These Highlights Do Not Include All The Information Needed To Use Insulin Glargine Safely And Effectively. See Full Prescribing Information For Insulin Glargine.
9835904f-64f5-42b3-9ac9-89e4aaa5182a
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Insulin glargine is indicated to improve glycemic control in adults and pediatric patients with type 1 diabetes mellitus and in adults with type 2 diabetes mellitus.
Indications and Usage
Insulin glargine is indicated to improve glycemic control in adults and pediatric patients with type 1 diabetes mellitus and in adults with type 2 diabetes mellitus.
Dosage and Administration
Individualize dosage based on metabolic needs, blood glucose monitoring, glycemic control, type of diabetes, and prior insulin use. ( 2.1 , 2.3 , 2.4 ) Administer subcutaneously into the abdominal area, thigh, or deltoid once daily at any time of day, but at the same time every day. ( 2.1 ) Do not dilute or mix with any other insulin or solution. ( 2.1 ) Rotate injection sites to reduce risk of lipodystrophy and localized cutaneous amyloidosis. ( 2.2 ) Closely monitor glucose when changing to Insulin glargine and during initial weeks thereafter. ( 2.4 )
Warnings and Precautions
Never share an Insulin glargine SoloStar prefilled pen between patients, even if the needle is changed. ( 5.1 ) Hyperglycemia or hypoglycemia with changes in insulin regimen : Make changes to a patient's insulin regimen (e.g., insulin strength, manufacturer, type, injection site or method of administration) under close medical supervision with increased frequency of blood glucose monitoring. ( 5.2 ) Hypoglycemia : May be life-threatening. Increase frequency of glucose monitoring with changes to: insulin dosage, coadministered glucose lowering medications, meal pattern, physical activity; and in patients with renal or hepatic impairment and hypoglycemia unawareness. ( 5.3 , 6.1 ) Medication Errors : Accidental mix-ups between insulin products can occur. Instruct patients to check insulin labels before injection. ( 5.4 , 6.3 ) Hypersensitivity reactions : Severe, life-threatening, generalized allergy, including anaphylaxis, can occur. Discontinue Insulin glargine. Monitor and treat if indicated. ( 5.5 , 6.1 ) Hypokalemia : May be life-threatening. Monitor potassium levels in patients at risk of hypokalemia and treat if indicated. ( 5.6 ) Fluid retention and heart failure with concomitant use of thiazolidinediones (TZDs) : Observe for signs and symptoms of heart failure; consider dosage reduction or discontinuation of TZD if heart failure occurs. ( 5.7 )
Contraindications
Insulin glargine is contraindicated: during episodes of hypoglycemia [see Warnings and Precautions (5.3) ] in patients with hypersensitivity to Insulin glargine or one of its excipients [see Warnings and Precautions (5.5) ]
Adverse Reactions
The following adverse reactions are discussed elsewhere: Hypoglycemia [see Warnings and Precautions (5.3) ] Hypersensitivity and allergic reactions [see Warnings and Precautions (5.5) ] Hypokalemia [see Warnings and Precautions (5.6) ]
Drug Interactions
Table 8 includes clinically significant drug interactions with Insulin glargine. Table 8: Clinically Significant Drug Interactions with Insulin Glargine Drugs that May Increase the Risk of Hypoglycemia Drugs : Antidiabetic agents, ACE inhibitors, angiotensin II receptor blocking agents, disopyramide, fibrates, fluoxetine, monoamine oxidase inhibitors, pentoxifylline, pramlintide, salicylates, somatostatin analogs (e.g., octreotide), and sulfonamide antibiotics. Intervention : Dose reductions and increased frequency of glucose monitoring may be required when Insulin glargine is coadministered with these drugs. Drugs that May Decrease the Blood Glucose Lowering Effect of Insulin Glargine Drugs : Atypical antipsychotics (e.g., olanzapine and clozapine), corticosteroids, danazol, diuretics, estrogens, glucagon, isoniazid, niacin, oral contraceptives, phenothiazines, progestogens (e.g., in oral contraceptives), protease inhibitors, somatropin, sympathomimetic agents (e.g., albuterol, epinephrine, terbutaline), and thyroid hormones. Intervention : Dose increases and increased frequency of glucose monitoring may be required when Insulin glargine is coadministered with these drugs. Drugs that May Increase or Decrease the Blood Glucose Lowering Effect of Insulin Glargine Drugs : Alcohol, beta-blockers, clonidine, and lithium salts. Pentamidine may cause hypoglycemia, which may sometimes be followed by hyperglycemia. Intervention : Dose adjustment and increased frequency of glucose monitoring may be required when Insulin glargine is coadministered with these drugs. Drugs that May Blunt Signs and Symptoms of Hypoglycemia Drugs : Beta-blockers, clonidine, guanethidine, and reserpine. Intervention : Increased frequency of glucose monitoring may be required when Insulin glargine is coadministered with these drugs.
Storage and Handling
Product: 50090-6178 NDC: 50090-6178-0 3 mL in a SYRINGE / 5 in a CARTON
How Supplied
Product: 50090-6178 NDC: 50090-6178-0 3 mL in a SYRINGE / 5 in a CARTON
Medication Information
Warnings and Precautions
Never share an Insulin glargine SoloStar prefilled pen between patients, even if the needle is changed. ( 5.1 ) Hyperglycemia or hypoglycemia with changes in insulin regimen : Make changes to a patient's insulin regimen (e.g., insulin strength, manufacturer, type, injection site or method of administration) under close medical supervision with increased frequency of blood glucose monitoring. ( 5.2 ) Hypoglycemia : May be life-threatening. Increase frequency of glucose monitoring with changes to: insulin dosage, coadministered glucose lowering medications, meal pattern, physical activity; and in patients with renal or hepatic impairment and hypoglycemia unawareness. ( 5.3 , 6.1 ) Medication Errors : Accidental mix-ups between insulin products can occur. Instruct patients to check insulin labels before injection. ( 5.4 , 6.3 ) Hypersensitivity reactions : Severe, life-threatening, generalized allergy, including anaphylaxis, can occur. Discontinue Insulin glargine. Monitor and treat if indicated. ( 5.5 , 6.1 ) Hypokalemia : May be life-threatening. Monitor potassium levels in patients at risk of hypokalemia and treat if indicated. ( 5.6 ) Fluid retention and heart failure with concomitant use of thiazolidinediones (TZDs) : Observe for signs and symptoms of heart failure; consider dosage reduction or discontinuation of TZD if heart failure occurs. ( 5.7 )
Indications and Usage
Insulin glargine is indicated to improve glycemic control in adults and pediatric patients with type 1 diabetes mellitus and in adults with type 2 diabetes mellitus.
Dosage and Administration
Individualize dosage based on metabolic needs, blood glucose monitoring, glycemic control, type of diabetes, and prior insulin use. ( 2.1 , 2.3 , 2.4 ) Administer subcutaneously into the abdominal area, thigh, or deltoid once daily at any time of day, but at the same time every day. ( 2.1 ) Do not dilute or mix with any other insulin or solution. ( 2.1 ) Rotate injection sites to reduce risk of lipodystrophy and localized cutaneous amyloidosis. ( 2.2 ) Closely monitor glucose when changing to Insulin glargine and during initial weeks thereafter. ( 2.4 )
Contraindications
Insulin glargine is contraindicated: during episodes of hypoglycemia [see Warnings and Precautions (5.3) ] in patients with hypersensitivity to Insulin glargine or one of its excipients [see Warnings and Precautions (5.5) ]
Adverse Reactions
The following adverse reactions are discussed elsewhere: Hypoglycemia [see Warnings and Precautions (5.3) ] Hypersensitivity and allergic reactions [see Warnings and Precautions (5.5) ] Hypokalemia [see Warnings and Precautions (5.6) ]
Drug Interactions
Table 8 includes clinically significant drug interactions with Insulin glargine. Table 8: Clinically Significant Drug Interactions with Insulin Glargine Drugs that May Increase the Risk of Hypoglycemia Drugs : Antidiabetic agents, ACE inhibitors, angiotensin II receptor blocking agents, disopyramide, fibrates, fluoxetine, monoamine oxidase inhibitors, pentoxifylline, pramlintide, salicylates, somatostatin analogs (e.g., octreotide), and sulfonamide antibiotics. Intervention : Dose reductions and increased frequency of glucose monitoring may be required when Insulin glargine is coadministered with these drugs. Drugs that May Decrease the Blood Glucose Lowering Effect of Insulin Glargine Drugs : Atypical antipsychotics (e.g., olanzapine and clozapine), corticosteroids, danazol, diuretics, estrogens, glucagon, isoniazid, niacin, oral contraceptives, phenothiazines, progestogens (e.g., in oral contraceptives), protease inhibitors, somatropin, sympathomimetic agents (e.g., albuterol, epinephrine, terbutaline), and thyroid hormones. Intervention : Dose increases and increased frequency of glucose monitoring may be required when Insulin glargine is coadministered with these drugs. Drugs that May Increase or Decrease the Blood Glucose Lowering Effect of Insulin Glargine Drugs : Alcohol, beta-blockers, clonidine, and lithium salts. Pentamidine may cause hypoglycemia, which may sometimes be followed by hyperglycemia. Intervention : Dose adjustment and increased frequency of glucose monitoring may be required when Insulin glargine is coadministered with these drugs. Drugs that May Blunt Signs and Symptoms of Hypoglycemia Drugs : Beta-blockers, clonidine, guanethidine, and reserpine. Intervention : Increased frequency of glucose monitoring may be required when Insulin glargine is coadministered with these drugs.
Storage and Handling
Product: 50090-6178 NDC: 50090-6178-0 3 mL in a SYRINGE / 5 in a CARTON
How Supplied
Product: 50090-6178 NDC: 50090-6178-0 3 mL in a SYRINGE / 5 in a CARTON
Description
Insulin glargine is indicated to improve glycemic control in adults and pediatric patients with type 1 diabetes mellitus and in adults with type 2 diabetes mellitus.
Section 42229-5
Limitations of Use
Insulin glargine is not recommended for the treatment of diabetic ketoacidosis.
Section 42230-3
| Patient Information Insulin glargine (IN-su-lin-GLAR-gine) (insulin glargine injection) for subcutaneous use, 100 Units/mL (U-100) |
|
|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration | Revised: December 2020 |
|
Do not share your syringes with other people, even if the needle has been changed. You may give other people a serious infection, or get a serious infection from them. What is Insulin glargine? Insulin glargine is a long-acting man-made-insulin used to control high blood sugar in adults with diabetes mellitus.
|
|
|
Who should not use Insulin glargine? Do not use Insulin glargine if you:
|
|
|
What should I tell my healthcare provider before using Insulin glargine? Before using Insulin glargine, tell your healthcare provider about all your medical conditions including if you:
Before you start using Insulin glargine, talk to your healthcare provider about low blood sugar and how to manage it. |
|
How should I use Insulin glargine?
|
|
Your dose of Insulin glargine may need to change because of:
|
|
|
What should I avoid while using Insulin glargine? While using Insulin glargine do not:
|
|
|
What are the possible side effects of Insulin glargine and other insulins? Insulin glargine may cause serious side effects that can lead to death, including:
|
|
|
General information about the safe and effective use of Insulin glargine.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Insulin glargine for a condition for which it was not prescribed. Do not give Insulin glargine to other people, even if they have the same symptoms that you have. It may harm them. This Patient Information leaflet summarizes the most important information about Insulin glargine. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about Insulin glargine that is written for healthcare professionals. For more information, go to www.winthropus.com or call 1-800-633-1610. |
|
What are the ingredients in Insulin glargine?
|
|
| Manufactured by: Winthrop U.S., a business of sanofi-aventis U.S. LLC, Bridgewater, NJ 08807 |
Section 59845-8
| Patient Information Insulin glargine (IN-su-lin-GLAR-gine) (insulin glargine injection) for subcutaneous use, 100 Units/mL (U-100) |
|
|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: December 2020 |
|
Do not share your Insulin glargine SoloStar® pen with other people, even if the needle has been changed. You may give other people a serious infection, or get a serious infection from them. What is Insulin glargine? Insulin glargine is a long-acting man-made insulin used to control high blood sugar in adults with diabetes mellitus.
|
|
|
Who should not use Insulin glargine? Do not use Insulin glargine if you:
|
|
|
What should I tell my healthcare provider before using Insulin glargine? Before using Insulin glargine, tell your healthcare provider about all your medical conditions including if you:
Before you start using Insulin glargine, talk to your healthcare provider about low blood sugar and how to manage it. |
|
How should I use Insulin glargine?
|
|
Your dose of Insulin glargine may need to change because of:
|
|
|
What should I avoid while using Insulin glargine? While using Insulin glargine do not:
|
|
|
What are the possible side effects of Insulin glargine and other insulins? Insulin glargine may cause serious side effects that can lead to death, including:
|
|
|
General information about the safe and effective use of Insulin glargine.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Insulin glargine for a condition for which it was not prescribed. Do not give Insulin glargine to other people, even if they have the same symptoms that you have. It may harm them. This Patient Information leaflet summarizes the most important information about Insulin glargine. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about Insulin glargine that is written for healthcare professionals. For more information about Insulin glargine call 1-800-633-1610 or go to the website www.winthropus.com. |
|
What are the ingredients in Insulin glargine?
|
|
| Manufactured by: Winthrop U.S., a business of sanofi-aventis U.S., LLC Bridgewater, NJ 08807 |
8.8 Obesity
In controlled clinical trials, subgroup analyses based on BMI did not show differences in safety and efficacy between Insulin glargine and NPH.
10 Overdosage
Excess insulin administration may cause hypoglycemia and hypokalemia [see Warnings and Precautions (5.3, 5.6)]. Mild episodes of hypoglycemia can usually be treated with oral carbohydrates. Adjustments in drug dosage, meal patterns, or exercise may be needed.
More severe episodes of hypoglycemia with coma, seizure, or neurologic impairment may be treated with intramuscular/subcutaneous glucagon or concentrated intravenous glucose. After apparent clinical recovery from hypoglycemia, continued observation and additional carbohydrate intake may be necessary to avoid recurrence of hypoglycemia. Hypokalemia must be corrected appropriately.
11 Description
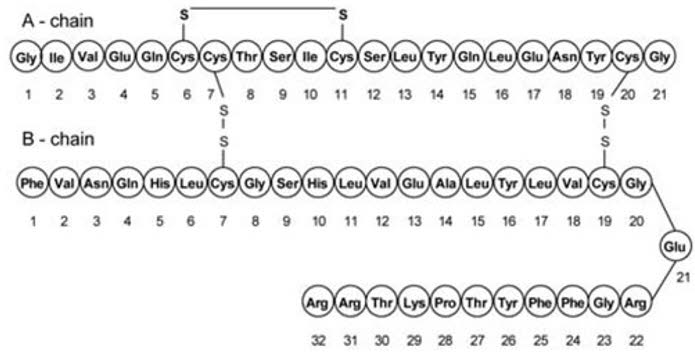
Insulin glargine (insulin glargine injection) is a sterile solution of insulin glargine for subcutaneous use. Insulin glargine is a recombinant human insulin analog that is a long-acting, parenteral blood-glucose-lowering agent [see Clinical Pharmacology (12)]. Insulin glargine has low aqueous solubility at neutral pH. At pH 4 insulin glargine is completely soluble. After injection into the subcutaneous tissue, the acidic solution is neutralized, leading to formation of microprecipitates from which small amounts of insulin glargine are slowly released, resulting in a relatively constant concentration/time profile over 24 hours with no pronounced peak. This profile allows once-daily dosing as a basal insulin. Insulin glargine is produced by recombinant DNA technology utilizing a non-pathogenic laboratory strain of Escherichia coli (K12) as the production organism. Insulin glargine differs from human insulin in that the amino acid asparagine at position A21 is replaced by glycine and two arginines are added to the C-terminus of the B-chain. Chemically, insulin glargine is 21A-Gly-30Ba-L-Arg-30Bb-L-Arg-human insulin and has the empirical formula C267H404N72O78S6 and a molecular weight of 6063. Insulin glargine has the following structural formula:
Insulin glargine consists of insulin glargine dissolved in a clear aqueous fluid. Each milliliter of insulin glargine injection contains 100 units (3.6378 mg) insulin glargine.
The 10 mL vial presentation contains the following inactive ingredients per mL: 30 mcg zinc, 2.7 mg m-cresol, 20 mg glycerol 85%, 20 mcg polysorbate 20, and water for injection.
The 3 mL prefilled pen presentation contains the following inactive ingredients per mL: 30 mcg zinc, 2.7 mg m-cresol, 20 mg glycerol 85%, and water for injection.
The pH is adjusted by addition of aqueous solutions of hydrochloric acid and sodium hydroxide. Insulin glargine has a pH of approximately 4.
5.6 Hypokalemia
All insulin products, including Insulin glargine, cause a shift in potassium from the extracellular to intracellular space, possibly leading to hypokalemia. Untreated hypokalemia may cause respiratory paralysis, ventricular arrhythmia, and death. Monitor potassium levels in patients at risk for hypokalemia, if indicated (e.g., patients using potassium-lowering medications, patients taking medications sensitive to serum potassium concentrations).
5.3 Hypoglycemia
Hypoglycemia is the most common adverse reaction associated with insulin, including Insulin glargine. Severe hypoglycemia can cause seizures, may be life-threatening or cause death. Hypoglycemia can impair concentration ability and reaction time; this may place an individual and others at risk in situations where these abilities are important (e.g., driving or operating other machinery).
Hypoglycemia can happen suddenly and symptoms may differ in each individual and change over time in the same individual. Symptomatic awareness of hypoglycemia may be less pronounced in patients with longstanding diabetes, in patients with diabetic nerve disease, in patients using medications that block the sympathetic nervous system (e.g., beta-blockers) [see Drug Interactions (7)], or in patients who experience recurrent hypoglycemia.
Insulin Glargine
8.4 Pediatric Use
The safety and effectiveness of Insulin glargine have been established in pediatric patients (age 6 to 15 years) with type 1 diabetes [see Clinical Studies (14.2)]. The safety and effectiveness of Insulin glargine in pediatric patients younger than 6 years of age with type 1 diabetes and pediatric patients with type 2 diabetes have not been established.
The dosage recommendation when changing to Insulin glargine in pediatric patients (age 6 to 15 years) with type 1 diabetes is the same as that described for adults [see Dosage and Administration (2.2, 2.4) , Clinical Studies (14)]. As in adults, the dosage of Insulin glargine must be individualized in pediatric patients (age 6 to 15 years) with type 1 diabetes based on metabolic needs and frequent monitoring of blood glucose.
In the pediatric clinical trial, pediatric patients (age 6 to 15 years) with type 1 diabetes had a higher incidence of severe symptomatic hypoglycemia compared to the adults in trials with type 1 diabetes [see Adverse Reactions (6.1)].
8.5 Geriatric Use
Of the total number of subjects in controlled clinical studies of patients with type 1 and type 2 diabetes who were treated with Insulin glargine, 15% were ≥65 years of age and 2% were ≥75 years of age. The only difference in safety or effectiveness in the subpopulation of patients ≥65 years of age compared to the entire study population was a higher incidence of cardiovascular events typically seen in an older population in the Insulin glargine and NPH treatment groups.
Nevertheless, caution should be exercised when Insulin glargine is administered to geriatric patients. In elderly patients with diabetes, the initial dosing, dose increments, and maintenance dosage should be conservative to avoid hypoglycemic reactions. Hypoglycemia may be difficult to recognize in the elderly.
6.2 Immunogenicity
As with all therapeutic proteins, there is potential for immunogenicity. All insulin products can elicit the formation of insulin antibodies. The presence of such insulin antibodies may increase or decrease the efficacy of insulin and may require adjustment of the insulin dose. In phase 3 clinical trials of Insulin glargine, increases in titers of antibodies to insulin were observed in NPH insulin and Insulin glargine treatment groups with similar incidences.
4 Contraindications
Insulin glargine is contraindicated:
- during episodes of hypoglycemia [see Warnings and Precautions (5.3)]
- in patients with hypersensitivity to Insulin glargine or one of its excipients [see Warnings and Precautions (5.5)]
6 Adverse Reactions
The following adverse reactions are discussed elsewhere:
- Hypoglycemia [see Warnings and Precautions (5.3)]
- Hypersensitivity and allergic reactions [see Warnings and Precautions (5.5)]
- Hypokalemia [see Warnings and Precautions (5.6)]
7 Drug Interactions
Table 8 includes clinically significant drug interactions with Insulin glargine.
| Drugs that May Increase the Risk of Hypoglycemia | |
| Drugs: | Antidiabetic agents, ACE inhibitors, angiotensin II receptor blocking agents, disopyramide, fibrates, fluoxetine, monoamine oxidase inhibitors, pentoxifylline, pramlintide, salicylates, somatostatin analogs (e.g., octreotide), and sulfonamide antibiotics. |
| Intervention: | Dose reductions and increased frequency of glucose monitoring may be required when Insulin glargine is coadministered with these drugs. |
| Drugs that May Decrease the Blood Glucose Lowering Effect of Insulin Glargine | |
| Drugs: | Atypical antipsychotics (e.g., olanzapine and clozapine), corticosteroids, danazol, diuretics, estrogens, glucagon, isoniazid, niacin, oral contraceptives, phenothiazines, progestogens (e.g., in oral contraceptives), protease inhibitors, somatropin, sympathomimetic agents (e.g., albuterol, epinephrine, terbutaline), and thyroid hormones. |
| Intervention: | Dose increases and increased frequency of glucose monitoring may be required when Insulin glargine is coadministered with these drugs. |
| Drugs that May Increase or Decrease the Blood Glucose Lowering Effect of Insulin Glargine | |
| Drugs: | Alcohol, beta-blockers, clonidine, and lithium salts. Pentamidine may cause hypoglycemia, which may sometimes be followed by hyperglycemia. |
| Intervention: | Dose adjustment and increased frequency of glucose monitoring may be required when Insulin glargine is coadministered with these drugs. |
| Drugs that May Blunt Signs and Symptoms of Hypoglycemia | |
| Drugs: | Beta-blockers, clonidine, guanethidine, and reserpine. |
| Intervention: | Increased frequency of glucose monitoring may be required when Insulin glargine is coadministered with these drugs. |
8.7 Renal Impairment
The effect of renal impairment on the pharmacokinetics of Insulin glargine has not been studied. Some studies with human insulin have shown increased circulating levels of insulin in patients with renal failure. Frequent glucose monitoring and dose adjustment may be necessary for Insulin glargine in patients with renal impairment [see Warnings and Precautions (5.3)].
Instructions for Use
Insulin glargine (IN-su-lin-GLAR-gine)
injection for subcutaneous use
10 mL Vial (100 Units/mL, U-100)
Read the Instructions for Use before you start taking Insulin glargine and each time you get a new Insulin glargine vial. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
Do not share your Insulin glargine syringes with other people even if the needle has been changed. You may give other people a serious infection, or get a serious infection from them.
Supplies needed to give your injection:
- an Insulin glargine 10 mL vial
- a U-100 insulin syringe and needle
- 2 alcohol swabs
- 1 sharps container for throwing away used needles and syringes. See "Disposing of used needles and syringes" at the end of these instructions.
Preparing your Insulin glargine dose:
- Wash your hands with soap and water or with alcohol.
- Check the Insulin glargine label to make sure you are taking the right type of insulin. This is especially important if you use more than 1 type of insulin.
- Check the insulin to make sure it is clear and colorless. Do not use Insulin glargine if it is colored or cloudy, or if you see particles in the solution.
- Do not use Insulin glargine after the expiration date stamped on the label or 28 days after you first use it.
- Always use a syringe that is marked for U-100 insulin. If you use a syringe other than a U-100 insulin syringe, you may get the wrong dose of insulin.
- Always use a new syringe or needle for each injection. Do not re-use or share your syringes or needles with other people. You may give other people a serious infection or get a serious infection from them.
Step 1:
If you are using a new vial, remove the protective cap. Do not remove the stopper.
Step 2:
Wipe the top of the vial with an alcohol swab. You do not have to shake the vial of Insulin glargine before use.
Step 3:
Draw air into the syringe equal to your insulin dose. Put the needle through the rubber top of the vial and push the plunger to inject the air into the vial.
Step 4:
Leave the syringe in the vial and turn both upside down. Hold the syringe and vial firmly in one hand. Make sure the tip of the needle is in the insulin. With your free hand, pull the plunger to withdraw the correct dose into the syringe.
Step 5:
Before you take the needle out of the vial, check the syringe for air bubbles. If bubbles are in the syringe, hold the syringe straight up and tap the side of the syringe until the bubbles float to the top. Push the bubbles out with the plunger and draw insulin back in until you have the correct dose.
Step 6:
Remove the needle from the vial. Do not let the needle touch anything. You are now ready to inject.
Giving your Insulin glargine injection:
- Inject your insulin exactly as your healthcare provider has shown you.
- Inject your insulin under the skin (subcutaneously) of your upper legs (thighs), upper arms, or stomach area (abdomen).
- Change (rotate) your injection sites within the area you choose for each dose to reduce your risk of getting lipodystrophy (pits in the skin or thickened skin) and localized cutaneous amyloidosis (skin with lumps) at the injection sites.
- Do not inject where the skin has pits, is thickened, or has lumps.
- Do not inject where the skin is tender, bruised, scaly or hard, or into scars or damaged skin.
Step 7:
Choosing your injection site: Insulin glargine is injected under the skin (subcutaneously) of your upper arm, thigh, or stomach area (abdomen). Wipe the skin with an alcohol swab to clean the injection site. Let the injection site dry before you inject your dose.
Step 8:
- Pinch the skin.
- Insert the needle in the way your healthcare provider showed you.
- Release the skin.
- Slowly push in the plunger of the syringe all the way, making sure you have injected all the insulin.
- Leave the needle in the skin for about 10 seconds.
Step 9:
- Pull the needle straight out of your skin.
- Gently press the injection site for several seconds. Do not rub the area.
- Do not recap the used needle. Recapping the needle can lead to a needle stick injury.
Disposing of used needles and syringes:
- Put your used needles and syringes in a FDA-cleared sharps disposal container right away after use. Do not throw away (dispose of) loose needles and syringes in your household trash.
- If you do not have a FDA-cleared sharps container, you may use a household container that is:
- made of a heavy-duty plastic,
- can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out,
- upright and stable during use,
- leak resistant, and
- properly labeled to warn of hazardous waste inside the container.
- When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal.
- Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container.
How should I store Insulin glargine?
- Store unused Insulin glargine vials in the refrigerator between 36°F to 46°F (2°C to 8°C).
- Store in-use (opened) Insulin glargine vials in a refrigerator or at room temperature below 86°F (30°C).
- Do not freeze Insulin glargine.
- Keep Insulin glargine out of direct heat and light.
- If a vial has been frozen or overheated, throw it away.
- The Insulin glargine vials you are using should be thrown away after 28 days, even if it still has insulin left in it.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Revised: December 2020
Winthrop U.S.,
a business of sanofi-aventis U.S. LLC
Bridgewater, NJ 08807
A SANOFI COMPANY
©2020 sanofi-aventis U.S. LLC
12.2 Pharmacodynamics
In clinical studies, the glucose-lowering effect on a molar basis (i.e., when given at the same doses) of intravenous insulin glargine is approximately the same as that for human insulin. Figure 1 shows results from a study in patients with type 1 diabetes conducted for a maximum of 24 hours after the injection. The median time between injection and the end of pharmacological effect was 14.5 hours (range: 9.5 to 19.3 hours) for NPH insulin, and 24 hours (range: 10.8 to >24.0 hours) (24 hours was the end of the observation period) for insulin glargine.
| Figure 1: Activity Profile in Patients with Type 1 Diabetes |
|---|
| * Determined as amount of glucose infused to maintain constant plasma glucose levels |
|
|
The duration of action after abdominal, deltoid, or thigh subcutaneous administration was similar. The time course of action of insulins, including Insulin glargine, may vary between individuals and within the same individual.
5.4 Medication Errors
Accidental mix-ups among insulin products, particularly between long-acting insulins and rapid-acting insulins, have been reported. To avoid medication errors between Insulin glargine and other insulins, instruct patients to always check the insulin label before each injection [see Adverse Reactions (6.3)].
8.6 Hepatic Impairment
The effect of hepatic impairment on the pharmacokinetics of Insulin glargine has not been studied. Frequent glucose monitoring and dose adjustment may be necessary for Insulin glargine in patients with hepatic impairment [see Warnings and Precautions (5.3)].
1 Indications and Usage
Insulin glargine is indicated to improve glycemic control in adults and pediatric patients with type 1 diabetes mellitus and in adults with type 2 diabetes mellitus.
12.1 Mechanism of Action
The primary activity of insulin, including insulin glargine, is regulation of glucose metabolism. Insulin and its analogs lower blood glucose by stimulating peripheral glucose uptake, especially by skeletal muscle and fat, and by inhibiting hepatic glucose production. Insulin inhibits lipolysis and proteolysis, and enhances protein synthesis.
5 Warnings and Precautions
- Never share an Insulin glargine SoloStar prefilled pen between patients, even if the needle is changed. (5.1)
- Hyperglycemia or hypoglycemia with changes in insulin regimen: Make changes to a patient's insulin regimen (e.g., insulin strength, manufacturer, type, injection site or method of administration) under close medical supervision with increased frequency of blood glucose monitoring. (5.2)
- Hypoglycemia: May be life-threatening. Increase frequency of glucose monitoring with changes to: insulin dosage, coadministered glucose lowering medications, meal pattern, physical activity; and in patients with renal or hepatic impairment and hypoglycemia unawareness. (5.3, 6.1)
- Medication Errors: Accidental mix-ups between insulin products can occur. Instruct patients to check insulin labels before injection. (5.4, 6.3)
- Hypersensitivity reactions: Severe, life-threatening, generalized allergy, including anaphylaxis, can occur. Discontinue Insulin glargine. Monitor and treat if indicated. (5.5, 6.1)
- Hypokalemia: May be life-threatening. Monitor potassium levels in patients at risk of hypokalemia and treat if indicated. (5.6)
- Fluid retention and heart failure with concomitant use of thiazolidinediones (TZDs): Observe for signs and symptoms of heart failure; consider dosage reduction or discontinuation of TZD if heart failure occurs. (5.7)
2 Dosage and Administration
- Individualize dosage based on metabolic needs, blood glucose monitoring, glycemic control, type of diabetes, and prior insulin use. (2.1, 2.3, 2.4)
- Administer subcutaneously into the abdominal area, thigh, or deltoid once daily at any time of day, but at the same time every day. (2.1)
- Do not dilute or mix with any other insulin or solution. (2.1)
- Rotate injection sites to reduce risk of lipodystrophy and localized cutaneous amyloidosis. (2.2)
- Closely monitor glucose when changing to Insulin glargine and during initial weeks thereafter. (2.4)
3 Dosage Forms and Strengths
Injection: 100 units per mL (U-100) available as:
- 10 mL multiple-dose vial (1,000 units/10 mL)
- 3 mL single-patient-use SoloStar prefilled pen (300 units/3 mL)
6.3 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of Insulin glargine. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Medication errors have been reported in which other insulins, particularly rapid-acting insulins, have been accidentally administered instead of Insulin glargine [see Patient Counseling Information (17)]. To avoid medication errors between Insulin glargine and other insulins, patients should be instructed to always verify the insulin label before each injection.
Localized cutaneous amyloidosis at the injection site has occurred. Hyperglycemia has been reported with repeated insulin injections into areas of localized cutaneous amyloidosis; hypoglycemia has been reported with a sudden change to an unaffected injection site.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of a drug cannot be directly compared to rates in the clinical trial of another drug and may not reflect the rates observed in practice.
The data in Table 1 reflect the exposure of 2327 patients with type 1 diabetes to Insulin glargine or NPH. The type 1 diabetes population had the following characteristics: Mean age was 38.5 years. Fifty-four percent were male, 96.9% were Caucasian, 1.8% were Black or African American and 2.7% were Hispanic. The mean BMI was 25.1 kg/m2.
The data in Table 2 reflect the exposure of 1563 patients with type 2 diabetes to Insulin glargine or NPH. The type 2 diabetes population had the following characteristics: Mean age was 59.3 years. Fifty-eight percent were male, 86.7% were Caucasian, 7.8% were Black or African American and 9% were Hispanic. The mean BMI was 29.2 kg/m2.
The frequencies of adverse events during Insulin glargine clinical trials in patients with type 1 diabetes mellitus and type 2 diabetes mellitus are listed in the tables below.
| Insulin glargine, % (n=1257) |
NPH, % (n=1070) |
|
|---|---|---|
| Upper respiratory tract infection | 22.4 | 23.1 |
| Infection Body system not specified
|
9.4 | 10.3 |
| Accidental injury | 5.7 | 6.4 |
| Headache | 5.5 | 4.7 |
| Insulin glargine, % (n=849) |
NPH, % (n=714) |
|
|---|---|---|
| Upper respiratory tract infection | 11.4 | 13.3 |
| Infection Body system not specified
|
10.4 | 11.6 |
| Retinal vascular disorder | 5.8 | 7.4 |
| Insulin glargine, % (n=514) |
NPH, % (n=503) |
|
|---|---|---|
| Upper respiratory tract infection | 29.0 | 33.6 |
| Edema peripheral | 20.0 | 22.7 |
| Hypertension | 19.6 | 18.9 |
| Influenza | 18.7 | 19.5 |
| Sinusitis | 18.5 | 17.9 |
| Cataract | 18.1 | 15.9 |
| Bronchitis | 15.2 | 14.1 |
| Arthralgia | 14.2 | 16.1 |
| Pain in extremity | 13.0 | 13.1 |
| Back pain | 12.8 | 12.3 |
| Cough | 12.1 | 7.4 |
| Urinary tract infection | 10.7 | 10.1 |
| Diarrhea | 10.7 | 10.3 |
| Depression | 10.5 | 9.7 |
| Headache | 10.3 | 9.3 |
| Insulin glargine, % (n=174) |
NPH, % (n=175) |
|
|---|---|---|
| Infection Body system not specified
|
13.8 | 17.7 |
| Upper respiratory tract infection | 13.8 | 16.0 |
| Pharyngitis | 7.5 | 8.6 |
| Rhinitis | 5.2 | 5.1 |
2.2 General Dosing Instructions
- Individualize and adjust the dosage of Insulin glargine based on the individual's metabolic needs, blood glucose monitoring results and glycemic control goal.
- Dosage adjustments may be needed with changes in physical activity, changes in meal patterns (i.e., macronutrient content or timing of food intake), during acute illness, or changes in renal or hepatic function. Dosage adjustments should only be made under medical supervision with appropriate glucose monitoring [see Warnings and Precautions (5.2)].
14.1 Overview of Clinical Studies
The safety and effectiveness of Insulin glargine given once-daily at bedtime was compared to that of once-daily and twice-daily NPH insulin in open-label, randomized, active-controlled, parallel studies of 2,327 adult patients and 349 pediatric patients with type 1 diabetes mellitus and 1,563 adult patients with type 2 diabetes mellitus (see Tables 9–11). In general, the reduction in glycated hemoglobin (HbA1c) with Insulin glargine was similar to that with NPH insulin.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
16 How Supplied/storage and Handling
Product: 50090-6178
NDC: 50090-6178-0 3 mL in a SYRINGE / 5 in a CARTON
2.1 Important Administration Instructions
- Administer Insulin glargine subcutaneously once daily at any time of day but at the same time every day.
- Prior to initiation of Insulin glargine, train patients on proper use and injection technique.
- Patient should follow the Instructions for Use to correctly administer Insulin glargine.
- Administer Insulin glargine subcutaneously into the abdominal area, thigh, or deltoid, and rotate injection sites within the same region from one injection to the next to reduce the risk of lipodystrophy and localized cutaneous amyloidosis. Do not inject into areas of lipodystrophy or localized cutaneous amyloidosis [see Warnings and Precautions (5.2), Adverse Reactions (6)].
- During changes to a patient's insulin regimen, increase the frequency of blood glucose monitoring [see Warnings and Precautions (5.2)].
- Visually inspect Insulin glargine vials and SoloStar prefilled pens for particulate matter and discoloration prior to administration. Only use if the solution is clear and colorless with no visible particles.
- The Insulin glargine SoloStar prefilled pen dials in 1-unit increments.
- Use Insulin glargine SoloStar prefilled pen with caution in patients with visual impairment who may rely on audible clicks to dial their dose.
- Refrigerate unused (unopened) Insulin glargine vials and SoloStar® prefilled pens.
- Do not administer intravenously or via an insulin pump.
- Do not dilute or mix Insulin glargine with any other insulin or solution.
- The SoloStar prefilled pen is for single patient use only [see Warnings and Precautions (5.1)].
5.5 Hypersensitivity and Allergic Reactions
Severe, life-threatening, generalized allergy, including anaphylaxis, can occur with insulin products, including Insulin glargine. If hypersensitivity reactions occur, discontinue Insulin glargine; treat per standard of care and monitor until symptoms and signs resolve [see Adverse Reactions (6.1)]. Insulin glargine is contraindicated in patients who have had hypersensitivity reactions to insulin glargine or one of the excipients [see Contraindications (4)].
14.3 Clinical Studies in Adults With Type 2 Diabetes
In a randomized, controlled clinical study (Study E) (n=570), Insulin glargine was evaluated for 52 weeks in combination with oral antidiabetic medications (a sulfonylurea, metformin, acarbose, or combinations of these drugs). The average age was 59.5 years. The majority of patients were White (92.8%) and 53.7% were male. The mean BMI was approximately 29.1 kg/m2. The mean duration of diabetes was 10.3 years. Insulin glargine administered once daily at bedtime was as effective as NPH insulin administered once daily at bedtime in reducing HbA1c and fasting glucose (Table 11). The rate of severe symptomatic hypoglycemia was similar in Insulin glargine and NPH insulin treated patients [see Adverse Reactions (6.1)].
In a randomized, controlled clinical study (Study F), in patients with type 2 diabetes not using oral antidiabetic medications (n=518), a basal-bolus regimen of Insulin glargine once daily at bedtime or NPH insulin administered once or twice daily was evaluated for 28 weeks. Regular human insulin was used before meals, as needed. The average age was 59.3 years. The majority of patients were White (80.7%) and 60% were male. The mean BMI was approximately 30.5 kg/m2. The mean duration of diabetes was 13.7 years. Insulin glargine had similar effectiveness as either once- or twice-daily NPH insulin in reducing HbA1c and fasting glucose (Table 11) with a similar incidence of hypoglycemia [see Adverse Reactions (6.1)].
In a randomized, controlled clinical study (Study G), patients with type 2 diabetes were randomized to 5 years of treatment with once-daily Insulin glargine or twice-daily NPH insulin. For patients not previously treated with insulin, the starting dose of Insulin glargine or NPH insulin was 10 units daily. Patients who were already treated with NPH insulin either continued on the same total daily NPH insulin dose or started Insulin glargine at a dose that was 80% of the total previous NPH insulin dose. The primary endpoint for this study was a comparison of the progression of diabetic retinopathy by 3 or more steps on the Early Treatment Diabetic Retinopathy Study (ETDRS) scale. HbA1c change from baseline was a secondary endpoint. Similar glycemic control in the 2 treatment groups was desired in order to not confound the interpretation of the retinal data. Patients or study personnel used an algorithm to adjust the Insulin glargine and NPH insulin doses to a target fasting plasma glucose ≤100 mg/dL. After the Insulin glargine or NPH insulin dose was adjusted, other antidiabetic agents, including premeal insulin were to be adjusted or added. The average age was 55.1 years. The majority of patients were White (85.3%) and 53.9% were male. The mean BMI was approximately 34.3 kg/m2. The mean duration of diabetes was 10.8 years. The Insulin glargine group had a smaller mean reduction from baseline in HbA1c compared to the NPH insulin group, which may be explained by the lower daily basal insulin doses in the Insulin glargine group (Table 11). The incidences of severe symptomatic hypoglycemia were similar between groups [see Adverse Reactions (6.1)].
| Study E | Study F | Study G | ||||
|---|---|---|---|---|---|---|
| Treatment duration | 52 weeks | 28 weeks | 5 years | |||
| Treatment in combination with | Oral agents | Regular insulin | Regular insulin | |||
| Insulin glargine | NPH | Insulin glargine | NPH | Insulin glargine | NPH | |
| Number of subjects treated | 289 | 281 | 259 | 259 | 513 | 504 |
| HbA1c | ||||||
| Baseline mean | 9.0 | 8.9 | 8.6 | 8.5 | 8.4 | 8.3 |
| Adjusted mean change from baseline | -0.5 | -0.4 | -0.4 | -0.6 | -0.6 | -0.8 |
| Insulin glargine – NPH | -0.1 | +0.2 | +0.2 | |||
| 95% CI for Treatment difference | (-0.3; +0.1) | (0.0; +0.4) | (+0.1; +0.4) | |||
|
Basal insulin dose In Study G, the baseline dose of basal or total insulin was the first available on-treatment dose prescribed during the study (on visit month 1.5).
|
||||||
| Baseline mean | 14 | 15 | 44.1 | 45.5 | 39 | 44 |
| Mean change from baseline | +12 | +9 | -1 | +7 | +23 | +30 |
| Total insulin dose | ||||||
| Baseline mean | 14 | 15 | 64 | 67 | 48 | 53 |
| Mean change from baseline | +12 | +9 | +10 | +13 | +41 | +40 |
| Fasting blood glucose (mg/dL) | ||||||
| Baseline mean | 179 | 180 | 164 | 166 | 190 | 180 |
| Adj. mean change from baseline | -49 | -46 | -24 | -22 | -45 | -44 |
| Body weight (kg) | ||||||
| Baseline mean | 83.5 | 82.1 | 89.6 | 90.7 | 100 | 99 |
| Adj. mean change from baseline | 2.0 | 1.9 | 0.4 | 1.4 | 3.7 | 4.8 |
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In mice and rats, standard two-year carcinogenicity studies with insulin glargine were performed at doses up to 0.455 mg/kg, which was for the rat approximately 65 times the recommended human subcutaneous starting dose of 0.2 units/kg/day (0.007 mg/kg/day) on a mg/kg basis. Histiocytomas were found at injection sites in male rats and mice in acid vehicle containing groups and are considered a response to chronic tissue irritation and inflammation in rodents. These tumors were not found in female animals, in saline control, or insulin comparator groups using a different vehicle.
Insulin glargine was not mutagenic in tests for detection of gene mutations in bacteria and mammalian cells (Ames and HGPRT-test) and in tests for detection of chromosomal aberrations (cytogenetics in vitro in V79 cells and in vivo in Chinese hamsters).
In a combined fertility and prenatal and postnatal study in male and female rats at subcutaneous doses up to 0.36 mg/kg/day, which was approximately 50 times the recommended human subcutaneous starting dose of 0.2 units/kg/day (0.007 mg/kg/day) maternal toxicity due to dose-dependent hypoglycemia, including some deaths, was observed. Consequently, a reduction of the rearing rate occurred in the high-dose group only. Similar effects were observed with NPH insulin.
2.4 Changing to Insulin Glargine From Other Insulin Therapies
- If changing patients from once-daily TOUJEO (insulin glargine) 300 units/mL to once-daily Insulin glargine, the recommended initial Insulin glargine dose is 80% of the TOUJEO dose that is being discontinued. This dose reduction will lower the likelihood of hypoglycemia [see Warnings and Precautions (5.3)].
- If changing from a treatment regimen with an intermediate or long-acting insulin to a regimen with Insulin glargine, a change in the dose of the basal insulin may be required and the amount and timing of the shorter-acting insulins and doses of any oral antidiabetic drugs may need to be adjusted.
- If changing patients from once-daily NPH insulin to once-daily Insulin glargine, the recommended initial Insulin glargine dose is the same as the dose of NPH that is being discontinued.
- If changing patients from twice-daily NPH insulin to once-daily Insulin glargine, the recommended initial Insulin glargine dosage is 80% of the total NPH dose that is being discontinued. This dosage reduction will lower the likelihood of hypoglycemia [see Warnings and Precautions (5.3)].
5.2 Hyperglycemia Or Hypoglycemia With Changes in Insulin Regimen
Changes in an insulin regimen (e.g., insulin strength, manufacturer, type, injection site or method of administration) may affect glycemic control and predispose to hypoglycemia [see Warnings and Precautions (5.3)] or hyperglycemia. Repeated insulin injections into areas of lipodystrophy or localized cutaneous amyloidosis have been reported to result in hyperglycemia; and a sudden change in the injection site (to unaffected area) has been reported to result in hypoglycemia [see Adverse Reactions (6)].
Make any changes to a patient's insulin regimen under close medical supervision with increased frequency of blood glucose monitoring. Advise patients who have repeatedly injected into areas of lipodystrophy or localized cutaneous amyloidosis to change the injection site to unaffected areas and closely monitor for hypoglycemia. For patients with type 2 diabetes, dosage adjustments of concomitant oral and antidiabetic products may be needed.
14.2 Clinical Studies in Adult and Pediatric Patients With Type 1 Diabetes
In two clinical studies (Studies A and B), patients with type 1 diabetes (Study A n=585, Study B n=534) were randomized to 28 weeks of basal-bolus treatment with Insulin glargine or NPH insulin. Regular human insulin was administered before each meal. Insulin glargine was administered at bedtime. NPH insulin was administered either as once daily at bedtime or in the morning and at bedtime when used twice daily.
In Study A, the average age was 39.2 years. The majority of patients were White (99%) and 55.7% were male. The mean BMI was approximately 24.9 kg/m2. The mean duration of diabetes was 15.5 years.
In Study B, the average age was 38.5 years. The majority of patients were White (95.3%) and 50.6% were male. The mean BMI was approximately 25.8 kg/m2. The mean duration of diabetes was 17.4 years.
In another clinical study (Study C), patients with type 1 diabetes (n=619) were randomized to 16 weeks of basal-bolus treatment with Insulin glargine or NPH insulin. Insulin lispro was used before each meal. Insulin glargine was administered once daily at bedtime and NPH insulin was administered once or twice daily. The average age was 39.2 years. The majority of patients were White (96.9%) and 50.6% were male. The mean BMI was approximately 25.6 kg/m2. The mean duration of diabetes was 18.5 years.
In these 3 studies, Insulin glargine and NPH insulin had similar effects on HbA1c (Table 9) with a similar overall rate of severe symptomatic hypoglycemia [see Adverse Reactions (6.1)].
| Study A | Study B | Study C | ||||
|---|---|---|---|---|---|---|
| Treatment duration | 28 weeks | 28 weeks | 16 weeks | |||
| Treatment in combination with | Regular insulin | Regular insulin | Insulin lispro | |||
| Insulin glargine | NPH | Insulin glargine | NPH | Insulin glargine | NPH | |
| Number of subjects treated | 292 | 293 | 264 | 270 | 310 | 309 |
| HbA1c | ||||||
| Baseline HbA1c | 8.0 | 8.0 | 7.7 | 7.7 | 7.6 | 7.7 |
| Adjusted mean change at trial end | +0.2 | +0.1 | -0.2 | -0.2 | -0.1 | -0.1 |
| Treatment Difference (95% CI) | +0.1 (0.0; +0.2) | +0.1 (-0.1; +0.2) | 0.0 (-0.1; +0.1) | |||
| Basal insulin dose | ||||||
| Baseline mean | 21 | 23 | 29 | 29 | 28 | 28 |
| Mean change from baseline | -2 | 0 | -4 | +2 | -5 | +1 |
| Total insulin dose | ||||||
| Baseline mean | 48 | 52 | 50 | 51 | 50 | 50 |
| Mean change from baseline | -1 | 0 | 0 | +4 | -3 | 0 |
| Fasting blood glucose (mg/dL) | ||||||
| Baseline mean | 167 | 166 | 166 | 175 | 175 | 173 |
| Adj. mean change from baseline | -21 | -16 | -20 | -17 | -29 | -12 |
| Body weight (kg) | ||||||
| Baseline mean | 73.2 | 74.8 | 75.5 | 75.0 | 74.8 | 75.6 |
| Mean change from baseline | 0.1 | -0.0 | 0.7 | 1.0 | 0.1 | 0.5 |
5.7 Fluid Retention and Heart Failure With Concomitant Use of Ppar Gamma Agonists
Thiazolidinediones (TZDs), which are peroxisome proliferator-activated receptor (PPAR)-gamma agonists, can cause dose-related fluid retention, particularly when used in combination with insulin. Fluid retention may lead to or exacerbate heart failure. Patients treated with insulin, including Insulin glargine, and a PPAR-gamma agonist should be observed for signs and symptoms of heart failure. If heart failure develops, it should be managed according to current standards of care, and discontinuation or dose reduction of the PPAR-gamma agonist must be considered.
5.1 Never Share An Insulin Glargine Solostar Prefilled Pen, Syringe, Or Needle Between Patients
Insulin glargine SoloStar prefilled pens must never be shared between patients, even if the needle is changed. Patients using Insulin glargine vials must never re-use or share needles or syringes with another person. Sharing poses a risk for transmission of blood-borne pathogens.
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use
Insulin glargine is not recommended for the treatment of diabetic ketoacidosis.
Section 42230-3 (42230-3)
| Patient Information Insulin glargine (IN-su-lin-GLAR-gine) (insulin glargine injection) for subcutaneous use, 100 Units/mL (U-100) |
|
|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration | Revised: December 2020 |
|
Do not share your syringes with other people, even if the needle has been changed. You may give other people a serious infection, or get a serious infection from them. What is Insulin glargine? Insulin glargine is a long-acting man-made-insulin used to control high blood sugar in adults with diabetes mellitus.
|
|
|
Who should not use Insulin glargine? Do not use Insulin glargine if you:
|
|
|
What should I tell my healthcare provider before using Insulin glargine? Before using Insulin glargine, tell your healthcare provider about all your medical conditions including if you:
Before you start using Insulin glargine, talk to your healthcare provider about low blood sugar and how to manage it. |
|
How should I use Insulin glargine?
|
|
Your dose of Insulin glargine may need to change because of:
|
|
|
What should I avoid while using Insulin glargine? While using Insulin glargine do not:
|
|
|
What are the possible side effects of Insulin glargine and other insulins? Insulin glargine may cause serious side effects that can lead to death, including:
|
|
|
General information about the safe and effective use of Insulin glargine.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Insulin glargine for a condition for which it was not prescribed. Do not give Insulin glargine to other people, even if they have the same symptoms that you have. It may harm them. This Patient Information leaflet summarizes the most important information about Insulin glargine. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about Insulin glargine that is written for healthcare professionals. For more information, go to www.winthropus.com or call 1-800-633-1610. |
|
What are the ingredients in Insulin glargine?
|
|
| Manufactured by: Winthrop U.S., a business of sanofi-aventis U.S. LLC, Bridgewater, NJ 08807 |
Section 59845-8 (59845-8)
| Patient Information Insulin glargine (IN-su-lin-GLAR-gine) (insulin glargine injection) for subcutaneous use, 100 Units/mL (U-100) |
|
|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: December 2020 |
|
Do not share your Insulin glargine SoloStar® pen with other people, even if the needle has been changed. You may give other people a serious infection, or get a serious infection from them. What is Insulin glargine? Insulin glargine is a long-acting man-made insulin used to control high blood sugar in adults with diabetes mellitus.
|
|
|
Who should not use Insulin glargine? Do not use Insulin glargine if you:
|
|
|
What should I tell my healthcare provider before using Insulin glargine? Before using Insulin glargine, tell your healthcare provider about all your medical conditions including if you:
Before you start using Insulin glargine, talk to your healthcare provider about low blood sugar and how to manage it. |
|
How should I use Insulin glargine?
|
|
Your dose of Insulin glargine may need to change because of:
|
|
|
What should I avoid while using Insulin glargine? While using Insulin glargine do not:
|
|
|
What are the possible side effects of Insulin glargine and other insulins? Insulin glargine may cause serious side effects that can lead to death, including:
|
|
|
General information about the safe and effective use of Insulin glargine.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Insulin glargine for a condition for which it was not prescribed. Do not give Insulin glargine to other people, even if they have the same symptoms that you have. It may harm them. This Patient Information leaflet summarizes the most important information about Insulin glargine. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about Insulin glargine that is written for healthcare professionals. For more information about Insulin glargine call 1-800-633-1610 or go to the website www.winthropus.com. |
|
What are the ingredients in Insulin glargine?
|
|
| Manufactured by: Winthrop U.S., a business of sanofi-aventis U.S., LLC Bridgewater, NJ 08807 |
8.8 Obesity
In controlled clinical trials, subgroup analyses based on BMI did not show differences in safety and efficacy between Insulin glargine and NPH.
10 Overdosage (10 OVERDOSAGE)
Excess insulin administration may cause hypoglycemia and hypokalemia [see Warnings and Precautions (5.3, 5.6)]. Mild episodes of hypoglycemia can usually be treated with oral carbohydrates. Adjustments in drug dosage, meal patterns, or exercise may be needed.
More severe episodes of hypoglycemia with coma, seizure, or neurologic impairment may be treated with intramuscular/subcutaneous glucagon or concentrated intravenous glucose. After apparent clinical recovery from hypoglycemia, continued observation and additional carbohydrate intake may be necessary to avoid recurrence of hypoglycemia. Hypokalemia must be corrected appropriately.
11 Description (11 DESCRIPTION)
Insulin glargine (insulin glargine injection) is a sterile solution of insulin glargine for subcutaneous use. Insulin glargine is a recombinant human insulin analog that is a long-acting, parenteral blood-glucose-lowering agent [see Clinical Pharmacology (12)]. Insulin glargine has low aqueous solubility at neutral pH. At pH 4 insulin glargine is completely soluble. After injection into the subcutaneous tissue, the acidic solution is neutralized, leading to formation of microprecipitates from which small amounts of insulin glargine are slowly released, resulting in a relatively constant concentration/time profile over 24 hours with no pronounced peak. This profile allows once-daily dosing as a basal insulin. Insulin glargine is produced by recombinant DNA technology utilizing a non-pathogenic laboratory strain of Escherichia coli (K12) as the production organism. Insulin glargine differs from human insulin in that the amino acid asparagine at position A21 is replaced by glycine and two arginines are added to the C-terminus of the B-chain. Chemically, insulin glargine is 21A-Gly-30Ba-L-Arg-30Bb-L-Arg-human insulin and has the empirical formula C267H404N72O78S6 and a molecular weight of 6063. Insulin glargine has the following structural formula:
Insulin glargine consists of insulin glargine dissolved in a clear aqueous fluid. Each milliliter of insulin glargine injection contains 100 units (3.6378 mg) insulin glargine.
The 10 mL vial presentation contains the following inactive ingredients per mL: 30 mcg zinc, 2.7 mg m-cresol, 20 mg glycerol 85%, 20 mcg polysorbate 20, and water for injection.
The 3 mL prefilled pen presentation contains the following inactive ingredients per mL: 30 mcg zinc, 2.7 mg m-cresol, 20 mg glycerol 85%, and water for injection.
The pH is adjusted by addition of aqueous solutions of hydrochloric acid and sodium hydroxide. Insulin glargine has a pH of approximately 4.
5.6 Hypokalemia
All insulin products, including Insulin glargine, cause a shift in potassium from the extracellular to intracellular space, possibly leading to hypokalemia. Untreated hypokalemia may cause respiratory paralysis, ventricular arrhythmia, and death. Monitor potassium levels in patients at risk for hypokalemia, if indicated (e.g., patients using potassium-lowering medications, patients taking medications sensitive to serum potassium concentrations).
5.3 Hypoglycemia
Hypoglycemia is the most common adverse reaction associated with insulin, including Insulin glargine. Severe hypoglycemia can cause seizures, may be life-threatening or cause death. Hypoglycemia can impair concentration ability and reaction time; this may place an individual and others at risk in situations where these abilities are important (e.g., driving or operating other machinery).
Hypoglycemia can happen suddenly and symptoms may differ in each individual and change over time in the same individual. Symptomatic awareness of hypoglycemia may be less pronounced in patients with longstanding diabetes, in patients with diabetic nerve disease, in patients using medications that block the sympathetic nervous system (e.g., beta-blockers) [see Drug Interactions (7)], or in patients who experience recurrent hypoglycemia.
Insulin Glargine (INSULIN GLARGINE)
8.4 Pediatric Use
The safety and effectiveness of Insulin glargine have been established in pediatric patients (age 6 to 15 years) with type 1 diabetes [see Clinical Studies (14.2)]. The safety and effectiveness of Insulin glargine in pediatric patients younger than 6 years of age with type 1 diabetes and pediatric patients with type 2 diabetes have not been established.
The dosage recommendation when changing to Insulin glargine in pediatric patients (age 6 to 15 years) with type 1 diabetes is the same as that described for adults [see Dosage and Administration (2.2, 2.4) , Clinical Studies (14)]. As in adults, the dosage of Insulin glargine must be individualized in pediatric patients (age 6 to 15 years) with type 1 diabetes based on metabolic needs and frequent monitoring of blood glucose.
In the pediatric clinical trial, pediatric patients (age 6 to 15 years) with type 1 diabetes had a higher incidence of severe symptomatic hypoglycemia compared to the adults in trials with type 1 diabetes [see Adverse Reactions (6.1)].
8.5 Geriatric Use
Of the total number of subjects in controlled clinical studies of patients with type 1 and type 2 diabetes who were treated with Insulin glargine, 15% were ≥65 years of age and 2% were ≥75 years of age. The only difference in safety or effectiveness in the subpopulation of patients ≥65 years of age compared to the entire study population was a higher incidence of cardiovascular events typically seen in an older population in the Insulin glargine and NPH treatment groups.
Nevertheless, caution should be exercised when Insulin glargine is administered to geriatric patients. In elderly patients with diabetes, the initial dosing, dose increments, and maintenance dosage should be conservative to avoid hypoglycemic reactions. Hypoglycemia may be difficult to recognize in the elderly.
6.2 Immunogenicity
As with all therapeutic proteins, there is potential for immunogenicity. All insulin products can elicit the formation of insulin antibodies. The presence of such insulin antibodies may increase or decrease the efficacy of insulin and may require adjustment of the insulin dose. In phase 3 clinical trials of Insulin glargine, increases in titers of antibodies to insulin were observed in NPH insulin and Insulin glargine treatment groups with similar incidences.
4 Contraindications (4 CONTRAINDICATIONS)
Insulin glargine is contraindicated:
- during episodes of hypoglycemia [see Warnings and Precautions (5.3)]
- in patients with hypersensitivity to Insulin glargine or one of its excipients [see Warnings and Precautions (5.5)]
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed elsewhere:
- Hypoglycemia [see Warnings and Precautions (5.3)]
- Hypersensitivity and allergic reactions [see Warnings and Precautions (5.5)]
- Hypokalemia [see Warnings and Precautions (5.6)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Table 8 includes clinically significant drug interactions with Insulin glargine.
| Drugs that May Increase the Risk of Hypoglycemia | |
| Drugs: | Antidiabetic agents, ACE inhibitors, angiotensin II receptor blocking agents, disopyramide, fibrates, fluoxetine, monoamine oxidase inhibitors, pentoxifylline, pramlintide, salicylates, somatostatin analogs (e.g., octreotide), and sulfonamide antibiotics. |
| Intervention: | Dose reductions and increased frequency of glucose monitoring may be required when Insulin glargine is coadministered with these drugs. |
| Drugs that May Decrease the Blood Glucose Lowering Effect of Insulin Glargine | |
| Drugs: | Atypical antipsychotics (e.g., olanzapine and clozapine), corticosteroids, danazol, diuretics, estrogens, glucagon, isoniazid, niacin, oral contraceptives, phenothiazines, progestogens (e.g., in oral contraceptives), protease inhibitors, somatropin, sympathomimetic agents (e.g., albuterol, epinephrine, terbutaline), and thyroid hormones. |
| Intervention: | Dose increases and increased frequency of glucose monitoring may be required when Insulin glargine is coadministered with these drugs. |
| Drugs that May Increase or Decrease the Blood Glucose Lowering Effect of Insulin Glargine | |
| Drugs: | Alcohol, beta-blockers, clonidine, and lithium salts. Pentamidine may cause hypoglycemia, which may sometimes be followed by hyperglycemia. |
| Intervention: | Dose adjustment and increased frequency of glucose monitoring may be required when Insulin glargine is coadministered with these drugs. |
| Drugs that May Blunt Signs and Symptoms of Hypoglycemia | |
| Drugs: | Beta-blockers, clonidine, guanethidine, and reserpine. |
| Intervention: | Increased frequency of glucose monitoring may be required when Insulin glargine is coadministered with these drugs. |
8.7 Renal Impairment
The effect of renal impairment on the pharmacokinetics of Insulin glargine has not been studied. Some studies with human insulin have shown increased circulating levels of insulin in patients with renal failure. Frequent glucose monitoring and dose adjustment may be necessary for Insulin glargine in patients with renal impairment [see Warnings and Precautions (5.3)].
Instructions for Use
Insulin glargine (IN-su-lin-GLAR-gine)
injection for subcutaneous use
10 mL Vial (100 Units/mL, U-100)
Read the Instructions for Use before you start taking Insulin glargine and each time you get a new Insulin glargine vial. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
Do not share your Insulin glargine syringes with other people even if the needle has been changed. You may give other people a serious infection, or get a serious infection from them.
Supplies needed to give your injection:
- an Insulin glargine 10 mL vial
- a U-100 insulin syringe and needle
- 2 alcohol swabs
- 1 sharps container for throwing away used needles and syringes. See "Disposing of used needles and syringes" at the end of these instructions.
Preparing your Insulin glargine dose:
- Wash your hands with soap and water or with alcohol.
- Check the Insulin glargine label to make sure you are taking the right type of insulin. This is especially important if you use more than 1 type of insulin.
- Check the insulin to make sure it is clear and colorless. Do not use Insulin glargine if it is colored or cloudy, or if you see particles in the solution.
- Do not use Insulin glargine after the expiration date stamped on the label or 28 days after you first use it.
- Always use a syringe that is marked for U-100 insulin. If you use a syringe other than a U-100 insulin syringe, you may get the wrong dose of insulin.
- Always use a new syringe or needle for each injection. Do not re-use or share your syringes or needles with other people. You may give other people a serious infection or get a serious infection from them.
Step 1:
If you are using a new vial, remove the protective cap. Do not remove the stopper.
Step 2:
Wipe the top of the vial with an alcohol swab. You do not have to shake the vial of Insulin glargine before use.
Step 3:
Draw air into the syringe equal to your insulin dose. Put the needle through the rubber top of the vial and push the plunger to inject the air into the vial.
Step 4:
Leave the syringe in the vial and turn both upside down. Hold the syringe and vial firmly in one hand. Make sure the tip of the needle is in the insulin. With your free hand, pull the plunger to withdraw the correct dose into the syringe.
Step 5:
Before you take the needle out of the vial, check the syringe for air bubbles. If bubbles are in the syringe, hold the syringe straight up and tap the side of the syringe until the bubbles float to the top. Push the bubbles out with the plunger and draw insulin back in until you have the correct dose.
Step 6:
Remove the needle from the vial. Do not let the needle touch anything. You are now ready to inject.
Giving your Insulin glargine injection:
- Inject your insulin exactly as your healthcare provider has shown you.
- Inject your insulin under the skin (subcutaneously) of your upper legs (thighs), upper arms, or stomach area (abdomen).
- Change (rotate) your injection sites within the area you choose for each dose to reduce your risk of getting lipodystrophy (pits in the skin or thickened skin) and localized cutaneous amyloidosis (skin with lumps) at the injection sites.
- Do not inject where the skin has pits, is thickened, or has lumps.
- Do not inject where the skin is tender, bruised, scaly or hard, or into scars or damaged skin.
Step 7:
Choosing your injection site: Insulin glargine is injected under the skin (subcutaneously) of your upper arm, thigh, or stomach area (abdomen). Wipe the skin with an alcohol swab to clean the injection site. Let the injection site dry before you inject your dose.
Step 8:
- Pinch the skin.
- Insert the needle in the way your healthcare provider showed you.
- Release the skin.
- Slowly push in the plunger of the syringe all the way, making sure you have injected all the insulin.
- Leave the needle in the skin for about 10 seconds.
Step 9:
- Pull the needle straight out of your skin.
- Gently press the injection site for several seconds. Do not rub the area.
- Do not recap the used needle. Recapping the needle can lead to a needle stick injury.
Disposing of used needles and syringes:
- Put your used needles and syringes in a FDA-cleared sharps disposal container right away after use. Do not throw away (dispose of) loose needles and syringes in your household trash.
- If you do not have a FDA-cleared sharps container, you may use a household container that is:
- made of a heavy-duty plastic,
- can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out,
- upright and stable during use,
- leak resistant, and
- properly labeled to warn of hazardous waste inside the container.
- When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal.
- Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container.
How should I store Insulin glargine?
- Store unused Insulin glargine vials in the refrigerator between 36°F to 46°F (2°C to 8°C).
- Store in-use (opened) Insulin glargine vials in a refrigerator or at room temperature below 86°F (30°C).
- Do not freeze Insulin glargine.
- Keep Insulin glargine out of direct heat and light.
- If a vial has been frozen or overheated, throw it away.
- The Insulin glargine vials you are using should be thrown away after 28 days, even if it still has insulin left in it.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Revised: December 2020
Winthrop U.S.,
a business of sanofi-aventis U.S. LLC
Bridgewater, NJ 08807
A SANOFI COMPANY
©2020 sanofi-aventis U.S. LLC
12.2 Pharmacodynamics
In clinical studies, the glucose-lowering effect on a molar basis (i.e., when given at the same doses) of intravenous insulin glargine is approximately the same as that for human insulin. Figure 1 shows results from a study in patients with type 1 diabetes conducted for a maximum of 24 hours after the injection. The median time between injection and the end of pharmacological effect was 14.5 hours (range: 9.5 to 19.3 hours) for NPH insulin, and 24 hours (range: 10.8 to >24.0 hours) (24 hours was the end of the observation period) for insulin glargine.
| Figure 1: Activity Profile in Patients with Type 1 Diabetes |
|---|
| * Determined as amount of glucose infused to maintain constant plasma glucose levels |
|
|
The duration of action after abdominal, deltoid, or thigh subcutaneous administration was similar. The time course of action of insulins, including Insulin glargine, may vary between individuals and within the same individual.
5.4 Medication Errors
Accidental mix-ups among insulin products, particularly between long-acting insulins and rapid-acting insulins, have been reported. To avoid medication errors between Insulin glargine and other insulins, instruct patients to always check the insulin label before each injection [see Adverse Reactions (6.3)].
8.6 Hepatic Impairment
The effect of hepatic impairment on the pharmacokinetics of Insulin glargine has not been studied. Frequent glucose monitoring and dose adjustment may be necessary for Insulin glargine in patients with hepatic impairment [see Warnings and Precautions (5.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Insulin glargine is indicated to improve glycemic control in adults and pediatric patients with type 1 diabetes mellitus and in adults with type 2 diabetes mellitus.
12.1 Mechanism of Action
The primary activity of insulin, including insulin glargine, is regulation of glucose metabolism. Insulin and its analogs lower blood glucose by stimulating peripheral glucose uptake, especially by skeletal muscle and fat, and by inhibiting hepatic glucose production. Insulin inhibits lipolysis and proteolysis, and enhances protein synthesis.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Never share an Insulin glargine SoloStar prefilled pen between patients, even if the needle is changed. (5.1)
- Hyperglycemia or hypoglycemia with changes in insulin regimen: Make changes to a patient's insulin regimen (e.g., insulin strength, manufacturer, type, injection site or method of administration) under close medical supervision with increased frequency of blood glucose monitoring. (5.2)
- Hypoglycemia: May be life-threatening. Increase frequency of glucose monitoring with changes to: insulin dosage, coadministered glucose lowering medications, meal pattern, physical activity; and in patients with renal or hepatic impairment and hypoglycemia unawareness. (5.3, 6.1)
- Medication Errors: Accidental mix-ups between insulin products can occur. Instruct patients to check insulin labels before injection. (5.4, 6.3)
- Hypersensitivity reactions: Severe, life-threatening, generalized allergy, including anaphylaxis, can occur. Discontinue Insulin glargine. Monitor and treat if indicated. (5.5, 6.1)
- Hypokalemia: May be life-threatening. Monitor potassium levels in patients at risk of hypokalemia and treat if indicated. (5.6)
- Fluid retention and heart failure with concomitant use of thiazolidinediones (TZDs): Observe for signs and symptoms of heart failure; consider dosage reduction or discontinuation of TZD if heart failure occurs. (5.7)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Individualize dosage based on metabolic needs, blood glucose monitoring, glycemic control, type of diabetes, and prior insulin use. (2.1, 2.3, 2.4)
- Administer subcutaneously into the abdominal area, thigh, or deltoid once daily at any time of day, but at the same time every day. (2.1)
- Do not dilute or mix with any other insulin or solution. (2.1)
- Rotate injection sites to reduce risk of lipodystrophy and localized cutaneous amyloidosis. (2.2)
- Closely monitor glucose when changing to Insulin glargine and during initial weeks thereafter. (2.4)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: 100 units per mL (U-100) available as:
- 10 mL multiple-dose vial (1,000 units/10 mL)
- 3 mL single-patient-use SoloStar prefilled pen (300 units/3 mL)
6.3 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of Insulin glargine. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Medication errors have been reported in which other insulins, particularly rapid-acting insulins, have been accidentally administered instead of Insulin glargine [see Patient Counseling Information (17)]. To avoid medication errors between Insulin glargine and other insulins, patients should be instructed to always verify the insulin label before each injection.
Localized cutaneous amyloidosis at the injection site has occurred. Hyperglycemia has been reported with repeated insulin injections into areas of localized cutaneous amyloidosis; hypoglycemia has been reported with a sudden change to an unaffected injection site.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of a drug cannot be directly compared to rates in the clinical trial of another drug and may not reflect the rates observed in practice.
The data in Table 1 reflect the exposure of 2327 patients with type 1 diabetes to Insulin glargine or NPH. The type 1 diabetes population had the following characteristics: Mean age was 38.5 years. Fifty-four percent were male, 96.9% were Caucasian, 1.8% were Black or African American and 2.7% were Hispanic. The mean BMI was 25.1 kg/m2.
The data in Table 2 reflect the exposure of 1563 patients with type 2 diabetes to Insulin glargine or NPH. The type 2 diabetes population had the following characteristics: Mean age was 59.3 years. Fifty-eight percent were male, 86.7% were Caucasian, 7.8% were Black or African American and 9% were Hispanic. The mean BMI was 29.2 kg/m2.
The frequencies of adverse events during Insulin glargine clinical trials in patients with type 1 diabetes mellitus and type 2 diabetes mellitus are listed in the tables below.
| Insulin glargine, % (n=1257) |
NPH, % (n=1070) |
|
|---|---|---|
| Upper respiratory tract infection | 22.4 | 23.1 |
| Infection Body system not specified
|
9.4 | 10.3 |
| Accidental injury | 5.7 | 6.4 |
| Headache | 5.5 | 4.7 |
| Insulin glargine, % (n=849) |
NPH, % (n=714) |
|
|---|---|---|
| Upper respiratory tract infection | 11.4 | 13.3 |
| Infection Body system not specified
|
10.4 | 11.6 |
| Retinal vascular disorder | 5.8 | 7.4 |
| Insulin glargine, % (n=514) |
NPH, % (n=503) |
|
|---|---|---|
| Upper respiratory tract infection | 29.0 | 33.6 |
| Edema peripheral | 20.0 | 22.7 |
| Hypertension | 19.6 | 18.9 |
| Influenza | 18.7 | 19.5 |
| Sinusitis | 18.5 | 17.9 |
| Cataract | 18.1 | 15.9 |
| Bronchitis | 15.2 | 14.1 |
| Arthralgia | 14.2 | 16.1 |
| Pain in extremity | 13.0 | 13.1 |
| Back pain | 12.8 | 12.3 |
| Cough | 12.1 | 7.4 |
| Urinary tract infection | 10.7 | 10.1 |
| Diarrhea | 10.7 | 10.3 |
| Depression | 10.5 | 9.7 |
| Headache | 10.3 | 9.3 |
| Insulin glargine, % (n=174) |
NPH, % (n=175) |
|
|---|---|---|
| Infection Body system not specified
|
13.8 | 17.7 |
| Upper respiratory tract infection | 13.8 | 16.0 |
| Pharyngitis | 7.5 | 8.6 |
| Rhinitis | 5.2 | 5.1 |
2.2 General Dosing Instructions
- Individualize and adjust the dosage of Insulin glargine based on the individual's metabolic needs, blood glucose monitoring results and glycemic control goal.
- Dosage adjustments may be needed with changes in physical activity, changes in meal patterns (i.e., macronutrient content or timing of food intake), during acute illness, or changes in renal or hepatic function. Dosage adjustments should only be made under medical supervision with appropriate glucose monitoring [see Warnings and Precautions (5.2)].
14.1 Overview of Clinical Studies
The safety and effectiveness of Insulin glargine given once-daily at bedtime was compared to that of once-daily and twice-daily NPH insulin in open-label, randomized, active-controlled, parallel studies of 2,327 adult patients and 349 pediatric patients with type 1 diabetes mellitus and 1,563 adult patients with type 2 diabetes mellitus (see Tables 9–11). In general, the reduction in glycated hemoglobin (HbA1c) with Insulin glargine was similar to that with NPH insulin.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Product: 50090-6178
NDC: 50090-6178-0 3 mL in a SYRINGE / 5 in a CARTON
2.1 Important Administration Instructions
- Administer Insulin glargine subcutaneously once daily at any time of day but at the same time every day.
- Prior to initiation of Insulin glargine, train patients on proper use and injection technique.
- Patient should follow the Instructions for Use to correctly administer Insulin glargine.
- Administer Insulin glargine subcutaneously into the abdominal area, thigh, or deltoid, and rotate injection sites within the same region from one injection to the next to reduce the risk of lipodystrophy and localized cutaneous amyloidosis. Do not inject into areas of lipodystrophy or localized cutaneous amyloidosis [see Warnings and Precautions (5.2), Adverse Reactions (6)].
- During changes to a patient's insulin regimen, increase the frequency of blood glucose monitoring [see Warnings and Precautions (5.2)].
- Visually inspect Insulin glargine vials and SoloStar prefilled pens for particulate matter and discoloration prior to administration. Only use if the solution is clear and colorless with no visible particles.
- The Insulin glargine SoloStar prefilled pen dials in 1-unit increments.
- Use Insulin glargine SoloStar prefilled pen with caution in patients with visual impairment who may rely on audible clicks to dial their dose.
- Refrigerate unused (unopened) Insulin glargine vials and SoloStar® prefilled pens.
- Do not administer intravenously or via an insulin pump.
- Do not dilute or mix Insulin glargine with any other insulin or solution.
- The SoloStar prefilled pen is for single patient use only [see Warnings and Precautions (5.1)].
5.5 Hypersensitivity and Allergic Reactions
Severe, life-threatening, generalized allergy, including anaphylaxis, can occur with insulin products, including Insulin glargine. If hypersensitivity reactions occur, discontinue Insulin glargine; treat per standard of care and monitor until symptoms and signs resolve [see Adverse Reactions (6.1)]. Insulin glargine is contraindicated in patients who have had hypersensitivity reactions to insulin glargine or one of the excipients [see Contraindications (4)].
14.3 Clinical Studies in Adults With Type 2 Diabetes (14.3 Clinical Studies in Adults with Type 2 Diabetes)
In a randomized, controlled clinical study (Study E) (n=570), Insulin glargine was evaluated for 52 weeks in combination with oral antidiabetic medications (a sulfonylurea, metformin, acarbose, or combinations of these drugs). The average age was 59.5 years. The majority of patients were White (92.8%) and 53.7% were male. The mean BMI was approximately 29.1 kg/m2. The mean duration of diabetes was 10.3 years. Insulin glargine administered once daily at bedtime was as effective as NPH insulin administered once daily at bedtime in reducing HbA1c and fasting glucose (Table 11). The rate of severe symptomatic hypoglycemia was similar in Insulin glargine and NPH insulin treated patients [see Adverse Reactions (6.1)].
In a randomized, controlled clinical study (Study F), in patients with type 2 diabetes not using oral antidiabetic medications (n=518), a basal-bolus regimen of Insulin glargine once daily at bedtime or NPH insulin administered once or twice daily was evaluated for 28 weeks. Regular human insulin was used before meals, as needed. The average age was 59.3 years. The majority of patients were White (80.7%) and 60% were male. The mean BMI was approximately 30.5 kg/m2. The mean duration of diabetes was 13.7 years. Insulin glargine had similar effectiveness as either once- or twice-daily NPH insulin in reducing HbA1c and fasting glucose (Table 11) with a similar incidence of hypoglycemia [see Adverse Reactions (6.1)].
In a randomized, controlled clinical study (Study G), patients with type 2 diabetes were randomized to 5 years of treatment with once-daily Insulin glargine or twice-daily NPH insulin. For patients not previously treated with insulin, the starting dose of Insulin glargine or NPH insulin was 10 units daily. Patients who were already treated with NPH insulin either continued on the same total daily NPH insulin dose or started Insulin glargine at a dose that was 80% of the total previous NPH insulin dose. The primary endpoint for this study was a comparison of the progression of diabetic retinopathy by 3 or more steps on the Early Treatment Diabetic Retinopathy Study (ETDRS) scale. HbA1c change from baseline was a secondary endpoint. Similar glycemic control in the 2 treatment groups was desired in order to not confound the interpretation of the retinal data. Patients or study personnel used an algorithm to adjust the Insulin glargine and NPH insulin doses to a target fasting plasma glucose ≤100 mg/dL. After the Insulin glargine or NPH insulin dose was adjusted, other antidiabetic agents, including premeal insulin were to be adjusted or added. The average age was 55.1 years. The majority of patients were White (85.3%) and 53.9% were male. The mean BMI was approximately 34.3 kg/m2. The mean duration of diabetes was 10.8 years. The Insulin glargine group had a smaller mean reduction from baseline in HbA1c compared to the NPH insulin group, which may be explained by the lower daily basal insulin doses in the Insulin glargine group (Table 11). The incidences of severe symptomatic hypoglycemia were similar between groups [see Adverse Reactions (6.1)].
| Study E | Study F | Study G | ||||
|---|---|---|---|---|---|---|
| Treatment duration | 52 weeks | 28 weeks | 5 years | |||
| Treatment in combination with | Oral agents | Regular insulin | Regular insulin | |||
| Insulin glargine | NPH | Insulin glargine | NPH | Insulin glargine | NPH | |
| Number of subjects treated | 289 | 281 | 259 | 259 | 513 | 504 |
| HbA1c | ||||||
| Baseline mean | 9.0 | 8.9 | 8.6 | 8.5 | 8.4 | 8.3 |
| Adjusted mean change from baseline | -0.5 | -0.4 | -0.4 | -0.6 | -0.6 | -0.8 |
| Insulin glargine – NPH | -0.1 | +0.2 | +0.2 | |||
| 95% CI for Treatment difference | (-0.3; +0.1) | (0.0; +0.4) | (+0.1; +0.4) | |||
|
Basal insulin dose In Study G, the baseline dose of basal or total insulin was the first available on-treatment dose prescribed during the study (on visit month 1.5).
|
||||||
| Baseline mean | 14 | 15 | 44.1 | 45.5 | 39 | 44 |
| Mean change from baseline | +12 | +9 | -1 | +7 | +23 | +30 |
| Total insulin dose | ||||||
| Baseline mean | 14 | 15 | 64 | 67 | 48 | 53 |
| Mean change from baseline | +12 | +9 | +10 | +13 | +41 | +40 |
| Fasting blood glucose (mg/dL) | ||||||
| Baseline mean | 179 | 180 | 164 | 166 | 190 | 180 |
| Adj. mean change from baseline | -49 | -46 | -24 | -22 | -45 | -44 |
| Body weight (kg) | ||||||
| Baseline mean | 83.5 | 82.1 | 89.6 | 90.7 | 100 | 99 |
| Adj. mean change from baseline | 2.0 | 1.9 | 0.4 | 1.4 | 3.7 | 4.8 |
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In mice and rats, standard two-year carcinogenicity studies with insulin glargine were performed at doses up to 0.455 mg/kg, which was for the rat approximately 65 times the recommended human subcutaneous starting dose of 0.2 units/kg/day (0.007 mg/kg/day) on a mg/kg basis. Histiocytomas were found at injection sites in male rats and mice in acid vehicle containing groups and are considered a response to chronic tissue irritation and inflammation in rodents. These tumors were not found in female animals, in saline control, or insulin comparator groups using a different vehicle.
Insulin glargine was not mutagenic in tests for detection of gene mutations in bacteria and mammalian cells (Ames and HGPRT-test) and in tests for detection of chromosomal aberrations (cytogenetics in vitro in V79 cells and in vivo in Chinese hamsters).
In a combined fertility and prenatal and postnatal study in male and female rats at subcutaneous doses up to 0.36 mg/kg/day, which was approximately 50 times the recommended human subcutaneous starting dose of 0.2 units/kg/day (0.007 mg/kg/day) maternal toxicity due to dose-dependent hypoglycemia, including some deaths, was observed. Consequently, a reduction of the rearing rate occurred in the high-dose group only. Similar effects were observed with NPH insulin.
2.4 Changing to Insulin Glargine From Other Insulin Therapies (2.4 Changing to Insulin Glargine from Other Insulin Therapies)
- If changing patients from once-daily TOUJEO (insulin glargine) 300 units/mL to once-daily Insulin glargine, the recommended initial Insulin glargine dose is 80% of the TOUJEO dose that is being discontinued. This dose reduction will lower the likelihood of hypoglycemia [see Warnings and Precautions (5.3)].
- If changing from a treatment regimen with an intermediate or long-acting insulin to a regimen with Insulin glargine, a change in the dose of the basal insulin may be required and the amount and timing of the shorter-acting insulins and doses of any oral antidiabetic drugs may need to be adjusted.
- If changing patients from once-daily NPH insulin to once-daily Insulin glargine, the recommended initial Insulin glargine dose is the same as the dose of NPH that is being discontinued.
- If changing patients from twice-daily NPH insulin to once-daily Insulin glargine, the recommended initial Insulin glargine dosage is 80% of the total NPH dose that is being discontinued. This dosage reduction will lower the likelihood of hypoglycemia [see Warnings and Precautions (5.3)].
5.2 Hyperglycemia Or Hypoglycemia With Changes in Insulin Regimen (5.2 Hyperglycemia or Hypoglycemia with Changes in Insulin Regimen)
Changes in an insulin regimen (e.g., insulin strength, manufacturer, type, injection site or method of administration) may affect glycemic control and predispose to hypoglycemia [see Warnings and Precautions (5.3)] or hyperglycemia. Repeated insulin injections into areas of lipodystrophy or localized cutaneous amyloidosis have been reported to result in hyperglycemia; and a sudden change in the injection site (to unaffected area) has been reported to result in hypoglycemia [see Adverse Reactions (6)].
Make any changes to a patient's insulin regimen under close medical supervision with increased frequency of blood glucose monitoring. Advise patients who have repeatedly injected into areas of lipodystrophy or localized cutaneous amyloidosis to change the injection site to unaffected areas and closely monitor for hypoglycemia. For patients with type 2 diabetes, dosage adjustments of concomitant oral and antidiabetic products may be needed.
14.2 Clinical Studies in Adult and Pediatric Patients With Type 1 Diabetes (14.2 Clinical Studies in Adult and Pediatric Patients with Type 1 Diabetes)
In two clinical studies (Studies A and B), patients with type 1 diabetes (Study A n=585, Study B n=534) were randomized to 28 weeks of basal-bolus treatment with Insulin glargine or NPH insulin. Regular human insulin was administered before each meal. Insulin glargine was administered at bedtime. NPH insulin was administered either as once daily at bedtime or in the morning and at bedtime when used twice daily.
In Study A, the average age was 39.2 years. The majority of patients were White (99%) and 55.7% were male. The mean BMI was approximately 24.9 kg/m2. The mean duration of diabetes was 15.5 years.
In Study B, the average age was 38.5 years. The majority of patients were White (95.3%) and 50.6% were male. The mean BMI was approximately 25.8 kg/m2. The mean duration of diabetes was 17.4 years.
In another clinical study (Study C), patients with type 1 diabetes (n=619) were randomized to 16 weeks of basal-bolus treatment with Insulin glargine or NPH insulin. Insulin lispro was used before each meal. Insulin glargine was administered once daily at bedtime and NPH insulin was administered once or twice daily. The average age was 39.2 years. The majority of patients were White (96.9%) and 50.6% were male. The mean BMI was approximately 25.6 kg/m2. The mean duration of diabetes was 18.5 years.
In these 3 studies, Insulin glargine and NPH insulin had similar effects on HbA1c (Table 9) with a similar overall rate of severe symptomatic hypoglycemia [see Adverse Reactions (6.1)].
| Study A | Study B | Study C | ||||
|---|---|---|---|---|---|---|
| Treatment duration | 28 weeks | 28 weeks | 16 weeks | |||
| Treatment in combination with | Regular insulin | Regular insulin | Insulin lispro | |||
| Insulin glargine | NPH | Insulin glargine | NPH | Insulin glargine | NPH | |
| Number of subjects treated | 292 | 293 | 264 | 270 | 310 | 309 |
| HbA1c | ||||||
| Baseline HbA1c | 8.0 | 8.0 | 7.7 | 7.7 | 7.6 | 7.7 |
| Adjusted mean change at trial end | +0.2 | +0.1 | -0.2 | -0.2 | -0.1 | -0.1 |
| Treatment Difference (95% CI) | +0.1 (0.0; +0.2) | +0.1 (-0.1; +0.2) | 0.0 (-0.1; +0.1) | |||
| Basal insulin dose | ||||||
| Baseline mean | 21 | 23 | 29 | 29 | 28 | 28 |
| Mean change from baseline | -2 | 0 | -4 | +2 | -5 | +1 |
| Total insulin dose | ||||||
| Baseline mean | 48 | 52 | 50 | 51 | 50 | 50 |
| Mean change from baseline | -1 | 0 | 0 | +4 | -3 | 0 |
| Fasting blood glucose (mg/dL) | ||||||
| Baseline mean | 167 | 166 | 166 | 175 | 175 | 173 |
| Adj. mean change from baseline | -21 | -16 | -20 | -17 | -29 | -12 |
| Body weight (kg) | ||||||
| Baseline mean | 73.2 | 74.8 | 75.5 | 75.0 | 74.8 | 75.6 |
| Mean change from baseline | 0.1 | -0.0 | 0.7 | 1.0 | 0.1 | 0.5 |
5.7 Fluid Retention and Heart Failure With Concomitant Use of Ppar Gamma Agonists (5.7 Fluid Retention and Heart Failure with Concomitant Use of PPAR-gamma Agonists)
Thiazolidinediones (TZDs), which are peroxisome proliferator-activated receptor (PPAR)-gamma agonists, can cause dose-related fluid retention, particularly when used in combination with insulin. Fluid retention may lead to or exacerbate heart failure. Patients treated with insulin, including Insulin glargine, and a PPAR-gamma agonist should be observed for signs and symptoms of heart failure. If heart failure develops, it should be managed according to current standards of care, and discontinuation or dose reduction of the PPAR-gamma agonist must be considered.
5.1 Never Share An Insulin Glargine Solostar Prefilled Pen, Syringe, Or Needle Between Patients (5.1 Never Share an Insulin Glargine SoloStar Prefilled Pen, Syringe, or Needle Between Patients)
Insulin glargine SoloStar prefilled pens must never be shared between patients, even if the needle is changed. Patients using Insulin glargine vials must never re-use or share needles or syringes with another person. Sharing poses a risk for transmission of blood-borne pathogens.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:39:13.037965 · Updated: 2026-03-14T21:56:58.197015