These Highlights Do Not Include All The Information Needed To Use Famotidine Injection Safely And Effectively. See Full Prescribing Information For Famotidine Injection.
9710995d-b518-40ce-bc2b-8b5dcf002c31
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Famotidine Injection is indicated for use in hospitalized adults, or as an alternative to oral famotidine in adults, for the treatment of: active duodenal ulcer (DU). active gastric ulcer (GU). symptomatic nonerosive gastroesophageal reflux disease (GERD). erosive esophagitis due to GERD, diagnosed by endoscopy. treatment of pathological hypersecretory conditions (e.g., Zollinger-Ellison syndrome, multiple endocrine neoplasias). reduction of the risk of duodenal ulcer recurrence.
Indications and Usage
Famotidine Injection is indicated for use in hospitalized adults, or as an alternative to oral famotidine in adults, for the treatment of: active duodenal ulcer (DU). active gastric ulcer (GU). symptomatic nonerosive gastroesophageal reflux disease (GERD). erosive esophagitis due to GERD, diagnosed by endoscopy. treatment of pathological hypersecretory conditions (e.g., Zollinger-Ellison syndrome, multiple endocrine neoplasias). reduction of the risk of duodenal ulcer recurrence.
Dosage and Administration
Administration Information ( 2.1 ) Famotidine Injection is intended for use in adult and pediatric hospitalized patients, or as an alternative to oral famotidine. Discontinue as soon as the patient is able to tolerate oral treatment and switch to an appropriate oral medication. Recommended Dosage ( 2.2 ) Adults: 20 mg every 12 hours. For pathological hypersecretory conditions titrate the dosage to individual patient needs. Pediatric Patients 1 year of age and older: 0.25 mg/kg every 12 hours; titrate to a maximum of 0.5 mg/kg every 12 hours (up to maximum of 20 mg every 12 hours) based on clinical response and/or gastric pH determination and endoscopy. Administer as an intravenous injection over at least 2 minutes or an intravenous infusion over 15 minutes to 30 minutes. Refer to the prescribing information for oral famotidine products for the recommended duration of famotidine treatment. Renal Impairment ( 2.3 ) See the full prescribing information for the recommended dosage for adult patients with moderate or severe renal impairment.
Warnings and Precautions
Central Nervous System (CNS) Adverse Reactions : Reported in elderly patients and patients with moderate and severe renal impairment; monitor elderly patients for CNS adverse reactions. ( 5.1 , 8.5 , 8.6 ) Concurrent GI Malignancy : Absence of GI symptoms does not preclude the presence of gastric malignancy; evaluate prior to initiating therapy. ( 5.2 ) Risk of Benzyl Alcohol Toxicity in Neonates : Famotidine Injection is not approved in neonates. Serious and fatal adverse reactions have been reported in low-birth weight and preterm neonates who received benzyl-alcohol-containing drugs intravenously. The minimum amount of benzyl alcohol at which these serious adverse reactions may occur is not known. ( 5.3 )
Contraindications
Famotidine Injection is contraindicated in patients with a history of serious hypersensitivity reactions (e.g., anaphylaxis) to famotidine or other H 2 -receptor antagonists.
Adverse Reactions
Most common adverse reactions (>1%) are: headache, dizziness, constipation, and diarrhea. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Sagent Pharmaceuticals at 1-866-625-1618 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Drug Interactions
Drugs Dependent on Gastric pH for Absorption : Systemic exposure of the concomitant drug may be significantly reduced leading to loss of efficacy. See the prescribing information for other drugs dependent on gastric pH for absorption. ( 7.1 ) Tizanidine (CYP1A2) Substrate : Potential for substantial increases in blood concentrations of tizanidine resulting in hypotension, bradycardia or excessive drowsiness; avoid concomitant use, if possible. ( 7.2 )
Storage and Handling
Famotidine Injection is a clear, colorless, sterile solution supplied premixed and ready-to-use as follows: Famotidine Injection (Preservative-free) NDC (4 mg per mL) Package Factor 25021-755-05 20 mg per 5 mL Single-Dose Vial 25 vials per carton Famotidine Injection (Preservative) NDC (4 mg per mL) Package Factor 25021-756-10 40 mg per 10 mL Multi-Dose Vial 10 vials per carton 25021-756-50 200 mg per 50 mL Multi-Dose Vial 1 vial per carton
How Supplied
Famotidine Injection is a clear, colorless, sterile solution supplied premixed and ready-to-use as follows: Famotidine Injection (Preservative-free) NDC (4 mg per mL) Package Factor 25021-755-05 20 mg per 5 mL Single-Dose Vial 25 vials per carton Famotidine Injection (Preservative) NDC (4 mg per mL) Package Factor 25021-756-10 40 mg per 10 mL Multi-Dose Vial 10 vials per carton 25021-756-50 200 mg per 50 mL Multi-Dose Vial 1 vial per carton
Medication Information
Warnings and Precautions
Central Nervous System (CNS) Adverse Reactions : Reported in elderly patients and patients with moderate and severe renal impairment; monitor elderly patients for CNS adverse reactions. ( 5.1 , 8.5 , 8.6 ) Concurrent GI Malignancy : Absence of GI symptoms does not preclude the presence of gastric malignancy; evaluate prior to initiating therapy. ( 5.2 ) Risk of Benzyl Alcohol Toxicity in Neonates : Famotidine Injection is not approved in neonates. Serious and fatal adverse reactions have been reported in low-birth weight and preterm neonates who received benzyl-alcohol-containing drugs intravenously. The minimum amount of benzyl alcohol at which these serious adverse reactions may occur is not known. ( 5.3 )
Indications and Usage
Famotidine Injection is indicated for use in hospitalized adults, or as an alternative to oral famotidine in adults, for the treatment of: active duodenal ulcer (DU). active gastric ulcer (GU). symptomatic nonerosive gastroesophageal reflux disease (GERD). erosive esophagitis due to GERD, diagnosed by endoscopy. treatment of pathological hypersecretory conditions (e.g., Zollinger-Ellison syndrome, multiple endocrine neoplasias). reduction of the risk of duodenal ulcer recurrence.
Dosage and Administration
Administration Information ( 2.1 ) Famotidine Injection is intended for use in adult and pediatric hospitalized patients, or as an alternative to oral famotidine. Discontinue as soon as the patient is able to tolerate oral treatment and switch to an appropriate oral medication. Recommended Dosage ( 2.2 ) Adults: 20 mg every 12 hours. For pathological hypersecretory conditions titrate the dosage to individual patient needs. Pediatric Patients 1 year of age and older: 0.25 mg/kg every 12 hours; titrate to a maximum of 0.5 mg/kg every 12 hours (up to maximum of 20 mg every 12 hours) based on clinical response and/or gastric pH determination and endoscopy. Administer as an intravenous injection over at least 2 minutes or an intravenous infusion over 15 minutes to 30 minutes. Refer to the prescribing information for oral famotidine products for the recommended duration of famotidine treatment. Renal Impairment ( 2.3 ) See the full prescribing information for the recommended dosage for adult patients with moderate or severe renal impairment.
Contraindications
Famotidine Injection is contraindicated in patients with a history of serious hypersensitivity reactions (e.g., anaphylaxis) to famotidine or other H 2 -receptor antagonists.
Adverse Reactions
Most common adverse reactions (>1%) are: headache, dizziness, constipation, and diarrhea. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Sagent Pharmaceuticals at 1-866-625-1618 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Drug Interactions
Drugs Dependent on Gastric pH for Absorption : Systemic exposure of the concomitant drug may be significantly reduced leading to loss of efficacy. See the prescribing information for other drugs dependent on gastric pH for absorption. ( 7.1 ) Tizanidine (CYP1A2) Substrate : Potential for substantial increases in blood concentrations of tizanidine resulting in hypotension, bradycardia or excessive drowsiness; avoid concomitant use, if possible. ( 7.2 )
Storage and Handling
Famotidine Injection is a clear, colorless, sterile solution supplied premixed and ready-to-use as follows: Famotidine Injection (Preservative-free) NDC (4 mg per mL) Package Factor 25021-755-05 20 mg per 5 mL Single-Dose Vial 25 vials per carton Famotidine Injection (Preservative) NDC (4 mg per mL) Package Factor 25021-756-10 40 mg per 10 mL Multi-Dose Vial 10 vials per carton 25021-756-50 200 mg per 50 mL Multi-Dose Vial 1 vial per carton
How Supplied
Famotidine Injection is a clear, colorless, sterile solution supplied premixed and ready-to-use as follows: Famotidine Injection (Preservative-free) NDC (4 mg per mL) Package Factor 25021-755-05 20 mg per 5 mL Single-Dose Vial 25 vials per carton Famotidine Injection (Preservative) NDC (4 mg per mL) Package Factor 25021-756-10 40 mg per 10 mL Multi-Dose Vial 10 vials per carton 25021-756-50 200 mg per 50 mL Multi-Dose Vial 1 vial per carton
Description
Famotidine Injection is indicated for use in hospitalized adults, or as an alternative to oral famotidine in adults, for the treatment of: active duodenal ulcer (DU). active gastric ulcer (GU). symptomatic nonerosive gastroesophageal reflux disease (GERD). erosive esophagitis due to GERD, diagnosed by endoscopy. treatment of pathological hypersecretory conditions (e.g., Zollinger-Ellison syndrome, multiple endocrine neoplasias). reduction of the risk of duodenal ulcer recurrence.
Section 42229-5
Pediatric Patients 1 Year of Age and Older
Famotidine Injection is indicated in hospitalized pediatric patients 1 year of age and older, or as an alternative to oral famotidine in pediatric patients 1 year of age and older, for the treatment of peptic ulcer disease.
Section 44425-7
Storage Conditions
Store at room temperature between 20° to 25°C (68° to 77°F); excursions permitted between 15° and 30°C (59° and 86°F). [See USP Controlled Room Temperature.]
If not used immediately, store diluted solutions of Famotidine Injection between 2° and 8°C (36° and 46°F) for up to 48 hours [see Dosage and Administration (2.4)].
Do not freeze.
Protect from light. Retain in carton until time of use.
The container closure is not made with natural rubber latex.
Section 51945-4
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Vial Label
NDC 25021-755-05
Rx only
Famotidine Injection
20 mg per 5 mL
(4 mg per mL)
For Intravenous Use
5 mL Single-Dose Vial
Discard Unused Portion
10 Overdosage
Adverse reactions reported in cases of overdosage are similar to the adverse reactions reported with use of the recommended dosage [see Adverse Reactions (6)]. In the event of overdosage, treatment should be symptomatic and supportive. In the event of overdosage, monitor the patient, and employ supportive therapy. Due to low binding to plasma proteins, famotidine is eliminated by hemodialysis. There is limited experience on the usefulness of hemodialysis as a treatment for famotidine overdosage.
Consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdose management recommendations.
11 Description
The active ingredient in Famotidine Injection is a histamine H2-receptor antagonist.
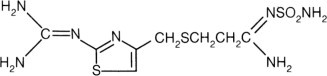
Famotidine, USP is N'-(aminosulfonyl)-3-[[[2-[ ( diaminomethylene )amino ]-4 thiazolyl]methyl]thio]propanimidamide. The empirical formula of famotidine is C8H15N7O2S3 and its molecular weight is 337.45. Its structural formula is:
Famotidine, USP is a white to pale yellow crystalline compound that is freely soluble in glacial acetic acid, slightly soluble in methanol, very slightly soluble in water, and practically insoluble in ethanol.
Famotidine Injection is supplied as a premixed, ready-to-use, sterile solution for intravenous injection. Each mL contains 4 mg of famotidine and the following inactive ingredients: L-aspartic acid 1.6 mg, mannitol 8 mg, sodium chloride 7 mg, and Water for Injection q.s. 1 mL. The multiple-dose vials of 10 mL and 50 mL also contain benzyl alcohol 0.36% (3.6 mg per mL) added as a preservative. The pH ranges for all three concentrations range from 5.7 to 6.4 and may have been adjusted with sodium hydroxide.
8.5 Geriatric Use
Of the 1,442 patients treated with oral famotidine in clinical studies, approximately 10% were 65 and older. In these studies, no overall differences in safety or effectiveness were observed between elderly and younger patients. In elderly patients, there are no clinically significant age-related changes in the pharmacokinetics of famotidine [see Clinical Pharmacology (12.3)].
In postmarketing experience, CNS adverse reactions have been reported in elderly patients with and without renal impairment receiving famotidine. Monitor elderly patients for CNS adverse reactions [see Warnings and Precautions (5.1)]. Famotidine is known to be substantially excreted by the kidney, and the risk of adverse reactions to Famotidine Injection may be greater in elderly patients, particularly those with impaired renal function [see Dosage and Administration (2.3) and Use in Specific Populations (8.6)].
4 Contraindications
Famotidine Injection is contraindicated in patients with a history of serious hypersensitivity reactions (e.g., anaphylaxis) to famotidine or other H2-receptor antagonists.
6 Adverse Reactions
Most common adverse reactions (>1%) are: headache, dizziness, constipation, and diarrhea. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Sagent Pharmaceuticals at 1-866-625-1618 or FDA at 1-800-FDA-1088 or
www.fda.gov/medwatch
.
7 Drug Interactions
- Drugs Dependent on Gastric pH for Absorption: Systemic exposure of the concomitant drug may be significantly reduced leading to loss of efficacy. See the prescribing information for other drugs dependent on gastric pH for absorption. (7.1)
- Tizanidine (CYP1A2) Substrate: Potential for substantial increases in blood concentrations of tizanidine resulting in hypotension, bradycardia or excessive drowsiness; avoid concomitant use, if possible. (7.2)
8.6 Renal Impairment
CNS adverse reactions and prolonged QT intervals have been reported in patients with moderate and severe renal impairment [see Warnings and Precautions (5.1)]. The clearance of famotidine is reduced in adults with moderate and severe renal impairment compared to adults with normal renal function [see Clinical Pharmacology (12.3)]. The recommended dosage in adults with moderate or severe renal impairment (creatinine clearance <60 mL/minutes) is less than the recommended dosage in adults with normal renal function [see Dosage and Administration (2.3)]. The recommended dosage in adults with mild renal impairment (creatinine clearance ≥60 mL/minute) is the same as the recommended dosage in adults with normal renal function.
Data are not available to establish a safe and effective dosage in pediatric patients 1 year to less than 17 years of age with renal impairment for the treatment of peptic ulcer disease.
12.3 Pharmacokinetics
Plasma levels after multiple doses are similar to those after single doses.
1 Indications and Usage
Famotidine Injection is indicated for use in hospitalized adults, or as an alternative to oral famotidine in adults, for the treatment of:
- active duodenal ulcer (DU).
- active gastric ulcer (GU).
- symptomatic nonerosive gastroesophageal reflux disease (GERD).
- erosive esophagitis due to GERD, diagnosed by endoscopy.
- treatment of pathological hypersecretory conditions (e.g., Zollinger-Ellison syndrome, multiple endocrine neoplasias).
- reduction of the risk of duodenal ulcer recurrence.
12.1 Mechanism of Action
Famotidine is a competitive inhibitor of histamine H2-receptors. The primary clinically important pharmacologic activity of famotidine is inhibition of gastric secretion. Both the acid concentration and volume of gastric secretion are suppressed by famotidine, while changes in pepsin secretion are proportional to volume output.
5 Warnings and Precautions
- Central Nervous System (CNS) Adverse Reactions: Reported in elderly patients and patients with moderate and severe renal impairment; monitor elderly patients for CNS adverse reactions. (5.1, 8.5, 8.6)
- Concurrent GI Malignancy: Absence of GI symptoms does not preclude the presence of gastric malignancy; evaluate prior to initiating therapy. (5.2)
- Risk of Benzyl Alcohol Toxicity in Neonates: Famotidine Injection is not approved in neonates. Serious and fatal adverse reactions have been reported in low-birth weight and preterm neonates who received benzyl-alcohol-containing drugs intravenously. The minimum amount of benzyl alcohol at which these serious adverse reactions may occur is not known. (5.3)
2 Dosage and Administration
Administration Information (2.1)
- Famotidine Injection is intended for use in adult and pediatric hospitalized patients, or as an alternative to oral famotidine.
- Discontinue as soon as the patient is able to tolerate oral treatment and switch to an appropriate oral medication.
Recommended Dosage (2.2)
-
Adults: 20 mg every 12 hours.
- For pathological hypersecretory conditions titrate the dosage to individual patient needs.
- Pediatric Patients 1 year of age and older: 0.25 mg/kg every 12 hours; titrate to a maximum of 0.5 mg/kg every 12 hours (up to maximum of 20 mg every 12 hours) based on clinical response and/or gastric pH determination and endoscopy.
- Administer as an intravenous injection over at least 2 minutes or an intravenous infusion over 15 minutes to 30 minutes.
- Refer to the prescribing information for oral famotidine products for the recommended duration of famotidine treatment.
Renal Impairment (2.3)
- See the full prescribing information for the recommended dosage for adult patients with moderate or severe renal impairment.
3 Dosage Forms and Strengths
Injection: a clear, colorless, solution supplied ready-to-use as:
- 20 mg/5 mL (4 mg/mL) single-dose vial
- 40 mg/10 mL (4 mg/mL) multi-dose vial
- 200 mg/50 mL (4 mg/mL) multi-dose vial
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of famotidine. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiovascular: arrhythmia, AV block, prolonged QT interval
Gastrointestinal: cholestatic jaundice, hepatitis
Hematologic: agranulocytosis, pancytopenia, leukopenia
Hypersensitivity: anaphylaxis, angioedema, facial edema, urticaria
Musculoskeletal: rhabdomyolysis, muscle cramps
Nervous System/Psychiatric: confusion, agitation, paresthesia
Respiratory: interstitial pneumonia
Skin: toxic epidermal necrolysis/Stevens-Johnson syndrome
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of Famotidine Injection has been established based on adequate and well-controlled studies of an oral famotidine product. The following is a summary of the adverse reactions reported in those studies.
Oral famotidine was studied in 7 U.S. and international placebo- and active-controlled trials in approximately 2,500 patients. A total of 1,442 patients were treated with oral famotidine, including 302 treated with 40 mg twice daily, 456 treated with 20 mg twice daily, 461 treated with 40 mg once daily, and 396 treated with 20 mg once daily. The population was 17 to 91 years old, fairly well distributed between sex and race; however, the predominant race was White.
Adverse reactions reported in ≥1% of patients treated with oral famotidine in clinical trials were: headache, dizziness, constipation, and diarrhea.
The following other adverse reactions were reported in less than 1% of patients treated with oral famotidine in clinical trials:
Body as a Whole: fever, asthenia, fatigue
Cardiovascular: palpitations
Gastrointestinal: cholestatic jaundice, elevated liver enzymes, vomiting, nausea, abdominal discomfort, anorexia, dry mouth
Hematologic: thrombocytopenia
Hypersensitivity: orbital edema, rash, conjunctival injection, bronchospasm
Musculoskeletal: musculoskeletal pain, arthralgia
Nervous System/Psychiatric: seizure, hallucinations, depression, anxiety, decreased libido, insomnia, somnolence
Respiratory: interstitial pneumonia
Skin: pruritus, dry skin, flushing
Special Senses: tinnitus, taste disorder
Other: impotence
Adverse reactions reported with oral famotidine may also occur with Famotidine Injection. In addition, transient irritation at the injection site was reported with intravenous famotidine.
5.2 Concurrent Gastric Malignancy
In adults, symptomatic response to therapy with Famotidine Injection does not preclude the presence of gastric malignancy. Consider evaluation for gastric malignancy in adult patients who have a suboptimal response or an early symptomatic relapse after completing treatment with Famotidine Injection.
7.2 Tizanidine (cyp1a2 Substrate)
Although not studied clinically, famotidine is considered a weak CYP1A2 inhibitor and may lead to substantial increases in blood concentrations of tizanidine, a CYP1A2 substrate. Avoid concomitant use with Famotidine Injection. If concomitant use is necessary, monitor for hypotension, bradycardia or excessive drowsiness. Refer to the full prescribing information for tizanidine.
16 How Supplied/storage and Handling
Famotidine Injection is a clear, colorless, sterile solution supplied premixed and ready-to-use as follows:
| Famotidine Injection (Preservative-free) | ||
| NDC | (4 mg per mL) | Package Factor |
| 25021-755-05 | 20 mg per 5 mL Single-Dose Vial | 25 vials per carton |
| Famotidine Injection (Preservative) | ||
| NDC | (4 mg per mL) | Package Factor |
| 25021-756-10 | 40 mg per 10 mL Multi-Dose Vial | 10 vials per carton |
| 25021-756-50 | 200 mg per 50 mL Multi-Dose Vial | 1 vial per carton |
2.1 Important Administration Information
- Famotidine Injection is intended for use in adult and pediatric hospitalized patients, or as an alternative to oral famotidine.
- Discontinue Famotidine Injection as soon as the patient is able to tolerate oral treatment and switch to an appropriate oral medication.
5.1 Central Nervous System Adverse Reactions
Central nervous system (CNS) adverse reactions, including confusion, delirium, hallucinations, disorientation, agitation, seizures, and lethargy, have been reported in elderly patients and patients with moderate and severe renal impairment treated with famotidine. Monitor elderly patients for CNS adverse reactions [see Use in Specific Populations (8.5)]. Dosage adjustments are recommended in adult patients with moderate and severe renal impairment due to higher famotidine systemic exposure compared to patients with normal renal function [see Dosage and Administration (2.3), Use in Specific Populations (8.6), and Clinical Pharmacology (12.3)].
5.3 Risk of Benzyl Alcohol Toxicity in Neonates
Famotidine Injection is not approved in neonates. Serious adverse reactions, including fatal reactions, of new onset or worsening metabolic acidosis that progressed to neurotoxicity, and in some cases gasping syndrome, have been reported in low-birth weight neonates (less than 2,500 grams) and preterm neonates (gestational age less than 34 weeks) who received benzyl alcohol (BA)-containing drugs intravenously. Gasping syndrome is a life-threatening condition in neonates caused by BA toxicity and is primarily characterized by multiorgan dysfunction secondary to metabolic acidosis, which leads to gasping respirations and death. The minimum amount of BA at which these serious adverse reactions, including fatal reactions, may occur is not known (Famotidine Injection contains 3.6 mg of BA per mL) [see Use in Specific Populations (8.4)].
7.1 Drugs Dependent On Gastric Ph for Absorption
Famotidine can reduce the absorption of other drugs due to its effect on reducing intragastric acidity, leading to loss of efficacy of the concomitant drug. See the prescribing information for other drugs dependent on gastric pH for absorption.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
The carcinogenic potential of intravenously administered famotidine has not been determined. In a 106-week study in rats and a 92-week study in mice given oral doses of up to 2,000 mg/kg/day there was no evidence of carcinogenic potential for famotidine.
Famotidine was negative in the bacterial mutagen test (Ames test) using Salmonella typhimurium and Escherichia coli with or without rat liver enzyme activation at concentrations up to 10,000 mcg/plate. In in vivo studies in mice, with a micronucleus test and a chromosomal aberration test, no evidence of a mutagenic effect was observed.
In studies with rats given oral doses of up to 2,000 mg/kg/day or intravenous doses of up to 200 mg/kg/day, fertility and reproductive performance were not affected.
2.2 Recommended Dosage in Adults and Pediatric Patients 1 Year of Age and Older
The recommended dosage of Famotidine Injection in adults and pediatric patients 1 year of age and older is shown in Tables 1 and 2, respectively.
Administer Famotidine Injection as an intravenous injection over at least 2 minutes or as an intravenous infusion over 15 minutes to 30 minutes [see Dosage and Administration (2.4)].
Refer to the prescribing information for oral famotidine products for the recommended duration of famotidine treatment.
|
1 Refer to the prescribing information for oral famotidine products for the recommended duration of famotidine treatment. |
|
| Indication | Recommended Dosage |
| Active Duodenal Ulcer | 20 mg every 12 hours |
| Active Gastric Ulcer | |
| Symptomatic Nonerosive GERD | |
| Erosive Esophagitis Diagnosed by Endoscopy | |
| Reduction of the Risk of Duodenal Ulcer Recurrence | |
| Pathological Hypersecretory Conditions | Starting dosage is 20 mg every 12 hours; titrate the dosage to individual patient needs |
|
1 Refer to the prescribing information for oral famotidine products for the recommended duration of famotidine treatment. |
|
| Indication | Recommended Dosage |
| Peptic Ulcer Disease |
|
Structured Label Content
Section 42229-5 (42229-5)
Pediatric Patients 1 Year of Age and Older
Famotidine Injection is indicated in hospitalized pediatric patients 1 year of age and older, or as an alternative to oral famotidine in pediatric patients 1 year of age and older, for the treatment of peptic ulcer disease.
Section 44425-7 (44425-7)
Storage Conditions
Store at room temperature between 20° to 25°C (68° to 77°F); excursions permitted between 15° and 30°C (59° and 86°F). [See USP Controlled Room Temperature.]
If not used immediately, store diluted solutions of Famotidine Injection between 2° and 8°C (36° and 46°F) for up to 48 hours [see Dosage and Administration (2.4)].
Do not freeze.
Protect from light. Retain in carton until time of use.
The container closure is not made with natural rubber latex.
Section 51945-4 (51945-4)
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Vial Label
NDC 25021-755-05
Rx only
Famotidine Injection
20 mg per 5 mL
(4 mg per mL)
For Intravenous Use
5 mL Single-Dose Vial
Discard Unused Portion
10 Overdosage (10 OVERDOSAGE)
Adverse reactions reported in cases of overdosage are similar to the adverse reactions reported with use of the recommended dosage [see Adverse Reactions (6)]. In the event of overdosage, treatment should be symptomatic and supportive. In the event of overdosage, monitor the patient, and employ supportive therapy. Due to low binding to plasma proteins, famotidine is eliminated by hemodialysis. There is limited experience on the usefulness of hemodialysis as a treatment for famotidine overdosage.
Consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdose management recommendations.
11 Description (11 DESCRIPTION)
The active ingredient in Famotidine Injection is a histamine H2-receptor antagonist.
Famotidine, USP is N'-(aminosulfonyl)-3-[[[2-[ ( diaminomethylene )amino ]-4 thiazolyl]methyl]thio]propanimidamide. The empirical formula of famotidine is C8H15N7O2S3 and its molecular weight is 337.45. Its structural formula is:
Famotidine, USP is a white to pale yellow crystalline compound that is freely soluble in glacial acetic acid, slightly soluble in methanol, very slightly soluble in water, and practically insoluble in ethanol.
Famotidine Injection is supplied as a premixed, ready-to-use, sterile solution for intravenous injection. Each mL contains 4 mg of famotidine and the following inactive ingredients: L-aspartic acid 1.6 mg, mannitol 8 mg, sodium chloride 7 mg, and Water for Injection q.s. 1 mL. The multiple-dose vials of 10 mL and 50 mL also contain benzyl alcohol 0.36% (3.6 mg per mL) added as a preservative. The pH ranges for all three concentrations range from 5.7 to 6.4 and may have been adjusted with sodium hydroxide.
8.5 Geriatric Use
Of the 1,442 patients treated with oral famotidine in clinical studies, approximately 10% were 65 and older. In these studies, no overall differences in safety or effectiveness were observed between elderly and younger patients. In elderly patients, there are no clinically significant age-related changes in the pharmacokinetics of famotidine [see Clinical Pharmacology (12.3)].
In postmarketing experience, CNS adverse reactions have been reported in elderly patients with and without renal impairment receiving famotidine. Monitor elderly patients for CNS adverse reactions [see Warnings and Precautions (5.1)]. Famotidine is known to be substantially excreted by the kidney, and the risk of adverse reactions to Famotidine Injection may be greater in elderly patients, particularly those with impaired renal function [see Dosage and Administration (2.3) and Use in Specific Populations (8.6)].
4 Contraindications (4 CONTRAINDICATIONS)
Famotidine Injection is contraindicated in patients with a history of serious hypersensitivity reactions (e.g., anaphylaxis) to famotidine or other H2-receptor antagonists.
6 Adverse Reactions (6 ADVERSE REACTIONS)
Most common adverse reactions (>1%) are: headache, dizziness, constipation, and diarrhea. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Sagent Pharmaceuticals at 1-866-625-1618 or FDA at 1-800-FDA-1088 or
www.fda.gov/medwatch
.
7 Drug Interactions (7 DRUG INTERACTIONS)
- Drugs Dependent on Gastric pH for Absorption: Systemic exposure of the concomitant drug may be significantly reduced leading to loss of efficacy. See the prescribing information for other drugs dependent on gastric pH for absorption. (7.1)
- Tizanidine (CYP1A2) Substrate: Potential for substantial increases in blood concentrations of tizanidine resulting in hypotension, bradycardia or excessive drowsiness; avoid concomitant use, if possible. (7.2)
8.6 Renal Impairment
CNS adverse reactions and prolonged QT intervals have been reported in patients with moderate and severe renal impairment [see Warnings and Precautions (5.1)]. The clearance of famotidine is reduced in adults with moderate and severe renal impairment compared to adults with normal renal function [see Clinical Pharmacology (12.3)]. The recommended dosage in adults with moderate or severe renal impairment (creatinine clearance <60 mL/minutes) is less than the recommended dosage in adults with normal renal function [see Dosage and Administration (2.3)]. The recommended dosage in adults with mild renal impairment (creatinine clearance ≥60 mL/minute) is the same as the recommended dosage in adults with normal renal function.
Data are not available to establish a safe and effective dosage in pediatric patients 1 year to less than 17 years of age with renal impairment for the treatment of peptic ulcer disease.
12.3 Pharmacokinetics
Plasma levels after multiple doses are similar to those after single doses.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Famotidine Injection is indicated for use in hospitalized adults, or as an alternative to oral famotidine in adults, for the treatment of:
- active duodenal ulcer (DU).
- active gastric ulcer (GU).
- symptomatic nonerosive gastroesophageal reflux disease (GERD).
- erosive esophagitis due to GERD, diagnosed by endoscopy.
- treatment of pathological hypersecretory conditions (e.g., Zollinger-Ellison syndrome, multiple endocrine neoplasias).
- reduction of the risk of duodenal ulcer recurrence.
12.1 Mechanism of Action
Famotidine is a competitive inhibitor of histamine H2-receptors. The primary clinically important pharmacologic activity of famotidine is inhibition of gastric secretion. Both the acid concentration and volume of gastric secretion are suppressed by famotidine, while changes in pepsin secretion are proportional to volume output.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Central Nervous System (CNS) Adverse Reactions: Reported in elderly patients and patients with moderate and severe renal impairment; monitor elderly patients for CNS adverse reactions. (5.1, 8.5, 8.6)
- Concurrent GI Malignancy: Absence of GI symptoms does not preclude the presence of gastric malignancy; evaluate prior to initiating therapy. (5.2)
- Risk of Benzyl Alcohol Toxicity in Neonates: Famotidine Injection is not approved in neonates. Serious and fatal adverse reactions have been reported in low-birth weight and preterm neonates who received benzyl-alcohol-containing drugs intravenously. The minimum amount of benzyl alcohol at which these serious adverse reactions may occur is not known. (5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Administration Information (2.1)
- Famotidine Injection is intended for use in adult and pediatric hospitalized patients, or as an alternative to oral famotidine.
- Discontinue as soon as the patient is able to tolerate oral treatment and switch to an appropriate oral medication.
Recommended Dosage (2.2)
-
Adults: 20 mg every 12 hours.
- For pathological hypersecretory conditions titrate the dosage to individual patient needs.
- Pediatric Patients 1 year of age and older: 0.25 mg/kg every 12 hours; titrate to a maximum of 0.5 mg/kg every 12 hours (up to maximum of 20 mg every 12 hours) based on clinical response and/or gastric pH determination and endoscopy.
- Administer as an intravenous injection over at least 2 minutes or an intravenous infusion over 15 minutes to 30 minutes.
- Refer to the prescribing information for oral famotidine products for the recommended duration of famotidine treatment.
Renal Impairment (2.3)
- See the full prescribing information for the recommended dosage for adult patients with moderate or severe renal impairment.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: a clear, colorless, solution supplied ready-to-use as:
- 20 mg/5 mL (4 mg/mL) single-dose vial
- 40 mg/10 mL (4 mg/mL) multi-dose vial
- 200 mg/50 mL (4 mg/mL) multi-dose vial
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of famotidine. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiovascular: arrhythmia, AV block, prolonged QT interval
Gastrointestinal: cholestatic jaundice, hepatitis
Hematologic: agranulocytosis, pancytopenia, leukopenia
Hypersensitivity: anaphylaxis, angioedema, facial edema, urticaria
Musculoskeletal: rhabdomyolysis, muscle cramps
Nervous System/Psychiatric: confusion, agitation, paresthesia
Respiratory: interstitial pneumonia
Skin: toxic epidermal necrolysis/Stevens-Johnson syndrome
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of Famotidine Injection has been established based on adequate and well-controlled studies of an oral famotidine product. The following is a summary of the adverse reactions reported in those studies.
Oral famotidine was studied in 7 U.S. and international placebo- and active-controlled trials in approximately 2,500 patients. A total of 1,442 patients were treated with oral famotidine, including 302 treated with 40 mg twice daily, 456 treated with 20 mg twice daily, 461 treated with 40 mg once daily, and 396 treated with 20 mg once daily. The population was 17 to 91 years old, fairly well distributed between sex and race; however, the predominant race was White.
Adverse reactions reported in ≥1% of patients treated with oral famotidine in clinical trials were: headache, dizziness, constipation, and diarrhea.
The following other adverse reactions were reported in less than 1% of patients treated with oral famotidine in clinical trials:
Body as a Whole: fever, asthenia, fatigue
Cardiovascular: palpitations
Gastrointestinal: cholestatic jaundice, elevated liver enzymes, vomiting, nausea, abdominal discomfort, anorexia, dry mouth
Hematologic: thrombocytopenia
Hypersensitivity: orbital edema, rash, conjunctival injection, bronchospasm
Musculoskeletal: musculoskeletal pain, arthralgia
Nervous System/Psychiatric: seizure, hallucinations, depression, anxiety, decreased libido, insomnia, somnolence
Respiratory: interstitial pneumonia
Skin: pruritus, dry skin, flushing
Special Senses: tinnitus, taste disorder
Other: impotence
Adverse reactions reported with oral famotidine may also occur with Famotidine Injection. In addition, transient irritation at the injection site was reported with intravenous famotidine.
5.2 Concurrent Gastric Malignancy
In adults, symptomatic response to therapy with Famotidine Injection does not preclude the presence of gastric malignancy. Consider evaluation for gastric malignancy in adult patients who have a suboptimal response or an early symptomatic relapse after completing treatment with Famotidine Injection.
7.2 Tizanidine (cyp1a2 Substrate) (7.2 Tizanidine (CYP1A2 Substrate))
Although not studied clinically, famotidine is considered a weak CYP1A2 inhibitor and may lead to substantial increases in blood concentrations of tizanidine, a CYP1A2 substrate. Avoid concomitant use with Famotidine Injection. If concomitant use is necessary, monitor for hypotension, bradycardia or excessive drowsiness. Refer to the full prescribing information for tizanidine.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Famotidine Injection is a clear, colorless, sterile solution supplied premixed and ready-to-use as follows:
| Famotidine Injection (Preservative-free) | ||
| NDC | (4 mg per mL) | Package Factor |
| 25021-755-05 | 20 mg per 5 mL Single-Dose Vial | 25 vials per carton |
| Famotidine Injection (Preservative) | ||
| NDC | (4 mg per mL) | Package Factor |
| 25021-756-10 | 40 mg per 10 mL Multi-Dose Vial | 10 vials per carton |
| 25021-756-50 | 200 mg per 50 mL Multi-Dose Vial | 1 vial per carton |
2.1 Important Administration Information
- Famotidine Injection is intended for use in adult and pediatric hospitalized patients, or as an alternative to oral famotidine.
- Discontinue Famotidine Injection as soon as the patient is able to tolerate oral treatment and switch to an appropriate oral medication.
5.1 Central Nervous System Adverse Reactions
Central nervous system (CNS) adverse reactions, including confusion, delirium, hallucinations, disorientation, agitation, seizures, and lethargy, have been reported in elderly patients and patients with moderate and severe renal impairment treated with famotidine. Monitor elderly patients for CNS adverse reactions [see Use in Specific Populations (8.5)]. Dosage adjustments are recommended in adult patients with moderate and severe renal impairment due to higher famotidine systemic exposure compared to patients with normal renal function [see Dosage and Administration (2.3), Use in Specific Populations (8.6), and Clinical Pharmacology (12.3)].
5.3 Risk of Benzyl Alcohol Toxicity in Neonates
Famotidine Injection is not approved in neonates. Serious adverse reactions, including fatal reactions, of new onset or worsening metabolic acidosis that progressed to neurotoxicity, and in some cases gasping syndrome, have been reported in low-birth weight neonates (less than 2,500 grams) and preterm neonates (gestational age less than 34 weeks) who received benzyl alcohol (BA)-containing drugs intravenously. Gasping syndrome is a life-threatening condition in neonates caused by BA toxicity and is primarily characterized by multiorgan dysfunction secondary to metabolic acidosis, which leads to gasping respirations and death. The minimum amount of BA at which these serious adverse reactions, including fatal reactions, may occur is not known (Famotidine Injection contains 3.6 mg of BA per mL) [see Use in Specific Populations (8.4)].
7.1 Drugs Dependent On Gastric Ph for Absorption (7.1 Drugs Dependent on Gastric pH for Absorption)
Famotidine can reduce the absorption of other drugs due to its effect on reducing intragastric acidity, leading to loss of efficacy of the concomitant drug. See the prescribing information for other drugs dependent on gastric pH for absorption.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
The carcinogenic potential of intravenously administered famotidine has not been determined. In a 106-week study in rats and a 92-week study in mice given oral doses of up to 2,000 mg/kg/day there was no evidence of carcinogenic potential for famotidine.
Famotidine was negative in the bacterial mutagen test (Ames test) using Salmonella typhimurium and Escherichia coli with or without rat liver enzyme activation at concentrations up to 10,000 mcg/plate. In in vivo studies in mice, with a micronucleus test and a chromosomal aberration test, no evidence of a mutagenic effect was observed.
In studies with rats given oral doses of up to 2,000 mg/kg/day or intravenous doses of up to 200 mg/kg/day, fertility and reproductive performance were not affected.
2.2 Recommended Dosage in Adults and Pediatric Patients 1 Year of Age and Older
The recommended dosage of Famotidine Injection in adults and pediatric patients 1 year of age and older is shown in Tables 1 and 2, respectively.
Administer Famotidine Injection as an intravenous injection over at least 2 minutes or as an intravenous infusion over 15 minutes to 30 minutes [see Dosage and Administration (2.4)].
Refer to the prescribing information for oral famotidine products for the recommended duration of famotidine treatment.
|
1 Refer to the prescribing information for oral famotidine products for the recommended duration of famotidine treatment. |
|
| Indication | Recommended Dosage |
| Active Duodenal Ulcer | 20 mg every 12 hours |
| Active Gastric Ulcer | |
| Symptomatic Nonerosive GERD | |
| Erosive Esophagitis Diagnosed by Endoscopy | |
| Reduction of the Risk of Duodenal Ulcer Recurrence | |
| Pathological Hypersecretory Conditions | Starting dosage is 20 mg every 12 hours; titrate the dosage to individual patient needs |
|
1 Refer to the prescribing information for oral famotidine products for the recommended duration of famotidine treatment. |
|
| Indication | Recommended Dosage |
| Peptic Ulcer Disease |
|
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:53:47.209019 · Updated: 2026-03-14T22:47:25.384104