Surgicept Waterless Surgical Hand Scrub & Healthcare Personnel Handwash – Alcohol Solution
96fe2cc7-e812-4d39-8f11-bd3cc7411a21
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Antiseptic
Medication Information
Purpose
Antiseptic
Dosage and Administration
Directions
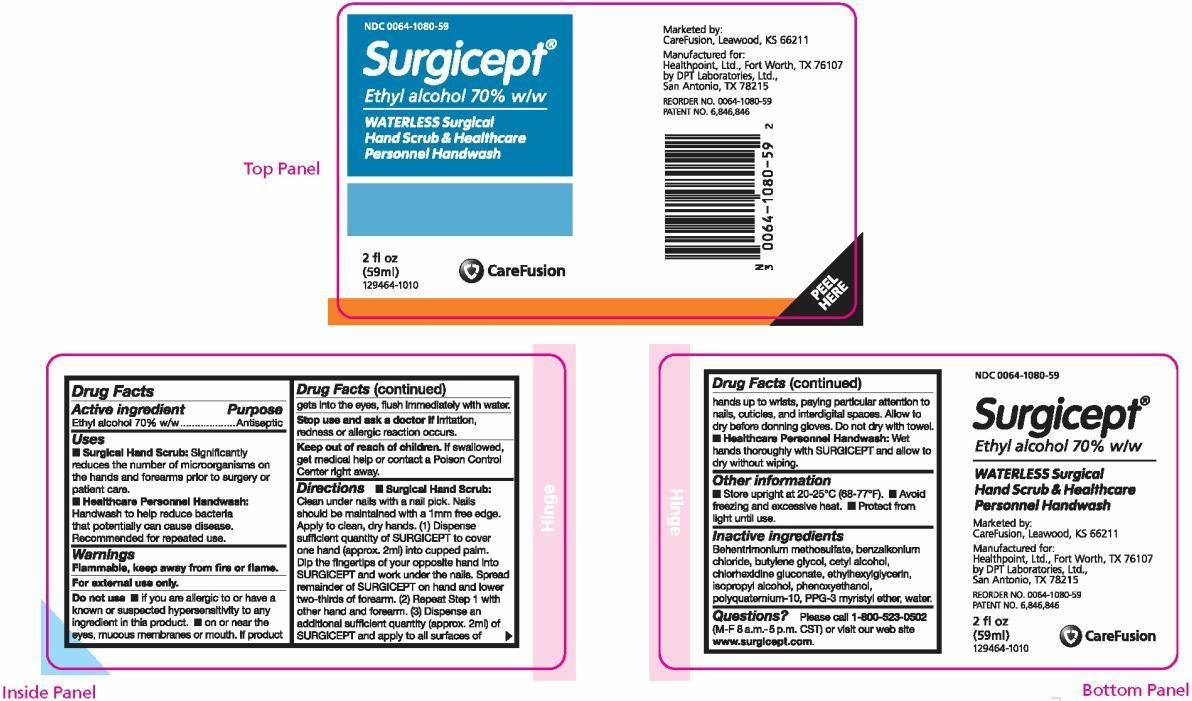
- Surgical Hand Scrub: Clean under nails with a nail pick. Nails should be maintained with a 1mm free edge. Apply to clean, dry hands. (1) Dispense sufficient quantity of SURGICEPT to cover one hand (approx. 2ml) into cupped palm. Dip the fingertips of your opposite hand into SURGICEPT and work under the nails. Spread remainder of SURGICEPT on hand and lower two-thirds of forearm. (2) Repeat Step 1 with other hand and forearm. (3) Dispense an additional sufficient quantity (approx. 2ml) of SURGICEPT and apply to all surfaces of hands up to wrists, paying particular attention to nails, cuticles, and interdigital spaces. Allow to dry before donning gloves. Do not dry with a towel.
- Healthcare Personnel Handwash: Wet hands thoroughly with SURGICEPT and allow to dry without wiping.
Description
Drug Facts
Uses
- Surgical Hand Scrub: Significantly reduces the number of microorganisms on the hands and forearms prior to surgery or patient care.
- Healthcare Personnel Handwash: Handwash to help reduce bacteria that potentially can cause disease. Recommended for repeated use.
Section 42229-5
Drug Facts
Section 50565-1
Other information
- Store upright at 20-25°C (68-77°F)
- Avoid freezing and excessive heat.
- Protect from light until use.
Warnings
- Flammable, keep away from fire or flame.
- For external use only.
-
Do not use
- if you are allergic to or have a known or suspected hypersensitivity to any ingredient in this product.
- on or near the eyes, mucous membranes or mouth. If product gets into the eyes, flush immediately with water.
- Stop use and ask a doctor if irritation, redness or allergic reaction occurs.
- Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Questions?
Please call 1-800-523-0502 (M-F 8 a.m.-5 p.m. CST) or visit our web site www.surgicept.com.
PRINCIPAL DISPLAY PANEL
NDC 0064-1080-59
Surgicept ®
Ethyl alcohol 70% w/w
WATERLESS Surgical Hand Scrub & Healthcare Personnel Handwash
2 fl oz (59 ml)
129464-1010
Marketed by:
CareFusion, Leawood, KS 66211
Manufactured for:
Healthpoint, Ltd., Fort Worth, TX 76107
by DPT Laboratories, Ltd., San Antonio, TX 78215
REORDER NO. 0064-1080-59
PATENT NO. 6,846,846
Active Ingredient
Ethyl alcohol 70% w/w
Inactive Ingredients
Behentrimonium methosulfate, benzalkonium chloride, butylene glycol, cetyl alcohol, chlorhexidine gluconate, ethylhexylglycerin, isopropyl alcohol, phenoxyethanol, polyquaternium-10, PPG-3 myristyl ether, water.
Package Label.principal Display Panel
Structured Label Content
Uses
- Surgical Hand Scrub: Significantly reduces the number of microorganisms on the hands and forearms prior to surgery or patient care.
- Healthcare Personnel Handwash: Handwash to help reduce bacteria that potentially can cause disease. Recommended for repeated use.
Dosage and Administration (34068-7)
Directions
- Surgical Hand Scrub: Clean under nails with a nail pick. Nails should be maintained with a 1mm free edge. Apply to clean, dry hands. (1) Dispense sufficient quantity of SURGICEPT to cover one hand (approx. 2ml) into cupped palm. Dip the fingertips of your opposite hand into SURGICEPT and work under the nails. Spread remainder of SURGICEPT on hand and lower two-thirds of forearm. (2) Repeat Step 1 with other hand and forearm. (3) Dispense an additional sufficient quantity (approx. 2ml) of SURGICEPT and apply to all surfaces of hands up to wrists, paying particular attention to nails, cuticles, and interdigital spaces. Allow to dry before donning gloves. Do not dry with a towel.
- Healthcare Personnel Handwash: Wet hands thoroughly with SURGICEPT and allow to dry without wiping.
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Other information
- Store upright at 20-25°C (68-77°F)
- Avoid freezing and excessive heat.
- Protect from light until use.
Purpose
Antiseptic
Warnings
- Flammable, keep away from fire or flame.
- For external use only.
-
Do not use
- if you are allergic to or have a known or suspected hypersensitivity to any ingredient in this product.
- on or near the eyes, mucous membranes or mouth. If product gets into the eyes, flush immediately with water.
- Stop use and ask a doctor if irritation, redness or allergic reaction occurs.
- Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Questions?
Please call 1-800-523-0502 (M-F 8 a.m.-5 p.m. CST) or visit our web site www.surgicept.com.
PRINCIPAL DISPLAY PANEL
NDC 0064-1080-59
Surgicept ®
Ethyl alcohol 70% w/w
WATERLESS Surgical Hand Scrub & Healthcare Personnel Handwash
2 fl oz (59 ml)
129464-1010
Marketed by:
CareFusion, Leawood, KS 66211
Manufactured for:
Healthpoint, Ltd., Fort Worth, TX 76107
by DPT Laboratories, Ltd., San Antonio, TX 78215
REORDER NO. 0064-1080-59
PATENT NO. 6,846,846
Active Ingredient (Active ingredient)
Ethyl alcohol 70% w/w
Inactive Ingredients (Inactive ingredients)
Behentrimonium methosulfate, benzalkonium chloride, butylene glycol, cetyl alcohol, chlorhexidine gluconate, ethylhexylglycerin, isopropyl alcohol, phenoxyethanol, polyquaternium-10, PPG-3 myristyl ether, water.
Package Label.principal Display Panel (PACKAGE LABEL.PRINCIPAL DISPLAY PANEL)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:24.863466 · Updated: 2026-03-14T22:54:51.534540