Skin Tag Relief
98fb9d4a-d0d2-425b-878b-927eebf53067
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Cutaneous Skin Tags Relief
Medication Information
Purpose
Cutaneous Skin Tags Relief
Description
Drug Facts
Uses
Symptomatic treatment of Skin Tags.
Section 42229-5
Drug Facts
Section 50565-1
Keep out of the reach of children. In case of ingestion call Poison Control Center hotline immediately at 1-800-222-1222.
Section 50567-7
Avoid using near the eyes or mouth.
Section 50570-1
Do not use if tamper evident seal is broken.
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Warnings
For external use only.
Directions
Apply 3 times daily to affected area. Skin tags will dry and flake away over several week period. Some individuals may be sensitive to essential oil. Skin test for tolerability. If irritation or reaction occurs, discontinue use.
Other Ingredients
Cedar Leaf Oil, Melaleuca Alternifollia Leaf Oil (Tea Tree Oil), Ricinus Communis Seed Oil (Castor Oil).
Active Ingredients
Thuja Occidentalis 6X (HPUS) 1%
The letters HPUS indicate the component(s) in this product is (are) officially monographed in the Homeopathic Pharmacopeia of the United States.
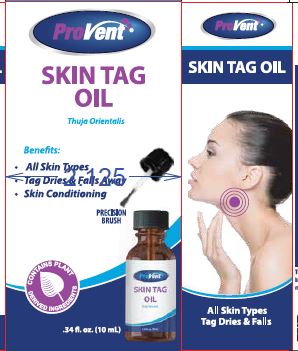
Principal Display Panel 10 Ml Bottle Carton
Structured Label Content
Uses
Symptomatic treatment of Skin Tags.
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of the reach of children. In case of ingestion call Poison Control Center hotline immediately at 1-800-222-1222.
Section 50567-7 (50567-7)
Avoid using near the eyes or mouth.
Section 50570-1 (50570-1)
Do not use if tamper evident seal is broken.
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Purpose
Cutaneous Skin Tags Relief
Warnings
For external use only.
Directions
Apply 3 times daily to affected area. Skin tags will dry and flake away over several week period. Some individuals may be sensitive to essential oil. Skin test for tolerability. If irritation or reaction occurs, discontinue use.
Other Ingredients (Other ingredients)
Cedar Leaf Oil, Melaleuca Alternifollia Leaf Oil (Tea Tree Oil), Ricinus Communis Seed Oil (Castor Oil).
Active Ingredients
Thuja Occidentalis 6X (HPUS) 1%
The letters HPUS indicate the component(s) in this product is (are) officially monographed in the Homeopathic Pharmacopeia of the United States.
Principal Display Panel 10 Ml Bottle Carton (PRINCIPAL DISPLAY PANEL - 10 mL Bottle Carton)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:11:36.322331 · Updated: 2026-03-14T23:17:01.647677