jafra daily skin brightening antiperspirant roll-on
96669750-7355-4992-a44c-d25d2164cd9f
34390-5
HUMAN OTC DRUG LABEL
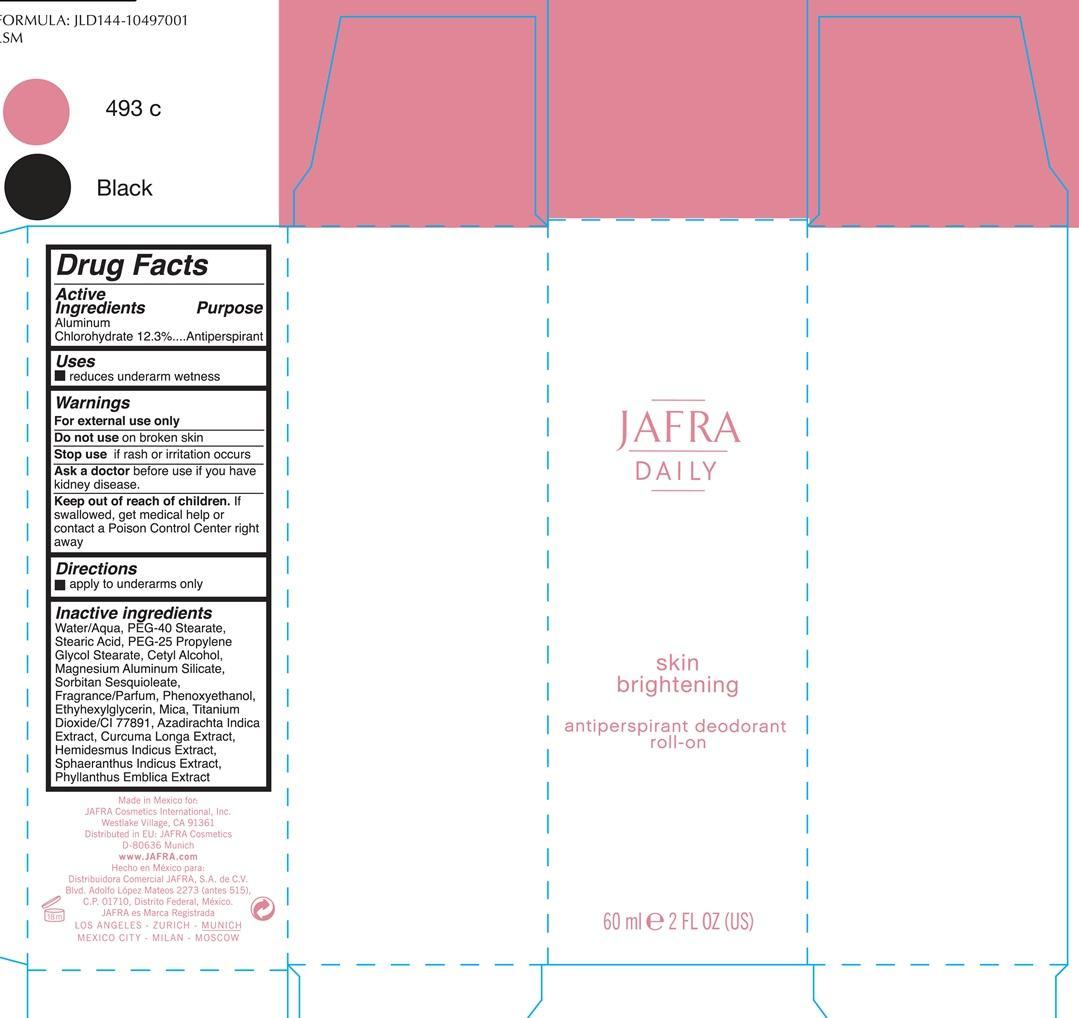
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredients Purpose Aluminum Chlorohydrate 12.3% Antiperspirant
Medication Information
Warnings and Precautions
Warnings
For external use only
Do not use on broken skin
Ask a doctor before before use if you have kidney disease
Active Ingredient
Active ingredients Purpose
Aluminum Chlorohydrate 12.3% Antiperspirant
Indications and Usage
Stop use if rash or irritation occurs
Dosage and Administration
Directions
Apply to underarms only
Description
Active ingredients Purpose Aluminum Chlorohydrate 12.3% Antiperspirant
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 51727-6
Water/Aqua, PEG-40 Stearate, PEG-25 Propylene Glycol Stearate, Cetyl Alcohol, Magnesium Aluminum Silicate, Sorbitan Sesquioleate, Fragrance/parfum, Phenoxyethanol, Ehtylhexylhexyl Glycerin, Mica, Titanium Dioxide/CI 77891, Azadirachta Indica Extract, Curcuma Longa, Hemisdesmus Indicus, Sphaeranthus Indicus Extract, Phyllanthus Emblica Extract
Section 51945-4
Jafra
Daily
Skin Brightening
antiperspirant deodorant roll-on
60 ml 2 FL OZ
Section 55105-1
Uses
reduces underarm wetness
Structured Label Content
Warnings and Precautions (34071-1)
Warnings
For external use only
Do not use on broken skin
Ask a doctor before before use if you have kidney disease
Active Ingredient (55106-9)
Active ingredients Purpose
Aluminum Chlorohydrate 12.3% Antiperspirant
Indications and Usage (34067-9)
Stop use if rash or irritation occurs
Dosage and Administration (34068-7)
Directions
Apply to underarms only
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 51727-6 (51727-6)
Water/Aqua, PEG-40 Stearate, PEG-25 Propylene Glycol Stearate, Cetyl Alcohol, Magnesium Aluminum Silicate, Sorbitan Sesquioleate, Fragrance/parfum, Phenoxyethanol, Ehtylhexylhexyl Glycerin, Mica, Titanium Dioxide/CI 77891, Azadirachta Indica Extract, Curcuma Longa, Hemisdesmus Indicus, Sphaeranthus Indicus Extract, Phyllanthus Emblica Extract
Section 51945-4 (51945-4)
Jafra
Daily
Skin Brightening
antiperspirant deodorant roll-on
60 ml 2 FL OZ
Section 55105-1 (55105-1)
Uses
reduces underarm wetness
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:44.868128 · Updated: 2026-03-14T22:55:25.290849