These Highlights Do Not Include All The Information Needed To Use Pirfenidone Capsules Safely And Effectively. See Full Prescribing Information For Pirfenidone Capsules.
945d4337-7d18-4d84-807b-8ae13750c76d
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dosage and Administration ( 2.3 ) 02/2023 Warnings and Precautions ( 5.3 ) 02/2023
Indications and Usage
Pirfenidone capsules are indicated for the treatment of idiopathic pulmonary fibrosis (IPF).
Dosage and Administration
• Take with food. • Recommended dosage: 801 mg three times daily (2403 mg/day). ( 2 ) • Upon initiation of treatment, titrate to the full dosage of 2403 mg/day over a 14-day period as follows: Treatment days Dosage Days 1 through 7 267 mg three times daily (801 mg/day) Days 8 through 14 534 mg three times daily (1602 mg/day) Days 15 onward 801 mg three times daily (2403 mg/day) • Consider temporary dosage reduction, treatment interruption, or discontinuation for management of adverse reactions. ( 2.3 , 5.1 , 5.2 , 5.3 , 5.4 ) • Prior to treatment, conduct liver function tests. ( 2.1 )
Warnings and Precautions
• Elevated liver enzymes and drug-induced liver injury: ALT, AST, and bilirubin elevations have occurred with pirfenidone including cases of drug-induced liver injury. In the postmarketing setting, non-serious and serious cases of drug-induced liver injury, including severe liver injury with fatal outcomes, have been reported. Monitor ALT, AST, and bilirubin before and during treatment. Temporary dosage reductions or discontinuations may be required. ( 2.1 , 5.1 ) • Photosensitivity and rash: Photosensitivity and rash have been noted with pirfenidone. Avoid exposure to sunlight and sunlamps. Wear sunscreen and protective clothing daily. Temporary dosage reductions or discontinuations may be required. ( 5.2 ) • Severe Cutaneous Adverse Reactions (SCAR): Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug reactions with eosinophilia and systemic symptoms (DRESS) have been reported in association with the use of pirfenidone in the postmarketing setting. Interrupt pirfenidone in case of signs or symptoms of SCAR. Permanently discontinue pirfenidone if a SCAR is confirmed. ( 5.3 ) • Gastrointestinal disorders: Nausea, vomiting, diarrhea, dyspepsia, gastro-esophageal reflux disease, and abdominal pain have occurred with pirfenidone. Temporary dosage reductions or discontinuations may be required. ( 5.4 )
Contraindications
None.
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling: • Liver Enzyme Elevations and Drug-Induced Liver Injury [see Warnings and Precautions ( 5.1 )] • Photosensitivity Reaction or Rash [see Warnings and Precautions ( 5.2 )] • Severe Cutaneous Adverse Reactions [see Warnings and Precautions ( 5.3 )] • Gastrointestinal Disorders [see Warnings and Precautions ( 5.4 )]
Drug Interactions
Moderate (e.g., ciprofloxacin) and strong inhibitors of CYP1A2 (e.g., fluvoxamine) increase systemic exposure of pirfenidone and may alter the adverse reaction profile of pirfenidone. Discontinue fluvoxamine prior to administration of pirfenidone or reduce to 267 mg three times a day. Consider dosage reduction with use of ciprofloxacin. ( 7.1 )
Storage and Handling
Pirfenidone capsules are white hard gelatin capsules contain 267 mg of pirfenidone. The cap of the capsule is printed with "PN267" in black ink. The capsules are supplied as follows: NDC 0781-2158-32, bottle for a 30-day supply containing 270 capsules and closed with child-resistant closure
How Supplied
Pirfenidone capsules are white hard gelatin capsules contain 267 mg of pirfenidone. The cap of the capsule is printed with "PN267" in black ink. The capsules are supplied as follows: NDC 0781-2158-32, bottle for a 30-day supply containing 270 capsules and closed with child-resistant closure
Medication Information
Warnings and Precautions
• Elevated liver enzymes and drug-induced liver injury: ALT, AST, and bilirubin elevations have occurred with pirfenidone including cases of drug-induced liver injury. In the postmarketing setting, non-serious and serious cases of drug-induced liver injury, including severe liver injury with fatal outcomes, have been reported. Monitor ALT, AST, and bilirubin before and during treatment. Temporary dosage reductions or discontinuations may be required. ( 2.1 , 5.1 ) • Photosensitivity and rash: Photosensitivity and rash have been noted with pirfenidone. Avoid exposure to sunlight and sunlamps. Wear sunscreen and protective clothing daily. Temporary dosage reductions or discontinuations may be required. ( 5.2 ) • Severe Cutaneous Adverse Reactions (SCAR): Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug reactions with eosinophilia and systemic symptoms (DRESS) have been reported in association with the use of pirfenidone in the postmarketing setting. Interrupt pirfenidone in case of signs or symptoms of SCAR. Permanently discontinue pirfenidone if a SCAR is confirmed. ( 5.3 ) • Gastrointestinal disorders: Nausea, vomiting, diarrhea, dyspepsia, gastro-esophageal reflux disease, and abdominal pain have occurred with pirfenidone. Temporary dosage reductions or discontinuations may be required. ( 5.4 )
Indications and Usage
Pirfenidone capsules are indicated for the treatment of idiopathic pulmonary fibrosis (IPF).
Dosage and Administration
• Take with food. • Recommended dosage: 801 mg three times daily (2403 mg/day). ( 2 ) • Upon initiation of treatment, titrate to the full dosage of 2403 mg/day over a 14-day period as follows: Treatment days Dosage Days 1 through 7 267 mg three times daily (801 mg/day) Days 8 through 14 534 mg three times daily (1602 mg/day) Days 15 onward 801 mg three times daily (2403 mg/day) • Consider temporary dosage reduction, treatment interruption, or discontinuation for management of adverse reactions. ( 2.3 , 5.1 , 5.2 , 5.3 , 5.4 ) • Prior to treatment, conduct liver function tests. ( 2.1 )
Contraindications
None.
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling: • Liver Enzyme Elevations and Drug-Induced Liver Injury [see Warnings and Precautions ( 5.1 )] • Photosensitivity Reaction or Rash [see Warnings and Precautions ( 5.2 )] • Severe Cutaneous Adverse Reactions [see Warnings and Precautions ( 5.3 )] • Gastrointestinal Disorders [see Warnings and Precautions ( 5.4 )]
Drug Interactions
Moderate (e.g., ciprofloxacin) and strong inhibitors of CYP1A2 (e.g., fluvoxamine) increase systemic exposure of pirfenidone and may alter the adverse reaction profile of pirfenidone. Discontinue fluvoxamine prior to administration of pirfenidone or reduce to 267 mg three times a day. Consider dosage reduction with use of ciprofloxacin. ( 7.1 )
Storage and Handling
Pirfenidone capsules are white hard gelatin capsules contain 267 mg of pirfenidone. The cap of the capsule is printed with "PN267" in black ink. The capsules are supplied as follows: NDC 0781-2158-32, bottle for a 30-day supply containing 270 capsules and closed with child-resistant closure
How Supplied
Pirfenidone capsules are white hard gelatin capsules contain 267 mg of pirfenidone. The cap of the capsule is printed with "PN267" in black ink. The capsules are supplied as follows: NDC 0781-2158-32, bottle for a 30-day supply containing 270 capsules and closed with child-resistant closure
Description
Dosage and Administration ( 2.3 ) 02/2023 Warnings and Precautions ( 5.3 ) 02/2023
Section 42229-5
Dosage Modification due to Elevated Liver Enzymes
Dosage modifications or interruptions may also be necessary when liver enzyme and bilirubin elevations are exhibited. For liver enzyme elevations, modify the dosage as follows:
If a patient exhibits >3 but ≤5 × the upper limit of normal (ULN) ALT and/or AST without symptoms or hyperbilirubinemia after starting pirfenidone capsules therapy:
-
•Discontinue confounding medications, exclude other causes, and monitor the patient closely.
-
•Repeat liver chemistry tests as clinically indicated.
-
•The full daily dosage may be maintained, if clinically appropriate, or reduced or interrupted (e.g., until liver chemistry tests are within normal limits) with subsequent re-titration to the full dosage as tolerated.
If a patient exhibits >3 but ≤5 × ULN ALT and/or AST accompanied by symptoms or hyperbilirubinemia:
-
•Permanently discontinue pirfenidone capsules.
-
•Do not rechallenge patient with pirfenidone capsules.
If a patient exhibits >5 × ULN ALT and/or AST:
-
•Permanently discontinue pirfenidone capsules.
-
•Do not rechallenge patient with pirfenidone capsules.
Section 42230-3
|
Patient Information
(pir-FEN-i-done) |
|
What are pirfenidone capsules?
|
|
Before you take pirfenidone capsules, tell your doctor about all of your medical conditions, including if you:
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. |
|
How should I take pirfenidone capsules?
|
|
What should I avoid while taking pirfenidone capsules?
|
|
What are the possible side effects of pirfenidone capsules? Pirfenidone capsules may cause serious side effects, including:
The most common side effects of pirfenidone capsules include feeling tired, insomnia, upper respiratory tract infections, sinusitis, headache, dizziness, decreased weight and decreased or loss of appetite. These are not all the possible side effects of pirfenidone capsules. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store pirfenidone capsules?
Safely throw away any pirfenidone capsules that is out of date or no longer needed. Keep pirfenidone capsules and all medicines out of reach of children. |
|
General information about the safe and effective use of pirfenidone capsules. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use pirfenidone capsules for a condition for which it was not prescribed. Do not give pirfenidone capsules to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or doctor for information about pirfenidone capsules that is written for health professionals. |
|
What are the ingredients in pirfenidone capsules? Active ingredient: pirfenidone Inactive ingredients: croscarmellose sodium, hypromellose, magnesium stearate, and pregelatinized starch. Capsule Shell: gelatin and titanium dioxide Capsule Black Printing Ink: iron oxide black, potassium hydroxide, propylene glycol, shellac and strong ammonia solution. For more information, contact Sandoz Inc., at 1-800-525-8747 Finished Drug Product Manufactured by Novartis d.o.o., 1000 Ljubljana, Slovenia for Sandoz Inc., Princeton, NJ 08540 |
This Patient Information has been approved by the U.S. Food and Drug Administration Rev. October 2024
Section 44425-7
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Keep the bottle tightly closed. Do not use if the seal over the bottle opening is broken or missing. Safely throw away any pirfenidone capsules that is out of date or no longer needed.
8.8 Smokers
Smoking causes decreased exposure to pirfenidone [see Clinical Pharmacology (12.3)], which may alter the efficacy profile of pirfenidone. Instruct patients to stop smoking prior to treatment with pirfenidone and to avoid smoking when using pirfenidone.
10 Overdosage
There is limited clinical experience with overdosage. Multiple dosages of pirfenidone up to a maximum tolerated dose of 4005 mg per day were administered as five 267 mg capsules three times daily to healthy adult volunteers over a 12-day dose escalation.
In the event of a suspected overdosage, appropriate supportive medical care should be provided, including monitoring of vital signs and observation of the clinical status of the patient.
8.2 Lactation
Risk Summary
No information is available on the presence of pirfenidone in human milk, the effects of the drug on the breastfed infant, or the effects of the drug on milk production. The lack of clinical data during lactation precludes clear determination of the risk of pirfenidone to an infant during lactation; therefore, the developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for pirfenidone and the potential adverse effects on the breastfed child from pirfenidone or from the underlying maternal condition.
Data
Animal Data: A study with radio-labeled pirfenidone in rats has shown that pirfenidone or its metabolites are excreted in milk. There are no data on the presence of pirfenidone or its metabolites in human milk, the effects of pirfenidone on the breastfed child, or its effects on milk production.
11 Description
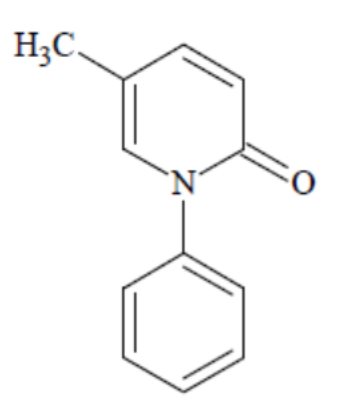
Pirfenidone belongs to the chemical class of pyridone. Pirfenidone capsules are available as white hard gelatin capsules containing 267 mg of pirfenidone for oral administration.
Pirfenidone has a molecular formula of C12H11NO and a molecular weight of 185.23. Pirfenidone has the following structural formula, which has been referred to as 5-methyl-1-phenyl-2-1(H)-pyridone or 5-methyl-1-phenyl-2-(1H)-pyridone.
Pirfenidone is a white to pale yellow crystalline, non-hygroscopic powder. It is more soluble in methanol, ethyl alcohol, acetone and chloroform than in water and 1.0 N HCl. The melting point is approximately 108°C.
Pirfenidone capsule contains pirfenidone and the following inactive ingredients: croscarmellose sodium, hypromellose, magnesium stearate, and pregelatinized starch.
In addition, the capsule shell contains gelatin and titanium dioxide. The capsule black printing ink includes iron oxide black, potassium hydroxide, propylene glycol, shellac and strong ammonia solution.
8.4 Pediatric Use
Safety and effectiveness of pirfenidone in pediatric patients have not been established.
8.5 Geriatric Use
Of the total number of subjects in the clinical studies receiving pirfenidone capsules, 714 (67%) were 65 years old and over, while 231 (22%) were 75 years old and over. No overall differences in safety or effectiveness were observed between older and younger patients. No dosage adjustment is required based upon age.
14 Clinical Studies
The efficacy of pirfenidone was evaluated in patients with IPF in three phase 3, randomized, double-blind, placebo-controlled, multicenter trials (Studies 1, 2, and 3).
Study 1 was a 52-week trial comparing pirfenidone 2403 mg/day (n=278) versus placebo (n=277) in patients with IPF. Study 2 and Study 3 were nearly identical to each other in design, with few exceptions, including an intermediate dose treatment arm in Study 2. Study 2 compared treatment with either pirfenidone 2403 mg/day (n=174) or pirfenidone 1197 mg/day (n=87) to placebo (n=174), while Study 3 compared pirfenidone 2403 mg/day (n=171) to placebo (n=173). Study drug was administered three times daily with food for a minimum of 72 weeks. Patients continued on treatment until the last patient completed 72 weeks of treatment, which included observations to approximately 120 weeks of study treatment. The primary endpoint was the change in percent predicted forced vital capacity (%FVC) from baseline to study end, measured at 52 weeks in Study 1, and at 72 weeks in Studies 2 and 3.
Studies 1, 2 and 3 enrolled adult patients who had a clinical and radiographic diagnosis of IPF (with or without accompanying surgical lung biopsy), without evidence or suspicion of an alternative diagnosis for interstitial lung disease. Eligible patients were to have %FVC greater than or equal to 50% at baseline and a percent predicted diffusing capacity of the lungs for carbon monoxide (%DLCO) greater than or equal to 30% (Study 1) or 35% (Studies 2 and 3) at baseline. In all three trials, over 80% of patients completed study treatment.
A total of 1247 patients with IPF were randomized to receive pirfenidone 2403 mg/day (n=623) or placebo (n=624) in these three trials. Baseline characteristics were generally balanced across treatment groups. The study population ranged from 40 to 80 years of age (mean age 67 years). Most patients were male (74%), white (95%), and current or former smokers (65%). Approximately 93% of patients met criteria for definite IPF on high resolution computed tomography (HRCT). Baseline mean %FVC and %DLCO were 72% and 46%, respectively. Approximately 15% subjects discontinued from each treatment group.
4 Contraindications
None.
6 Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling:
-
•Liver Enzyme Elevations and Drug-Induced Liver Injury [see Warnings and Precautions (5.1)]
-
•Photosensitivity Reaction or Rash [see Warnings and Precautions (5.2)]
-
•Severe Cutaneous Adverse Reactions [see Warnings and Precautions (5.3)]
-
•Gastrointestinal Disorders [see Warnings and Precautions (5.4)]
7 Drug Interactions
Moderate (e.g., ciprofloxacin) and strong inhibitors of CYP1A2 (e.g., fluvoxamine) increase systemic exposure of pirfenidone and may alter the adverse reaction profile of pirfenidone. Discontinue fluvoxamine prior to administration of pirfenidone or reduce to 267 mg three times a day. Consider dosage reduction with use of ciprofloxacin. (7.1)
7.2 Cyp1a2 Inducers
The concomitant use of pirfenidone and a CYP1A2 inducer may decrease the exposure of pirfenidone and this may lead to loss of efficacy. Therefore, discontinue use of strong CYP1A2 inducers prior to pirfenidone treatment and avoid the concomitant use of pirfenidone and a strong CYP1A2 inducer [see Clinical Pharmacology (12.3)].
8.7 Renal Impairment
Pirfenidone should be used with caution in patients with mild (CLcr 50 to 80 mL/min), moderate (CLcr 30 to 50 mL/min), or severe (CLcr less than 30 mL/min) renal impairment [see Clinical Pharmacology (12.3)]. Monitor for adverse reactions and consider dosage modification or discontinuation of pirfenidone as needed [see Dosage and Administration (2.3)]. The safety, efficacy, and pharmacokinetics of pirfenidone have not been studied in patients with end-stage renal disease requiring dialysis. Use of pirfenidone in patients with end-stage renal diseases requiring dialysis is not recommended.
12.2 Pharmacodynamics
Cardiac Electrophysiology
The effect of pirfenidone on QT interval was evaluated in a randomized, placebo, and positive controlled parallel study in 160 healthy adult volunteers. Volunteers received pirfenidone 2403 mg/day (recommended dose) and 4005 mg/day (1.6 times recommended dose) or placebo for 10 days or a single dose of 400 mg moxifloxacin (active control).
Relative to placebo, the maximum mean change from baseline in study-specific QT interval was 3.2 milliseconds (ms) and 2.2 ms for pirfenidone 2403 mg/day and 4005 mg/day, respectively. No volunteer had a QTc interval greater than 480 ms or change from baseline greater than 60 ms. Although there was no evidence that pirfenidone prolonged the QTc interval in this study, a definitive conclusion may not be drawn as the positive control (moxifloxacin) did not perform as expected in this study, and pirfenidone at 4005 mg/day (1.7 times the maximum recommended dose) did not cover the maximum pirfenidone exposure increase with co-administration of fluvoxamine, a strong CYP1A2 inhibitor.
7.1 Cyp1a2 Inhibitors
Pirfenidone is metabolized primarily (70 to 80%) via CYP1A2 with minor contributions from other CYP isoenzymes including CYP2C9, 2C19, 2D6 and 2E1.
2.2 Recommended Dosage
The recommended daily maintenance dosage of pirfenidone capsules are 801 mg three times daily for a total of 2403 mg/day. Doses should be taken with food at the same time each day.
Upon initiation of treatment, titrate to the full dosage of 2403 mg/day over a 14-day period as follows:
| Treatment days | Dosage |
|---|---|
|
Days 1 through 7 |
267 mg three times daily (801 mg/day) |
|
Days 8 through 14 |
534 mg three times daily (1602 mg/day) |
|
Days 15 onward |
801 mg three times daily (2403 mg/day) |
Dosages above 2403 mg/day are not recommended for any patient. Patients should not take 2 doses at the same time to make up for a missed dose. Patients should not take more than 3 doses per day.
8.6 Hepatic Impairment
Pirfenidone should be used with caution in patients with mild (Child Pugh Class A) to moderate (Child Pugh Class B) hepatic impairment. Monitor for adverse reactions and consider dosage modification or discontinuation of pirfenidone as needed [see Dosage and Administration (2.3)].
The safety, efficacy, and pharmacokinetics of pirfenidone have not been studied in patients with severe hepatic impairment. Pirfenidone is not recommended for use in patients with severe (Child Pugh Class C) hepatic impairment [see Clinical Pharmacology (12.3)].
1 Indications and Usage
Pirfenidone capsules are indicated for the treatment of idiopathic pulmonary fibrosis (IPF).
Principal Display Panel
NDC 0781-2158-32
Pirfenidone Capsules
267 mg
Rx Only
270 Capsules
SANDOZ
12.1 Mechanism of Action
The mechanism of action of pirfenidone in the treatment of IPF has not been established.
5 Warnings and Precautions
-
•Elevated liver enzymes and drug-induced liver injury: ALT, AST, and bilirubin elevations have occurred with pirfenidone including cases of drug-induced liver injury. In the postmarketing setting, non-serious and serious cases of drug-induced liver injury, including severe liver injury with fatal outcomes, have been reported. Monitor ALT, AST, and bilirubin before and during treatment. Temporary dosage reductions or discontinuations may be required. (2.1, 5.1)
-
•Photosensitivity and rash: Photosensitivity and rash have been noted with pirfenidone. Avoid exposure to sunlight and sunlamps. Wear sunscreen and protective clothing daily. Temporary dosage reductions or discontinuations may be required. (5.2)
-
•Severe Cutaneous Adverse Reactions (SCAR): Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug reactions with eosinophilia and systemic symptoms (DRESS) have been reported in association with the use of pirfenidone in the postmarketing setting. Interrupt pirfenidone in case of signs or symptoms of SCAR. Permanently discontinue pirfenidone if a SCAR is confirmed. (5.3)
-
•Gastrointestinal disorders: Nausea, vomiting, diarrhea, dyspepsia, gastro-esophageal reflux disease, and abdominal pain have occurred with pirfenidone. Temporary dosage reductions or discontinuations may be required. (5.4)
2 Dosage and Administration
-
•Take with food.
-
•Recommended dosage: 801 mg three times daily (2403 mg/day). (2)
-
•Upon initiation of treatment, titrate to the full dosage of 2403 mg/day over a 14-day period as follows:
| Treatment days | Dosage |
|---|---|
|
Days 1 through 7 |
267 mg three times daily (801 mg/day) |
|
Days 8 through 14 |
534 mg three times daily (1602 mg/day) |
|
Days 15 onward |
801 mg three times daily (2403 mg/day) |
3 Dosage Forms and Strengths
Capsules: 267 mg, White hard gelatin capsules printed with PN267 on the cap of the capsule in black ink.
6.2 Postmarketing Experience
In addition to adverse reactions identified from clinical trials the following adverse reactions have been identified during post-approval use of pirfenidone. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency.
Blood and Lymphatic System Disorders: Agranulocytosis
Hepatobiliary Disorders: Drug-induced liver injury
Immune System Disorders: Angioedema
Skin and Subcutaneous Tissue Disorders: Severe Cutaneous Adverse Reactions (SCAR)
8 Use in Specific Populations
-
•Hepatic Impairment: Monitor for adverse reactions and consider dosage modification or discontinuation of pirfenidone as needed. Pirfenidone is not recommended for use in patients with severe hepatic impairment. (8.6, 12.3)
-
•Renal Impairment: Monitor for adverse reactions and consider dosage modification or discontinuation of pirfenidone as needed. Pirfenidone is not recommended for use in patients with end stage renal disease on dialysis. (8.7, 12.3)
-
•Smokers: Decreased exposure has been noted in smokers which may alter the efficacy profile of pirfenidone. (8.8)
5.4 Gastrointestinal Disorders
In the clinical studies, gastrointestinal events of nausea, diarrhea, dyspepsia, vomiting, gastro-esophageal reflux disease, and abdominal pain were more frequently reported by patients in the pirfenidone treatment groups than in those taking placebo. Dosage reduction or interruption for gastrointestinal events was required in 18.5% of patients in the 2403 mg/day group, as compared to 5.8% of patients in the placebo group; 2.2% of patients in the pirfenidone 2403 mg/day group discontinued treatment due to a gastrointestinal event, as compared to 1.0% in the placebo group. The most common (>2%) gastrointestinal events that led to dosage reduction or interruption were nausea, diarrhea, vomiting, and dyspepsia. The incidence of gastrointestinal events was highest early in the course of treatment (with highest incidence occurring during the initial 3 months) and decreased over time. Dosage modifications may be necessary in some cases of gastrointestinal adverse reactions [see Dosage and Administration (2.3)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of pirfenidone has been evaluated in more than 1400 subjects with over 170 subjects exposed to pirfenidone for more than 5 years in clinical trials.
Pirfenidone was studied in 3 randomized, double-blind, placebo-controlled trials (Studies 1, 2, and 3) in which a total of 623 patients received 2403 mg/day of pirfenidone and 624 patients received placebo. Subjects ages ranged from 40 to 80 years (mean age of 67 years). Most patients were male (74%) and Caucasian (95%). The mean duration of exposure to pirfenidone was 62 weeks (range: 2 to 118 weeks) in these 3 trials.
At the recommended dosage of 2403 mg/day, 14.6% of patients on pirfenidone compared to 9.6% on placebo permanently discontinued treatment because of an adverse event. The most common (>1%) adverse reactions leading to discontinuation were rash and nausea. The most common (>3%) adverse reactions leading to dosage reduction or interruption were rash, nausea, diarrhea, and photosensitivity reaction.
The most common adverse reactions with an incidence of ≥10% and more frequent in the pirfenidone than placebo treatment group are listed in Table 2 .
-
Table 2. Adverse Reactions Occurring in ≥10% of Pirfenidone-Treated Patients and More Commonly Than Placebo in Studies 1, 2, and 3
| Adverse Reaction | % of Patients (0 to 118 Weeks) | |
|---|---|---|
|
Pirfenidone 2403 mg/day
(N = 623) |
Placebo
(N = 624) |
|
|
Nausea |
36% |
16% |
|
Rash |
30% |
10% |
|
Abdominal Pain Includes abdominal pain, upper abdominal pain, abdominal distension, and stomach discomfort.
|
24% |
15% |
|
Upper Respiratory Tract Infection |
27% |
25% |
|
Diarrhea |
26% |
20% |
|
Fatigue |
26% |
19% |
|
Headache |
22% |
19% |
|
Decreased Appetite |
21% |
8% |
|
Dyspepsia |
19% |
7% |
|
Dizziness |
18% |
11% |
|
Vomiting |
13% |
6% |
|
Gastro-esophageal Reflux Disease |
11% |
7% |
|
Sinusitis |
11% |
10% |
|
Insomnia |
10% |
7% |
|
Weight Decreased |
10% |
5% |
|
Arthralgia |
10% |
7% |
Adverse reactions occurring in ≥5 to <10% of pirfenidone-treated patients and more commonly than placebo are photosensitivity reaction (9% vs. 1%), pruritus (8% vs. 5%), asthenia (6% vs. 4%), dysgeusia (6% vs. 2%), and non-cardiac chest pain (5% vs. 4%).
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling ( Patient Information ).
16 How Supplied/storage and Handling
Pirfenidone capsules are white hard gelatin capsules contain 267 mg of pirfenidone. The cap of the capsule is printed with "PN267" in black ink. The capsules are supplied as follows:
-
NDC 0781-2158-32, bottle for a 30-day supply containing 270 capsules and closed with child-resistant closure
5.2 Photosensitivity Reaction Or Rash
Patients treated with pirfenidone 2403 mg/day in the three Phase 3 studies had a higher incidence of photosensitivity reactions (9%) compared with patients treated with placebo (1%). The majority of the photosensitivity reactions occurred during the initial 6 months. Instruct patients to avoid or minimize exposure to sunlight (including sunlamps), to use a sunblock (SPF 50 or higher), and to wear clothing that protects against sun exposure. Additionally, instruct patients to avoid concomitant medications known to cause photosensitivity. Dosage reduction or discontinuation may be necessary in some cases of photosensitivity reaction or rash [see Dosage and Administration (2.3) ].
5.3 Severe Cutaneous Adverse Reactions
Severe cutaneous adverse reactions (SCAR), including Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug reaction with eosinophilia and systemic symptoms (DRESS), have been reported in association with the use of pirfenidone in the postmarketing setting. If signs or symptoms of SCAR occur, interrupt pirfenidone treatment until the etiology of the reaction has been determined. Consultation with a dermatologist is recommended. If a SCAR is confirmed, permanently discontinue pirfenidone.
2.3 Dosage Modifications Due to Adverse Reactions
Patients who miss 14 or more days of pirfenidone capsules should re-initiate treatment by undergoing the initial 2-week titration regimen up to the full maintenance dosage [see Dosage and Administration (2.2) ]. For treatment interruption of less than 14 days, the dosage prior to the interruption can be resumed.
If patients experience significant adverse reactions (i.e., gastrointestinal, photosensitivity reaction or rash, severe cutaneous adverse reactions (SCAR)), consider temporary dosage reductions or interruptions of pirfenidone capsules to allow for resolution of symptoms. If a SCAR is confirmed, permanently discontinue pirfenidone capsules [see Warnings and Precautions (5.1, 5.2, 5.3, 5.4)] .
2.4 Dosage Modifications Due to Drug Interactions
Strong CYP1A2 Inhibitors (e.g., fluvoxamine, enoxacin)
Reduce pirfenidone capsules to 267 mg three times a day (801 mg/day).
Moderate CYP1A2 Inhibitors (e.g., ciprofloxacin)
With use of ciprofloxacin at a dosage of 750 mg twice daily, reduce pirfenidone capsules to 534 mg three times a day (1602 mg/day).
2.1 Testing Prior to Pirfenidone Capsules Administration
Conduct liver function tests prior to initiating treatment with pirfenidone capsules [see Warnings and Precautions (5.1) ].
5.1 Elevated Liver Enzymes and Drug Induced Liver Injury
Cases of drug-induced liver injury (DILI) have been observed with pirfenidone. In the postmarketing period, non-serious and serious cases of DILI, including severe liver injury with fatal outcome, have been reported. Patients treated with pirfenidone 2403 mg/day in three Phase 3 trials had a higher incidence of elevations in ALT or AST ≥3 × ULN than placebo patients (3.7% vs. 0.8%, respectively). Elevations ≥10 × ULN in ALT or AST occurred in 0.3% of patients in the pirfenidone 2403 mg/day group and in 0.2% of patients in the placebo group. Increases in ALT and AST ≥3 × ULN were reversible with dose modification or treatment discontinuation.
Conduct liver function tests (ALT, AST, and bilirubin) prior to the initiation of therapy with pirfenidone, monthly for the first 6 months, every 3 months thereafter, and as clinically indicated. Measure liver function tests promptly in patients who report symptoms that may indicate liver injury, including fatigue, anorexia, right upper abdominal discomfort, dark urine, or jaundice. Dosage modification or interruption may be necessary for liver enzyme elevations [see Dosage and Administration (2.1, 2.3)].
Structured Label Content
Section 42229-5 (42229-5)
Dosage Modification due to Elevated Liver Enzymes
Dosage modifications or interruptions may also be necessary when liver enzyme and bilirubin elevations are exhibited. For liver enzyme elevations, modify the dosage as follows:
If a patient exhibits >3 but ≤5 × the upper limit of normal (ULN) ALT and/or AST without symptoms or hyperbilirubinemia after starting pirfenidone capsules therapy:
-
•Discontinue confounding medications, exclude other causes, and monitor the patient closely.
-
•Repeat liver chemistry tests as clinically indicated.
-
•The full daily dosage may be maintained, if clinically appropriate, or reduced or interrupted (e.g., until liver chemistry tests are within normal limits) with subsequent re-titration to the full dosage as tolerated.
If a patient exhibits >3 but ≤5 × ULN ALT and/or AST accompanied by symptoms or hyperbilirubinemia:
-
•Permanently discontinue pirfenidone capsules.
-
•Do not rechallenge patient with pirfenidone capsules.
If a patient exhibits >5 × ULN ALT and/or AST:
-
•Permanently discontinue pirfenidone capsules.
-
•Do not rechallenge patient with pirfenidone capsules.
Section 42230-3 (42230-3)
|
Patient Information
(pir-FEN-i-done) |
|
What are pirfenidone capsules?
|
|
Before you take pirfenidone capsules, tell your doctor about all of your medical conditions, including if you:
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. |
|
How should I take pirfenidone capsules?
|
|
What should I avoid while taking pirfenidone capsules?
|
|
What are the possible side effects of pirfenidone capsules? Pirfenidone capsules may cause serious side effects, including:
The most common side effects of pirfenidone capsules include feeling tired, insomnia, upper respiratory tract infections, sinusitis, headache, dizziness, decreased weight and decreased or loss of appetite. These are not all the possible side effects of pirfenidone capsules. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store pirfenidone capsules?
Safely throw away any pirfenidone capsules that is out of date or no longer needed. Keep pirfenidone capsules and all medicines out of reach of children. |
|
General information about the safe and effective use of pirfenidone capsules. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use pirfenidone capsules for a condition for which it was not prescribed. Do not give pirfenidone capsules to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or doctor for information about pirfenidone capsules that is written for health professionals. |
|
What are the ingredients in pirfenidone capsules? Active ingredient: pirfenidone Inactive ingredients: croscarmellose sodium, hypromellose, magnesium stearate, and pregelatinized starch. Capsule Shell: gelatin and titanium dioxide Capsule Black Printing Ink: iron oxide black, potassium hydroxide, propylene glycol, shellac and strong ammonia solution. For more information, contact Sandoz Inc., at 1-800-525-8747 Finished Drug Product Manufactured by Novartis d.o.o., 1000 Ljubljana, Slovenia for Sandoz Inc., Princeton, NJ 08540 |
This Patient Information has been approved by the U.S. Food and Drug Administration Rev. October 2024
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Keep the bottle tightly closed. Do not use if the seal over the bottle opening is broken or missing. Safely throw away any pirfenidone capsules that is out of date or no longer needed.
8.8 Smokers
Smoking causes decreased exposure to pirfenidone [see Clinical Pharmacology (12.3)], which may alter the efficacy profile of pirfenidone. Instruct patients to stop smoking prior to treatment with pirfenidone and to avoid smoking when using pirfenidone.
10 Overdosage (10 OVERDOSAGE)
There is limited clinical experience with overdosage. Multiple dosages of pirfenidone up to a maximum tolerated dose of 4005 mg per day were administered as five 267 mg capsules three times daily to healthy adult volunteers over a 12-day dose escalation.
In the event of a suspected overdosage, appropriate supportive medical care should be provided, including monitoring of vital signs and observation of the clinical status of the patient.
8.2 Lactation
Risk Summary
No information is available on the presence of pirfenidone in human milk, the effects of the drug on the breastfed infant, or the effects of the drug on milk production. The lack of clinical data during lactation precludes clear determination of the risk of pirfenidone to an infant during lactation; therefore, the developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for pirfenidone and the potential adverse effects on the breastfed child from pirfenidone or from the underlying maternal condition.
Data
Animal Data: A study with radio-labeled pirfenidone in rats has shown that pirfenidone or its metabolites are excreted in milk. There are no data on the presence of pirfenidone or its metabolites in human milk, the effects of pirfenidone on the breastfed child, or its effects on milk production.
11 Description (11 DESCRIPTION)
Pirfenidone belongs to the chemical class of pyridone. Pirfenidone capsules are available as white hard gelatin capsules containing 267 mg of pirfenidone for oral administration.
Pirfenidone has a molecular formula of C12H11NO and a molecular weight of 185.23. Pirfenidone has the following structural formula, which has been referred to as 5-methyl-1-phenyl-2-1(H)-pyridone or 5-methyl-1-phenyl-2-(1H)-pyridone.
Pirfenidone is a white to pale yellow crystalline, non-hygroscopic powder. It is more soluble in methanol, ethyl alcohol, acetone and chloroform than in water and 1.0 N HCl. The melting point is approximately 108°C.
Pirfenidone capsule contains pirfenidone and the following inactive ingredients: croscarmellose sodium, hypromellose, magnesium stearate, and pregelatinized starch.
In addition, the capsule shell contains gelatin and titanium dioxide. The capsule black printing ink includes iron oxide black, potassium hydroxide, propylene glycol, shellac and strong ammonia solution.
8.4 Pediatric Use
Safety and effectiveness of pirfenidone in pediatric patients have not been established.
8.5 Geriatric Use
Of the total number of subjects in the clinical studies receiving pirfenidone capsules, 714 (67%) were 65 years old and over, while 231 (22%) were 75 years old and over. No overall differences in safety or effectiveness were observed between older and younger patients. No dosage adjustment is required based upon age.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of pirfenidone was evaluated in patients with IPF in three phase 3, randomized, double-blind, placebo-controlled, multicenter trials (Studies 1, 2, and 3).
Study 1 was a 52-week trial comparing pirfenidone 2403 mg/day (n=278) versus placebo (n=277) in patients with IPF. Study 2 and Study 3 were nearly identical to each other in design, with few exceptions, including an intermediate dose treatment arm in Study 2. Study 2 compared treatment with either pirfenidone 2403 mg/day (n=174) or pirfenidone 1197 mg/day (n=87) to placebo (n=174), while Study 3 compared pirfenidone 2403 mg/day (n=171) to placebo (n=173). Study drug was administered three times daily with food for a minimum of 72 weeks. Patients continued on treatment until the last patient completed 72 weeks of treatment, which included observations to approximately 120 weeks of study treatment. The primary endpoint was the change in percent predicted forced vital capacity (%FVC) from baseline to study end, measured at 52 weeks in Study 1, and at 72 weeks in Studies 2 and 3.
Studies 1, 2 and 3 enrolled adult patients who had a clinical and radiographic diagnosis of IPF (with or without accompanying surgical lung biopsy), without evidence or suspicion of an alternative diagnosis for interstitial lung disease. Eligible patients were to have %FVC greater than or equal to 50% at baseline and a percent predicted diffusing capacity of the lungs for carbon monoxide (%DLCO) greater than or equal to 30% (Study 1) or 35% (Studies 2 and 3) at baseline. In all three trials, over 80% of patients completed study treatment.
A total of 1247 patients with IPF were randomized to receive pirfenidone 2403 mg/day (n=623) or placebo (n=624) in these three trials. Baseline characteristics were generally balanced across treatment groups. The study population ranged from 40 to 80 years of age (mean age 67 years). Most patients were male (74%), white (95%), and current or former smokers (65%). Approximately 93% of patients met criteria for definite IPF on high resolution computed tomography (HRCT). Baseline mean %FVC and %DLCO were 72% and 46%, respectively. Approximately 15% subjects discontinued from each treatment group.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in greater detail in other sections of the labeling:
-
•Liver Enzyme Elevations and Drug-Induced Liver Injury [see Warnings and Precautions (5.1)]
-
•Photosensitivity Reaction or Rash [see Warnings and Precautions (5.2)]
-
•Severe Cutaneous Adverse Reactions [see Warnings and Precautions (5.3)]
-
•Gastrointestinal Disorders [see Warnings and Precautions (5.4)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Moderate (e.g., ciprofloxacin) and strong inhibitors of CYP1A2 (e.g., fluvoxamine) increase systemic exposure of pirfenidone and may alter the adverse reaction profile of pirfenidone. Discontinue fluvoxamine prior to administration of pirfenidone or reduce to 267 mg three times a day. Consider dosage reduction with use of ciprofloxacin. (7.1)
7.2 Cyp1a2 Inducers (7.2 CYP1A2 Inducers)
The concomitant use of pirfenidone and a CYP1A2 inducer may decrease the exposure of pirfenidone and this may lead to loss of efficacy. Therefore, discontinue use of strong CYP1A2 inducers prior to pirfenidone treatment and avoid the concomitant use of pirfenidone and a strong CYP1A2 inducer [see Clinical Pharmacology (12.3)].
8.7 Renal Impairment
Pirfenidone should be used with caution in patients with mild (CLcr 50 to 80 mL/min), moderate (CLcr 30 to 50 mL/min), or severe (CLcr less than 30 mL/min) renal impairment [see Clinical Pharmacology (12.3)]. Monitor for adverse reactions and consider dosage modification or discontinuation of pirfenidone as needed [see Dosage and Administration (2.3)]. The safety, efficacy, and pharmacokinetics of pirfenidone have not been studied in patients with end-stage renal disease requiring dialysis. Use of pirfenidone in patients with end-stage renal diseases requiring dialysis is not recommended.
12.2 Pharmacodynamics
Cardiac Electrophysiology
The effect of pirfenidone on QT interval was evaluated in a randomized, placebo, and positive controlled parallel study in 160 healthy adult volunteers. Volunteers received pirfenidone 2403 mg/day (recommended dose) and 4005 mg/day (1.6 times recommended dose) or placebo for 10 days or a single dose of 400 mg moxifloxacin (active control).
Relative to placebo, the maximum mean change from baseline in study-specific QT interval was 3.2 milliseconds (ms) and 2.2 ms for pirfenidone 2403 mg/day and 4005 mg/day, respectively. No volunteer had a QTc interval greater than 480 ms or change from baseline greater than 60 ms. Although there was no evidence that pirfenidone prolonged the QTc interval in this study, a definitive conclusion may not be drawn as the positive control (moxifloxacin) did not perform as expected in this study, and pirfenidone at 4005 mg/day (1.7 times the maximum recommended dose) did not cover the maximum pirfenidone exposure increase with co-administration of fluvoxamine, a strong CYP1A2 inhibitor.
7.1 Cyp1a2 Inhibitors (7.1 CYP1A2 Inhibitors)
Pirfenidone is metabolized primarily (70 to 80%) via CYP1A2 with minor contributions from other CYP isoenzymes including CYP2C9, 2C19, 2D6 and 2E1.
2.2 Recommended Dosage
The recommended daily maintenance dosage of pirfenidone capsules are 801 mg three times daily for a total of 2403 mg/day. Doses should be taken with food at the same time each day.
Upon initiation of treatment, titrate to the full dosage of 2403 mg/day over a 14-day period as follows:
| Treatment days | Dosage |
|---|---|
|
Days 1 through 7 |
267 mg three times daily (801 mg/day) |
|
Days 8 through 14 |
534 mg three times daily (1602 mg/day) |
|
Days 15 onward |
801 mg three times daily (2403 mg/day) |
Dosages above 2403 mg/day are not recommended for any patient. Patients should not take 2 doses at the same time to make up for a missed dose. Patients should not take more than 3 doses per day.
8.6 Hepatic Impairment
Pirfenidone should be used with caution in patients with mild (Child Pugh Class A) to moderate (Child Pugh Class B) hepatic impairment. Monitor for adverse reactions and consider dosage modification or discontinuation of pirfenidone as needed [see Dosage and Administration (2.3)].
The safety, efficacy, and pharmacokinetics of pirfenidone have not been studied in patients with severe hepatic impairment. Pirfenidone is not recommended for use in patients with severe (Child Pugh Class C) hepatic impairment [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Pirfenidone capsules are indicated for the treatment of idiopathic pulmonary fibrosis (IPF).
Principal Display Panel
NDC 0781-2158-32
Pirfenidone Capsules
267 mg
Rx Only
270 Capsules
SANDOZ
12.1 Mechanism of Action
The mechanism of action of pirfenidone in the treatment of IPF has not been established.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Elevated liver enzymes and drug-induced liver injury: ALT, AST, and bilirubin elevations have occurred with pirfenidone including cases of drug-induced liver injury. In the postmarketing setting, non-serious and serious cases of drug-induced liver injury, including severe liver injury with fatal outcomes, have been reported. Monitor ALT, AST, and bilirubin before and during treatment. Temporary dosage reductions or discontinuations may be required. (2.1, 5.1)
-
•Photosensitivity and rash: Photosensitivity and rash have been noted with pirfenidone. Avoid exposure to sunlight and sunlamps. Wear sunscreen and protective clothing daily. Temporary dosage reductions or discontinuations may be required. (5.2)
-
•Severe Cutaneous Adverse Reactions (SCAR): Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug reactions with eosinophilia and systemic symptoms (DRESS) have been reported in association with the use of pirfenidone in the postmarketing setting. Interrupt pirfenidone in case of signs or symptoms of SCAR. Permanently discontinue pirfenidone if a SCAR is confirmed. (5.3)
-
•Gastrointestinal disorders: Nausea, vomiting, diarrhea, dyspepsia, gastro-esophageal reflux disease, and abdominal pain have occurred with pirfenidone. Temporary dosage reductions or discontinuations may be required. (5.4)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
-
•Take with food.
-
•Recommended dosage: 801 mg three times daily (2403 mg/day). (2)
-
•Upon initiation of treatment, titrate to the full dosage of 2403 mg/day over a 14-day period as follows:
| Treatment days | Dosage |
|---|---|
|
Days 1 through 7 |
267 mg three times daily (801 mg/day) |
|
Days 8 through 14 |
534 mg three times daily (1602 mg/day) |
|
Days 15 onward |
801 mg three times daily (2403 mg/day) |
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Capsules: 267 mg, White hard gelatin capsules printed with PN267 on the cap of the capsule in black ink.
6.2 Postmarketing Experience
In addition to adverse reactions identified from clinical trials the following adverse reactions have been identified during post-approval use of pirfenidone. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency.
Blood and Lymphatic System Disorders: Agranulocytosis
Hepatobiliary Disorders: Drug-induced liver injury
Immune System Disorders: Angioedema
Skin and Subcutaneous Tissue Disorders: Severe Cutaneous Adverse Reactions (SCAR)
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
-
•Hepatic Impairment: Monitor for adverse reactions and consider dosage modification or discontinuation of pirfenidone as needed. Pirfenidone is not recommended for use in patients with severe hepatic impairment. (8.6, 12.3)
-
•Renal Impairment: Monitor for adverse reactions and consider dosage modification or discontinuation of pirfenidone as needed. Pirfenidone is not recommended for use in patients with end stage renal disease on dialysis. (8.7, 12.3)
-
•Smokers: Decreased exposure has been noted in smokers which may alter the efficacy profile of pirfenidone. (8.8)
5.4 Gastrointestinal Disorders
In the clinical studies, gastrointestinal events of nausea, diarrhea, dyspepsia, vomiting, gastro-esophageal reflux disease, and abdominal pain were more frequently reported by patients in the pirfenidone treatment groups than in those taking placebo. Dosage reduction or interruption for gastrointestinal events was required in 18.5% of patients in the 2403 mg/day group, as compared to 5.8% of patients in the placebo group; 2.2% of patients in the pirfenidone 2403 mg/day group discontinued treatment due to a gastrointestinal event, as compared to 1.0% in the placebo group. The most common (>2%) gastrointestinal events that led to dosage reduction or interruption were nausea, diarrhea, vomiting, and dyspepsia. The incidence of gastrointestinal events was highest early in the course of treatment (with highest incidence occurring during the initial 3 months) and decreased over time. Dosage modifications may be necessary in some cases of gastrointestinal adverse reactions [see Dosage and Administration (2.3)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of pirfenidone has been evaluated in more than 1400 subjects with over 170 subjects exposed to pirfenidone for more than 5 years in clinical trials.
Pirfenidone was studied in 3 randomized, double-blind, placebo-controlled trials (Studies 1, 2, and 3) in which a total of 623 patients received 2403 mg/day of pirfenidone and 624 patients received placebo. Subjects ages ranged from 40 to 80 years (mean age of 67 years). Most patients were male (74%) and Caucasian (95%). The mean duration of exposure to pirfenidone was 62 weeks (range: 2 to 118 weeks) in these 3 trials.
At the recommended dosage of 2403 mg/day, 14.6% of patients on pirfenidone compared to 9.6% on placebo permanently discontinued treatment because of an adverse event. The most common (>1%) adverse reactions leading to discontinuation were rash and nausea. The most common (>3%) adverse reactions leading to dosage reduction or interruption were rash, nausea, diarrhea, and photosensitivity reaction.
The most common adverse reactions with an incidence of ≥10% and more frequent in the pirfenidone than placebo treatment group are listed in Table 2 .
-
Table 2. Adverse Reactions Occurring in ≥10% of Pirfenidone-Treated Patients and More Commonly Than Placebo in Studies 1, 2, and 3
| Adverse Reaction | % of Patients (0 to 118 Weeks) | |
|---|---|---|
|
Pirfenidone 2403 mg/day
(N = 623) |
Placebo
(N = 624) |
|
|
Nausea |
36% |
16% |
|
Rash |
30% |
10% |
|
Abdominal Pain Includes abdominal pain, upper abdominal pain, abdominal distension, and stomach discomfort.
|
24% |
15% |
|
Upper Respiratory Tract Infection |
27% |
25% |
|
Diarrhea |
26% |
20% |
|
Fatigue |
26% |
19% |
|
Headache |
22% |
19% |
|
Decreased Appetite |
21% |
8% |
|
Dyspepsia |
19% |
7% |
|
Dizziness |
18% |
11% |
|
Vomiting |
13% |
6% |
|
Gastro-esophageal Reflux Disease |
11% |
7% |
|
Sinusitis |
11% |
10% |
|
Insomnia |
10% |
7% |
|
Weight Decreased |
10% |
5% |
|
Arthralgia |
10% |
7% |
Adverse reactions occurring in ≥5 to <10% of pirfenidone-treated patients and more commonly than placebo are photosensitivity reaction (9% vs. 1%), pruritus (8% vs. 5%), asthenia (6% vs. 4%), dysgeusia (6% vs. 2%), and non-cardiac chest pain (5% vs. 4%).
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling ( Patient Information ).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Pirfenidone capsules are white hard gelatin capsules contain 267 mg of pirfenidone. The cap of the capsule is printed with "PN267" in black ink. The capsules are supplied as follows:
-
NDC 0781-2158-32, bottle for a 30-day supply containing 270 capsules and closed with child-resistant closure
5.2 Photosensitivity Reaction Or Rash (5.2 Photosensitivity Reaction or Rash)
Patients treated with pirfenidone 2403 mg/day in the three Phase 3 studies had a higher incidence of photosensitivity reactions (9%) compared with patients treated with placebo (1%). The majority of the photosensitivity reactions occurred during the initial 6 months. Instruct patients to avoid or minimize exposure to sunlight (including sunlamps), to use a sunblock (SPF 50 or higher), and to wear clothing that protects against sun exposure. Additionally, instruct patients to avoid concomitant medications known to cause photosensitivity. Dosage reduction or discontinuation may be necessary in some cases of photosensitivity reaction or rash [see Dosage and Administration (2.3) ].
5.3 Severe Cutaneous Adverse Reactions
Severe cutaneous adverse reactions (SCAR), including Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and drug reaction with eosinophilia and systemic symptoms (DRESS), have been reported in association with the use of pirfenidone in the postmarketing setting. If signs or symptoms of SCAR occur, interrupt pirfenidone treatment until the etiology of the reaction has been determined. Consultation with a dermatologist is recommended. If a SCAR is confirmed, permanently discontinue pirfenidone.
2.3 Dosage Modifications Due to Adverse Reactions (2.3 Dosage Modifications due to Adverse Reactions)
Patients who miss 14 or more days of pirfenidone capsules should re-initiate treatment by undergoing the initial 2-week titration regimen up to the full maintenance dosage [see Dosage and Administration (2.2) ]. For treatment interruption of less than 14 days, the dosage prior to the interruption can be resumed.
If patients experience significant adverse reactions (i.e., gastrointestinal, photosensitivity reaction or rash, severe cutaneous adverse reactions (SCAR)), consider temporary dosage reductions or interruptions of pirfenidone capsules to allow for resolution of symptoms. If a SCAR is confirmed, permanently discontinue pirfenidone capsules [see Warnings and Precautions (5.1, 5.2, 5.3, 5.4)] .
2.4 Dosage Modifications Due to Drug Interactions (2.4 Dosage Modifications due to Drug Interactions)
Strong CYP1A2 Inhibitors (e.g., fluvoxamine, enoxacin)
Reduce pirfenidone capsules to 267 mg three times a day (801 mg/day).
Moderate CYP1A2 Inhibitors (e.g., ciprofloxacin)
With use of ciprofloxacin at a dosage of 750 mg twice daily, reduce pirfenidone capsules to 534 mg three times a day (1602 mg/day).
2.1 Testing Prior to Pirfenidone Capsules Administration
Conduct liver function tests prior to initiating treatment with pirfenidone capsules [see Warnings and Precautions (5.1) ].
5.1 Elevated Liver Enzymes and Drug Induced Liver Injury (5.1 Elevated Liver Enzymes and Drug-Induced Liver Injury)
Cases of drug-induced liver injury (DILI) have been observed with pirfenidone. In the postmarketing period, non-serious and serious cases of DILI, including severe liver injury with fatal outcome, have been reported. Patients treated with pirfenidone 2403 mg/day in three Phase 3 trials had a higher incidence of elevations in ALT or AST ≥3 × ULN than placebo patients (3.7% vs. 0.8%, respectively). Elevations ≥10 × ULN in ALT or AST occurred in 0.3% of patients in the pirfenidone 2403 mg/day group and in 0.2% of patients in the placebo group. Increases in ALT and AST ≥3 × ULN were reversible with dose modification or treatment discontinuation.
Conduct liver function tests (ALT, AST, and bilirubin) prior to the initiation of therapy with pirfenidone, monthly for the first 6 months, every 3 months thereafter, and as clinically indicated. Measure liver function tests promptly in patients who report symptoms that may indicate liver injury, including fatigue, anorexia, right upper abdominal discomfort, dark urine, or jaundice. Dosage modification or interruption may be necessary for liver enzyme elevations [see Dosage and Administration (2.1, 2.3)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:45.413456 · Updated: 2026-03-14T22:30:51.485924