These Highlights Do Not Include All The Information Needed To Use Folic Acid Oral Solution Safely And Effectively. See Full Prescribing Information For Quiofic.
96618bf8-6c03-4df3-9896-b7ef27ca392a
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
FOLIC ACID oral solution is indicated for the treatment of megaloblastic anemias due to folic acid deficiency in adult and pediatric patients.
Indications and Usage
FOLIC ACID oral solution is indicated for the treatment of megaloblastic anemias due to folic acid deficiency in adult and pediatric patients.
Dosage and Administration
Recommended starting dosage in adults and pediatric patients (regardless of age) is up to 1 mg orally daily. ( 2 ) Maintenance dosage ( 2 ) - Pediatric patients birth to 23 months: 0.1 mg orally daily - Pediatric patients 2 years to less than 4 years: up to 0.3 mg orally daily - Adults and pediatric patients 4 years and older: 0.4 mg orally daily - Pregnant and Lactating Women: 0.8 mg orally daily; but never less than 0.1 mg orally per day
Warnings and Precautions
Folic acid in doses above 0.1 mg daily may obscure pernicious anemia in that hematologic remission can occur while neurologic manifestations remain progressive. This may result in severe nervous system damage before the correct diagnosis is made. ( 5.1 )
Contraindications
FOLIC ACID oral solution is contraindicated in patients with a history of a hypersensitivity reaction to folic acid or any of the ingredients of QUIOFIC [see Description (11) ].
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Risk of Obscuring Diagnosis of Pernicious Anemia [see Warnings and Precautions (5.1) ] The following adverse reactions associated with the use of folic acid were identified in clinical studies or post marketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Adverse Reactions: Hypersensitivity reactions including rash, itching, malaise, and bronchospasm. Gastrointestinal reactions (nausea, anorexia, abdominal distension, flatulence, dysgeusia) Neurological reactions (altered sleep patterns, difficulty concentrating, irritability, overactivity, excitement, depression, confusion, impaired judgement) Decreased vitamin B 12 serum levels (with prolonged folic acid therapy). Increased seizures in patients with epilepsy receiving phenobarbital, primidone, or diphenylhydantoin
Drug Interactions
Anticonvulsant action of phenytoin is antagonized by folic acid. ( 7 ) Multiple drug classes, including anticonvulsants, antibiotic/antimicrobial agents, folate antagonists, GI-binding agents, and oral contraceptives, may reduce folic acid absorption or folate levels. ( 7 )
Storage and Handling
FOLIC ACID Oral Solution, 0.2 mg/mL is a yellow solution with a mixed berry flavor supplied in amber PET bottles with a child-resistant closure containing 75 mL of oral solution (NDC 69499-501-75). Store at 20°C to 25°C (68°F to 77°F) [see USP Controlled Room Temperature]. Store and dispense in the original container to protect from light. Discard unused portion 30 days after first opening.
How Supplied
FOLIC ACID Oral Solution, 0.2 mg/mL is a yellow solution with a mixed berry flavor supplied in amber PET bottles with a child-resistant closure containing 75 mL of oral solution (NDC 69499-501-75). Store at 20°C to 25°C (68°F to 77°F) [see USP Controlled Room Temperature]. Store and dispense in the original container to protect from light. Discard unused portion 30 days after first opening.
Medication Information
Warnings and Precautions
Folic acid in doses above 0.1 mg daily may obscure pernicious anemia in that hematologic remission can occur while neurologic manifestations remain progressive. This may result in severe nervous system damage before the correct diagnosis is made. ( 5.1 )
Indications and Usage
FOLIC ACID oral solution is indicated for the treatment of megaloblastic anemias due to folic acid deficiency in adult and pediatric patients.
Dosage and Administration
Recommended starting dosage in adults and pediatric patients (regardless of age) is up to 1 mg orally daily. ( 2 ) Maintenance dosage ( 2 ) - Pediatric patients birth to 23 months: 0.1 mg orally daily - Pediatric patients 2 years to less than 4 years: up to 0.3 mg orally daily - Adults and pediatric patients 4 years and older: 0.4 mg orally daily - Pregnant and Lactating Women: 0.8 mg orally daily; but never less than 0.1 mg orally per day
Contraindications
FOLIC ACID oral solution is contraindicated in patients with a history of a hypersensitivity reaction to folic acid or any of the ingredients of QUIOFIC [see Description (11) ].
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Risk of Obscuring Diagnosis of Pernicious Anemia [see Warnings and Precautions (5.1) ] The following adverse reactions associated with the use of folic acid were identified in clinical studies or post marketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Adverse Reactions: Hypersensitivity reactions including rash, itching, malaise, and bronchospasm. Gastrointestinal reactions (nausea, anorexia, abdominal distension, flatulence, dysgeusia) Neurological reactions (altered sleep patterns, difficulty concentrating, irritability, overactivity, excitement, depression, confusion, impaired judgement) Decreased vitamin B 12 serum levels (with prolonged folic acid therapy). Increased seizures in patients with epilepsy receiving phenobarbital, primidone, or diphenylhydantoin
Drug Interactions
Anticonvulsant action of phenytoin is antagonized by folic acid. ( 7 ) Multiple drug classes, including anticonvulsants, antibiotic/antimicrobial agents, folate antagonists, GI-binding agents, and oral contraceptives, may reduce folic acid absorption or folate levels. ( 7 )
Storage and Handling
FOLIC ACID Oral Solution, 0.2 mg/mL is a yellow solution with a mixed berry flavor supplied in amber PET bottles with a child-resistant closure containing 75 mL of oral solution (NDC 69499-501-75). Store at 20°C to 25°C (68°F to 77°F) [see USP Controlled Room Temperature]. Store and dispense in the original container to protect from light. Discard unused portion 30 days after first opening.
How Supplied
FOLIC ACID Oral Solution, 0.2 mg/mL is a yellow solution with a mixed berry flavor supplied in amber PET bottles with a child-resistant closure containing 75 mL of oral solution (NDC 69499-501-75). Store at 20°C to 25°C (68°F to 77°F) [see USP Controlled Room Temperature]. Store and dispense in the original container to protect from light. Discard unused portion 30 days after first opening.
Description
FOLIC ACID oral solution is indicated for the treatment of megaloblastic anemias due to folic acid deficiency in adult and pediatric patients.
Section 42229-5
Folic acid may interfere with gastrointestinal absorption of methotrexate. Folic acid therapy in folate-deficient individuals may decrease serum levels of phenytoin. Folic acid may also interfere with the absorption and effectiveness of antibiotic tetracycline.
Folic acid supplements are usually avoided on the day of oral methotrexate administration. Generally, the time of administration of these drugs should be separated from folic acid.
10 Overdosage
Consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdose management recommendations.
11 Description
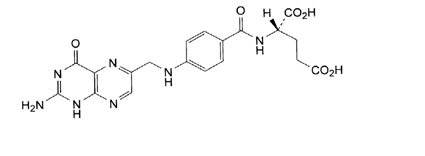
Folic acid, N-[p-[[(2-amino-4- hydroxy-6-pteridinyl) methyl]- amino] benzoyl]-L glutamic acid, is a B complex vitamin containing a pteridine moiety linked by a methylene bridge to para-aminobenzoic acid, which is joined by a peptide linkage to glutamic acid. Conjugates of folic acid are present in a wide variety of foods, particularly liver, kidneys, yeast, and leafy green vegetables. Commercially available folic acid is prepared synthetically. Folic acid, USP occurs as a yellow or yellowish-orange crystalline powder and is very slightly soluble in water and insoluble in alcohol. Folic acid, USP is readily soluble in dilute solutions of alkali hydroxides and carbonates, and solutions of the drug may be prepared with the aid of sodium hydroxide or sodium carbonate, thereby forming the soluble sodium salt of folic acid (sodium folate). Aqueous solutions of folic acid are heat sensitive and rapidly decompose in the presence of light and/or riboflavin; solutions should be stored in a cool place protected from light.
The structural formula of folic acid is as follows:
M.W. 441.40
Each 1 mL of solution, for oral administration, contains 0.2 mg folic acid, USP.
FOLIC ACID Oral Solution, 0.2 mg/mL contains the following inactive ingredients: edetate disodium, methylparaben, mixed berry flavor, propylene glycol, propylparaben, purified water, sodium phosphate dibasic, sodium phosphate monobasic, and sorbitol solution.
4 Contraindications
FOLIC ACID oral solution is contraindicated in patients with a history of a hypersensitivity reaction to folic acid or any of the ingredients of QUIOFIC [see Description (11)].
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Risk of Obscuring Diagnosis of Pernicious Anemia [see Warnings and Precautions (5.1)]
The following adverse reactions associated with the use of folic acid were identified in clinical studies or post marketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Adverse Reactions:
- Hypersensitivity reactions including rash, itching, malaise, and bronchospasm.
- Gastrointestinal reactions (nausea, anorexia, abdominal distension, flatulence, dysgeusia)
- Neurological reactions (altered sleep patterns, difficulty concentrating, irritability, overactivity, excitement, depression, confusion, impaired judgement)
- Decreased vitamin B12 serum levels (with prolonged folic acid therapy).
- Increased seizures in patients with epilepsy receiving phenobarbital, primidone, or diphenylhydantoin
7 Drug Interactions
12.3 Pharmacokinetics
Absorption
Folic acid is absorbed from the small intestine, primarily from the proximal portion. Folic acid appears in the plasma approximately 15 to 30 minutes after an oral dose; peak levels are generally reached within 1 hour.
Distribution
Folic acid as well as its physiologically active metabolite, L-5-methyl tetrahydro folate (L-5-MTHF), can be bound to plasma proteins when entering the systemic circulation. The fraction of folic acid bound to plasma proteins, particularly albumin, ranges between ~50% to 64%. The volume of distribution of L-5-MTHF is estimated to be 32.0 L. Tetrahydrofolic acid derivatives are distributed to all body tissues but are stored primarily in the liver. Cerebrospinal fluid levels of folic acid are several times greater than serum levels of the drug.
Metabolism
The first step in the metabolism of folic acid is its reduction to dihydrofolate via dihydrofolate reductase. Subsequently, dihydrofolate is further reduced to tetrahydrofolate (THF). THF is further converted to methylene-THF by serine-hydroxymethyltransferase and reduced to physiologically active L-5-MTHF via methylenetetrahydrofolate reductase (MTHFR).
Elimination
After intravenous administration, folic acid is rapidly cleared from the plasma. Following oral administration, majority of the dose was recovered in the urine. A majority of the metabolic products appeared in the urine after 6 hours; excretion was generally complete within 24 hours. Small amounts of orally administered folic acid have also been recovered in the feces.
2.2 Recommended Dosing
Initial Dosing
The recommended starting dosage of FOLIC ACID oral solution in pediatric and adult patients is up to 1 mg orally daily. FOLIC ACID oral solution can be taken with or without food. Rule out pernicious anemia prior to use of any doses greater than 0.4 mg (except during pregnancy and lactation).
Maintenance Dosing
When clinical symptoms have subsided and the blood picture has become normal, use a daily maintenance level as follows:
- Pediatric patients aged birth to 23 months: 0.1 mg orally daily
- Pediatric patients aged 2 years to less than 4 years: up to 0.3 mg orally daily
- Pediatric patients aged 4 years and older and adult patients: 0.4 mg orally daily
- Pregnant and Lactating Women: 0.8 mg orally daily; but never less than 0.1 mg orally per day
Higher maintenance doses may be needed in the presence of alcoholism, hemolytic anemia, anticonvulsant therapy, or chronic infection.
Monitor patients frequently for relapse and adjust dose accordingly.
1 Indications and Usage
FOLIC ACID oral solution is indicated for the treatment of megaloblastic anemias due to folic acid deficiency in adult and pediatric patients.
12.1 Mechanism of Action
Folic acid acts on megaloblastic bone marrow to produce a normoblastic marrow.
In man, an exogenous source of folate is required for nucleoprotein synthesis and the maintenance of normal erythropoiesis. Folic acid is the precursor of tetrahydrofolic acid, which is involved as a cofactor for transformylation reactions in the biosynthesis of purines and thymidylates of nucleic acids. Impairment of thymidylate synthesis in patients with folic acid deficiency is thought to account for the defective deoxyribonucleic acid (DNA) synthesis that leads to megaloblast formation and megaloblastic and macrocytic anemias.
5 Warnings and Precautions
- Folic acid in doses above 0.1 mg daily may obscure pernicious anemia in that hematologic remission can occur while neurologic manifestations remain progressive. This may result in severe nervous system damage before the correct diagnosis is made. (5.1)
2 Dosage and Administration
- Recommended starting dosage in adults and pediatric patients (regardless of age) is up to 1 mg orally daily. (2)
- Maintenance dosage (2)
- Pediatric patients birth to 23 months: 0.1 mg orally daily
- Pediatric patients 2 years to less than 4 years: up to 0.3 mg orally daily
- Adults and pediatric patients 4 years and older: 0.4 mg orally daily
- Pregnant and Lactating Women: 0.8 mg orally daily; but never less than 0.1 mg orally per day
3 Dosage Forms and Strengths
Oral Solution: 0.2 mg/mL; yellow solution with a mixed berry flavor.
17 Patient Counseling Information
Physicians should advise patients and caregivers about the following for safe use of FOLIC ACID oral solution:
- Advise patients to report signs or symptoms of hypersensitivity reaction [see Contraindications (4)].
- Instruct patients and caregivers to use an oral dosing syringe to correctly measure the prescribed amount of medication.
- Inform patients that oral dosing syringes may be obtained from their pharmacy.
Manufactured for: Ayurax, LLC
Madisonville, LA 70447
3105 R0126
16 How Supplied/storage and Handling
FOLIC ACID Oral Solution, 0.2 mg/mL is a yellow solution with a mixed berry flavor supplied in amber PET bottles with a child-resistant closure containing 75 mL of oral solution (NDC 69499-501-75).
Store at 20°C to 25°C (68°F to 77°F) [see USP Controlled Room Temperature].
Store and dispense in the original container to protect from light.
Discard unused portion 30 days after first opening.
Principal Display Panel Bottle Label
NDC 73308-501-75
Rx Only
FOLIC ACID ORAL SOLUTION
0.2 mg/mL
75 mL
2.1 Important Administration Information
Instruct patients or caregivers to use an oral dosing syringe to correctly measure the prescribed amount of medication. Inform patients that oral dosing syringes may be obtained from their pharmacy.
5.1 Risk of Obscuring Diagnosis of Pernicious Anemia
The use of single-agent FOLIC ACID oral solution (without the use of vitamin B12) is not recommended for the treatment of pernicious anemia and other megaloblastic anemias in which vitamin B12 is deficient.
Folic acid in doses above 0.1 mg daily may obscure pernicious anemia in that hematologic remission can occur while neurologic manifestations remain progressive. There is a potential danger in administering folic acid to patients with undiagnosed anemia, since folic acid may obscure the diagnosis of pernicious anemia by alleviating the hematologic manifestations of the disease while allowing the neurologic complications to progress. This may result in severe nervous system damage before the correct diagnosis is made. Adequate doses of vitamin B12 may prevent, halt, or improve the neurologic changes caused by pernicious anemia.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals to evaluate carcinogenic potential and studies to evaluate the mutagenic potential or effect on fertility have not been conducted.
Structured Label Content
Section 42229-5 (42229-5)
Folic acid may interfere with gastrointestinal absorption of methotrexate. Folic acid therapy in folate-deficient individuals may decrease serum levels of phenytoin. Folic acid may also interfere with the absorption and effectiveness of antibiotic tetracycline.
Folic acid supplements are usually avoided on the day of oral methotrexate administration. Generally, the time of administration of these drugs should be separated from folic acid.
10 Overdosage (10 OVERDOSAGE)
Consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdose management recommendations.
11 Description (11 DESCRIPTION)
Folic acid, N-[p-[[(2-amino-4- hydroxy-6-pteridinyl) methyl]- amino] benzoyl]-L glutamic acid, is a B complex vitamin containing a pteridine moiety linked by a methylene bridge to para-aminobenzoic acid, which is joined by a peptide linkage to glutamic acid. Conjugates of folic acid are present in a wide variety of foods, particularly liver, kidneys, yeast, and leafy green vegetables. Commercially available folic acid is prepared synthetically. Folic acid, USP occurs as a yellow or yellowish-orange crystalline powder and is very slightly soluble in water and insoluble in alcohol. Folic acid, USP is readily soluble in dilute solutions of alkali hydroxides and carbonates, and solutions of the drug may be prepared with the aid of sodium hydroxide or sodium carbonate, thereby forming the soluble sodium salt of folic acid (sodium folate). Aqueous solutions of folic acid are heat sensitive and rapidly decompose in the presence of light and/or riboflavin; solutions should be stored in a cool place protected from light.
The structural formula of folic acid is as follows:
M.W. 441.40
Each 1 mL of solution, for oral administration, contains 0.2 mg folic acid, USP.
FOLIC ACID Oral Solution, 0.2 mg/mL contains the following inactive ingredients: edetate disodium, methylparaben, mixed berry flavor, propylene glycol, propylparaben, purified water, sodium phosphate dibasic, sodium phosphate monobasic, and sorbitol solution.
4 Contraindications (4 CONTRAINDICATIONS)
FOLIC ACID oral solution is contraindicated in patients with a history of a hypersensitivity reaction to folic acid or any of the ingredients of QUIOFIC [see Description (11)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Risk of Obscuring Diagnosis of Pernicious Anemia [see Warnings and Precautions (5.1)]
The following adverse reactions associated with the use of folic acid were identified in clinical studies or post marketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Adverse Reactions:
- Hypersensitivity reactions including rash, itching, malaise, and bronchospasm.
- Gastrointestinal reactions (nausea, anorexia, abdominal distension, flatulence, dysgeusia)
- Neurological reactions (altered sleep patterns, difficulty concentrating, irritability, overactivity, excitement, depression, confusion, impaired judgement)
- Decreased vitamin B12 serum levels (with prolonged folic acid therapy).
- Increased seizures in patients with epilepsy receiving phenobarbital, primidone, or diphenylhydantoin
7 Drug Interactions (7 DRUG INTERACTIONS)
12.3 Pharmacokinetics
Absorption
Folic acid is absorbed from the small intestine, primarily from the proximal portion. Folic acid appears in the plasma approximately 15 to 30 minutes after an oral dose; peak levels are generally reached within 1 hour.
Distribution
Folic acid as well as its physiologically active metabolite, L-5-methyl tetrahydro folate (L-5-MTHF), can be bound to plasma proteins when entering the systemic circulation. The fraction of folic acid bound to plasma proteins, particularly albumin, ranges between ~50% to 64%. The volume of distribution of L-5-MTHF is estimated to be 32.0 L. Tetrahydrofolic acid derivatives are distributed to all body tissues but are stored primarily in the liver. Cerebrospinal fluid levels of folic acid are several times greater than serum levels of the drug.
Metabolism
The first step in the metabolism of folic acid is its reduction to dihydrofolate via dihydrofolate reductase. Subsequently, dihydrofolate is further reduced to tetrahydrofolate (THF). THF is further converted to methylene-THF by serine-hydroxymethyltransferase and reduced to physiologically active L-5-MTHF via methylenetetrahydrofolate reductase (MTHFR).
Elimination
After intravenous administration, folic acid is rapidly cleared from the plasma. Following oral administration, majority of the dose was recovered in the urine. A majority of the metabolic products appeared in the urine after 6 hours; excretion was generally complete within 24 hours. Small amounts of orally administered folic acid have also been recovered in the feces.
2.2 Recommended Dosing
Initial Dosing
The recommended starting dosage of FOLIC ACID oral solution in pediatric and adult patients is up to 1 mg orally daily. FOLIC ACID oral solution can be taken with or without food. Rule out pernicious anemia prior to use of any doses greater than 0.4 mg (except during pregnancy and lactation).
Maintenance Dosing
When clinical symptoms have subsided and the blood picture has become normal, use a daily maintenance level as follows:
- Pediatric patients aged birth to 23 months: 0.1 mg orally daily
- Pediatric patients aged 2 years to less than 4 years: up to 0.3 mg orally daily
- Pediatric patients aged 4 years and older and adult patients: 0.4 mg orally daily
- Pregnant and Lactating Women: 0.8 mg orally daily; but never less than 0.1 mg orally per day
Higher maintenance doses may be needed in the presence of alcoholism, hemolytic anemia, anticonvulsant therapy, or chronic infection.
Monitor patients frequently for relapse and adjust dose accordingly.
1 Indications and Usage (1 INDICATIONS AND USAGE)
FOLIC ACID oral solution is indicated for the treatment of megaloblastic anemias due to folic acid deficiency in adult and pediatric patients.
12.1 Mechanism of Action
Folic acid acts on megaloblastic bone marrow to produce a normoblastic marrow.
In man, an exogenous source of folate is required for nucleoprotein synthesis and the maintenance of normal erythropoiesis. Folic acid is the precursor of tetrahydrofolic acid, which is involved as a cofactor for transformylation reactions in the biosynthesis of purines and thymidylates of nucleic acids. Impairment of thymidylate synthesis in patients with folic acid deficiency is thought to account for the defective deoxyribonucleic acid (DNA) synthesis that leads to megaloblast formation and megaloblastic and macrocytic anemias.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Folic acid in doses above 0.1 mg daily may obscure pernicious anemia in that hematologic remission can occur while neurologic manifestations remain progressive. This may result in severe nervous system damage before the correct diagnosis is made. (5.1)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Recommended starting dosage in adults and pediatric patients (regardless of age) is up to 1 mg orally daily. (2)
- Maintenance dosage (2)
- Pediatric patients birth to 23 months: 0.1 mg orally daily
- Pediatric patients 2 years to less than 4 years: up to 0.3 mg orally daily
- Adults and pediatric patients 4 years and older: 0.4 mg orally daily
- Pregnant and Lactating Women: 0.8 mg orally daily; but never less than 0.1 mg orally per day
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Oral Solution: 0.2 mg/mL; yellow solution with a mixed berry flavor.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Physicians should advise patients and caregivers about the following for safe use of FOLIC ACID oral solution:
- Advise patients to report signs or symptoms of hypersensitivity reaction [see Contraindications (4)].
- Instruct patients and caregivers to use an oral dosing syringe to correctly measure the prescribed amount of medication.
- Inform patients that oral dosing syringes may be obtained from their pharmacy.
Manufactured for: Ayurax, LLC
Madisonville, LA 70447
3105 R0126
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
FOLIC ACID Oral Solution, 0.2 mg/mL is a yellow solution with a mixed berry flavor supplied in amber PET bottles with a child-resistant closure containing 75 mL of oral solution (NDC 69499-501-75).
Store at 20°C to 25°C (68°F to 77°F) [see USP Controlled Room Temperature].
Store and dispense in the original container to protect from light.
Discard unused portion 30 days after first opening.
Principal Display Panel Bottle Label (PRINCIPAL DISPLAY PANEL - Bottle Label)
NDC 73308-501-75
Rx Only
FOLIC ACID ORAL SOLUTION
0.2 mg/mL
75 mL
2.1 Important Administration Information
Instruct patients or caregivers to use an oral dosing syringe to correctly measure the prescribed amount of medication. Inform patients that oral dosing syringes may be obtained from their pharmacy.
5.1 Risk of Obscuring Diagnosis of Pernicious Anemia
The use of single-agent FOLIC ACID oral solution (without the use of vitamin B12) is not recommended for the treatment of pernicious anemia and other megaloblastic anemias in which vitamin B12 is deficient.
Folic acid in doses above 0.1 mg daily may obscure pernicious anemia in that hematologic remission can occur while neurologic manifestations remain progressive. There is a potential danger in administering folic acid to patients with undiagnosed anemia, since folic acid may obscure the diagnosis of pernicious anemia by alleviating the hematologic manifestations of the disease while allowing the neurologic complications to progress. This may result in severe nervous system damage before the correct diagnosis is made. Adequate doses of vitamin B12 may prevent, halt, or improve the neurologic changes caused by pernicious anemia.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals to evaluate carcinogenic potential and studies to evaluate the mutagenic potential or effect on fertility have not been conducted.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T02:18:21.754661 · Updated: 2026-03-14T22:49:56.242164