These Highlights Do Not Include All The Information Needed To Use Zeposia Safely And Effectively. See Full Prescribing Information For Zeposia.
93ce2fab-edfb-4804-8074-963071de51e4
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dosage and Administration ( 2.1 ) 6/2024 Warnings and Precautions ( 5.2 , 5.8 , 5.9 ) 6/2024 Warnings and Precautions ( 5.4 , 5.6 ) 8/2024
Indications and Usage
ZEPOSIA is indicated for the treatment of: • relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults. • moderately to severely active ulcerative colitis (UC) in adults.
Dosage and Administration
• Assessments are required prior to initiating ZEPOSIA. ( 2.1 ) • Titration is required for treatment initiation. ( 2.2 ) • The recommended maintenance dosage is 0.92 mg orally once daily. ( 2.2 ) • The recommended maintenance dosage in patients with mild or moderate chronic hepatic impairment (Child-Pugh class A or B) is 0.92 mg once every other day. ( 2.3 ) • If a dose is missed within the first 2 weeks of treatment, reinitiate with the titration regimen. If a dose is missed after the first 2 weeks of treatment, continue treatment as planned. ( 2.4 )
Warnings and Precautions
• Infections : ZEPOSIA may increase the risk of infections. Obtain a complete blood count (CBC) before initiation of treatment. Monitor for infection during treatment and for 3 months after discontinuation. Do not start ZEPOSIA in patients with active infections. ( 5.1 ) • Progressive Multifocal Leukoencephalopathy (PML) : Withhold ZEPOSIA at the first sign or symptom suggestive of PML. ( 5.2 ) • Bradyarrhythmia and Atrioventricular Conduction Delays : ZEPOSIA may result in transient decrease in heart rate; titration is required for treatment initiation. Check an electrocardiogram (ECG) to assess for preexisting cardiac conduction abnormalities before starting ZEPOSIA. Consider cardiology consultation for conduction abnormalities or concomitant use with other drugs that decrease heart rate. ( 2.1 , 2.2 , 5.3 , 7 ) • Liver Injury : Obtain liver enzyme results before initiation and periodically during treatment. Discontinue if there is evidence of liver injury without other cause. ( 5.4 ) • Fetal Risk : Women of childbearing potential should use effective contraception during treatment and for 3 months after stopping ZEPOSIA. ( 5.5 , 8.3 ) • Increased Blood Pressure (BP) : Monitor BP during treatment. ( 5.6 ) • Respiratory Effects : May cause a decline in pulmonary function. Assess pulmonary function (e.g., spirometry) if clinically indicated. ( 5.7 ) • Macular Edema : Increases the risk of macular edema. Obtain a baseline evaluation of the fundus, including the macula, near the start of treatment with ZEPOSIA. Conduct an evaluation of the fundus, including the macula, periodically while on therapy and any time there is a change in vision. Consider discontinuing ZEPOSIA if macular edema develops. Diabetes mellitus and uveitis increase the risk. ( 5.8 ) • Cutaneous Malignancies : Skin examination prior to or shortly after the start of treatment and periodically thereafter is recommended. Suspicious skin lesions should be evaluated. ( 5.9 )
Contraindications
ZEPOSIA is contraindicated in patients who: • In the last 6 months, have experienced a myocardial infarction, unstable angina, stroke, transient ischemic attack (TIA), decompensated heart failure requiring hospitalization, or Class III or IV heart failure [see Warnings and Precautions (5.3) ] • Have the presence of Mobitz type II second-degree or third degree atrioventricular (AV) block, sick sinus syndrome, or sino-atrial block, unless the patient has a functioning pacemaker [see Warnings and Precautions (5.3) ] • Have severe untreated sleep apnea [see Warnings and Precautions (5.3) ] • Are taking a monoamine oxidase (MAO) inhibitor [see Drug Interactions (7) ]
Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling: • Infections [see Warnings and Precautions (5.1) ] • Progressive Multifocal Leukoencephalopathy [see Warnings and Precautions (5.2) ] • Bradyarrhythmia and Atrioventricular Conduction Delays [see Warnings and Precautions (5.3) ] • Liver Injury [see Warnings and Precautions (5.4) ] • Fetal Risk [see Warnings and Precautions (5.5) ] • Increased Blood Pressure [see Warnings and Precautions (5.6) ] • Respiratory Effects [see Warnings and Precautions (5.7) ] • Macular Edema [see Warnings and Precautions (5.8) ] • Cutaneous Malignancies [see Warnings and Precautions (5.9) ] • Posterior Reversible Encephalopathy Syndrome [see Warnings and Precautions (5.10) ] • Unintended Additive Immunosuppressive Effects from Prior Treatment with Immunosuppressive or Immune-Modulating Drugs [see Warnings and Precautions (5.11) ] • Severe Increase in Multiple Sclerosis Disability after Stopping ZEPOSIA [see Warnings and Precautions (5.12) ] • Immune System Effects after Stopping ZEPOSIA [see Warnings and Precautions (5.13) ]
Drug Interactions
Tables 5 and 6 include drugs with clinically important drug and vaccine interactions when administered concomitantly with ZEPOSIA and instructions for preventing or managing them. Table 5: Clinically Relevant Interactions Affecting Drugs and Vaccines Co-administered with ZEPOSIA Anti-Neoplastic, Immune-Modulating, or Non-Corticosteroid Immunosuppressive Therapies Clinical Impact: ZEPOSIA has not been studied in combination with anti-neoplastic, immune-modulating, or non-corticosteroid immunosuppressive therapies with the exception of cyclosporine, which had no pharmacokinetic interaction [see Clinical Pharmacology (12.3) ] . Prevention or Management: Caution should be used during concomitant administration because of the risk of additive immune effects during such therapy and in the weeks following administration [see Warnings and Precautions (5.1) ] . When switching from drugs with prolonged immune effects, the half-life and mode of action of these drugs must be considered in order to avoid unintended additive immunosuppressive effects [see Warnings and Precautions (5.11) ] . Alemtuzumab : Initiating treatment with ZEPOSIA after alemtuzumab is not recommended because of the characteristics and duration of alemtuzumab immune suppressive effects. Beta interferon or glatiramer acetate : ZEPOSIA can generally be started immediately after discontinuation of beta interferon or glatiramer acetate. Anti-Arrhythmic Drugs, QT Prolonging Drugs, Drugs That May Decrease Heart Rate Clinical Impact: ZEPOSIA has not been studied in patients taking QT prolonging drugs. Class Ia (e.g., quinidine, procainamide) and Class III (e.g., amiodarone, sotalol) anti-arrhythmic drugs have been associated with cases of Torsades de Pointes in patients with bradycardia. Prevention or Management: If treatment with ZEPOSIA is considered in patients on Class Ia or Class III anti-arrhythmic drugs, advice from a cardiologist should be sought [see Warnings and Precautions (5.3) ] . Because of the potential additive effects on heart rate, treatment with ZEPOSIA should generally not be initiated in patients who are concurrently treated with QT prolonging drugs with known arrhythmogenic properties [see Warnings and Precautions (5.3) ]. If treatment initiation with ZEPOSIA is considered in patients on QT prolonging drugs, advice from a cardiologist should be sought. Combination Beta Blocker and Calcium Channel Blocker Clinical Impact : The co-administration of ZEPOSIA with both a beta blocker and a calcium channel blocker has not been studied. However, there is a potential of additive effects on heart rate. Prevention or Management: Treatment with ZEPOSIA should generally not be initiated in patients who are concurrently treated with both a heart rate lowering calcium channel blocker (e.g., verapamil, diltiazem) and beta blocker [see Warnings and Precautions (5.3) ]. If treatment initiation with ZEPOSIA is considered in patients on both a heart rate lowering calcium channel blocker and beta blocker, advice from a cardiologist should be sought. Vaccination Clinical Impact: During, and for up to three months after, discontinuation of treatment with ZEPOSIA, vaccinations may be less effective. The use of live attenuated vaccines may carry the risk of infection. Prevention or Management: Live attenuated vaccines should be avoided during ZEPOSIA treatment and for up to 3 months after discontinuation of treatment with ZEPOSIA [see Warnings and Precautions (5.1) ] . Table 6: Clinically Relevant Interactions Affecting ZEPOSIA When Co-administered with Other Drugs Monoamine Oxidase (MAO) Inhibitors Clinical Impact: The effect of MAO inhibition on ozanimod and/or its metabolites has not been studied clinically. Potential effects on efficacy or safety with co-administration of MAO inhibitors because of altered exposures of ozanimod and/or its metabolites cannot be ruled out. Prevention or Management: Co-administration of ZEPOSIA with MAO inhibitors (e.g., selegiline, phenelzine, linezolid) is contraindicated. At least 14 days should elapse between discontinuation of ZEPOSIA and initiation of treatment with MAO inhibitors. Strong CYP2C8 Inhibitors Clinical Impact: Co-administration of ZEPOSIA with strong CYP2C8 inhibitors increases the exposure of the active metabolites of ozanimod [see Clinical Pharmacology (12.3) ] , which may increase the risk of ZEPOSIA adverse reactions. Prevention or Management: Co-administration of ZEPOSIA with strong CYP2C8 inhibitors (e.g., gemfibrozil) is not recommended. Strong CYP2C8 Inducers Clinical Impact: Co-administration of ZEPOSIA with strong CYP2C8 inducers (e.g., rifampin) reduces the exposure of the major active metabolites of ozanimod [see Clinical Pharmacology (12.3) ] , which may decrease the efficacy of ZEPOSIA. Prevention or Management: Co-administration of ZEPOSIA with strong CYP2C8 inducers should be avoided.
How Supplied
ZEPOSIA is available as capsules in the following dosage strengths: • 0.23 mg ozanimod: light grey opaque body/light grey opaque cap imprinted with black ink "OZA" on the cap and "0.23 mg" on the body • 0.46 mg ozanimod: light grey opaque body/orange opaque cap imprinted with black ink "OZA" on the cap and "0.46 mg" on the body • 0.92 mg ozanimod: orange opaque body/orange opaque cap imprinted with black ink "OZA" on the cap and "0.92 mg" on the body Capsules are supplied in the following strengths and package configurations: Package configuration Tablet strength NDC number Bottles of 30 0.92 mg ozanimod 59572-820-30 7-Day Starter Pack 7-capsule starter pack containing: (4) 0.23 mg ozanimod capsules and (3) 0.46 ozanimod mg capsules 59572-810-07 Starter Kit (7-Day Starter Pack and 0.92 mg 30-count Bottle) 37-capsule starter kit including: one 7-capsule starter pack containing: (4) 0.23 mg ozanimod capsules and (3) 0.46 mg ozanimod capsules and one bottle containing: (30) 0.92 mg ozanimod capsules 59572-890-91 59572-890-07 59572-890-30 Starter Kit (7-Day Starter Pack and 0.92 mg 21-count Bottle) 28-capsule starter kit including: one 7-capsule starter pack containing: (4) 0.23 mg ozanimod capsules and (3) 0.46 mg ozanimod capsules and one bottle containing: (21) 0.92 mg ozanimod capsules 59572-890-28 59572-890-07 59572-890-21
Medication Information
Warnings and Precautions
• Infections : ZEPOSIA may increase the risk of infections. Obtain a complete blood count (CBC) before initiation of treatment. Monitor for infection during treatment and for 3 months after discontinuation. Do not start ZEPOSIA in patients with active infections. ( 5.1 ) • Progressive Multifocal Leukoencephalopathy (PML) : Withhold ZEPOSIA at the first sign or symptom suggestive of PML. ( 5.2 ) • Bradyarrhythmia and Atrioventricular Conduction Delays : ZEPOSIA may result in transient decrease in heart rate; titration is required for treatment initiation. Check an electrocardiogram (ECG) to assess for preexisting cardiac conduction abnormalities before starting ZEPOSIA. Consider cardiology consultation for conduction abnormalities or concomitant use with other drugs that decrease heart rate. ( 2.1 , 2.2 , 5.3 , 7 ) • Liver Injury : Obtain liver enzyme results before initiation and periodically during treatment. Discontinue if there is evidence of liver injury without other cause. ( 5.4 ) • Fetal Risk : Women of childbearing potential should use effective contraception during treatment and for 3 months after stopping ZEPOSIA. ( 5.5 , 8.3 ) • Increased Blood Pressure (BP) : Monitor BP during treatment. ( 5.6 ) • Respiratory Effects : May cause a decline in pulmonary function. Assess pulmonary function (e.g., spirometry) if clinically indicated. ( 5.7 ) • Macular Edema : Increases the risk of macular edema. Obtain a baseline evaluation of the fundus, including the macula, near the start of treatment with ZEPOSIA. Conduct an evaluation of the fundus, including the macula, periodically while on therapy and any time there is a change in vision. Consider discontinuing ZEPOSIA if macular edema develops. Diabetes mellitus and uveitis increase the risk. ( 5.8 ) • Cutaneous Malignancies : Skin examination prior to or shortly after the start of treatment and periodically thereafter is recommended. Suspicious skin lesions should be evaluated. ( 5.9 )
Indications and Usage
ZEPOSIA is indicated for the treatment of: • relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults. • moderately to severely active ulcerative colitis (UC) in adults.
Dosage and Administration
• Assessments are required prior to initiating ZEPOSIA. ( 2.1 ) • Titration is required for treatment initiation. ( 2.2 ) • The recommended maintenance dosage is 0.92 mg orally once daily. ( 2.2 ) • The recommended maintenance dosage in patients with mild or moderate chronic hepatic impairment (Child-Pugh class A or B) is 0.92 mg once every other day. ( 2.3 ) • If a dose is missed within the first 2 weeks of treatment, reinitiate with the titration regimen. If a dose is missed after the first 2 weeks of treatment, continue treatment as planned. ( 2.4 )
Contraindications
ZEPOSIA is contraindicated in patients who: • In the last 6 months, have experienced a myocardial infarction, unstable angina, stroke, transient ischemic attack (TIA), decompensated heart failure requiring hospitalization, or Class III or IV heart failure [see Warnings and Precautions (5.3) ] • Have the presence of Mobitz type II second-degree or third degree atrioventricular (AV) block, sick sinus syndrome, or sino-atrial block, unless the patient has a functioning pacemaker [see Warnings and Precautions (5.3) ] • Have severe untreated sleep apnea [see Warnings and Precautions (5.3) ] • Are taking a monoamine oxidase (MAO) inhibitor [see Drug Interactions (7) ]
Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling: • Infections [see Warnings and Precautions (5.1) ] • Progressive Multifocal Leukoencephalopathy [see Warnings and Precautions (5.2) ] • Bradyarrhythmia and Atrioventricular Conduction Delays [see Warnings and Precautions (5.3) ] • Liver Injury [see Warnings and Precautions (5.4) ] • Fetal Risk [see Warnings and Precautions (5.5) ] • Increased Blood Pressure [see Warnings and Precautions (5.6) ] • Respiratory Effects [see Warnings and Precautions (5.7) ] • Macular Edema [see Warnings and Precautions (5.8) ] • Cutaneous Malignancies [see Warnings and Precautions (5.9) ] • Posterior Reversible Encephalopathy Syndrome [see Warnings and Precautions (5.10) ] • Unintended Additive Immunosuppressive Effects from Prior Treatment with Immunosuppressive or Immune-Modulating Drugs [see Warnings and Precautions (5.11) ] • Severe Increase in Multiple Sclerosis Disability after Stopping ZEPOSIA [see Warnings and Precautions (5.12) ] • Immune System Effects after Stopping ZEPOSIA [see Warnings and Precautions (5.13) ]
Drug Interactions
Tables 5 and 6 include drugs with clinically important drug and vaccine interactions when administered concomitantly with ZEPOSIA and instructions for preventing or managing them. Table 5: Clinically Relevant Interactions Affecting Drugs and Vaccines Co-administered with ZEPOSIA Anti-Neoplastic, Immune-Modulating, or Non-Corticosteroid Immunosuppressive Therapies Clinical Impact: ZEPOSIA has not been studied in combination with anti-neoplastic, immune-modulating, or non-corticosteroid immunosuppressive therapies with the exception of cyclosporine, which had no pharmacokinetic interaction [see Clinical Pharmacology (12.3) ] . Prevention or Management: Caution should be used during concomitant administration because of the risk of additive immune effects during such therapy and in the weeks following administration [see Warnings and Precautions (5.1) ] . When switching from drugs with prolonged immune effects, the half-life and mode of action of these drugs must be considered in order to avoid unintended additive immunosuppressive effects [see Warnings and Precautions (5.11) ] . Alemtuzumab : Initiating treatment with ZEPOSIA after alemtuzumab is not recommended because of the characteristics and duration of alemtuzumab immune suppressive effects. Beta interferon or glatiramer acetate : ZEPOSIA can generally be started immediately after discontinuation of beta interferon or glatiramer acetate. Anti-Arrhythmic Drugs, QT Prolonging Drugs, Drugs That May Decrease Heart Rate Clinical Impact: ZEPOSIA has not been studied in patients taking QT prolonging drugs. Class Ia (e.g., quinidine, procainamide) and Class III (e.g., amiodarone, sotalol) anti-arrhythmic drugs have been associated with cases of Torsades de Pointes in patients with bradycardia. Prevention or Management: If treatment with ZEPOSIA is considered in patients on Class Ia or Class III anti-arrhythmic drugs, advice from a cardiologist should be sought [see Warnings and Precautions (5.3) ] . Because of the potential additive effects on heart rate, treatment with ZEPOSIA should generally not be initiated in patients who are concurrently treated with QT prolonging drugs with known arrhythmogenic properties [see Warnings and Precautions (5.3) ]. If treatment initiation with ZEPOSIA is considered in patients on QT prolonging drugs, advice from a cardiologist should be sought. Combination Beta Blocker and Calcium Channel Blocker Clinical Impact : The co-administration of ZEPOSIA with both a beta blocker and a calcium channel blocker has not been studied. However, there is a potential of additive effects on heart rate. Prevention or Management: Treatment with ZEPOSIA should generally not be initiated in patients who are concurrently treated with both a heart rate lowering calcium channel blocker (e.g., verapamil, diltiazem) and beta blocker [see Warnings and Precautions (5.3) ]. If treatment initiation with ZEPOSIA is considered in patients on both a heart rate lowering calcium channel blocker and beta blocker, advice from a cardiologist should be sought. Vaccination Clinical Impact: During, and for up to three months after, discontinuation of treatment with ZEPOSIA, vaccinations may be less effective. The use of live attenuated vaccines may carry the risk of infection. Prevention or Management: Live attenuated vaccines should be avoided during ZEPOSIA treatment and for up to 3 months after discontinuation of treatment with ZEPOSIA [see Warnings and Precautions (5.1) ] . Table 6: Clinically Relevant Interactions Affecting ZEPOSIA When Co-administered with Other Drugs Monoamine Oxidase (MAO) Inhibitors Clinical Impact: The effect of MAO inhibition on ozanimod and/or its metabolites has not been studied clinically. Potential effects on efficacy or safety with co-administration of MAO inhibitors because of altered exposures of ozanimod and/or its metabolites cannot be ruled out. Prevention or Management: Co-administration of ZEPOSIA with MAO inhibitors (e.g., selegiline, phenelzine, linezolid) is contraindicated. At least 14 days should elapse between discontinuation of ZEPOSIA and initiation of treatment with MAO inhibitors. Strong CYP2C8 Inhibitors Clinical Impact: Co-administration of ZEPOSIA with strong CYP2C8 inhibitors increases the exposure of the active metabolites of ozanimod [see Clinical Pharmacology (12.3) ] , which may increase the risk of ZEPOSIA adverse reactions. Prevention or Management: Co-administration of ZEPOSIA with strong CYP2C8 inhibitors (e.g., gemfibrozil) is not recommended. Strong CYP2C8 Inducers Clinical Impact: Co-administration of ZEPOSIA with strong CYP2C8 inducers (e.g., rifampin) reduces the exposure of the major active metabolites of ozanimod [see Clinical Pharmacology (12.3) ] , which may decrease the efficacy of ZEPOSIA. Prevention or Management: Co-administration of ZEPOSIA with strong CYP2C8 inducers should be avoided.
How Supplied
ZEPOSIA is available as capsules in the following dosage strengths: • 0.23 mg ozanimod: light grey opaque body/light grey opaque cap imprinted with black ink "OZA" on the cap and "0.23 mg" on the body • 0.46 mg ozanimod: light grey opaque body/orange opaque cap imprinted with black ink "OZA" on the cap and "0.46 mg" on the body • 0.92 mg ozanimod: orange opaque body/orange opaque cap imprinted with black ink "OZA" on the cap and "0.92 mg" on the body Capsules are supplied in the following strengths and package configurations: Package configuration Tablet strength NDC number Bottles of 30 0.92 mg ozanimod 59572-820-30 7-Day Starter Pack 7-capsule starter pack containing: (4) 0.23 mg ozanimod capsules and (3) 0.46 ozanimod mg capsules 59572-810-07 Starter Kit (7-Day Starter Pack and 0.92 mg 30-count Bottle) 37-capsule starter kit including: one 7-capsule starter pack containing: (4) 0.23 mg ozanimod capsules and (3) 0.46 mg ozanimod capsules and one bottle containing: (30) 0.92 mg ozanimod capsules 59572-890-91 59572-890-07 59572-890-30 Starter Kit (7-Day Starter Pack and 0.92 mg 21-count Bottle) 28-capsule starter kit including: one 7-capsule starter pack containing: (4) 0.23 mg ozanimod capsules and (3) 0.46 mg ozanimod capsules and one bottle containing: (21) 0.92 mg ozanimod capsules 59572-890-28 59572-890-07 59572-890-21
Description
Dosage and Administration ( 2.1 ) 6/2024 Warnings and Precautions ( 5.2 , 5.8 , 5.9 ) 6/2024 Warnings and Precautions ( 5.4 , 5.6 ) 8/2024
Section 42229-5
Cardiac Evaluation
Obtain an electrocardiogram (ECG) to determine whether preexisting conduction abnormalities are present. In patients with certain preexisting conditions, advice from a cardiologist should be sought [see Warnings and Precautions (5.3)].
Complete Blood Count
Obtain a recent (i.e., within the last 6 months or after discontinuation of prior MS or UC therapy) complete blood count (CBC), including lymphocyte count [see Warnings and Precautions (5.1)].
Liver Function Tests
Obtain recent (i.e., within the last 6 months) transaminase and bilirubin levels [see Warnings and Precautions (5.4)].
Ophthalmic Assessment
Obtain a baseline evaluation of the fundus, including the macula, near the start of treatment with ZEPOSIA [see Warnings and Precautions (5.8)].
Skin Examination
Obtain a baseline skin examination prior to or shortly after initiation of ZEPOSIA. If a suspicious skin lesion is observed, it should be promptly evaluated [see Warnings and Precautions (5.9)].
Current or Prior Medications
-
•If patients are taking anti-neoplastic, non-corticosteroid immunosuppressive, or immune-modulating therapies, or if there is a history of prior use of these drugs, consider possible unintended additive immunosuppressive effects before initiating treatment with ZEPOSIA [see Warnings and Precautions (5.1) and Drug Interactions (7)].
-
•Determine if patients are taking drugs that could slow heart rate or atrioventricular conduction [see Warnings and Precautions (5.3) and Drug Interactions (7)].
Vaccinations
Patients without a healthcare professional-confirmed history of chickenpox or without documentation of a full course of vaccination against varicella zoster virus (VZV) should be tested for antibodies to VZV before initiating ZEPOSIA; VZV vaccination of antibody-negative patients is recommended prior to commencing treatment with ZEPOSIA [see Warnings and Precautions (5.1) and Drug Interactions (7)].
If live attenuated vaccine immunizations are required, administer at least 1 month prior to initiation of ZEPOSIA.
Section 42231-1
|
MEDICATION GUIDE
|
|||
|
Read this Medication Guide before you start taking ZEPOSIA and each time you get a refill. There may be new information. This Medication Guide does not take the place of talking with your healthcare provider about your medical condition or treatment. |
|||
|
What is the most important information I should know about ZEPOSIA?
|
|||
|
|
||
|
Your healthcare provider may delay starting or may stop your ZEPOSIA treatment if you have an infection. |
|||
|
|||
|
|
||
|
|||
|
|
||
|
Follow directions from your healthcare provider when starting ZEPOSIA and when you miss a dose. See "How should I take ZEPOSIA?". |
|||
|
See "What are possible side effects of ZEPOSIA?" for more information about side effects. |
|||
|
What is ZEPOSIA?
It is not known if ZEPOSIA is safe and effective in children. |
|||
|
Do not take ZEPOSIA if you:
Talk to your healthcare provider before taking ZEPOSIA if you have any of these conditions or do not know if you have any of these conditions. |
|||
|
Before taking ZEPOSIA, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take or have recently taken, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Using ZEPOSIA with other medicines can cause serious side effects. Especially tell your healthcare provider if you take or have taken:
You should not receive live vaccines during treatment with ZEPOSIA, for at least 1 month before taking ZEPOSIA and for 3 months after you stop taking ZEPOSIA. Vaccines may not work as well when given during treatment with ZEPOSIA. |
|||
|
How should I take ZEPOSIA?
|
|||
|
Days 1-4 |
Take 0.23 mg (capsule in light grey color) 1 time a day |
||
|
Days 5-7 |
Take 0.46 mg (capsule in half-light grey and half-orange color) 1 time a day |
||
|
Days 8 and thereafter |
Take 0.92 mg (capsule in orange color) 1 time a day, or as directed by your healthcare provider |
||
|
|||
|
What are the possible side effects of ZEPOSIA?
|
|||
|
|
||
|
|||
|
|
||
|
|||
|
|
||
|
|||
|
The most common side effects of ZEPOSIA can include: |
|||
|
|
|
|
|
These are not all of the possible side effects of ZEPOSIA. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
|
How should I store ZEPOSIA?
Keep ZEPOSIA and all medicines out of the reach of children. |
|||
|
General information about the safe and effective use of ZEPOSIA.
|
|||
|
What are the ingredients in ZEPOSIA?
|
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Revised: 8/2024
ZEPMG.009
Section 43683-2
16.2 Storage
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
8.1 Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to ZEPOSIA during pregnancy. Healthcare providers are encouraged to register patients on-line, or pregnant women may register themselves at https://www.zeposiapregnancyregistry.com/or by calling 1-877-301-9314. Currently this registry is enrolling women with MS. Information regarding registration of pregnant women with UC will be made available in the future.
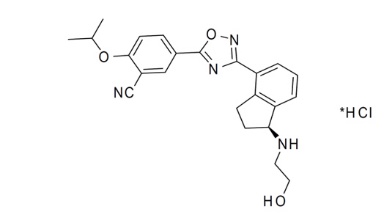
11 Description
ZEPOSIA contains ozanimod, a sphingosine 1-phosphate receptor modulator and is supplied as ozanimod hydrochloride (HCl).
The chemical name of ozanimod HCl is 5-(3-{(1S)-1-[(2-hydroxyethyl)amino]-2,3-dihydro-1H-inden-4-yl}-1,2,4-oxadiazol-5-yl)-2-[(propan-2-yl)oxy]benzonitrile, monohydrochloride.
Ozanimod HCl is a white to off-white solid that is freely soluble in water and alcohol with a molecular weight of 440.92 g/mol.
The chemical structure is:
ZEPOSIA capsules are provided as hard gelatin capsules for oral administration, containing 0.23, 0.46, or 0.92 mg of ozanimod (equivalent to 0.25, 0.5, and 1 mg ozanimod HCl, respectively). ZEPOSIA capsules consist of the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, and microcrystalline cellulose. The capsule shell, imprinted with black ink, contains the following inactive ingredients: black iron oxide, gelatin, red iron oxide, titanium dioxide, and yellow iron oxide.
5.5 Fetal Risk
There are no adequate and well-controlled studies in pregnant women. Based on animal studies, ZEPOSIA may cause fetal harm [see Use in Specific Populations (8.1)]. Because it takes approximately 3 months to eliminate ZEPOSIA from the body, women of childbearing potential should use effective contraception to avoid pregnancy during treatment and for 3 months after stopping ZEPOSIA [see Use in Specific Populations (8.3)].
5.4 Liver Injury
Clinically significant liver injury, including acute liver failure requiring transplant, has occurred in patients treated with ZEPOSIA in the postmarketing setting. Signs of liver injury, including elevated serum hepatic enzymes and elevated total bilirubin, have occurred as early as ten days after the first dose.
In MS Study 1 and Study 2, elevations of ALT to 5-fold the upper limit of normal (ULN) or greater occurred in 1.6% of patients treated with ZEPOSIA 0.92 mg and 1.3% of patients who received IFN beta-1a. Elevations of 3-fold the ULN or greater occurred in 5.5% of patients treated with ZEPOSIA and 3.1% of patients who received IFN beta-1a. The median time to an elevation of 3-fold the ULN was 6 months. The majority (79%) of patients continued treatment with ZEPOSIA with values returning to less than 3-fold the ULN within approximately 2-4 weeks. ZEPOSIA was discontinued for a confirmed elevation greater than 5-fold the ULN. Overall, the discontinuation rate because of elevations in hepatic enzymes was 1.1% of patients with MS treated with ZEPOSIA 0.92 mg and 0.8% of patients who received IFN beta-1a.
In UC Study 1, elevations of ALT to 5-fold the ULN or greater occurred in 0.9% of patients treated with ZEPOSIA 0.92 mg and 0.5% of patients who received placebo, and in UC Study 2 elevations occurred in 0.9% of patients and no patients, respectively. In UC Study 1, elevations of ALT to 3-fold the ULN or greater occurred in 2.6% of UC patients treated with ZEPOSIA 0.92 mg and 0.5% of patients who received placebo, and in UC Study 2 elevations occurred in 2.3% of patients and no patients, respectively. In controlled and uncontrolled UC studies, the majority (96%) of patients with ALT greater than 3-fold the ULN continued treatment with ZEPOSIA with values returning to less than 3-fold the ULN within approximately 2 to 4 weeks. Overall, the discontinuation rate because of elevations in hepatic enzymes was 0.4% in patients treated with ZEPOSIA 0.92 mg, and none in patients who received placebo in the controlled UC studies.
Individuals with an AST or ALT greater than 1.5-fold ULN were excluded from MS Study 1 and Study 2 and greater than 2 times the ULN for UC Study 1 and Study 3. There are no data to establish that patients with preexisting liver disease are at increased risk to develop elevated liver function test values when taking ZEPOSIA. Dosage adjustment in patients with mild or moderate hepatic impairment (Child-Pugh class A or B) is required [see Dosage and Administration (2.3)], and use of ZEPOSIA in patients with severe hepatic impairment (Child-Pugh class C) is not recommended [see Use in Specific Populations (8.6)].
Obtain transaminase and bilirubin levels, if not recently available (i.e., within 6 months), before initiation of ZEPOSIA. Obtain transaminase levels and total bilirubin levels periodically during treatment and until two months after ZEPOSIA discontinuation.
Patients should be monitored for signs and symptoms of any hepatic injury. Patients who develop symptoms suggestive of hepatic dysfunction, such as unexplained nausea, vomiting, abdominal pain, fatigue, anorexia, or jaundice and/or dark urine, should have hepatic enzymes promptly checked, and ZEPOSIA should be interrupted. Treatment should not be resumed if a plausible alternative etiology for the signs and symptoms cannot be established, because these patients are at risk for severe drug-induced liver injury.
16.1 How Supplied
ZEPOSIA is available as capsules in the following dosage strengths:
-
•0.23 mg ozanimod: light grey opaque body/light grey opaque cap imprinted with black ink "OZA" on the cap and "0.23 mg" on the body
-
•0.46 mg ozanimod: light grey opaque body/orange opaque cap imprinted with black ink "OZA" on the cap and "0.46 mg" on the body
-
•0.92 mg ozanimod: orange opaque body/orange opaque cap imprinted with black ink "OZA" on the cap and "0.92 mg" on the body
Capsules are supplied in the following strengths and package configurations:
|
Package configuration |
Tablet strength |
NDC number |
|
Bottles of 30 |
0.92 mg ozanimod |
59572-820-30 |
|
7-Day Starter Pack |
7-capsule starter pack containing: (4) 0.23 mg ozanimod capsules and (3) 0.46 ozanimod mg capsules |
59572-810-07 |
|
Starter Kit |
37-capsule starter kit including: |
59572-890-91 |
|
Starter Kit (7-Day Starter Pack and 0.92 mg 21-count Bottle) |
28-capsule starter kit including: |
59572-890-28 |
5.8 Macular Edema
S1P receptor modulators, including ZEPOSIA, have been associated with an increased risk of macular edema. Obtain a baseline evaluation of the fundus, including the macula, near the start of treatment with ZEPOSIA. Perform an examination of the fundus, including the macula, periodically while on therapy and any time there is a change in vision.
In MS Study 1 and Study 2, macular edema was observed in 0.3% of patients treated with ZEPOSIA and in 0.3% of patients who received IFN beta-1a. Macular edema was reported in a total of 1 (0.2%) patient in UC Study 1 and Study 3, and in 1 (0.4%) patient in UC Study 2 treated with ZEPOSIA, and in no patients who received placebo.
Continuation of ZEPOSIA therapy in patients with macular edema has not been evaluated. Macular edema over an extended period of time (i.e., 6 months) can lead to permanent visual loss. Consider discontinuing ZEPOSIA if macular edema develops; this decision should include an assessment of the potential benefits and risks for the individual patient. The risk of recurrence after rechallenge has not been evaluated.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of ZEPOSIA did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. No clinically significant differences in the pharmacokinetics of ozanimod and CC112273 were observed based on age [see Clinical Pharmacology (12.3)]. Monitor elderly patients for cardiac and hepatic adverse reactions, because of the greater frequency of reduced cardiac and hepatic function in the elderly population.
4 Contraindications
ZEPOSIA is contraindicated in patients who:
-
•In the last 6 months, have experienced a myocardial infarction, unstable angina, stroke, transient ischemic attack (TIA), decompensated heart failure requiring hospitalization, or Class III or IV heart failure [see Warnings and Precautions (5.3) ]
-
•Have the presence of Mobitz type II second-degree or third degree atrioventricular (AV) block, sick sinus syndrome, or sino-atrial block, unless the patient has a functioning pacemaker [see Warnings and Precautions (5.3)]
-
•Have severe untreated sleep apnea [see Warnings and Precautions (5.3)]
-
•Are taking a monoamine oxidase (MAO) inhibitor [see Drug Interactions (7)]
6 Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling:
-
•Infections [see Warnings and Precautions (5.1)]
-
•Progressive Multifocal Leukoencephalopathy [see Warnings and Precautions (5.2)]
-
•Bradyarrhythmia and Atrioventricular Conduction Delays [see Warnings and Precautions (5.3)]
-
•Liver Injury [see Warnings and Precautions (5.4) ]
-
•Fetal Risk [see Warnings and Precautions (5.5) ]
-
•Increased Blood Pressure [see Warnings and Precautions (5.6) ]
-
•Respiratory Effects [see Warnings and Precautions (5.7) ]
-
•Macular Edema [see Warnings and Precautions (5.8) ]
-
•Cutaneous Malignancies [see Warnings and Precautions (5.9)]
-
•Posterior Reversible Encephalopathy Syndrome [see Warnings and Precautions (5.10) ]
-
•Unintended Additive Immunosuppressive Effects from Prior Treatment with Immunosuppressive or Immune-Modulating Drugs [see Warnings and Precautions (5.11) ]
-
•Severe Increase in Multiple Sclerosis Disability after Stopping ZEPOSIA [see Warnings and Precautions (5.12) ]
-
•Immune System Effects after Stopping ZEPOSIA [see Warnings and Precautions (5.13) ]
7 Drug Interactions
Tables 5 and 6 include drugs with clinically important drug and vaccine interactions when administered concomitantly with ZEPOSIA and instructions for preventing or managing them.
|
Anti-Neoplastic, Immune-Modulating, or Non-Corticosteroid Immunosuppressive Therapies |
|
|
Clinical Impact: |
ZEPOSIA has not been studied in combination with anti-neoplastic, immune-modulating, or non-corticosteroid immunosuppressive therapies with the exception of cyclosporine, which had no pharmacokinetic interaction [see Clinical Pharmacology (12.3)]. |
|
Prevention or Management: |
Caution should be used during concomitant administration because of the risk of additive immune effects during such therapy and in the weeks following administration [see Warnings and Precautions (5.1)]. |
|
Anti-Arrhythmic Drugs, QT Prolonging Drugs, Drugs That May Decrease Heart Rate |
|
|
Clinical Impact: |
ZEPOSIA has not been studied in patients taking QT prolonging drugs. |
|
Prevention or Management: |
If treatment with ZEPOSIA is considered in patients on Class Ia or Class III anti-arrhythmic drugs, advice from a cardiologist should be sought [see Warnings and Precautions (5.3)]. |
|
Combination Beta Blocker and Calcium Channel Blocker |
|
|
Clinical Impact: |
The co-administration of ZEPOSIA with both a beta blocker and a calcium channel blocker has not been studied. However, there is a potential of additive effects on heart rate. |
|
Prevention or Management: |
Treatment with ZEPOSIA should generally not be initiated in patients who are concurrently treated with both a heart rate lowering calcium channel blocker (e.g., verapamil, diltiazem) and beta blocker [see Warnings and Precautions (5.3)]. If treatment initiation with ZEPOSIA is considered in patients on both a heart rate lowering calcium channel blocker and beta blocker, advice from a cardiologist should be sought. |
|
Vaccination |
|
|
Clinical Impact: |
During, and for up to three months after, discontinuation of treatment with ZEPOSIA, vaccinations may be less effective. The use of live attenuated vaccines may carry the risk of infection. |
|
Prevention or Management: |
Live attenuated vaccines should be avoided during ZEPOSIA treatment and for up to 3 months after discontinuation of treatment with ZEPOSIA [see Warnings and Precautions (5.1)]. |
|
Monoamine Oxidase (MAO) Inhibitors |
|
|
Clinical Impact: |
The effect of MAO inhibition on ozanimod and/or its metabolites has not been studied clinically. Potential effects on efficacy or safety with co-administration of MAO inhibitors because of altered exposures of ozanimod and/or its metabolites cannot be ruled out. |
|
Prevention or Management: |
Co-administration of ZEPOSIA with MAO inhibitors (e.g., selegiline, phenelzine, linezolid) is contraindicated. At least 14 days should elapse between discontinuation of ZEPOSIA and initiation of treatment with MAO inhibitors. |
|
Strong CYP2C8 Inhibitors |
|
|
Clinical Impact: |
Co-administration of ZEPOSIA with strong CYP2C8 inhibitors increases the exposure of the active metabolites of ozanimod [see Clinical Pharmacology (12.3)], which may increase the risk of ZEPOSIA adverse reactions. |
|
Prevention or Management: |
Co-administration of ZEPOSIA with strong CYP2C8 inhibitors (e.g., gemfibrozil) is not recommended. |
|
Strong CYP2C8 Inducers |
|
|
Clinical Impact: |
Co-administration of ZEPOSIA with strong CYP2C8 inducers (e.g., rifampin) reduces the exposure of the major active metabolites of ozanimod [see Clinical Pharmacology (12.3)], which may decrease the efficacy of ZEPOSIA. |
|
Prevention or Management: |
Co-administration of ZEPOSIA with strong CYP2C8 inducers should be avoided. |
12.3 Pharmacokinetics
The steady state exposure parameters of ozanimod and its major active metabolite, CC112273 are summarized in Table 7. Population pharmacokinetic analysis indicated no meaningful differences in these pharmacokinetic parameters in patients with relapsing MS or UC.
|
a Mean [coefficient of variation (CV%)] following ozanimod 0.92 mg once daily dose in relapsing MS patients, unless otherwise specified. b In healthy subjects. Cmax,ss = maximum observed plasma concentration at steady state, AUCtau,ss = area under the plasma concentration-time curve during a dosage interval at steady state. |
||
|
Parameters |
Ozanimod |
CC112273 |
|
Cmax,ss |
0.244 ng/mL (31.8%) |
6.98 ng/mL (42.7%) |
|
AUCtau,ss |
4.46 ng*h/mL (31.8%) |
143.77 ng*h/mL (39.2%) |
|
Dose Proportionality |
The Cmax and AUC increases proportionally over the ozanimod dose range from 0.46 mg to 0.92 mg. |
|
|
Time to Steady State |
102 hours (28.2%)b |
45 days (45%) |
|
Accumulation Ratio |
2.40 (21.1%) b |
16 (101%) |
8.6 Hepatic Impairment
In patients with mild (Child-Pugh class A) or moderate hepatic impairment (Child-Pugh class B), the exposures of ozanimod and its active metabolites are higher than those in healthy controls [see Clinical Pharmacology (12.3)], which may increase the risk of adverse reactions. Therefore, dosage adjustment in patients with mild or moderate hepatic impairment is required [see Dosage and Administration (2.3)].
The pharmacokinetics of ozanimod and its active metabolites were not evaluated in patients with severe hepatic impairment (Child-Pugh class C). Therefore, use of ZEPOSIA in patients with severe hepatic impairment is not recommended.
1 Indications and Usage
ZEPOSIA is indicated for the treatment of:
-
•relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults.
-
•moderately to severely active ulcerative colitis (UC) in adults.
14.1 Multiple Sclerosis
The efficacy of ZEPOSIA was demonstrated in 2 randomized, double-blind, double-dummy, parallel-group, active comparator-controlled clinical trials of similar design, in patients with relapsing forms of MS [MS Study 1 (NCT02294058) and MS Study 2 (NCT02047734)]. Patients in Study 1 were treated until the last enrolled patient completed 1 year of treatment. Patients in Study 2 were treated for 24 months. Both studies included patients who had experienced at least 1 relapse within the prior year, or 1 relapse within the prior 2 years with evidence of at least a gadolinium-enhancing (GdE) lesion in the prior year, and had an Expanded Disability Status Scale (EDSS) score from 0 to 5.0 at baseline. Patients with primary progressive MS were excluded.
Patients were randomized to receive either ZEPOSIA 0.92 mg given orally once daily, beginning with a dosage titration [see Dosage and Administration (2.2)], or interferon (IFN) beta-1a, the active comparator, 30 mcg given intramuscularly once weekly. Neurological evaluations were performed at baseline, every 3 months, and at the time of a suspected relapse. Brain MRI scans were performed at baseline, 6 months (Study 1), 1 year (Studies 1 and 2), and 2 years (Study 2).
The primary endpoint of both Study 1 and Study 2 was the annualized relapse rate (ARR) over the treatment period (Study 1) and 24 months (Study 2). Additional outcome measures included: 1) the number of new or enlarging MRI T2 hyperintense lesions over 12 and 24 months, 2) the number of MRI T1 Gadolinium-enhancing (Gd+) lesions at 12 and 24 months, and 3) the time to confirmed disability progression, defined as at least a 1-point increase from baseline EDSS confirmed after 3 months and after 6 months. Confirmed disability progression was evaluated in a pooled analysis of Studies 1 and 2.
In Study 1, a total of 895 patients were randomized to receive ZEPOSIA (n=447) or IFN beta-1a (n=448); of these patients, 94% who received ZEPOSIA and 92% who received IFN beta-1a completed the study. The mean age was 35.4 years, 99.8% were White, and 65% were female. The mean time since MS symptom-onset was 6.9 years, and the median EDSS score at baseline was 2.5; 31% had been treated with a non-steroid therapy for MS. At baseline, the mean number of relapses in the prior year was 1.3 and 48% of patients had one or more T1 Gd-enhancing lesions (mean 1.8) on their baseline MRI scan.
In Study 2, a total of 874 patients were randomized to receive ZEPOSIA (n=433) or IFN beta-1a (n=441); of these patients, 90% who received ZEPOSIA and 85% who received IFN beta-1a completed the study. The mean age was 35.6 years, 98% were White, and 68% were female. The mean time since MS symptom onset was 6.6 years, and the median EDSS score at baseline was 2.5; 29% of patients had been treated with a non-steroid therapy for MS. At baseline, the mean number of relapses in the prior year was 1.3 and 43% of patients had one or more T1 Gd-enhancing lesions (mean 1.7).
The ARR was statistically significantly lower in patients treated with ZEPOSIA 0.92 mg than in patients who received IFN beta-1a 30 mcg IM. The number of new or enlarging T2 lesions and the number of GdE lesions were statistically significantly lower in patients treated with ZEPOSIA 0.92 mg than in patients who received IFN beta-1a.
There was no statistically significant difference in the three-month and six-month confirmed disability progression between ZEPOSIA and IFN beta-1a-treated patients over 2 years.
The results for Study 1 and Study 2 are shown in Table 8.
|
a Through the treatment period (mean duration 13.6 months). b Over treatment period for Study 1 and over 24 months for Study 2. c Disability progression defined as 1-point increase in Expanded Disability Status Scale (EDSS) confirmed 3 months or 6 months later. d Prospective planned pooled analysis of Studies 1 and 2. e Not statistically significant. f Over 12 months for Study 1 and over 24 months for Study 2. g At 12 months for Study 1 and at 24 months for Study 2. |
||||
|
Endpoints |
Study 1 |
Study 2 |
||
|
ZEPOSIA
|
IFN beta-1a
|
ZEPOSIA
|
IFN beta-1a
|
|
|
Clinical Endpoints |
||||
|
Annualized Relapse Rate (Primary Endpoint) |
0.181a |
0.350 a |
0.172 |
0.276 |
|
Relative Reduction |
48% (p<0.0001) |
38% (p<0.0001) |
||
|
Percentage of patients without relapseb |
78% |
66% |
76% |
64% |
|
Proportion of Patients with 3-Month Confirmed Disability Progressionc,d |
7.6% ZEPOSIA vs. 7.8% IFN beta-1a |
|||
|
Hazard Ratio |
0.95 (p=0.77)e |
|||
|
MRI Endpoints |
||||
|
Mean number of new or enlarging T2 hyperintense lesions per MRIf |
1.47 |
2.84 |
1.84 |
3.18 |
|
Relative Reduction |
48% (p<0.0001) |
42% (p<0.0001) |
||
|
Mean number of T1 Gd-enhancing lesionsg |
0.16 |
0.43 |
0.18 |
0.37 |
|
Relative Reduction |
63% (p<0.0001) |
53% (p=0.0006) |
A similar effect of ZEPOSIA on the ARR compared to IFN beta-1a was observed in exploratory subgroups defined by sex, age, prior non-steroid therapy for MS, and baseline disease activity.
14.2 Ulcerative Colitis
The efficacy and safety of ZEPOSIA were evaluated in two multicenter, randomized, double-blind, placebo-controlled clinical studies [UC Study 1 (induction) and UC Study 2 (maintenance) (NCT02435992)] in adult patients with moderately to severely active ulcerative colitis.
5.7 Respiratory Effects
Dose-dependent reductions in absolute forced expiratory volume over 1 second (FEV1) were observed in MS patients treated with ZEPOSIA as early as 3 months after treatment initiation. In the MS pooled analyses of Study 1 and Study 2, the decline in absolute FEV1 from baseline in patients treated with ZEPOSIA compared to patients who received IFN beta-1a was 60 mL (95% CI: -100, -20) at 12 months. The mean difference in percent predicted FEV1 at 12 months between patients treated with ZEPOSIA and patients who received IFN beta-1a was 1.9% (95% CI: -2.9, -0.8). Dose-dependent reductions in forced vital capacity (FVC) (absolute value and %-predicted) were also seen at Month 3 in pooled analyses comparing patients treated with ZEPOSIA to patients who received IFN beta-1a [60 mL, 95% CI (-110, -10); 1.4%, 95% CI: (-2.6, -0.2)], though significant reductions were not seen at other timepoints. There is insufficient information to determine the reversibility of the decrease in FEV1 or FVC after drug discontinuation. One patient in MS Study 1 discontinued ZEPOSIA because of dyspnea.
In UC Study 1 the mean difference in decline in absolute FEV1 from baseline in patients treated with ZEPOSIA compared to patients who received placebo was 22 mL (95% CI: -84, 39) at 10 weeks. The mean difference in percent predicted normal (PPN) FEV1 at 10 weeks between patients treated with ZEPOSIA compared to those who received placebo was 0.8% (95% CI: -2.6, 1.0). The difference in reductions in FVC (absolute value and %-predicted) seen at Week 10 in UC Study 1, comparing patients who were treated with ZEPOSIA to those who received placebo was 44 mL, 95% CI (-114, 26); 0.5%, 95% CI (-2.3, 1.2), respectively. There is insufficient information to determine the reversibility of observed decreases in FEV1 or FVC after discontinuation of ZEPOSIA, or whether changes could be progressive with continued use.
Spirometric evaluation of respiratory function should be performed during therapy with ZEPOSIA, if clinically indicated.
12.1 Mechanism of Action
Ozanimod is a sphingosine 1-phosphate (S1P) receptor modulator that binds with high affinity to S1P receptors 1 and 5. Ozanimod blocks the capacity of lymphocytes to egress from lymph nodes, reducing the number of lymphocytes in peripheral blood. Ozanimod has minimal or no activity on S1P2, S1P3, and S1P4. The mechanism by which ozanimod exerts therapeutic effects in multiple sclerosis and ulcerative colitis is unknown but may involve the reduction of lymphocyte migration into the central nervous system and intestine.
5 Warnings and Precautions
-
•Infections: ZEPOSIA may increase the risk of infections. Obtain a complete blood count (CBC) before initiation of treatment. Monitor for infection during treatment and for 3 months after discontinuation. Do not start ZEPOSIA in patients with active infections. (5.1)
-
•Progressive Multifocal Leukoencephalopathy (PML): Withhold ZEPOSIA at the first sign or symptom suggestive of PML. (5.2)
-
•Bradyarrhythmia and Atrioventricular Conduction Delays: ZEPOSIA may result in transient decrease in heart rate; titration is required for treatment initiation. Check an electrocardiogram (ECG) to assess for preexisting cardiac conduction abnormalities before starting ZEPOSIA. Consider cardiology consultation for conduction abnormalities or concomitant use with other drugs that decrease heart rate. (2.1, 2.2, 5.3, 7)
-
•Liver Injury: Obtain liver enzyme results before initiation and periodically during treatment. Discontinue if there is evidence of liver injury without other cause. (5.4)
-
•Fetal Risk: Women of childbearing potential should use effective contraception during treatment and for 3 months after stopping ZEPOSIA. (5.5, 8.3)
-
•Increased Blood Pressure (BP): Monitor BP during treatment. (5.6)
-
•Respiratory Effects: May cause a decline in pulmonary function. Assess pulmonary function (e.g., spirometry) if clinically indicated. (5.7)
-
•Macular Edema: Increases the risk of macular edema. Obtain a baseline evaluation of the fundus, including the macula, near the start of treatment with ZEPOSIA. Conduct an evaluation of the fundus, including the macula, periodically while on therapy and any time there is a change in vision. Consider discontinuing ZEPOSIA if macular edema develops. Diabetes mellitus and uveitis increase the risk. (5.8)
-
•Cutaneous Malignancies: Skin examination prior to or shortly after the start of treatment and periodically thereafter is recommended. Suspicious skin lesions should be evaluated. (5.9)
5.9 Cutaneous Malignancies
The risk of cutaneous malignancies (including basal cell carcinoma (BCC), squamous cell carcinoma (SCC), and melanoma) is increased in patients treated with S1P receptor modulators. Cases of BCC, SCC, and melanoma have been reported in patients treated with ZEPOSIA; melanoma and BCC were reported in controlled trials [see Adverse Reactions (6.1) ] . Kaposi’s sarcoma and Merkel cell carcinoma have also been reported in patients treated with S1P receptor modulators in the postmarketing setting.
Skin examinations are recommended prior to or shortly after the start of treatment and periodically thereafter for all patients, particularly those with risk factors for skin cancer. Providers and patients are advised to monitor for suspicious skin lesions. If a suspicious skin lesion is observed, it should be promptly evaluated. As usual for patients with increased risk for skin cancer, exposure to sunlight and ultraviolet light should be limited by wearing protective clothing and using a sunscreen with a high protection factor. Concomitant phototherapy with UV-B radiation or PUVA photochemotherapy is not recommended in patients taking ZEPOSIA.
2 Dosage and Administration
-
•Assessments are required prior to initiating ZEPOSIA. (2.1)
-
•Titration is required for treatment initiation. (2.2)
-
•The recommended maintenance dosage is 0.92 mg orally once daily. (2.2)
-
•The recommended maintenance dosage in patients with mild or moderate chronic hepatic impairment (Child-Pugh class A or B) is 0.92 mg once every other day. (2.3)
-
•If a dose is missed within the first 2 weeks of treatment, reinitiate with the titration regimen. If a dose is missed after the first 2 weeks of treatment, continue treatment as planned. (2.4)
Package/label Display Panel
Rx only
NDC 59572-890-28
ZEPOSIA®
(ozanimod) capsules
STARTER KIT
STEP 1: (Days 1-7)
Starting at Day 1, complete the 7-Day Starter Pack
Days 1-4: Take one 0.23 mg capsule orally once daily
Days 5-7: Take one 0.46 mg capsule orally once daily
STEP 2: (Day 8 and thereafter)
After completing the 7-Day Starter Pack, begin
taking one 0.92 mg capsule a day or as directed
from the 21-count bottle.
This Starter Kit contains 28 capsules for titration
over 7 days up to prescribed maintenance dose
of one 0.92 mg capsule taken once daily or as
directed. The contents of the Starter Kit, which
are not to be sold separately, are as follows:
Four 0.23 mg capsules
Three 0.46 mg capsules
Twenty One 0.92 mg capsules
28 CAPSULES
Dispense the accompanying Medication Guide to each patient.
Bristol Myers Squibb
3 Dosage Forms and Strengths
Capsules:
-
•0.23 mg ozanimod: light grey opaque body/light grey opaque cap imprinted with black ink "OZA" on the cap and "0.23 mg" on the body
-
•0.46 mg ozanimod: light grey opaque body/orange opaque cap imprinted with black ink "OZA" on the cap and "0.46 mg" on the body
-
•0.92 mg ozanimod: orange opaque body/orange opaque cap imprinted with black ink "OZA" on the cap and "0.92 mg" on the body
5.6 Increased Blood Pressure
In MS Study 1 and Study 2, patients treated with ZEPOSIA had an average increase of approximately 1 to 2 mm Hg in systolic pressure over patients who received IFN beta-1a, and no effect on diastolic pressure. The increase in systolic pressure was first detected after approximately 3 months of treatment and persisted throughout treatment. Hypertension was reported as an adverse reaction in 3.9% of patients treated with ZEPOSIA 0.92 mg and in 2.1% of patients who received IFN beta-1a. Two patients treated with ZEPOSIA in MS Study 1 and one patient treated with interferon (IFN) beta-1a in Study 2 experienced a hypertensive crisis that was not clearly influenced by a concomitant medication.
The mean increase in systolic blood pressure (SBP) and diastolic blood pressure (DBP) in UC patients treated with ZEPOSIA is similar to patients with MS. In UC Study 1 and Study 3, the average increase from baseline in SBP was 3.7 mm Hg in patients treated with ZEPOSIA and 2.3 mm Hg in patients treated with placebo. In UC Study 2, the average increase from baseline in SBP was 5.1 mm Hg in patients treated with ZEPOSIA and 1.5 mm Hg in patients treated with placebo. There was no effect on DBP.
Hypertension was reported as an adverse reaction in 1.2% of patients treated with ZEPOSIA 0.92 mg and none in patients treated with placebo in UC Study 1 and Study 3, and in 2.2% and 2.2% of patients in UC Study 2, respectively. Hypertensive crisis was reported in two patients receiving ZEPOSIA and one patient receiving placebo.
Blood pressure should be monitored during treatment with ZEPOSIA and managed appropriately.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of ZEPOSIA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Hepatobiliary Disorders: Liver injury [see Warnings and Precautions (5.4)]
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Principal Display Panel Kit Carton
Rx only
NDC 59572-890-91
ZEPOSIA®
(ozanimod) capsules
STARTER KIT
STEP 1: (Days 1-7)
Starting at Day 1, complete the 7-Day Starter Pack
Days 1-4: Take one 0.23 mg capsule orally once daily
Days 5-7: Take one 0.46 mg capsule orally once daily
STEP 2: (Day 8 and thereafter)
After completing the 7-Day Starter Pack, begin taking
one 0.92 mg capsule once daily or as directed from the 30-count bottle.
This Starter Kit contains 37 capsules for titration
over 7 days up to prescribed maintenance dose
of one 0.92 mg capsule taken once daily or as directed. The
contents of the Starter Kit, which are not to be
sold separately, are as follows:
Four 0.23 mg capsules
Three 0.46 mg capsules
Thirty 0.92 mg capsules
37 CAPSULES
Dispense the accompanying Medication Guide to each patient.
Bristol Myers Squibb
2.1 Assessments Prior to First Dose of Zeposia
Before initiation of treatment with ZEPOSIA, assess the following:
5.2 Progressive Multifocal Leukoencephalopathy
Progressive multifocal leukoencephalopathy (PML) is an opportunistic viral infection of the brain caused by the JC virus (JCV) that typically occurs in patients who are immunocompromised, and that usually leads to death or severe disability. Typical symptoms associated with PML are diverse, progress over days to weeks, and include progressive weakness on one side of the body or clumsiness of limbs, disturbance of vision, and changes in thinking, memory, and orientation leading to confusion and personality changes.
PML has been reported in patients treated with S1P receptor modulators, including ZEPOSIA, and other multiple sclerosis (MS) and UC therapies and has been associated with some risk factors (e.g., immunocompromised patients, polytherapy with immunosuppressants, duration of use). Based on data from patients with MS, longer treatment duration increases the risk of PML in patients treated with S1P receptor modulators, and the majority of PML cases have occurred in patients treated with S1P receptor modulators for at least 18 months. Physicians should be vigilant for clinical symptoms or MRI findings that may be suggestive of PML. MRI findings may be apparent before clinical signs or symptoms. If PML is suspected, treatment with ZEPOSIA should be suspended until PML has been excluded by an appropriate diagnostic evaluation.
If PML is confirmed, treatment with ZEPOSIA should be discontinued.
Immune reconstitution inflammatory syndrome (IRIS) has been reported in MS patients treated with S1P receptor modulators who developed PML and subsequently discontinued treatment. IRIS presents as a clinical decline in the patient’s condition that may be rapid, can lead to serious neurological complications or death, and is often associated with characteristic changes on MRI. The time to onset of IRIS in patients with PML was generally within a few months after S1P receptor modulator discontinuation. Monitoring for development of IRIS and appropriate treatment of the associated inflammation should be undertaken.
5.10 Posterior Reversible Encephalopathy Syndrome
Rare cases of posterior reversible encephalopathy syndrome (PRES) have been reported in patients receiving a S1P receptor modulator. In MS controlled clinical trials with ZEPOSIA, one case of PRES was reported. Should a ZEPOSIA-treated patient develop any unexpected neurological or psychiatric symptoms/signs (e.g., cognitive deficits, behavioral changes, cortical visual disturbances, or any other neurological cortical symptoms/signs), any symptom/sign suggestive of an increase of intracranial pressure, or accelerated neurological deterioration, the physician should promptly schedule a complete physical and neurological examination and should consider an MRI. Symptoms of PRES are usually reversible but may evolve into ischemic stroke or cerebral hemorrhage. Delay in diagnosis and treatment may lead to permanent neurological sequelae. If PRES is suspected, treatment with ZEPOSIA should be discontinued.
5.13 Immune System Effects After Stopping Zeposia
After discontinuing ZEPOSIA, the median time for peripheral blood lymphocytes to return to the normal range was approximately 30 days, with approximately 80% to 90% of patients in the normal range within 3 months [see Clinical Pharmacology (12.2)]. Use of immunosuppressants within this period may lead to an additive effect on the immune system, and therefore caution should be applied when initiating other drugs 4 weeks after the last dose of ZEPOSIA [see Drug Interactions (7)].
Principal Display Panel 0.92 Mg Capsule Bottle Label
NDC 59572-820-30
ZEPOSIA®
(ozanimod) capsules
0.92 mg
Dispense the accompanying
Medication Guide to each patient.
Rx only
30 Capsules
Bristol-Myers Squibb
2.4 Reinitiation of Zeposia After Treatment Interruption
If a dose of ZEPOSIA is missed during the first 2 weeks of treatment, reinitiate treatment using the titration regimen [see Dosage and Administration (2.2)].
If a dose of ZEPOSIA is missed after the first 2 weeks of treatment, continue with the treatment as planned.
2.3 Recommended Dosage in Patients With Hepatic Impairment
In patients with mild or moderate hepatic impairment (Child-Pugh class A or B), initiate ZEPOSIA with a 7-day titration, as shown in Table 1. After initial titration, the recommended dosage of ZEPOSIA in these patients is 0.92 mg taken orally once every other day, starting on Day 8 [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
5.3 Bradyarrhythmia and Atrioventricular Conduction Delays
Since initiation of ZEPOSIA may result in a transient decrease in heart rate and atrioventricular conduction delays, an up-titration scheme should be used to reach the maintenance dosage of ZEPOSIA [see Dosage and Administration (2.2) and Clinical Pharmacology (12.2)].
ZEPOSIA was not studied in patients who had:
-
•A myocardial infarction, unstable angina, stroke, TIA, or decompensated heart failure requiring hospitalization within the last 6 months
-
•New York Heart Association Class III / IV heart failure
-
•Cardiac conduction or rhythm disorders, including sick sinus syndrome, significant QT prolongation (QTcF > 450 msec in males, > 470 msec in females), risk factors for QT prolongation, or other conduction abnormalities or cardiac condition that in the opinion of the treating investigator could jeopardize the patient's health
-
•Other pre-existing stable cardiac conditions without clearance from a cardiologist
-
•Severe untreated sleep apnea
-
•A resting heart rate less than 55 beats per minute (bpm) at baseline
Principal Display Panel 7 Capsule Starter Pack Kit Carton
NDC 59572-810-07
Rx only
ZEPOSIA®
(ozanimod) capsules
7-DAY
STARTER PACK
This pack contains 7 capsules for dosing
over 7 days. The contents of this pack are
as follows:
Four 0.23 mg capsules
Three 0.46 mg capsules
7 CAPSULES
Dispense the accompanying Medication Guide to each patient.
Bristol-Myers Squibb
2.2 Recommended Dosage for Multiple Sclerosis and Ulcerative Colitis
Initiate ZEPOSIA with a 7-day titration, as shown in Table 1 [see Warnings and Precautions (5.3) ]. After initial titration, the recommended dosage of ZEPOSIA is 0.92 mg taken orally once daily starting on Day 8.
Swallow ZEPOSIA capsules whole, with or without food [see Clinical Pharmacology (12.3)].
|
Days 1-4 |
0.23 mg once daily |
|
Days 5-7 |
0.46 mg once daily |
|
Day 8 and thereafter |
0.92 mg once daily* |
*Patients with mild to moderate hepatic impairment (Child-Pugh class A or B) should take 0.92 mg once every other day [see Recommended Dosage in Patients with Hepatic Impairment (2.3) .]
5.12 Severe Increase in Multiple Sclerosis Disability After Stopping Zeposia
In MS, severe exacerbation of disease, including disease rebound, has been rarely reported after discontinuation of a S1P receptor modulator. The possibility of severe exacerbation of disease should be considered after stopping ZEPOSIA treatment. Patients should be observed for a severe increase in disability upon ZEPOSIA discontinuation and appropriate treatment should be instituted, as required.
After stopping ZEPOSIA in the setting of PML, monitor for development of immune reconstitution inflammatory syndrome (PML-IRIS) [see Warnings and Precautions (5.2) ].
5.11 Unintended Additive Immunosuppressive Effects From Prior Treatment With Immunosuppressive Or Immune Modulating Drugs
When switching from drugs with prolonged immune effects, the half-life and mode of action of these drugs must be considered to avoid unintended additive immunosuppressive effects while at the same time minimizing risk of disease reactivation, when initiating ZEPOSIA.
Initiating treatment with ZEPOSIA after treatment with alemtuzumab is not recommended [see Drug Interactions (7)].
Structured Label Content
Section 42229-5 (42229-5)
Cardiac Evaluation
Obtain an electrocardiogram (ECG) to determine whether preexisting conduction abnormalities are present. In patients with certain preexisting conditions, advice from a cardiologist should be sought [see Warnings and Precautions (5.3)].
Complete Blood Count
Obtain a recent (i.e., within the last 6 months or after discontinuation of prior MS or UC therapy) complete blood count (CBC), including lymphocyte count [see Warnings and Precautions (5.1)].
Liver Function Tests
Obtain recent (i.e., within the last 6 months) transaminase and bilirubin levels [see Warnings and Precautions (5.4)].
Ophthalmic Assessment
Obtain a baseline evaluation of the fundus, including the macula, near the start of treatment with ZEPOSIA [see Warnings and Precautions (5.8)].
Skin Examination
Obtain a baseline skin examination prior to or shortly after initiation of ZEPOSIA. If a suspicious skin lesion is observed, it should be promptly evaluated [see Warnings and Precautions (5.9)].
Current or Prior Medications
-
•If patients are taking anti-neoplastic, non-corticosteroid immunosuppressive, or immune-modulating therapies, or if there is a history of prior use of these drugs, consider possible unintended additive immunosuppressive effects before initiating treatment with ZEPOSIA [see Warnings and Precautions (5.1) and Drug Interactions (7)].
-
•Determine if patients are taking drugs that could slow heart rate or atrioventricular conduction [see Warnings and Precautions (5.3) and Drug Interactions (7)].
Vaccinations
Patients without a healthcare professional-confirmed history of chickenpox or without documentation of a full course of vaccination against varicella zoster virus (VZV) should be tested for antibodies to VZV before initiating ZEPOSIA; VZV vaccination of antibody-negative patients is recommended prior to commencing treatment with ZEPOSIA [see Warnings and Precautions (5.1) and Drug Interactions (7)].
If live attenuated vaccine immunizations are required, administer at least 1 month prior to initiation of ZEPOSIA.
Section 42231-1 (42231-1)
|
MEDICATION GUIDE
|
|||
|
Read this Medication Guide before you start taking ZEPOSIA and each time you get a refill. There may be new information. This Medication Guide does not take the place of talking with your healthcare provider about your medical condition or treatment. |
|||
|
What is the most important information I should know about ZEPOSIA?
|
|||
|
|
||
|
Your healthcare provider may delay starting or may stop your ZEPOSIA treatment if you have an infection. |
|||
|
|||
|
|
||
|
|||
|
|
||
|
Follow directions from your healthcare provider when starting ZEPOSIA and when you miss a dose. See "How should I take ZEPOSIA?". |
|||
|
See "What are possible side effects of ZEPOSIA?" for more information about side effects. |
|||
|
What is ZEPOSIA?
It is not known if ZEPOSIA is safe and effective in children. |
|||
|
Do not take ZEPOSIA if you:
Talk to your healthcare provider before taking ZEPOSIA if you have any of these conditions or do not know if you have any of these conditions. |
|||
|
Before taking ZEPOSIA, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take or have recently taken, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Using ZEPOSIA with other medicines can cause serious side effects. Especially tell your healthcare provider if you take or have taken:
You should not receive live vaccines during treatment with ZEPOSIA, for at least 1 month before taking ZEPOSIA and for 3 months after you stop taking ZEPOSIA. Vaccines may not work as well when given during treatment with ZEPOSIA. |
|||
|
How should I take ZEPOSIA?
|
|||
|
Days 1-4 |
Take 0.23 mg (capsule in light grey color) 1 time a day |
||
|
Days 5-7 |
Take 0.46 mg (capsule in half-light grey and half-orange color) 1 time a day |
||
|
Days 8 and thereafter |
Take 0.92 mg (capsule in orange color) 1 time a day, or as directed by your healthcare provider |
||
|
|||
|
What are the possible side effects of ZEPOSIA?
|
|||
|
|
||
|
|||
|
|
||
|
|||
|
|
||
|
|||
|
The most common side effects of ZEPOSIA can include: |
|||
|
|
|
|
|
These are not all of the possible side effects of ZEPOSIA. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
|
How should I store ZEPOSIA?
Keep ZEPOSIA and all medicines out of the reach of children. |
|||
|
General information about the safe and effective use of ZEPOSIA.
|
|||
|
What are the ingredients in ZEPOSIA?
|
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Revised: 8/2024
ZEPMG.009
Section 43683-2 (43683-2)
16.2 Storage
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
8.1 Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to ZEPOSIA during pregnancy. Healthcare providers are encouraged to register patients on-line, or pregnant women may register themselves at https://www.zeposiapregnancyregistry.com/or by calling 1-877-301-9314. Currently this registry is enrolling women with MS. Information regarding registration of pregnant women with UC will be made available in the future.
11 Description (11 DESCRIPTION)
ZEPOSIA contains ozanimod, a sphingosine 1-phosphate receptor modulator and is supplied as ozanimod hydrochloride (HCl).
The chemical name of ozanimod HCl is 5-(3-{(1S)-1-[(2-hydroxyethyl)amino]-2,3-dihydro-1H-inden-4-yl}-1,2,4-oxadiazol-5-yl)-2-[(propan-2-yl)oxy]benzonitrile, monohydrochloride.
Ozanimod HCl is a white to off-white solid that is freely soluble in water and alcohol with a molecular weight of 440.92 g/mol.
The chemical structure is:
ZEPOSIA capsules are provided as hard gelatin capsules for oral administration, containing 0.23, 0.46, or 0.92 mg of ozanimod (equivalent to 0.25, 0.5, and 1 mg ozanimod HCl, respectively). ZEPOSIA capsules consist of the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, and microcrystalline cellulose. The capsule shell, imprinted with black ink, contains the following inactive ingredients: black iron oxide, gelatin, red iron oxide, titanium dioxide, and yellow iron oxide.
5.5 Fetal Risk
There are no adequate and well-controlled studies in pregnant women. Based on animal studies, ZEPOSIA may cause fetal harm [see Use in Specific Populations (8.1)]. Because it takes approximately 3 months to eliminate ZEPOSIA from the body, women of childbearing potential should use effective contraception to avoid pregnancy during treatment and for 3 months after stopping ZEPOSIA [see Use in Specific Populations (8.3)].
5.4 Liver Injury
Clinically significant liver injury, including acute liver failure requiring transplant, has occurred in patients treated with ZEPOSIA in the postmarketing setting. Signs of liver injury, including elevated serum hepatic enzymes and elevated total bilirubin, have occurred as early as ten days after the first dose.
In MS Study 1 and Study 2, elevations of ALT to 5-fold the upper limit of normal (ULN) or greater occurred in 1.6% of patients treated with ZEPOSIA 0.92 mg and 1.3% of patients who received IFN beta-1a. Elevations of 3-fold the ULN or greater occurred in 5.5% of patients treated with ZEPOSIA and 3.1% of patients who received IFN beta-1a. The median time to an elevation of 3-fold the ULN was 6 months. The majority (79%) of patients continued treatment with ZEPOSIA with values returning to less than 3-fold the ULN within approximately 2-4 weeks. ZEPOSIA was discontinued for a confirmed elevation greater than 5-fold the ULN. Overall, the discontinuation rate because of elevations in hepatic enzymes was 1.1% of patients with MS treated with ZEPOSIA 0.92 mg and 0.8% of patients who received IFN beta-1a.
In UC Study 1, elevations of ALT to 5-fold the ULN or greater occurred in 0.9% of patients treated with ZEPOSIA 0.92 mg and 0.5% of patients who received placebo, and in UC Study 2 elevations occurred in 0.9% of patients and no patients, respectively. In UC Study 1, elevations of ALT to 3-fold the ULN or greater occurred in 2.6% of UC patients treated with ZEPOSIA 0.92 mg and 0.5% of patients who received placebo, and in UC Study 2 elevations occurred in 2.3% of patients and no patients, respectively. In controlled and uncontrolled UC studies, the majority (96%) of patients with ALT greater than 3-fold the ULN continued treatment with ZEPOSIA with values returning to less than 3-fold the ULN within approximately 2 to 4 weeks. Overall, the discontinuation rate because of elevations in hepatic enzymes was 0.4% in patients treated with ZEPOSIA 0.92 mg, and none in patients who received placebo in the controlled UC studies.
Individuals with an AST or ALT greater than 1.5-fold ULN were excluded from MS Study 1 and Study 2 and greater than 2 times the ULN for UC Study 1 and Study 3. There are no data to establish that patients with preexisting liver disease are at increased risk to develop elevated liver function test values when taking ZEPOSIA. Dosage adjustment in patients with mild or moderate hepatic impairment (Child-Pugh class A or B) is required [see Dosage and Administration (2.3)], and use of ZEPOSIA in patients with severe hepatic impairment (Child-Pugh class C) is not recommended [see Use in Specific Populations (8.6)].
Obtain transaminase and bilirubin levels, if not recently available (i.e., within 6 months), before initiation of ZEPOSIA. Obtain transaminase levels and total bilirubin levels periodically during treatment and until two months after ZEPOSIA discontinuation.
Patients should be monitored for signs and symptoms of any hepatic injury. Patients who develop symptoms suggestive of hepatic dysfunction, such as unexplained nausea, vomiting, abdominal pain, fatigue, anorexia, or jaundice and/or dark urine, should have hepatic enzymes promptly checked, and ZEPOSIA should be interrupted. Treatment should not be resumed if a plausible alternative etiology for the signs and symptoms cannot be established, because these patients are at risk for severe drug-induced liver injury.
16.1 How Supplied
ZEPOSIA is available as capsules in the following dosage strengths:
-
•0.23 mg ozanimod: light grey opaque body/light grey opaque cap imprinted with black ink "OZA" on the cap and "0.23 mg" on the body
-
•0.46 mg ozanimod: light grey opaque body/orange opaque cap imprinted with black ink "OZA" on the cap and "0.46 mg" on the body
-
•0.92 mg ozanimod: orange opaque body/orange opaque cap imprinted with black ink "OZA" on the cap and "0.92 mg" on the body
Capsules are supplied in the following strengths and package configurations:
|
Package configuration |
Tablet strength |
NDC number |
|
Bottles of 30 |
0.92 mg ozanimod |
59572-820-30 |
|
7-Day Starter Pack |
7-capsule starter pack containing: (4) 0.23 mg ozanimod capsules and (3) 0.46 ozanimod mg capsules |
59572-810-07 |
|
Starter Kit |
37-capsule starter kit including: |
59572-890-91 |
|
Starter Kit (7-Day Starter Pack and 0.92 mg 21-count Bottle) |
28-capsule starter kit including: |
59572-890-28 |
5.8 Macular Edema
S1P receptor modulators, including ZEPOSIA, have been associated with an increased risk of macular edema. Obtain a baseline evaluation of the fundus, including the macula, near the start of treatment with ZEPOSIA. Perform an examination of the fundus, including the macula, periodically while on therapy and any time there is a change in vision.
In MS Study 1 and Study 2, macular edema was observed in 0.3% of patients treated with ZEPOSIA and in 0.3% of patients who received IFN beta-1a. Macular edema was reported in a total of 1 (0.2%) patient in UC Study 1 and Study 3, and in 1 (0.4%) patient in UC Study 2 treated with ZEPOSIA, and in no patients who received placebo.
Continuation of ZEPOSIA therapy in patients with macular edema has not been evaluated. Macular edema over an extended period of time (i.e., 6 months) can lead to permanent visual loss. Consider discontinuing ZEPOSIA if macular edema develops; this decision should include an assessment of the potential benefits and risks for the individual patient. The risk of recurrence after rechallenge has not been evaluated.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of ZEPOSIA did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. No clinically significant differences in the pharmacokinetics of ozanimod and CC112273 were observed based on age [see Clinical Pharmacology (12.3)]. Monitor elderly patients for cardiac and hepatic adverse reactions, because of the greater frequency of reduced cardiac and hepatic function in the elderly population.
4 Contraindications (4 CONTRAINDICATIONS)
ZEPOSIA is contraindicated in patients who:
-
•In the last 6 months, have experienced a myocardial infarction, unstable angina, stroke, transient ischemic attack (TIA), decompensated heart failure requiring hospitalization, or Class III or IV heart failure [see Warnings and Precautions (5.3) ]
-
•Have the presence of Mobitz type II second-degree or third degree atrioventricular (AV) block, sick sinus syndrome, or sino-atrial block, unless the patient has a functioning pacemaker [see Warnings and Precautions (5.3)]
-
•Have severe untreated sleep apnea [see Warnings and Precautions (5.3)]
-
•Are taking a monoamine oxidase (MAO) inhibitor [see Drug Interactions (7)]
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described elsewhere in the labeling:
-
•Infections [see Warnings and Precautions (5.1)]
-
•Progressive Multifocal Leukoencephalopathy [see Warnings and Precautions (5.2)]
-
•Bradyarrhythmia and Atrioventricular Conduction Delays [see Warnings and Precautions (5.3)]
-
•Liver Injury [see Warnings and Precautions (5.4) ]
-
•Fetal Risk [see Warnings and Precautions (5.5) ]
-
•Increased Blood Pressure [see Warnings and Precautions (5.6) ]
-
•Respiratory Effects [see Warnings and Precautions (5.7) ]
-
•Macular Edema [see Warnings and Precautions (5.8) ]
-
•Cutaneous Malignancies [see Warnings and Precautions (5.9)]
-
•Posterior Reversible Encephalopathy Syndrome [see Warnings and Precautions (5.10) ]
-
•Unintended Additive Immunosuppressive Effects from Prior Treatment with Immunosuppressive or Immune-Modulating Drugs [see Warnings and Precautions (5.11) ]
-
•Severe Increase in Multiple Sclerosis Disability after Stopping ZEPOSIA [see Warnings and Precautions (5.12) ]
-
•Immune System Effects after Stopping ZEPOSIA [see Warnings and Precautions (5.13) ]
7 Drug Interactions (7 DRUG INTERACTIONS)
Tables 5 and 6 include drugs with clinically important drug and vaccine interactions when administered concomitantly with ZEPOSIA and instructions for preventing or managing them.
|
Anti-Neoplastic, Immune-Modulating, or Non-Corticosteroid Immunosuppressive Therapies |
|
|
Clinical Impact: |
ZEPOSIA has not been studied in combination with anti-neoplastic, immune-modulating, or non-corticosteroid immunosuppressive therapies with the exception of cyclosporine, which had no pharmacokinetic interaction [see Clinical Pharmacology (12.3)]. |
|
Prevention or Management: |
Caution should be used during concomitant administration because of the risk of additive immune effects during such therapy and in the weeks following administration [see Warnings and Precautions (5.1)]. |
|
Anti-Arrhythmic Drugs, QT Prolonging Drugs, Drugs That May Decrease Heart Rate |
|
|
Clinical Impact: |
ZEPOSIA has not been studied in patients taking QT prolonging drugs. |
|
Prevention or Management: |
If treatment with ZEPOSIA is considered in patients on Class Ia or Class III anti-arrhythmic drugs, advice from a cardiologist should be sought [see Warnings and Precautions (5.3)]. |
|
Combination Beta Blocker and Calcium Channel Blocker |
|
|
Clinical Impact: |
The co-administration of ZEPOSIA with both a beta blocker and a calcium channel blocker has not been studied. However, there is a potential of additive effects on heart rate. |
|
Prevention or Management: |
Treatment with ZEPOSIA should generally not be initiated in patients who are concurrently treated with both a heart rate lowering calcium channel blocker (e.g., verapamil, diltiazem) and beta blocker [see Warnings and Precautions (5.3)]. If treatment initiation with ZEPOSIA is considered in patients on both a heart rate lowering calcium channel blocker and beta blocker, advice from a cardiologist should be sought. |
|
Vaccination |
|
|
Clinical Impact: |
During, and for up to three months after, discontinuation of treatment with ZEPOSIA, vaccinations may be less effective. The use of live attenuated vaccines may carry the risk of infection. |
|
Prevention or Management: |
Live attenuated vaccines should be avoided during ZEPOSIA treatment and for up to 3 months after discontinuation of treatment with ZEPOSIA [see Warnings and Precautions (5.1)]. |
|
Monoamine Oxidase (MAO) Inhibitors |
|
|
Clinical Impact: |
The effect of MAO inhibition on ozanimod and/or its metabolites has not been studied clinically. Potential effects on efficacy or safety with co-administration of MAO inhibitors because of altered exposures of ozanimod and/or its metabolites cannot be ruled out. |
|
Prevention or Management: |
Co-administration of ZEPOSIA with MAO inhibitors (e.g., selegiline, phenelzine, linezolid) is contraindicated. At least 14 days should elapse between discontinuation of ZEPOSIA and initiation of treatment with MAO inhibitors. |
|
Strong CYP2C8 Inhibitors |
|
|
Clinical Impact: |
Co-administration of ZEPOSIA with strong CYP2C8 inhibitors increases the exposure of the active metabolites of ozanimod [see Clinical Pharmacology (12.3)], which may increase the risk of ZEPOSIA adverse reactions. |
|
Prevention or Management: |
Co-administration of ZEPOSIA with strong CYP2C8 inhibitors (e.g., gemfibrozil) is not recommended. |
|
Strong CYP2C8 Inducers |
|
|
Clinical Impact: |
Co-administration of ZEPOSIA with strong CYP2C8 inducers (e.g., rifampin) reduces the exposure of the major active metabolites of ozanimod [see Clinical Pharmacology (12.3)], which may decrease the efficacy of ZEPOSIA. |
|
Prevention or Management: |
Co-administration of ZEPOSIA with strong CYP2C8 inducers should be avoided. |
12.3 Pharmacokinetics
The steady state exposure parameters of ozanimod and its major active metabolite, CC112273 are summarized in Table 7. Population pharmacokinetic analysis indicated no meaningful differences in these pharmacokinetic parameters in patients with relapsing MS or UC.
|
a Mean [coefficient of variation (CV%)] following ozanimod 0.92 mg once daily dose in relapsing MS patients, unless otherwise specified. b In healthy subjects. Cmax,ss = maximum observed plasma concentration at steady state, AUCtau,ss = area under the plasma concentration-time curve during a dosage interval at steady state. |
||
|
Parameters |
Ozanimod |
CC112273 |
|
Cmax,ss |
0.244 ng/mL (31.8%) |
6.98 ng/mL (42.7%) |
|
AUCtau,ss |
4.46 ng*h/mL (31.8%) |
143.77 ng*h/mL (39.2%) |
|
Dose Proportionality |
The Cmax and AUC increases proportionally over the ozanimod dose range from 0.46 mg to 0.92 mg. |
|
|
Time to Steady State |
102 hours (28.2%)b |
45 days (45%) |
|
Accumulation Ratio |
2.40 (21.1%) b |
16 (101%) |
8.6 Hepatic Impairment
In patients with mild (Child-Pugh class A) or moderate hepatic impairment (Child-Pugh class B), the exposures of ozanimod and its active metabolites are higher than those in healthy controls [see Clinical Pharmacology (12.3)], which may increase the risk of adverse reactions. Therefore, dosage adjustment in patients with mild or moderate hepatic impairment is required [see Dosage and Administration (2.3)].
The pharmacokinetics of ozanimod and its active metabolites were not evaluated in patients with severe hepatic impairment (Child-Pugh class C). Therefore, use of ZEPOSIA in patients with severe hepatic impairment is not recommended.
1 Indications and Usage (1 INDICATIONS AND USAGE)
ZEPOSIA is indicated for the treatment of:
-
•relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults.
-
•moderately to severely active ulcerative colitis (UC) in adults.
14.1 Multiple Sclerosis
The efficacy of ZEPOSIA was demonstrated in 2 randomized, double-blind, double-dummy, parallel-group, active comparator-controlled clinical trials of similar design, in patients with relapsing forms of MS [MS Study 1 (NCT02294058) and MS Study 2 (NCT02047734)]. Patients in Study 1 were treated until the last enrolled patient completed 1 year of treatment. Patients in Study 2 were treated for 24 months. Both studies included patients who had experienced at least 1 relapse within the prior year, or 1 relapse within the prior 2 years with evidence of at least a gadolinium-enhancing (GdE) lesion in the prior year, and had an Expanded Disability Status Scale (EDSS) score from 0 to 5.0 at baseline. Patients with primary progressive MS were excluded.
Patients were randomized to receive either ZEPOSIA 0.92 mg given orally once daily, beginning with a dosage titration [see Dosage and Administration (2.2)], or interferon (IFN) beta-1a, the active comparator, 30 mcg given intramuscularly once weekly. Neurological evaluations were performed at baseline, every 3 months, and at the time of a suspected relapse. Brain MRI scans were performed at baseline, 6 months (Study 1), 1 year (Studies 1 and 2), and 2 years (Study 2).
The primary endpoint of both Study 1 and Study 2 was the annualized relapse rate (ARR) over the treatment period (Study 1) and 24 months (Study 2). Additional outcome measures included: 1) the number of new or enlarging MRI T2 hyperintense lesions over 12 and 24 months, 2) the number of MRI T1 Gadolinium-enhancing (Gd+) lesions at 12 and 24 months, and 3) the time to confirmed disability progression, defined as at least a 1-point increase from baseline EDSS confirmed after 3 months and after 6 months. Confirmed disability progression was evaluated in a pooled analysis of Studies 1 and 2.
In Study 1, a total of 895 patients were randomized to receive ZEPOSIA (n=447) or IFN beta-1a (n=448); of these patients, 94% who received ZEPOSIA and 92% who received IFN beta-1a completed the study. The mean age was 35.4 years, 99.8% were White, and 65% were female. The mean time since MS symptom-onset was 6.9 years, and the median EDSS score at baseline was 2.5; 31% had been treated with a non-steroid therapy for MS. At baseline, the mean number of relapses in the prior year was 1.3 and 48% of patients had one or more T1 Gd-enhancing lesions (mean 1.8) on their baseline MRI scan.
In Study 2, a total of 874 patients were randomized to receive ZEPOSIA (n=433) or IFN beta-1a (n=441); of these patients, 90% who received ZEPOSIA and 85% who received IFN beta-1a completed the study. The mean age was 35.6 years, 98% were White, and 68% were female. The mean time since MS symptom onset was 6.6 years, and the median EDSS score at baseline was 2.5; 29% of patients had been treated with a non-steroid therapy for MS. At baseline, the mean number of relapses in the prior year was 1.3 and 43% of patients had one or more T1 Gd-enhancing lesions (mean 1.7).
The ARR was statistically significantly lower in patients treated with ZEPOSIA 0.92 mg than in patients who received IFN beta-1a 30 mcg IM. The number of new or enlarging T2 lesions and the number of GdE lesions were statistically significantly lower in patients treated with ZEPOSIA 0.92 mg than in patients who received IFN beta-1a.
There was no statistically significant difference in the three-month and six-month confirmed disability progression between ZEPOSIA and IFN beta-1a-treated patients over 2 years.
The results for Study 1 and Study 2 are shown in Table 8.
|
a Through the treatment period (mean duration 13.6 months). b Over treatment period for Study 1 and over 24 months for Study 2. c Disability progression defined as 1-point increase in Expanded Disability Status Scale (EDSS) confirmed 3 months or 6 months later. d Prospective planned pooled analysis of Studies 1 and 2. e Not statistically significant. f Over 12 months for Study 1 and over 24 months for Study 2. g At 12 months for Study 1 and at 24 months for Study 2. |
||||
|
Endpoints |
Study 1 |
Study 2 |
||
|
ZEPOSIA
|
IFN beta-1a
|
ZEPOSIA
|
IFN beta-1a
|
|
|
Clinical Endpoints |
||||
|
Annualized Relapse Rate (Primary Endpoint) |
0.181a |
0.350 a |
0.172 |
0.276 |
|
Relative Reduction |
48% (p<0.0001) |
38% (p<0.0001) |
||
|
Percentage of patients without relapseb |
78% |
66% |
76% |
64% |
|
Proportion of Patients with 3-Month Confirmed Disability Progressionc,d |
7.6% ZEPOSIA vs. 7.8% IFN beta-1a |
|||
|
Hazard Ratio |
0.95 (p=0.77)e |
|||
|
MRI Endpoints |
||||
|
Mean number of new or enlarging T2 hyperintense lesions per MRIf |
1.47 |
2.84 |
1.84 |
3.18 |
|
Relative Reduction |
48% (p<0.0001) |
42% (p<0.0001) |
||
|
Mean number of T1 Gd-enhancing lesionsg |
0.16 |
0.43 |
0.18 |
0.37 |
|
Relative Reduction |
63% (p<0.0001) |
53% (p=0.0006) |
A similar effect of ZEPOSIA on the ARR compared to IFN beta-1a was observed in exploratory subgroups defined by sex, age, prior non-steroid therapy for MS, and baseline disease activity.
14.2 Ulcerative Colitis
The efficacy and safety of ZEPOSIA were evaluated in two multicenter, randomized, double-blind, placebo-controlled clinical studies [UC Study 1 (induction) and UC Study 2 (maintenance) (NCT02435992)] in adult patients with moderately to severely active ulcerative colitis.
5.7 Respiratory Effects
Dose-dependent reductions in absolute forced expiratory volume over 1 second (FEV1) were observed in MS patients treated with ZEPOSIA as early as 3 months after treatment initiation. In the MS pooled analyses of Study 1 and Study 2, the decline in absolute FEV1 from baseline in patients treated with ZEPOSIA compared to patients who received IFN beta-1a was 60 mL (95% CI: -100, -20) at 12 months. The mean difference in percent predicted FEV1 at 12 months between patients treated with ZEPOSIA and patients who received IFN beta-1a was 1.9% (95% CI: -2.9, -0.8). Dose-dependent reductions in forced vital capacity (FVC) (absolute value and %-predicted) were also seen at Month 3 in pooled analyses comparing patients treated with ZEPOSIA to patients who received IFN beta-1a [60 mL, 95% CI (-110, -10); 1.4%, 95% CI: (-2.6, -0.2)], though significant reductions were not seen at other timepoints. There is insufficient information to determine the reversibility of the decrease in FEV1 or FVC after drug discontinuation. One patient in MS Study 1 discontinued ZEPOSIA because of dyspnea.
In UC Study 1 the mean difference in decline in absolute FEV1 from baseline in patients treated with ZEPOSIA compared to patients who received placebo was 22 mL (95% CI: -84, 39) at 10 weeks. The mean difference in percent predicted normal (PPN) FEV1 at 10 weeks between patients treated with ZEPOSIA compared to those who received placebo was 0.8% (95% CI: -2.6, 1.0). The difference in reductions in FVC (absolute value and %-predicted) seen at Week 10 in UC Study 1, comparing patients who were treated with ZEPOSIA to those who received placebo was 44 mL, 95% CI (-114, 26); 0.5%, 95% CI (-2.3, 1.2), respectively. There is insufficient information to determine the reversibility of observed decreases in FEV1 or FVC after discontinuation of ZEPOSIA, or whether changes could be progressive with continued use.
Spirometric evaluation of respiratory function should be performed during therapy with ZEPOSIA, if clinically indicated.
12.1 Mechanism of Action
Ozanimod is a sphingosine 1-phosphate (S1P) receptor modulator that binds with high affinity to S1P receptors 1 and 5. Ozanimod blocks the capacity of lymphocytes to egress from lymph nodes, reducing the number of lymphocytes in peripheral blood. Ozanimod has minimal or no activity on S1P2, S1P3, and S1P4. The mechanism by which ozanimod exerts therapeutic effects in multiple sclerosis and ulcerative colitis is unknown but may involve the reduction of lymphocyte migration into the central nervous system and intestine.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Infections: ZEPOSIA may increase the risk of infections. Obtain a complete blood count (CBC) before initiation of treatment. Monitor for infection during treatment and for 3 months after discontinuation. Do not start ZEPOSIA in patients with active infections. (5.1)
-
•Progressive Multifocal Leukoencephalopathy (PML): Withhold ZEPOSIA at the first sign or symptom suggestive of PML. (5.2)
-
•Bradyarrhythmia and Atrioventricular Conduction Delays: ZEPOSIA may result in transient decrease in heart rate; titration is required for treatment initiation. Check an electrocardiogram (ECG) to assess for preexisting cardiac conduction abnormalities before starting ZEPOSIA. Consider cardiology consultation for conduction abnormalities or concomitant use with other drugs that decrease heart rate. (2.1, 2.2, 5.3, 7)
-
•Liver Injury: Obtain liver enzyme results before initiation and periodically during treatment. Discontinue if there is evidence of liver injury without other cause. (5.4)
-
•Fetal Risk: Women of childbearing potential should use effective contraception during treatment and for 3 months after stopping ZEPOSIA. (5.5, 8.3)
-
•Increased Blood Pressure (BP): Monitor BP during treatment. (5.6)
-
•Respiratory Effects: May cause a decline in pulmonary function. Assess pulmonary function (e.g., spirometry) if clinically indicated. (5.7)
-
•Macular Edema: Increases the risk of macular edema. Obtain a baseline evaluation of the fundus, including the macula, near the start of treatment with ZEPOSIA. Conduct an evaluation of the fundus, including the macula, periodically while on therapy and any time there is a change in vision. Consider discontinuing ZEPOSIA if macular edema develops. Diabetes mellitus and uveitis increase the risk. (5.8)
-
•Cutaneous Malignancies: Skin examination prior to or shortly after the start of treatment and periodically thereafter is recommended. Suspicious skin lesions should be evaluated. (5.9)
5.9 Cutaneous Malignancies
The risk of cutaneous malignancies (including basal cell carcinoma (BCC), squamous cell carcinoma (SCC), and melanoma) is increased in patients treated with S1P receptor modulators. Cases of BCC, SCC, and melanoma have been reported in patients treated with ZEPOSIA; melanoma and BCC were reported in controlled trials [see Adverse Reactions (6.1) ] . Kaposi’s sarcoma and Merkel cell carcinoma have also been reported in patients treated with S1P receptor modulators in the postmarketing setting.
Skin examinations are recommended prior to or shortly after the start of treatment and periodically thereafter for all patients, particularly those with risk factors for skin cancer. Providers and patients are advised to monitor for suspicious skin lesions. If a suspicious skin lesion is observed, it should be promptly evaluated. As usual for patients with increased risk for skin cancer, exposure to sunlight and ultraviolet light should be limited by wearing protective clothing and using a sunscreen with a high protection factor. Concomitant phototherapy with UV-B radiation or PUVA photochemotherapy is not recommended in patients taking ZEPOSIA.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
-
•Assessments are required prior to initiating ZEPOSIA. (2.1)
-
•Titration is required for treatment initiation. (2.2)
-
•The recommended maintenance dosage is 0.92 mg orally once daily. (2.2)
-
•The recommended maintenance dosage in patients with mild or moderate chronic hepatic impairment (Child-Pugh class A or B) is 0.92 mg once every other day. (2.3)
-
•If a dose is missed within the first 2 weeks of treatment, reinitiate with the titration regimen. If a dose is missed after the first 2 weeks of treatment, continue treatment as planned. (2.4)
Package/label Display Panel (Package/Label Display Panel)
Rx only
NDC 59572-890-28
ZEPOSIA®
(ozanimod) capsules
STARTER KIT
STEP 1: (Days 1-7)
Starting at Day 1, complete the 7-Day Starter Pack
Days 1-4: Take one 0.23 mg capsule orally once daily
Days 5-7: Take one 0.46 mg capsule orally once daily
STEP 2: (Day 8 and thereafter)
After completing the 7-Day Starter Pack, begin
taking one 0.92 mg capsule a day or as directed
from the 21-count bottle.
This Starter Kit contains 28 capsules for titration
over 7 days up to prescribed maintenance dose
of one 0.92 mg capsule taken once daily or as
directed. The contents of the Starter Kit, which
are not to be sold separately, are as follows:
Four 0.23 mg capsules
Three 0.46 mg capsules
Twenty One 0.92 mg capsules
28 CAPSULES
Dispense the accompanying Medication Guide to each patient.
Bristol Myers Squibb
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Capsules:
-
•0.23 mg ozanimod: light grey opaque body/light grey opaque cap imprinted with black ink "OZA" on the cap and "0.23 mg" on the body
-
•0.46 mg ozanimod: light grey opaque body/orange opaque cap imprinted with black ink "OZA" on the cap and "0.46 mg" on the body
-
•0.92 mg ozanimod: orange opaque body/orange opaque cap imprinted with black ink "OZA" on the cap and "0.92 mg" on the body
5.6 Increased Blood Pressure
In MS Study 1 and Study 2, patients treated with ZEPOSIA had an average increase of approximately 1 to 2 mm Hg in systolic pressure over patients who received IFN beta-1a, and no effect on diastolic pressure. The increase in systolic pressure was first detected after approximately 3 months of treatment and persisted throughout treatment. Hypertension was reported as an adverse reaction in 3.9% of patients treated with ZEPOSIA 0.92 mg and in 2.1% of patients who received IFN beta-1a. Two patients treated with ZEPOSIA in MS Study 1 and one patient treated with interferon (IFN) beta-1a in Study 2 experienced a hypertensive crisis that was not clearly influenced by a concomitant medication.
The mean increase in systolic blood pressure (SBP) and diastolic blood pressure (DBP) in UC patients treated with ZEPOSIA is similar to patients with MS. In UC Study 1 and Study 3, the average increase from baseline in SBP was 3.7 mm Hg in patients treated with ZEPOSIA and 2.3 mm Hg in patients treated with placebo. In UC Study 2, the average increase from baseline in SBP was 5.1 mm Hg in patients treated with ZEPOSIA and 1.5 mm Hg in patients treated with placebo. There was no effect on DBP.
Hypertension was reported as an adverse reaction in 1.2% of patients treated with ZEPOSIA 0.92 mg and none in patients treated with placebo in UC Study 1 and Study 3, and in 2.2% and 2.2% of patients in UC Study 2, respectively. Hypertensive crisis was reported in two patients receiving ZEPOSIA and one patient receiving placebo.
Blood pressure should be monitored during treatment with ZEPOSIA and managed appropriately.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of ZEPOSIA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Hepatobiliary Disorders: Liver injury [see Warnings and Precautions (5.4)]
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Principal Display Panel Kit Carton (PRINCIPAL DISPLAY PANEL - Kit Carton)
Rx only
NDC 59572-890-91
ZEPOSIA®
(ozanimod) capsules
STARTER KIT
STEP 1: (Days 1-7)
Starting at Day 1, complete the 7-Day Starter Pack
Days 1-4: Take one 0.23 mg capsule orally once daily
Days 5-7: Take one 0.46 mg capsule orally once daily
STEP 2: (Day 8 and thereafter)
After completing the 7-Day Starter Pack, begin taking
one 0.92 mg capsule once daily or as directed from the 30-count bottle.
This Starter Kit contains 37 capsules for titration
over 7 days up to prescribed maintenance dose
of one 0.92 mg capsule taken once daily or as directed. The
contents of the Starter Kit, which are not to be
sold separately, are as follows:
Four 0.23 mg capsules
Three 0.46 mg capsules
Thirty 0.92 mg capsules
37 CAPSULES
Dispense the accompanying Medication Guide to each patient.
Bristol Myers Squibb
2.1 Assessments Prior to First Dose of Zeposia (2.1 Assessments Prior to First Dose of ZEPOSIA)
Before initiation of treatment with ZEPOSIA, assess the following:
5.2 Progressive Multifocal Leukoencephalopathy
Progressive multifocal leukoencephalopathy (PML) is an opportunistic viral infection of the brain caused by the JC virus (JCV) that typically occurs in patients who are immunocompromised, and that usually leads to death or severe disability. Typical symptoms associated with PML are diverse, progress over days to weeks, and include progressive weakness on one side of the body or clumsiness of limbs, disturbance of vision, and changes in thinking, memory, and orientation leading to confusion and personality changes.
PML has been reported in patients treated with S1P receptor modulators, including ZEPOSIA, and other multiple sclerosis (MS) and UC therapies and has been associated with some risk factors (e.g., immunocompromised patients, polytherapy with immunosuppressants, duration of use). Based on data from patients with MS, longer treatment duration increases the risk of PML in patients treated with S1P receptor modulators, and the majority of PML cases have occurred in patients treated with S1P receptor modulators for at least 18 months. Physicians should be vigilant for clinical symptoms or MRI findings that may be suggestive of PML. MRI findings may be apparent before clinical signs or symptoms. If PML is suspected, treatment with ZEPOSIA should be suspended until PML has been excluded by an appropriate diagnostic evaluation.
If PML is confirmed, treatment with ZEPOSIA should be discontinued.
Immune reconstitution inflammatory syndrome (IRIS) has been reported in MS patients treated with S1P receptor modulators who developed PML and subsequently discontinued treatment. IRIS presents as a clinical decline in the patient’s condition that may be rapid, can lead to serious neurological complications or death, and is often associated with characteristic changes on MRI. The time to onset of IRIS in patients with PML was generally within a few months after S1P receptor modulator discontinuation. Monitoring for development of IRIS and appropriate treatment of the associated inflammation should be undertaken.
5.10 Posterior Reversible Encephalopathy Syndrome
Rare cases of posterior reversible encephalopathy syndrome (PRES) have been reported in patients receiving a S1P receptor modulator. In MS controlled clinical trials with ZEPOSIA, one case of PRES was reported. Should a ZEPOSIA-treated patient develop any unexpected neurological or psychiatric symptoms/signs (e.g., cognitive deficits, behavioral changes, cortical visual disturbances, or any other neurological cortical symptoms/signs), any symptom/sign suggestive of an increase of intracranial pressure, or accelerated neurological deterioration, the physician should promptly schedule a complete physical and neurological examination and should consider an MRI. Symptoms of PRES are usually reversible but may evolve into ischemic stroke or cerebral hemorrhage. Delay in diagnosis and treatment may lead to permanent neurological sequelae. If PRES is suspected, treatment with ZEPOSIA should be discontinued.
5.13 Immune System Effects After Stopping Zeposia (5.13 Immune System Effects after Stopping ZEPOSIA)
After discontinuing ZEPOSIA, the median time for peripheral blood lymphocytes to return to the normal range was approximately 30 days, with approximately 80% to 90% of patients in the normal range within 3 months [see Clinical Pharmacology (12.2)]. Use of immunosuppressants within this period may lead to an additive effect on the immune system, and therefore caution should be applied when initiating other drugs 4 weeks after the last dose of ZEPOSIA [see Drug Interactions (7)].
Principal Display Panel 0.92 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 0.92 mg Capsule Bottle Label)
NDC 59572-820-30
ZEPOSIA®
(ozanimod) capsules
0.92 mg
Dispense the accompanying
Medication Guide to each patient.
Rx only
30 Capsules
Bristol-Myers Squibb
2.4 Reinitiation of Zeposia After Treatment Interruption (2.4 Reinitiation of ZEPOSIA after Treatment Interruption)
If a dose of ZEPOSIA is missed during the first 2 weeks of treatment, reinitiate treatment using the titration regimen [see Dosage and Administration (2.2)].
If a dose of ZEPOSIA is missed after the first 2 weeks of treatment, continue with the treatment as planned.
2.3 Recommended Dosage in Patients With Hepatic Impairment (2.3 Recommended Dosage in Patients with Hepatic Impairment)
In patients with mild or moderate hepatic impairment (Child-Pugh class A or B), initiate ZEPOSIA with a 7-day titration, as shown in Table 1. After initial titration, the recommended dosage of ZEPOSIA in these patients is 0.92 mg taken orally once every other day, starting on Day 8 [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
5.3 Bradyarrhythmia and Atrioventricular Conduction Delays
Since initiation of ZEPOSIA may result in a transient decrease in heart rate and atrioventricular conduction delays, an up-titration scheme should be used to reach the maintenance dosage of ZEPOSIA [see Dosage and Administration (2.2) and Clinical Pharmacology (12.2)].
ZEPOSIA was not studied in patients who had:
-
•A myocardial infarction, unstable angina, stroke, TIA, or decompensated heart failure requiring hospitalization within the last 6 months
-
•New York Heart Association Class III / IV heart failure
-
•Cardiac conduction or rhythm disorders, including sick sinus syndrome, significant QT prolongation (QTcF > 450 msec in males, > 470 msec in females), risk factors for QT prolongation, or other conduction abnormalities or cardiac condition that in the opinion of the treating investigator could jeopardize the patient's health
-
•Other pre-existing stable cardiac conditions without clearance from a cardiologist
-
•Severe untreated sleep apnea
-
•A resting heart rate less than 55 beats per minute (bpm) at baseline
Principal Display Panel 7 Capsule Starter Pack Kit Carton (PRINCIPAL DISPLAY PANEL - 7 Capsule Starter Pack Kit Carton)
NDC 59572-810-07
Rx only
ZEPOSIA®
(ozanimod) capsules
7-DAY
STARTER PACK
This pack contains 7 capsules for dosing
over 7 days. The contents of this pack are
as follows:
Four 0.23 mg capsules
Three 0.46 mg capsules
7 CAPSULES
Dispense the accompanying Medication Guide to each patient.
Bristol-Myers Squibb
2.2 Recommended Dosage for Multiple Sclerosis and Ulcerative Colitis
Initiate ZEPOSIA with a 7-day titration, as shown in Table 1 [see Warnings and Precautions (5.3) ]. After initial titration, the recommended dosage of ZEPOSIA is 0.92 mg taken orally once daily starting on Day 8.
Swallow ZEPOSIA capsules whole, with or without food [see Clinical Pharmacology (12.3)].
|
Days 1-4 |
0.23 mg once daily |
|
Days 5-7 |
0.46 mg once daily |
|
Day 8 and thereafter |
0.92 mg once daily* |
*Patients with mild to moderate hepatic impairment (Child-Pugh class A or B) should take 0.92 mg once every other day [see Recommended Dosage in Patients with Hepatic Impairment (2.3) .]
5.12 Severe Increase in Multiple Sclerosis Disability After Stopping Zeposia (5.12 Severe Increase in Multiple Sclerosis Disability after Stopping ZEPOSIA)
In MS, severe exacerbation of disease, including disease rebound, has been rarely reported after discontinuation of a S1P receptor modulator. The possibility of severe exacerbation of disease should be considered after stopping ZEPOSIA treatment. Patients should be observed for a severe increase in disability upon ZEPOSIA discontinuation and appropriate treatment should be instituted, as required.
After stopping ZEPOSIA in the setting of PML, monitor for development of immune reconstitution inflammatory syndrome (PML-IRIS) [see Warnings and Precautions (5.2) ].
5.11 Unintended Additive Immunosuppressive Effects From Prior Treatment With Immunosuppressive Or Immune Modulating Drugs (5.11 Unintended Additive Immunosuppressive Effects from Prior Treatment with Immunosuppressive or Immune-Modulating Drugs)
When switching from drugs with prolonged immune effects, the half-life and mode of action of these drugs must be considered to avoid unintended additive immunosuppressive effects while at the same time minimizing risk of disease reactivation, when initiating ZEPOSIA.
Initiating treatment with ZEPOSIA after treatment with alemtuzumab is not recommended [see Drug Interactions (7)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:42:50.199978 · Updated: 2026-03-14T22:11:10.971269