Headache 4798l
994a037b-7165-3a1e-e053-2a95a90ad7ff
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
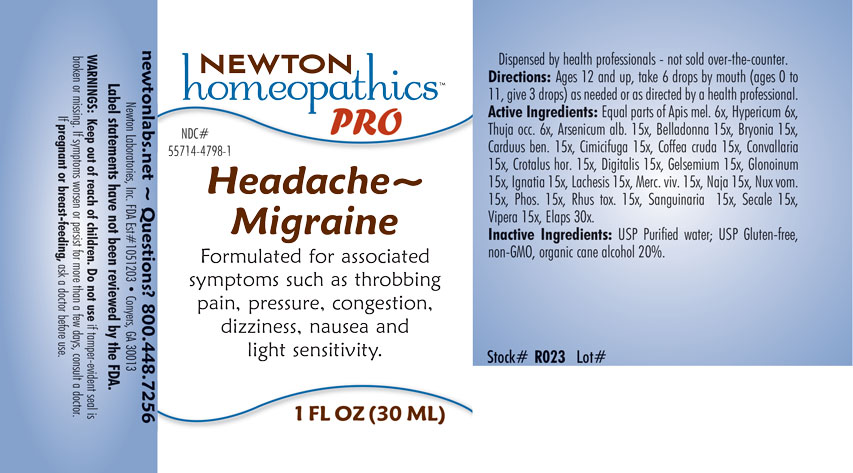
Formulated for associated symptoms such as throbbing pain, pressure, congestion, dizziness, nausea and light sensitivity.
Purpose
Formulated for associated symptoms such as throbbing pain, pressure, congestion, dizziness, nausea and light sensitivity.
Medication Information
Purpose
Formulated for associated symptoms such as throbbing pain, pressure, congestion, dizziness, nausea and light sensitivity.
Description
Formulated for associated symptoms such as throbbing pain, pressure, congestion, dizziness, nausea and light sensitivity.
Package Label
Warnings Section
WARNINGS: Keep out of reach of children. Do not use if tamper-evident seal is broken or missing. If symptoms worsen or persist for more than a few days, consult a doctor. If pregnant or breast-feeding, ask a doctor before use.
Questions Section
newtonlabs.net – Questions? 800.448.7256
Newton Laboratories, Inc. FDA Est # 1051203 - Conyers, GA 30013
Otc Purpose Section
Formulated for associated symptoms such as throbbing pain, pressure, congestion, dizziness, nausea and light sensitivity.
Inactive Ingredient Section
USP Purified water. USP Gluten-free, non-GMO, organic cane alcohol 20%.
Indications & Usage Section
Formulated for associated symptoms such as throbbing pain, pressure, congestion, dizziness, nausea and light sensitivity.
Dosage & Administration Section
Directions: Ages 12 and up, take 6 drops by mouth (ages 0 to 11, give 3 drops) as needed or as directed by a health professional.
Otc Active Ingredient Section
Equal parts of Apis mellifica 6x, Hypericum perforatum 6x, Thuja occidentalis 6x, Arsenicum album 15x, Belladonna 15x, Bryonia 15x, Carduus benedictus 15x, Cimicifuga racemosa 15x, Coffea cruda 15x, Convallaria majalis 15x, Crotalus horridus 15x, Digitalis purpurea 15x, Gelsemium sempervirens 15x, Glonoinum 15x, Ignatia amara 15x, Lachesis mutus 15x, Mercurius vivus 15x, Naja tripudians 15x, Nux vomica 15x, Phosphorus 15x, Rhus toxicodendron 15x, Sanguinaria canadensis 15x, Secale cornutum 15x, Vipera berus 15x, Elaps corallinus 30x.
Otc Pregnancy Or Breast Feeding Section
If pregnant or breast-feeding, ask a doctor before use.
Otc Keep Out of Reach of Children Section
Keep out of reach of children
Structured Label Content
Package Label (PACKAGE LABEL)
Warnings Section (WARNINGS SECTION)
WARNINGS: Keep out of reach of children. Do not use if tamper-evident seal is broken or missing. If symptoms worsen or persist for more than a few days, consult a doctor. If pregnant or breast-feeding, ask a doctor before use.
Questions Section (QUESTIONS SECTION)
newtonlabs.net – Questions? 800.448.7256
Newton Laboratories, Inc. FDA Est # 1051203 - Conyers, GA 30013
Otc Purpose Section (OTC - PURPOSE SECTION)
Formulated for associated symptoms such as throbbing pain, pressure, congestion, dizziness, nausea and light sensitivity.
Inactive Ingredient Section (INACTIVE INGREDIENT SECTION)
USP Purified water. USP Gluten-free, non-GMO, organic cane alcohol 20%.
Indications & Usage Section (INDICATIONS & USAGE SECTION)
Formulated for associated symptoms such as throbbing pain, pressure, congestion, dizziness, nausea and light sensitivity.
Dosage & Administration Section (DOSAGE & ADMINISTRATION SECTION)
Directions: Ages 12 and up, take 6 drops by mouth (ages 0 to 11, give 3 drops) as needed or as directed by a health professional.
Otc Active Ingredient Section (OTC - ACTIVE INGREDIENT SECTION)
Equal parts of Apis mellifica 6x, Hypericum perforatum 6x, Thuja occidentalis 6x, Arsenicum album 15x, Belladonna 15x, Bryonia 15x, Carduus benedictus 15x, Cimicifuga racemosa 15x, Coffea cruda 15x, Convallaria majalis 15x, Crotalus horridus 15x, Digitalis purpurea 15x, Gelsemium sempervirens 15x, Glonoinum 15x, Ignatia amara 15x, Lachesis mutus 15x, Mercurius vivus 15x, Naja tripudians 15x, Nux vomica 15x, Phosphorus 15x, Rhus toxicodendron 15x, Sanguinaria canadensis 15x, Secale cornutum 15x, Vipera berus 15x, Elaps corallinus 30x.
Otc Pregnancy Or Breast Feeding Section (OTC - PREGNANCY OR BREAST FEEDING SECTION)
If pregnant or breast-feeding, ask a doctor before use.
Otc Keep Out of Reach of Children Section (OTC - KEEP OUT OF REACH OF CHILDREN SECTION)
Keep out of reach of children
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:09:56.607123 · Updated: 2026-03-14T23:15:01.065113