73288-003
94043f4e-fe47-4b6f-8354-30697daaed81
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
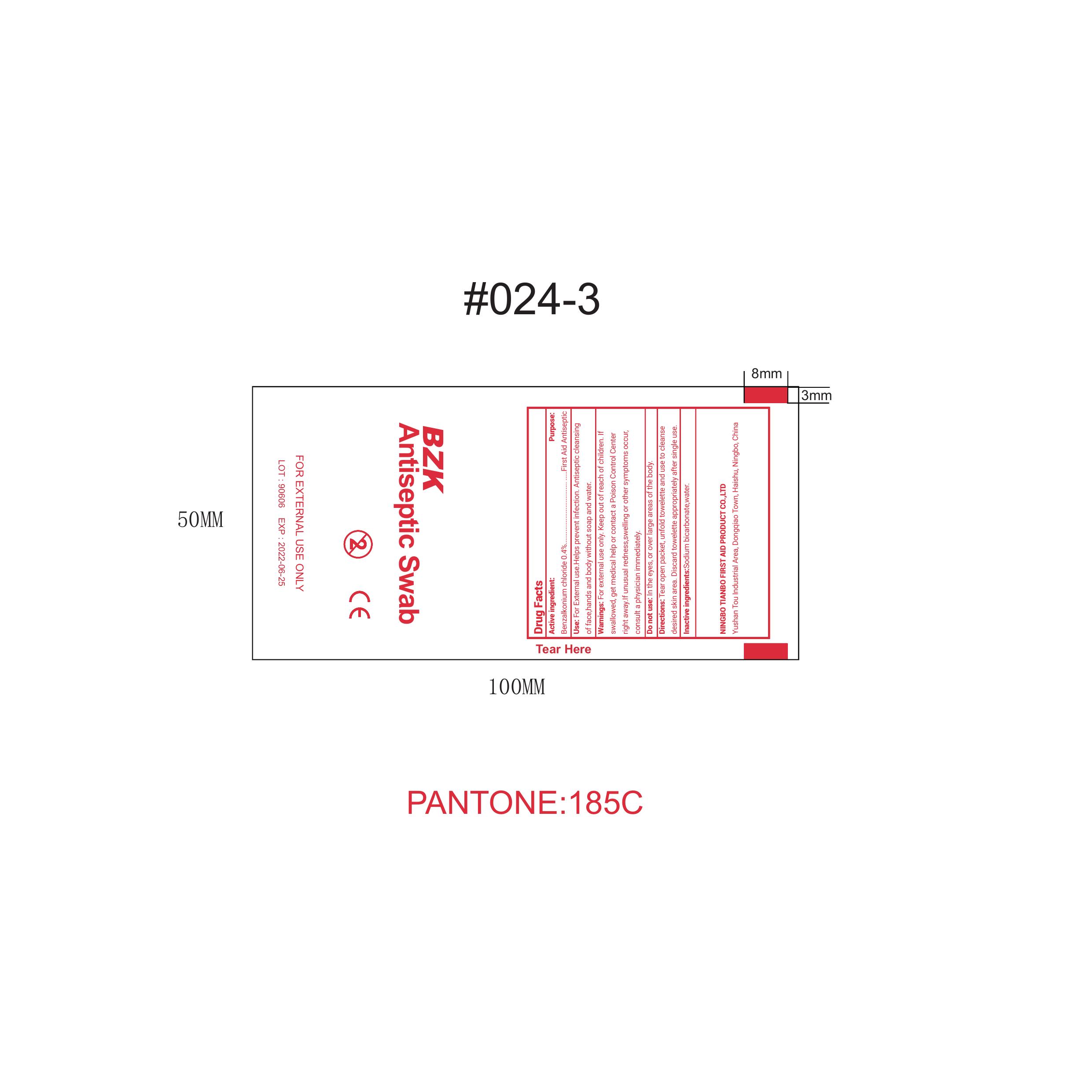
Active Ingredient: Benzalkonium Chloride 0.4%
Medication Information
Warnings and Precautions
Warnings:
For external use only
Indications and Usage
Uses: For external use. Help prevent infection. Antiseptic cleasing of face, hand, and body withour soap and water.
Dosage and Administration
Directions: Tear open packet, unfold towelette and use to clease desire skin area. Discard towelette appropriately after single use.
Description
Active Ingredient: Benzalkonium Chloride 0.4%
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away. If usual redness, swelling, or other symptoms occur, consult a physician immediately.
Section 51727-6
Inactive Ingredient
Sodium Bicarbonate, Water
Section 51945-4
Section 55105-1
Purpose: First Aide Antiseptic
Section 55106-9
Active Ingredient: Benzalkonium Chloride 0.4%
Section 60561-8
Do not use in the eyes or over large area of the body.
Structured Label Content
Indications and Usage (34067-9)
Uses: For external use. Help prevent infection. Antiseptic cleasing of face, hand, and body withour soap and water.
Dosage and Administration (34068-7)
Directions: Tear open packet, unfold towelette and use to clease desire skin area. Discard towelette appropriately after single use.
Warnings and Precautions (34071-1)
Warnings:
For external use only
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away. If usual redness, swelling, or other symptoms occur, consult a physician immediately.
Section 51727-6 (51727-6)
Inactive Ingredient
Sodium Bicarbonate, Water
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Purpose: First Aide Antiseptic
Section 55106-9 (55106-9)
Active Ingredient: Benzalkonium Chloride 0.4%
Section 60561-8 (60561-8)
Do not use in the eyes or over large area of the body.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:11.610411 · Updated: 2026-03-14T23:12:21.703136