65517-0001 - Alcohol Prep Pads
953b0dd6-a667-4146-815e-6845aba0309b
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Medication Information
Warnings and Precautions
Warnings
- For External Use Only
- Flammable, Keep away from fire or flame
Indications and Usage
Apply topically as needed to cleanse
Dosage and Administration
Directions
Wipe injection site vigorously and discard.
Description
Drug Facts
Section 42229-5
Drug Facts
Section 44425-7
Other Information
Store at Room Temperature 15 - 30 C (59 - 86 F)
Section 50565-1
Keep out of reach of children
If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop Use
If irritation and redness develop. If condition persists, consult your health care practitioner.
Section 50570-1
Do Not Use
- with electrocautery procedures
- In the Eyes. If contact occurs, flush eyes with water
Section 51727-6
Inactive Ingredient
purified water
Section 51945-4
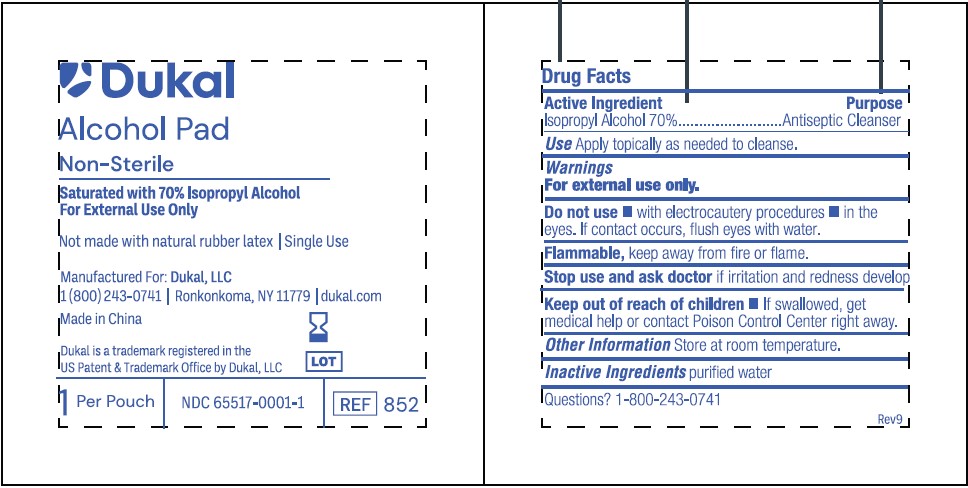
Dukal
Alcohol Pad
Non-Sterile
Saturated with 70% Isopropyl Alcohol
For External Use Only
Not made with natural rubber latex / Single Use
Manufactured For: Dukal, LLC
1(800)243-0741 /Ronkonkoma, NY 11779 / dukal.com
Made in China
Dukal is a trademark registered in the
US Patent & Trademark Office by Dukal, LLC
1 Per Pouch / NDC 65517-0001-1 / REF 852
Section 55105-1
Purpose
Antiseptic Cleanser
Section 55106-9
Active Ingredients
Isopropyl Alcohol 70%
Indications & Usage Section
Updated to match the label claim
Structured Label Content
Indications and Usage (34067-9)
Apply topically as needed to cleanse
Dosage and Administration (34068-7)
Directions
Wipe injection site vigorously and discard.
Warnings and Precautions (34071-1)
Warnings
- For External Use Only
- Flammable, Keep away from fire or flame
Section 42229-5 (42229-5)
Drug Facts
Section 44425-7 (44425-7)
Other Information
Store at Room Temperature 15 - 30 C (59 - 86 F)
Section 50565-1 (50565-1)
Keep out of reach of children
If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop Use
If irritation and redness develop. If condition persists, consult your health care practitioner.
Section 50570-1 (50570-1)
Do Not Use
- with electrocautery procedures
- In the Eyes. If contact occurs, flush eyes with water
Section 51727-6 (51727-6)
Inactive Ingredient
purified water
Section 51945-4 (51945-4)
Dukal
Alcohol Pad
Non-Sterile
Saturated with 70% Isopropyl Alcohol
For External Use Only
Not made with natural rubber latex / Single Use
Manufactured For: Dukal, LLC
1(800)243-0741 /Ronkonkoma, NY 11779 / dukal.com
Made in China
Dukal is a trademark registered in the
US Patent & Trademark Office by Dukal, LLC
1 Per Pouch / NDC 65517-0001-1 / REF 852
Section 55105-1 (55105-1)
Purpose
Antiseptic Cleanser
Section 55106-9 (55106-9)
Active Ingredients
Isopropyl Alcohol 70%
Indications & Usage Section
Updated to match the label claim
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:18.019403 · Updated: 2026-03-14T23:10:34.945513