These Highlights Do Not Include All The Information Needed To Use Dutasteride Capsules Safely And Effectively. See Full Prescribing Information For Dutasteride Capsules.
947644ea-bc35-41df-8a7a-874360c90192
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Warnings and Precautions, Evaluation for Other Urological Diseases ( 5.3 ) 03/2012
Indications and Usage
Dutasteride is a 5 alpha-reductase inhibitor indicated for the treatment of symptomatic benign prostatic hyperplasia (BPH) in men with an enlarged prostate to: ( 1.1 ) improve symptoms, reduce the risk of acute urinary retention, and reduce the risk of the need for BPH-related surgery. Dutasteride in combination with the alpha adrenergic antagonist, tamsulosin, is indicated for the treatment of symptomatic BPH in men with an enlarged prostate. ( 1.2 ) Limitations of Use: Dutasteride is not approved for the prevention of prostate cancer. ( 1.3 )
Dosage and Administration
The capsules should be swallowed whole and not chewed or opened, as contact with the capsule contents may result in irritation of the oropharyngeal mucosa. Dutasteride may be administered with or without food.
Warnings and Precautions
Dutasteride reduces serum prostate-specific antigen (PSA) concentration by approximately 50%. However, any confirmed increase in PSA while on dutasteride may signal the presence of prostate cancer and should be evaluated, even if those values are still within the normal range for untreated men. ( 5.1 ) Dutasteride may increase the risk of high-grade prostate cancer. ( 5.2 , 6.1 ) Prior to initiating treatment with dutasteride, consideration should be given to other urological conditions that may cause similar symptoms. ( 5.3 ) Women who are pregnant or could become pregnant should not handle dutasteride capsules due to potential risk to a male fetus. ( 5.4 , 8.1 ) Patients should not donate blood until 6 months after their last dose of dutasteride. ( 5.5 )
Contraindications
Dutasteride is contraindicated for use in: Pregnancy. In animal reproduction and developmental toxicity trials, dutasteride inhibited development of male fetus external genitalia. Therefore, dutasteride may cause fetal harm when administered to a pregnant woman. If dutasteride is used during pregnancy or if the patient becomes pregnant while taking dutasteride, the patient should be apprised of the potential hazard to the fetus [see Warnings and Precautions (5.4) , Use in Specific Populations (8.1) ] . Women of childbearing potential [see Warnings and Precautions (5.4) , Use in Specific Populations (8.1) ] . Pediatric patients [see Use in Specific Populations (8.4) ] . Patients with previously demonstrated clinically significant hypersensitivity (e.g., serious skin reactions, angioedema) to dutasteride or other 5 alpha-reductase inhibitors [see Adverse Reactions (6.2) ] .
Adverse Reactions
The most common adverse reactions, reported in ≥1% of patients treated with dutasteride and more commonly than in subjects treated with placebo, are impotence, decreased libido, ejaculation disorders, and breast disorders. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Banyan Pharma LLC at 1-888-328-0749 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Use with caution in patients taking potent, chronic CYP3A4 enzyme inhibitors (e.g., ritonavir). ( 7 )
Storage and Handling
Dutasteride capsules 0.5 mg are light brown, oblong, soft gelatin capsule printed with black 'AV5' and containing a clear, slightly yellow to pale amber oily liquid packaged in bottles of 30 (NDC 84091-100-30) and 90 (NDC 84091-100-90) with child-resistant closures.
How Supplied
Dutasteride capsules 0.5 mg are light brown, oblong, soft gelatin capsule printed with black 'AV5' and containing a clear, slightly yellow to pale amber oily liquid packaged in bottles of 30 (NDC 84091-100-30) and 90 (NDC 84091-100-90) with child-resistant closures.
Patient Information
Dutasteride Capsules (doo-TAS-ter-ide) Dutasteride is for use by men only. Read this patient information before you start taking dutasteride and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or your treatment. What is dutasteride? Dutasteride is a prescription medicine that contains dutasteride. Dutasteride is used to treat the symptoms of benign prostatic hyperplasia (BPH) in men with an enlarged prostate to: improve symptoms reduce the risk of acute urinary retention (a complete blockage of urine flow) reduce the risk of the need for BPH-related surgery Who should NOT take dutasteride? Do Not Take Dutasteride if you are: pregnant or could become pregnant. Dutasteride may harm your unborn baby. Pregnant women should not touch dutasteride capsules. If a woman who is pregnant with a male baby gets enough dutasteride in her body by swallowing or touching dutasteride, the male baby may be born with sex organs that are not normal. If a pregnant woman or woman of childbearing potential comes in contact with leaking dutasteride capsules, the contact area should be washed immediately with soap and water. a child or a teenager. allergic to dutasteride or any of the ingredients in dutasteride. See the end of this leaflet for a complete list of ingredients in dutasteride. allergic to other 5 alpha-reductase inhibitors, for example, finasteride tablets. What should I tell my healthcare provider before taking dutasteride? Before you take dutasteride, tell your healthcare provider if you: have liver problems Tell your healthcare provider about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements. Dutasteride and other medicines may affect each other, causing side effects. Dutasteride may affect the way other medicines work, and other medicines may affect how dutasteride works. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. How should I take dutasteride? Take 1 dutasteride capsule once a day. Swallow dutasteride capsules whole. Do not crush, chew, or open dutasteride capsules because the contents of the capsule may irritate your lips, mouth, or throat. You can take dutasteride with or without food. If you miss a dose, you may take it later that day. Do not make up the missed dose by taking 2 doses the next day. What should I avoid while taking dutasteride? You should not donate blood while taking dutasteride or for 6 months after you have stopped dutasteride. This is important to prevent pregnant women from receiving dutasteride through blood transfusions. What are the possible side effects of dutasteride? Dutasteride may cause serious side effects, including: Rare and serious allergic reactions, including: swelling of your face, tongue, or throat serious skin reactions, such as skin peeling Get medical help right away if you have these serious allergic reactions. Higher chance of a more serious form of prostate cancer. The most common side effects of dutasteride include: trouble getting or keeping an erection (impotence) Some of these events may continue after you stop taking dustasteride. a decrease in sex drive (libido) ejaculation problems enlarged or painful breasts. If you notice breast lumps or nipple discharge, you should talk to your healthcare provider. Depressed mood has been reported in patients receiving dutasteride. Dutasteride has been shown to reduce sperm count, semen volume, and sperm movement. However, the effect of dutasteride on male fertility is not known. Prostate-Specific Antigen (PSA) Test: Your healthcare provider may check you for other prostate problems, including prostate cancer before you start and while you take dutasteride. A blood test called PSA (prostate-specific antigen) is sometimes used to see if you might have prostate cancer. Dutasteride will reduce the amount of PSA measured in your blood. Your healthcare provider is aware of this effect and can still use PSA to see if you might have prostate cancer. Increases in your PSA levels while on treatment with dutasteride (even if the PSA levels are in the normal range) should be evaluated by your healthcare provider. Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects with dutasteride. For more information, ask you healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. How should I store dutasteride? Store dutasteride capsules at room temperature (59°F to 86°F or 15°C to 30°C). Dutasteride capsules may become deformed and/or discolored if kept at high temperatures. Do not use dutasteride if your capsules are deformed, discolored, or leaking. Safely throw away medicine that is no longer needed. Keep dutasteride and all medicines out of the reach of children. Medicines are sometimes prescribed for purposes other than those listed in a patient leaflet. Do not use dutasteride for a condition for which it was not prescribed. Do not give dutasteride to other people, even if they have the same symptoms that you have. It may harm them. This patient information leaflet summarizes the most important information about dutasteride. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about dutasteride that is written for health professionals. For more information, contact Banyan Pharma LLC at 1-888-328-0749. What are the ingredients in dutasteride capsules? Active ingredient: dutasteride Inactive ingredients: glycerol, gelatin, titanium dioxide, yellow iron oxide, red iron oxide, mono and di-glycerides, butylhydroxytoluene, lecithin, medium chain triglycerides and black ink. The non-volatile ingredients of the black ink are ammonium hydroxide, black iron oxide, propylene glycol and shellac. How does dutasteride work? Prostate growth is caused by a hormone in the blood called dihydrotestosterone (DHT). Dutasteride lowers DHT production in the body, leading to shrinkage of the enlarged prostate in most men. While some men have fewer problems and symptoms after 3 months of treatment with dutasteride, a treatment period of at least 6 months is usually necessary to see if dutasteride will work for you. Distributed by: Banyan Pharma LLC Hoboken, NJ, 07030 Manufacturered by: Esjay Pharma Pvt. Ltd. Made in India Revised: 12/2025
Medication Information
Warnings and Precautions
Dutasteride reduces serum prostate-specific antigen (PSA) concentration by approximately 50%. However, any confirmed increase in PSA while on dutasteride may signal the presence of prostate cancer and should be evaluated, even if those values are still within the normal range for untreated men. ( 5.1 ) Dutasteride may increase the risk of high-grade prostate cancer. ( 5.2 , 6.1 ) Prior to initiating treatment with dutasteride, consideration should be given to other urological conditions that may cause similar symptoms. ( 5.3 ) Women who are pregnant or could become pregnant should not handle dutasteride capsules due to potential risk to a male fetus. ( 5.4 , 8.1 ) Patients should not donate blood until 6 months after their last dose of dutasteride. ( 5.5 )
Indications and Usage
Dutasteride is a 5 alpha-reductase inhibitor indicated for the treatment of symptomatic benign prostatic hyperplasia (BPH) in men with an enlarged prostate to: ( 1.1 ) improve symptoms, reduce the risk of acute urinary retention, and reduce the risk of the need for BPH-related surgery. Dutasteride in combination with the alpha adrenergic antagonist, tamsulosin, is indicated for the treatment of symptomatic BPH in men with an enlarged prostate. ( 1.2 ) Limitations of Use: Dutasteride is not approved for the prevention of prostate cancer. ( 1.3 )
Dosage and Administration
The capsules should be swallowed whole and not chewed or opened, as contact with the capsule contents may result in irritation of the oropharyngeal mucosa. Dutasteride may be administered with or without food.
Contraindications
Dutasteride is contraindicated for use in: Pregnancy. In animal reproduction and developmental toxicity trials, dutasteride inhibited development of male fetus external genitalia. Therefore, dutasteride may cause fetal harm when administered to a pregnant woman. If dutasteride is used during pregnancy or if the patient becomes pregnant while taking dutasteride, the patient should be apprised of the potential hazard to the fetus [see Warnings and Precautions (5.4) , Use in Specific Populations (8.1) ] . Women of childbearing potential [see Warnings and Precautions (5.4) , Use in Specific Populations (8.1) ] . Pediatric patients [see Use in Specific Populations (8.4) ] . Patients with previously demonstrated clinically significant hypersensitivity (e.g., serious skin reactions, angioedema) to dutasteride or other 5 alpha-reductase inhibitors [see Adverse Reactions (6.2) ] .
Adverse Reactions
The most common adverse reactions, reported in ≥1% of patients treated with dutasteride and more commonly than in subjects treated with placebo, are impotence, decreased libido, ejaculation disorders, and breast disorders. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Banyan Pharma LLC at 1-888-328-0749 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Use with caution in patients taking potent, chronic CYP3A4 enzyme inhibitors (e.g., ritonavir). ( 7 )
Storage and Handling
Dutasteride capsules 0.5 mg are light brown, oblong, soft gelatin capsule printed with black 'AV5' and containing a clear, slightly yellow to pale amber oily liquid packaged in bottles of 30 (NDC 84091-100-30) and 90 (NDC 84091-100-90) with child-resistant closures.
How Supplied
Dutasteride capsules 0.5 mg are light brown, oblong, soft gelatin capsule printed with black 'AV5' and containing a clear, slightly yellow to pale amber oily liquid packaged in bottles of 30 (NDC 84091-100-30) and 90 (NDC 84091-100-90) with child-resistant closures.
Patient Information
Dutasteride Capsules (doo-TAS-ter-ide)
Dutasteride is for use by men only.
Read this patient information before you start taking dutasteride and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or your treatment.
What is dutasteride?
Dutasteride is a prescription medicine that contains dutasteride. Dutasteride is used to treat the symptoms of benign prostatic hyperplasia (BPH) in men with an enlarged prostate to:
- improve symptoms
- reduce the risk of acute urinary retention (a complete blockage of urine flow)
- reduce the risk of the need for BPH-related surgery
Who should NOT take dutasteride?
Do Not Take Dutasteride if you are:
- pregnant or could become pregnant. Dutasteride may harm your unborn baby. Pregnant women should not touch dutasteride capsules. If a woman who is pregnant with a male baby gets enough dutasteride in her body by swallowing or touching dutasteride, the male baby may be born with sex organs that are not normal. If a pregnant woman or woman of childbearing potential comes in contact with leaking dutasteride capsules, the contact area should be washed immediately with soap and water.
- a child or a teenager.
- allergic to dutasteride or any of the ingredients in dutasteride. See the end of this leaflet for a complete list of ingredients in dutasteride.
- allergic to other 5 alpha-reductase inhibitors, for example, finasteride tablets.
What should I tell my healthcare provider before taking dutasteride?
Before you take dutasteride, tell your healthcare provider if you:
- have liver problems
Tell your healthcare provider about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements. Dutasteride and other medicines may affect each other, causing side effects. Dutasteride may affect the way other medicines work, and other medicines may affect how dutasteride works.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine.
How should I take dutasteride?
- Take 1 dutasteride capsule once a day.
- Swallow dutasteride capsules whole. Do not crush, chew, or open dutasteride capsules because the contents of the capsule may irritate your lips, mouth, or throat.
- You can take dutasteride with or without food.
- If you miss a dose, you may take it later that day. Do not make up the missed dose by taking 2 doses the next day.
What should I avoid while taking dutasteride?
- You should not donate blood while taking dutasteride or for 6 months after you have stopped dutasteride. This is important to prevent pregnant women from receiving dutasteride through blood transfusions.
What are the possible side effects of dutasteride?
Dutasteride may cause serious side effects, including:
-
Rare and serious allergic reactions, including:
- swelling of your face, tongue, or throat
- serious skin reactions, such as skin peeling
Get medical help right away if you have these serious allergic reactions.
- Higher chance of a more serious form of prostate cancer.
The most common side effects of dutasteride include:
- trouble getting or keeping an erection (impotence)Some of these events may continue after you stop taking dustasteride.
- a decrease in sex drive (libido)
- ejaculation problems
- enlarged or painful breasts. If you notice breast lumps or nipple discharge, you should talk to your healthcare provider.
Depressed mood has been reported in patients receiving dutasteride.
Dutasteride has been shown to reduce sperm count, semen volume, and sperm movement. However, the effect of dutasteride on male fertility is not known.
Prostate-Specific Antigen (PSA) Test: Your healthcare provider may check you for other prostate problems, including prostate cancer before you start and while you take dutasteride. A blood test called PSA (prostate-specific antigen) is sometimes used to see if you might have prostate cancer. Dutasteride will reduce the amount of PSA measured in your blood. Your healthcare provider is aware of this effect and can still use PSA to see if you might have prostate cancer. Increases in your PSA levels while on treatment with dutasteride (even if the PSA levels are in the normal range) should be evaluated by your healthcare provider.
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects with dutasteride. For more information, ask you healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store dutasteride?
- Store dutasteride capsules at room temperature (59°F to 86°F or 15°C to 30°C).
- Dutasteride capsules may become deformed and/or discolored if kept at high temperatures.
- Do not use dutasteride if your capsules are deformed, discolored, or leaking.
- Safely throw away medicine that is no longer needed.
Keep dutasteride and all medicines out of the reach of children.
Medicines are sometimes prescribed for purposes other than those listed in a patient leaflet. Do not use dutasteride for a condition for which it was not prescribed. Do not give dutasteride to other people, even if they have the same symptoms that you have. It may harm them.
This patient information leaflet summarizes the most important information about dutasteride. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about dutasteride that is written for health professionals.
For more information, contact Banyan Pharma LLC at 1-888-328-0749.
What are the ingredients in dutasteride capsules?
Active ingredient: dutasteride
Inactive ingredients: glycerol, gelatin, titanium dioxide, yellow iron oxide, red iron oxide, mono and di-glycerides, butylhydroxytoluene, lecithin, medium chain triglycerides and black ink. The non-volatile ingredients of the black ink are ammonium hydroxide, black iron oxide, propylene glycol and shellac.
How does dutasteride work?
Prostate growth is caused by a hormone in the blood called dihydrotestosterone (DHT). Dutasteride lowers DHT production in the body, leading to shrinkage of the enlarged prostate in most men. While some men have fewer problems and symptoms after 3 months of treatment with dutasteride, a treatment period of at least 6 months is usually necessary to see if dutasteride will work for you.
Distributed by: Banyan Pharma LLC Hoboken, NJ, 07030
Manufacturered by:
Esjay Pharma Pvt. Ltd.
Made in India
Revised: 12/2025
Description
Warnings and Precautions, Evaluation for Other Urological Diseases ( 5.3 ) 03/2012
Section 34077-8
Teratogenic Effects
Section 42229-5
Monotherapy: Over 4,300 male subjects with BPH were randomly assigned to receive placebo or 0.5-mg daily doses of dutasteride in 3 identical 2-year, placebo-controlled, double-blind, Phase 3 treatment trials, each followed by a 2-year open-label extension. During the double-blind treatment period, 2,167 male subjects were exposed to dutasteride, including 1,772 exposed for 1 year and 1,510 exposed for 2 years. When including the open-label extensions, 1,009 male subjects were exposed to dutasteride for 3 years and 812 were exposed for 4 years. The population was aged 47 to 94 years (mean age: 66 years) and greater than 90% were Caucasian. Table 1 summarizes clinical adverse reactions reported in at least 1% of subjects receiving dutasteride and at a higher incidence than subjects receiving placebo.
| Adverse Reaction Dutasteride (n) Placebo (n) |
Adverse Reaction Time of Onset | |||
|---|---|---|---|---|
| Months 0-6 (n = 2,167) (n = 2,158) |
Months 7-12 (n = 1,901) (n = 1,922) |
Months 13-18 (n = 1,725) (n = 1,714) |
Months 19-24 (n = 1,605) (n = 1,555) |
|
| Impotence These sexual adverse reactions are associated with dutasteride treatment (including monotherapy and combination with tamsulosin). These adverse reactions may persist after treatment discontinuation. The role of dutasteride in this persistence is unknown.
|
||||
| Dutasteride | 4.7% | 1.4% | 1.0% | 0.8% |
| Placebo | 1.7% | 1.5% | 0.5% | 0.9% |
| Decreased libido | ||||
| Dutasteride | 3.0% | 0.7% | 0.3% | 0.3% |
| Placebo | 1.4% | 0.6% | 0.2% | 0.1% |
| Ejaculation disorders | ||||
| Dutasteride | 1.4% | 0.5% | 0.5% | 0.1% |
| Placebo | 0.5% | 0.3% | 0.1% | 0.0% |
| Breast disorders Includes breast tenderness and breast enlargement.
|
||||
| Dutasteride | 0.5% | 0.8% | 1.1% | 0.6% |
| Placebo | 0.2% | 0.3% | 0.3% | 0.1% |
Section 43683-2
| Warnings and Precautions, Evaluation for Other Urological Diseases (5.3) | 03/2012 |
Section 44425-7
Store at 25°C (77°F); excursions permitted to 15-30°C (59-86°F) [see USP Controlled Room Temperature].
Dutasteride is absorbed through the skin. Dutasteride capsules should not be handled by women who are pregnant or who could become pregnant because of the potential for absorption of dutasteride and the subsequent potential risk to a developing male fetus [see Warnings and Precautions (5.4)].
7.5 Digoxin
Dutasteride does not alter the steady-state pharmacokinetics of digoxin when administered concomitantly at a dose of 0.5 mg/day for 3 weeks [see Clinical Pharmacology (12.3)].
7.6 Warfarin
Concomitant administration of dutasteride 0.5 mg/day for 3 weeks with warfarin does not alter the steady-state pharmacokinetics of the S- or R-warfarin isomers or alter the effect of warfarin on prothrombin time [see Clinical Pharmacology (12.3)].
10 Overdosage
In volunteer trials, single doses of dutasteride up to 40 mg (80 times the therapeutic dose) for 7 days have been administered without significant safety concerns. In a clinical trial, daily doses of 5 mg (10 times the therapeutic dose) were administered to 60 subjects for 6 months with no additional adverse effects to those seen at therapeutic doses of 0.5 mg.
There is no specific antidote for dutasteride. Therefore, in cases of suspected overdosage, symptomatic and supportive treatment should be given as appropriate, taking the long half-life of dutasteride into consideration.
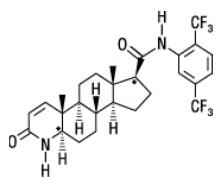
11 Description
Dutasteride is a synthetic 4-azasteroid compound that is a selective inhibitor of both the type 1 and type 2 isoforms of steroid 5 alpha-reductase, an intracellular enzyme that converts testosterone to DHT.
Dutasteride is chemically designated as (5α,17β)-N-{2,5 bis(trifluoromethyl)phenyl}-3-oxo-4-azaandrost-1-ene-17-carboxamide. The empirical formula of dutasteride is C27H30F6N2O2, representing a molecular weight of 528.5 with the following structural formula:
Dutasteride is a white to pale yellow powder with a melting point of 242° to 250°C. It is soluble in ethanol (44 mg/mL), methanol (64 mg/mL), and polyethylene glycol 400 (3 mg/mL), but it is insoluble in water.
Each dutasteride capsule, administered orally, contains 0.5 mg of dutasteride dissolved in a mixture of mono-di-glycerides of caprylic/capric acid and butylhydroxytoluene. The inactive excipients are glycerol, gelatin, titanium dioxide, yellow iron oxide, red iron oxide, mono and di-glycerides, butylhydroxytoluene, lecithin, medium chain triglycerides and black ink. The non-volatile ingredients of the black ink are ammonium hydroxide, black iron oxide, propylene glycol and shellac.
1.1 Monotherapy
Dutasteride capsules are indicated for the treatment of symptomatic benign prostatic hyperplasia (BPH) in men with an enlarged prostate to:
- improve symptoms,
- reduce the risk of acute urinary retention (AUR), and
- reduce the risk of the need for BPH-related surgery.
2.1 Monotherapy
The recommended dose of dutasteride is 1 capsule (0.5 mg) taken once daily.
14.1 Monotherapy
Dutasteride 0.5 mg/day (n = 2,167) or placebo (n = 2,158) was evaluated in male subjects with BPH in three 2-year multicenter, placebo-controlled, double-blind trials, each with 2-year open-label extensions (n = 2,340). More than 90% of the trial population was Caucasian. Subjects were at least 50 years of age with a serum PSA ≥1.5 ng/mL and <10 ng/mL and BPH diagnosed by medical history and physical examination, including enlarged prostate (≥30 cc) and BPH symptoms that were moderate to severe according to the American Urological Association Symptom Index (AUA-SI). Most of the 4,325 subjects randomly assigned to receive either dutasteride or placebo completed 2 years of double-blind treatment (70% and 67%, respectively). Most of the 2,340 subjects in the trial extensions completed 2 additional years of open-label treatment (71%).
8.4 Pediatric Use
Dutasteride is contraindicated for use in pediatric patients. Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Of 2,167 male subjects treated with dutasteride in 3 clinical trials, 60% were aged 65 years and older and 15% were aged 75 years and older. No overall differences in safety or efficacy were observed between these subjects and younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out [see Clinical Pharmacology (12.3)].
5.5 Blood Donation
Men being treated with dutasteride should not donate blood until at least 6 months have passed following their last dose. The purpose of this deferred period is to prevent administration of dutasteride to a pregnant female transfusion recipient.
7.4 Cholestyramine
Administration of a single 5-mg dose of dutasteride followed 1 hour later by 12 g of cholestyramine does not affect the relative bioavailability of dutasteride [see Clinical Pharmacology (12.3)].
17.1 Psa Monitoring
Physicians should inform patients that dutasteride reduces serum PSA levels by approximately 50% within 3 to 6 months of therapy, although it may vary for each individual. For patients undergoing PSA screening, increases in PSA levels while on treatment with dutasteride may signal the presence of prostate cancer and should be evaluated by a healthcare provider [see Warnings and Precautions (5.1)].
17.4 Blood Donation
Physicians should inform men treated with dutasteride that they should not donate blood until at least 6 months following their last dose to prevent pregnant women from receiving dutasteride through blood transfusion [see Warnings and Precautions (5.5)]. Serum levels of dutasteride are detectable for 4 to 6 months after treatment ends [see Clinical Pharmacology (12.3)].
4 Contraindications
Dutasteride is contraindicated for use in:
- Pregnancy. In animal reproduction and developmental toxicity trials, dutasteride inhibited development of male fetus external genitalia. Therefore, dutasteride may cause fetal harm when administered to a pregnant woman. If dutasteride is used during pregnancy or if the patient becomes pregnant while taking dutasteride, the patient should be apprised of the potential hazard to the fetus [see Warnings and Precautions (5.4), Use in Specific Populations (8.1)].
- Women of childbearing potential [see Warnings and Precautions (5.4), Use in Specific Populations (8.1)].
- Pediatric patients [see Use in Specific Populations (8.4)].
- Patients with previously demonstrated clinically significant hypersensitivity (e.g., serious skin reactions, angioedema) to dutasteride or other 5 alpha-reductase inhibitors [see Adverse Reactions (6.2)].
6 Adverse Reactions
The most common adverse reactions, reported in ≥1% of patients treated with dutasteride and more commonly than in subjects treated with placebo, are impotence, decreased libido, ejaculation disorders, and breast disorders. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Banyan Pharma LLC at 1-888-328-0749 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions
Use with caution in patients taking potent, chronic CYP3A4 enzyme inhibitors (e.g., ritonavir). (7)
8.3 Nursing Mothers
Dutasteride is contraindicated for use in women of childbearing potential, including nursing women. It is not known whether dutasteride is excreted in human milk.
8.6 Renal Impairment
No dose adjustment is necessary for dutasteride in patients with renal impairment [see Clinical Pharmacology (12.3)].
1.3 Limitations of Use
Dutasteride is not approved for the prevention of prostate cancer.
8.7 Hepatic Impairment
The effect of hepatic impairment on dutasteride pharmacokinetics has not been studied. Because dutasteride is extensively metabolized, exposure could be higher in hepatically impaired patients. However, in a clinical trial where 60 subjects received 5 mg (10 times the therapeutic dose) daily for 24 weeks, no additional adverse events were observed compared with those observed at the therapeutic dose of 0.5 mg [see Clinical Pharmacology (12.3)].
1 Indications and Usage
Dutasteride is a 5 alpha-reductase inhibitor indicated for the treatment of symptomatic benign prostatic hyperplasia (BPH) in men with an enlarged prostate to: (1.1)
- improve symptoms,
- reduce the risk of acute urinary retention, and
- reduce the risk of the need for BPH-related surgery.
Dutasteride in combination with the alpha adrenergic antagonist, tamsulosin, is indicated for the treatment of symptomatic BPH in men with an enlarged prostate. (1.2)
Limitations of Use: Dutasteride is not approved for the prevention of prostate cancer. (1.3)
12.1 Mechanism of Action
Dutasteride inhibits the conversion of testosterone to dihydrotestosterone (DHT). DHT is the androgen primarily responsible for the initial development and subsequent enlargement of the prostate gland. Testosterone is converted to DHT by the enzyme 5 alpha-reductase, which exists as 2 isoforms, type 1 and type 2. The type 2 isoenzyme is primarily active in the reproductive tissues, while the type 1 isoenzyme is also responsible for testosterone conversion in the skin and liver.
Dutasteride is a competitive and specific inhibitor of both type 1 and type 2 5 alpha-reductase isoenzymes, with which it forms a stable enzyme complex. Dissociation from this complex has been evaluated under in vitro and in vivo conditions and is extremely slow. Dutasteride does not bind to the human androgen receptor.
5 Warnings and Precautions
- Dutasteride reduces serum prostate-specific antigen (PSA) concentration by approximately 50%. However, any confirmed increase in PSA while on dutasteride may signal the presence of prostate cancer and should be evaluated, even if those values are still within the normal range for untreated men. (5.1)
- Dutasteride may increase the risk of high-grade prostate cancer. (5.2, 6.1)
- Prior to initiating treatment with dutasteride, consideration should be given to other urological conditions that may cause similar symptoms. (5.3)
- Women who are pregnant or could become pregnant should not handle dutasteride capsules due to potential risk to a male fetus. (5.4, 8.1)
- Patients should not donate blood until 6 months after their last dose of dutasteride. (5.5)
2 Dosage and Administration
The capsules should be swallowed whole and not chewed or opened, as contact with the capsule contents may result in irritation of the oropharyngeal mucosa. Dutasteride may be administered with or without food.
3 Dosage Forms and Strengths
0.5-mg, light brown, oblong, soft gelatin capsule printed with black 'AV5' and containing a clear, slightly yellow to pale amber oily liquid.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of dutasteride. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. These reactions have been chosen for inclusion due to a combination of their seriousness, frequency of reporting, or potential causal connection to dutasteride.
Immune System Disorders: Hypersensitivity reactions, including rash, pruritus, urticaria, localized edema, serious skin reactions, and angioedema.
Neoplasms: Male breast cancer.
Psychiatric Disorders: Depressed mood.
Reproductive System and Breast Disorders: Testicular pain and testicular swelling.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trial of another drug and may not reflect the rates observed in practice.
From clinical trials with dutasteride as monotherapy or in combination with tamsulosin:
- The most common adverse reactions reported in subjects receiving dutasteride were impotence, decreased libido, breast disorders (including breast enlargement and tenderness), and ejaculation disorders. The most common adverse reactions reported in subjects receiving combination therapy (dutasteride plus tamsulosin) were impotence, decreased libido, breast disorders (including breast enlargement and tenderness), ejaculation disorders, and dizziness. Ejaculation disorders occurred significantly more in subjects receiving combination therapy (11%) compared with those receiving dutasteride (2%) or tamsulosin (4%) as monotherapy.
- Trial withdrawal due to adverse reactions occurred in 4% of subjects receiving dutasteride and 3% of subjects receiving placebo in placebo-controlled trials with dutasteride. The most common adverse reaction leading to trial withdrawal was impotence (1%).
- In the clinical trial evaluating the combination therapy, trial withdrawal due to adverse reactions occurred in 6% of subjects receiving combination therapy (dutasteride plus tamsulosin) and 4% of subjects receiving dutasteride or tamsulosin as monotherapy. The most common adverse reaction in all treatment arms leading to trial withdrawal was erectile dysfunction (1% to 1.5%).
7.3 Calcium Channel Antagonists
Coadministration of verapamil or diltiazem decreases dutasteride clearance and leads to increased exposure to dutasteride. The change in dutasteride exposure is not considered to be clinically significant. No dose adjustment is recommended [see Clinical Pharmacology (12.3)].
7.2 Alpha Adrenergic Antagonists
The administration of dutasteride in combination with tamsulosin or terazosin has no effect on the steady-state pharmacokinetics of either alpha adrenergic antagonist. The effect of administration of tamsulosin or terazosin on dutasteride pharmacokinetic parameters has not been evaluated.
17 Patient Counseling Information
See FDA-approved patient labeling (Patient Information).
7.1 Cytochrome P450 3a Inhibitors
Dutasteride is extensively metabolized in humans by the CYP3A4 and CYP3A5 isoenzymes. The effect of potent CYP3A4 inhibitors on dutasteride has not been studied. Because of the potential for drug-drug interactions, use caution when prescribing dutasteride to patients taking potent, chronic CYP3A4 enzyme inhibitors (e.g., ritonavir) [see Clinical Pharmacology (12.3)].
5.6 Effect On Semen Characteristics
The effects of dutasteride 0.5 mg/day on semen characteristics were evaluated in normal volunteers aged 18 to 52 (n = 27 dutasteride, n = 23 placebo) throughout 52 weeks of treatment and 24 weeks of post-treatment follow-up. At 52 weeks, the mean percent reductions from baseline in total sperm count, semen volume, and sperm motility were 23%, 26%, and 18%, respectively, in the dutasteride group when adjusted for changes from baseline in the placebo group. Sperm concentration and sperm morphology were unaffected. After 24 weeks of follow-up, the mean percent change in total sperm count in the dutasteride group remained 23% lower than baseline. While mean values for all semen parameters at all time-points remained within the normal ranges and did not meet predefined criteria for a clinically significant change (30%), 2 subjects in the dutasteride group had decreases in sperm count of greater than 90% from baseline at 52 weeks, with partial recovery at the 24-week follow-up. The clinical significance of dutasteride's effect on semen characteristics for an individual patient's fertility is not known.
16 How Supplied/storage and Handling
Dutasteride capsules 0.5 mg are light brown, oblong, soft gelatin capsule printed with black 'AV5' and containing a clear, slightly yellow to pale amber oily liquid packaged in bottles of 30 (NDC 84091-100-30) and 90 (NDC 84091-100-90) with child-resistant closures.
Principal Display Panel 90 Capsules
NDC 84091-100-90
Dutasteride Capsules
0.5 mg
WARNING: DUTASTERIDE SHOULD NOT BE
USED BY WOMEN OR CHILDREN.
Woman who are or may potentially be pregnant
should not use or handle Dutasteride Capsules
(see prescribing information). If contact is
made with leaking capsules, wash immediately
with soap and water.
Rx Only
90 Softgel Capsules
Distributed by:
Banyan Pharma LLC
Made in India
5.4 Exposure of Women—risk to Male Fetus
Dutasteride capsules should not be handled by a woman who is pregnant or who could become pregnant. Dutasteride is absorbed through the skin and could result in unintended fetal exposure. If a woman who is pregnant or who could become pregnant comes in contact with leaking dutasteride capsules, the contact area should be washed immediately with soap and water [see Use in Specific Populations (8.1)].
17.3 Exposure of Women—risk to Male Fetus
Physicians should inform patients that dutasteride capsules should not be handled by a woman who is pregnant or who could become pregnant because of the potential for absorption of dutasteride and the subsequent potential risk to a developing male fetus. Dutasteride is absorbed through the skin and could result in unintended fetal exposure. If a pregnant woman or woman of childbearing potential comes in contact with leaking dutasteride capsules, the contact area should be washed immediately with soap and water [see Warnings and Precautions (5.4), Use in Specific Populations (8.1)].
5.3 Evaluation for Other Urological Diseases
Prior to initiating treatment with dutasteride, consideration should be given to other urological conditions that may cause similar symptoms. In addition, BPH and prostate cancer may coexist.
1.2 Combination With Alpha Adrenergic Antagonist
Dutasteride in combination with the alpha adrenergic antagonist, tamsulosin, is indicated for the treatment of symptomatic BPH in men with an enlarged prostate.
2.2 Combination With Alpha Adrenergic Antagonist
The recommended dose of dutasteride is 1 capsule (0.5 mg) taken once daily and tamsulosin 0.4 mg taken once daily.
5.2 Increased Risk of High Grade Prostate Cancer
In men aged 50 to 75 years with a prior negative biopsy for prostate cancer and a baseline PSA between 2.5 ng/mL and 10.0 ng/mL taking dutasteride in the 4-year Reduction by dutasteride of Prostate Cancer Events (REDUCE) trial, there was an increased incidence of Gleason score 8-10 prostate cancer compared with men taking placebo (dutasteride 1.0% versus placebo 0.5%) [see Indications and Usage (1.3), Adverse Reactions (6.1)]. In a 7-year placebo-controlled clinical trial with another 5 alpha-reductase inhibitor (finasteride 5 mg), similar results for Gleason score 8-10 prostate cancer were observed (finasteride 1.8% versus placebo 1.1%).
5 alpha-reductase inhibitors may increase the risk of development of high-grade prostate cancer. Whether the effect of 5 alpha-reductase inhibitors to reduce prostate volume or trial-related factors impacted the results of these trials has not been established.
17.2 Increased Risk of High Grade Prostate Cancer
Physicians should inform patients that there was an increase in high-grade prostate cancer in men treated with 5 alpha-reductase inhibitors (which are indicated for BPH treatment), including dutasteride, compared with those treated with placebo in trials looking at the use of these drugs to reduce the risk of prostate cancer [see Indications and Usage (1.3), Warnings and Precautions (5.2), Adverse Reactions (6.1)].
14.2 Combination With Alpha Blocker Therapy (combat)
The efficacy of combination therapy (dutasteride 0.5 mg/day plus tamsulosin 0.4 mg/day, n = 1,610) was compared with dutasteride alone (n = 1,623) or tamsulosin alone (n = 1,611) in a 4-year multicenter, randomized, double-blind trial. Trial entry criteria were similar to the double-blind, placebo-controlled monotherapy efficacy trials described above in section 14.1. Eighty-eight percent (88%) of the enrolled trial population was Caucasian. Approximately 52% of subjects had previous exposure to 5 alpha-reductase inhibitor or alpha adrenergic antagonist treatment. Of the 4,844 subjects randomly assigned to receive treatment, 69% of subjects in the combination group, 67% in the group receiving dutasteride, and 61% in the tamsulosin group completed 4 years of double-blind treatment.
5.1 Effects On Prostate Specific Antigen (psa) and the Use of Psa in Prostate Cancer Detection
In clinical trials, dutasteride reduced serum PSA concentration by approximately 50% within 3 to 6 months of treatment. This decrease was predictable over the entire range of PSA values in patients with symptomatic BPH, although it may vary in individuals. Dutasteride may also cause decreases in serum PSA in the presence of prostate cancer.
To interpret serial PSAs in men taking dutasteride, a new PSA baseline should be established at least 3 months after starting treatment and PSA monitored periodically thereafter. Any confirmed increase from the lowest PSA value while on dutasteride may signal the presence of prostate cancer and should be evaluated, even if PSA levels are still within the normal range for men not taking a 5 alpha-reductase inhibitor. Noncompliance with dutasteride may also affect PSA test results.
To interpret an isolated PSA value in a man treated with dutasteride for 3 months or more, the PSA value should be doubled for comparison with normal values in untreated men.
The free-to-total PSA ratio (percent free PSA) remains constant, even under the influence of dutasteride. If clinicians elect to use percent free PSA as an aid in the detection of prostate cancer in men receiving dutasteride, no adjustment to its value appears necessary.
Coadministration of dutasteride and tamsulosin resulted in similar changes to serum PSA as dutasteride monotherapy.
Structured Label Content
Section 34077-8 (34077-8)
Teratogenic Effects
Section 42229-5 (42229-5)
Monotherapy: Over 4,300 male subjects with BPH were randomly assigned to receive placebo or 0.5-mg daily doses of dutasteride in 3 identical 2-year, placebo-controlled, double-blind, Phase 3 treatment trials, each followed by a 2-year open-label extension. During the double-blind treatment period, 2,167 male subjects were exposed to dutasteride, including 1,772 exposed for 1 year and 1,510 exposed for 2 years. When including the open-label extensions, 1,009 male subjects were exposed to dutasteride for 3 years and 812 were exposed for 4 years. The population was aged 47 to 94 years (mean age: 66 years) and greater than 90% were Caucasian. Table 1 summarizes clinical adverse reactions reported in at least 1% of subjects receiving dutasteride and at a higher incidence than subjects receiving placebo.
| Adverse Reaction Dutasteride (n) Placebo (n) |
Adverse Reaction Time of Onset | |||
|---|---|---|---|---|
| Months 0-6 (n = 2,167) (n = 2,158) |
Months 7-12 (n = 1,901) (n = 1,922) |
Months 13-18 (n = 1,725) (n = 1,714) |
Months 19-24 (n = 1,605) (n = 1,555) |
|
| Impotence These sexual adverse reactions are associated with dutasteride treatment (including monotherapy and combination with tamsulosin). These adverse reactions may persist after treatment discontinuation. The role of dutasteride in this persistence is unknown.
|
||||
| Dutasteride | 4.7% | 1.4% | 1.0% | 0.8% |
| Placebo | 1.7% | 1.5% | 0.5% | 0.9% |
| Decreased libido | ||||
| Dutasteride | 3.0% | 0.7% | 0.3% | 0.3% |
| Placebo | 1.4% | 0.6% | 0.2% | 0.1% |
| Ejaculation disorders | ||||
| Dutasteride | 1.4% | 0.5% | 0.5% | 0.1% |
| Placebo | 0.5% | 0.3% | 0.1% | 0.0% |
| Breast disorders Includes breast tenderness and breast enlargement.
|
||||
| Dutasteride | 0.5% | 0.8% | 1.1% | 0.6% |
| Placebo | 0.2% | 0.3% | 0.3% | 0.1% |
Section 43683-2 (43683-2)
| Warnings and Precautions, Evaluation for Other Urological Diseases (5.3) | 03/2012 |
Section 44425-7 (44425-7)
Store at 25°C (77°F); excursions permitted to 15-30°C (59-86°F) [see USP Controlled Room Temperature].
Dutasteride is absorbed through the skin. Dutasteride capsules should not be handled by women who are pregnant or who could become pregnant because of the potential for absorption of dutasteride and the subsequent potential risk to a developing male fetus [see Warnings and Precautions (5.4)].
7.5 Digoxin
Dutasteride does not alter the steady-state pharmacokinetics of digoxin when administered concomitantly at a dose of 0.5 mg/day for 3 weeks [see Clinical Pharmacology (12.3)].
7.6 Warfarin
Concomitant administration of dutasteride 0.5 mg/day for 3 weeks with warfarin does not alter the steady-state pharmacokinetics of the S- or R-warfarin isomers or alter the effect of warfarin on prothrombin time [see Clinical Pharmacology (12.3)].
10 Overdosage (10 OVERDOSAGE)
In volunteer trials, single doses of dutasteride up to 40 mg (80 times the therapeutic dose) for 7 days have been administered without significant safety concerns. In a clinical trial, daily doses of 5 mg (10 times the therapeutic dose) were administered to 60 subjects for 6 months with no additional adverse effects to those seen at therapeutic doses of 0.5 mg.
There is no specific antidote for dutasteride. Therefore, in cases of suspected overdosage, symptomatic and supportive treatment should be given as appropriate, taking the long half-life of dutasteride into consideration.
11 Description (11 DESCRIPTION)
Dutasteride is a synthetic 4-azasteroid compound that is a selective inhibitor of both the type 1 and type 2 isoforms of steroid 5 alpha-reductase, an intracellular enzyme that converts testosterone to DHT.
Dutasteride is chemically designated as (5α,17β)-N-{2,5 bis(trifluoromethyl)phenyl}-3-oxo-4-azaandrost-1-ene-17-carboxamide. The empirical formula of dutasteride is C27H30F6N2O2, representing a molecular weight of 528.5 with the following structural formula:
Dutasteride is a white to pale yellow powder with a melting point of 242° to 250°C. It is soluble in ethanol (44 mg/mL), methanol (64 mg/mL), and polyethylene glycol 400 (3 mg/mL), but it is insoluble in water.
Each dutasteride capsule, administered orally, contains 0.5 mg of dutasteride dissolved in a mixture of mono-di-glycerides of caprylic/capric acid and butylhydroxytoluene. The inactive excipients are glycerol, gelatin, titanium dioxide, yellow iron oxide, red iron oxide, mono and di-glycerides, butylhydroxytoluene, lecithin, medium chain triglycerides and black ink. The non-volatile ingredients of the black ink are ammonium hydroxide, black iron oxide, propylene glycol and shellac.
1.1 Monotherapy
Dutasteride capsules are indicated for the treatment of symptomatic benign prostatic hyperplasia (BPH) in men with an enlarged prostate to:
- improve symptoms,
- reduce the risk of acute urinary retention (AUR), and
- reduce the risk of the need for BPH-related surgery.
2.1 Monotherapy
The recommended dose of dutasteride is 1 capsule (0.5 mg) taken once daily.
14.1 Monotherapy
Dutasteride 0.5 mg/day (n = 2,167) or placebo (n = 2,158) was evaluated in male subjects with BPH in three 2-year multicenter, placebo-controlled, double-blind trials, each with 2-year open-label extensions (n = 2,340). More than 90% of the trial population was Caucasian. Subjects were at least 50 years of age with a serum PSA ≥1.5 ng/mL and <10 ng/mL and BPH diagnosed by medical history and physical examination, including enlarged prostate (≥30 cc) and BPH symptoms that were moderate to severe according to the American Urological Association Symptom Index (AUA-SI). Most of the 4,325 subjects randomly assigned to receive either dutasteride or placebo completed 2 years of double-blind treatment (70% and 67%, respectively). Most of the 2,340 subjects in the trial extensions completed 2 additional years of open-label treatment (71%).
8.4 Pediatric Use
Dutasteride is contraindicated for use in pediatric patients. Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Of 2,167 male subjects treated with dutasteride in 3 clinical trials, 60% were aged 65 years and older and 15% were aged 75 years and older. No overall differences in safety or efficacy were observed between these subjects and younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out [see Clinical Pharmacology (12.3)].
5.5 Blood Donation
Men being treated with dutasteride should not donate blood until at least 6 months have passed following their last dose. The purpose of this deferred period is to prevent administration of dutasteride to a pregnant female transfusion recipient.
7.4 Cholestyramine
Administration of a single 5-mg dose of dutasteride followed 1 hour later by 12 g of cholestyramine does not affect the relative bioavailability of dutasteride [see Clinical Pharmacology (12.3)].
17.1 Psa Monitoring (17.1 PSA Monitoring)
Physicians should inform patients that dutasteride reduces serum PSA levels by approximately 50% within 3 to 6 months of therapy, although it may vary for each individual. For patients undergoing PSA screening, increases in PSA levels while on treatment with dutasteride may signal the presence of prostate cancer and should be evaluated by a healthcare provider [see Warnings and Precautions (5.1)].
17.4 Blood Donation
Physicians should inform men treated with dutasteride that they should not donate blood until at least 6 months following their last dose to prevent pregnant women from receiving dutasteride through blood transfusion [see Warnings and Precautions (5.5)]. Serum levels of dutasteride are detectable for 4 to 6 months after treatment ends [see Clinical Pharmacology (12.3)].
4 Contraindications (4 CONTRAINDICATIONS)
Dutasteride is contraindicated for use in:
- Pregnancy. In animal reproduction and developmental toxicity trials, dutasteride inhibited development of male fetus external genitalia. Therefore, dutasteride may cause fetal harm when administered to a pregnant woman. If dutasteride is used during pregnancy or if the patient becomes pregnant while taking dutasteride, the patient should be apprised of the potential hazard to the fetus [see Warnings and Precautions (5.4), Use in Specific Populations (8.1)].
- Women of childbearing potential [see Warnings and Precautions (5.4), Use in Specific Populations (8.1)].
- Pediatric patients [see Use in Specific Populations (8.4)].
- Patients with previously demonstrated clinically significant hypersensitivity (e.g., serious skin reactions, angioedema) to dutasteride or other 5 alpha-reductase inhibitors [see Adverse Reactions (6.2)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The most common adverse reactions, reported in ≥1% of patients treated with dutasteride and more commonly than in subjects treated with placebo, are impotence, decreased libido, ejaculation disorders, and breast disorders. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Banyan Pharma LLC at 1-888-328-0749 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions (7 DRUG INTERACTIONS)
Use with caution in patients taking potent, chronic CYP3A4 enzyme inhibitors (e.g., ritonavir). (7)
8.3 Nursing Mothers
Dutasteride is contraindicated for use in women of childbearing potential, including nursing women. It is not known whether dutasteride is excreted in human milk.
Patient Information
Dutasteride Capsules (doo-TAS-ter-ide)
Dutasteride is for use by men only.
Read this patient information before you start taking dutasteride and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or your treatment.
What is dutasteride?
Dutasteride is a prescription medicine that contains dutasteride. Dutasteride is used to treat the symptoms of benign prostatic hyperplasia (BPH) in men with an enlarged prostate to:
- improve symptoms
- reduce the risk of acute urinary retention (a complete blockage of urine flow)
- reduce the risk of the need for BPH-related surgery
Who should NOT take dutasteride?
Do Not Take Dutasteride if you are:
- pregnant or could become pregnant. Dutasteride may harm your unborn baby. Pregnant women should not touch dutasteride capsules. If a woman who is pregnant with a male baby gets enough dutasteride in her body by swallowing or touching dutasteride, the male baby may be born with sex organs that are not normal. If a pregnant woman or woman of childbearing potential comes in contact with leaking dutasteride capsules, the contact area should be washed immediately with soap and water.
- a child or a teenager.
- allergic to dutasteride or any of the ingredients in dutasteride. See the end of this leaflet for a complete list of ingredients in dutasteride.
- allergic to other 5 alpha-reductase inhibitors, for example, finasteride tablets.
What should I tell my healthcare provider before taking dutasteride?
Before you take dutasteride, tell your healthcare provider if you:
- have liver problems
Tell your healthcare provider about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements. Dutasteride and other medicines may affect each other, causing side effects. Dutasteride may affect the way other medicines work, and other medicines may affect how dutasteride works.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine.
How should I take dutasteride?
- Take 1 dutasteride capsule once a day.
- Swallow dutasteride capsules whole. Do not crush, chew, or open dutasteride capsules because the contents of the capsule may irritate your lips, mouth, or throat.
- You can take dutasteride with or without food.
- If you miss a dose, you may take it later that day. Do not make up the missed dose by taking 2 doses the next day.
What should I avoid while taking dutasteride?
- You should not donate blood while taking dutasteride or for 6 months after you have stopped dutasteride. This is important to prevent pregnant women from receiving dutasteride through blood transfusions.
What are the possible side effects of dutasteride?
Dutasteride may cause serious side effects, including:
-
Rare and serious allergic reactions, including:
- swelling of your face, tongue, or throat
- serious skin reactions, such as skin peeling
Get medical help right away if you have these serious allergic reactions.
- Higher chance of a more serious form of prostate cancer.
The most common side effects of dutasteride include:
- trouble getting or keeping an erection (impotence)Some of these events may continue after you stop taking dustasteride.
- a decrease in sex drive (libido)
- ejaculation problems
- enlarged or painful breasts. If you notice breast lumps or nipple discharge, you should talk to your healthcare provider.
Depressed mood has been reported in patients receiving dutasteride.
Dutasteride has been shown to reduce sperm count, semen volume, and sperm movement. However, the effect of dutasteride on male fertility is not known.
Prostate-Specific Antigen (PSA) Test: Your healthcare provider may check you for other prostate problems, including prostate cancer before you start and while you take dutasteride. A blood test called PSA (prostate-specific antigen) is sometimes used to see if you might have prostate cancer. Dutasteride will reduce the amount of PSA measured in your blood. Your healthcare provider is aware of this effect and can still use PSA to see if you might have prostate cancer. Increases in your PSA levels while on treatment with dutasteride (even if the PSA levels are in the normal range) should be evaluated by your healthcare provider.
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects with dutasteride. For more information, ask you healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store dutasteride?
- Store dutasteride capsules at room temperature (59°F to 86°F or 15°C to 30°C).
- Dutasteride capsules may become deformed and/or discolored if kept at high temperatures.
- Do not use dutasteride if your capsules are deformed, discolored, or leaking.
- Safely throw away medicine that is no longer needed.
Keep dutasteride and all medicines out of the reach of children.
Medicines are sometimes prescribed for purposes other than those listed in a patient leaflet. Do not use dutasteride for a condition for which it was not prescribed. Do not give dutasteride to other people, even if they have the same symptoms that you have. It may harm them.
This patient information leaflet summarizes the most important information about dutasteride. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about dutasteride that is written for health professionals.
For more information, contact Banyan Pharma LLC at 1-888-328-0749.
What are the ingredients in dutasteride capsules?
Active ingredient: dutasteride
Inactive ingredients: glycerol, gelatin, titanium dioxide, yellow iron oxide, red iron oxide, mono and di-glycerides, butylhydroxytoluene, lecithin, medium chain triglycerides and black ink. The non-volatile ingredients of the black ink are ammonium hydroxide, black iron oxide, propylene glycol and shellac.
How does dutasteride work?
Prostate growth is caused by a hormone in the blood called dihydrotestosterone (DHT). Dutasteride lowers DHT production in the body, leading to shrinkage of the enlarged prostate in most men. While some men have fewer problems and symptoms after 3 months of treatment with dutasteride, a treatment period of at least 6 months is usually necessary to see if dutasteride will work for you.
Distributed by: Banyan Pharma LLC Hoboken, NJ, 07030
Manufacturered by:
Esjay Pharma Pvt. Ltd.
Made in India
Revised: 12/2025
8.6 Renal Impairment
No dose adjustment is necessary for dutasteride in patients with renal impairment [see Clinical Pharmacology (12.3)].
1.3 Limitations of Use
Dutasteride is not approved for the prevention of prostate cancer.
8.7 Hepatic Impairment
The effect of hepatic impairment on dutasteride pharmacokinetics has not been studied. Because dutasteride is extensively metabolized, exposure could be higher in hepatically impaired patients. However, in a clinical trial where 60 subjects received 5 mg (10 times the therapeutic dose) daily for 24 weeks, no additional adverse events were observed compared with those observed at the therapeutic dose of 0.5 mg [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Dutasteride is a 5 alpha-reductase inhibitor indicated for the treatment of symptomatic benign prostatic hyperplasia (BPH) in men with an enlarged prostate to: (1.1)
- improve symptoms,
- reduce the risk of acute urinary retention, and
- reduce the risk of the need for BPH-related surgery.
Dutasteride in combination with the alpha adrenergic antagonist, tamsulosin, is indicated for the treatment of symptomatic BPH in men with an enlarged prostate. (1.2)
Limitations of Use: Dutasteride is not approved for the prevention of prostate cancer. (1.3)
12.1 Mechanism of Action
Dutasteride inhibits the conversion of testosterone to dihydrotestosterone (DHT). DHT is the androgen primarily responsible for the initial development and subsequent enlargement of the prostate gland. Testosterone is converted to DHT by the enzyme 5 alpha-reductase, which exists as 2 isoforms, type 1 and type 2. The type 2 isoenzyme is primarily active in the reproductive tissues, while the type 1 isoenzyme is also responsible for testosterone conversion in the skin and liver.
Dutasteride is a competitive and specific inhibitor of both type 1 and type 2 5 alpha-reductase isoenzymes, with which it forms a stable enzyme complex. Dissociation from this complex has been evaluated under in vitro and in vivo conditions and is extremely slow. Dutasteride does not bind to the human androgen receptor.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Dutasteride reduces serum prostate-specific antigen (PSA) concentration by approximately 50%. However, any confirmed increase in PSA while on dutasteride may signal the presence of prostate cancer and should be evaluated, even if those values are still within the normal range for untreated men. (5.1)
- Dutasteride may increase the risk of high-grade prostate cancer. (5.2, 6.1)
- Prior to initiating treatment with dutasteride, consideration should be given to other urological conditions that may cause similar symptoms. (5.3)
- Women who are pregnant or could become pregnant should not handle dutasteride capsules due to potential risk to a male fetus. (5.4, 8.1)
- Patients should not donate blood until 6 months after their last dose of dutasteride. (5.5)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
The capsules should be swallowed whole and not chewed or opened, as contact with the capsule contents may result in irritation of the oropharyngeal mucosa. Dutasteride may be administered with or without food.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
0.5-mg, light brown, oblong, soft gelatin capsule printed with black 'AV5' and containing a clear, slightly yellow to pale amber oily liquid.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of dutasteride. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. These reactions have been chosen for inclusion due to a combination of their seriousness, frequency of reporting, or potential causal connection to dutasteride.
Immune System Disorders: Hypersensitivity reactions, including rash, pruritus, urticaria, localized edema, serious skin reactions, and angioedema.
Neoplasms: Male breast cancer.
Psychiatric Disorders: Depressed mood.
Reproductive System and Breast Disorders: Testicular pain and testicular swelling.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trial of another drug and may not reflect the rates observed in practice.
From clinical trials with dutasteride as monotherapy or in combination with tamsulosin:
- The most common adverse reactions reported in subjects receiving dutasteride were impotence, decreased libido, breast disorders (including breast enlargement and tenderness), and ejaculation disorders. The most common adverse reactions reported in subjects receiving combination therapy (dutasteride plus tamsulosin) were impotence, decreased libido, breast disorders (including breast enlargement and tenderness), ejaculation disorders, and dizziness. Ejaculation disorders occurred significantly more in subjects receiving combination therapy (11%) compared with those receiving dutasteride (2%) or tamsulosin (4%) as monotherapy.
- Trial withdrawal due to adverse reactions occurred in 4% of subjects receiving dutasteride and 3% of subjects receiving placebo in placebo-controlled trials with dutasteride. The most common adverse reaction leading to trial withdrawal was impotence (1%).
- In the clinical trial evaluating the combination therapy, trial withdrawal due to adverse reactions occurred in 6% of subjects receiving combination therapy (dutasteride plus tamsulosin) and 4% of subjects receiving dutasteride or tamsulosin as monotherapy. The most common adverse reaction in all treatment arms leading to trial withdrawal was erectile dysfunction (1% to 1.5%).
7.3 Calcium Channel Antagonists
Coadministration of verapamil or diltiazem decreases dutasteride clearance and leads to increased exposure to dutasteride. The change in dutasteride exposure is not considered to be clinically significant. No dose adjustment is recommended [see Clinical Pharmacology (12.3)].
7.2 Alpha Adrenergic Antagonists
The administration of dutasteride in combination with tamsulosin or terazosin has no effect on the steady-state pharmacokinetics of either alpha adrenergic antagonist. The effect of administration of tamsulosin or terazosin on dutasteride pharmacokinetic parameters has not been evaluated.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
See FDA-approved patient labeling (Patient Information).
7.1 Cytochrome P450 3a Inhibitors (7.1 Cytochrome P450 3A Inhibitors)
Dutasteride is extensively metabolized in humans by the CYP3A4 and CYP3A5 isoenzymes. The effect of potent CYP3A4 inhibitors on dutasteride has not been studied. Because of the potential for drug-drug interactions, use caution when prescribing dutasteride to patients taking potent, chronic CYP3A4 enzyme inhibitors (e.g., ritonavir) [see Clinical Pharmacology (12.3)].
5.6 Effect On Semen Characteristics (5.6 Effect on Semen Characteristics)
The effects of dutasteride 0.5 mg/day on semen characteristics were evaluated in normal volunteers aged 18 to 52 (n = 27 dutasteride, n = 23 placebo) throughout 52 weeks of treatment and 24 weeks of post-treatment follow-up. At 52 weeks, the mean percent reductions from baseline in total sperm count, semen volume, and sperm motility were 23%, 26%, and 18%, respectively, in the dutasteride group when adjusted for changes from baseline in the placebo group. Sperm concentration and sperm morphology were unaffected. After 24 weeks of follow-up, the mean percent change in total sperm count in the dutasteride group remained 23% lower than baseline. While mean values for all semen parameters at all time-points remained within the normal ranges and did not meet predefined criteria for a clinically significant change (30%), 2 subjects in the dutasteride group had decreases in sperm count of greater than 90% from baseline at 52 weeks, with partial recovery at the 24-week follow-up. The clinical significance of dutasteride's effect on semen characteristics for an individual patient's fertility is not known.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Dutasteride capsules 0.5 mg are light brown, oblong, soft gelatin capsule printed with black 'AV5' and containing a clear, slightly yellow to pale amber oily liquid packaged in bottles of 30 (NDC 84091-100-30) and 90 (NDC 84091-100-90) with child-resistant closures.
Principal Display Panel 90 Capsules (PRINCIPAL DISPLAY PANEL - 90 Capsules)
NDC 84091-100-90
Dutasteride Capsules
0.5 mg
WARNING: DUTASTERIDE SHOULD NOT BE
USED BY WOMEN OR CHILDREN.
Woman who are or may potentially be pregnant
should not use or handle Dutasteride Capsules
(see prescribing information). If contact is
made with leaking capsules, wash immediately
with soap and water.
Rx Only
90 Softgel Capsules
Distributed by:
Banyan Pharma LLC
Made in India
5.4 Exposure of Women—risk to Male Fetus (5.4 Exposure of Women—Risk to Male Fetus)
Dutasteride capsules should not be handled by a woman who is pregnant or who could become pregnant. Dutasteride is absorbed through the skin and could result in unintended fetal exposure. If a woman who is pregnant or who could become pregnant comes in contact with leaking dutasteride capsules, the contact area should be washed immediately with soap and water [see Use in Specific Populations (8.1)].
17.3 Exposure of Women—risk to Male Fetus (17.3 Exposure of Women—Risk to Male Fetus)
Physicians should inform patients that dutasteride capsules should not be handled by a woman who is pregnant or who could become pregnant because of the potential for absorption of dutasteride and the subsequent potential risk to a developing male fetus. Dutasteride is absorbed through the skin and could result in unintended fetal exposure. If a pregnant woman or woman of childbearing potential comes in contact with leaking dutasteride capsules, the contact area should be washed immediately with soap and water [see Warnings and Precautions (5.4), Use in Specific Populations (8.1)].
5.3 Evaluation for Other Urological Diseases
Prior to initiating treatment with dutasteride, consideration should be given to other urological conditions that may cause similar symptoms. In addition, BPH and prostate cancer may coexist.
1.2 Combination With Alpha Adrenergic Antagonist
Dutasteride in combination with the alpha adrenergic antagonist, tamsulosin, is indicated for the treatment of symptomatic BPH in men with an enlarged prostate.
2.2 Combination With Alpha Adrenergic Antagonist
The recommended dose of dutasteride is 1 capsule (0.5 mg) taken once daily and tamsulosin 0.4 mg taken once daily.
5.2 Increased Risk of High Grade Prostate Cancer (5.2 Increased Risk of High-Grade Prostate Cancer)
In men aged 50 to 75 years with a prior negative biopsy for prostate cancer and a baseline PSA between 2.5 ng/mL and 10.0 ng/mL taking dutasteride in the 4-year Reduction by dutasteride of Prostate Cancer Events (REDUCE) trial, there was an increased incidence of Gleason score 8-10 prostate cancer compared with men taking placebo (dutasteride 1.0% versus placebo 0.5%) [see Indications and Usage (1.3), Adverse Reactions (6.1)]. In a 7-year placebo-controlled clinical trial with another 5 alpha-reductase inhibitor (finasteride 5 mg), similar results for Gleason score 8-10 prostate cancer were observed (finasteride 1.8% versus placebo 1.1%).
5 alpha-reductase inhibitors may increase the risk of development of high-grade prostate cancer. Whether the effect of 5 alpha-reductase inhibitors to reduce prostate volume or trial-related factors impacted the results of these trials has not been established.
17.2 Increased Risk of High Grade Prostate Cancer (17.2 Increased Risk of High-Grade Prostate Cancer)
Physicians should inform patients that there was an increase in high-grade prostate cancer in men treated with 5 alpha-reductase inhibitors (which are indicated for BPH treatment), including dutasteride, compared with those treated with placebo in trials looking at the use of these drugs to reduce the risk of prostate cancer [see Indications and Usage (1.3), Warnings and Precautions (5.2), Adverse Reactions (6.1)].
14.2 Combination With Alpha Blocker Therapy (combat) (14.2 Combination With Alpha-Blocker Therapy (CombAT))
The efficacy of combination therapy (dutasteride 0.5 mg/day plus tamsulosin 0.4 mg/day, n = 1,610) was compared with dutasteride alone (n = 1,623) or tamsulosin alone (n = 1,611) in a 4-year multicenter, randomized, double-blind trial. Trial entry criteria were similar to the double-blind, placebo-controlled monotherapy efficacy trials described above in section 14.1. Eighty-eight percent (88%) of the enrolled trial population was Caucasian. Approximately 52% of subjects had previous exposure to 5 alpha-reductase inhibitor or alpha adrenergic antagonist treatment. Of the 4,844 subjects randomly assigned to receive treatment, 69% of subjects in the combination group, 67% in the group receiving dutasteride, and 61% in the tamsulosin group completed 4 years of double-blind treatment.
5.1 Effects On Prostate Specific Antigen (psa) and the Use of Psa in Prostate Cancer Detection (5.1 Effects on Prostate-Specific Antigen (PSA) and the Use of PSA in Prostate Cancer Detection)
In clinical trials, dutasteride reduced serum PSA concentration by approximately 50% within 3 to 6 months of treatment. This decrease was predictable over the entire range of PSA values in patients with symptomatic BPH, although it may vary in individuals. Dutasteride may also cause decreases in serum PSA in the presence of prostate cancer.
To interpret serial PSAs in men taking dutasteride, a new PSA baseline should be established at least 3 months after starting treatment and PSA monitored periodically thereafter. Any confirmed increase from the lowest PSA value while on dutasteride may signal the presence of prostate cancer and should be evaluated, even if PSA levels are still within the normal range for men not taking a 5 alpha-reductase inhibitor. Noncompliance with dutasteride may also affect PSA test results.
To interpret an isolated PSA value in a man treated with dutasteride for 3 months or more, the PSA value should be doubled for comparison with normal values in untreated men.
The free-to-total PSA ratio (percent free PSA) remains constant, even under the influence of dutasteride. If clinicians elect to use percent free PSA as an aid in the detection of prostate cancer in men receiving dutasteride, no adjustment to its value appears necessary.
Coadministration of dutasteride and tamsulosin resulted in similar changes to serum PSA as dutasteride monotherapy.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:45.669608 · Updated: 2026-03-14T22:41:36.835741