 20°C to 25°C (68°F to 77°F); Excursion permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature]. DO NOT FREEZE.
How Supplied
Rocuronium Bromide Injection is a clear, colorless to yellow/orange solution, free from visible particulate matter and available in single-dose vials in the following packaging configurations: Product Code Unit of Sale Strength Each 442105 NDC 65219-442-05 Unit of 10 50 mg per 5 mL (10 mg per mL) NDC 65219-442-02 Single-Dose Vial RF442105 NDC 65219-695-05 Unit of 10 50 mg per 5 mL (10 mg per mL) NDC 65219-695-01 Single-Dose Vial This product is RFID-enabled. 442110 NDC 65219-444-10 Unit of 10 100 mg per 10 mL (10 mg per mL) NDC 65219-444-04 Single-Dose Vial RF442110 NDC 65219-697-10 Unit of 10 100 mg per 10 mL (10 mg per mL) NDC 65219-697-01 Single-Dose Vial This product is RFID-enabled. The container closure is not made with natural rubber latex. Rocuronium Bromide Injection should be stored at 20°C to 25°C (68°F to 77°F); Excursion permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature]. DO NOT FREEZE.
Description
Rocuronium Bromide Injection is indicated as an adjunct to general anesthesia to facilitate both rapid sequence and routine tracheal intubation, and to provide skeletal muscle relaxation during surgery or mechanical ventilation.
Section 42229-5
Risk of Medication Errors
Accidental administration of neuromuscular blocking agents may be fatal. Store Rocuronium Bromide Injection with the cap and ferrule intact and in a manner that minimizes the possibility of selecting the wrong product [see Warnings and Precautions (5.3)].
Section 51945-4
PACKAGE LABEL - PRINCIPAL DISPLAY - Rocuronium 5 mL Single-Dose Vial Label
NDC 65219-442-02
5 mL
Single-Dose Vial.
Discard Unused Portion
Rocuronium
Bromide Injection
50 mg per 5 mL
(10 mg per mL)
WARNING:
Paralyzing Agent
For intravenous use only
Rx only
7.9 Propofol
The use of propofol for induction and maintenance of anesthesia does not alter the clinical duration or recovery characteristics following recommended doses of Rocuronium Bromide Injection.
10 Overdosage
Overdosage with neuromuscular blocking agents may result in neuromuscular block beyond the time needed for surgery and anesthesia. The primary treatment is maintenance of a patent airway, controlled ventilation, and adequate sedation until recovery of normal neuromuscular function is assured. Once evidence of recovery from neuromuscular block is observed, further recovery may be facilitated by administration of an anticholinesterase agent in conjunction with an appropriate anticholinergic agent.
7.6 Magnesium
Magnesium salts administered for the management of pre-eclampsia or eclampsia of pregnancy may enhance neuromuscular blockade [see Warnings and Precautions (5.10)].
11 Description
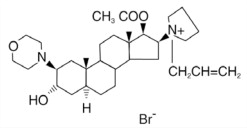
Rocuronium bromide is a nondepolarizing neuromuscular blocking agent with a rapid to intermediate onset depending on dose and intermediate duration. Rocuronium bromide is chemically designated as 1-[17β-(acetyloxy)-3α-hydroxy-2β-(4-morpholinyl)-5α-androstan-16β-yl]-1-(2-propenyl) pyrrolidinium bromide.
The structural formula is:
Rocuronium bromide USP is an almost white to pale yellow, hygroscopic powder. Rocuronium bromide is soluble in water and dichloromethane, slightly soluble in ethanol and methanol. The chemical formula is C32H53BrN2O4 with a molecular weight of 609.70. The partition coefficient of rocuronium bromide in n-octanol/water is 0.5 at 20°C.
Rocuronium Bromide Injection is supplied as a sterile, nonpyrogenic, isotonic solution that is clear, colorless to yellow/orange, for intravenous use only. Each mL contains 10 mg rocuronium bromide (equivalent to 8.69 mg of rocuronium) and inactive ingredients 17.1 mcL hydrochloric acid, 8 mg sodium chloride. The solution pH is adjusted to 2.8 to 3.2 with hydrochloric acid and/or sodium hydroxide.
7.10 Quinidine
Injection of quinidine during recovery from use of muscle relaxants is associated with recurrent paralysis. This possibility must also be considered for Rocuronium Bromide Injection [see Warnings and Precautions (5.10)].
5.2 Anaphylaxis
Severe anaphylactic reactions to neuromuscular blocking agents, including Rocuronium Bromide Injection, have been reported. These reactions have, in some cases (including cases with Rocuronium Bromide Injection), been life threatening and fatal. Due to the potential severity of these reactions, the necessary precautions, such as the immediate availability of appropriate emergency treatment, should be taken. Precautions should also be taken in those patients who have had previous anaphylactic reactions to other neuromuscular blocking agents, since cross-reactivity between neuromuscular blocking agents, both depolarizing and nondepolarizing, has been reported.
7.1 Antibiotics
Drugs which may enhance the neuromuscular blocking action of nondepolarizing agents such as Rocuronium Bromide Injection include certain antibiotics (e.g., aminoglycosides; vancomycin; tetracyclines; bacitracin; polymyxins; colistin; and sodium colistimethate). If these antibiotics are used in conjunction with Rocuronium Bromide Injection, prolongation of neuromuscular block may occur.
7.8 Procainamide
Procainamide has been shown to increase the duration of neuromuscular block and decrease infusion requirements of neuromuscular blocking agents [see Warnings and Precautions (5.10)].
8.4 Pediatric Use
The use of another rocuronium bromide injection product has been studied in pediatric patients 3 months to 14 years of age under halothane anesthesia. Of the pediatric patients anesthetized with halothane who did not receive atropine for induction, about 80% experienced a transient increase (30% or greater) in heart rate after intubation. One of the 19 infants anesthetized with halothane and fentanyl who received atropine for induction experienced this magnitude of change [see Dosage and Administration (2.6), Clinical Studies (14.3)].
Another rocuronium bromide injection product was also studied in pediatric patients up to 17 years of age, including neonates, under sevoflurane (induction) and isoflurane/nitrous oxide (maintenance) anesthesia. Onset time and clinical duration varied with dose, the age of the patient, and anesthetic technique. The overall analysis of ECG data in pediatric patients indicated that the concomitant use of Rocuronium Bromide Injection with general anesthetic agents can prolong the QTc interval. The data also suggest that Rocuronium Bromide Injection may increase heart rate. However, it was not possible to conclusively identify an effect of rocuronium bromide independent of that of anesthesia and other factors. Additionally, when examining plasma levels of rocuronium bromide in correlation to QTc interval prolongation, no relationship was observed [see Dosage and Administration (2.6), Warnings and Precautions (5.9), Clinical Studies (14.3)].
Rocuronium Bromide Injection is not recommended for rapid sequence intubation in pediatric patients. Recommendations for use in pediatric patients are discussed in other sections of labeling [see Dosage and Administration (2.6) Clinical Pharmacology (12.2)].
8.5 Geriatric Use
Another rocuronium bromide injection product was administered to 140 geriatric patients (65 years of age or older) in U.S. clinical trials and 128 geriatric patients in European clinical trials. The observed pharmacokinetic profile of rocuronium bromide for geriatric patients (n=20) was similar to that for other adult surgical patients [see Clinical Pharmacology (12.3)]. However, onset time and duration of action of rocuronium bromide were slightly longer for geriatric patients (n=43) in clinical trials. Clinical experiences and recommendations for use of Rocuronium Bromide Injection in geriatric patients are discussed in other sections of the labeling [see Dosage and Administration (2.6), Warnings and Precautions (5.5), Clinical Pharmacology (12.2), Clinical Studies (14.2)].
5.14 Extravasation
If extravasation occurs after Rocuronium Bromide Injection administration, it may be associated with signs or symptoms of local irritation. The Rocuronium Bromide Injection or infusion should be terminated immediately and restarted in another vein.
14 Clinical Studies
The effectiveness of Rocuronium Bromide Injection has been established based on adequate and well-controlled studies of another formulation of rocuronium bromide injection in adult patients. Below is a display of the efficacy results of the adequate and well-controlled studies of the other formulation of rocuronium bromide injection.
In US clinical studies, a total of 1137 patients received another rocuronium bromide injection, product, including 176 pediatric, 140 geriatric, 55 obstetric, and 766 other adults. Most patients (90%) were ASA physical status I or II, about 9% were ASA III, and 10 patients (undergoing coronary artery bypass grafting or valvular surgery) were ASA IV. In European clinical studies, a total of 1394 patients received rocuronium bromide injection, including 52 pediatric, 128 geriatric (65 years or greater), and 1214 other adults.
14.1 Adult Patients
Intubation using doses of rocuronium bromide injection 0.6 to 0.85 mg/kg was evaluated in 203 adults in 11 clinical studies. Excellent to good intubating conditions were generally achieved within 2 minutes and maximum block occurred within 3 minutes in most patients. Doses within this range provide clinical relaxation for a median (range) time of 33 (14-85) minutes under opioid/nitrous oxide/oxygen anesthesia. Larger doses (0.9 and 1.2 mg/kg) were evaluated in 2 studies with 19 and 16 patients under opioid/nitrous oxide/oxygen anesthesia and provided 58 (27-111) and 67 (38-160) minutes of clinical relaxation, respectively.
4 Contraindications
Rocuronium Bromide Injection is contraindicated in patients known to have hypersensitivity (e.g., anaphylaxis) to rocuronium bromide or other neuromuscular blocking agents [see Warnings and Precautions (5.2)].
6 Adverse Reactions
In clinical trials, the most common adverse reactions (2%) are transient hypotension and hypertension.
The following adverse reactions are described, or described in greater detail, in other sections:
7 Drug Interactions
- Succinylcholine: Use before succinylcholine has not been studied. (7.11)
- Nondepolarizing muscle relaxants: Interactions have been observed. (7.7)
- Enhanced Rocuronium Bromide Injection activity possible: Inhalation anesthetics (7.3), certain antibiotics (7.1), quinidine (7.10), magnesium (7.6), lithium (7.4), local anesthetics (7.5), procainamide (7.8)
- Reduced Rocuronium Bromide Injection activity possible: Anticonvulsants. (7.2)
7.2 Anticonvulsants
In 2 of 4 patients who received chronic anticonvulsant therapy, apparent resistance to the effects of another rocuronium bromide injection product was observed in the form of diminished magnitude of neuromuscular block or shortened clinical duration. As with other nondepolarizing neuromuscular blocking drugs, if Rocuronium Bromide Injection is administered to patients chronically receiving anticonvulsant agents such as carbamazepine or phenytoin, shorter durations of neuromuscular block may occur and infusion rates may be higher due to the development of resistance to nondepolarizing muscle relaxants. While the mechanism for development of this resistance is not known, receptor up-regulation may be a contributing factor [see Warnings and Precautions (5.10)].
7.11 Succinylcholine
The use of Rocuronium Bromide Injection before succinylcholine, for the purpose of attenuating some of the side effects of succinylcholine, has not been studied.
If Rocuronium Bromide Injection is administered following administration of succinylcholine, it should not be given until recovery from succinylcholine has been observed. The median duration of action of Rocuronium Bromide Injection 0.6 mg/kg administered after a 1 mg/kg dose of succinylcholine when T1 returned to 75% of control was 36 minutes (range: 14-57, n=12) vs. 28 minutes (range: 17-51, n=12) without succinylcholine.
12.2 Pharmacodynamics
The Rocuronium Bromide Injection dose required to produce 95% suppression (ED95) of the first [T1] mechanomyographic [MMG] response of the adductor pollicis muscle [thumb] to indirect supramaximal train-of-four stimulation of the ulnar nerve) during opioid/nitrous oxide/oxygen anesthesia is approximately 0.3 mg/kg. Patient variability around the ED95 dose suggests that 50% of patients will exhibit T1 depression of 91% to 97%.
Table 4 presents intubating conditions in patients with intubation initiated at 60 to 70 seconds.
|
* Excludes patients undergoing Cesarean section. |
||
|
† Pediatric patients were under halothane anesthesia. |
||
|
Excellent intubating conditions=jaw relaxed, vocal cords apart and immobile, no diaphragmatic movement. |
||
|
Good intubating conditions=same as excellent but with some diaphragmatic movement. |
||
| rocuronium bromide injection Dose (mg/kg) Administered Over 5 sec | Percent of Patients with Excellent or Good Intubating Conditions |
Time to Completion of Intubation (min) |
| Adults* 18 to 64 yrs 0.45 (n=43) 0.6 (n=51) |
86% 96% |
1.6 (1.0-7.0) 1.6 (1.0-3.2) |
| Infants† 3 mo to 1 yr 0.6 (n=18) |
100% | 1.0 (1.0-1.5) |
| Pediatric† 1 to 12 yrs 0.6 (n=12) |
100% | 1.0 (0.5-2.3) |
Table 5 presents the time to onset and clinical duration for the initial dose of Rocuronium Bromide Injection under opioid/nitrous oxide/oxygen anesthesia in adults and geriatric patients, and under halothane anesthesia in pediatric patients.
|
n=the number of patients who had time to maximum block recorded. |
|||
|
Clinical duration=time until return to 25% of control T1. Patients receiving doses of 0.45 mg/kg who achieved less than 90% block (16% of these patients) had about 12 to 15 minutes to 25% recovery. |
|||
| rocuronium bromide injection Dose (mg/kg) Administered Over 5 sec | Time to ≥80% Block (min) | Time to Maximum Block (min) | Clinical Duration (min) |
| Adults 18 to 64 yrs |
1.3 (0.8-6.2) |
3.0 (1.3-8.2) |
22 (12-31) |
| 0.45 (n=50) | |||
| 0.6 (n=142) | 1.0 (0.4-6.0) | 1.8 (0.6-13.0) | 31 (15-85) |
| 0.9 (n=20) | 1.1 (0.3-3.8) | 1.4 (0.8-6.2) | 58 (27-111) |
| 1.2 (n=18) | 0.7 (0.4-1.7) | 1.0 (0.6-4.7) | 67 (38-160) |
| Geriatric ≥65 yrs |
2.3 (1.0-8.3) |
3.7 (1.3-11.3) |
46 (22-73) |
| 0.6 (n=31) | |||
| 0.9 (n=5) | 2.0 (1.0-3.0) | 2.5 (1.2-5.0) | 62 (49-75) |
| 1.2 (n=7) | 1.0 (0.8-3.5) | 1.3 (1.2-4.7) | 94 (64-138) |
| Infants 3 mo to 1 yr 0.6 (n=17) 0.8 (n=9) |
— — |
0.8 (0.3-3.0) 0.7 (0.5-0.8) |
41 (24-68) 40 (27-70) |
| Pediatric 1 to 12 yrs 0.6 (n=27) 0.8 (n=18) |
0.8 (0.4-2.0) — |
1.0 (0.5-3.3) 0.5 (0.3-1.0) |
26 (17-39) 30 (17-56) |
Table 6 presents the time to onset and clinical duration for the initial dose of Rocuronium Bromide Injection (rocuronium bromide) Injection under sevoflurane (induction) and isoflurane/nitrous oxide (maintenance) anesthesia in pediatric patients.
|
n=the number of patients with the highest number of observations for time to maximum block or reappearance T3. |
||
| rocuronium bromide injection Dose (mg/kg) Administered Over 5 sec | Time to Maximum Block (min) | Time to Reappearance T3 (min) |
| Neonates birth to <28 days |
1.1 (0.6-2.2) |
40.3 (32.5-62.6) |
| 0.45 (n=5) | ||
| 0.6 (n=10) | 1.0 (0.2-2.1) | 49.7 (16.6-119.0) |
| 1 (n=6) | 0.6 (0.3-1.8) | 114.4 (92.6-136.3) |
| Infants 28 days to ≤3 mo |
0.5 (0.4-1.3) |
49.1 (13.5-79.9) |
| 0.45 (n=9) | ||
| 0.6 (n=11) | 0.4 (0.2-0.8) | 59.8 (32.3-87.8) |
| 1 (n=5) | 0.3 (0.2-0.7) | 103.3 (90.8-155.4) |
| Toddlers >3 mo to ≤2 yrs |
0.8 (0.3-1.9) |
39.2 (16.9-59.4) |
| 0.45 (n=17) | ||
| 0.6 (n=29) | 0.6 (0.2-1.6) | 44.2 (18.9-68.8) |
| 1 (n=15) | 0.5 (0.2-1.5) | 72.0 (36.2-128.2) |
| Children >2 yrs to ≤11 yrs |
0.9 (0.4-1.9) |
21.5 (17.5-38.0) |
| 0.45 (n=14) | ||
| 0.6 (n=37) | 0.8 (0.3-1.7) | 36.7 (20.1-65.9) |
| 1 (n=16) | 0.7 (0.4-1.2) | 53.1 (31.2-89.9) |
| Adolescents >11 to ≤17 yrs |
1.0 (0.5-1.7) |
37.5 (18.3-65.7) |
| 0.45 (n=18) | ||
| 0.6 (n=31) | 0.9 (0.2-2.1) | 41.4 (16.3-91.2) |
| 1 (n=14) | 0.7 (0.5-1.2) | 67.1 (25.6-93.8) |
The time to 80% or greater block and clinical duration as a function of dose are presented in
Figures 1
and
2.
Figure 1: Time to 80% or Greater Block vs. Initial Dose of rocuronium bromide injection by Age Group (Median, 25th and 75th Percentile, and Individual Values)
Figure 2: Duration of Clinical Effect vs. Initial Dose of rocuronium bromide injection by Age Group (Median, 25th and 75th Percentile, and Individual Values)
The clinical durations for the first 5 maintenance doses, in patients receiving 5 or more maintenance doses are represented in
Figure 3
[see Dosage and Administration (2.4)].
Figure 3: Duration of Clinical Effect vs. Number of rocuronium bromide injection Maintenance Doses, by Dose
Once spontaneous recovery has reached 25% of control T1, the neuromuscular block produced by rocuronium bromide injection is readily reversed with anticholinesterase agents, e.g., edrophonium or neostigmine.
The median spontaneous recovery from 25% to 75% T1 was 13 minutes in adult patients. When neuromuscular block was reversed in 36 adults at a T1 of 22% to 27%, recovery to a T1 of 89 (50-132)% and T4/T1 of 69 (38-92)% was achieved within 5 minutes. Only 5 of 320 adults reversed received an additional dose of reversal agent. The median (range) dose of neostigmine was 0.04 (0.01-0.09) mg/kg and the median (range) dose of edrophonium was 0.5 (0.3-1.0) mg/kg.
In geriatric patients (n=51) reversed with neostigmine, the median T4/T1 increased from 40% to 88% in 5 minutes.
In clinical trials with halothane, pediatric patients (n=27) who received 0.5 mg/kg edrophonium had increases in the median T4/T1 from 37% at reversal to 93% after 2 minutes. Pediatric patients (n=58) who received 1 mg/kg edrophonium had increases in the median T4/T1 from 72% at reversal to 100% after 2 minutes. Infants (n=10) who were reversed with 0.03 mg/kg neostigmine recovered from 25% to 75% T1 within 4 minutes.
There were no reports of less than satisfactory clinical recovery of neuromuscular function.
The neuromuscular blocking action of Rocuronium Bromide Injection may be enhanced in the presence of potent inhalation anesthetics [see Drug Interactions (7.3)].
7.4 Lithium Carbonate
Lithium has been shown to increase the duration of neuromuscular block and decrease infusion requirements of neuromuscular blocking agents [see Warnings and Precautions (5.10)].
7.5 Local Anesthetics
Local anesthetics have been shown to increase the duration of neuromuscular block and decrease infusion requirements of neuromuscular blocking agents [see Warnings and Precautions (5.10)].
2.4 Maintenance Dosing
Maintenance doses of 0.1, 0.15, and 0.2 mg/kg Rocuronium Bromide Injection, administered at 25% recovery of control T1 (defined as 3 twitches of train-of-four), provide a median (range) of 12 (2-31), 17 (6-50), and 24 (7-69) minutes of clinical duration under opioid/nitrous oxide/oxygen anesthesia [see Clinical Pharmacology (12.2)]. In all cases, dosing should be guided based on the clinical duration following initial dose or prior maintenance dose and not administered until recovery of neuromuscular function is evident. A clinically insignificant cumulation of effect with repetitive maintenance dosing has been observed [see Clinical Pharmacology (12.2)].
5.5 Residual Paralysis
To prevent complications resulting from residual paralysis from Rocuronium Bromide Injection, it is recommended to extubate only after the patient has recovered sufficiently from neuromuscular block. Geriatric patients (65 years or older) may be at increased risk for residual neuromuscular block. Other factors which could cause residual paralysis after extubation in the post- operative phase (such as drug interactions or patient condition) should also be considered. If not used as part of standard clinical practice the use of a reversal agent should be considered, especially in those cases where residual paralysis is more likely to occur.
1 Indications and Usage
Rocuronium Bromide Injection is indicated as an adjunct to general anesthesia to facilitate both rapid sequence and routine tracheal intubation, and to provide skeletal muscle relaxation during surgery or mechanical ventilation.
14.2 Geriatric Patients
Rocuronium bromide injection was evaluated in 55 geriatric patients (ages 65-80 years) in 6 clinical studies. Doses of 0.6 mg/kg provided excellent to good intubating conditions in a median (range) time of 2.3 (1-8) minutes. Recovery times from 25% to 75% after these doses were not prolonged in geriatric patients compared to other adult patients [see Dosage and Administration (2.6) and Use in Specific Populations (8.5)].
14.3 Pediatric Patients
Rocuronium bromide injection 0.45, 0.6, or 1 mg/kg was evaluated under sevoflurane (induction) and isoflurane/nitrous oxide (maintenance) anesthesia for intubation in 326 patients in 2 studies. In 1 of these studies maintenance bolus and infusion requirements were evaluated in 137 patients. In all age groups, doses of 0.6 mg/kg provided time to maximum block in about 1 minute. Across all age groups, median (range) time to reappearance of T3 for doses of 0.6 mg/kg was shortest in the children [36.7 (20.1-65.9) minutes] and longest in infants [59.8 (32.3-87.8) minutes]. For pediatric patients older than 3 months, the time to recovery was shorter after stopping infusion maintenance when compared with bolus maintenance [see Dosage and Administration (2.6) and Use in Specific Populations (8.4)].
Rocuronium bromide injection 0.6 or 0.8 mg/kg was evaluated for intubation in 75 pediatric patients (n=28; age 3-12 months, n=47; age 1-12 years) in 3 studies using halothane (1%-5%) and nitrous oxide (60%-70%) in oxygen. Doses of 0.6 mg/kg provided a median (range) time to maximum block of 1 (0.5-3.3) minute(s). This dose provided a median (range) time of clinical relaxation of 41 (24-68) minutes in 3-month to 1-year-old infants and 26 (17-39) minutes in 1- to 12-year-old pediatric patients [see Dosage and Administration (2.6) and Use in Specific Populations (8.4)].
12.1 Mechanism of Action
Rocuronium Bromide Injection is a nondepolarizing neuromuscular blocking agent with a rapid to intermediate onset depending on dose and intermediate duration. It acts by competing for cholinergic receptors at the motor end-plate. This action is antagonized by acetylcholinesterase inhibitors, such as neostigmine and edrophonium.
5 Warnings and Precautions
- Appropriate Administration and Monitoring: Use only if facilities for intubation, mechanical ventilation, oxygen therapy, and an antagonist are immediately available. (5.1)
- Anaphylaxis: Severe anaphylaxis has been reported. Consider cross-reactivity among neuromuscular blocking agents. (5.2)
- Risk of Death due to Medication Errors: Accidental administration can cause death. (5.3)
- Need for Adequate Anesthesia: Must be accompanied by adequate anesthesia or sedation. (5.4)
- Residual Paralysis: Consider using a reversal agent in cases where residual paralysis is more likely to occur. (5.5)
5.7 Malignant Hyperthermia
Rocuronium Bromide Injection has not been studied in malignant hyperthermia (MH)-susceptible patients. In an animal study in MH-susceptible swine, the administration of Rocuronium Bromide Injection did not appear to trigger malignant hyperthermia.
Because Rocuronium Bromide Injection is always used with other agents, and the occurrence of MH during anesthesia is possible even in the absence of known triggering agents, clinicians should be familiar with early signs, confirmatory diagnosis, and treatment of MH prior to the start of any anesthetic [see Adverse Reactions (6.2)].
7.3 Inhalation Anesthetics
Use of inhalation anesthetics (enflurane > isoflurane > halothane) has been shown to enhance the activity of other neuromuscular blocking agents. Isoflurane and enflurane may also prolong the duration of action of initial and maintenance doses of Rocuronium Bromide Injection and decrease the average infusion requirement of Rocuronium Bromide Injection by 40% compared to opioid/nitrous oxide/oxygen anesthesia. No definite interaction between rocuronium bromide injection and halothane has been demonstrated. In one study, use of enflurane in 10 patients who received another rocuronium bromide injection product resulted in a 20% increase in mean clinical duration of the initial intubating dose, and a 37% increase in the duration of subsequent maintenance doses, when compared in the same study to 10 patients under opioid/nitrous oxide/oxygen anesthesia. The clinical duration of initial doses of another rocuronium bromide injection product (0.57 to 0.85 mg/kg) under enflurane or isoflurane anesthesia, as used clinically, was increased by 11% and 23%, respectively. The duration of maintenance doses was affected to a greater extent, increasing by 30% to 50% under either enflurane or isoflurane anesthesia.
Potentiation by these agents was also observed with respect to the infusion rates of rocuronium bromide injection required to maintain approximately 95% neuromuscular block. Under isoflurane and enflurane anesthesia, the infusion rates were decreased by approximately 40% compared to opioid/nitrous oxide/oxygen anesthesia. The median spontaneous recovery time (from 25% to 75% of control T1) was not affected by halothane but is prolonged by enflurane (15% longer) and isoflurane (62% longer). Reversal-induced recovery of Rocuronium Bromide Injection neuromuscular block is minimally affected by anesthetic technique [see Dosage and Administration (2.6) and Warnings and Precautions (5.10)].
2 Dosage and Administration
Rocuronium Bromide Injection should only be administered by experienced clinicians or trained individuals supervised by an experienced clinician familiar with the use, actions, characteristics, and complications of neuromuscular blocking agents. (2.1)
- Individualize the dose for each patient. (2.1)
- Peripheral nerve stimulator recommended for determination of drug response and need for additional doses, and to evaluate recovery. (2.1)
- Store Rocuronium Bromide Injection with cap and ferrule intact and in a manner that minimizes the possibility of selecting the wrong product. (2.1)
- Tracheal intubation: Recommended initial dose is 0.6 mg/kg. (2.2)
- Rapid sequence intubation: 0.6 to 1.2 mg/kg. (2.3)
- Maintenance doses: Guided by response to prior dose, not administered until recovery is evident. (2.4)
- Continuous infusion: Initial rate of 10 to 12 mcg/kg/min. Start only after early evidence of spontaneous recovery from an intubating dose. (2.5)
3 Dosage Forms and Strengths
Rocuronium Bromide Injection is a clear, colorless to yellow/orange solution, free from visible particulate matter available as:
- 50 mg/5 mL (10 mg/mL), single-dose vials
- 100 mg/10 mL (10 mg per mL), single-dose vials
5.9 Qt Interval Prolongation
The overall analysis of ECG data in pediatric patients indicates that the concomitant use of Rocuronium Bromide Injection with general anesthetic agents can prolong the QTc interval [see Clinical Studies (14.3)].
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of rocuronium bromide injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune system disorders: In clinical practice, there have been reports of severe allergic reactions (anaphylactic reactions and shock) with rocuronium bromide injection, including some that have been life-threatening and fatal [see Warnings and Precautions (5.2)].
General disorders and administration site conditions: There have been reports of malignant hyperthermia with the use of rocuronium bromide injection [see Warnings and Precautions (5.7)].
2.3 Rapid Sequence Intubation
In appropriately premedicated and adequately anesthetized patients, Rocuronium Bromide Injection 0.6 to 1.2 mg/kg will provide excellent or good intubating conditions in most patients in less than 2 minutes [see Clinical Studies (14.1)].
8 Use in Specific Populations
2.5 Use By Continuous Infusion
Infusion at an initial rate of 10 to 12 mcg/kg/min of Rocuronium Bromide Injection should be initiated only after early evidence of spontaneous recovery from an intubating dose. Due to rapid redistribution [see Clinical Pharmacology (12.3)] and the associated rapid spontaneous recovery, initiation of the infusion after substantial return of neuromuscular function (more than 10% of controlT1) may necessitate additional bolus doses to maintain adequate block for surgery.
Upon reaching the desired level of neuromuscular block, the infusion of Rocuronium Bromide Injection must be individualized for each patient. The rate of administration should be adjusted according to the patient's twitch response as monitored with the use of a peripheral nerve stimulator. In clinical trials, infusion rates have ranged from 4 to 16 mcg/kg/min.
Inhalation anesthetics, particularly enflurane and isoflurane, may enhance the neuromuscular blocking action of nondepolarizing muscle relaxants. In the presence of steady-state concentrations of enflurane or isoflurane, it may be necessary to reduce the rate of infusion by 30% to 50%, at 45 to 60 minutes after the intubating dose.
Spontaneous recovery and reversal of neuromuscular blockade following discontinuation of Rocuronium Bromide Injection infusion may be expected to proceed at rates comparable to that following comparable total doses administered by repetitive bolus injections [see Clinical Pharmacology (12.2)].
Infusion solutions of Rocuronium Bromide Injection can be prepared by mixing Rocuronium Bromide Injection with an appropriate infusion solution such as 5% glucose in water or lactated Ringers [see Dosage and Administration (2.7)]. These infusion solutions should be used within 24 hours of mixing. Unused portions of infusion solutions should be discarded.
Infusion rates of Rocuronium Bromide Injection can be individualized for each patient using the following tables for 3 different concentrations of Rocuronium Bromide Injection solution as guidelines:
|
* 50 mg Rocuronium Bromide Injection in 100 mL solution. |
|||||||||||
| Patient Weight | Drug Delivery Rate (mcg/kg/min) | ||||||||||
| (kg) | (lbs) | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 12 | 14 | 16 |
| Infusion Delivery Rate (mL/hr) | |||||||||||
| 10 | 22 | 4.8 | 6 | 7.2 | 8.4 | 9.6 | 10.8 | 12 | 14.4 | 16.8 | 19.2 |
| 15 | 33 | 7.2 | 9 | 10.8 | 12.6 | 14.4 | 16.2 | 18 | 21.6 | 25.2 | 28.8 |
| 20 | 44 | 9.6 | 12 | 14.4 | 16.8 | 19.2 | 21.6 | 24 | 28.8 | 33.6 | 38.4 |
| 25 | 55 | 12 | 15 | 18 | 21 | 24 | 27 | 30 | 36 | 42 | 48 |
| 35 | 77 | 16.8 | 21 | 25.2 | 29.4 | 33.6 | 37.8 | 42 | 50.4 | 58.8 | 67.2 |
| 50 | 110 | 24 | 30 | 36 | 42 | 48 | 54 | 60 | 72 | 84 | 96 |
| 60 | 132 | 28.8 | 36 | 43.2 | 50.4 | 57.6 | 64.8 | 72 | 86.4 | 100.8 | 115.2 |
| 70 | 154 | 33.6 | 42 | 50.4 | 58.8 | 67.2 | 75.6 | 84 | 100.8 | 117.6 | 134.4 |
| 80 | 176 | 38.4 | 48 | 57.6 | 67.2 | 76.8 | 86.4 | 96 | 115.2 | 134.4 | 153.6 |
| 90 | 198 | 43.2 | 54 | 64.8 | 75.6 | 86.4 | 97.2 | 108 | 129.6 | 151.2 | 172.8 |
| 100 | 220 | 48 | 60 | 72 | 84 | 96 | 108 | 120 | 144 | 168 | 192 |
|
* 100 mg Rocuronium Bromide Injection in 100 mL solution. |
|||||||||||
| Patient | Weight | Drug Delivery Rate (mcg/kg/min) | |||||||||
| (kg) | (lbs) | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 12 | 14 | 16 |
| Infusion Delivery Rate (mL/hr) | |||||||||||
| 10 | 22 | 2.4 | 3 | 3.6 | 4.2 | 4.8 | 5.4 | 6 | 7.2 | 8.4 | 9.6 |
| 15 | 33 | 3.6 | 4.5 | 5.4 | 6.3 | 7.2 | 8.1 | 9 | 10.8 | 12.6 | 14.4 |
| 20 | 44 | 4.8 | 6 | 7.2 | 8.4 | 9.6 | 10.8 | 12 | 14.4 | 16.8 | 19.2 |
| 25 | 55 | 6 | 7.5 | 9 | 10.5 | 12 | 13.5 | 15 | 18 | 21 | 24 |
| 35 | 77 | 8.4 | 10.5 | 12.6 | 14.7 | 16.8 | 18.9 | 21 | 25.2 | 29.4 | 33.6 |
| 50 | 110 | 12 | 15 | 18 | 21 | 24 | 27 | 30 | 36 | 42 | 48 |
| 60 | 132 | 14.4 | 18 | 21.6 | 25.2 | 28.8 | 32.4 | 36 | 43.2 | 50.4 | 57.6 |
| 70 | 154 | 16.8 | 21 | 25.2 | 29.4 | 33.6 | 37.8 | 42 | 50.4 | 58.8 | 67.2 |
| 80 | 176 | 19.2 | 24 | 28.8 | 33.6 | 38.4 | 43.2 | 48 | 57.6 | 67.2 | 76.8 |
| 90 | 198 | 21.6 | 27 | 32.4 | 37.8 | 43.2 | 48.6 | 54 | 64.8 | 75.6 | 86.4 |
| 100 | 220 | 24 | 30 | 36 | 42 | 48 | 54 | 60 | 72 | 84 | 96 |
|
* 500 mg Rocuronium Bromide Injection in 100 mL solution. |
|||||||||||
| Patient | Weight | Drug Delivery Rate (mcg/kg/min) | |||||||||
| (kg) | (lbs) | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 12 | 14 | 16 |
| Infusion Delivery Rate (mL/hr) | |||||||||||
| 10 | 22 | 0.5 | 0.6 | 0.7 | 0.8 | 1 | 1.1 | 1.2 | 1.4 | 1.7 | 1.9 |
| 15 | 33 | 0.7 | 0.9 | 1.1 | 1.3 | 1.4 | 1.6 | 1 8 | 2.2 | 2.5 | 2.9 |
| 20 | 44 | 1 | 1.2 | 1.4 | 1.7 | 1.9 | 2.2 | 2.4 | 2.9 | 3.4 | 3.8 |
| 25 | 55 | 1.2 | 1.5 | 1.8 | 2.1 | 2.4 | 2.7 | 3 | 3.6 | 4.2 | 4.8 |
| 35 | 77 | 1.7 | 2.1 | 2.5 | 2.9 | 3.4 | 3.8 | 4.2 | 5 | 5.9 | 6.7 |
| 50 | 110 | 2.4 | 3 | 3.6 | 4.2 | 4.8 | 5.4 | 6 | 7.2 | 8.4 | 9.6 |
| 60 | 132 | 2.9 | 3.6 | 4.3 | 5 | 5.8 | 6.5 | 7.2 | 8.6 | 10.1 | 11.5 |
| 70 | 154 | 3.4 | 4.2 | 5 | 5.9 | 6.7 | 7.6 | 8.4 | 10.1 | 11.8 | 13.4 |
| 80 | 176 | 3.8 | 4.8 | 5.8 | 6.7 | 7.7 | 8.6 | 9.6 | 11.5 | 13.4 | 15.4 |
| 90 | 198 | 4.3 | 5.4 | 6.5 | 7.6 | 8.6 | 9.7 | 10.8 | 13 | 15.1 | 17.3 |
| 100 | 220 | 4.8 | 6 | 7.2 | 8.4 | 9.6 | 10.8 | 12 | 14.4 | 16.8 | 19.2 |
5.8 Prolonged Circulation Time
Conditions associated with an increased circulatory delayed time, e.g., cardiovascular disease or advanced age, in patients treated with Rocuronium Bromide Injection may be associated with a delay in onset time [see Dosage and Administration (2.6)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Clinical studies of another rocuronium bromide injection product in the U.S. (n=1137) and Europe (n=1394) totaled 2531 patients. The following adverse reactions were reported in patients administered another rocuronium bromide injection (all events judged by investigators during the clinical trials to have a possible causal relationship):
Adverse reactions in greater than 1% of patients: None
Adverse reactions in less than 1% of patients (probably related or relationship unknown):
-
Cardiovascular: arrhythmia, abnormal electrocardiogram, tachycardia
-
Digestive: nausea, vomiting
-
Respiratory: asthma (bronchospasm, wheezing, or rhonchi), hiccup
-
Skin and Appendages: rash, injection site edema, pruritus
In the European studies of another rocuronium bromide injection product, the most commonly reported reactions were transient hypotension (2%) and hypertension (2%); these are in greater frequency than the US studies (0.1% and 0.1%). Changes in heart rate and blood pressure were defined differently from in the US studies in which changes in cardiovascular parameters were not considered as adverse events unless judged by the investigator as unexpected, clinically significant, or thought to be histamine related.
In a clinical study of another rocuronium bromide injection product in patients with clinically significant cardiovascular disease undergoing coronary artery bypass graft, hypertension and tachycardia were reported in some patients, but these occurrences were less frequent in patients receiving beta or calcium channel-blocking drugs. In some patients, the use of another rocuronium bromide injection product was associated with transient increases (30% or greater) in pulmonary vascular resistance. In another clinical study of patients undergoing abdominal aortic surgery, transient increases (30% or greater) in pulmonary vascular resistance were observed in about 24% of patients who received another rocuronium bromide injection product at 0.6 or 0.9 mg/kg.
In pediatric patient studies worldwide of another rocuronium bromide injection product (n=704), tachycardia occurred at an incidence of 5.3% (n=37), and it was judged by the investigator as related in 10 cases (1.4%).
2.2 Dose for Tracheal Intubation
The recommended initial dose of Rocuronium Bromide Injection, regardless of anesthetic technique, is 0.6 mg/kg. Neuromuscular block sufficient for intubation (80% block or greater) is attained in a median (range) time of 1 (0.4-6) minute(s) and most patients have intubation completed within 2 minutes. Maximum blockade is achieved in most patients in less than 3 minutes. This dose may be expected to provide 31 (15-85) minutes of clinical relaxation under opioid/nitrous oxide/oxygen anesthesia. Under halothane, isoflurane, and enflurane anesthesia, some extension of the period of clinical relaxation should be expected [see Drug Interactions (7.3)].
A lower dose of Rocuronium Bromide Injection (0.45 mg/kg) may be used. Neuromuscular block sufficient for intubation (80% block or greater) is attained in a median (range) time of 1.3 (0.8-6.2) minute(s), and most patients have intubation completed within 2 minutes. Maximum blockade is achieved in most patients in less than 4 minutes. This dose may be expected to provide 22 (12-31) minutes of clinical relaxation under opioid/nitrous oxide/oxygen anesthesia. Patients receiving this low dose of 0.45 mg/kg who achieve less than 90% block (about 16% of these patients) may have a more rapid time to 25% recovery, 12 to 15 minutes.
A large bolus dose of 0.9 or 1.2 mg/kg can be administered under opioid/nitrous oxide/oxygen anesthesia without adverse effects to the cardiovascular system [see Clinical Pharmacology (12.2)].
5.4 Need for Adequate Anesthesia
Rocuronium Bromide Injection has no known effect on consciousness, pain threshold, or cerebration. Therefore, its administration must be accompanied by adequate anesthesia or sedation.
17 Patient Counseling Information
Obtain information about your patient's medical history, current medications, any history of hypersensitivity to rocuronium bromide or other neuromuscular blocking agents. If applicable, inform your patients that certain medical conditions and medications might influence how Rocuronium Bromide Injection works [see Warnings and Precautions (5.1), Drug Interactions (7)].
In addition, inform your patient that severe anaphylactic reactions to neuromuscular blocking agents, including Rocuronium Bromide Injection, have been reported. Because allergic cross-reactivity has been reported in this class, request information from your patients about previous anaphylactic reactions to other neuromuscular blocking agents [see Warnings and Precautions (5.2)].
Manufactured by:
www.fresenius-kabi.com/us
451851
8.7 Patients With Renal Impairment
Due to the limited role of the kidney in the excretion of rocuronium bromide, usual dosing recommendations Rocuronium Bromide Injection for should be followed. In patients with renal dysfunction, the duration of neuromuscular blockade was not prolonged; however, there was substantial individual variability (range: 22-90 minutes) [see Clinical Pharmacology (12.3)].
16 How Supplied/storage and Handling
Rocuronium Bromide Injection is a clear, colorless to yellow/orange solution, free from visible particulate matter and available in single-dose vials in the following packaging configurations:
| Product Code | Unit of Sale | Strength | Each |
| 442105 | NDC 65219-442-05 Unit of 10 |
50 mg per 5 mL (10 mg per mL) |
NDC 65219-442-02 Single-Dose Vial |
| RF442105 | NDC 65219-695-05 Unit of 10 |
50 mg per 5 mL (10 mg per mL) |
NDC 65219-695-01 Single-Dose Vial This product is RFID-enabled. |
| 442110 | NDC 65219-444-10 Unit of 10 |
100 mg per 10 mL (10 mg per mL) |
NDC 65219-444-04 Single-Dose Vial |
| RF442110 | NDC 65219-697-10 Unit of 10 |
100 mg per 10 mL (10 mg per mL) |
NDC 65219-697-01 Single-Dose Vial This product is RFID-enabled. |
The container closure is not made with natural rubber latex.
Rocuronium Bromide Injection should be stored at 20°C to 25°C (68°F to 77°F); Excursion permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature]. DO NOT FREEZE.
5.13 Use in Patients With Myasthenia
In patients with myasthenia gravis or myasthenic (Eaton-Lambert) syndrome, small doses of nondepolarizing neuromuscular blocking agents, including Rocuronium Bromide Injection, may have profound effects. In such patients, a peripheral nerve stimulator and use of a small test dose may be of value in monitoring the response to administration of muscle relaxants.
7.7 Nondepolarizing Muscle Relaxants
There are no controlled studies documenting the use of Rocuronium Bromide Injection before or after other nondepolarizing muscle relaxants. Interactions have been observed when other nondepolarizing muscle relaxants have been administered in succession.
8.6 Patients With Hepatic Impairment
Because rocuronium bromide is primarily excreted by the liver, it should be used with caution in patients with clinically significant hepatic impairment. Another rocuronium bromide injection product (0.6 mg/kg) was studied in a limited number of patients (n=9) with clinically significant hepatic impairment under steady-state isoflurane anesthesia. After rocuronium bromide injection administration (0.6 mg/kg), the median (range) clinical duration of 60 (35-166) minutes was moderately prolonged compared to 42 minutes in patients with normal hepatic function. The median recovery time of 53 minutes was also prolonged in patients with cirrhosis compared to 20 minutes in patients with normal hepatic function. Four of 8 patients with cirrhosis, who received the other rocuronium bromide injection product (0.6 mg/kg) under opioid/nitrous oxide/oxygen anesthesia, did not achieve complete block. These findings are consistent with the increase in volume of distribution at steady state observed in patients with significant hepatic impairment [see Clinical Pharmacology (12.3)]. If used for rapid sequence induction in patients with ascites, an increased initial dosage of Rocuronium Bromide Injection may be necessary to assure complete block. Duration will be prolonged in these cases. The use of Rocuronium Bromide Injection doses higher than 0.6 mg/kg has not been studied [see Dosage and Administration (2.6)].
5.3 Risk of Death Due to Medication Errors
Administration of Rocuronium Bromide Injection results in paralysis, which may lead to respiratory arrest and death, a progression that may be more likely to occur in a patient for whom it is not intended. Confirm proper selection of intended product and avoid confusion with other injectable solutions that are present in critical care and other clinical settings. If another healthcare provider is administering Rocuronium Bromide Injection, ensure that the intended dose is clearly labeled and communicated.
5.6 Long Term Use in An Intensive Care Unit
Rocuronium Bromide Injection has not been studied for long-term use in the intensive care unit (ICU). As with other nondepolarizing neuromuscular blocking drugs, apparent tolerance to Rocuronium Bromide Injection may develop during chronic administration in the ICU. While the mechanism for development of this resistance is not known, receptor up-regulation may be a contributing factor. It is strongly recommended that neuromuscular transmission be monitored continuously during Rocuronium Bromide Injection administration and recovery with the help of a nerve stimulator. Additional doses of Rocuronium Bromide Injection or any other neuromuscular blocking agent should not be given until there is a definite response (one twitch of the train-of-four) to nerve stimulation. Prolonged paralysis and/or skeletal muscle weakness may be noted during initial attempts to wean from the ventilator patients who have chronically received neuromuscular blocking drugs in the ICU.
Myopathy after long-term administration of other nondepolarizing neuromuscular blocking agents in the ICU alone or in combination with corticosteroid therapy has been reported. Therefore, for patients receiving both neuromuscular blocking agents (including Rocuronium Bromide Injection) and corticosteroids, the period of use of the neuromuscular blocking agent should be limited as much as possible and only used in the setting where in the opinion of the prescribing physician, the specific advantages of the drug outweigh the risk.
5.11 Incompatibility With Alkaline Solutions
Rocuronium Bromide Injection, which has an acid pH, should not be mixed with alkaline solutions (e.g., barbiturate solutions) in the same syringe or administered simultaneously during intravenous infusion through the same needle.
5.1 Appropriate Administration and Monitoring
Rocuronium Bromide Injection should be administered in carefully adjusted dosages by or under the supervision of experienced clinicians who are familiar with the drug's actions and the possible complications of its use. Rocuronium Bromide Injection should not be administered unless facilities for intubation, mechanical ventilation, oxygen therapy, and an antagonist are immediately available. It is recommended that clinicians administering neuromuscular blocking agents such as Rocuronium Bromide Injection employ a peripheral nerve stimulator to monitor drug effect, need for additional doses, adequacy of spontaneous recovery or antagonism, and to decrease the complications of overdosage if additional doses are administered.
5.12 Increase in Pulmonary Vascular Resistance
Rocuronium Bromide Injection may be associated with increased pulmonary vascular resistance, so caution is appropriate in patients with pulmonary hypertension or valvular heart disease [see Clinical Studies (14.1)].
2.1 Important Dosing and Administration Information
Rocuronium Bromide Injection is for intravenous use only.
Rocuronium Bromide Injection should only be administered by experienced clinicians or trained individuals supervised by an experienced clinician familiar with the use, actions, characteristics, and complications of neuromuscular blocking agents. Doses of Rocuronium Bromide Injection should be individualized and a peripheral nerve stimulator should be used to monitor drug effect, need for additional doses, adequacy of spontaneous recovery or antagonism, and to decrease the complications of overdosage if additional doses are administered.
The dosage information which follows is derived from studies based upon units of drug per unit of body weight. It is intended to serve as an initial guide to clinicians familiar with other neuromuscular blocking agents to acquire experience with Rocuronium Bromide Injection.
In patients in whom potentiation of, or resistance to, neuromuscular block is anticipated, a dose adjustment should be considered [see Dosage and Administration (2.6), Warnings and Precautions (5.10, 5.13), Drug Interactions (7.2, 7.3, 7.4, 7.5, 7.6, 7.8, 7.10), Use in Specific Populations (8.6)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies in animals have not been performed with rocuronium bromide to evaluate carcinogenic potential or impairment of fertility. Mutagenicity studies (Ames test, analysis of chromosomal aberrations in mammalian cells, and micronucleus test) conducted with rocuronium bromide did not suggest mutagenic potential.
Structured Label Content
Section 42229-5 (42229-5)
Risk of Medication Errors
Accidental administration of neuromuscular blocking agents may be fatal. Store Rocuronium Bromide Injection with the cap and ferrule intact and in a manner that minimizes the possibility of selecting the wrong product [see Warnings and Precautions (5.3)].
Section 51945-4 (51945-4)
PACKAGE LABEL - PRINCIPAL DISPLAY - Rocuronium 5 mL Single-Dose Vial Label
NDC 65219-442-02
5 mL
Single-Dose Vial.
Discard Unused Portion
Rocuronium
Bromide Injection
50 mg per 5 mL
(10 mg per mL)
WARNING:
Paralyzing Agent
For intravenous use only
Rx only
7.9 Propofol
The use of propofol for induction and maintenance of anesthesia does not alter the clinical duration or recovery characteristics following recommended doses of Rocuronium Bromide Injection.
10 Overdosage (10 OVERDOSAGE)
Overdosage with neuromuscular blocking agents may result in neuromuscular block beyond the time needed for surgery and anesthesia. The primary treatment is maintenance of a patent airway, controlled ventilation, and adequate sedation until recovery of normal neuromuscular function is assured. Once evidence of recovery from neuromuscular block is observed, further recovery may be facilitated by administration of an anticholinesterase agent in conjunction with an appropriate anticholinergic agent.
7.6 Magnesium
Magnesium salts administered for the management of pre-eclampsia or eclampsia of pregnancy may enhance neuromuscular blockade [see Warnings and Precautions (5.10)].
11 Description (11 DESCRIPTION)
Rocuronium bromide is a nondepolarizing neuromuscular blocking agent with a rapid to intermediate onset depending on dose and intermediate duration. Rocuronium bromide is chemically designated as 1-[17β-(acetyloxy)-3α-hydroxy-2β-(4-morpholinyl)-5α-androstan-16β-yl]-1-(2-propenyl) pyrrolidinium bromide.
The structural formula is:
Rocuronium bromide USP is an almost white to pale yellow, hygroscopic powder. Rocuronium bromide is soluble in water and dichloromethane, slightly soluble in ethanol and methanol. The chemical formula is C32H53BrN2O4 with a molecular weight of 609.70. The partition coefficient of rocuronium bromide in n-octanol/water is 0.5 at 20°C.
Rocuronium Bromide Injection is supplied as a sterile, nonpyrogenic, isotonic solution that is clear, colorless to yellow/orange, for intravenous use only. Each mL contains 10 mg rocuronium bromide (equivalent to 8.69 mg of rocuronium) and inactive ingredients 17.1 mcL hydrochloric acid, 8 mg sodium chloride. The solution pH is adjusted to 2.8 to 3.2 with hydrochloric acid and/or sodium hydroxide.
7.10 Quinidine
Injection of quinidine during recovery from use of muscle relaxants is associated with recurrent paralysis. This possibility must also be considered for Rocuronium Bromide Injection [see Warnings and Precautions (5.10)].
5.2 Anaphylaxis
Severe anaphylactic reactions to neuromuscular blocking agents, including Rocuronium Bromide Injection, have been reported. These reactions have, in some cases (including cases with Rocuronium Bromide Injection), been life threatening and fatal. Due to the potential severity of these reactions, the necessary precautions, such as the immediate availability of appropriate emergency treatment, should be taken. Precautions should also be taken in those patients who have had previous anaphylactic reactions to other neuromuscular blocking agents, since cross-reactivity between neuromuscular blocking agents, both depolarizing and nondepolarizing, has been reported.
7.1 Antibiotics
Drugs which may enhance the neuromuscular blocking action of nondepolarizing agents such as Rocuronium Bromide Injection include certain antibiotics (e.g., aminoglycosides; vancomycin; tetracyclines; bacitracin; polymyxins; colistin; and sodium colistimethate). If these antibiotics are used in conjunction with Rocuronium Bromide Injection, prolongation of neuromuscular block may occur.
7.8 Procainamide
Procainamide has been shown to increase the duration of neuromuscular block and decrease infusion requirements of neuromuscular blocking agents [see Warnings and Precautions (5.10)].
8.4 Pediatric Use
The use of another rocuronium bromide injection product has been studied in pediatric patients 3 months to 14 years of age under halothane anesthesia. Of the pediatric patients anesthetized with halothane who did not receive atropine for induction, about 80% experienced a transient increase (30% or greater) in heart rate after intubation. One of the 19 infants anesthetized with halothane and fentanyl who received atropine for induction experienced this magnitude of change [see Dosage and Administration (2.6), Clinical Studies (14.3)].
Another rocuronium bromide injection product was also studied in pediatric patients up to 17 years of age, including neonates, under sevoflurane (induction) and isoflurane/nitrous oxide (maintenance) anesthesia. Onset time and clinical duration varied with dose, the age of the patient, and anesthetic technique. The overall analysis of ECG data in pediatric patients indicated that the concomitant use of Rocuronium Bromide Injection with general anesthetic agents can prolong the QTc interval. The data also suggest that Rocuronium Bromide Injection may increase heart rate. However, it was not possible to conclusively identify an effect of rocuronium bromide independent of that of anesthesia and other factors. Additionally, when examining plasma levels of rocuronium bromide in correlation to QTc interval prolongation, no relationship was observed [see Dosage and Administration (2.6), Warnings and Precautions (5.9), Clinical Studies (14.3)].
Rocuronium Bromide Injection is not recommended for rapid sequence intubation in pediatric patients. Recommendations for use in pediatric patients are discussed in other sections of labeling [see Dosage and Administration (2.6) Clinical Pharmacology (12.2)].
8.5 Geriatric Use
Another rocuronium bromide injection product was administered to 140 geriatric patients (65 years of age or older) in U.S. clinical trials and 128 geriatric patients in European clinical trials. The observed pharmacokinetic profile of rocuronium bromide for geriatric patients (n=20) was similar to that for other adult surgical patients [see Clinical Pharmacology (12.3)]. However, onset time and duration of action of rocuronium bromide were slightly longer for geriatric patients (n=43) in clinical trials. Clinical experiences and recommendations for use of Rocuronium Bromide Injection in geriatric patients are discussed in other sections of the labeling [see Dosage and Administration (2.6), Warnings and Precautions (5.5), Clinical Pharmacology (12.2), Clinical Studies (14.2)].
5.14 Extravasation
If extravasation occurs after Rocuronium Bromide Injection administration, it may be associated with signs or symptoms of local irritation. The Rocuronium Bromide Injection or infusion should be terminated immediately and restarted in another vein.
14 Clinical Studies (14 CLINICAL STUDIES)
The effectiveness of Rocuronium Bromide Injection has been established based on adequate and well-controlled studies of another formulation of rocuronium bromide injection in adult patients. Below is a display of the efficacy results of the adequate and well-controlled studies of the other formulation of rocuronium bromide injection.
In US clinical studies, a total of 1137 patients received another rocuronium bromide injection, product, including 176 pediatric, 140 geriatric, 55 obstetric, and 766 other adults. Most patients (90%) were ASA physical status I or II, about 9% were ASA III, and 10 patients (undergoing coronary artery bypass grafting or valvular surgery) were ASA IV. In European clinical studies, a total of 1394 patients received rocuronium bromide injection, including 52 pediatric, 128 geriatric (65 years or greater), and 1214 other adults.
14.1 Adult Patients
Intubation using doses of rocuronium bromide injection 0.6 to 0.85 mg/kg was evaluated in 203 adults in 11 clinical studies. Excellent to good intubating conditions were generally achieved within 2 minutes and maximum block occurred within 3 minutes in most patients. Doses within this range provide clinical relaxation for a median (range) time of 33 (14-85) minutes under opioid/nitrous oxide/oxygen anesthesia. Larger doses (0.9 and 1.2 mg/kg) were evaluated in 2 studies with 19 and 16 patients under opioid/nitrous oxide/oxygen anesthesia and provided 58 (27-111) and 67 (38-160) minutes of clinical relaxation, respectively.
4 Contraindications (4 CONTRAINDICATIONS)
Rocuronium Bromide Injection is contraindicated in patients known to have hypersensitivity (e.g., anaphylaxis) to rocuronium bromide or other neuromuscular blocking agents [see Warnings and Precautions (5.2)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
In clinical trials, the most common adverse reactions (2%) are transient hypotension and hypertension.
The following adverse reactions are described, or described in greater detail, in other sections:
7 Drug Interactions (7 DRUG INTERACTIONS)
- Succinylcholine: Use before succinylcholine has not been studied. (7.11)
- Nondepolarizing muscle relaxants: Interactions have been observed. (7.7)
- Enhanced Rocuronium Bromide Injection activity possible: Inhalation anesthetics (7.3), certain antibiotics (7.1), quinidine (7.10), magnesium (7.6), lithium (7.4), local anesthetics (7.5), procainamide (7.8)
- Reduced Rocuronium Bromide Injection activity possible: Anticonvulsants. (7.2)
7.2 Anticonvulsants
In 2 of 4 patients who received chronic anticonvulsant therapy, apparent resistance to the effects of another rocuronium bromide injection product was observed in the form of diminished magnitude of neuromuscular block or shortened clinical duration. As with other nondepolarizing neuromuscular blocking drugs, if Rocuronium Bromide Injection is administered to patients chronically receiving anticonvulsant agents such as carbamazepine or phenytoin, shorter durations of neuromuscular block may occur and infusion rates may be higher due to the development of resistance to nondepolarizing muscle relaxants. While the mechanism for development of this resistance is not known, receptor up-regulation may be a contributing factor [see Warnings and Precautions (5.10)].
7.11 Succinylcholine
The use of Rocuronium Bromide Injection before succinylcholine, for the purpose of attenuating some of the side effects of succinylcholine, has not been studied.
If Rocuronium Bromide Injection is administered following administration of succinylcholine, it should not be given until recovery from succinylcholine has been observed. The median duration of action of Rocuronium Bromide Injection 0.6 mg/kg administered after a 1 mg/kg dose of succinylcholine when T1 returned to 75% of control was 36 minutes (range: 14-57, n=12) vs. 28 minutes (range: 17-51, n=12) without succinylcholine.
12.2 Pharmacodynamics
The Rocuronium Bromide Injection dose required to produce 95% suppression (ED95) of the first [T1] mechanomyographic [MMG] response of the adductor pollicis muscle [thumb] to indirect supramaximal train-of-four stimulation of the ulnar nerve) during opioid/nitrous oxide/oxygen anesthesia is approximately 0.3 mg/kg. Patient variability around the ED95 dose suggests that 50% of patients will exhibit T1 depression of 91% to 97%.
Table 4 presents intubating conditions in patients with intubation initiated at 60 to 70 seconds.
|
* Excludes patients undergoing Cesarean section. |
||
|
† Pediatric patients were under halothane anesthesia. |
||
|
Excellent intubating conditions=jaw relaxed, vocal cords apart and immobile, no diaphragmatic movement. |
||
|
Good intubating conditions=same as excellent but with some diaphragmatic movement. |
||
| rocuronium bromide injection Dose (mg/kg) Administered Over 5 sec | Percent of Patients with Excellent or Good Intubating Conditions |
Time to Completion of Intubation (min) |
| Adults* 18 to 64 yrs 0.45 (n=43) 0.6 (n=51) |
86% 96% |
1.6 (1.0-7.0) 1.6 (1.0-3.2) |
| Infants† 3 mo to 1 yr 0.6 (n=18) |
100% | 1.0 (1.0-1.5) |
| Pediatric† 1 to 12 yrs 0.6 (n=12) |
100% | 1.0 (0.5-2.3) |
Table 5 presents the time to onset and clinical duration for the initial dose of Rocuronium Bromide Injection under opioid/nitrous oxide/oxygen anesthesia in adults and geriatric patients, and under halothane anesthesia in pediatric patients.
|
n=the number of patients who had time to maximum block recorded. |
|||
|
Clinical duration=time until return to 25% of control T1. Patients receiving doses of 0.45 mg/kg who achieved less than 90% block (16% of these patients) had about 12 to 15 minutes to 25% recovery. |
|||
| rocuronium bromide injection Dose (mg/kg) Administered Over 5 sec | Time to ≥80% Block (min) | Time to Maximum Block (min) | Clinical Duration (min) |
| Adults 18 to 64 yrs |
1.3 (0.8-6.2) |
3.0 (1.3-8.2) |
22 (12-31) |
| 0.45 (n=50) | |||
| 0.6 (n=142) | 1.0 (0.4-6.0) | 1.8 (0.6-13.0) | 31 (15-85) |
| 0.9 (n=20) | 1.1 (0.3-3.8) | 1.4 (0.8-6.2) | 58 (27-111) |
| 1.2 (n=18) | 0.7 (0.4-1.7) | 1.0 (0.6-4.7) | 67 (38-160) |
| Geriatric ≥65 yrs |
2.3 (1.0-8.3) |
3.7 (1.3-11.3) |
46 (22-73) |
| 0.6 (n=31) | |||
| 0.9 (n=5) | 2.0 (1.0-3.0) | 2.5 (1.2-5.0) | 62 (49-75) |
| 1.2 (n=7) | 1.0 (0.8-3.5) | 1.3 (1.2-4.7) | 94 (64-138) |
| Infants 3 mo to 1 yr 0.6 (n=17) 0.8 (n=9) |
— — |
0.8 (0.3-3.0) 0.7 (0.5-0.8) |
41 (24-68) 40 (27-70) |
| Pediatric 1 to 12 yrs 0.6 (n=27) 0.8 (n=18) |
0.8 (0.4-2.0) — |
1.0 (0.5-3.3) 0.5 (0.3-1.0) |
26 (17-39) 30 (17-56) |
Table 6 presents the time to onset and clinical duration for the initial dose of Rocuronium Bromide Injection (rocuronium bromide) Injection under sevoflurane (induction) and isoflurane/nitrous oxide (maintenance) anesthesia in pediatric patients.
|
n=the number of patients with the highest number of observations for time to maximum block or reappearance T3. |
||
| rocuronium bromide injection Dose (mg/kg) Administered Over 5 sec | Time to Maximum Block (min) | Time to Reappearance T3 (min) |
| Neonates birth to <28 days |
1.1 (0.6-2.2) |
40.3 (32.5-62.6) |
| 0.45 (n=5) | ||
| 0.6 (n=10) | 1.0 (0.2-2.1) | 49.7 (16.6-119.0) |
| 1 (n=6) | 0.6 (0.3-1.8) | 114.4 (92.6-136.3) |
| Infants 28 days to ≤3 mo |
0.5 (0.4-1.3) |
49.1 (13.5-79.9) |
| 0.45 (n=9) | ||
| 0.6 (n=11) | 0.4 (0.2-0.8) | 59.8 (32.3-87.8) |
| 1 (n=5) | 0.3 (0.2-0.7) | 103.3 (90.8-155.4) |
| Toddlers >3 mo to ≤2 yrs |
0.8 (0.3-1.9) |
39.2 (16.9-59.4) |
| 0.45 (n=17) | ||
| 0.6 (n=29) | 0.6 (0.2-1.6) | 44.2 (18.9-68.8) |
| 1 (n=15) | 0.5 (0.2-1.5) | 72.0 (36.2-128.2) |
| Children >2 yrs to ≤11 yrs |
0.9 (0.4-1.9) |
21.5 (17.5-38.0) |
| 0.45 (n=14) | ||
| 0.6 (n=37) | 0.8 (0.3-1.7) | 36.7 (20.1-65.9) |
| 1 (n=16) | 0.7 (0.4-1.2) | 53.1 (31.2-89.9) |
| Adolescents >11 to ≤17 yrs |
1.0 (0.5-1.7) |
37.5 (18.3-65.7) |
| 0.45 (n=18) | ||
| 0.6 (n=31) | 0.9 (0.2-2.1) | 41.4 (16.3-91.2) |
| 1 (n=14) | 0.7 (0.5-1.2) | 67.1 (25.6-93.8) |
The time to 80% or greater block and clinical duration as a function of dose are presented in
Figures 1
and
2.
Figure 1: Time to 80% or Greater Block vs. Initial Dose of rocuronium bromide injection by Age Group (Median, 25th and 75th Percentile, and Individual Values)
Figure 2: Duration of Clinical Effect vs. Initial Dose of rocuronium bromide injection by Age Group (Median, 25th and 75th Percentile, and Individual Values)
The clinical durations for the first 5 maintenance doses, in patients receiving 5 or more maintenance doses are represented in
Figure 3
[see Dosage and Administration (2.4)].
Figure 3: Duration of Clinical Effect vs. Number of rocuronium bromide injection Maintenance Doses, by Dose
Once spontaneous recovery has reached 25% of control T1, the neuromuscular block produced by rocuronium bromide injection is readily reversed with anticholinesterase agents, e.g., edrophonium or neostigmine.
The median spontaneous recovery from 25% to 75% T1 was 13 minutes in adult patients. When neuromuscular block was reversed in 36 adults at a T1 of 22% to 27%, recovery to a T1 of 89 (50-132)% and T4/T1 of 69 (38-92)% was achieved within 5 minutes. Only 5 of 320 adults reversed received an additional dose of reversal agent. The median (range) dose of neostigmine was 0.04 (0.01-0.09) mg/kg and the median (range) dose of edrophonium was 0.5 (0.3-1.0) mg/kg.
In geriatric patients (n=51) reversed with neostigmine, the median T4/T1 increased from 40% to 88% in 5 minutes.
In clinical trials with halothane, pediatric patients (n=27) who received 0.5 mg/kg edrophonium had increases in the median T4/T1 from 37% at reversal to 93% after 2 minutes. Pediatric patients (n=58) who received 1 mg/kg edrophonium had increases in the median T4/T1 from 72% at reversal to 100% after 2 minutes. Infants (n=10) who were reversed with 0.03 mg/kg neostigmine recovered from 25% to 75% T1 within 4 minutes.
There were no reports of less than satisfactory clinical recovery of neuromuscular function.
The neuromuscular blocking action of Rocuronium Bromide Injection may be enhanced in the presence of potent inhalation anesthetics [see Drug Interactions (7.3)].
7.4 Lithium Carbonate
Lithium has been shown to increase the duration of neuromuscular block and decrease infusion requirements of neuromuscular blocking agents [see Warnings and Precautions (5.10)].
7.5 Local Anesthetics
Local anesthetics have been shown to increase the duration of neuromuscular block and decrease infusion requirements of neuromuscular blocking agents [see Warnings and Precautions (5.10)].
2.4 Maintenance Dosing
Maintenance doses of 0.1, 0.15, and 0.2 mg/kg Rocuronium Bromide Injection, administered at 25% recovery of control T1 (defined as 3 twitches of train-of-four), provide a median (range) of 12 (2-31), 17 (6-50), and 24 (7-69) minutes of clinical duration under opioid/nitrous oxide/oxygen anesthesia [see Clinical Pharmacology (12.2)]. In all cases, dosing should be guided based on the clinical duration following initial dose or prior maintenance dose and not administered until recovery of neuromuscular function is evident. A clinically insignificant cumulation of effect with repetitive maintenance dosing has been observed [see Clinical Pharmacology (12.2)].
5.5 Residual Paralysis
To prevent complications resulting from residual paralysis from Rocuronium Bromide Injection, it is recommended to extubate only after the patient has recovered sufficiently from neuromuscular block. Geriatric patients (65 years or older) may be at increased risk for residual neuromuscular block. Other factors which could cause residual paralysis after extubation in the post- operative phase (such as drug interactions or patient condition) should also be considered. If not used as part of standard clinical practice the use of a reversal agent should be considered, especially in those cases where residual paralysis is more likely to occur.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Rocuronium Bromide Injection is indicated as an adjunct to general anesthesia to facilitate both rapid sequence and routine tracheal intubation, and to provide skeletal muscle relaxation during surgery or mechanical ventilation.
14.2 Geriatric Patients
Rocuronium bromide injection was evaluated in 55 geriatric patients (ages 65-80 years) in 6 clinical studies. Doses of 0.6 mg/kg provided excellent to good intubating conditions in a median (range) time of 2.3 (1-8) minutes. Recovery times from 25% to 75% after these doses were not prolonged in geriatric patients compared to other adult patients [see Dosage and Administration (2.6) and Use in Specific Populations (8.5)].
14.3 Pediatric Patients
Rocuronium bromide injection 0.45, 0.6, or 1 mg/kg was evaluated under sevoflurane (induction) and isoflurane/nitrous oxide (maintenance) anesthesia for intubation in 326 patients in 2 studies. In 1 of these studies maintenance bolus and infusion requirements were evaluated in 137 patients. In all age groups, doses of 0.6 mg/kg provided time to maximum block in about 1 minute. Across all age groups, median (range) time to reappearance of T3 for doses of 0.6 mg/kg was shortest in the children [36.7 (20.1-65.9) minutes] and longest in infants [59.8 (32.3-87.8) minutes]. For pediatric patients older than 3 months, the time to recovery was shorter after stopping infusion maintenance when compared with bolus maintenance [see Dosage and Administration (2.6) and Use in Specific Populations (8.4)].
Rocuronium bromide injection 0.6 or 0.8 mg/kg was evaluated for intubation in 75 pediatric patients (n=28; age 3-12 months, n=47; age 1-12 years) in 3 studies using halothane (1%-5%) and nitrous oxide (60%-70%) in oxygen. Doses of 0.6 mg/kg provided a median (range) time to maximum block of 1 (0.5-3.3) minute(s). This dose provided a median (range) time of clinical relaxation of 41 (24-68) minutes in 3-month to 1-year-old infants and 26 (17-39) minutes in 1- to 12-year-old pediatric patients [see Dosage and Administration (2.6) and Use in Specific Populations (8.4)].
12.1 Mechanism of Action
Rocuronium Bromide Injection is a nondepolarizing neuromuscular blocking agent with a rapid to intermediate onset depending on dose and intermediate duration. It acts by competing for cholinergic receptors at the motor end-plate. This action is antagonized by acetylcholinesterase inhibitors, such as neostigmine and edrophonium.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Appropriate Administration and Monitoring: Use only if facilities for intubation, mechanical ventilation, oxygen therapy, and an antagonist are immediately available. (5.1)
- Anaphylaxis: Severe anaphylaxis has been reported. Consider cross-reactivity among neuromuscular blocking agents. (5.2)
- Risk of Death due to Medication Errors: Accidental administration can cause death. (5.3)
- Need for Adequate Anesthesia: Must be accompanied by adequate anesthesia or sedation. (5.4)
- Residual Paralysis: Consider using a reversal agent in cases where residual paralysis is more likely to occur. (5.5)
5.7 Malignant Hyperthermia
Rocuronium Bromide Injection has not been studied in malignant hyperthermia (MH)-susceptible patients. In an animal study in MH-susceptible swine, the administration of Rocuronium Bromide Injection did not appear to trigger malignant hyperthermia.
Because Rocuronium Bromide Injection is always used with other agents, and the occurrence of MH during anesthesia is possible even in the absence of known triggering agents, clinicians should be familiar with early signs, confirmatory diagnosis, and treatment of MH prior to the start of any anesthetic [see Adverse Reactions (6.2)].
7.3 Inhalation Anesthetics
Use of inhalation anesthetics (enflurane > isoflurane > halothane) has been shown to enhance the activity of other neuromuscular blocking agents. Isoflurane and enflurane may also prolong the duration of action of initial and maintenance doses of Rocuronium Bromide Injection and decrease the average infusion requirement of Rocuronium Bromide Injection by 40% compared to opioid/nitrous oxide/oxygen anesthesia. No definite interaction between rocuronium bromide injection and halothane has been demonstrated. In one study, use of enflurane in 10 patients who received another rocuronium bromide injection product resulted in a 20% increase in mean clinical duration of the initial intubating dose, and a 37% increase in the duration of subsequent maintenance doses, when compared in the same study to 10 patients under opioid/nitrous oxide/oxygen anesthesia. The clinical duration of initial doses of another rocuronium bromide injection product (0.57 to 0.85 mg/kg) under enflurane or isoflurane anesthesia, as used clinically, was increased by 11% and 23%, respectively. The duration of maintenance doses was affected to a greater extent, increasing by 30% to 50% under either enflurane or isoflurane anesthesia.
Potentiation by these agents was also observed with respect to the infusion rates of rocuronium bromide injection required to maintain approximately 95% neuromuscular block. Under isoflurane and enflurane anesthesia, the infusion rates were decreased by approximately 40% compared to opioid/nitrous oxide/oxygen anesthesia. The median spontaneous recovery time (from 25% to 75% of control T1) was not affected by halothane but is prolonged by enflurane (15% longer) and isoflurane (62% longer). Reversal-induced recovery of Rocuronium Bromide Injection neuromuscular block is minimally affected by anesthetic technique [see Dosage and Administration (2.6) and Warnings and Precautions (5.10)].
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Rocuronium Bromide Injection should only be administered by experienced clinicians or trained individuals supervised by an experienced clinician familiar with the use, actions, characteristics, and complications of neuromuscular blocking agents. (2.1)
- Individualize the dose for each patient. (2.1)
- Peripheral nerve stimulator recommended for determination of drug response and need for additional doses, and to evaluate recovery. (2.1)
- Store Rocuronium Bromide Injection with cap and ferrule intact and in a manner that minimizes the possibility of selecting the wrong product. (2.1)
- Tracheal intubation: Recommended initial dose is 0.6 mg/kg. (2.2)
- Rapid sequence intubation: 0.6 to 1.2 mg/kg. (2.3)
- Maintenance doses: Guided by response to prior dose, not administered until recovery is evident. (2.4)
- Continuous infusion: Initial rate of 10 to 12 mcg/kg/min. Start only after early evidence of spontaneous recovery from an intubating dose. (2.5)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Rocuronium Bromide Injection is a clear, colorless to yellow/orange solution, free from visible particulate matter available as:
- 50 mg/5 mL (10 mg/mL), single-dose vials
- 100 mg/10 mL (10 mg per mL), single-dose vials
5.9 Qt Interval Prolongation (5.9 QT Interval Prolongation)
The overall analysis of ECG data in pediatric patients indicates that the concomitant use of Rocuronium Bromide Injection with general anesthetic agents can prolong the QTc interval [see Clinical Studies (14.3)].
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of rocuronium bromide injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune system disorders: In clinical practice, there have been reports of severe allergic reactions (anaphylactic reactions and shock) with rocuronium bromide injection, including some that have been life-threatening and fatal [see Warnings and Precautions (5.2)].
General disorders and administration site conditions: There have been reports of malignant hyperthermia with the use of rocuronium bromide injection [see Warnings and Precautions (5.7)].
2.3 Rapid Sequence Intubation
In appropriately premedicated and adequately anesthetized patients, Rocuronium Bromide Injection 0.6 to 1.2 mg/kg will provide excellent or good intubating conditions in most patients in less than 2 minutes [see Clinical Studies (14.1)].
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
2.5 Use By Continuous Infusion (2.5 Use by Continuous Infusion)
Infusion at an initial rate of 10 to 12 mcg/kg/min of Rocuronium Bromide Injection should be initiated only after early evidence of spontaneous recovery from an intubating dose. Due to rapid redistribution [see Clinical Pharmacology (12.3)] and the associated rapid spontaneous recovery, initiation of the infusion after substantial return of neuromuscular function (more than 10% of controlT1) may necessitate additional bolus doses to maintain adequate block for surgery.
Upon reaching the desired level of neuromuscular block, the infusion of Rocuronium Bromide Injection must be individualized for each patient. The rate of administration should be adjusted according to the patient's twitch response as monitored with the use of a peripheral nerve stimulator. In clinical trials, infusion rates have ranged from 4 to 16 mcg/kg/min.
Inhalation anesthetics, particularly enflurane and isoflurane, may enhance the neuromuscular blocking action of nondepolarizing muscle relaxants. In the presence of steady-state concentrations of enflurane or isoflurane, it may be necessary to reduce the rate of infusion by 30% to 50%, at 45 to 60 minutes after the intubating dose.
Spontaneous recovery and reversal of neuromuscular blockade following discontinuation of Rocuronium Bromide Injection infusion may be expected to proceed at rates comparable to that following comparable total doses administered by repetitive bolus injections [see Clinical Pharmacology (12.2)].
Infusion solutions of Rocuronium Bromide Injection can be prepared by mixing Rocuronium Bromide Injection with an appropriate infusion solution such as 5% glucose in water or lactated Ringers [see Dosage and Administration (2.7)]. These infusion solutions should be used within 24 hours of mixing. Unused portions of infusion solutions should be discarded.
Infusion rates of Rocuronium Bromide Injection can be individualized for each patient using the following tables for 3 different concentrations of Rocuronium Bromide Injection solution as guidelines:
|
* 50 mg Rocuronium Bromide Injection in 100 mL solution. |
|||||||||||
| Patient Weight | Drug Delivery Rate (mcg/kg/min) | ||||||||||
| (kg) | (lbs) | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 12 | 14 | 16 |
| Infusion Delivery Rate (mL/hr) | |||||||||||
| 10 | 22 | 4.8 | 6 | 7.2 | 8.4 | 9.6 | 10.8 | 12 | 14.4 | 16.8 | 19.2 |
| 15 | 33 | 7.2 | 9 | 10.8 | 12.6 | 14.4 | 16.2 | 18 | 21.6 | 25.2 | 28.8 |
| 20 | 44 | 9.6 | 12 | 14.4 | 16.8 | 19.2 | 21.6 | 24 | 28.8 | 33.6 | 38.4 |
| 25 | 55 | 12 | 15 | 18 | 21 | 24 | 27 | 30 | 36 | 42 | 48 |
| 35 | 77 | 16.8 | 21 | 25.2 | 29.4 | 33.6 | 37.8 | 42 | 50.4 | 58.8 | 67.2 |
| 50 | 110 | 24 | 30 | 36 | 42 | 48 | 54 | 60 | 72 | 84 | 96 |
| 60 | 132 | 28.8 | 36 | 43.2 | 50.4 | 57.6 | 64.8 | 72 | 86.4 | 100.8 | 115.2 |
| 70 | 154 | 33.6 | 42 | 50.4 | 58.8 | 67.2 | 75.6 | 84 | 100.8 | 117.6 | 134.4 |
| 80 | 176 | 38.4 | 48 | 57.6 | 67.2 | 76.8 | 86.4 | 96 | 115.2 | 134.4 | 153.6 |
| 90 | 198 | 43.2 | 54 | 64.8 | 75.6 | 86.4 | 97.2 | 108 | 129.6 | 151.2 | 172.8 |
| 100 | 220 | 48 | 60 | 72 | 84 | 96 | 108 | 120 | 144 | 168 | 192 |
|
* 100 mg Rocuronium Bromide Injection in 100 mL solution. |
|||||||||||
| Patient | Weight | Drug Delivery Rate (mcg/kg/min) | |||||||||
| (kg) | (lbs) | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 12 | 14 | 16 |
| Infusion Delivery Rate (mL/hr) | |||||||||||
| 10 | 22 | 2.4 | 3 | 3.6 | 4.2 | 4.8 | 5.4 | 6 | 7.2 | 8.4 | 9.6 |
| 15 | 33 | 3.6 | 4.5 | 5.4 | 6.3 | 7.2 | 8.1 | 9 | 10.8 | 12.6 | 14.4 |
| 20 | 44 | 4.8 | 6 | 7.2 | 8.4 | 9.6 | 10.8 | 12 | 14.4 | 16.8 | 19.2 |
| 25 | 55 | 6 | 7.5 | 9 | 10.5 | 12 | 13.5 | 15 | 18 | 21 | 24 |
| 35 | 77 | 8.4 | 10.5 | 12.6 | 14.7 | 16.8 | 18.9 | 21 | 25.2 | 29.4 | 33.6 |
| 50 | 110 | 12 | 15 | 18 | 21 | 24 | 27 | 30 | 36 | 42 | 48 |
| 60 | 132 | 14.4 | 18 | 21.6 | 25.2 | 28.8 | 32.4 | 36 | 43.2 | 50.4 | 57.6 |
| 70 | 154 | 16.8 | 21 | 25.2 | 29.4 | 33.6 | 37.8 | 42 | 50.4 | 58.8 | 67.2 |
| 80 | 176 | 19.2 | 24 | 28.8 | 33.6 | 38.4 | 43.2 | 48 | 57.6 | 67.2 | 76.8 |
| 90 | 198 | 21.6 | 27 | 32.4 | 37.8 | 43.2 | 48.6 | 54 | 64.8 | 75.6 | 86.4 |
| 100 | 220 | 24 | 30 | 36 | 42 | 48 | 54 | 60 | 72 | 84 | 96 |
|
* 500 mg Rocuronium Bromide Injection in 100 mL solution. |
|||||||||||
| Patient | Weight | Drug Delivery Rate (mcg/kg/min) | |||||||||
| (kg) | (lbs) | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 12 | 14 | 16 |
| Infusion Delivery Rate (mL/hr) | |||||||||||
| 10 | 22 | 0.5 | 0.6 | 0.7 | 0.8 | 1 | 1.1 | 1.2 | 1.4 | 1.7 | 1.9 |
| 15 | 33 | 0.7 | 0.9 | 1.1 | 1.3 | 1.4 | 1.6 | 1 8 | 2.2 | 2.5 | 2.9 |
| 20 | 44 | 1 | 1.2 | 1.4 | 1.7 | 1.9 | 2.2 | 2.4 | 2.9 | 3.4 | 3.8 |
| 25 | 55 | 1.2 | 1.5 | 1.8 | 2.1 | 2.4 | 2.7 | 3 | 3.6 | 4.2 | 4.8 |
| 35 | 77 | 1.7 | 2.1 | 2.5 | 2.9 | 3.4 | 3.8 | 4.2 | 5 | 5.9 | 6.7 |
| 50 | 110 | 2.4 | 3 | 3.6 | 4.2 | 4.8 | 5.4 | 6 | 7.2 | 8.4 | 9.6 |
| 60 | 132 | 2.9 | 3.6 | 4.3 | 5 | 5.8 | 6.5 | 7.2 | 8.6 | 10.1 | 11.5 |
| 70 | 154 | 3.4 | 4.2 | 5 | 5.9 | 6.7 | 7.6 | 8.4 | 10.1 | 11.8 | 13.4 |
| 80 | 176 | 3.8 | 4.8 | 5.8 | 6.7 | 7.7 | 8.6 | 9.6 | 11.5 | 13.4 | 15.4 |
| 90 | 198 | 4.3 | 5.4 | 6.5 | 7.6 | 8.6 | 9.7 | 10.8 | 13 | 15.1 | 17.3 |
| 100 | 220 | 4.8 | 6 | 7.2 | 8.4 | 9.6 | 10.8 | 12 | 14.4 | 16.8 | 19.2 |
5.8 Prolonged Circulation Time
Conditions associated with an increased circulatory delayed time, e.g., cardiovascular disease or advanced age, in patients treated with Rocuronium Bromide Injection may be associated with a delay in onset time [see Dosage and Administration (2.6)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Clinical studies of another rocuronium bromide injection product in the U.S. (n=1137) and Europe (n=1394) totaled 2531 patients. The following adverse reactions were reported in patients administered another rocuronium bromide injection (all events judged by investigators during the clinical trials to have a possible causal relationship):
Adverse reactions in greater than 1% of patients: None
Adverse reactions in less than 1% of patients (probably related or relationship unknown):
-
Cardiovascular: arrhythmia, abnormal electrocardiogram, tachycardia
-
Digestive: nausea, vomiting
-
Respiratory: asthma (bronchospasm, wheezing, or rhonchi), hiccup
-
Skin and Appendages: rash, injection site edema, pruritus
In the European studies of another rocuronium bromide injection product, the most commonly reported reactions were transient hypotension (2%) and hypertension (2%); these are in greater frequency than the US studies (0.1% and 0.1%). Changes in heart rate and blood pressure were defined differently from in the US studies in which changes in cardiovascular parameters were not considered as adverse events unless judged by the investigator as unexpected, clinically significant, or thought to be histamine related.
In a clinical study of another rocuronium bromide injection product in patients with clinically significant cardiovascular disease undergoing coronary artery bypass graft, hypertension and tachycardia were reported in some patients, but these occurrences were less frequent in patients receiving beta or calcium channel-blocking drugs. In some patients, the use of another rocuronium bromide injection product was associated with transient increases (30% or greater) in pulmonary vascular resistance. In another clinical study of patients undergoing abdominal aortic surgery, transient increases (30% or greater) in pulmonary vascular resistance were observed in about 24% of patients who received another rocuronium bromide injection product at 0.6 or 0.9 mg/kg.
In pediatric patient studies worldwide of another rocuronium bromide injection product (n=704), tachycardia occurred at an incidence of 5.3% (n=37), and it was judged by the investigator as related in 10 cases (1.4%).
2.2 Dose for Tracheal Intubation
The recommended initial dose of Rocuronium Bromide Injection, regardless of anesthetic technique, is 0.6 mg/kg. Neuromuscular block sufficient for intubation (80% block or greater) is attained in a median (range) time of 1 (0.4-6) minute(s) and most patients have intubation completed within 2 minutes. Maximum blockade is achieved in most patients in less than 3 minutes. This dose may be expected to provide 31 (15-85) minutes of clinical relaxation under opioid/nitrous oxide/oxygen anesthesia. Under halothane, isoflurane, and enflurane anesthesia, some extension of the period of clinical relaxation should be expected [see Drug Interactions (7.3)].
A lower dose of Rocuronium Bromide Injection (0.45 mg/kg) may be used. Neuromuscular block sufficient for intubation (80% block or greater) is attained in a median (range) time of 1.3 (0.8-6.2) minute(s), and most patients have intubation completed within 2 minutes. Maximum blockade is achieved in most patients in less than 4 minutes. This dose may be expected to provide 22 (12-31) minutes of clinical relaxation under opioid/nitrous oxide/oxygen anesthesia. Patients receiving this low dose of 0.45 mg/kg who achieve less than 90% block (about 16% of these patients) may have a more rapid time to 25% recovery, 12 to 15 minutes.
A large bolus dose of 0.9 or 1.2 mg/kg can be administered under opioid/nitrous oxide/oxygen anesthesia without adverse effects to the cardiovascular system [see Clinical Pharmacology (12.2)].
5.4 Need for Adequate Anesthesia
Rocuronium Bromide Injection has no known effect on consciousness, pain threshold, or cerebration. Therefore, its administration must be accompanied by adequate anesthesia or sedation.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Obtain information about your patient's medical history, current medications, any history of hypersensitivity to rocuronium bromide or other neuromuscular blocking agents. If applicable, inform your patients that certain medical conditions and medications might influence how Rocuronium Bromide Injection works [see Warnings and Precautions (5.1), Drug Interactions (7)].
In addition, inform your patient that severe anaphylactic reactions to neuromuscular blocking agents, including Rocuronium Bromide Injection, have been reported. Because allergic cross-reactivity has been reported in this class, request information from your patients about previous anaphylactic reactions to other neuromuscular blocking agents [see Warnings and Precautions (5.2)].
Manufactured by:
www.fresenius-kabi.com/us
451851
8.7 Patients With Renal Impairment (8.7 Patients with Renal Impairment)
Due to the limited role of the kidney in the excretion of rocuronium bromide, usual dosing recommendations Rocuronium Bromide Injection for should be followed. In patients with renal dysfunction, the duration of neuromuscular blockade was not prolonged; however, there was substantial individual variability (range: 22-90 minutes) [see Clinical Pharmacology (12.3)].
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Rocuronium Bromide Injection is a clear, colorless to yellow/orange solution, free from visible particulate matter and available in single-dose vials in the following packaging configurations:
| Product Code | Unit of Sale | Strength | Each |
| 442105 | NDC 65219-442-05 Unit of 10 |
50 mg per 5 mL (10 mg per mL) |
NDC 65219-442-02 Single-Dose Vial |
| RF442105 | NDC 65219-695-05 Unit of 10 |
50 mg per 5 mL (10 mg per mL) |
NDC 65219-695-01 Single-Dose Vial This product is RFID-enabled. |
| 442110 | NDC 65219-444-10 Unit of 10 |
100 mg per 10 mL (10 mg per mL) |
NDC 65219-444-04 Single-Dose Vial |
| RF442110 | NDC 65219-697-10 Unit of 10 |
100 mg per 10 mL (10 mg per mL) |
NDC 65219-697-01 Single-Dose Vial This product is RFID-enabled. |
The container closure is not made with natural rubber latex.
Rocuronium Bromide Injection should be stored at 20°C to 25°C (68°F to 77°F); Excursion permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature]. DO NOT FREEZE.
5.13 Use in Patients With Myasthenia (5.13 Use in Patients with Myasthenia)
In patients with myasthenia gravis or myasthenic (Eaton-Lambert) syndrome, small doses of nondepolarizing neuromuscular blocking agents, including Rocuronium Bromide Injection, may have profound effects. In such patients, a peripheral nerve stimulator and use of a small test dose may be of value in monitoring the response to administration of muscle relaxants.
7.7 Nondepolarizing Muscle Relaxants
There are no controlled studies documenting the use of Rocuronium Bromide Injection before or after other nondepolarizing muscle relaxants. Interactions have been observed when other nondepolarizing muscle relaxants have been administered in succession.
8.6 Patients With Hepatic Impairment (8.6 Patients with Hepatic Impairment)
Because rocuronium bromide is primarily excreted by the liver, it should be used with caution in patients with clinically significant hepatic impairment. Another rocuronium bromide injection product (0.6 mg/kg) was studied in a limited number of patients (n=9) with clinically significant hepatic impairment under steady-state isoflurane anesthesia. After rocuronium bromide injection administration (0.6 mg/kg), the median (range) clinical duration of 60 (35-166) minutes was moderately prolonged compared to 42 minutes in patients with normal hepatic function. The median recovery time of 53 minutes was also prolonged in patients with cirrhosis compared to 20 minutes in patients with normal hepatic function. Four of 8 patients with cirrhosis, who received the other rocuronium bromide injection product (0.6 mg/kg) under opioid/nitrous oxide/oxygen anesthesia, did not achieve complete block. These findings are consistent with the increase in volume of distribution at steady state observed in patients with significant hepatic impairment [see Clinical Pharmacology (12.3)]. If used for rapid sequence induction in patients with ascites, an increased initial dosage of Rocuronium Bromide Injection may be necessary to assure complete block. Duration will be prolonged in these cases. The use of Rocuronium Bromide Injection doses higher than 0.6 mg/kg has not been studied [see Dosage and Administration (2.6)].
5.3 Risk of Death Due to Medication Errors (5.3 Risk of Death due to Medication Errors)
Administration of Rocuronium Bromide Injection results in paralysis, which may lead to respiratory arrest and death, a progression that may be more likely to occur in a patient for whom it is not intended. Confirm proper selection of intended product and avoid confusion with other injectable solutions that are present in critical care and other clinical settings. If another healthcare provider is administering Rocuronium Bromide Injection, ensure that the intended dose is clearly labeled and communicated.
5.6 Long Term Use in An Intensive Care Unit (5.6 Long-Term Use in an Intensive Care Unit)
Rocuronium Bromide Injection has not been studied for long-term use in the intensive care unit (ICU). As with other nondepolarizing neuromuscular blocking drugs, apparent tolerance to Rocuronium Bromide Injection may develop during chronic administration in the ICU. While the mechanism for development of this resistance is not known, receptor up-regulation may be a contributing factor. It is strongly recommended that neuromuscular transmission be monitored continuously during Rocuronium Bromide Injection administration and recovery with the help of a nerve stimulator. Additional doses of Rocuronium Bromide Injection or any other neuromuscular blocking agent should not be given until there is a definite response (one twitch of the train-of-four) to nerve stimulation. Prolonged paralysis and/or skeletal muscle weakness may be noted during initial attempts to wean from the ventilator patients who have chronically received neuromuscular blocking drugs in the ICU.
Myopathy after long-term administration of other nondepolarizing neuromuscular blocking agents in the ICU alone or in combination with corticosteroid therapy has been reported. Therefore, for patients receiving both neuromuscular blocking agents (including Rocuronium Bromide Injection) and corticosteroids, the period of use of the neuromuscular blocking agent should be limited as much as possible and only used in the setting where in the opinion of the prescribing physician, the specific advantages of the drug outweigh the risk.
5.11 Incompatibility With Alkaline Solutions (5.11 Incompatibility with Alkaline Solutions)
Rocuronium Bromide Injection, which has an acid pH, should not be mixed with alkaline solutions (e.g., barbiturate solutions) in the same syringe or administered simultaneously during intravenous infusion through the same needle.
5.1 Appropriate Administration and Monitoring
Rocuronium Bromide Injection should be administered in carefully adjusted dosages by or under the supervision of experienced clinicians who are familiar with the drug's actions and the possible complications of its use. Rocuronium Bromide Injection should not be administered unless facilities for intubation, mechanical ventilation, oxygen therapy, and an antagonist are immediately available. It is recommended that clinicians administering neuromuscular blocking agents such as Rocuronium Bromide Injection employ a peripheral nerve stimulator to monitor drug effect, need for additional doses, adequacy of spontaneous recovery or antagonism, and to decrease the complications of overdosage if additional doses are administered.
5.12 Increase in Pulmonary Vascular Resistance
Rocuronium Bromide Injection may be associated with increased pulmonary vascular resistance, so caution is appropriate in patients with pulmonary hypertension or valvular heart disease [see Clinical Studies (14.1)].
2.1 Important Dosing and Administration Information
Rocuronium Bromide Injection is for intravenous use only.
Rocuronium Bromide Injection should only be administered by experienced clinicians or trained individuals supervised by an experienced clinician familiar with the use, actions, characteristics, and complications of neuromuscular blocking agents. Doses of Rocuronium Bromide Injection should be individualized and a peripheral nerve stimulator should be used to monitor drug effect, need for additional doses, adequacy of spontaneous recovery or antagonism, and to decrease the complications of overdosage if additional doses are administered.
The dosage information which follows is derived from studies based upon units of drug per unit of body weight. It is intended to serve as an initial guide to clinicians familiar with other neuromuscular blocking agents to acquire experience with Rocuronium Bromide Injection.
In patients in whom potentiation of, or resistance to, neuromuscular block is anticipated, a dose adjustment should be considered [see Dosage and Administration (2.6), Warnings and Precautions (5.10, 5.13), Drug Interactions (7.2, 7.3, 7.4, 7.5, 7.6, 7.8, 7.10), Use in Specific Populations (8.6)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies in animals have not been performed with rocuronium bromide to evaluate carcinogenic potential or impairment of fertility. Mutagenicity studies (Ames test, analysis of chromosomal aberrations in mammalian cells, and micronucleus test) conducted with rocuronium bromide did not suggest mutagenic potential.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:27.774336 · Updated: 2026-03-14T22:29:58.342833