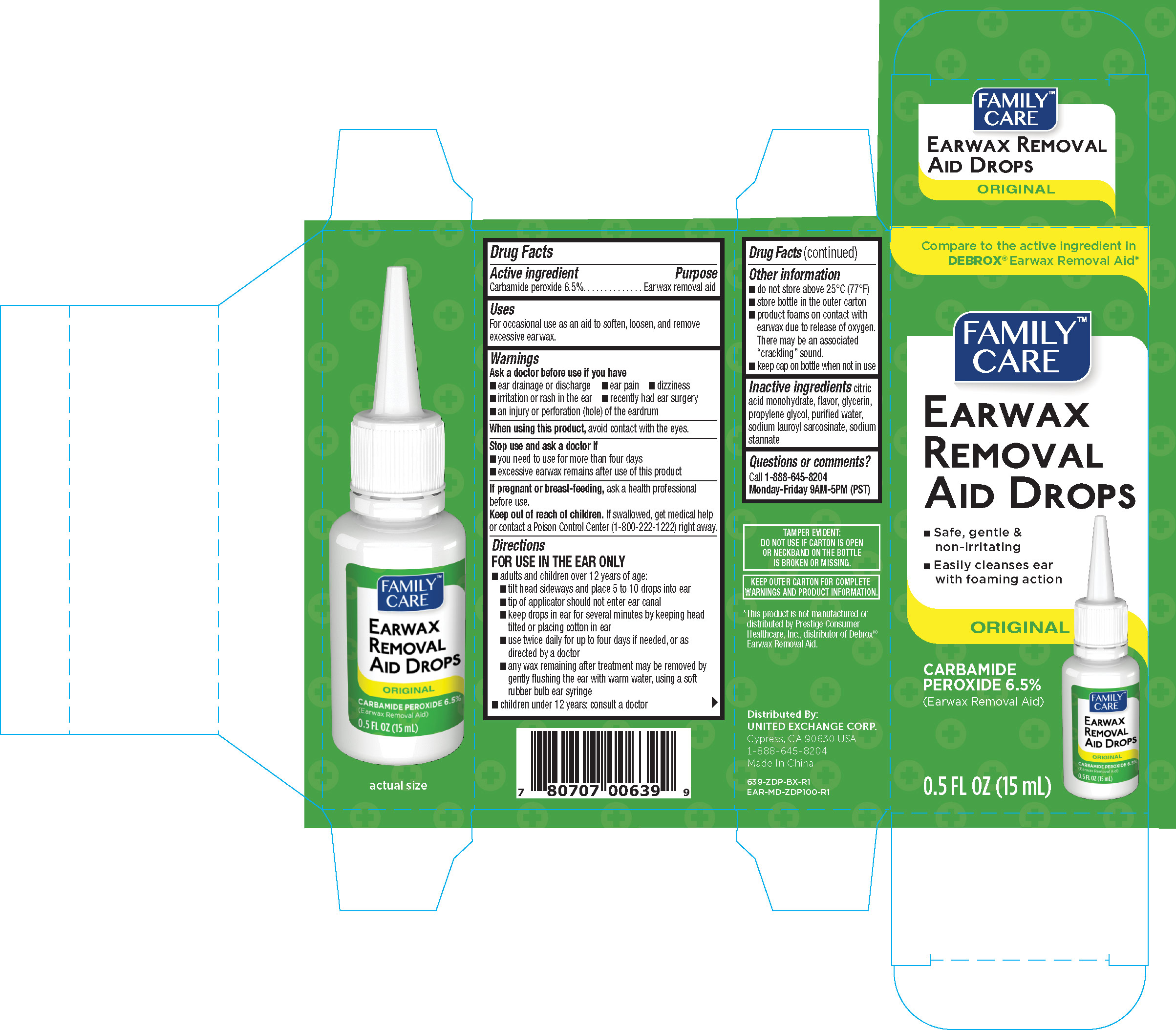
family care earwax removal aid drops 0.5 oz 639 zdp
958741f5-47c1-339c-e053-2a95a90a3f5f
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredient Purpose Carbamide peroxide 6.5%.................................Ear wax removal
Medication Information
Warnings and Precautions
Warnings
Ask a doctor before use if you have
- ear drainage or discharge
- ear pain
- dizziness
- irritation or rash in the ear
- recently had ear surgery
- an injury or perforation (hole) of the eardrum
Active Ingredient
Active ingredient Purpose
Carbamide peroxide 6.5%.................................Ear wax removal
Indications and Usage
Directions
FOR USE IN THE EAR ONLY
- adults and children over 12 years of age:
- tilt head sideways and place 5 to 10 drops into ear
- tip of applicator should not enter ear canal
- keep drops in ear for several minutes by keeping head tilted or placing cotton in ear
- use twice daily for up to four days if needed, or as directed by a doctor
- any way remaining after treatment may be removed by gently flushing the ear with warm water, using a soft rubber bulb ear syringe
- children under 12 years: consult a doctor
Dosage and Administration
Distributed by:
United Exchange Corp.
Cypress, CA 90630
Made in China
Description
Active ingredient Purpose Carbamide peroxide 6.5%.................................Ear wax removal
Section 44425-7
Other information
- do not store above 25°C (77°F)
- store bottle in the outer carton
- product foams on contact with earwax due to release of oxygen. There may be associated "crackling" sound
- keep cap on bottle when not in use
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Section 50566-9
Stop use and ask a doctor if
- you need to use for more than four days
- excessive earwax remains after use of this product
Section 51727-6
Inactive ingredients
citric acid monohydrate, flavor, glycerin, propylene glycol, purified water, sodium lauroyl sarcosinate, sodium stannate
Section 51945-4
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1
Uses
For occasional use as an aid to soften, loosen, and remove excessive ear wax.
Structured Label Content
Warnings and Precautions (34071-1)
Warnings
Ask a doctor before use if you have
- ear drainage or discharge
- ear pain
- dizziness
- irritation or rash in the ear
- recently had ear surgery
- an injury or perforation (hole) of the eardrum
Active Ingredient (55106-9)
Active ingredient Purpose
Carbamide peroxide 6.5%.................................Ear wax removal
Indications and Usage (34067-9)
Directions
FOR USE IN THE EAR ONLY
- adults and children over 12 years of age:
- tilt head sideways and place 5 to 10 drops into ear
- tip of applicator should not enter ear canal
- keep drops in ear for several minutes by keeping head tilted or placing cotton in ear
- use twice daily for up to four days if needed, or as directed by a doctor
- any way remaining after treatment may be removed by gently flushing the ear with warm water, using a soft rubber bulb ear syringe
- children under 12 years: consult a doctor
Dosage and Administration (34068-7)
Distributed by:
United Exchange Corp.
Cypress, CA 90630
Made in China
Section 44425-7 (44425-7)
Other information
- do not store above 25°C (77°F)
- store bottle in the outer carton
- product foams on contact with earwax due to release of oxygen. There may be associated "crackling" sound
- keep cap on bottle when not in use
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if
- you need to use for more than four days
- excessive earwax remains after use of this product
Section 51727-6 (51727-6)
Inactive ingredients
citric acid monohydrate, flavor, glycerin, propylene glycol, purified water, sodium lauroyl sarcosinate, sodium stannate
Section 51945-4 (51945-4)
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1 (55105-1)
Uses
For occasional use as an aid to soften, loosen, and remove excessive ear wax.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:03.339672 · Updated: 2026-03-14T23:13:11.905913