Letrozole Tablets, Usp
93f317f1-96d1-498e-e053-2995a90ac60d
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Contraindications ( 4 ) 7/2017 Warnings and Precautions, Embryo-Fetal Toxicity ( 5.6 ) 7/2017
Indications and Usage
Letrozole tablets are an aromatase inhibitor indicated for: Adjuvant treatment of postmenopausal women with hormone receptor positive early breast cancer ( 1.1 ) Extended adjuvant treatment of postmenopausal women with early breast cancer who have received prior standard adjuvant tamoxifen therapy ( 1.2 ) First and second-line treatment of postmenopausal women with hormone receptor positive or unknown advanced breast cancer ( 1.3 )
Dosage and Administration
Letrozole tablets are taken orally without regard to meals ( 2 ): Recommended dose: 2.5 mg once daily ( 2.1 ) Patients with cirrhosis or severe hepatic impairment: 2.5 mg every other day ( 2.5 , 5.3 )
Warnings and Precautions
Decreases in bone mineral density may occur. Consider bone mineral density monitoring ( 5.1 ) Increases in total cholesterol may occur. Consider cholesterol monitoring. ( 5.2 ) Fatigue, dizziness and somnolence may occur. Exercise caution when operating machinery ( 5.4 ) Embryo-Fetal toxicity: Can cause fetal harm when administered to pregnant women. Obtain a pregnancy test in females of reproductive potential. Advise females of reproductive potential to use effective contraception ( 5.6 , 8.1 , 8.3 )
Contraindications
Pregnancy: Letrozole can cause fetal harm [see Use in Specific Populations (8.1) ] . Pregnancy: Letrozole can cause fetal harm [see Use in Specific Populations (8.1) ] . Known hypersensitivity to the active substance, or to any of the excipients [see Adverse Reactions (6) ] . Known hypersensitivity to the active substance, or to any of the excipients [see Adverse Reactions (6) ] .
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling. Bone effects [see Warnings and Precautions (5.1) ] Increases in cholesterol [see Warnings and Precautions (5.2) ] Fatigue and Dizziness [see Warnings and Precautions (5.4) ]
Storage and Handling
Dispensed in Unit Dose Package. For Institutional Use Only. 2.5 milligram tablets NDC 50268-476-15 (10 tablets per card, 5 cards per carton)
How Supplied
Dispensed in Unit Dose Package. For Institutional Use Only. 2.5 milligram tablets NDC 50268-476-15 (10 tablets per card, 5 cards per carton)
Medication Information
Warnings and Precautions
Decreases in bone mineral density may occur. Consider bone mineral density monitoring ( 5.1 ) Increases in total cholesterol may occur. Consider cholesterol monitoring. ( 5.2 ) Fatigue, dizziness and somnolence may occur. Exercise caution when operating machinery ( 5.4 ) Embryo-Fetal toxicity: Can cause fetal harm when administered to pregnant women. Obtain a pregnancy test in females of reproductive potential. Advise females of reproductive potential to use effective contraception ( 5.6 , 8.1 , 8.3 )
Indications and Usage
Letrozole tablets are an aromatase inhibitor indicated for: Adjuvant treatment of postmenopausal women with hormone receptor positive early breast cancer ( 1.1 ) Extended adjuvant treatment of postmenopausal women with early breast cancer who have received prior standard adjuvant tamoxifen therapy ( 1.2 ) First and second-line treatment of postmenopausal women with hormone receptor positive or unknown advanced breast cancer ( 1.3 )
Dosage and Administration
Letrozole tablets are taken orally without regard to meals ( 2 ): Recommended dose: 2.5 mg once daily ( 2.1 ) Patients with cirrhosis or severe hepatic impairment: 2.5 mg every other day ( 2.5 , 5.3 )
Contraindications
Pregnancy: Letrozole can cause fetal harm [see Use in Specific Populations (8.1) ] . Pregnancy: Letrozole can cause fetal harm [see Use in Specific Populations (8.1) ] . Known hypersensitivity to the active substance, or to any of the excipients [see Adverse Reactions (6) ] . Known hypersensitivity to the active substance, or to any of the excipients [see Adverse Reactions (6) ] .
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling. Bone effects [see Warnings and Precautions (5.1) ] Increases in cholesterol [see Warnings and Precautions (5.2) ] Fatigue and Dizziness [see Warnings and Precautions (5.4) ]
Storage and Handling
Dispensed in Unit Dose Package. For Institutional Use Only. 2.5 milligram tablets NDC 50268-476-15 (10 tablets per card, 5 cards per carton)
How Supplied
Dispensed in Unit Dose Package. For Institutional Use Only. 2.5 milligram tablets NDC 50268-476-15 (10 tablets per card, 5 cards per carton)
Description
Contraindications ( 4 ) 7/2017 Warnings and Precautions, Embryo-Fetal Toxicity ( 5.6 ) 7/2017
Section 42229-5
Adjuvant Treatment of Early Breast Cancer
In study, BIG 1-98, the median treatment duration of adjuvant treatment was 60 months and the median duration of follow-up for safety was 96 months for patients receiving letrozole tablets and tamoxifen.
Certain adverse reactions were prospectively specified for analysis (see Table 1), based on the known pharmacologic properties and side effect profiles of the two drugs.
Adverse reactions were analyzed irrespective of whether a symptom was present or absent at baseline. Most adverse reactions reported (approximately 75% of patients who reported AEs) were Grade 1 or Grade 2 applying the Common Toxicity Criteria (CTC)Version 2.0/Common Terminology Criteria for Adverse Events (CTCAE), version 3.0. Table 1 describes adverse reactions (Grades 1-4 and Grades 3-4) irrespective of relationship to study treatment in the adjuvant trial for the monotherapy arms analysis (safety population).
| Adverse Reactions | Grades 1-4 | Grades 3-4 | ||||||
|---|---|---|---|---|---|---|---|---|
| Letrozole tablets
N=2448 n (%) |
Tamoxifen
N=2447 n (%) |
Letrozole tablets
N=2448 n (%) |
Tamoxifen
N=2447 n (%) |
|||||
| TIA = Transient ischemic attack | ||||||||
| Note: Cardiovascular events (including cerebrovascular and thromboembolic events), skeletal and urogenital/endometrial events and second primary malignancies were collected life -long. All of these events were assumed to be of CTC Grade 3 to 5 and were not individually graded | ||||||||
| Patients with any adverse reaction | 2309 | (94.3) | 2212 | (90.4) | 636 | (26.0) | 606 | (24.8) |
| Hypercholesterolemia
Target events pre-specified for analysis
|
1280 | (52.3) | 700 | (28.6) | 11 | (0.4) | 6 | (0.2) |
| Hot flashes | 819 | (33.5) | 929 | (38.0) | - | - | - | - |
| Arthralgia/arthritis | 621 | (25.4) | 504 | (20.6) | 84 | (3.4) | 50 | (2.0) |
| Bone fractures
At median follow-up of 96 months (i.e. any time after randomization) for letrozole tablets (range up to 144 months) and 95 months for tamoxifen (range up to 143 months )
|
361 | (14.7) | 280 | (11.4) | - | - | - | - |
| Night sweats | 356 | (14.5) | 426 | (17.4) | - | - | - | - |
| Weight increase | 317 | (12.9) | 378 | (15.4) | 27 | (1.1) | 39 | (1.6) |
| Nausea | 284 | (11.6) | 277 | (11.3) | 6 | (0.2) | 9 | (0.4) |
| Bone fractures
Events pre-printed on CRF
At median treatment duration of 60 months (i.e. during treatment + 30 days after discontinuation of treatment) for letrozole tablets and tamoxifen (range up to 68 months)
|
249 | (10.2) | 175 | (7.2) | - | - | - | - |
| Fatigue (lethargy, malaise, asthenia) | 235 | (9.6) | 250 | (10.2) | 6 | (0.2) | 7 | (0.3) |
| Myalgia | 221 | (9.0) | 212 | (8.7) | 18 | (0.7) | 14 | (0.6) |
| Vaginal bleeding | 129 | (5.3) | 320 | (13.1) | 1 | (<0.1) | 8 | (0.3) |
| Edema | 164 | (6.7) | 160 | (6.5) | 3 | (0.1) | 1 | (<0.1) |
| Weight decrease | 140 | (5.7) | 129 | (5.3) | 8 | (0.3) | 5 | (0.2) |
| Osteoporosis | 126 | (5.1) | 67 | (2.7) | 10 | (0.4) | 5 | (0.2) |
| Back pain | 125 | (5.1) | 136 | (5.6) | 7 | (0.3) | 11 | (0.4) |
| Bone pain | 123 | (5.0) | 109 | (4.5) | 6 | (0.2) | 4 | (0.2) |
| Depression | 119 | (4.9) | 114 | (4.7) | 16 | (0.7) | 14 | (0.6) |
| Vaginal irritation | 112 | (4.6) | 77 | (3.1) | 2 | (<0.1) | 2 | (<0.1) |
| Headache | 105 | (4.3) | 94 | (3.8) | 8 | (0.3) | 4 | (0.2) |
| Pain in extremity | 103 | (4.2) | 79 | (3.2) | 6 | (0.2) | 4 | (0.2) |
| Osteopenia | 87 | (3.6) | 76 | (3.1) | 0 | - | 3 | (0.1) |
| Dizziness/light-headedness | 84 | (3.4) | 80 | (3.3) | 1 | (<0.1) | 6 | (0.2) |
| Alopecia | 83 | (3.4) | 84 | (3.4) | - | - | - | - |
| Vomiting | 80 | (3.3) | 80 | (3.3) | 3 | (0.1) | 5 | (0.2) |
| Cataract | 49 | (2.0) | 54 | (2.2) | 16 | (0.7) | 17 | (0.7) |
| Constipation | 49 | (2.0) | 71 | (2.9) | 3 | (0.1) | 1 | (<0.1) |
| Myocardial infarction | 42 | (1.7) | 28 | (1.1) | - | - | - | - |
| Breast pain | 37 | (1.5) | 43 | (1.8) | 1 | (<0.1) | - | - |
| Anorexia | 20 | (0.8) | 20 | (0.8) | 1 | (<0.1) | 1 | (<0.1) |
| Endometrial proliferation disorders | 14 | (0.6) | 86 | (3.5) | 0 | - | 14 | (0.6) |
| Ovarian cyst | 11 | (0.4) | 18 | (0.7) | 4 | (0.2) | 4 | (0.2) |
| Endometrial hyperplasia/cancer | 11 | (0.4) | 72 | (2.9) | - | - | - | - |
| Endometrial hyperplasia/cancer
,
Excluding women who had undergone hysterectomy before study entry
|
6/1909 | (0.3) | 57/1943 | (2.9) | - | - | - | - |
| Other endometrial disorders | 2 | (<0.1) | 3 | (0.1) | 0 | - | 0 | - |
| Myocardial infarction | 24 | (1.0) | 12 | (0.5) | - | - | - | - |
| Myocardial ischemia | 6 | (0.2) | 9 | (0.4) | - | - | - | - |
| Cerebrovascular accident/TIA | 74 | (3.0) | 68 | (2.8) | - | - | - | - |
| Cerebrovascular accident/TIA | 51 | (2.1) | 47 | (1.9) | - | - | - | - |
| Angina requiring surgery | 35 | (1.4) | 33 | (1.3) | - | - | - | - |
| Angina requiring surgery | 25 | (1.0) | 25 | (1.0) | - | - | - | - |
| Thromboembolic event | 79 | (3.2) | 113 | (4.6) | - | - | - | - |
| Thromboembolic event | 51 | (2.1) | 89 | (3.6) | - | - | - | - |
| Cardiac failure | 39 | (1.6) | 34 | (1.4) | - | - | - | - |
| Cardiac failure | 27 | (1.1) | 15 | (0.6) | - | - | - | - |
| Hypertension | 160 | (6.5) | 175 | (7.2) | - | - | - | - |
| Hypertension | 138 | (5.6) | 139 | (5.7) | - | - | - | - |
| Other cardiovascular | 172 | (7.0) | 174 | (7.1) | - | - | - | - |
| Other cardiovascular | 120 | (4.9) | 119 | (4.9) | - | - | - | - |
| Second primary malignancy | 129 | (5.3) | 150 | (6.1) | - | - | - | - |
| Second primary malignancy | 54 | (2.2) | 79 | (3.2) | - | - | - | - |
When considering all grades during study treatment, a higher incidence of events was seen for letrozole tablets regarding fractures (10.1% vs 7.1%), myocardial infarctions (1.0% vs 0.5%), and arthralgia (25.2% vs 20.4%) (letrozole tablets vs tamoxifen respectively). A higher incidence was seen for tamoxifen regarding thromboembolic events (2.1% vs 3.6%), endometrial hyperplasia/cancer (0.3% vs 2.9%), and endometrial proliferation disorders (0.3% vs 1.8%) (letrozole tablets vs tamoxifen respectively).
At a median follow-up of 96 months, a higher incidence of events was seen for letrozole tablets (14.7%) than for tamoxifen (11.4%) regarding fractures. A higher incidence was seen for tamoxifen compared to letrozole tablets regarding thromboembolic events (4.6% vs 3.2%), and endometrial hyperplasia or cancer (2.9% vs 0.4%) (tamoxifen vs letrozole tablets, respectively).
Section 43683-2
Section 44425-7
Store at 25°C (77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
10 Overdosage
Isolated cases of letrozole tablets overdose have been reported. In these instances, the highest single dose ingested was 62.5 mg or 25 tablets. While no serious adverse reactions were reported in these cases, because of the limited data available, no firm recommendations for treatment can be made. However, emesis could be induced if the patient is alert. In general, supportive care and frequent monitoring of vital signs are also appropriate. In single-dose studies, the highest dose used was 30 mg, which was well tolerated; in multiple-dose trials, the largest dose of 10 mg was well tolerated.
Lethality was observed in mice and rats following single oral doses that were equal to or greater than 2,000 mg/kg (about 4,000 to 8,000 times the daily maximum recommended human dose on a mg/m 2 basis); death was associated with reduced motor activity, ataxia and dyspnea. Lethality was observed in cats following single IV doses that were equal to or greater than 10 mg/kg (about 50 times the daily maximum recommended human dose on a mg/m 2 basis); death was preceded by depressed blood pressure and arrhythmias.
11 Description
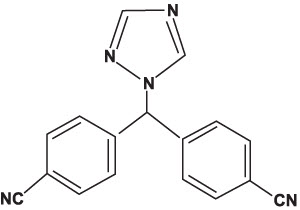
Letrozole tablets for oral administration contain 2.5 mg of letrozole, a nonsteroidal aromatase inhibitor (inhibitor of estrogen synthesis). It is chemically described as 4,4'-(1H-1,2,4-Triazol-1-ylmethylene)dibenzonitrile, and its structural formula is
Letrozole is a white to yellowish crystalline powder, practically odorless, freely soluble in dichloromethane, slightly soluble in ethanol, and practically insoluble in water. It has a molecular weight of 285.31, empirical formula C 17H 11N 5, and a melting range of 184°C to 185°C.
Letrozole tablets are available as 2.5 mg tablets for oral administration.
Inactive Ingredients: Colloidal silicon dioxide, ferric oxide (yellow), hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, pregelatinized starch, polyethylene glycol, sodium starch glycolate, talc and titanium dioxide.
5.2 Cholesterol
Consideration should be given to monitoring serum cholesterol. In the adjuvant trial (BIG 1-98), hypercholesterolemia was reported in 52.3% of letrozole patients and 28.6% of tamoxifen patients. Grade 3-4 hypercholesterolemia was reported in 0.4% of letrozole patients and 0.1% of tamoxifen patients. Also in the adjuvant setting, an increase of greater than or equal to 1.5 × upper limit of normal (ULN) in total cholesterol (generally nonfasting) was observed in patients on monotherapy who had baseline total serum cholesterol within the normal range (i.e., less than =1.5 × ULN) in 155/1843 (8.4%) patients on letrozole vs 71/1840 (3.9%) patients on tamoxifen. Lipid lowering medications were required for 29% of patients on letrozole and 20% on tamoxifen [see Adverse Reactions (6)] .
5.1 Bone Effects
Use of letrozole tablets may cause decreases in bone mineral density (BMD). Consideration should be given to monitoring BMD. Results of a safety study to evaluate safety in the adjuvant setting comparing the effect on lumbar spine (L2-L4) BMD of adjuvant treatment with letrozole to that with tamoxifen showed at 24 months a median decrease in lumbar spine BMD of 4.1% in the letrozole arm compared to a median increase of 0.3% in the tamoxifen arm (difference = 4.4%) ( P<0.0001) [see Adverse Reactions (6)] . Updated results from the BMD substudy (MA-17B) in the extended adjuvant setting demonstrated that at 2 years patients receiving letrozole had a median decrease from baseline of 3.8% in hip BMD compared to a median decrease of 2.0% in the placebo group. The changes from baseline in lumbar spine BMD in letrozole and placebo treated groups were not significantly different [see Adverse Reactions (6)].
In the adjuvant trial (BIG 1-98) the incidence of bone fractures at any time after randomization was 14.7% for letrozole and 11.4% for tamoxifen at a median follow-up of 96 months. The incidence of osteoporosis was 5.1% for letrozole and 2.7% for tamoxifen [see Adverse Reactions (6)] . In the extended adjuvant trial (MA-17), the incidence of bone fractures at any time after randomization was 13.3% for letrozole and 7.8% for placebo at a median follow-up of 62 months. The incidence of new osteoporosis was 14.5% for letrozole and 7.8% for placebo [see Adverse Reactions (6)] .
8.4 Pediatric Use
The safety and effectiveness in pediatric patients have not been established.
Letrozole administration to young (postnatal day 7) rats for 12 weeks duration at 0.003, 0.03, 0.3 mg/kg/day by oral gavage resulted in adverse skeletal/growth effects (bone maturation, bone mineral density) and neuroendocrine and reproductive developmental perturbations of the hypothalamic-pituitary axis. Administration of 0.3 mg/kg/day resulted in AUC values that were similar to the AUC in adult patients receiving the recommended dose of 2.5 mg/day. Decreased fertility was accompanied by hypertrophy of the hypophysis and testicular changes that included degeneration of the seminiferous tubular epithelium and atrophy of the female reproductive tract. Young rats in this study were allowed to recover following discontinuation of letrozole treatment for 42 days. Histopathological changes were not reversible at clinically relevant exposures.
8.5 Geriatric Use
The median age of patients in all studies of first-line and second-line treatment of metastatic breast cancer was 64-65 years. About 1/3 of the patients were greater than or equal to 70 years old. In the first-line study, patients greater than or equal to 70 years of age experienced longer time to tumor progression and higher response rates than patients less than 70.
For the extended adjuvant setting (MA-17), more than 5,100 postmenopausal women were enrolled in the clinical study. In total, 41% of patients were aged 65 years or older at enrollment, while 12% were 75 or older. In the extended adjuvant setting, no overall differences in safety or efficacy were observed between these older patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
In the adjuvant setting (BIG 1-98), more than 8,000 postmenopausal women were enrolled in the clinical study. In total, 36% of patients were aged 65 years or older at enrollment, while 12% were 75 or older. More adverse reactions were generally reported in elderly patients irrespective of study treatment allocation. However, in comparison to tamoxifen, no overall differences with regard to the safety and efficacy profiles were observed between elderly patients and younger patients.
4 Contraindications
- Pregnancy: Letrozole can cause fetal harm [see Use in Specific Populations (8.1)] . Pregnancy: Letrozole can cause fetal harm [see Use in Specific Populations (8.1)] .
- Known hypersensitivity to the active substance, or to any of the excipients [see Adverse Reactions (6)] . Known hypersensitivity to the active substance, or to any of the excipients [see Adverse Reactions (6)] .
6 Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the labeling.
- Bone effects [see Warnings and Precautions (5.1)]
- Increases in cholesterol [see Warnings and Precautions (5.2)]
- Fatigue and Dizziness [see Warnings and Precautions (5.4)]
2.1 Recommended Dose
The recommended dose of letrozole tablets is one 2.5 mg tablet administered once a day, without regard to meals.
12.2 Pharmacodynamics
In postmenopausal patients with advanced breast cancer, daily doses of 0.1 mg to 5 mg letrozole tablets suppress plasma concentrations of estradiol, estrone, and estrone sulfate by 75% to 95% from baseline with maximal suppression achieved within two-three days. Suppression is dose-related, with doses of 0.5 mg and higher giving many values of estrone and estrone sulfate that were below the limit of detection in the assays. Estrogen suppression was maintained throughout treatment in all patients treated at 0.5 mg or higher.
Letrozole is highly specific in inhibiting aromatase activity. There is no impairment of adrenal steroidogenesis. No clinically-relevant changes were found in the plasma concentrations of cortisol, aldosterone, 11-deoxycortisol, 17-hydroxy-progesterone, ACTH or in plasma renin activity among postmenopausal patients treated with a daily dose of letrozole tablets 0.1 mg to 5 mg. The ACTH stimulation test performed after 6 and 12 weeks of treatment with daily doses of 0.1, 0.25, 0.5, 1, 2.5, and 5 mg did not indicate any attenuation of aldosterone or cortisol production. Glucocorticoid or mineralocorticoid supplementation is, therefore, not necessary.
No changes were noted in plasma concentrations of androgens (androstenedione and testosterone) among healthy postmenopausal women after 0.1, 0.5, and 2.5 mg single doses of letrozole tablets or in plasma concentrations of androstenedione among postmenopausal patients treated with daily doses of 0.1 mg to 5 mg. This indicates that the blockade of estrogen biosynthesis does not lead to accumulation of androgenic precursors. Plasma levels of LH and FSH were not affected by letrozole in patients, nor was thyroid function as evaluated by TSH levels, T3 uptake, and T4 levels.
5.3 Hepatic Impairment
Subjects with cirrhosis and severe hepatic impairment who were dosed with 2.5 mg of letrozole tablets experienced approximately twice the exposure to letrozole tablets as healthy volunteers with normal liver function [see Clinical Pharmacology (12.3)]. Therefore, a dose reduction is recommended for this patient population. The effect of hepatic impairment on letrozole tablets exposure in cancer patients with elevated bilirubin levels has not been determined. [see Dosage and Administration (2.5)].
1 Indications and Usage
Letrozole tablets are an aromatase inhibitor indicated for:
- Adjuvant treatment of postmenopausal women with hormone receptor positive early breast cancer ( 1.1)
- Extended adjuvant treatment of postmenopausal women with early breast cancer who have received prior standard adjuvant tamoxifen therapy ( 1.2)
- First and second-line treatment of postmenopausal women with hormone receptor positive or unknown advanced breast cancer ( 1.3)
12.1 Mechanism of Action
The growth of some cancers of the breast is stimulated or maintained by estrogens. Treatment of breast cancer thought to be hormonally responsive (i.e., estrogen and/or progesterone receptor positive or receptor unknown) has included a variety of efforts to decrease estrogen levels (ovariectomy, adrenalectomy, hypophysectomy) or inhibit estrogen effects (antiestrogens and progestational agents). These interventions lead to decreased tumor mass or delayed progression of tumor growth in some women.
In postmenopausal women, estrogens are mainly derived from the action of the aromatase enzyme, which converts adrenal androgens (primarily androstenedione and testosterone) to estrone and estradiol. The suppression of estrogen biosynthesis in peripheral tissues and in the cancer tissue itself can therefore be achieved by specifically inhibiting the aromatase enzyme.
Letrozole is a nonsteroidal competitive inhibitor of the aromatase enzyme system; it inhibits the conversion of androgens to estrogens. In adult nontumor- and tumor-bearing female animals, letrozole is as effective as ovariectomy in reducing uterine weight, elevating serum LH, and causing the regression of estrogen-dependent tumors. In contrast to ovariectomy, treatment with letrozole does not lead to an increase in serum FSH. Letrozole selectively inhibits gonadal steroidogenesis but has no significant effect on adrenal mineralocorticoid or glucocorticoid synthesis.
Letrozole inhibits the aromatase enzyme by competitively binding to the heme of the cytochrome P450 subunit of the enzyme, resulting in a reduction of estrogen biosynthesis in all tissues. Treatment of women with letrozole significantly lowers serum estrone, estradiol and estrone sulfate and has not been shown to significantly affect adrenal corticosteroid synthesis, aldosterone synthesis, or synthesis of thyroid hormones.
5.4 Fatigue and Dizziness
Because fatigue, dizziness, and somnolence have been reported with the use of letrozole tablets, caution is advised when driving or using machinery until it is known how the patient reacts to letrozole tablets use.
5.6 Embryo Fetal Toxicity
Based on post-marketing reports, findings from animal studies and the mechanism of action, letrozole tablets can cause fetal harm and is contraindicated for use in pregnant women. In post-marketing reports, use of letrozole during pregnancy resulted in cases of spontaneous abortions and congenital birth defects. Letrozole caused embryo-fetal toxicities in rats and rabbits at maternal exposures that were below the maximum recommended human dose (MHRD) on a mg/m 2 basis. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during therapy with letrozole tablets and for at least 3 weeks after the last dose [see Postmarketing Experience (6.2), Use in Specific Populations (8.1, 8.3) and Clinical Pharmacology (12.1)] .
5 Warnings and Precautions
- Decreases in bone mineral density may occur. Consider bone mineral density monitoring ( 5.1)
- Increases in total cholesterol may occur. Consider cholesterol monitoring. ( 5.2)
- Fatigue, dizziness and somnolence may occur. Exercise caution when operating machinery ( 5.4)
- Embryo-Fetal toxicity: Can cause fetal harm when administered to pregnant women. Obtain a pregnancy test in females of reproductive potential. Advise females of reproductive potential to use effective contraception ( 5.6, 8.1, 8.3)
2 Dosage and Administration
2.6 Use in Renal Impairment
No dosage adjustment is required for patients with renal impairment if creatinine clearance is greater than or equal to 10 mL/min. [see Clinical Pharmacology (12.3)] .
3 Dosage Forms and Strengths
2.5 mg tablets: Yellow colored, film-coated, round shaped tablets debossed with one side 'N' and other side 'L'.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of letrozole tablets. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Eye Disorders: blurred vision
- Hepatobiliary Disorders: increased hepatic enzymes, hepatitis
- Immune System Disorders: anaphylactic reactions, hypersensitivity reactions
- Nervous System Disorders: carpal tunnel syndrome, trigger finger
- Pregnancy: spontaneous abortions, congenital birth defects
- Skin and subcutaneous disorders: angioedema, toxic epidermal necrolysis, erythema multiforme
To report SUSPECTED ADVERSE REACTIONS contact AvKARE at 1-855-361-3993; email [email protected]; or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
2.5 Use in Hepatic Impairment
No dosage adjustment is recommended for patients with mild to moderate hepatic impairment, although letrozole tablets blood concentrations were modestly increased in subjects with moderate hepatic impairment due to cirrhosis. The dose of letrozole tablets in patients with cirrhosis and severe hepatic dysfunction should be reduced by 50% [see Warnings and Precautions (5.3)]. The recommended dose of letrozole tablets for such patients is 2.5 mg administered every other day. The effect of hepatic impairment on letrozole tablets exposure in noncirrhotic cancer patients with elevated bilirubin levels has not been determined.
8 Use in Specific Populations
- Lactation: Advise not to breastfeed. ( 8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reactions rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.5 Laboratory Test Abnormalities
No dose-related effect of letrozole tablets on any hematologic or clinical chemistry parameter was evident. Moderate decreases in lymphocyte counts, of uncertain clinical significance, were observed in some patients receiving letrozole tablets 2.5 mg. This depression was transient in about half of those affected. Two patients on letrozole tablets developed thrombocytopenia; relationship to the study drug was unclear. Patient withdrawal due to laboratory abnormalities, whether related to study treatment or not was infrequent.
16 How Supplied/storage and Handling
Dispensed in Unit Dose Package. For Institutional Use Only.
| 2.5 milligram tablets | |
|
NDC 50268-476-15 (10 tablets per card, 5 cards per carton) |
|
1.1 Adjuvant Treatment of Early Breast Cancer
Letrozole tablets are indicated for the adjuvant treatment of postmenopausal women with hormone receptor positive early breast cancer.
Principal Display Panel 30 Tablet Bottle Label
14.4 First Line Treatment of Advanced Breast Cancer
A randomized, double-blind, multinational trial (P025) compared letrozole tablets 2.5 mg with tamoxifen 20 mg in 916 postmenopausal patients with locally advanced (Stage IIIB or loco-regional recurrence not amenable to treatment with surgery or radiation) or metastatic breast cancer. Time to progression (TTP) was the primary endpoint of the trial. Selected baseline characteristics for this study are shown in Table 11.
| Baseline Status | Letrozole tablets
N=458 |
Tamoxifen
N=458 |
|---|---|---|
| Stage of Disease | ||
| IIIB | 6% | 7% |
| IV | 93% | 92% |
| Receptor Status | ||
| ER and PgR Positive | 38% | 41% |
| ER or PgR Positive | 26% | 26% |
| Both Unknown | 34% | 33% |
| ER - or PgR -/Other Unknown | <1% | 0 |
| Previous Antiestrogen Therapy | ||
| Adjuvant | 19% | 18% |
| None | 81% | 82% |
| Dominant Site of Disease | ||
| Soft Tissue | 25% | 25% |
| Bone | 32% | 29% |
| Viscera | 43% | 46% |
Letrozole tablets were superior to tamoxifen in TTP and rate of objective tumor response (see Table 12).
Table 12 summarizes the results of the trial, with a total median follow-up of approximately 32 months. (All analyses are unadjusted and use 2-sided P-values.)
| Letrozole tablets
2.5 mg N=453 |
Tamoxifen
20 mg N=454 |
Hazard or Odds
Ratio (95% CI) P-Value (2-Sided) |
|
|---|---|---|---|
| Median Time to Progression | 9.4 months | 6.0 months | 0.72 (0.62, 0.83)
Hazard ratio
P<0.0001 |
| Objective Response Rate | |||
| (CR + PR) | 145 (32%) | 95 (21%) | 1.77 (1.31, 2.39)
Odds ratio
P=0.0002 |
| (CR) | 42 (9%) | 15 (3%) | 2.99 (1.63, 5.47)
P=0.0004 |
| Duration of Objective Response | |||
| Median | 18 months
(N=145) |
16 months
(N=95) |
|
| Overall Survival | 35 months
(N=458) |
32 months
(N=458) |
P=0.5136
Overall log-rank test
|
Figure 2 shows the Kaplan-Meier curves for TTP.
Table 13 shows results in the subgroup of women who had received prior antiestrogen adjuvant therapy, Table 14, results by disease site and Table 15, the results by receptor status.
| Variable | Letrozole tablets
2.5 mg N=84 |
Tamoxifen
20 mg N=83 |
|---|---|---|
| Median Time to Progression (95% CI) | 8.9 months (6.2, 12.5) | 5.9 months (3.2, 6.2) |
| Hazard Ratio for TTP (95% CI) | 0.60 (0.43, 0.84) | |
| Objective Response Rate | ||
| (CR + PR) | 22 (26%) | 7 (8%) |
| Odds Ratio for Response (95% CI) | 3.85 (1.50, 9.60) |
Hazard ratio less than 1 or odds ratio greater than 1 favors letrozole tablets; hazard ratio greater than 1 or odds ratio less than 1 favors tamoxifen.
| Letrozole tablets
2.5 mg |
Tamoxifen
20 mg |
|
|---|---|---|
| Dominant Disease Site | ||
| Soft Tissue: | N=113 | N=115 |
| Median TTP | 12.1 months | 6.4 months |
| Objective Response Rate | 50% | 34% |
| Bone: | N=145 | N=131 |
| Median TTP | 9.5 months | 6.3 months |
| Objective Response Rate | 23% | 15% |
| Viscera: | N=195 | N=208 |
| Median TTP | 8.3 months | 4.6 months |
| Objective Response Rate | 28% | 17% |
| Variable | Letrozole tablets
2.5 mg |
Tamoxifen
20 mg |
|---|---|---|
| Receptor Positive | N=294 | N=305 |
| Median Time to Progression (95% CI) | 9.4 months (8.9, 11.8) | 6.0 months (5.1, 8.5) |
| Hazard Ratio for TTP (95% CI) | 0.69 (0.58, 0.83) | |
| Objective Response Rate (CR+PR) | 97 (33%) | 66 (22%) |
| Odds Ratio for Response (95% CI) | 1.78 (1.20, 2.60) | |
| Receptor Unknown | N=159 | N=149 |
| Median Time to Progression (95% CI) | 9.2 months (6.1, 12.3) | 6.0 months (4.1, 6.4) |
| Hazard Ratio for TTP (95% CI) | 0.77 (0.60, 0.99) | |
| Objective Response Rate (CR+PR) | 48 (30%) | 29 (20%) |
| Odds Ratio for Response (95% CI) | 1.79 (1.10, 3.00) |
Hazard ratio less than 1 or odds ratio greater than 1 favors letrozole tablets; hazard ratio greater than 1 or odds ratio less than 1 favors tamoxifen.
Figure 3 shows the Kaplan-Meier curves for survival.
Legend: Randomized letrozole tablets: n=458, events 57%, median overall survival 35 months (95% CI 32 to 38 months)
Randomized tamoxifen: n=458, events 57%, median overall survival 32 months (95% CI 28 to 37 months)
Overall log-rank P=0.5136 (i.e., there was no significant difference between treatment arms in overall survival).
The median overall survival was 35 months for the letrozole tablets group and 32 months for the tamoxifen group, with a P-value 0.5136. Study design allowed patients to cross over upon progression to the other therapy. Approximately 50% of patients crossed over to the opposite treatment arm and almost all patients who crossed over had done so by 36 months. The median time to crossover was 17 months (letrozole tablets to tamoxifen) and 13 months (tamoxifen to letrozole tablets). In patients who did not cross over to the opposite treatment arm, median survival was 35 months with letrozole tablets (n=219, 95% CI 29 to 43 months) vs 20 months with tamoxifen (n=229, 95% CI 16 to 26 months).
14.5 Second Line Treatment of Advanced Breast Cancer
Letrozole tablets were initially studied at doses of 0.1 mg to 5.0 mg daily in six non-comparative trials (AR/BC1, P01, AR/ST1, AR/PS1, AR/ES1 and NJO-03) in 181 postmenopausal estrogen/progesterone receptor positive or unknown advanced breast cancer patients previously treated with at least antiestrogen therapy. Patients had received other hormonal therapies and also may have received cytotoxic therapy. Eight (20%) of forty patients treated with letrozole tablets 2.5 mg daily in trials achieved an objective tumor response (complete or partial response).
Two large randomized, controlled, multinational (predominantly European) trials (AR/BC2, AR/BC3) were conducted in patients with advanced breast cancer who had progressed despite antiestrogen therapy. Patients were randomized to letrozole tablets 0.5 mg daily, letrozole tablets 2.5 mg daily, or a comparator [megestrol acetate 160 mg daily in one study (AR/BC2); and aminoglutethimide 250 mg twice a day with corticosteroid supplementation in the other study (AR/BC3)]. In each study over 60% of the patients had received therapeutic antiestrogens, and about one-fifth of these patients had had an objective response. The megestrol acetate controlled study was double-blind; the other study was open label. Selected baseline characteristics for each study are shown in Table 16.
| Parameter | megestrol acetate study | aminoglutethimide study |
|---|---|---|
| No. of Participants | 552 | 557 |
| Receptor Status | ||
| ER/PR Positive | 57% | 56% |
| ER/PR Unknown | 43% | 44% |
| Previous Therapy | ||
| Adjuvant Only | 33% | 38% |
| Therapeutic +/- Adj. | 66% | 62% |
| Sites of Disease | ||
| Soft Tissue | 56% | 50% |
| Bone | 50% | 55% |
| Viscera | 40% | 44% |
Confirmed objective tumor response (complete response plus partial response) was the primary endpoint of the trials. Responses were measured according to the Union Internationale Contre le Cancer (UICC) criteria and verified by independent, blinded review. All responses were confirmed by a second evaluation 4 to 12 weeks after the documentation of the initial response.
Table 17 shows the results for the first trial (AR/BC2), with a minimum follow-up of 15 months, that compared letrozole tablets 0.5 mg, letrozole tablets 2.5 mg, and megestrol acetate 160 mg daily. (All analyses are unadjusted.)
| Letrozole tablets
0.5 mg N=188 |
Letrozole tablets
2.5 mg N=174 |
megestrol acetate
N=190 |
|
|---|---|---|---|
| Objective Response (CR + PR) | 22 (11.7%) | 41 (23.6%) | 31 (16.3%) |
| Median Duration of Response | 552 days | (Not reached) | 561 days |
| Median Time to Progression | 154 days | 170 days | 168 days |
| Median Survival | 633 days | 730 days | 659 days |
| Odds Ratio for Response | Letrozole tablets 2.5: Letrozole tablets 0.5=2.33
(95% CI: 1.32, 4.17); P=0.004 Two-sided
P-value
|
Letrozole tablets 2.5: megestrol=1.58
(95% CI: 0.94, 2.66); P=0.08 |
|
| Relative Risk of Progression | Letrozole tablets 2.5: Letrozole tablets 0.5=0.81
(95% CI: 0.63, 1.03); P=0.09 |
Letrozole tablets 2.5: megestrol=0.77
(95% CI: 0.60, 0.98); P=0.03 |
The Kaplan-Meier curves for progression for the megestrol acetate study are shown in Figure 4.
The results for the study comparing letrozole tablets to aminoglutethimide (AR/BC3), with a minimum follow-up of 9 months, are shown in Table 18. (Unadjusted analyses are used.)
| Letrozole tablets
0.5 mg N=193 |
Letrozole tablets
2.5 mg N=185 |
aminoglutethimide
N=179 |
|
|---|---|---|---|
| Objective Response (CR + PR) | 34 (17.6%) | 34 (18.4%) | 22 (12.3%) |
| Median Duration of Response | 619 days | 706 days | 450 days |
| Median Time to Progression | 103 days | 123 days | 112 days |
| Median Survival | 636 days | 792 days | 592 days |
| Odds Ratio for Response | Letrozole tablets 2.5: Letrozole tablets 0.5=1.05
(95% CI: 0.62, 1.79); P=0.85 Two-sided
P-value
|
Letrozole tablets 2.5: aminoglutethimide=1.61
(95% CI: 0.90, 2.87); P=0.11 |
|
| Relative Risk of Progression | Letrozole tablets 2.5: Letrozole tablets 0.5=0.86
(95% CI: 0.68, 1.11); P=0.25 |
Letrozole tablets 2.5: aminoglutethimide=0.74
(95% CI: 0.57, 0.94); P=0.02 |
The Kaplan-Meier curves for progression for the aminoglutethimide study is shown in Figure 5
Figure 5 Kaplan-Meier Estimates of Time to Progression (Aminoglutethimide Study)
2.2 Use in Adjuvant Treatment of Early Breast Cancer
In the adjuvant setting, the optimal duration of treatment with letrozole is unknown. In both the adjuvant study and the postapproval adjuvant study, median treatment duration was 5 years. Treatment should be discontinued at relapse [see Clinical Studies (14.1)].
1.2 Extended Adjuvant Treatment of Early Breast Cancer
Letrozole tablets are indicated for the extended adjuvant treatment of early breast cancer in postmenopausal women, who have received 5 years of adjuvant tamoxifen therapy. The effectiveness of letrozole tablets in extended adjuvant treatment of early breast cancer is based on an analysis of disease-free survival in patients treated with letrozole tablets for a median of 60 months [see Clinical Studies (14.2, 14.3)] .
14.1 Updated Adjuvant Treatment of Early Breast Cancer
In a multicenter study (BIG 1-98, NCT00004205) enrolling over 8,000 postmenopausal women with resected, receptor-positive early breast cancer, one of the following treatments was randomized in a double-blind manner:
Option 1:
- tamoxifen for 5 years
- Letrozole tablets for 5 years
- tamoxifen for 2 years followed by letrozole tablets for 3 years
- Letrozole tablets for 2 years followed by tamoxifen for 3 years
Option 2:
- tamoxifen for 5 years
- Letrozole tablets for 5 years
The study in the adjuvant setting, BIG 1-98 was designed to answer two primary questions: whether letrozole tablets for 5 years was superior to Tamoxifen for 5 years (Primary Core Analysis) and whether switching endocrine treatments at 2 years was superior to continuing the same agent for a total of 5 years (Sequential Treatments Analysis). Selected baseline characteristics for the study population are shown in Table 6.
The primary endpoint of this trial was disease-free survival (DFS) (i.e., interval between randomization and earliest occurrence of a local, regional, or distant recurrence, or invasive contralateral breast cancer, or death from any cause). The secondary endpoints were overall survival (OS), systemic disease-free survival (SDFS), invasive contralateral breast cancer, time to breast cancer recurrence (TBR) and time to distant metastasis (TDM).
The Primary Core Analysis (PCA) included all patients and all follow-up in the monotherapy arms in both randomization options, but follow-up in the two sequential treatments arms was truncated 30 days after switching treatments. The PCA was conducted at a median treatment duration of 24 months and a median follow-up of 26 months. Letrozole tablets were superior to tamoxifen in all endpoints except overall survival and contralateral breast cancer [e.g., DFS: hazard ratio, HR 0.79; 95% CI (0.68, 0.92); P=0.002; SDFS: HR 0.83; 95% CI (0.70, 0.97); TDM: HR 0.73; 95% CI (0.60, 0.88); OS: HR 0.86; 95% CI (0.70, 1.06)].
In 2005, based on recommendations by the independent Data Monitoring Committee, the tamoxifen arms were unblinded and patients were allowed to complete initial adjuvant therapy with letrozole tablets (if they had received tamoxifen for at least 2 years) or to start extended adjuvant treatment with letrozole tablets (if they had received tamoxifen for at least 4.5 years) if they remained alive and disease-free. In total, 632 patients crossed to letrozole tablets or another aromatase inhibitor. Approximately 70% (448) of these 632 patients crossed to letrozole tablets to complete initial adjuvant therapy and most of these crossed in years 3 to 4. All of these patients were in Option 1. A total of 184 patients started extended adjuvant therapy with letrozole tablets (172 patients) or with another aromatase inhibitor (12 patients). To explore the impact of this selective crossover, results from analyses censoring follow-up at the date of the selective crossover (in the tamoxifen arm) are presented for the MAA.
The PCA allowed the results of letrozole tablets for 5 years compared with tamoxifen for 5 years to be reported in 2005 after a median follow-up of only 26 months. The design of the PCA is not optimal to evaluate the effect of letrozole tablets after a longer time (because follow-up was truncated in two arms at around 25 months). The MAA (ignoring the two sequential treatment arms) provided follow-up equally as long in each treatment and did not over-emphasize early recurrences as the PCA did. The MAA thus provides the clinically appropriate updated efficacy results in answer to the first primary question, despite the confounding of the tamoxifen reference arm by the selective crossover to letrozole tablets. The updated results for the MAA are summarized in Table 7. Median follow-up for this analysis is 73 months.
The Sequential Treatments Analysis (STA) addresses the second primary question of the study. The primary analysis for the STA was from switch (or equivalent time-point in monotherapy arms) + 30 days (STA-S) with a two-sided test applied to each pair-wise comparison at the 2.5% level. Additional analyses were conducted from randomization (STA-R) but these comparisons (added in light of changing medical practice) were under-powered for efficacy.
| Primary Core Analysis
(PCA) |
Monotherapy Arms Analysis
(MAA) |
|||
|---|---|---|---|---|
| Characteristic | Letrozole tablets
N=4003 n (%) |
Tamoxifen
N=4007 n (%) |
Letrozole tablets
N=2463 n (%) |
Tamoxifen
N=2459 n (%) |
| Age (median, years) | 61 | 61 | 61 | 61 |
| Age range (years) | 38-89 | 39-90 | 38-88 | 39-90 |
| Hormone receptor status (%) | ||||
| ER+ and/or PgR+ | 99.7 | 99.7 | 99.7 | 99.7 |
| Both unknown | 0.3 | 0.3 | 0.3 | 0.3 |
| Nodal status (%) | ||||
| Node negative | 52 | 52 | 50 | 52 |
| Node positive | 41 | 41 | 43 | 41 |
| Nodal status unknown | 7 | 7 | 7 | 7 |
| Prior adjuvant chemotherapy (%) | 24 | 24 | 24 | 24 |
| Letrozole tablets
N=2463 |
Tamoxifen
N=2459 |
Hazard ratio | |||||
|---|---|---|---|---|---|---|---|
| Events
(%) |
5-year rate | Events
(%) |
5-year rate | (95% CI) | P | ||
| ITT analysis ignores selective crossover in tamoxifen arms. | |||||||
| Censored analysis censors follow-up at the date of selective crossover in 632 patients who crossed to letrozole tablets or another aromatase inhibitor after the tamoxifen arms were unblinded in 2005. | |||||||
| Definition of: | |||||||
| Disease-free survival
Disease-free survival: Interval from randomization to earliest event of invasive loco-regional recurrence, distant metastasis, invasive contralateral breast cancer, or death without a prior event.
|
ITT | 445 (18.1) | 87.4 | 500 (20.3) | 84.7 | 0.87 (0.76, 0.99) | 0.03 |
| Censor | 445 | 87.4 | 483 | 84.2 | 0.84 (0.73, 0.95) | ||
| 0 positive nodes | ITT | 165 | 92.2 | 189 | 90.3 | 0.88 (0.72, 1.09) | |
| 1-3 positive nodes | ITT | 151 | 85.6 | 163 | 83.0 | 0.85 (0.68, 1.06) | |
| >=4 positive nodes | ITT | 123 | 71.2 | 142 | 62.6 | 0.81 (0.64, 1.03) | |
| Adjuvant chemotherapy | ITT | 119 | 86.4 | 150 | 80.6 | 0.77 (0.60, 0.98) | |
| No chemotherapy | ITT | 326 | 87.8 | 350 | 86.1 | 0.91 (0.78, 1.06) | |
| Systemic DFS
Systemic disease-free survival: Interval from randomization to invasive regional recurrence, distant metastasis, or death without a prior cancer event.
|
ITT | 401 | 88.5 | 446 | 86.6 | 0.88 (0.77, 1.01) | |
| Time to distant metastasis
Time to distant metastasis: Interval from randomization to distant metastasis.
|
ITT | 257 | 92.4 | 298 | 90.1 | 0.85 (0.72, 1.00) | |
| Adjuvant chemotherapy | ITT | 84 | - | 109 | - | 0.75 (0.56, 1.00) | |
| No chemotherapy | ITT | 173 | - | 189 | - | 0.90 (0.73, 1.11) | |
| Distant DFS
Distant disease-free survival: Interval from randomization to earlier event of relapse in a distant site or death from any cause
|
ITT | 385 | 89.0 | 432 | 87.1 | 0.87 (0.76, 1.00) | |
| Contralateral breast cancer | ITT | 34 | 99.2 | 44 | 98.6 | 0.76 (0.49, 1.19) | |
| Overall survival | ITT | 303 | 91.8 | 343 | 90.9 | 0.87 (0.75, 1.02) | |
| Censor | 303 | 91.8 | 338 | 90.1 | 0.82 (0.70, 0.96) | ||
| 0 positive nodes | ITT | 107 | 95.2 | 121 | 94.8 | 0.90 (0.69, 1.16) | |
| 1-3 positive nodes | ITT | 99 | 90.8 | 114 | 90.6 | 0.81 (0.62, 1.06) | |
| >=4 positive nodes | ITT | 92 | 80.2 | 104 | 73.6 | 0.86 (0.65, 1.14) | |
| Adjuvant chemotherapy | ITT | 76 | 91.5 | 96 | 88.4 | 0.79 (0.58, 1.06) | |
| No chemotherapy | ITT | 227 | 91.9 | 247 | 91.8 | 0.91 (0.76, 1.08) |
Figure 1 shows the Kaplan-Meier curves for Disease-Free Survival Monotherapy Analysis
DFS events defined as loco-regional recurrence, distant metastasis, invasive contralateral breast cancer, or death from any cause (i.e., definition excludes second non-breast primary cancers).
The medians of overall survival for both arms were not reached for the MAA. There was no statistically significant difference in overall survival. The hazard ratio for survival in the letrozole tablets arm compared to the tamoxifen arm was 0.87, with 95% CI (0.75, 1.02) (see Table 7).
There were no significant differences in DFS, OS, SDFS, and Distant DFS from switch in the Sequential Treatments Analysis with respect to either monotherapy (e.g., [Tamoxifen 2 years followed by] letrozole tablets 3 years versus tamoxifen beyond 2 years, DFS HR 0.89; 97.5% CI 0.68, 1.15 and [letrozole tablets 2 years followed by] tamoxifen 3 years versus letrozole tablets beyond 2 years, DFS HR 0.93; 97.5% CI 0.71, 1.22).
There were no significant differences in DFS, OS, SDFS, and Distant DFS from randomization in the Sequential Treatments Analyses.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
A conventional carcinogenesis study in mice at doses of 0.6 to 60 mg/kg/day (about 1 to 100 times the daily maximum recommended human dose on a mg/m 2 basis) administered by oral gavage for up to 2 years revealed a dose-related increase in the incidence of benign ovarian stromal tumors. The incidence of combined hepatocellular adenoma and carcinoma showed a significant trend in females when the high dose group was excluded due to low survival. In a separate study, plasma AUC 0-12hr levels in mice at 60 mg/kg/day were 55 times higher than the AUC 0-24hr level in breast cancer patients at the recommended dose. The carcinogenicity study in rats at oral doses of 0.1 to 10 mg/kg/day (about 0.4 to 40 times the daily maximum recommended human dose on a mg/m 2 basis) for up to 2 years also produced an increase in the incidence of benign ovarian stromal tumors at 10 mg/kg/day. Ovarian hyperplasia was observed in females at doses equal to or greater than 0.1 mg/kg/day. At 10 mg/kg/day, plasma AUC 0-24hr levels in rats were 80 times higher than the level in breast cancer patients at the recommended dose. The benign ovarian stromal tumors observed in mice and rats were considered to be related to the pharmacological inhibition of estrogen synthesis and may be due to increased luteinizing hormone resulting from the decrease in circulating estrogen.
Letrozole tablets were not mutagenic in in vitro tests (Ames and E.coli bacterial tests) but were observed to be a potential clastogen in in vitro assays (CHO K1 and CCL 61 Chinese hamster ovary cells). Letrozole was not clastogenic in vivo (micronucleus test in rats).
In a fertility and early embryonic development toxicity study in female rats, oral administration of letrozole starting 2 weeks before mating until pregnancy day 6 resulted in an increase in pre-implantation loss at doses ≥ 0.03 mg/kg/day (approximately 0.1 times the maximum recommended human dose on a mg/m 2 basis). In repeat-dose toxicity studies, administration of letrozole caused sexual inactivity in females and atrophy of the reproductive tract in males and females at doses of 0.6, 0.1 and 0.03 mg/kg in mice, rats and dogs, respectively (approximately 1, 0.4 and 0.4 times the daily maximum recommended human dose on a mg/m 2 basis, respectively).
1.3 First and Second Line Treatment of Advanced Breast Cancer
Letrozole tablets are indicated for first-line treatment of postmenopausal women with hormone receptor positive or unknown, locally advanced or metastatic breast cancer. Letrozole tablets are also indicated for the treatment of advanced breast cancer in postmenopausal women with disease progression following antiestrogen therapy [see Clinical Studies (14.4, 14.5)].
2.3 Use in Extended Adjuvant Treatment of Early Breast Cancer
In the extended adjuvant setting, the optimal treatment duration with letrozole tablets is not known. The planned duration of treatment in the study was 5 years. In the final updated analysis, conducted at a median follow-up of 62 months, the median treatment duration for letrozole tablets was 60 months. Seventy-one (71%) percent of patients were treated for at least 3 years and 58% of patients completed at least 4.5 years of extended adjuvant treatment. The treatment should be discontinued at tumor relapse [see Clinical Studies (14.2)].
2.4 Use in First and Second Line Treatment of Advanced Breast Cancer
In patients with advanced disease, treatment with letrozole tablets should continue until tumor progression is evident. [see Clinical Studies (14.4, 14.5)]
14.2 Extended Adjuvant Treatment of Early Breast Cancer, Median Treatment Duration of 24 Months
A double-blind, randomized, placebo-controlled trial (MA-17, NCT00003140) of letrozole tablets was performed in over 5,100 postmenopausal women with receptor-positive or unknown primary breast cancer who were disease free after 5 years of adjuvant treatment with tamoxifen.
The planned duration of treatment for patients in the study was 5 years, but the trial was terminated early because of an interim analysis showing a favorable letrozole tablets effect on time without recurrence or contralateral breast cancer. At the time of unblinding, women had been followed for a median of 28 months, 30% of patients had completed 3 or more years of follow-up and less than 1% of patients had completed 5 years of follow-up.
Selected baseline characteristics for the study population are shown in Table 8.
| Baseline Status | Letrozole tablets
N=2582 |
Placebo
N=2586 |
|---|---|---|
| Hormone Receptor Status (%) | ||
| ER+ and/or PgR+ | 98 | 98 |
| Both Unknown | 2 | 2 |
| Nodal Status (%) | ||
| Node Negative | 50 | 50 |
| Node Positive | 46 | 46 |
| Nodal Status Unknown | 4 | 4 |
| Chemotherapy | 46 | 46 |
Table 9 shows the study results. Disease-free survival was measured as the time from randomization to the earliest event of loco-regional or distant recurrence of the primary disease or development of contralateral breast cancer or death. DFS by hormone receptor status, nodal status and adjuvant chemotherapy were similar to the overall results. Data were premature for an analysis of survival.
| Letrozole tablets
N = 2582 |
Placebo
N = 2586 |
Hazard Ratio
(95% CI) |
P-Value | |
|---|---|---|---|---|
| CI = confidence interval for hazard ratio. Hazard ratio of less than 1.0 indicates difference in favor of letrozole tablets (lesser risk of recurrence); hazard ratio greater than 1.0 indicates difference in favor of placebo (higher risk of recurrence with letrozole tablets). | ||||
| P-value based on stratified log-rank test. | ||||
|
Disease Free Survival (DFS)
First event of loco-regional recurrence, distant relapse, contralateral breast cancer or death from any cause. Events
|
122 (4.7%) | 193 (7.5%) | 0.62 (0.49, 0.78)
Analysis stratified by receptor status, nodal status and prior adjuvant chemotherapy (stratification factors as at randomization).
|
0.00003 |
| Local Breast Recurrence | 9 | 22 | ||
| Local Chest Wall Recurrence | 2 | 8 | ||
| Regional Recurrence | 7 | 4 | ||
| Distant Recurrence | 55 | 92 | 0.61 (0.44 - 0.84) | 0.003 |
| Contralateral Breast Cancer | 19 | 29 | ||
| Deaths Without Recurrence or Contralateral Breast Cancer | 30 | 38 |
14.3 Updated Analyses of Extended Adjuvant Treatment of Early Breast Cancer, Median Treatment Duration of 60 Months
| Letrozole tablets
N = 2582 (%) |
Placebo
N = 2586 (%) |
Hazard Ratio
Adjusted by receptor status, nodal status and prior chemotherapy
(95% CI) |
P-Value
Stratified log-rank test, stratified by receptor status, nodal status and prior chemotherapy
|
|
|---|---|---|---|---|
|
Disease Free Survival (DFS) events
DFS events defined as earliest of loco-regional recurrence, distant metastasis, contralateral breast cancer or death from any cause, and ignoring switches to letrozole tablets in 60% of the placebo arm.
|
344 (13.3) | 402 (15.5) | 0.89 (0.77, 1.03) | 0.12 |
|
Breast cancer recurrence
(Protocol definition of DFS events Protocol definition does not include deaths from any cause )
|
209 | 286 | 0.75 (0.63, 0.89) | 0.001 |
| Local Breast Recurrence | 15 | 44 | ||
| Local Chest Wall Recurrence | 6 | 14 | ||
| Regional Recurrence | 10 | 8 | ||
| Distant Recurrence | 140 | 167 | ||
| Distant Recurrence
(first or subsequent events) |
142 | 169 | 0.88 (0.70, 1.10) | 0.246 |
| Contralateral Breast Cancer | 37 | 53 | ||
| Deaths Without Recurrence or Contralateral Breast Cancer | 135 | 116 |
Updated analyses were conducted at a median follow-up of 62 months. In the letrozole tablets arm, 71% of the patients were treated for at least 3 years and 58% of patients completed at least 4.5 years of extended adjuvant treatment. After the unblinding of the study at a median follow-up of 28 months, approximately 60% of the selected patients in the placebo arm opted to switch to letrozole tablets.
In this updated analysis shown in Table 10, letrozole tablets significantly reduced the risk of breast cancer recurrence or contralateral breast cancer compared with placebo (HR 0.75; 95% CI 0.63, 0.89; P=0.001). However, in the updated DFS analysis (interval between randomization and earliest event of loco-regional recurrence, distant metastasis, contralateral breast cancer, or death from any cause) the treatment difference was heavily diluted by 60% of the patients in the placebo arm switching to letrozole tablets and accounting for 64% of the total placebo patient-years of follow-up. Ignoring these switches, the risk of DFS event was reduced by a non-significant 11% (HR 0.89; 95% CI 0.77, 1.03). There was no significant difference in distant disease-free survival or overall survival.
Structured Label Content
Section 42229-5 (42229-5)
Adjuvant Treatment of Early Breast Cancer
In study, BIG 1-98, the median treatment duration of adjuvant treatment was 60 months and the median duration of follow-up for safety was 96 months for patients receiving letrozole tablets and tamoxifen.
Certain adverse reactions were prospectively specified for analysis (see Table 1), based on the known pharmacologic properties and side effect profiles of the two drugs.
Adverse reactions were analyzed irrespective of whether a symptom was present or absent at baseline. Most adverse reactions reported (approximately 75% of patients who reported AEs) were Grade 1 or Grade 2 applying the Common Toxicity Criteria (CTC)Version 2.0/Common Terminology Criteria for Adverse Events (CTCAE), version 3.0. Table 1 describes adverse reactions (Grades 1-4 and Grades 3-4) irrespective of relationship to study treatment in the adjuvant trial for the monotherapy arms analysis (safety population).
| Adverse Reactions | Grades 1-4 | Grades 3-4 | ||||||
|---|---|---|---|---|---|---|---|---|
| Letrozole tablets
N=2448 n (%) |
Tamoxifen
N=2447 n (%) |
Letrozole tablets
N=2448 n (%) |
Tamoxifen
N=2447 n (%) |
|||||
| TIA = Transient ischemic attack | ||||||||
| Note: Cardiovascular events (including cerebrovascular and thromboembolic events), skeletal and urogenital/endometrial events and second primary malignancies were collected life -long. All of these events were assumed to be of CTC Grade 3 to 5 and were not individually graded | ||||||||
| Patients with any adverse reaction | 2309 | (94.3) | 2212 | (90.4) | 636 | (26.0) | 606 | (24.8) |
| Hypercholesterolemia
Target events pre-specified for analysis
|
1280 | (52.3) | 700 | (28.6) | 11 | (0.4) | 6 | (0.2) |
| Hot flashes | 819 | (33.5) | 929 | (38.0) | - | - | - | - |
| Arthralgia/arthritis | 621 | (25.4) | 504 | (20.6) | 84 | (3.4) | 50 | (2.0) |
| Bone fractures
At median follow-up of 96 months (i.e. any time after randomization) for letrozole tablets (range up to 144 months) and 95 months for tamoxifen (range up to 143 months )
|
361 | (14.7) | 280 | (11.4) | - | - | - | - |
| Night sweats | 356 | (14.5) | 426 | (17.4) | - | - | - | - |
| Weight increase | 317 | (12.9) | 378 | (15.4) | 27 | (1.1) | 39 | (1.6) |
| Nausea | 284 | (11.6) | 277 | (11.3) | 6 | (0.2) | 9 | (0.4) |
| Bone fractures
Events pre-printed on CRF
At median treatment duration of 60 months (i.e. during treatment + 30 days after discontinuation of treatment) for letrozole tablets and tamoxifen (range up to 68 months)
|
249 | (10.2) | 175 | (7.2) | - | - | - | - |
| Fatigue (lethargy, malaise, asthenia) | 235 | (9.6) | 250 | (10.2) | 6 | (0.2) | 7 | (0.3) |
| Myalgia | 221 | (9.0) | 212 | (8.7) | 18 | (0.7) | 14 | (0.6) |
| Vaginal bleeding | 129 | (5.3) | 320 | (13.1) | 1 | (<0.1) | 8 | (0.3) |
| Edema | 164 | (6.7) | 160 | (6.5) | 3 | (0.1) | 1 | (<0.1) |
| Weight decrease | 140 | (5.7) | 129 | (5.3) | 8 | (0.3) | 5 | (0.2) |
| Osteoporosis | 126 | (5.1) | 67 | (2.7) | 10 | (0.4) | 5 | (0.2) |
| Back pain | 125 | (5.1) | 136 | (5.6) | 7 | (0.3) | 11 | (0.4) |
| Bone pain | 123 | (5.0) | 109 | (4.5) | 6 | (0.2) | 4 | (0.2) |
| Depression | 119 | (4.9) | 114 | (4.7) | 16 | (0.7) | 14 | (0.6) |
| Vaginal irritation | 112 | (4.6) | 77 | (3.1) | 2 | (<0.1) | 2 | (<0.1) |
| Headache | 105 | (4.3) | 94 | (3.8) | 8 | (0.3) | 4 | (0.2) |
| Pain in extremity | 103 | (4.2) | 79 | (3.2) | 6 | (0.2) | 4 | (0.2) |
| Osteopenia | 87 | (3.6) | 76 | (3.1) | 0 | - | 3 | (0.1) |
| Dizziness/light-headedness | 84 | (3.4) | 80 | (3.3) | 1 | (<0.1) | 6 | (0.2) |
| Alopecia | 83 | (3.4) | 84 | (3.4) | - | - | - | - |
| Vomiting | 80 | (3.3) | 80 | (3.3) | 3 | (0.1) | 5 | (0.2) |
| Cataract | 49 | (2.0) | 54 | (2.2) | 16 | (0.7) | 17 | (0.7) |
| Constipation | 49 | (2.0) | 71 | (2.9) | 3 | (0.1) | 1 | (<0.1) |
| Myocardial infarction | 42 | (1.7) | 28 | (1.1) | - | - | - | - |
| Breast pain | 37 | (1.5) | 43 | (1.8) | 1 | (<0.1) | - | - |
| Anorexia | 20 | (0.8) | 20 | (0.8) | 1 | (<0.1) | 1 | (<0.1) |
| Endometrial proliferation disorders | 14 | (0.6) | 86 | (3.5) | 0 | - | 14 | (0.6) |
| Ovarian cyst | 11 | (0.4) | 18 | (0.7) | 4 | (0.2) | 4 | (0.2) |
| Endometrial hyperplasia/cancer | 11 | (0.4) | 72 | (2.9) | - | - | - | - |
| Endometrial hyperplasia/cancer
,
Excluding women who had undergone hysterectomy before study entry
|
6/1909 | (0.3) | 57/1943 | (2.9) | - | - | - | - |
| Other endometrial disorders | 2 | (<0.1) | 3 | (0.1) | 0 | - | 0 | - |
| Myocardial infarction | 24 | (1.0) | 12 | (0.5) | - | - | - | - |
| Myocardial ischemia | 6 | (0.2) | 9 | (0.4) | - | - | - | - |
| Cerebrovascular accident/TIA | 74 | (3.0) | 68 | (2.8) | - | - | - | - |
| Cerebrovascular accident/TIA | 51 | (2.1) | 47 | (1.9) | - | - | - | - |
| Angina requiring surgery | 35 | (1.4) | 33 | (1.3) | - | - | - | - |
| Angina requiring surgery | 25 | (1.0) | 25 | (1.0) | - | - | - | - |
| Thromboembolic event | 79 | (3.2) | 113 | (4.6) | - | - | - | - |
| Thromboembolic event | 51 | (2.1) | 89 | (3.6) | - | - | - | - |
| Cardiac failure | 39 | (1.6) | 34 | (1.4) | - | - | - | - |
| Cardiac failure | 27 | (1.1) | 15 | (0.6) | - | - | - | - |
| Hypertension | 160 | (6.5) | 175 | (7.2) | - | - | - | - |
| Hypertension | 138 | (5.6) | 139 | (5.7) | - | - | - | - |
| Other cardiovascular | 172 | (7.0) | 174 | (7.1) | - | - | - | - |
| Other cardiovascular | 120 | (4.9) | 119 | (4.9) | - | - | - | - |
| Second primary malignancy | 129 | (5.3) | 150 | (6.1) | - | - | - | - |
| Second primary malignancy | 54 | (2.2) | 79 | (3.2) | - | - | - | - |
When considering all grades during study treatment, a higher incidence of events was seen for letrozole tablets regarding fractures (10.1% vs 7.1%), myocardial infarctions (1.0% vs 0.5%), and arthralgia (25.2% vs 20.4%) (letrozole tablets vs tamoxifen respectively). A higher incidence was seen for tamoxifen regarding thromboembolic events (2.1% vs 3.6%), endometrial hyperplasia/cancer (0.3% vs 2.9%), and endometrial proliferation disorders (0.3% vs 1.8%) (letrozole tablets vs tamoxifen respectively).
At a median follow-up of 96 months, a higher incidence of events was seen for letrozole tablets (14.7%) than for tamoxifen (11.4%) regarding fractures. A higher incidence was seen for tamoxifen compared to letrozole tablets regarding thromboembolic events (4.6% vs 3.2%), and endometrial hyperplasia or cancer (2.9% vs 0.4%) (tamoxifen vs letrozole tablets, respectively).
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Store at 25°C (77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
10 Overdosage (10 OVERDOSAGE)
Isolated cases of letrozole tablets overdose have been reported. In these instances, the highest single dose ingested was 62.5 mg or 25 tablets. While no serious adverse reactions were reported in these cases, because of the limited data available, no firm recommendations for treatment can be made. However, emesis could be induced if the patient is alert. In general, supportive care and frequent monitoring of vital signs are also appropriate. In single-dose studies, the highest dose used was 30 mg, which was well tolerated; in multiple-dose trials, the largest dose of 10 mg was well tolerated.
Lethality was observed in mice and rats following single oral doses that were equal to or greater than 2,000 mg/kg (about 4,000 to 8,000 times the daily maximum recommended human dose on a mg/m 2 basis); death was associated with reduced motor activity, ataxia and dyspnea. Lethality was observed in cats following single IV doses that were equal to or greater than 10 mg/kg (about 50 times the daily maximum recommended human dose on a mg/m 2 basis); death was preceded by depressed blood pressure and arrhythmias.
11 Description (11 DESCRIPTION)
Letrozole tablets for oral administration contain 2.5 mg of letrozole, a nonsteroidal aromatase inhibitor (inhibitor of estrogen synthesis). It is chemically described as 4,4'-(1H-1,2,4-Triazol-1-ylmethylene)dibenzonitrile, and its structural formula is
Letrozole is a white to yellowish crystalline powder, practically odorless, freely soluble in dichloromethane, slightly soluble in ethanol, and practically insoluble in water. It has a molecular weight of 285.31, empirical formula C 17H 11N 5, and a melting range of 184°C to 185°C.
Letrozole tablets are available as 2.5 mg tablets for oral administration.
Inactive Ingredients: Colloidal silicon dioxide, ferric oxide (yellow), hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, pregelatinized starch, polyethylene glycol, sodium starch glycolate, talc and titanium dioxide.
5.2 Cholesterol
Consideration should be given to monitoring serum cholesterol. In the adjuvant trial (BIG 1-98), hypercholesterolemia was reported in 52.3% of letrozole patients and 28.6% of tamoxifen patients. Grade 3-4 hypercholesterolemia was reported in 0.4% of letrozole patients and 0.1% of tamoxifen patients. Also in the adjuvant setting, an increase of greater than or equal to 1.5 × upper limit of normal (ULN) in total cholesterol (generally nonfasting) was observed in patients on monotherapy who had baseline total serum cholesterol within the normal range (i.e., less than =1.5 × ULN) in 155/1843 (8.4%) patients on letrozole vs 71/1840 (3.9%) patients on tamoxifen. Lipid lowering medications were required for 29% of patients on letrozole and 20% on tamoxifen [see Adverse Reactions (6)] .
5.1 Bone Effects
Use of letrozole tablets may cause decreases in bone mineral density (BMD). Consideration should be given to monitoring BMD. Results of a safety study to evaluate safety in the adjuvant setting comparing the effect on lumbar spine (L2-L4) BMD of adjuvant treatment with letrozole to that with tamoxifen showed at 24 months a median decrease in lumbar spine BMD of 4.1% in the letrozole arm compared to a median increase of 0.3% in the tamoxifen arm (difference = 4.4%) ( P<0.0001) [see Adverse Reactions (6)] . Updated results from the BMD substudy (MA-17B) in the extended adjuvant setting demonstrated that at 2 years patients receiving letrozole had a median decrease from baseline of 3.8% in hip BMD compared to a median decrease of 2.0% in the placebo group. The changes from baseline in lumbar spine BMD in letrozole and placebo treated groups were not significantly different [see Adverse Reactions (6)].
In the adjuvant trial (BIG 1-98) the incidence of bone fractures at any time after randomization was 14.7% for letrozole and 11.4% for tamoxifen at a median follow-up of 96 months. The incidence of osteoporosis was 5.1% for letrozole and 2.7% for tamoxifen [see Adverse Reactions (6)] . In the extended adjuvant trial (MA-17), the incidence of bone fractures at any time after randomization was 13.3% for letrozole and 7.8% for placebo at a median follow-up of 62 months. The incidence of new osteoporosis was 14.5% for letrozole and 7.8% for placebo [see Adverse Reactions (6)] .
8.4 Pediatric Use
The safety and effectiveness in pediatric patients have not been established.
Letrozole administration to young (postnatal day 7) rats for 12 weeks duration at 0.003, 0.03, 0.3 mg/kg/day by oral gavage resulted in adverse skeletal/growth effects (bone maturation, bone mineral density) and neuroendocrine and reproductive developmental perturbations of the hypothalamic-pituitary axis. Administration of 0.3 mg/kg/day resulted in AUC values that were similar to the AUC in adult patients receiving the recommended dose of 2.5 mg/day. Decreased fertility was accompanied by hypertrophy of the hypophysis and testicular changes that included degeneration of the seminiferous tubular epithelium and atrophy of the female reproductive tract. Young rats in this study were allowed to recover following discontinuation of letrozole treatment for 42 days. Histopathological changes were not reversible at clinically relevant exposures.
8.5 Geriatric Use
The median age of patients in all studies of first-line and second-line treatment of metastatic breast cancer was 64-65 years. About 1/3 of the patients were greater than or equal to 70 years old. In the first-line study, patients greater than or equal to 70 years of age experienced longer time to tumor progression and higher response rates than patients less than 70.
For the extended adjuvant setting (MA-17), more than 5,100 postmenopausal women were enrolled in the clinical study. In total, 41% of patients were aged 65 years or older at enrollment, while 12% were 75 or older. In the extended adjuvant setting, no overall differences in safety or efficacy were observed between these older patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
In the adjuvant setting (BIG 1-98), more than 8,000 postmenopausal women were enrolled in the clinical study. In total, 36% of patients were aged 65 years or older at enrollment, while 12% were 75 or older. More adverse reactions were generally reported in elderly patients irrespective of study treatment allocation. However, in comparison to tamoxifen, no overall differences with regard to the safety and efficacy profiles were observed between elderly patients and younger patients.
4 Contraindications (4 CONTRAINDICATIONS)
- Pregnancy: Letrozole can cause fetal harm [see Use in Specific Populations (8.1)] . Pregnancy: Letrozole can cause fetal harm [see Use in Specific Populations (8.1)] .
- Known hypersensitivity to the active substance, or to any of the excipients [see Adverse Reactions (6)] . Known hypersensitivity to the active substance, or to any of the excipients [see Adverse Reactions (6)] .
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in greater detail in other sections of the labeling.
- Bone effects [see Warnings and Precautions (5.1)]
- Increases in cholesterol [see Warnings and Precautions (5.2)]
- Fatigue and Dizziness [see Warnings and Precautions (5.4)]
2.1 Recommended Dose
The recommended dose of letrozole tablets is one 2.5 mg tablet administered once a day, without regard to meals.
12.2 Pharmacodynamics
In postmenopausal patients with advanced breast cancer, daily doses of 0.1 mg to 5 mg letrozole tablets suppress plasma concentrations of estradiol, estrone, and estrone sulfate by 75% to 95% from baseline with maximal suppression achieved within two-three days. Suppression is dose-related, with doses of 0.5 mg and higher giving many values of estrone and estrone sulfate that were below the limit of detection in the assays. Estrogen suppression was maintained throughout treatment in all patients treated at 0.5 mg or higher.
Letrozole is highly specific in inhibiting aromatase activity. There is no impairment of adrenal steroidogenesis. No clinically-relevant changes were found in the plasma concentrations of cortisol, aldosterone, 11-deoxycortisol, 17-hydroxy-progesterone, ACTH or in plasma renin activity among postmenopausal patients treated with a daily dose of letrozole tablets 0.1 mg to 5 mg. The ACTH stimulation test performed after 6 and 12 weeks of treatment with daily doses of 0.1, 0.25, 0.5, 1, 2.5, and 5 mg did not indicate any attenuation of aldosterone or cortisol production. Glucocorticoid or mineralocorticoid supplementation is, therefore, not necessary.
No changes were noted in plasma concentrations of androgens (androstenedione and testosterone) among healthy postmenopausal women after 0.1, 0.5, and 2.5 mg single doses of letrozole tablets or in plasma concentrations of androstenedione among postmenopausal patients treated with daily doses of 0.1 mg to 5 mg. This indicates that the blockade of estrogen biosynthesis does not lead to accumulation of androgenic precursors. Plasma levels of LH and FSH were not affected by letrozole in patients, nor was thyroid function as evaluated by TSH levels, T3 uptake, and T4 levels.
5.3 Hepatic Impairment
Subjects with cirrhosis and severe hepatic impairment who were dosed with 2.5 mg of letrozole tablets experienced approximately twice the exposure to letrozole tablets as healthy volunteers with normal liver function [see Clinical Pharmacology (12.3)]. Therefore, a dose reduction is recommended for this patient population. The effect of hepatic impairment on letrozole tablets exposure in cancer patients with elevated bilirubin levels has not been determined. [see Dosage and Administration (2.5)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Letrozole tablets are an aromatase inhibitor indicated for:
- Adjuvant treatment of postmenopausal women with hormone receptor positive early breast cancer ( 1.1)
- Extended adjuvant treatment of postmenopausal women with early breast cancer who have received prior standard adjuvant tamoxifen therapy ( 1.2)
- First and second-line treatment of postmenopausal women with hormone receptor positive or unknown advanced breast cancer ( 1.3)
12.1 Mechanism of Action
The growth of some cancers of the breast is stimulated or maintained by estrogens. Treatment of breast cancer thought to be hormonally responsive (i.e., estrogen and/or progesterone receptor positive or receptor unknown) has included a variety of efforts to decrease estrogen levels (ovariectomy, adrenalectomy, hypophysectomy) or inhibit estrogen effects (antiestrogens and progestational agents). These interventions lead to decreased tumor mass or delayed progression of tumor growth in some women.
In postmenopausal women, estrogens are mainly derived from the action of the aromatase enzyme, which converts adrenal androgens (primarily androstenedione and testosterone) to estrone and estradiol. The suppression of estrogen biosynthesis in peripheral tissues and in the cancer tissue itself can therefore be achieved by specifically inhibiting the aromatase enzyme.
Letrozole is a nonsteroidal competitive inhibitor of the aromatase enzyme system; it inhibits the conversion of androgens to estrogens. In adult nontumor- and tumor-bearing female animals, letrozole is as effective as ovariectomy in reducing uterine weight, elevating serum LH, and causing the regression of estrogen-dependent tumors. In contrast to ovariectomy, treatment with letrozole does not lead to an increase in serum FSH. Letrozole selectively inhibits gonadal steroidogenesis but has no significant effect on adrenal mineralocorticoid or glucocorticoid synthesis.
Letrozole inhibits the aromatase enzyme by competitively binding to the heme of the cytochrome P450 subunit of the enzyme, resulting in a reduction of estrogen biosynthesis in all tissues. Treatment of women with letrozole significantly lowers serum estrone, estradiol and estrone sulfate and has not been shown to significantly affect adrenal corticosteroid synthesis, aldosterone synthesis, or synthesis of thyroid hormones.
5.4 Fatigue and Dizziness
Because fatigue, dizziness, and somnolence have been reported with the use of letrozole tablets, caution is advised when driving or using machinery until it is known how the patient reacts to letrozole tablets use.
5.6 Embryo Fetal Toxicity (5.6 Embryo-Fetal Toxicity)
Based on post-marketing reports, findings from animal studies and the mechanism of action, letrozole tablets can cause fetal harm and is contraindicated for use in pregnant women. In post-marketing reports, use of letrozole during pregnancy resulted in cases of spontaneous abortions and congenital birth defects. Letrozole caused embryo-fetal toxicities in rats and rabbits at maternal exposures that were below the maximum recommended human dose (MHRD) on a mg/m 2 basis. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during therapy with letrozole tablets and for at least 3 weeks after the last dose [see Postmarketing Experience (6.2), Use in Specific Populations (8.1, 8.3) and Clinical Pharmacology (12.1)] .
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Decreases in bone mineral density may occur. Consider bone mineral density monitoring ( 5.1)
- Increases in total cholesterol may occur. Consider cholesterol monitoring. ( 5.2)
- Fatigue, dizziness and somnolence may occur. Exercise caution when operating machinery ( 5.4)
- Embryo-Fetal toxicity: Can cause fetal harm when administered to pregnant women. Obtain a pregnancy test in females of reproductive potential. Advise females of reproductive potential to use effective contraception ( 5.6, 8.1, 8.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
2.6 Use in Renal Impairment
No dosage adjustment is required for patients with renal impairment if creatinine clearance is greater than or equal to 10 mL/min. [see Clinical Pharmacology (12.3)] .
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
2.5 mg tablets: Yellow colored, film-coated, round shaped tablets debossed with one side 'N' and other side 'L'.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of letrozole tablets. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Eye Disorders: blurred vision
- Hepatobiliary Disorders: increased hepatic enzymes, hepatitis
- Immune System Disorders: anaphylactic reactions, hypersensitivity reactions
- Nervous System Disorders: carpal tunnel syndrome, trigger finger
- Pregnancy: spontaneous abortions, congenital birth defects
- Skin and subcutaneous disorders: angioedema, toxic epidermal necrolysis, erythema multiforme
To report SUSPECTED ADVERSE REACTIONS contact AvKARE at 1-855-361-3993; email [email protected]; or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
2.5 Use in Hepatic Impairment
No dosage adjustment is recommended for patients with mild to moderate hepatic impairment, although letrozole tablets blood concentrations were modestly increased in subjects with moderate hepatic impairment due to cirrhosis. The dose of letrozole tablets in patients with cirrhosis and severe hepatic dysfunction should be reduced by 50% [see Warnings and Precautions (5.3)]. The recommended dose of letrozole tablets for such patients is 2.5 mg administered every other day. The effect of hepatic impairment on letrozole tablets exposure in noncirrhotic cancer patients with elevated bilirubin levels has not been determined.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Lactation: Advise not to breastfeed. ( 8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reactions rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.5 Laboratory Test Abnormalities
No dose-related effect of letrozole tablets on any hematologic or clinical chemistry parameter was evident. Moderate decreases in lymphocyte counts, of uncertain clinical significance, were observed in some patients receiving letrozole tablets 2.5 mg. This depression was transient in about half of those affected. Two patients on letrozole tablets developed thrombocytopenia; relationship to the study drug was unclear. Patient withdrawal due to laboratory abnormalities, whether related to study treatment or not was infrequent.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Dispensed in Unit Dose Package. For Institutional Use Only.
| 2.5 milligram tablets | |
|
NDC 50268-476-15 (10 tablets per card, 5 cards per carton) |
|
1.1 Adjuvant Treatment of Early Breast Cancer
Letrozole tablets are indicated for the adjuvant treatment of postmenopausal women with hormone receptor positive early breast cancer.
Principal Display Panel 30 Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 30 Tablet Bottle Label)
14.4 First Line Treatment of Advanced Breast Cancer (14.4 First-Line Treatment of Advanced Breast Cancer)
A randomized, double-blind, multinational trial (P025) compared letrozole tablets 2.5 mg with tamoxifen 20 mg in 916 postmenopausal patients with locally advanced (Stage IIIB or loco-regional recurrence not amenable to treatment with surgery or radiation) or metastatic breast cancer. Time to progression (TTP) was the primary endpoint of the trial. Selected baseline characteristics for this study are shown in Table 11.
| Baseline Status | Letrozole tablets
N=458 |
Tamoxifen
N=458 |
|---|---|---|
| Stage of Disease | ||
| IIIB | 6% | 7% |
| IV | 93% | 92% |
| Receptor Status | ||
| ER and PgR Positive | 38% | 41% |
| ER or PgR Positive | 26% | 26% |
| Both Unknown | 34% | 33% |
| ER - or PgR -/Other Unknown | <1% | 0 |
| Previous Antiestrogen Therapy | ||
| Adjuvant | 19% | 18% |
| None | 81% | 82% |
| Dominant Site of Disease | ||
| Soft Tissue | 25% | 25% |
| Bone | 32% | 29% |
| Viscera | 43% | 46% |
Letrozole tablets were superior to tamoxifen in TTP and rate of objective tumor response (see Table 12).
Table 12 summarizes the results of the trial, with a total median follow-up of approximately 32 months. (All analyses are unadjusted and use 2-sided P-values.)
| Letrozole tablets
2.5 mg N=453 |
Tamoxifen
20 mg N=454 |
Hazard or Odds
Ratio (95% CI) P-Value (2-Sided) |
|
|---|---|---|---|
| Median Time to Progression | 9.4 months | 6.0 months | 0.72 (0.62, 0.83)
Hazard ratio
P<0.0001 |
| Objective Response Rate | |||
| (CR + PR) | 145 (32%) | 95 (21%) | 1.77 (1.31, 2.39)
Odds ratio
P=0.0002 |
| (CR) | 42 (9%) | 15 (3%) | 2.99 (1.63, 5.47)
P=0.0004 |
| Duration of Objective Response | |||
| Median | 18 months
(N=145) |
16 months
(N=95) |
|
| Overall Survival | 35 months
(N=458) |
32 months
(N=458) |
P=0.5136
Overall log-rank test
|
Figure 2 shows the Kaplan-Meier curves for TTP.
Table 13 shows results in the subgroup of women who had received prior antiestrogen adjuvant therapy, Table 14, results by disease site and Table 15, the results by receptor status.
| Variable | Letrozole tablets
2.5 mg N=84 |
Tamoxifen
20 mg N=83 |
|---|---|---|
| Median Time to Progression (95% CI) | 8.9 months (6.2, 12.5) | 5.9 months (3.2, 6.2) |
| Hazard Ratio for TTP (95% CI) | 0.60 (0.43, 0.84) | |
| Objective Response Rate | ||
| (CR + PR) | 22 (26%) | 7 (8%) |
| Odds Ratio for Response (95% CI) | 3.85 (1.50, 9.60) |
Hazard ratio less than 1 or odds ratio greater than 1 favors letrozole tablets; hazard ratio greater than 1 or odds ratio less than 1 favors tamoxifen.
| Letrozole tablets
2.5 mg |
Tamoxifen
20 mg |
|
|---|---|---|
| Dominant Disease Site | ||
| Soft Tissue: | N=113 | N=115 |
| Median TTP | 12.1 months | 6.4 months |
| Objective Response Rate | 50% | 34% |
| Bone: | N=145 | N=131 |
| Median TTP | 9.5 months | 6.3 months |
| Objective Response Rate | 23% | 15% |
| Viscera: | N=195 | N=208 |
| Median TTP | 8.3 months | 4.6 months |
| Objective Response Rate | 28% | 17% |
| Variable | Letrozole tablets
2.5 mg |
Tamoxifen
20 mg |
|---|---|---|
| Receptor Positive | N=294 | N=305 |
| Median Time to Progression (95% CI) | 9.4 months (8.9, 11.8) | 6.0 months (5.1, 8.5) |
| Hazard Ratio for TTP (95% CI) | 0.69 (0.58, 0.83) | |
| Objective Response Rate (CR+PR) | 97 (33%) | 66 (22%) |
| Odds Ratio for Response (95% CI) | 1.78 (1.20, 2.60) | |
| Receptor Unknown | N=159 | N=149 |
| Median Time to Progression (95% CI) | 9.2 months (6.1, 12.3) | 6.0 months (4.1, 6.4) |
| Hazard Ratio for TTP (95% CI) | 0.77 (0.60, 0.99) | |
| Objective Response Rate (CR+PR) | 48 (30%) | 29 (20%) |
| Odds Ratio for Response (95% CI) | 1.79 (1.10, 3.00) |
Hazard ratio less than 1 or odds ratio greater than 1 favors letrozole tablets; hazard ratio greater than 1 or odds ratio less than 1 favors tamoxifen.
Figure 3 shows the Kaplan-Meier curves for survival.
Legend: Randomized letrozole tablets: n=458, events 57%, median overall survival 35 months (95% CI 32 to 38 months)
Randomized tamoxifen: n=458, events 57%, median overall survival 32 months (95% CI 28 to 37 months)
Overall log-rank P=0.5136 (i.e., there was no significant difference between treatment arms in overall survival).
The median overall survival was 35 months for the letrozole tablets group and 32 months for the tamoxifen group, with a P-value 0.5136. Study design allowed patients to cross over upon progression to the other therapy. Approximately 50% of patients crossed over to the opposite treatment arm and almost all patients who crossed over had done so by 36 months. The median time to crossover was 17 months (letrozole tablets to tamoxifen) and 13 months (tamoxifen to letrozole tablets). In patients who did not cross over to the opposite treatment arm, median survival was 35 months with letrozole tablets (n=219, 95% CI 29 to 43 months) vs 20 months with tamoxifen (n=229, 95% CI 16 to 26 months).
14.5 Second Line Treatment of Advanced Breast Cancer (14.5 Second-Line Treatment of Advanced Breast Cancer)
Letrozole tablets were initially studied at doses of 0.1 mg to 5.0 mg daily in six non-comparative trials (AR/BC1, P01, AR/ST1, AR/PS1, AR/ES1 and NJO-03) in 181 postmenopausal estrogen/progesterone receptor positive or unknown advanced breast cancer patients previously treated with at least antiestrogen therapy. Patients had received other hormonal therapies and also may have received cytotoxic therapy. Eight (20%) of forty patients treated with letrozole tablets 2.5 mg daily in trials achieved an objective tumor response (complete or partial response).
Two large randomized, controlled, multinational (predominantly European) trials (AR/BC2, AR/BC3) were conducted in patients with advanced breast cancer who had progressed despite antiestrogen therapy. Patients were randomized to letrozole tablets 0.5 mg daily, letrozole tablets 2.5 mg daily, or a comparator [megestrol acetate 160 mg daily in one study (AR/BC2); and aminoglutethimide 250 mg twice a day with corticosteroid supplementation in the other study (AR/BC3)]. In each study over 60% of the patients had received therapeutic antiestrogens, and about one-fifth of these patients had had an objective response. The megestrol acetate controlled study was double-blind; the other study was open label. Selected baseline characteristics for each study are shown in Table 16.
| Parameter | megestrol acetate study | aminoglutethimide study |
|---|---|---|
| No. of Participants | 552 | 557 |
| Receptor Status | ||
| ER/PR Positive | 57% | 56% |
| ER/PR Unknown | 43% | 44% |
| Previous Therapy | ||
| Adjuvant Only | 33% | 38% |
| Therapeutic +/- Adj. | 66% | 62% |
| Sites of Disease | ||
| Soft Tissue | 56% | 50% |
| Bone | 50% | 55% |
| Viscera | 40% | 44% |
Confirmed objective tumor response (complete response plus partial response) was the primary endpoint of the trials. Responses were measured according to the Union Internationale Contre le Cancer (UICC) criteria and verified by independent, blinded review. All responses were confirmed by a second evaluation 4 to 12 weeks after the documentation of the initial response.
Table 17 shows the results for the first trial (AR/BC2), with a minimum follow-up of 15 months, that compared letrozole tablets 0.5 mg, letrozole tablets 2.5 mg, and megestrol acetate 160 mg daily. (All analyses are unadjusted.)
| Letrozole tablets
0.5 mg N=188 |
Letrozole tablets
2.5 mg N=174 |
megestrol acetate
N=190 |
|
|---|---|---|---|
| Objective Response (CR + PR) | 22 (11.7%) | 41 (23.6%) | 31 (16.3%) |
| Median Duration of Response | 552 days | (Not reached) | 561 days |
| Median Time to Progression | 154 days | 170 days | 168 days |
| Median Survival | 633 days | 730 days | 659 days |
| Odds Ratio for Response | Letrozole tablets 2.5: Letrozole tablets 0.5=2.33
(95% CI: 1.32, 4.17); P=0.004 Two-sided
P-value
|
Letrozole tablets 2.5: megestrol=1.58
(95% CI: 0.94, 2.66); P=0.08 |
|
| Relative Risk of Progression | Letrozole tablets 2.5: Letrozole tablets 0.5=0.81
(95% CI: 0.63, 1.03); P=0.09 |
Letrozole tablets 2.5: megestrol=0.77
(95% CI: 0.60, 0.98); P=0.03 |
The Kaplan-Meier curves for progression for the megestrol acetate study are shown in Figure 4.
The results for the study comparing letrozole tablets to aminoglutethimide (AR/BC3), with a minimum follow-up of 9 months, are shown in Table 18. (Unadjusted analyses are used.)
| Letrozole tablets
0.5 mg N=193 |
Letrozole tablets
2.5 mg N=185 |
aminoglutethimide
N=179 |
|
|---|---|---|---|
| Objective Response (CR + PR) | 34 (17.6%) | 34 (18.4%) | 22 (12.3%) |
| Median Duration of Response | 619 days | 706 days | 450 days |
| Median Time to Progression | 103 days | 123 days | 112 days |
| Median Survival | 636 days | 792 days | 592 days |
| Odds Ratio for Response | Letrozole tablets 2.5: Letrozole tablets 0.5=1.05
(95% CI: 0.62, 1.79); P=0.85 Two-sided
P-value
|
Letrozole tablets 2.5: aminoglutethimide=1.61
(95% CI: 0.90, 2.87); P=0.11 |
|
| Relative Risk of Progression | Letrozole tablets 2.5: Letrozole tablets 0.5=0.86
(95% CI: 0.68, 1.11); P=0.25 |
Letrozole tablets 2.5: aminoglutethimide=0.74
(95% CI: 0.57, 0.94); P=0.02 |
The Kaplan-Meier curves for progression for the aminoglutethimide study is shown in Figure 5
Figure 5 Kaplan-Meier Estimates of Time to Progression (Aminoglutethimide Study)
2.2 Use in Adjuvant Treatment of Early Breast Cancer
In the adjuvant setting, the optimal duration of treatment with letrozole is unknown. In both the adjuvant study and the postapproval adjuvant study, median treatment duration was 5 years. Treatment should be discontinued at relapse [see Clinical Studies (14.1)].
1.2 Extended Adjuvant Treatment of Early Breast Cancer
Letrozole tablets are indicated for the extended adjuvant treatment of early breast cancer in postmenopausal women, who have received 5 years of adjuvant tamoxifen therapy. The effectiveness of letrozole tablets in extended adjuvant treatment of early breast cancer is based on an analysis of disease-free survival in patients treated with letrozole tablets for a median of 60 months [see Clinical Studies (14.2, 14.3)] .
14.1 Updated Adjuvant Treatment of Early Breast Cancer
In a multicenter study (BIG 1-98, NCT00004205) enrolling over 8,000 postmenopausal women with resected, receptor-positive early breast cancer, one of the following treatments was randomized in a double-blind manner:
Option 1:
- tamoxifen for 5 years
- Letrozole tablets for 5 years
- tamoxifen for 2 years followed by letrozole tablets for 3 years
- Letrozole tablets for 2 years followed by tamoxifen for 3 years
Option 2:
- tamoxifen for 5 years
- Letrozole tablets for 5 years
The study in the adjuvant setting, BIG 1-98 was designed to answer two primary questions: whether letrozole tablets for 5 years was superior to Tamoxifen for 5 years (Primary Core Analysis) and whether switching endocrine treatments at 2 years was superior to continuing the same agent for a total of 5 years (Sequential Treatments Analysis). Selected baseline characteristics for the study population are shown in Table 6.
The primary endpoint of this trial was disease-free survival (DFS) (i.e., interval between randomization and earliest occurrence of a local, regional, or distant recurrence, or invasive contralateral breast cancer, or death from any cause). The secondary endpoints were overall survival (OS), systemic disease-free survival (SDFS), invasive contralateral breast cancer, time to breast cancer recurrence (TBR) and time to distant metastasis (TDM).
The Primary Core Analysis (PCA) included all patients and all follow-up in the monotherapy arms in both randomization options, but follow-up in the two sequential treatments arms was truncated 30 days after switching treatments. The PCA was conducted at a median treatment duration of 24 months and a median follow-up of 26 months. Letrozole tablets were superior to tamoxifen in all endpoints except overall survival and contralateral breast cancer [e.g., DFS: hazard ratio, HR 0.79; 95% CI (0.68, 0.92); P=0.002; SDFS: HR 0.83; 95% CI (0.70, 0.97); TDM: HR 0.73; 95% CI (0.60, 0.88); OS: HR 0.86; 95% CI (0.70, 1.06)].
In 2005, based on recommendations by the independent Data Monitoring Committee, the tamoxifen arms were unblinded and patients were allowed to complete initial adjuvant therapy with letrozole tablets (if they had received tamoxifen for at least 2 years) or to start extended adjuvant treatment with letrozole tablets (if they had received tamoxifen for at least 4.5 years) if they remained alive and disease-free. In total, 632 patients crossed to letrozole tablets or another aromatase inhibitor. Approximately 70% (448) of these 632 patients crossed to letrozole tablets to complete initial adjuvant therapy and most of these crossed in years 3 to 4. All of these patients were in Option 1. A total of 184 patients started extended adjuvant therapy with letrozole tablets (172 patients) or with another aromatase inhibitor (12 patients). To explore the impact of this selective crossover, results from analyses censoring follow-up at the date of the selective crossover (in the tamoxifen arm) are presented for the MAA.
The PCA allowed the results of letrozole tablets for 5 years compared with tamoxifen for 5 years to be reported in 2005 after a median follow-up of only 26 months. The design of the PCA is not optimal to evaluate the effect of letrozole tablets after a longer time (because follow-up was truncated in two arms at around 25 months). The MAA (ignoring the two sequential treatment arms) provided follow-up equally as long in each treatment and did not over-emphasize early recurrences as the PCA did. The MAA thus provides the clinically appropriate updated efficacy results in answer to the first primary question, despite the confounding of the tamoxifen reference arm by the selective crossover to letrozole tablets. The updated results for the MAA are summarized in Table 7. Median follow-up for this analysis is 73 months.
The Sequential Treatments Analysis (STA) addresses the second primary question of the study. The primary analysis for the STA was from switch (or equivalent time-point in monotherapy arms) + 30 days (STA-S) with a two-sided test applied to each pair-wise comparison at the 2.5% level. Additional analyses were conducted from randomization (STA-R) but these comparisons (added in light of changing medical practice) were under-powered for efficacy.
| Primary Core Analysis
(PCA) |
Monotherapy Arms Analysis
(MAA) |
|||
|---|---|---|---|---|
| Characteristic | Letrozole tablets
N=4003 n (%) |
Tamoxifen
N=4007 n (%) |
Letrozole tablets
N=2463 n (%) |
Tamoxifen
N=2459 n (%) |
| Age (median, years) | 61 | 61 | 61 | 61 |
| Age range (years) | 38-89 | 39-90 | 38-88 | 39-90 |
| Hormone receptor status (%) | ||||
| ER+ and/or PgR+ | 99.7 | 99.7 | 99.7 | 99.7 |
| Both unknown | 0.3 | 0.3 | 0.3 | 0.3 |
| Nodal status (%) | ||||
| Node negative | 52 | 52 | 50 | 52 |
| Node positive | 41 | 41 | 43 | 41 |
| Nodal status unknown | 7 | 7 | 7 | 7 |
| Prior adjuvant chemotherapy (%) | 24 | 24 | 24 | 24 |
| Letrozole tablets
N=2463 |
Tamoxifen
N=2459 |
Hazard ratio | |||||
|---|---|---|---|---|---|---|---|
| Events
(%) |
5-year rate | Events
(%) |
5-year rate | (95% CI) | P | ||
| ITT analysis ignores selective crossover in tamoxifen arms. | |||||||
| Censored analysis censors follow-up at the date of selective crossover in 632 patients who crossed to letrozole tablets or another aromatase inhibitor after the tamoxifen arms were unblinded in 2005. | |||||||
| Definition of: | |||||||
| Disease-free survival
Disease-free survival: Interval from randomization to earliest event of invasive loco-regional recurrence, distant metastasis, invasive contralateral breast cancer, or death without a prior event.
|
ITT | 445 (18.1) | 87.4 | 500 (20.3) | 84.7 | 0.87 (0.76, 0.99) | 0.03 |
| Censor | 445 | 87.4 | 483 | 84.2 | 0.84 (0.73, 0.95) | ||
| 0 positive nodes | ITT | 165 | 92.2 | 189 | 90.3 | 0.88 (0.72, 1.09) | |
| 1-3 positive nodes | ITT | 151 | 85.6 | 163 | 83.0 | 0.85 (0.68, 1.06) | |
| >=4 positive nodes | ITT | 123 | 71.2 | 142 | 62.6 | 0.81 (0.64, 1.03) | |
| Adjuvant chemotherapy | ITT | 119 | 86.4 | 150 | 80.6 | 0.77 (0.60, 0.98) | |
| No chemotherapy | ITT | 326 | 87.8 | 350 | 86.1 | 0.91 (0.78, 1.06) | |
| Systemic DFS
Systemic disease-free survival: Interval from randomization to invasive regional recurrence, distant metastasis, or death without a prior cancer event.
|
ITT | 401 | 88.5 | 446 | 86.6 | 0.88 (0.77, 1.01) | |
| Time to distant metastasis
Time to distant metastasis: Interval from randomization to distant metastasis.
|
ITT | 257 | 92.4 | 298 | 90.1 | 0.85 (0.72, 1.00) | |
| Adjuvant chemotherapy | ITT | 84 | - | 109 | - | 0.75 (0.56, 1.00) | |
| No chemotherapy | ITT | 173 | - | 189 | - | 0.90 (0.73, 1.11) | |
| Distant DFS
Distant disease-free survival: Interval from randomization to earlier event of relapse in a distant site or death from any cause
|
ITT | 385 | 89.0 | 432 | 87.1 | 0.87 (0.76, 1.00) | |
| Contralateral breast cancer | ITT | 34 | 99.2 | 44 | 98.6 | 0.76 (0.49, 1.19) | |
| Overall survival | ITT | 303 | 91.8 | 343 | 90.9 | 0.87 (0.75, 1.02) | |
| Censor | 303 | 91.8 | 338 | 90.1 | 0.82 (0.70, 0.96) | ||
| 0 positive nodes | ITT | 107 | 95.2 | 121 | 94.8 | 0.90 (0.69, 1.16) | |
| 1-3 positive nodes | ITT | 99 | 90.8 | 114 | 90.6 | 0.81 (0.62, 1.06) | |
| >=4 positive nodes | ITT | 92 | 80.2 | 104 | 73.6 | 0.86 (0.65, 1.14) | |
| Adjuvant chemotherapy | ITT | 76 | 91.5 | 96 | 88.4 | 0.79 (0.58, 1.06) | |
| No chemotherapy | ITT | 227 | 91.9 | 247 | 91.8 | 0.91 (0.76, 1.08) |
Figure 1 shows the Kaplan-Meier curves for Disease-Free Survival Monotherapy Analysis
DFS events defined as loco-regional recurrence, distant metastasis, invasive contralateral breast cancer, or death from any cause (i.e., definition excludes second non-breast primary cancers).
The medians of overall survival for both arms were not reached for the MAA. There was no statistically significant difference in overall survival. The hazard ratio for survival in the letrozole tablets arm compared to the tamoxifen arm was 0.87, with 95% CI (0.75, 1.02) (see Table 7).
There were no significant differences in DFS, OS, SDFS, and Distant DFS from switch in the Sequential Treatments Analysis with respect to either monotherapy (e.g., [Tamoxifen 2 years followed by] letrozole tablets 3 years versus tamoxifen beyond 2 years, DFS HR 0.89; 97.5% CI 0.68, 1.15 and [letrozole tablets 2 years followed by] tamoxifen 3 years versus letrozole tablets beyond 2 years, DFS HR 0.93; 97.5% CI 0.71, 1.22).
There were no significant differences in DFS, OS, SDFS, and Distant DFS from randomization in the Sequential Treatments Analyses.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
A conventional carcinogenesis study in mice at doses of 0.6 to 60 mg/kg/day (about 1 to 100 times the daily maximum recommended human dose on a mg/m 2 basis) administered by oral gavage for up to 2 years revealed a dose-related increase in the incidence of benign ovarian stromal tumors. The incidence of combined hepatocellular adenoma and carcinoma showed a significant trend in females when the high dose group was excluded due to low survival. In a separate study, plasma AUC 0-12hr levels in mice at 60 mg/kg/day were 55 times higher than the AUC 0-24hr level in breast cancer patients at the recommended dose. The carcinogenicity study in rats at oral doses of 0.1 to 10 mg/kg/day (about 0.4 to 40 times the daily maximum recommended human dose on a mg/m 2 basis) for up to 2 years also produced an increase in the incidence of benign ovarian stromal tumors at 10 mg/kg/day. Ovarian hyperplasia was observed in females at doses equal to or greater than 0.1 mg/kg/day. At 10 mg/kg/day, plasma AUC 0-24hr levels in rats were 80 times higher than the level in breast cancer patients at the recommended dose. The benign ovarian stromal tumors observed in mice and rats were considered to be related to the pharmacological inhibition of estrogen synthesis and may be due to increased luteinizing hormone resulting from the decrease in circulating estrogen.
Letrozole tablets were not mutagenic in in vitro tests (Ames and E.coli bacterial tests) but were observed to be a potential clastogen in in vitro assays (CHO K1 and CCL 61 Chinese hamster ovary cells). Letrozole was not clastogenic in vivo (micronucleus test in rats).
In a fertility and early embryonic development toxicity study in female rats, oral administration of letrozole starting 2 weeks before mating until pregnancy day 6 resulted in an increase in pre-implantation loss at doses ≥ 0.03 mg/kg/day (approximately 0.1 times the maximum recommended human dose on a mg/m 2 basis). In repeat-dose toxicity studies, administration of letrozole caused sexual inactivity in females and atrophy of the reproductive tract in males and females at doses of 0.6, 0.1 and 0.03 mg/kg in mice, rats and dogs, respectively (approximately 1, 0.4 and 0.4 times the daily maximum recommended human dose on a mg/m 2 basis, respectively).
1.3 First and Second Line Treatment of Advanced Breast Cancer (1.3 First and Second-Line Treatment of Advanced Breast Cancer)
Letrozole tablets are indicated for first-line treatment of postmenopausal women with hormone receptor positive or unknown, locally advanced or metastatic breast cancer. Letrozole tablets are also indicated for the treatment of advanced breast cancer in postmenopausal women with disease progression following antiestrogen therapy [see Clinical Studies (14.4, 14.5)].
2.3 Use in Extended Adjuvant Treatment of Early Breast Cancer
In the extended adjuvant setting, the optimal treatment duration with letrozole tablets is not known. The planned duration of treatment in the study was 5 years. In the final updated analysis, conducted at a median follow-up of 62 months, the median treatment duration for letrozole tablets was 60 months. Seventy-one (71%) percent of patients were treated for at least 3 years and 58% of patients completed at least 4.5 years of extended adjuvant treatment. The treatment should be discontinued at tumor relapse [see Clinical Studies (14.2)].
2.4 Use in First and Second Line Treatment of Advanced Breast Cancer (2.4 Use in First and Second-Line Treatment of Advanced Breast Cancer)
In patients with advanced disease, treatment with letrozole tablets should continue until tumor progression is evident. [see Clinical Studies (14.4, 14.5)]
14.2 Extended Adjuvant Treatment of Early Breast Cancer, Median Treatment Duration of 24 Months
A double-blind, randomized, placebo-controlled trial (MA-17, NCT00003140) of letrozole tablets was performed in over 5,100 postmenopausal women with receptor-positive or unknown primary breast cancer who were disease free after 5 years of adjuvant treatment with tamoxifen.
The planned duration of treatment for patients in the study was 5 years, but the trial was terminated early because of an interim analysis showing a favorable letrozole tablets effect on time without recurrence or contralateral breast cancer. At the time of unblinding, women had been followed for a median of 28 months, 30% of patients had completed 3 or more years of follow-up and less than 1% of patients had completed 5 years of follow-up.
Selected baseline characteristics for the study population are shown in Table 8.
| Baseline Status | Letrozole tablets
N=2582 |
Placebo
N=2586 |
|---|---|---|
| Hormone Receptor Status (%) | ||
| ER+ and/or PgR+ | 98 | 98 |
| Both Unknown | 2 | 2 |
| Nodal Status (%) | ||
| Node Negative | 50 | 50 |
| Node Positive | 46 | 46 |
| Nodal Status Unknown | 4 | 4 |
| Chemotherapy | 46 | 46 |
Table 9 shows the study results. Disease-free survival was measured as the time from randomization to the earliest event of loco-regional or distant recurrence of the primary disease or development of contralateral breast cancer or death. DFS by hormone receptor status, nodal status and adjuvant chemotherapy were similar to the overall results. Data were premature for an analysis of survival.
| Letrozole tablets
N = 2582 |
Placebo
N = 2586 |
Hazard Ratio
(95% CI) |
P-Value | |
|---|---|---|---|---|
| CI = confidence interval for hazard ratio. Hazard ratio of less than 1.0 indicates difference in favor of letrozole tablets (lesser risk of recurrence); hazard ratio greater than 1.0 indicates difference in favor of placebo (higher risk of recurrence with letrozole tablets). | ||||
| P-value based on stratified log-rank test. | ||||
|
Disease Free Survival (DFS)
First event of loco-regional recurrence, distant relapse, contralateral breast cancer or death from any cause. Events
|
122 (4.7%) | 193 (7.5%) | 0.62 (0.49, 0.78)
Analysis stratified by receptor status, nodal status and prior adjuvant chemotherapy (stratification factors as at randomization).
|
0.00003 |
| Local Breast Recurrence | 9 | 22 | ||
| Local Chest Wall Recurrence | 2 | 8 | ||
| Regional Recurrence | 7 | 4 | ||
| Distant Recurrence | 55 | 92 | 0.61 (0.44 - 0.84) | 0.003 |
| Contralateral Breast Cancer | 19 | 29 | ||
| Deaths Without Recurrence or Contralateral Breast Cancer | 30 | 38 |
14.3 Updated Analyses of Extended Adjuvant Treatment of Early Breast Cancer, Median Treatment Duration of 60 Months
| Letrozole tablets
N = 2582 (%) |
Placebo
N = 2586 (%) |
Hazard Ratio
Adjusted by receptor status, nodal status and prior chemotherapy
(95% CI) |
P-Value
Stratified log-rank test, stratified by receptor status, nodal status and prior chemotherapy
|
|
|---|---|---|---|---|
|
Disease Free Survival (DFS) events
DFS events defined as earliest of loco-regional recurrence, distant metastasis, contralateral breast cancer or death from any cause, and ignoring switches to letrozole tablets in 60% of the placebo arm.
|
344 (13.3) | 402 (15.5) | 0.89 (0.77, 1.03) | 0.12 |
|
Breast cancer recurrence
(Protocol definition of DFS events Protocol definition does not include deaths from any cause )
|
209 | 286 | 0.75 (0.63, 0.89) | 0.001 |
| Local Breast Recurrence | 15 | 44 | ||
| Local Chest Wall Recurrence | 6 | 14 | ||
| Regional Recurrence | 10 | 8 | ||
| Distant Recurrence | 140 | 167 | ||
| Distant Recurrence
(first or subsequent events) |
142 | 169 | 0.88 (0.70, 1.10) | 0.246 |
| Contralateral Breast Cancer | 37 | 53 | ||
| Deaths Without Recurrence or Contralateral Breast Cancer | 135 | 116 |
Updated analyses were conducted at a median follow-up of 62 months. In the letrozole tablets arm, 71% of the patients were treated for at least 3 years and 58% of patients completed at least 4.5 years of extended adjuvant treatment. After the unblinding of the study at a median follow-up of 28 months, approximately 60% of the selected patients in the placebo arm opted to switch to letrozole tablets.
In this updated analysis shown in Table 10, letrozole tablets significantly reduced the risk of breast cancer recurrence or contralateral breast cancer compared with placebo (HR 0.75; 95% CI 0.63, 0.89; P=0.001). However, in the updated DFS analysis (interval between randomization and earliest event of loco-regional recurrence, distant metastasis, contralateral breast cancer, or death from any cause) the treatment difference was heavily diluted by 60% of the patients in the placebo arm switching to letrozole tablets and accounting for 64% of the total placebo patient-years of follow-up. Ignoring these switches, the risk of DFS event was reduced by a non-significant 11% (HR 0.89; 95% CI 0.77, 1.03). There was no significant difference in distant disease-free survival or overall survival.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:39:50.042907 · Updated: 2026-03-14T22:43:50.676219