These Highlights Do Not Include All The Information Needed To Use Zymfentra Safely And Effectively. See Full Prescribing Information For Zymfentra.
93a193ab-9f96-4da6-aa7c-31212e81879a
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
WARNING: SERIOUS INFECTIONS and MALIGNANCY See full prescribing information for complete boxed warning Increased risk of serious infections leading to hospitalization or death, including tuberculosis (TB), bacterial sepsis, invasive fungal infections (such as hitoplasmosis) and infections due to ohter opportunistic pathogens. (5.1) Discontinue ZYMFENTRA if a patient develops a serious infection or sepsis. (5.1) Perform test for latent TB; if positive, start treatment for TB prior to starting ZYMFENTRA. Monitor all patients for active TB during treatment, even if initial latent TB test is negative. (5.1) Lymphoma and other malignancies, some fatal, have been reported in children and adolescent patients treated with tumor necrosis factor (TNF) blockers, including infliximab products. (5.2) Postmarketing cases of fatal hepatosplenic T-cell lymphoma (HSTCL) have been reported in patients treated with TNF blockers including infliximab products. Almost all had received azathioprine or 6-mercaptopurine concomitantly with a TNF blocker at or prior to diagnosis. The majority of cases were reported in patients with Crohn's disease or ulcerative colitis, most of whom were adolescent or young adult males. (5.2 )
Indications and Usage
ZYMFENTRA is a tumor necrosis factor (TNF) blocker indicated in adults for maintenance treatment of: moderately to severely active ulcerative colitis following treatment with an infliximab product administered intravenously. ( 1 ) moderately to severely active Crohn’s disease following with an infliximab products administered intraneously. ( 1 )
Dosage and Administration
Important Dosage Information. ( 2.1 ). ZYMFENTRA is indicated as maintenance treatment only, starting at Week 10 and thereafter. ▫ All pateitns must complete an intravenous induction regiemen with an infliximab product before starting ZYMFENTRA. ZYMFENTRA is for subcutaneous use only. Recommeneded Maintenance Dosage in Ulcerative Colitis and Crohn's Direase ( 2.2 ) Week 10 and thereafter : Inject 120 mg subcutaneously once every two weeks. To swift patients who are responding to maintenance therapy with an infliximab product administered intravenously, administer the first subcutaneous dose of ZYMFENTRA in place of the next scheduled intravenous infusion and every two weeks thereafter. See the full prescribing information on how to administer subcutaneously. ( 2.3 )
Warnings and Precautions
Serious injections, including invasive fungal infections: Avoid use of ZYMFENTRA in patients with an active infection. If infection develops during treatment, conduct a prompt and complete diagnostic workup appropriate for an immunocompromised patient and initiate antimicrobial therapy. If systemic illness develops in patients who reside or travel to regions where mycoses are endemic, consider empiric antifungal therapy. ( 5.1 ) Malignancies : The incidence of malignancies, including lymphoma, was greater in TNF blocker-treated patients than in controls. Consider the higher risk of hepatosplenic T-cell lymphoma (HSTCL) with combination therapy versus increased tisk of immunogenicity and hypersensitivity reactions with monotherapy. ( 5.2 ) Hepatitis B virus (HBV) reactivation: Test for HBV infection before starting ZYMFENTRA. Monitor HBV carriers during and several months after therapy for active HBV infection. If reactivation occurs, stop ZYMFENTRA and begin anti-viral therapy. ( 5.3 ) Hepatotoxicity: Severe hepatic reactions, some fatal or necessitating liver transplantation with infliximab products. Monitor hepatic enzymes and liver function tests every 3 to 4 months during treatment; investigate liver enzyme elevations and interrupt treatment if drug-induced liver injury is suspected. Instruct patients to seek immediate medical attention if symptoms develop. ( 5.4 ) Congestive heart failure (CHF) : New onset or worsening symptoms may occur. Avoid ZYMFENTRA in patients with CHF. Monitor for new or worsening symptoms if a decision is made to administer ZYMFENTRA. ( 5.5 ) Hematologic Reactions : Advise patients to seek immediate medical attention if signs and symtoms of cytopenia develop; consider stopping ZYMFENTRA if significant hematologic abnormalities devlop. ( 5.6 ) Hypersensitivity and Other Administration Reactions : Serious systemic hypersensitivity reactions including anaphylaxis; institute appropriate therapy and discontinue ZYMFENTRA. ( 4 , 5.7 ) Neurologic Reactions : Exacerbation or new onset CNS demyelinating disorders may occurs; consider discontinuation of ZYMFENTRA. ( 5.8 ) Risk of infection with concurrent administration of other biological products : Concurrent use with other immunosuppressive biological products may increase the risk of injection ( 5.9 ) Risk of additive immunosuppressive effects from prior biologic products : Consider the half-life and mode of action of prior biological products. ( 5.10 ) Autoimmunity : Formation of autoantibodies and development of lupus-like syndrome may occur; discontinue ZYMFENTRA if symptoms develop. ( 5.11 ) V accinations and Use of Live Vaccines/Therapeutic Infectious Agents: Prior to initiating ZYMFENTRA bring patients up to date with all vaccinations. Live vaccines or therapeutic infectious agents should not be given with ZYMFENTRA. A 6-month waiting period following birth is recommended before the administration of live vaccines to infants exposed in utero to infliximab products. ( 5.12 )
Contraindications
ZYMFENTRA is contraindicated in patients with a history of a severe hypersensitivity reaction to infliximab-dyyb, other infliximab products, any of the inactive ingredients in ZYMFENTRA, or any murine proteins. Reactions have included anaphylaxis [ see Warnings and Precautions ( 5.7 ) ].
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Serious infections [ see Warnings and Precautions ( 5.1 ) ] Malignancies [ see Warnings and Precautions ( 5.2 ) ] Hepatitis B virus reactivation [ see Warnings and Precautions ( 5.3 ) ] Hepatotoxicity [ see Warning and Precautions ( 5.4 ) ] Congestive heart failure [ see Warnings and Precautions ( 5.5 ) ] Hematologic reactions [ see Warnings and Precautions ( 5.6 ) ] Hypersensitivity and other administration reactions [ see Warnings and Precautions ( 5.7 ) ] Neruologic reactions [ see Warnings and Precautions ( 5.8 ) ] Autoimmunity [ see Warnings and Precautions ( 5.11 ) ]
Medication Information
Recent Major Changes
WARNING: SERIOUS INFECTIONS and MALIGNANCY See full prescribing information for complete boxed warning Increased risk of serious infections leading to hospitalization or death, including tuberculosis (TB), bacterial sepsis, invasive fungal infections (such as hitoplasmosis) and infections due to ohter opportunistic pathogens. (5.1) Discontinue ZYMFENTRA if a patient develops a serious infection or sepsis. (5.1) Perform test for latent TB; if positive, start treatment for TB prior to starting ZYMFENTRA. Monitor all patients for active TB during treatment, even if initial latent TB test is negative. (5.1) Lymphoma and other malignancies, some fatal, have been reported in children and adolescent patients treated with tumor necrosis factor (TNF) blockers, including infliximab products. (5.2) Postmarketing cases of fatal hepatosplenic T-cell lymphoma (HSTCL) have been reported in patients treated with TNF blockers including infliximab products. Almost all had received azathioprine or 6-mercaptopurine concomitantly with a TNF blocker at or prior to diagnosis. The majority of cases were reported in patients with Crohn's disease or ulcerative colitis, most of whom were adolescent or young adult males. (5.2 )
Warnings and Precautions
Serious injections, including invasive fungal infections: Avoid use of ZYMFENTRA in patients with an active infection. If infection develops during treatment, conduct a prompt and complete diagnostic workup appropriate for an immunocompromised patient and initiate antimicrobial therapy. If systemic illness develops in patients who reside or travel to regions where mycoses are endemic, consider empiric antifungal therapy. ( 5.1 ) Malignancies : The incidence of malignancies, including lymphoma, was greater in TNF blocker-treated patients than in controls. Consider the higher risk of hepatosplenic T-cell lymphoma (HSTCL) with combination therapy versus increased tisk of immunogenicity and hypersensitivity reactions with monotherapy. ( 5.2 ) Hepatitis B virus (HBV) reactivation: Test for HBV infection before starting ZYMFENTRA. Monitor HBV carriers during and several months after therapy for active HBV infection. If reactivation occurs, stop ZYMFENTRA and begin anti-viral therapy. ( 5.3 ) Hepatotoxicity: Severe hepatic reactions, some fatal or necessitating liver transplantation with infliximab products. Monitor hepatic enzymes and liver function tests every 3 to 4 months during treatment; investigate liver enzyme elevations and interrupt treatment if drug-induced liver injury is suspected. Instruct patients to seek immediate medical attention if symptoms develop. ( 5.4 ) Congestive heart failure (CHF) : New onset or worsening symptoms may occur. Avoid ZYMFENTRA in patients with CHF. Monitor for new or worsening symptoms if a decision is made to administer ZYMFENTRA. ( 5.5 ) Hematologic Reactions : Advise patients to seek immediate medical attention if signs and symtoms of cytopenia develop; consider stopping ZYMFENTRA if significant hematologic abnormalities devlop. ( 5.6 ) Hypersensitivity and Other Administration Reactions : Serious systemic hypersensitivity reactions including anaphylaxis; institute appropriate therapy and discontinue ZYMFENTRA. ( 4 , 5.7 ) Neurologic Reactions : Exacerbation or new onset CNS demyelinating disorders may occurs; consider discontinuation of ZYMFENTRA. ( 5.8 ) Risk of infection with concurrent administration of other biological products : Concurrent use with other immunosuppressive biological products may increase the risk of injection ( 5.9 ) Risk of additive immunosuppressive effects from prior biologic products : Consider the half-life and mode of action of prior biological products. ( 5.10 ) Autoimmunity : Formation of autoantibodies and development of lupus-like syndrome may occur; discontinue ZYMFENTRA if symptoms develop. ( 5.11 ) V accinations and Use of Live Vaccines/Therapeutic Infectious Agents: Prior to initiating ZYMFENTRA bring patients up to date with all vaccinations. Live vaccines or therapeutic infectious agents should not be given with ZYMFENTRA. A 6-month waiting period following birth is recommended before the administration of live vaccines to infants exposed in utero to infliximab products. ( 5.12 )
Indications and Usage
ZYMFENTRA is a tumor necrosis factor (TNF) blocker indicated in adults for maintenance treatment of: moderately to severely active ulcerative colitis following treatment with an infliximab product administered intravenously. ( 1 ) moderately to severely active Crohn’s disease following with an infliximab products administered intraneously. ( 1 )
Dosage and Administration
Important Dosage Information. ( 2.1 ). ZYMFENTRA is indicated as maintenance treatment only, starting at Week 10 and thereafter. ▫ All pateitns must complete an intravenous induction regiemen with an infliximab product before starting ZYMFENTRA. ZYMFENTRA is for subcutaneous use only. Recommeneded Maintenance Dosage in Ulcerative Colitis and Crohn's Direase ( 2.2 ) Week 10 and thereafter : Inject 120 mg subcutaneously once every two weeks. To swift patients who are responding to maintenance therapy with an infliximab product administered intravenously, administer the first subcutaneous dose of ZYMFENTRA in place of the next scheduled intravenous infusion and every two weeks thereafter. See the full prescribing information on how to administer subcutaneously. ( 2.3 )
Contraindications
ZYMFENTRA is contraindicated in patients with a history of a severe hypersensitivity reaction to infliximab-dyyb, other infliximab products, any of the inactive ingredients in ZYMFENTRA, or any murine proteins. Reactions have included anaphylaxis [ see Warnings and Precautions ( 5.7 ) ].
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Serious infections [ see Warnings and Precautions ( 5.1 ) ] Malignancies [ see Warnings and Precautions ( 5.2 ) ] Hepatitis B virus reactivation [ see Warnings and Precautions ( 5.3 ) ] Hepatotoxicity [ see Warning and Precautions ( 5.4 ) ] Congestive heart failure [ see Warnings and Precautions ( 5.5 ) ] Hematologic reactions [ see Warnings and Precautions ( 5.6 ) ] Hypersensitivity and other administration reactions [ see Warnings and Precautions ( 5.7 ) ] Neruologic reactions [ see Warnings and Precautions ( 5.8 ) ] Autoimmunity [ see Warnings and Precautions ( 5.11 ) ]
Description
WARNING: SERIOUS INFECTIONS and MALIGNANCY See full prescribing information for complete boxed warning Increased risk of serious infections leading to hospitalization or death, including tuberculosis (TB), bacterial sepsis, invasive fungal infections (such as hitoplasmosis) and infections due to ohter opportunistic pathogens. (5.1) Discontinue ZYMFENTRA if a patient develops a serious infection or sepsis. (5.1) Perform test for latent TB; if positive, start treatment for TB prior to starting ZYMFENTRA. Monitor all patients for active TB during treatment, even if initial latent TB test is negative. (5.1) Lymphoma and other malignancies, some fatal, have been reported in children and adolescent patients treated with tumor necrosis factor (TNF) blockers, including infliximab products. (5.2) Postmarketing cases of fatal hepatosplenic T-cell lymphoma (HSTCL) have been reported in patients treated with TNF blockers including infliximab products. Almost all had received azathioprine or 6-mercaptopurine concomitantly with a TNF blocker at or prior to diagnosis. The majority of cases were reported in patients with Crohn's disease or ulcerative colitis, most of whom were adolescent or young adult males. (5.2 )
Section 42229-5
SERIOUS INFECTIONS
Patients treated with TNF blockers, inculding ZYMFENTRA, are at increased risk for developing serious infections that may lead to hospitalization or death [see Warnings and Precautions (5.1), Adverse Reactions (6.1)]. Most patients who developed these infections were taking concomitant immunosuppressants such as methotrexate or corticosteroids.
Discontinue ZYMFENTRA if a patient develops a serious infection or sepsis.
Reported infections include:
- Active tuberculosis, including reactivation of latent tuberculosis. Patients with tuberculosis have frequently presented with disseminated or extrapulmonary disease. Test patientsfor latent tuberculosis before ZYMFENTRA use and during therapy. Initiate treatment for latent infection prior to ZYMFENTRA use.
- Invasive fungal infections, including histoplasmosis, coccidioidomycosis, candidiasis, aspergillosis, blastomycosis, and pneumocystosis. Patients with histoplasmosis or other invasive fungal infections may present with disseminated, rather than localized, disease. Antigen and antibody testing for histoplasmosis may be negative in some patients with active infection. Consider empiric anti-fungal therapy in patients at risk for invasive fungal infections who develop severe systemic illness.
- Bacterial, viral and other infections due to opportunistic pathogens, including Legionella and Listeria.
Carefully consider the risks and benefits of treatment with ZYMFENTRA prior to initiating therapy in patients with chronic or recurrent infection.
Closely monitor patients for the development of signs and symtoms of infection during and after treatment with ZYMFENTRA, including the possible development of tuberculosis in patients who tested negative for latent tuberculosis infection prior to initiating therapy.
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration Revised: 02/2024 | ||
|
MEDICATION GUIDE
ZYMFENTRATM (Zim fen' trah) (infliximab-dyyb) injection, for subcutaneous use |
||
| Read the Medication Guide that comes with ZYMFENTRA before you receive the first treatment, and before each time you receive a treatment of ZYMFENTRA. This Medication Guide does not take the place of talking with your doctor about your medical condition or treatment. | ||
|
What is the most important information I should know about ZYMFENTRA? ZYMFENTRA may cause serious side effects, including:
After starting ZYMFENTRA, if you have an infection, any sign of an infection including a fever, cough, flu-like symptoms, or have open cuts or sores on your body, call your doctor right away. ZYMFENTRA can make you more likely to get infections or make any infection that you have worse.
|
||
|
What is ZYMFENTRA?
ZYMFENTRA is a prescription medicine used as an injection uner the skin (subcutaneous injection) by adults for the maintenance treatment of:
|
||
Do not take ZYMFNETRA if you:
|
||
|
Before you receive ZYMFENTRA, tell your doctor all of your medical conditions, including if you:
If you have a baby and you were receiving ZYMFENTRA during your pregnancy, it is important to tell your baby's doctor and other healthcare professionals about your ZYMFENTRA use so they can decide when your baby should receive any vaccine. Certain vaccinations can cause infections.
|
||
How should I receive ZYMFENTRA?
- If your healthcare provider decides that you or your cargiver can give your injections of ZYMFENTRA at home, you or your caregiver should be shown the right way to prepare and inject ZYMFENTRA. - Do not try to inject ZYMFENTRA yourself until you or your caregiver have been shown how to inject ZYMFENTRA by your healthcare provider. - ZYMFENTRA is injected under your skin (subcutaneously) 1 time every two weeks. - Inject ZYMFENTRA under the skin (subcutaneous injection), in your upper arms, stomach area (abdomen), or upper legs (thighs). - Do no give an injection in an area of the skin that is tender, bruised, red or hard. - Use a different injection site each time you use ZYMFENTRA. - If you are not able to inject ZYMFENTRA at your regular scheduled time or you miss a dose of ZYMFENTRA, inject the dose as soon as possible. Then, inject your next dose every two weeks thereafter. If you are not sure when to inject ZYMFENTRA, call your healthcare provider. - If you inject more than prescribed, call your doctor right away. - Be sure to keep all of your scheduled follow-up appointments. Read the detailed Instructions for Use at the end of this Medication Guide for insructions about how to prepare and inject a dose of ZYMFENTRA, and how to properly throw away (dispose of) used needles and syringes. The syringe and needle must never be re-used. After the rubber stopper is puntured, ZYMFENTRA can become contaiminated by harmful bacteria which could cause an infection if re-used. Therefore, throw away any unused portion of ZYMFENTRA. |
||
|
What should I avoid while taking ZYMFENTRA?
|
||
|
What are the possible side effects of ZYMFENTRA? ZYMFENTRA can cause serious side effects, including (see "What is the most important information I should know about ZYMFENTRA ?"): Serious Infections
|
||
|
|
|
|
||
|
|
|
|
Liver Injury Some patients receiving infliximab products have developed serious liver problems. Tell your doctor if you have: |
||
|
|
|
|
Heart Failure If you have a heart problem called congestive heart failure, your doctor should check you closely while you are receiving ZYMFENTRA. Your congestive heart failure may get worse while you are receiving ZYMFENTRA. Be sure to tell your doctor of any new or worse symptoms including: |
||
|
|
|
|
Treatment with ZYMFENTRA may need to be stopped if you get new or worse congestive heart failure. Blood Probelms In some patiens receiving infliximab products, the body may not make enough of the blood cells tha help fight infections or help stop bleeding. Tell your doctor if you: |
||
|
|
|
|
Allergic Reactions
|
||
|
|
|
|
Some patients treated with infliximab products have had delayed allergic reactions. Tell your doctor right away if you have any of these signs of delayed reaction to ZYMFENTRA: |
||
|
|
|
|
Nervous System Disorders Some patients receiving infliximab products have developed problems with their nervous system. Tell your doctor if you have: |
||
|
|
|
|
Some patients have experienced a stroke within approximately 24 hours of their infusion with infliximab products. Tell your doctor right away if you have symptoms of a stroke which may include: numbness or weakness of the face, arm, or leg, especially on one side of the body; sudden confusion, trouble speaking or understanding; sudden trouble seeing in one or both eyes, sudden trouble walking, dizziness, loss of balance or coordination or a sudden, severe headache. Lupus-like Syndrome Some patient have developed symptoms that are like the symptoms of Lupus. If you develop any of the following symptoms, your doctor may decide to stop your treatment with ZYMFENTRA: |
||
|
|
|
|
The most common side effects of ZYMFENTRA include: |
||
|
|
|
|
Tell your doctor about any side effect that bothers you or does not go away.
|
||
|
How should I store ZYMFENTRA?
|
||
|
General information about ZYMFENTRA
|
||
|
What are the ingredients in ZYMFENTRA?
|
||
| Manufactured by: CELLTRION, Inc. 23, Academy-ro, Yeonsu-gu, Incheon, 22014, Republic of Korea U.S. License No. 1996 ©CELLTRION, Inc. Distributed by CELLTRION USA, Inc. One Evertrust Plaza Suite 1207, Jersey City, New Jersey, 07302, USA For more information call 1-888-804-3433 |
8.2 Lactation
11 Description
Infliximab-dyyb, a tumor necrosis factor (TNF) blocker, is a chimeric IgG1κ monoclonal antibody (composed of human constant and murine variable regions). It has a molecular weight of approximately 145.9 kDa. Infliximab-dyyb is produced by a recombinant murine myeloma cell line, SP2/0.
ZYMFENTRA (inifliximab-dyyb) injection for subcutaneous use is a sterile, preservative-free, clear to opalescent, colorless to pale brown solution.
ZYMFENTRA is supplied in a single-dose prefilled syringe with 29 gauge fixed 1/2 inch needle, prefilled syringe with 29 gauge fixed 1/2 inch needle with needle guard, or prefilled pen with 27 gauge fixed 1/2 inch needle.
Each mL of solution contains 120 mg infliximab-dyyb, acetic acid (0.19 mg), polysorbate 80 (0.5 mg), sodium acetate (0.56 mg), sorbitol (45 mg) and Water for Injection, USP. The pH is 5.0.
Crohn's Disease
The most common adverse reactions reported in ≥3% of subjects and at a higher rate than placebo in CD Trial I are shown in Table 2.
|
Table 2 Adverse Reactionsa in the Maintenance Phase of a Randomized, Double-Blind |
||
|
ZYMFENTRA 120 mg Subcutaneous Injectionb N=222 (%) |
Placebo N=101 (%) |
|
|
COVID-19 |
10 |
5 |
|
Headache |
8 |
4 |
|
Upper respiratory track infectionsdc |
7 |
3 |
|
Injection site reactiond |
5 |
1 |
|
Diarrhea |
5 |
1 |
|
Increased blood creatine phosphokinase |
4 |
2 |
|
Arthralgia |
4 |
3 |
|
Increased alaine aninotransferase |
4 |
1 |
| Hypertensionse | 3 | 2 |
| Urinary track infectionf | 3 | 2 |
| Neutropenia | 3 | 0 |
| Dizziness | 3 | 0 |
|
Leukopenia |
3 |
0 |
|
a reported in at least 3% of ZYMFENTRA-treated subjects and at a higher rate than placebo b ZYMFENTRA 120 mg administered subcutaneously starting at Week 10 and every 2 weeks thereafter for up to Week 54. c Includes: upper respiratory tract infection, acute sinusitis, chronic sinusitis, influenza like illness, nasopharyngitis, pharyngitis, pharyngitis streptococcal, rhinitis, rhinorrhea, rhinovirus infection, sinusitis, tonsillitis d Some subjects had multiple occurrences of injection site reactions. In this table, injection site reactions are counted only once per subject. Symptoms in individual subjects included one or more of injection site bruising, edema, erythema, induration, pain, pruritus, rash, swelling, warmth. e Includes: hypertension and essential hypertension f Includes: urinary tract infection, pyelonephritis |
5.2 Malignancies
Malignancies, some fatal, have been reported among children, adolescents and young adults who received treatment with TNF blockers (initiation of therapy ≤18 years of age), including infliximab products. AApproximately half of these cases were lymphomas, including Hodgkin’s and non-Hodgkin’s lymphoma. The other cases represented a variety of malignancies, including rare malignancies that are usually associated with immunosuppression and malignancies that are not usually observed in children and adolescents. The malignancies occurred after a median of 30 months (range 1 to 84 months) after the first dose of TNF blocker therapy. Most of the patients were receiving concomitant immunosuppressants. These cases were reported postmarketing and are derived from a variety of sources, including registries and spontaneous postmarketing reports.
5.11 Autoimmunity
Treatment with TNF blockers, iincluding ZYMFENTRA may result in the formation of autoantibodies and in the development of a lupus-like syndrome. If a patient develops symptoms suggestive of a lupus-like syndrome following treatment with ZYMFENTRA, discontinue treatment.
8.4 Pediatric Use
The safety and effectiveness in pediatric patients have not been established.
8.5 Geriatirc Use
Clinical trials of ZYMFENTRA did not include sufficient numbers of sugjects aged 65 and over (8 subjects with UC and 6 subjects with CD) to determined whether they respond differently from younger subjects.
5.4 Hepatotoxicity
Hepatobiliary disorders, including acute liver failure, jaundice abnormal hepatic function, hepatic steatosis, hepatitis, hepatotoxicity, hyperbilirubinemia and non-alcoholic fatty liver, have been reported in postmarketing data in patients receiving infliximab products. Autoimmune hepatitis has been diagnosed in some of these cases. Severe hepatic reactions occurred between two weeks to more than one year after initiation of infliximab products administered intravenously; elevations in hepatic aminotransferase levels were not noted prior to discovery of the liver injury in many of these cases. Some of these cases were fatal or necessitated liver transplantation.
In clinical trials, three subjects treated with ZYMFENTRA had drug induced liver injury based on hepatic transaminase elevations, including one subject with accompanying bilirubin elevation [see Adverse Reactions (6.2)].
Monitor hepatic enzymes and liver function tests every 3 to 4 months during treatment with ZYMFENTRA. Prompt investigation of the cause of liver enzyme elevation should be undertaken to identify potential cases of drug-induced liver injury. Interrupt treatment if drug-induced liver injury is suspected, until this diagnosis is excluded. Instruct patients to seek immediate medical attention if they experience symptoms suggestive of hepatic dysfuction.
Ulcerative Colitis
The most common adverse reactions reported in ≥3% of subjects and at a higher rate than Placebo in UC Trial I are shown in Table 1.
|
Table 1 Adverse Reactionsa in the Maintenance Phase of a Randomized, Double-Blind 54-Week Study of Subjects with UC (UC Tral I) |
||
|
ZYMFENTRA 120 mg Subcutaneous Injectionb N=296 (%) |
Placebo N=140 (%) |
|
|
COVID-19 |
10 |
6 |
|
Anemiad |
5 |
4 |
|
Arthralgia |
4 |
1 |
| Injection site reaction# | 3 | 2 |
|
Increased alanine aminotransferase |
3 |
1 |
|
Abominal painf |
3 |
1 |
|
areported in at least 3% of ZYMFENTRA-treated subjects and at a higher than placebo bZYMFENTRA 120 mg as a subcutaneous injection every two weeks starting Week 10 following 3 intravenous induction doses of inflixiab-dyyb cIncludes: COVID-19 and COVID-19 pneumonia dIncludes: anemia and iron deficiency anemia #Some subjects had multiple occurences of injection site reactions. In this table, injection site reactions are counted only once per subject. Symptoms in individual subjects included one or more of injection site bruising, edema, erythema, induration, pain, pruritus and swelling. fIncludes: abdominal pain, upper abdominal pain, lower abdominal pain, and abdominal discomfort |
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Difference in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of infliximab-dyyb or of other infliximab products.
In UC and CD clinical studies (UC Trial I and CD Trial I), approximately 64% subjects developed anti-drug antibodies to infliximab-dyyb following induction treatment with infliximab-dyyb intravenously and maintenance treatment with ZYMFENTRA by Week 54 [183/287 (64%) in subjects with UC, and 151/232 (65%) in subjects with CD] [see Clinical Studies (14)]. Among subjects with anti-drug antibodies, 92% had neutralizing antibodies [161/183 (88%) subjects with UC, and 147/151 (97%) subjects with CD]. Use of concomitant immunosuppressant agents (azathioprine, 6-mercaptopurine and methotrexate) appeared to reduce the frequency of ADA to infliximab-dyyb.
14 Clinical Studies
4 Contraindications
ZYMFENTRA is contraindicated in patients with a history of a severe hypersensitivity reaction to infliximab-dyyb, other infliximab products, any of the inactive ingredients in ZYMFENTRA, or any murine proteins. Reactions have included anaphylaxis [see Warnings and Precautions (5.7)].
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Serious infections [see Warnings and Precautions (5.1)]
- Malignancies [see Warnings and Precautions (5.2)]
- Hepatitis B virus reactivation [see Warnings and Precautions (5.3)]
- Hepatotoxicity [see Warning and Precautions (5.4)]
- Congestive heart failure [see Warnings and Precautions (5.5)]
- Hematologic reactions [see Warnings and Precautions (5.6)]
- Hypersensitivity and other administration reactions [see Warnings and Precautions (5.7)]
- Neruologic reactions [see Warnings and Precautions (5.8)]
- Autoimmunity [see Warnings and Precautions (5.11)]
7 Drug Interactions
12.3 Phamacokinetics
Following single and multiple subcutaneous dosing of infliximab-dyyb, exposures to infliximab-dyyb (i.e., AUC) are proportionally increased over the dose range from 120 mg to 240 mg (2 times the recommended dosage). After a single subcutaneous dose of infliximab-dyyb 120 mg in healthy subjects, the median (SD) Cmax and AUCinf were 10.0 (3.2) mcg/mL and 6945.6 (2830.2) mcg·h/mL, respectively. Following recommended subcutaneous maintenance dose of ZYMFENTRA (120 mg every 2 weeks) in adult subjects with UC and CD from Week 10 after intravenous induction treatment with infliximab-dyyb, steady-state was achieved by Week 22, and the mean (SD) trough serum concentrations of infliximab-dyyb at steady-state were 14.6 (7.8) mcg/mL and 14.6 (8.9) mcg/mL in subjects with UC and CD, respectively. Pharmacokinetics are comparable between healthy subjects, and subjects with UC or CD.
Instructions for Use
For subcutaneous use only
Read and follow Instructions for Use that come with your ZYMFENTRA Prefilled Syringe before using it and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or treatment.
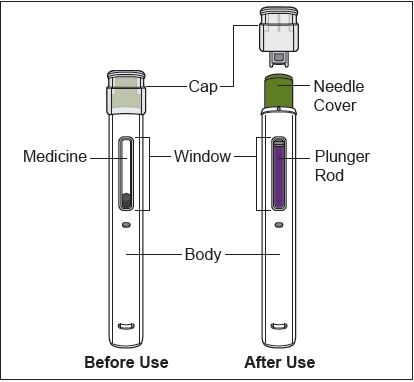
Figure A: Parts of ZYMFENTRA Prefilled Syringe
Caution! Do not remove the Cap until you are ready to inject. When you remove the Cap, do not recap the Prefilled Syringe.
Important Information
- Use the Prefilled Syringe only if your healthcare provider has trained you on the right way to prepare for and to give an injection.
- Ask your healthcare provider how often you will need to give an injection.
- Rotate the injection site each time you give an injection. Each new injection site should be at least 1.2 inches away from the previous injection site.
- Do not use Prefilled Syringe if it has been dropped or is visibly damaged.
- Do not reuse the Prefilled Syringe.
- Do not shake the Prefilled Syringe at any time.
- Do not share the Prefilled Syringe with anyone.
- Do not pull back on the plunger rod at any time.
- Only use each Prefilled Syringe for one injection.
How to store the Prefilled Syringe
- Store the Prefilled Syringe in a refrigerator between 36°F to 46°F (2°C to 8°C).
- Keep the Prefilled Syringe in the original carton to protect it from light.
- Do not use the Prefilled Syringe that has been left in direct sunlight.
- Do not freeze the Prefilled Syringe. If the Prefilled Pen has been frozen, do not use the Prefilled Syringe even if it is thawed.
- Do not warm the Prefilled Syringe using heat sources such as hot water or a microwave. Let the Prefilled Syringe naturally warm at room temperature between 68°F to 77°F (20°C to 25°C) for 30 minutes before giving an injection.
- When the Prefilled Syringe has reached room temperature, do not put it back in the refrigerator. The Prefilled Syringe must be thrown away (discarded) if not used within the 14 days.
- Keep the Prefilled Syringe and all medicines out of the reach of children.
Prepare for the Injection
1. Gather the supplies for the injection.
1a. Prepare a clean, flat surface, such as a table or countertop, in a well-lit area.
1b. Remove 1 Prefilled Syringe from the carton by holding the middle of the Prefilled Syringe Body.
- Do not touch the plunger rod.
Note: Return the carton with any unused Prefilled Syringe to the refrigerator immediately after taking out one Prefilled Syringe.
1c. Make sure you have the following supplies:
- Prefilled Syringe
- Alcohol swab
Not included in the carton:
- Cotton ball or gauze
- Adhesive bandage
- FDA-cleared sharps disposal container
|
2. Inspect the Prefilled Syringe. 2a. Make sure you have the correct medicine (ZYMFENTRA). 2b. Check the expiration (EXP) date on label of the Prefilled Syringe (See Figure B). 2c. Look at the Prefilled Syringe and make sure it is not cracked or damaged.
|
Figure B |
|
3. Inspect the Medicine. 3a. Look at the Prefilled Syringe Body and check the liquid is clear and colorless to pale brown (see Figure C).
|
Figure C |
|
4. Wait 30 minutes. 4a. Leave the Prefilled Syringe at room temperature 68°F to 77°F (20°C to 25°C) for 30 minutes to allow it to warm up (see Figure D).
|
Figure D |
|
5. Choose an injection site (see Figure E). 5a. You may inject into: - the front of your thighs. - the stomach area (abdomen) except for the 2 inches around the belly button (navel). - the outer area of the upper arm (caregiver only).
5b. Rotate the injection site each time you give an injection.
|
Figure E |
|
6. Wash your hands. 6a. Wash your hands with soap and water and dry them thoroughly (see Figure F). |
Figure F |
|
7. Clean the injection site. 7a. Clean the injection site with an alcohol swab using a circular motion (see Figure G).
|
Figure G |
|
Give the injection |
|
|
8. Remove the Cap. 8a. Hold the Prefilled Syringe by the injector body with the Cap on top using one hand. Gently pull the Cap straight off with the other hand.
8b. Throw away (dispose of) the Cap in an FDA cleared sharps container (see Step 12 and Figure H). |
Figure H |
|
9. Insert the Prefilled Syringe into the injection site. 9a. Hold the Prefilled Syringe by its body in one hand between your thumb and index finger. 9b. Gently pinch a fold of skin at the injection site with one hand. 9c. With a quick and “dart-like” motion, insert the Needle completely into the fold of the skin at a 45-defree angle (see Figure I).
|
Figure I |
|
10. Give the injection. 10a. After the Needle is inserted, release the pinched skin. 10b. Push the Plunger down slowly and as far as it will go until the Prefilled Syringe is empty (See Figure J). |
Figure J |
|
11. Remove the Prefilled Syringe from the injection site. 11a. Remove the Needle from your skin at the same angle it was inserted (see Figure K).
|
Figure K |
|
After the injection |
|
|
12. Throw away (dispose of) the Prefilled Syringe. 12a. Put the used Prefilled Syringe in an FDA-cleared sharps disposal container right away after use (see Figure L).
|
Figure L |
|
13. Care for the injection site. 13a. Treat the injection site by gently pressing a cotton ball or gauze to the site and apply an adhesive bandage, if necessary. |
|
|
Manufactured by: CELLTRION, Inc., 23, Academy-ro, Yeonsu-gu, Incheon, 22014, Republic of Korea Distributed by: CELLTRION USA, Inc., 1 Evertrust Plaza Suite 1207, Jersey City, NJ 07302 |
12.2 Pharmacodynamics
No formal pharmacodynamic studies have been conducted.
5.1 Serious Infections
Patients treated with ZYMFENTRA are at increased risk for developing serious infections involving various organ systems and sites that may lead to hospitalization or death.
Opportunistic infections due to bacterial, mycobacterial, invasive fungal, viral, or parasitic organisms including aspergillosis, blastomycosis, candidiasis, coccidioidomycosis, cryptococcosis, histoplasmosis, legionellosis, listeriosis, pneumocystosis, salmonellosis and tuberculosis have been reported with TNF blockers. Patients have frequently presented with disseminated rather than localized disease.
Treatment with ZYMFENTRA should not be initiated in patients with an active infection, including clinically important localized infections.Patients greater than 65 years of age, patients with comorbid-conditions and/or patients taking concomitant immunosuppressants such as corticosteroids or methotrexate may be at greater risk of infection. The risks and benefits of treatment should be considered prior to initiating therapy in patients:
- with chronic or recurrent infection;
- who have been exposed to tuberculosis;
- with a history of an opportunistic infection;
- who have resided or traveled in areas of endemic tuberculosis or endemic mycoses, such as histoplasmosis, coccidioidomycosis, or blastomycosis; or
- with underlying conditions that may predispose them to infection.
Instructions for Use
Read and follow the Instruction for Use that come with your ZYMFENTRA Prefilled Pen before you start using it and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or treatment.
Figure A: Parts of ZYMFENTRA Prefilled Pen
Important Information
- Use the Prefilled Pen only if your healthcare provider has trained you on the right way to prepare for and to give an injection.
- Ask your healthcare provider how often you will need to give an injection.
- Rotate the injection site each time you give an injection. Each new injection site should be at least 1.2 inches away from the previous injection site.
- Do not shake the Prefilled Pen at any time.
- Do not remove the Cap until you are ready to inject.
- Do not share the Prefilled Pen with anyone.
How to store the Prefilled Pen
-
Store the Prefilled Pen in a refrigerator between 36°F to 46°F (2°C to 8°C).
- Keep the Prefilled Pen in the original carton until use to protect it from light.
- Do not use the Prefilled Pen that has been left in direct sunlight.
- Do not freeze the Prefilled Pen. If the Prefilled Pen has been frozen, do not use the Prefilled Pen even if it is thawed.
- Do not warm the Prefilled Pen using heat sources such as hot water or a microwave. Let the Prefilled Pen naturally warm at room temperature between 68°F to 77°F (20°C to 25°C) for 30 minutes before giving an injection.
- When the Prefilled Pen has reached room temperature, do not put it back in the refrigerator. The Prefilled Pen must be thrown away (discarded) if not used within the 14 days.
- Keep the Prefilled Pen and all medicines out of the reach of children.
Prepare for the Injection
1. Gather the supplies for the injection.
1a. Prepare a clean, flat surface, such as a table or countertop, in a well-lit area.
1b. Remove 1 Prefilled Pen from the carton stored in your refrigerator.
Note: Return the carton with any unused Prefilled Pens to refrigerator immediately after taking out one Prefilled Pen.
1c. Make sure you have the following supplies:
- Prefilled Pen
- 1 Alcohol swab
Not included in the carton:
- 1 Cotton ball or gauze
- 1 Adhesive bandage
- FDA-cleared sharps disposal container
|
2. Inspect the Prefilled Pen. 2a. Make sure you have the correct medicine (ZYMFENTRA). 2b. Check the expiration (EXP) date on the label of the Prefilled Pen (See Figure B). 2c. Look at the Prefilled Pen and make sure it is not cracked or damaged.
|
Figure B |
|
3. Inspect the Medicine. 3a. Look through the Window and make sure that the liquid is clear, colorless to pale brown, and free of particles (see Figure C).
|
Figure C |
|
4. Wait 30 minutes. 4a. Leave the Prefilled Pen at room temperature 68°F to 77°F (20°C to 25°C) for 30 minutes to allow it to warm up (see Figure D).
|
Figure D |
|
5. Choose an injection site (see Figure E). 5a. You may inject into: - the front of your thighs. - the stomach area (abdomen) except for the 2 inches around the belly button (navel). - the outer area of the upper arm if you are a caregiver.
5b. Rotate the injection site each time you give an injection.
|
Figure E |
|
6. Wash your han 6a. Wash your hands with soap and water and dry them thoroughly (see Figure F). |
Figure F |
|
7. Clean the injection site. 7a. Clean the injection site with an alcohol swab using a circular motion (see Figure G).
|
Figure G |
|
Give the injection |
|
|
8. Remove the Cap. 8a. Hold the Prefilled Pen by the injector body with the Cap on top using one hand. Gently pull the Cap straight off with the other hand.
8b. Dispose of the Cap in an FDA cleared sharps container (see Step 12 and Figure H). |
Figure H |
|
9. Place the Prefilled Pen on the injection site. 9a. Hold the Prefilled Pen so that you can see the Window. 9b. Without pinching or stretching the skin, place the Prefilled Pen over the injection site at a 90-degree angle (see Figure I). |
Figure I |
|
10. Give the injection (see Figure J). 10a. Press the Prefilled Pen firmly against the skin.
10b. Keep holding the Prefilled Pen firmly against the skin and listen for the second loud "click." This can take up to 10 seconds.
10c. After you hear the second loud "click" continue to keep holding the Prefilled Pen firmly against the skin and count slowly to 5 to make sure you inject the full dose. 10d. Look at the Prefilled Pen and make sure that the purple Plunger Rod is filling the Window completely. |
Figure J |
|
11. Remove the Prefilled Pen from your skin. 11a. Remove the Prefilled Pen from your skin (see Figure K).
|
Figure K
Figure L |
|
After the injection |
|
|
12. Throw away (dispose of) the Prefilled Pen. 12a. Put the used Prefilled Pen in an FDA-cleared sharps disposal container right away after use (see Figure M).
|
Figure M |
|
13. Care for the injection site. 13a. Treat the injection site by gently pressing, not rubbing, a cotton ball or gauze to the site and apply an adhesive bandage, if necessary. |
|
|
Manufactured by: CELLTRION, Inc., 23, Academy-ro, Yeonsu-gu, Incheon, 22014, Republic of Korea Distributed by: CELLTRION USA, Inc., 1 Evertrust Plaza Suite 1207, Jersey City, NJ 07302 |
1 Indications and Usage
ZYMFENTRA is a tumor necrosis factor (TNF) blocker indicated in adults for maintenance treatment of:
- moderately to severely active ulcerative colitis following treatment with an infliximab product administered intravenously. (1)
- moderately to severely active Crohn’s disease following with an infliximab products administered intraneously. (1)
12 Clinical Phamacology
12.1 Mechanism of Action
Infliximab-dyyb neutralize the biological activity of TNFα by binding with high affinity to the soluble and transmembrane forms of TNFα and inhibit binding of TNFα with its receptors. Infliximab-dyyb has shown biological activities, such as TNFα neutralization activity and TNFα binding affinities, complement 1q (C1q) binding affinity and crystallizable fragment (Fc) receptor binding affinities in a wide variety of in vitro bioasays. The relationship of these biological response markers to the mechanism(s) by which infliximab-dyyb exerts its clinical effects is unknown.
5.8 Neurologic Reactions
Agents that inhibit TNF have been associated with central nervous system (CNS) manifestation of systemic vasculitis, seizure and new onset or exacerbation of clinical symptoms and/or radiographic evidence of CNS demyelinating disorders, including multiple sclerosis and optic neuritis, and peripheral demyelinating disorders, including Guillain-Barré syndrome. Avoid the use of ZYMFENTRA in patients with these neurologic disorders and consider discontinuation of ZYMFENTRA if these disorders develop.
13 Nonclinical Toxicology
5.6 Hematologic Reactions
Report of pancytopenia including aplastic anemia have been reported with TNF blocking agents. Cases of leukopenia, neutropenia, thrombocytopenia, and pancytopenia, some with a fatal outcome, have been reported in patients receiving infliximab products. Clinically significant events of neutropenia were noted in the clinical trials of ZYMFENTRA. The causal relationship to infliximab product therapy remains unclear. Although no high-risk group(s) has been identified, avoid ZYMFENTRA in patients who have ongoing or a history of significant hematologic abnormalities. Advise all patients to seek immediate medical attention if they develop signs and symptoms suggestive of blood dyscrasias or infection (e.g., persistent fever) during treatment with ZYMFENTRA. Consider discontinuation of ZYMFENTRA therapy in patients who develop significant hematologic abnormalities.
14.2 Adult Crohn's Disease
The safety and efficacy of ZYMFENTRA were assessed in a randomized, double-blind, placebo-controlled clinical studies (CD Trial I; NCT03945019) in adult subjects with moderately to severely active CD, defined as Crohn's Disease Activity Index (CDAI) score of 220 to 450 points, and a centrally-reveiwed Simplified Endoscopic Activity Score for Crohn’s Disease (SES-CD) of ≥6 points for ileal-colonic CD (or ≥4 points for isolated ileal disease).
Subjects had demonstrated an inadequate response or intolerance to treatment with corticosteroids and/or immunosuppressants. Subjects were permitted to use stable doses of oral amisosalicylates, oral corticosteroids (prednisone ≤20 mg/day or equivalent, budesonide ≤9 mg/day), CD-related antibiotics and/or immunomodulatory agents (azathioprine, 6-mercaptopurine, or methotrexate). Corticosteroid dose was tapered after Week 10.
All subjects received three intravenous induction doses of 5 mg/kg infliximab-dyyb at Week 0, 2 and 6. In order to be ranomized to treatment in CD Trial I, subjects had to be in clinical response at Week 10. Clinical response was defined as a decrease from baseline in CDAI of at least 100 points (i.e., CDAI-100 responders).
A total of 323 subjects were randomized at Week 10 in a double-blind fashion (2:1) to ZYMFENTRA 120 mg as a subcutaneous injection or placebo every 2 weeks.
At the time of randomization into thd double-blind phase (Week 10), 61% were receiving aminosalicylates, 40 % were receiving oral corticosteroids, and 32% were receiving immunomodulators including azathioprine, 6-mercaptopurine, or methotrexate.
A total of 11% of randomized subjects had prior exposure to biological products.
Subjects in the double-blind phase had a mean age of 35 years (range 18 to 75 years); 40% were female; and 91% identified as White, 4% identified as American Indian or Alaska Native, 4% identified as Asian, 0.3% as Black or African American, and 1% identified as another racial group.
The co-primary endopoints were clinical remission (based on CDAI) and endoscopic response at Week 54. Secondary endpoints included endoscopic remission, and corticosteroid-free remission at Week 54 (see Table 4).
2 Dosage and Administration
5 Warnings and Precautions
5.5 Congestive Heart Failure
Cases of worsening congestive heart failure (CHF) and new onset CHF, with and without identifiable precipitating factors (e.g., pre-existing cardiovascular disease), have been reported with TNF blockers, including infliximab products. Some of these patients have been under 50 years of age, and some cases had a fatal outcome. In several exploratory trials of other TNF blockers in the treatment of CHF, there were greater proportions of TNF-blocker-treated patients who had CHF exacerbations requiring hospitalization or increased mortality. ZYMFENTRA has not been studied in patients with a history of CHF. Avoid ZYMFENTRA in patients with CHF.
If a decision is made to administer ZYMFENTRA to patients with CHF, closely monitor patients during therapy for new or worsening symptoms of heart failure and discontinue ZYMFENTRA if symptoms appear.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of outside of the U.S with a non-U.S approved infliximab product administered subcutaneously. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Infections and infestations: cellulitis, disseminated tuberculosis, lower respiratory tract infection, pneumonia, sepsis
- Neoplasms benign, malignant and unspecified: breast cancer, gastric cancer, lung cancer
- Nervous system disorders: multiple sclerosis
- General disorders and administration site conditions: fatigue, malaise
The following additional adverse ractions have been identified during post-approval use of infliximab products administered intravenously.
- Neutropenia, agranuloxytosis (including infants exposed in utero to infliximab products), idiopathis thrombocytopenic purpura, thrombotic thrombocytopenic purpura.
- Interstitial lung disease (including pulmonary fibrosis/interstitial pneumonitis and rapidly progessive disease).
- Pericardial effusion, systemic and cutaneous vasculitis.
- Erythema multiforme, Stevens-Johnson Syndrome, toxic epidermal necrolysis, linear IgA bullous dermatosis (LABD), acute generalized exanthematous pustulosis (AGEP), new onset and worsening psoriasis (all subtypes including pustular, primarily palmoplantar), lichenoid react.
- Preipheral demyelinating disorders (such as Guillain-Barré syndrome, chronic inflammatory demyelinating polyneuropathy, and multifocal motor neuropathy) transverse myelitis, and neuropathies (additional neurologic reactions have also been observed).
- Acute liver failure, jaundice, hepatitis, and cholestasis.
- Serious infections and vaccine breakthrough infection including bovine tuberculosis (disseminated BCG infection) following vaccination in an infant exposed in utero to infliximab products.
- Malignancies, including leukemia, melanoma, Merkel cell carcinoma, and cervical cancer.
- Anaphylactic reactions, including anaphylactic shock, laryngeal/pharyngeal edema and severe bronchospasm, and seizure.
- Transient visual loss has been reported in association with infliximab products during or within 2 hours of infusion. Cerebrovascular accidents, myocardial ischemia/infarction (some fatal), and arrhythmia occurring within 24 hours of initiation of infusion have also been reported.
14.1 Adult Ulcerative Colitis
The safety and efficacy of ZYMFENTRA were assessed in a randomized, double-blind, placebo-controlled clinical trial (UC Trial I; NCT04205643) in adult subjects with moderately to severely active UC (defined as a modified Mayo score [mMS] between 5 to 9 with an endoscopic subscore [ES] of 2 or 3). The mMS is a 3-component Mayo score (0-9), which consists of following subscores (0 to 3 for each subscore): stool frequency (SFS), rectal bleeding (RBS), and findings on centrally read endoscopy score (ES). An ES of 2 was defined by marked erythema, lack of vascular pattern, any friability, and/or erosions, and a score of 3 was defined by spontaneous bleeding and ulceration.
Subjects had demonstrated an inadequate response or intolerance to treatment with corticosteroids alone or in combination with 6-mercaptopurine or azathioprine. Subjects were permitted to use stable doses of oral aminosalicylates, oral corticosteroids (prednisone ≤20 mg/day or equivalent, budesonide ≤9 mg/day), UC-related antibiotics, and/or immunomodulatory agents (azathioprine, 6-mercaptopurine, or methotrexate). Corticosteroid tapering was permitted after Week 10.
All subjects received three intravenous induction doses of 5 mg/kg of infliximab-dyyb at Weeks 0, 2 and 6. In order to be randomized to treatment in UC Trial I, subjects had to be in clinical response at Week 10. Clinical response was defined as a decrease from baseline in the mMS of at least 2 points and at least 30%, with an accompanying decrease in the RBS of at least 1 point or an aabsolute RBS of 0 or 1 point.
A total of 438 subjects were randomized at Week 10 in a double-blind fashion (2:1) to ZYMFENTRA 120 mg as a subcutaneous injection or placebo every two weeks.
At the time of randomization into the double-blind phase (Week 10), 92% were receiving aminosalicylates, 41% were receiving oral corticosteroids, and 22% were receiving immunomodulators including AZA, 6-MP or MTX.
A total of 10% of randomized subjects had prior exposure to biological poucts or JAK inhibitors.
Subjects in the boule-blind phase had a mean age of 39 years (range 18 to 75 years); 44% were female; and 98% identified as White, and 2% as American Indian or Alaska Native.
The primary endpoint was the proportion of subjects in clinical remission at Week 54. Wecondary endpoints included the proportion of subjects achieving histologic-endoscopic mucosal improvement and corticosteroid-fee remission at Week 54 (see Table 3).
8 Use in Specific Populations
3 Dosage Forms and Strengths
ZYMFENTRA (infliximab-dyyb) is a clear, colorless to pale brown solution available as follow:
- Injection: 120 mg/mL in a single-dose prefilled syringe.
- Injection: 120 mg/mL in a single-dose prefilled syringe with needle guard.
- Injection: 120 mg/mL in a single-dose prefilled pen.
6.1 Clinical Trials Experience
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
- The data described below reflect exposure to ZYMFENTRA in 518 adult subjects in two 54-weekrandomized, double-blind, placebo-controlled trials in subjects with moderately to severely active ulcerative colitis (UC) or Crohn’s disease (CD) (UC Trial I an CD Trial I). Subject who achieved clinical response following three induction doses of infliximab-dyyb administered as an intravenous infusion at Week 0, 2 and 6 were randomized 2:1 to ZYMFENTRA 120 mg or placebo as a subcutaneous injection every two weeks at Week 10 [see Clinical Studies (14.1 and 14.2)].
7.2 Cytochrome P450 Substrates
The formation of CYP450 enzymes may be suppressed by increased levels of cytokines (e.g., TNFα, IL-1, IL-6, IL-10, IFN) during chronic inflammation. Therefore, ZYMFENTRA, an antagonist of TNFα, could normalize the formation of CYP450 enzymes potentially resulting in a decrease in exposure of CYP450 substrates.
Upon initiation or discontinuation of TNF blockers, including ZYMFENTRA, in patients being treated with CYP450 substrates requiring therapeutic drug monitoring, monitor therpeutic parameters (e.g., INR for warfarin) or drug concentration (e.g., cyclosporine or theophylline). Dosage adjustment may be needed to maintain drug concentrations or parameters within the therapeutic range. See prescribing information for specific drugs.
2.1 Important Dosage Information
- ZYMFENTRA is indicated as maintenance treatment only, starting at Week 10 and thereafter.▫ All patients mush coplete an intravenous induction regimen with an infliximab product before starting ZYMFENTRA. For induction dosing information, see the corresponding full prescribing information for the chosen infliximab product.
- ZYMFENTRA is for subtutaneous use only.
5.3 Hepatitis B Virus Reactivation
Use of TNF blockers has been associated with reactivation of hepatitis B virus (HBV) in patients who are chronic carriers of this virus. In some instances, HBV reactivation occurring in conjunction with TNF blocker therapy has been fatal. The majority of these reports have occurred in patients concomitantly receiving other medications that suppress the immune system, which may also contribute to HBV reactivation.
Test patients for HBV infection before initiating TNF blocker therapy, including ZYMFENTRA. For patients who test positive for hepatitis B surface antigen, consultation with a physician with expertise in the treatment of hepatitis B is recommended. Adequate data are not available on the safety or efficacy of treating patients who are carriers of HBV with antiviral therapy in conjunction with TNF blocker therapy to prevent HBV reactivation. Patients who are carriers of HBV and require treatment with TNF blockers, including ZYMFENTRA, should be closely monitored for clinical and laboratory signs of active HBV infection throughout therapy and for several months following termination of therapy.
In patients who develop HBV reactivation, discontinue ZYMFENTRA and initiate antiviral therapy with appropriate supportive treatment. The safety of resuming TNF blocker therapy after HBV reactivation is controlled is not known. Therefore, prescribers should exercise caution when considering resumption of ZYMFENTRA in this situation and monitor patients closely.
17 Patient Counseling Information
Advise the patient or thier caregiver to read the FDA-Approved Patient Labeling (Medication Guide and Instructions for Use).
Patients or their caregivers should be advised of the potential benefits and risks of ZYMFENTRA. Healthcare providers should instruct their patients or their caregivers to read the Medication Guide before starting ZYMFENTRA therapy and to reread it each time they receive an injection.
2.3 Subcutaenous Administration Instructions
- ZYMFENTRA is intended for use under the guidance and supervision of a healthcare professional.
- If a healthcare professional determines that it is appropriate, patients may self-injection ZYMFENTRA or caregivers may injection ZYMFENTRA using either the ZYMFENTRA prefilled syringe, ZYMFENTRA prefilled syringe with needle guard, or ZYMFENTRA prefilled pen after proper training in subcutaenous injection technique.
- Parental drug products should be inspected visually for particulate matter and discoloration prior to administration, whether solution and container permit. ZYMFENTRA should be a clear, colorless to pale brwon solution. Do not use if particulates or discoloration is presnet.
- Inject into the front of the tights, the abdomen excepts for the 2 inches around the navel, or the outer area of the upper arms (caregiver only).
- Rotate the injectoin site each time an inejction is given. Allow at least 1.2 inches between the new injection site and the previous inejction site. Never inject into areas where the skin is red, bruised, tender, or indurated.
- Do not use the syringe or pen of it has been dropped or is visibly damaged. A damaged syringe may not function proprely.
- Do not resue or shake the syringe or pen at any time.
Anti Drug Antibody Effects On Pharmacokinetics
Subjects who were positive for anti-drug antibodies showed lower infliximab-dyyb trough seum concentrations of infliximab-dyyb by approximately 30 to 40% compared to subjects who were negative for anti-drug antibodies. In some subjects with high titers of anti-drug antibodies and positive neutralizing antibodies, trough seum concentrations of infliximab-dyyb were below the lower limit of quantitation (<0.1 mcg/mL). There was no identified clinically significant effect of anti-drug antibodies on the safety and effectiveness of ZYMFENTRA in Study UC I and Study CD I.
7.3 Live Vaccines/therapeutic Infectious Agents
It is recommended that live vaccines not be given concurrently with ZYMFENTRA. It is also recommended that live vaccines not be given to infants after in utero exposure to infliximab products for 6 months following birth [see Warnings and Precautions (5.12)].
It is recommended that therapeutic infectious agents not be given concurrently with ZYMFENTRA [see Warnings and Precautions (5.12)].
5.7 Hypersensitivity and Other Administration Reactions
In clinical trials of ZYMFENTRA, symptoms compatible with hypersensitivity reactions have been reported including bronchospasm, dyspnea, rash, and edema. In post-marketing experience, serious systemic hypersensitivity reactions (including anaphylaxis, hypotension, and serum sickness) have been reported following administration of infliximab products. If an anaphylactic or other clinically significant hypersensitivity reaction occurs, institute appropriate therapy and discontinue ZYMFENTRA. There are no data on the risks of using ZYMFENTRA in patients who have experienced a severe hypersensitivity reaction towards another TNF blocker; in these patients, caution is needed.
7.1 Other Biological Products Used to Treat Uc and Cd
The concurrent use of ZYMFENTRA with other immunosuppressive biological products used to treat UC and CD may increase the risk of infection and is not recommended [see Warnings and Precautions (5.9)].
Consier the half-life and mode of action of prior biologial products to avoid unintended additive immunosuppressive effects when initiating ZYMFENTRA [see Warnings and Precautions (5.10)].
Principal Display Panel 120 Mg/ml Prefilled Pen Carton
Zymfentra TM
infliximab-dyyb
Injection
120 mg/mL
FOR SUBCUTANEOUS USE ONLY
ATTENTION: Disponse the enclosed Medication Guide to each patient.
NDC 72606-025-01
Rx ony
1 prefilled pen + 2 alcohol preps
1 Single-Dose Prefilled Pen
CELLTRION USA, Inc.
Principal Display Panel 120 Mg/ml prefilled Pen Label
NDC 72606-025-01
Rx only
ZymfentraTM
infliximab–dyyb
injection
Subcutaneous use
CELLTRION Inc.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal studies of infliximab-dyyb have not been conducted to evaluate the carcinogenic potential or its effect on fertility.
Principal Display Panel 120 Mg/ml Prefilled Syringe Label
NDC 72606-025-05
Rx only
ZymfentraTM
infliximab–dyyb
injection
Subcutaneous use
CELLTRION, Inc.
Principal Display Panel 120 Mg/ml Prefilled Syringe Carton
ZymfentraTM
infliximab-dyyb
injection
120 mg/mL
FOR SUBCUTANEOUS USE ONLY
ATTENTION: Dispense the enclosed Medication Guide to each patient.
NDC x72606-025-05
Rx
1 prefilled syringe + 2 alcohol preps
1 Single-Dose Prefilled Syringe
CELLTRION USA, Inc.
5.12 Vaccinations and Use of Live Vaccines/therapeutic Infectious Agents
Principal Display Panel 120 Mg/ml Prefilled Syringe With Needle Guard Label
NDC 72606-025-09
Rx only
ZymfentraTM
infliximab–dyyb
injection
Subcutaneous use
CELLTRION, Inc.
5.10 Risk of Additive Immunosuppressive Effects From Prior Biological Products
Consider the half-life and mode of action of prior biological products to avoid unintended additive immunosuppressive effects when initiating ZYMFENTRA [see Drug Interactions (7.1)].
Principal Display Panel 120 Mg/ml Prefilled Syringe With Needle Guard Carton
ZymfentraTM
infliximab-dyyb
injection
120 mg/mL
FOR SUBCUTANEOUS USE ONLY
ATTENTION: Dispense the enclosed Medication Guide to each patient.
NDC 72606-025-09
1 prefilled syringe with needle guard
+ 2 alcohol preps
1 Single-Dose Prefilled Syringe with Needle Guard
CELLTRION USA, Inc.
5.9 Risk of Infection With Concurrent Administration With Other Biological Products
Serious infections and neutropenia have been reported with concurrent use of TNF-blockers and other immunosuppressive biological products (e.g., anakinra and abatacept). The concurrent use of ZYMFENTRA with other immunosuppressive biological products used to treat ulcerative colitis and Crohn’s disease may increase the risk of infection and is not recommended [see Drug Interactions (7.1)].
2.2 Recommeneded Dosage for Maintenance Treatment in Ulcerative Colitis Crohn's Disease
- Maintenance dosage starting at Week 10 and thereafter: 120 mg subcutaneously once every two weeks. To switch patients who are responding to maintenance therapy with an infliximab product administered intravenously, administer the first subcutaneous dose of ZYMFNETRA in place of the next scheuled intravenous infusion and every two weeks thereafter.
Structured Label Content
Recent Major Changes (34066-1)
WARNING: SERIOUS INFECTIONS and MALIGNANCY See full prescribing information for complete boxed warning Increased risk of serious infections leading to hospitalization or death, including tuberculosis (TB), bacterial sepsis, invasive fungal infections (such as hitoplasmosis) and infections due to ohter opportunistic pathogens. (5.1) Discontinue ZYMFENTRA if a patient develops a serious infection or sepsis. (5.1) Perform test for latent TB; if positive, start treatment for TB prior to starting ZYMFENTRA. Monitor all patients for active TB during treatment, even if initial latent TB test is negative. (5.1) Lymphoma and other malignancies, some fatal, have been reported in children and adolescent patients treated with tumor necrosis factor (TNF) blockers, including infliximab products. (5.2) Postmarketing cases of fatal hepatosplenic T-cell lymphoma (HSTCL) have been reported in patients treated with TNF blockers including infliximab products. Almost all had received azathioprine or 6-mercaptopurine concomitantly with a TNF blocker at or prior to diagnosis. The majority of cases were reported in patients with Crohn's disease or ulcerative colitis, most of whom were adolescent or young adult males. (5.2 )
Section 42229-5 (42229-5)
SERIOUS INFECTIONS
Patients treated with TNF blockers, inculding ZYMFENTRA, are at increased risk for developing serious infections that may lead to hospitalization or death [see Warnings and Precautions (5.1), Adverse Reactions (6.1)]. Most patients who developed these infections were taking concomitant immunosuppressants such as methotrexate or corticosteroids.
Discontinue ZYMFENTRA if a patient develops a serious infection or sepsis.
Reported infections include:
- Active tuberculosis, including reactivation of latent tuberculosis. Patients with tuberculosis have frequently presented with disseminated or extrapulmonary disease. Test patientsfor latent tuberculosis before ZYMFENTRA use and during therapy. Initiate treatment for latent infection prior to ZYMFENTRA use.
- Invasive fungal infections, including histoplasmosis, coccidioidomycosis, candidiasis, aspergillosis, blastomycosis, and pneumocystosis. Patients with histoplasmosis or other invasive fungal infections may present with disseminated, rather than localized, disease. Antigen and antibody testing for histoplasmosis may be negative in some patients with active infection. Consider empiric anti-fungal therapy in patients at risk for invasive fungal infections who develop severe systemic illness.
- Bacterial, viral and other infections due to opportunistic pathogens, including Legionella and Listeria.
Carefully consider the risks and benefits of treatment with ZYMFENTRA prior to initiating therapy in patients with chronic or recurrent infection.
Closely monitor patients for the development of signs and symtoms of infection during and after treatment with ZYMFENTRA, including the possible development of tuberculosis in patients who tested negative for latent tuberculosis infection prior to initiating therapy.
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration Revised: 02/2024 | ||
|
MEDICATION GUIDE
ZYMFENTRATM (Zim fen' trah) (infliximab-dyyb) injection, for subcutaneous use |
||
| Read the Medication Guide that comes with ZYMFENTRA before you receive the first treatment, and before each time you receive a treatment of ZYMFENTRA. This Medication Guide does not take the place of talking with your doctor about your medical condition or treatment. | ||
|
What is the most important information I should know about ZYMFENTRA? ZYMFENTRA may cause serious side effects, including:
After starting ZYMFENTRA, if you have an infection, any sign of an infection including a fever, cough, flu-like symptoms, or have open cuts or sores on your body, call your doctor right away. ZYMFENTRA can make you more likely to get infections or make any infection that you have worse.
|
||
|
What is ZYMFENTRA?
ZYMFENTRA is a prescription medicine used as an injection uner the skin (subcutaneous injection) by adults for the maintenance treatment of:
|
||
Do not take ZYMFNETRA if you:
|
||
|
Before you receive ZYMFENTRA, tell your doctor all of your medical conditions, including if you:
If you have a baby and you were receiving ZYMFENTRA during your pregnancy, it is important to tell your baby's doctor and other healthcare professionals about your ZYMFENTRA use so they can decide when your baby should receive any vaccine. Certain vaccinations can cause infections.
|
||
How should I receive ZYMFENTRA?
- If your healthcare provider decides that you or your cargiver can give your injections of ZYMFENTRA at home, you or your caregiver should be shown the right way to prepare and inject ZYMFENTRA. - Do not try to inject ZYMFENTRA yourself until you or your caregiver have been shown how to inject ZYMFENTRA by your healthcare provider. - ZYMFENTRA is injected under your skin (subcutaneously) 1 time every two weeks. - Inject ZYMFENTRA under the skin (subcutaneous injection), in your upper arms, stomach area (abdomen), or upper legs (thighs). - Do no give an injection in an area of the skin that is tender, bruised, red or hard. - Use a different injection site each time you use ZYMFENTRA. - If you are not able to inject ZYMFENTRA at your regular scheduled time or you miss a dose of ZYMFENTRA, inject the dose as soon as possible. Then, inject your next dose every two weeks thereafter. If you are not sure when to inject ZYMFENTRA, call your healthcare provider. - If you inject more than prescribed, call your doctor right away. - Be sure to keep all of your scheduled follow-up appointments. Read the detailed Instructions for Use at the end of this Medication Guide for insructions about how to prepare and inject a dose of ZYMFENTRA, and how to properly throw away (dispose of) used needles and syringes. The syringe and needle must never be re-used. After the rubber stopper is puntured, ZYMFENTRA can become contaiminated by harmful bacteria which could cause an infection if re-used. Therefore, throw away any unused portion of ZYMFENTRA. |
||
|
What should I avoid while taking ZYMFENTRA?
|
||
|
What are the possible side effects of ZYMFENTRA? ZYMFENTRA can cause serious side effects, including (see "What is the most important information I should know about ZYMFENTRA ?"): Serious Infections
|
||
|
|
|
|
||
|
|
|
|
Liver Injury Some patients receiving infliximab products have developed serious liver problems. Tell your doctor if you have: |
||
|
|
|
|
Heart Failure If you have a heart problem called congestive heart failure, your doctor should check you closely while you are receiving ZYMFENTRA. Your congestive heart failure may get worse while you are receiving ZYMFENTRA. Be sure to tell your doctor of any new or worse symptoms including: |
||
|
|
|
|
Treatment with ZYMFENTRA may need to be stopped if you get new or worse congestive heart failure. Blood Probelms In some patiens receiving infliximab products, the body may not make enough of the blood cells tha help fight infections or help stop bleeding. Tell your doctor if you: |
||
|
|
|
|
Allergic Reactions
|
||
|
|
|
|
Some patients treated with infliximab products have had delayed allergic reactions. Tell your doctor right away if you have any of these signs of delayed reaction to ZYMFENTRA: |
||
|
|
|
|
Nervous System Disorders Some patients receiving infliximab products have developed problems with their nervous system. Tell your doctor if you have: |
||
|
|
|
|
Some patients have experienced a stroke within approximately 24 hours of their infusion with infliximab products. Tell your doctor right away if you have symptoms of a stroke which may include: numbness or weakness of the face, arm, or leg, especially on one side of the body; sudden confusion, trouble speaking or understanding; sudden trouble seeing in one or both eyes, sudden trouble walking, dizziness, loss of balance or coordination or a sudden, severe headache. Lupus-like Syndrome Some patient have developed symptoms that are like the symptoms of Lupus. If you develop any of the following symptoms, your doctor may decide to stop your treatment with ZYMFENTRA: |
||
|
|
|
|
The most common side effects of ZYMFENTRA include: |
||
|
|
|
|
Tell your doctor about any side effect that bothers you or does not go away.
|
||
|
How should I store ZYMFENTRA?
|
||
|
General information about ZYMFENTRA
|
||
|
What are the ingredients in ZYMFENTRA?
|
||
| Manufactured by: CELLTRION, Inc. 23, Academy-ro, Yeonsu-gu, Incheon, 22014, Republic of Korea U.S. License No. 1996 ©CELLTRION, Inc. Distributed by CELLTRION USA, Inc. One Evertrust Plaza Suite 1207, Jersey City, New Jersey, 07302, USA For more information call 1-888-804-3433 |
8.2 Lactation
11 Description (11 DESCRIPTION)
Infliximab-dyyb, a tumor necrosis factor (TNF) blocker, is a chimeric IgG1κ monoclonal antibody (composed of human constant and murine variable regions). It has a molecular weight of approximately 145.9 kDa. Infliximab-dyyb is produced by a recombinant murine myeloma cell line, SP2/0.
ZYMFENTRA (inifliximab-dyyb) injection for subcutaneous use is a sterile, preservative-free, clear to opalescent, colorless to pale brown solution.
ZYMFENTRA is supplied in a single-dose prefilled syringe with 29 gauge fixed 1/2 inch needle, prefilled syringe with 29 gauge fixed 1/2 inch needle with needle guard, or prefilled pen with 27 gauge fixed 1/2 inch needle.
Each mL of solution contains 120 mg infliximab-dyyb, acetic acid (0.19 mg), polysorbate 80 (0.5 mg), sodium acetate (0.56 mg), sorbitol (45 mg) and Water for Injection, USP. The pH is 5.0.
Crohn's Disease
The most common adverse reactions reported in ≥3% of subjects and at a higher rate than placebo in CD Trial I are shown in Table 2.
|
Table 2 Adverse Reactionsa in the Maintenance Phase of a Randomized, Double-Blind |
||
|
ZYMFENTRA 120 mg Subcutaneous Injectionb N=222 (%) |
Placebo N=101 (%) |
|
|
COVID-19 |
10 |
5 |
|
Headache |
8 |
4 |
|
Upper respiratory track infectionsdc |
7 |
3 |
|
Injection site reactiond |
5 |
1 |
|
Diarrhea |
5 |
1 |
|
Increased blood creatine phosphokinase |
4 |
2 |
|
Arthralgia |
4 |
3 |
|
Increased alaine aninotransferase |
4 |
1 |
| Hypertensionse | 3 | 2 |
| Urinary track infectionf | 3 | 2 |
| Neutropenia | 3 | 0 |
| Dizziness | 3 | 0 |
|
Leukopenia |
3 |
0 |
|
a reported in at least 3% of ZYMFENTRA-treated subjects and at a higher rate than placebo b ZYMFENTRA 120 mg administered subcutaneously starting at Week 10 and every 2 weeks thereafter for up to Week 54. c Includes: upper respiratory tract infection, acute sinusitis, chronic sinusitis, influenza like illness, nasopharyngitis, pharyngitis, pharyngitis streptococcal, rhinitis, rhinorrhea, rhinovirus infection, sinusitis, tonsillitis d Some subjects had multiple occurrences of injection site reactions. In this table, injection site reactions are counted only once per subject. Symptoms in individual subjects included one or more of injection site bruising, edema, erythema, induration, pain, pruritus, rash, swelling, warmth. e Includes: hypertension and essential hypertension f Includes: urinary tract infection, pyelonephritis |
5.2 Malignancies
Malignancies, some fatal, have been reported among children, adolescents and young adults who received treatment with TNF blockers (initiation of therapy ≤18 years of age), including infliximab products. AApproximately half of these cases were lymphomas, including Hodgkin’s and non-Hodgkin’s lymphoma. The other cases represented a variety of malignancies, including rare malignancies that are usually associated with immunosuppression and malignancies that are not usually observed in children and adolescents. The malignancies occurred after a median of 30 months (range 1 to 84 months) after the first dose of TNF blocker therapy. Most of the patients were receiving concomitant immunosuppressants. These cases were reported postmarketing and are derived from a variety of sources, including registries and spontaneous postmarketing reports.
5.11 Autoimmunity
Treatment with TNF blockers, iincluding ZYMFENTRA may result in the formation of autoantibodies and in the development of a lupus-like syndrome. If a patient develops symptoms suggestive of a lupus-like syndrome following treatment with ZYMFENTRA, discontinue treatment.
8.4 Pediatric Use
The safety and effectiveness in pediatric patients have not been established.
8.5 Geriatirc Use
Clinical trials of ZYMFENTRA did not include sufficient numbers of sugjects aged 65 and over (8 subjects with UC and 6 subjects with CD) to determined whether they respond differently from younger subjects.
5.4 Hepatotoxicity
Hepatobiliary disorders, including acute liver failure, jaundice abnormal hepatic function, hepatic steatosis, hepatitis, hepatotoxicity, hyperbilirubinemia and non-alcoholic fatty liver, have been reported in postmarketing data in patients receiving infliximab products. Autoimmune hepatitis has been diagnosed in some of these cases. Severe hepatic reactions occurred between two weeks to more than one year after initiation of infliximab products administered intravenously; elevations in hepatic aminotransferase levels were not noted prior to discovery of the liver injury in many of these cases. Some of these cases were fatal or necessitated liver transplantation.
In clinical trials, three subjects treated with ZYMFENTRA had drug induced liver injury based on hepatic transaminase elevations, including one subject with accompanying bilirubin elevation [see Adverse Reactions (6.2)].
Monitor hepatic enzymes and liver function tests every 3 to 4 months during treatment with ZYMFENTRA. Prompt investigation of the cause of liver enzyme elevation should be undertaken to identify potential cases of drug-induced liver injury. Interrupt treatment if drug-induced liver injury is suspected, until this diagnosis is excluded. Instruct patients to seek immediate medical attention if they experience symptoms suggestive of hepatic dysfuction.
Ulcerative Colitis
The most common adverse reactions reported in ≥3% of subjects and at a higher rate than Placebo in UC Trial I are shown in Table 1.
|
Table 1 Adverse Reactionsa in the Maintenance Phase of a Randomized, Double-Blind 54-Week Study of Subjects with UC (UC Tral I) |
||
|
ZYMFENTRA 120 mg Subcutaneous Injectionb N=296 (%) |
Placebo N=140 (%) |
|
|
COVID-19 |
10 |
6 |
|
Anemiad |
5 |
4 |
|
Arthralgia |
4 |
1 |
| Injection site reaction# | 3 | 2 |
|
Increased alanine aminotransferase |
3 |
1 |
|
Abominal painf |
3 |
1 |
|
areported in at least 3% of ZYMFENTRA-treated subjects and at a higher than placebo bZYMFENTRA 120 mg as a subcutaneous injection every two weeks starting Week 10 following 3 intravenous induction doses of inflixiab-dyyb cIncludes: COVID-19 and COVID-19 pneumonia dIncludes: anemia and iron deficiency anemia #Some subjects had multiple occurences of injection site reactions. In this table, injection site reactions are counted only once per subject. Symptoms in individual subjects included one or more of injection site bruising, edema, erythema, induration, pain, pruritus and swelling. fIncludes: abdominal pain, upper abdominal pain, lower abdominal pain, and abdominal discomfort |
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Difference in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of infliximab-dyyb or of other infliximab products.
In UC and CD clinical studies (UC Trial I and CD Trial I), approximately 64% subjects developed anti-drug antibodies to infliximab-dyyb following induction treatment with infliximab-dyyb intravenously and maintenance treatment with ZYMFENTRA by Week 54 [183/287 (64%) in subjects with UC, and 151/232 (65%) in subjects with CD] [see Clinical Studies (14)]. Among subjects with anti-drug antibodies, 92% had neutralizing antibodies [161/183 (88%) subjects with UC, and 147/151 (97%) subjects with CD]. Use of concomitant immunosuppressant agents (azathioprine, 6-mercaptopurine and methotrexate) appeared to reduce the frequency of ADA to infliximab-dyyb.
14 Clinical Studies (14 CLINICAL STUDIES)
4 Contraindications (4 CONTRAINDICATIONS)
ZYMFENTRA is contraindicated in patients with a history of a severe hypersensitivity reaction to infliximab-dyyb, other infliximab products, any of the inactive ingredients in ZYMFENTRA, or any murine proteins. Reactions have included anaphylaxis [see Warnings and Precautions (5.7)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Serious infections [see Warnings and Precautions (5.1)]
- Malignancies [see Warnings and Precautions (5.2)]
- Hepatitis B virus reactivation [see Warnings and Precautions (5.3)]
- Hepatotoxicity [see Warning and Precautions (5.4)]
- Congestive heart failure [see Warnings and Precautions (5.5)]
- Hematologic reactions [see Warnings and Precautions (5.6)]
- Hypersensitivity and other administration reactions [see Warnings and Precautions (5.7)]
- Neruologic reactions [see Warnings and Precautions (5.8)]
- Autoimmunity [see Warnings and Precautions (5.11)]
7 Drug Interactions (7 DRUG INTERACTIONS)
12.3 Phamacokinetics
Following single and multiple subcutaneous dosing of infliximab-dyyb, exposures to infliximab-dyyb (i.e., AUC) are proportionally increased over the dose range from 120 mg to 240 mg (2 times the recommended dosage). After a single subcutaneous dose of infliximab-dyyb 120 mg in healthy subjects, the median (SD) Cmax and AUCinf were 10.0 (3.2) mcg/mL and 6945.6 (2830.2) mcg·h/mL, respectively. Following recommended subcutaneous maintenance dose of ZYMFENTRA (120 mg every 2 weeks) in adult subjects with UC and CD from Week 10 after intravenous induction treatment with infliximab-dyyb, steady-state was achieved by Week 22, and the mean (SD) trough serum concentrations of infliximab-dyyb at steady-state were 14.6 (7.8) mcg/mL and 14.6 (8.9) mcg/mL in subjects with UC and CD, respectively. Pharmacokinetics are comparable between healthy subjects, and subjects with UC or CD.
Instructions for Use (INSTRUCTIONS FOR USE)
For subcutaneous use only
Read and follow Instructions for Use that come with your ZYMFENTRA Prefilled Syringe before using it and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or treatment.
Figure A: Parts of ZYMFENTRA Prefilled Syringe
Caution! Do not remove the Cap until you are ready to inject. When you remove the Cap, do not recap the Prefilled Syringe.
Important Information
- Use the Prefilled Syringe only if your healthcare provider has trained you on the right way to prepare for and to give an injection.
- Ask your healthcare provider how often you will need to give an injection.
- Rotate the injection site each time you give an injection. Each new injection site should be at least 1.2 inches away from the previous injection site.
- Do not use Prefilled Syringe if it has been dropped or is visibly damaged.
- Do not reuse the Prefilled Syringe.
- Do not shake the Prefilled Syringe at any time.
- Do not share the Prefilled Syringe with anyone.
- Do not pull back on the plunger rod at any time.
- Only use each Prefilled Syringe for one injection.
How to store the Prefilled Syringe
- Store the Prefilled Syringe in a refrigerator between 36°F to 46°F (2°C to 8°C).
- Keep the Prefilled Syringe in the original carton to protect it from light.
- Do not use the Prefilled Syringe that has been left in direct sunlight.
- Do not freeze the Prefilled Syringe. If the Prefilled Pen has been frozen, do not use the Prefilled Syringe even if it is thawed.
- Do not warm the Prefilled Syringe using heat sources such as hot water or a microwave. Let the Prefilled Syringe naturally warm at room temperature between 68°F to 77°F (20°C to 25°C) for 30 minutes before giving an injection.
- When the Prefilled Syringe has reached room temperature, do not put it back in the refrigerator. The Prefilled Syringe must be thrown away (discarded) if not used within the 14 days.
- Keep the Prefilled Syringe and all medicines out of the reach of children.
Prepare for the Injection
1. Gather the supplies for the injection.
1a. Prepare a clean, flat surface, such as a table or countertop, in a well-lit area.
1b. Remove 1 Prefilled Syringe from the carton by holding the middle of the Prefilled Syringe Body.
- Do not touch the plunger rod.
Note: Return the carton with any unused Prefilled Syringe to the refrigerator immediately after taking out one Prefilled Syringe.
1c. Make sure you have the following supplies:
- Prefilled Syringe
- Alcohol swab
Not included in the carton:
- Cotton ball or gauze
- Adhesive bandage
- FDA-cleared sharps disposal container
|
2. Inspect the Prefilled Syringe. 2a. Make sure you have the correct medicine (ZYMFENTRA). 2b. Check the expiration (EXP) date on label of the Prefilled Syringe (See Figure B). 2c. Look at the Prefilled Syringe and make sure it is not cracked or damaged.
|
Figure B |
|
3. Inspect the Medicine. 3a. Look at the Prefilled Syringe Body and check the liquid is clear and colorless to pale brown (see Figure C).
|
Figure C |
|
4. Wait 30 minutes. 4a. Leave the Prefilled Syringe at room temperature 68°F to 77°F (20°C to 25°C) for 30 minutes to allow it to warm up (see Figure D).
|
Figure D |
|
5. Choose an injection site (see Figure E). 5a. You may inject into: - the front of your thighs. - the stomach area (abdomen) except for the 2 inches around the belly button (navel). - the outer area of the upper arm (caregiver only).
5b. Rotate the injection site each time you give an injection.
|
Figure E |
|
6. Wash your hands. 6a. Wash your hands with soap and water and dry them thoroughly (see Figure F). |
Figure F |
|
7. Clean the injection site. 7a. Clean the injection site with an alcohol swab using a circular motion (see Figure G).
|
Figure G |
|
Give the injection |
|
|
8. Remove the Cap. 8a. Hold the Prefilled Syringe by the injector body with the Cap on top using one hand. Gently pull the Cap straight off with the other hand.
8b. Throw away (dispose of) the Cap in an FDA cleared sharps container (see Step 12 and Figure H). |
Figure H |
|
9. Insert the Prefilled Syringe into the injection site. 9a. Hold the Prefilled Syringe by its body in one hand between your thumb and index finger. 9b. Gently pinch a fold of skin at the injection site with one hand. 9c. With a quick and “dart-like” motion, insert the Needle completely into the fold of the skin at a 45-defree angle (see Figure I).
|
Figure I |
|
10. Give the injection. 10a. After the Needle is inserted, release the pinched skin. 10b. Push the Plunger down slowly and as far as it will go until the Prefilled Syringe is empty (See Figure J). |
Figure J |
|
11. Remove the Prefilled Syringe from the injection site. 11a. Remove the Needle from your skin at the same angle it was inserted (see Figure K).
|
Figure K |
|
After the injection |
|
|
12. Throw away (dispose of) the Prefilled Syringe. 12a. Put the used Prefilled Syringe in an FDA-cleared sharps disposal container right away after use (see Figure L).
|
Figure L |
|
13. Care for the injection site. 13a. Treat the injection site by gently pressing a cotton ball or gauze to the site and apply an adhesive bandage, if necessary. |
|
|
Manufactured by: CELLTRION, Inc., 23, Academy-ro, Yeonsu-gu, Incheon, 22014, Republic of Korea Distributed by: CELLTRION USA, Inc., 1 Evertrust Plaza Suite 1207, Jersey City, NJ 07302 |
12.2 Pharmacodynamics
No formal pharmacodynamic studies have been conducted.
5.1 Serious Infections
Patients treated with ZYMFENTRA are at increased risk for developing serious infections involving various organ systems and sites that may lead to hospitalization or death.
Opportunistic infections due to bacterial, mycobacterial, invasive fungal, viral, or parasitic organisms including aspergillosis, blastomycosis, candidiasis, coccidioidomycosis, cryptococcosis, histoplasmosis, legionellosis, listeriosis, pneumocystosis, salmonellosis and tuberculosis have been reported with TNF blockers. Patients have frequently presented with disseminated rather than localized disease.
Treatment with ZYMFENTRA should not be initiated in patients with an active infection, including clinically important localized infections.Patients greater than 65 years of age, patients with comorbid-conditions and/or patients taking concomitant immunosuppressants such as corticosteroids or methotrexate may be at greater risk of infection. The risks and benefits of treatment should be considered prior to initiating therapy in patients:
- with chronic or recurrent infection;
- who have been exposed to tuberculosis;
- with a history of an opportunistic infection;
- who have resided or traveled in areas of endemic tuberculosis or endemic mycoses, such as histoplasmosis, coccidioidomycosis, or blastomycosis; or
- with underlying conditions that may predispose them to infection.
Instructions for Use (INSTRUCTIONS FOR USE )
Read and follow the Instruction for Use that come with your ZYMFENTRA Prefilled Pen before you start using it and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or treatment.
Figure A: Parts of ZYMFENTRA Prefilled Pen
Important Information
- Use the Prefilled Pen only if your healthcare provider has trained you on the right way to prepare for and to give an injection.
- Ask your healthcare provider how often you will need to give an injection.
- Rotate the injection site each time you give an injection. Each new injection site should be at least 1.2 inches away from the previous injection site.
- Do not shake the Prefilled Pen at any time.
- Do not remove the Cap until you are ready to inject.
- Do not share the Prefilled Pen with anyone.
How to store the Prefilled Pen
-
Store the Prefilled Pen in a refrigerator between 36°F to 46°F (2°C to 8°C).
- Keep the Prefilled Pen in the original carton until use to protect it from light.
- Do not use the Prefilled Pen that has been left in direct sunlight.
- Do not freeze the Prefilled Pen. If the Prefilled Pen has been frozen, do not use the Prefilled Pen even if it is thawed.
- Do not warm the Prefilled Pen using heat sources such as hot water or a microwave. Let the Prefilled Pen naturally warm at room temperature between 68°F to 77°F (20°C to 25°C) for 30 minutes before giving an injection.
- When the Prefilled Pen has reached room temperature, do not put it back in the refrigerator. The Prefilled Pen must be thrown away (discarded) if not used within the 14 days.
- Keep the Prefilled Pen and all medicines out of the reach of children.
Prepare for the Injection
1. Gather the supplies for the injection.
1a. Prepare a clean, flat surface, such as a table or countertop, in a well-lit area.
1b. Remove 1 Prefilled Pen from the carton stored in your refrigerator.
Note: Return the carton with any unused Prefilled Pens to refrigerator immediately after taking out one Prefilled Pen.
1c. Make sure you have the following supplies:
- Prefilled Pen
- 1 Alcohol swab
Not included in the carton:
- 1 Cotton ball or gauze
- 1 Adhesive bandage
- FDA-cleared sharps disposal container
|
2. Inspect the Prefilled Pen. 2a. Make sure you have the correct medicine (ZYMFENTRA). 2b. Check the expiration (EXP) date on the label of the Prefilled Pen (See Figure B). 2c. Look at the Prefilled Pen and make sure it is not cracked or damaged.
|
Figure B |
|
3. Inspect the Medicine. 3a. Look through the Window and make sure that the liquid is clear, colorless to pale brown, and free of particles (see Figure C).
|
Figure C |
|
4. Wait 30 minutes. 4a. Leave the Prefilled Pen at room temperature 68°F to 77°F (20°C to 25°C) for 30 minutes to allow it to warm up (see Figure D).
|
Figure D |
|
5. Choose an injection site (see Figure E). 5a. You may inject into: - the front of your thighs. - the stomach area (abdomen) except for the 2 inches around the belly button (navel). - the outer area of the upper arm if you are a caregiver.
5b. Rotate the injection site each time you give an injection.
|
Figure E |
|
6. Wash your han 6a. Wash your hands with soap and water and dry them thoroughly (see Figure F). |
Figure F |
|
7. Clean the injection site. 7a. Clean the injection site with an alcohol swab using a circular motion (see Figure G).
|
Figure G |
|
Give the injection |
|
|
8. Remove the Cap. 8a. Hold the Prefilled Pen by the injector body with the Cap on top using one hand. Gently pull the Cap straight off with the other hand.
8b. Dispose of the Cap in an FDA cleared sharps container (see Step 12 and Figure H). |
Figure H |
|
9. Place the Prefilled Pen on the injection site. 9a. Hold the Prefilled Pen so that you can see the Window. 9b. Without pinching or stretching the skin, place the Prefilled Pen over the injection site at a 90-degree angle (see Figure I). |
Figure I |
|
10. Give the injection (see Figure J). 10a. Press the Prefilled Pen firmly against the skin.
10b. Keep holding the Prefilled Pen firmly against the skin and listen for the second loud "click." This can take up to 10 seconds.
10c. After you hear the second loud "click" continue to keep holding the Prefilled Pen firmly against the skin and count slowly to 5 to make sure you inject the full dose. 10d. Look at the Prefilled Pen and make sure that the purple Plunger Rod is filling the Window completely. |
Figure J |
|
11. Remove the Prefilled Pen from your skin. 11a. Remove the Prefilled Pen from your skin (see Figure K).
|
Figure K
Figure L |
|
After the injection |
|
|
12. Throw away (dispose of) the Prefilled Pen. 12a. Put the used Prefilled Pen in an FDA-cleared sharps disposal container right away after use (see Figure M).
|
Figure M |
|
13. Care for the injection site. 13a. Treat the injection site by gently pressing, not rubbing, a cotton ball or gauze to the site and apply an adhesive bandage, if necessary. |
|
|
Manufactured by: CELLTRION, Inc., 23, Academy-ro, Yeonsu-gu, Incheon, 22014, Republic of Korea Distributed by: CELLTRION USA, Inc., 1 Evertrust Plaza Suite 1207, Jersey City, NJ 07302 |
1 Indications and Usage (1 INDICATIONS AND USAGE)
ZYMFENTRA is a tumor necrosis factor (TNF) blocker indicated in adults for maintenance treatment of:
- moderately to severely active ulcerative colitis following treatment with an infliximab product administered intravenously. (1)
- moderately to severely active Crohn’s disease following with an infliximab products administered intraneously. (1)
12 Clinical Phamacology (12 CLINICAL PHAMACOLOGY)
12.1 Mechanism of Action
Infliximab-dyyb neutralize the biological activity of TNFα by binding with high affinity to the soluble and transmembrane forms of TNFα and inhibit binding of TNFα with its receptors. Infliximab-dyyb has shown biological activities, such as TNFα neutralization activity and TNFα binding affinities, complement 1q (C1q) binding affinity and crystallizable fragment (Fc) receptor binding affinities in a wide variety of in vitro bioasays. The relationship of these biological response markers to the mechanism(s) by which infliximab-dyyb exerts its clinical effects is unknown.
5.8 Neurologic Reactions
Agents that inhibit TNF have been associated with central nervous system (CNS) manifestation of systemic vasculitis, seizure and new onset or exacerbation of clinical symptoms and/or radiographic evidence of CNS demyelinating disorders, including multiple sclerosis and optic neuritis, and peripheral demyelinating disorders, including Guillain-Barré syndrome. Avoid the use of ZYMFENTRA in patients with these neurologic disorders and consider discontinuation of ZYMFENTRA if these disorders develop.
13 Nonclinical Toxicology (13 NONCLINICAL TOXICOLOGY)
5.6 Hematologic Reactions
Report of pancytopenia including aplastic anemia have been reported with TNF blocking agents. Cases of leukopenia, neutropenia, thrombocytopenia, and pancytopenia, some with a fatal outcome, have been reported in patients receiving infliximab products. Clinically significant events of neutropenia were noted in the clinical trials of ZYMFENTRA. The causal relationship to infliximab product therapy remains unclear. Although no high-risk group(s) has been identified, avoid ZYMFENTRA in patients who have ongoing or a history of significant hematologic abnormalities. Advise all patients to seek immediate medical attention if they develop signs and symptoms suggestive of blood dyscrasias or infection (e.g., persistent fever) during treatment with ZYMFENTRA. Consider discontinuation of ZYMFENTRA therapy in patients who develop significant hematologic abnormalities.
14.2 Adult Crohn's Disease
The safety and efficacy of ZYMFENTRA were assessed in a randomized, double-blind, placebo-controlled clinical studies (CD Trial I; NCT03945019) in adult subjects with moderately to severely active CD, defined as Crohn's Disease Activity Index (CDAI) score of 220 to 450 points, and a centrally-reveiwed Simplified Endoscopic Activity Score for Crohn’s Disease (SES-CD) of ≥6 points for ileal-colonic CD (or ≥4 points for isolated ileal disease).
Subjects had demonstrated an inadequate response or intolerance to treatment with corticosteroids and/or immunosuppressants. Subjects were permitted to use stable doses of oral amisosalicylates, oral corticosteroids (prednisone ≤20 mg/day or equivalent, budesonide ≤9 mg/day), CD-related antibiotics and/or immunomodulatory agents (azathioprine, 6-mercaptopurine, or methotrexate). Corticosteroid dose was tapered after Week 10.
All subjects received three intravenous induction doses of 5 mg/kg infliximab-dyyb at Week 0, 2 and 6. In order to be ranomized to treatment in CD Trial I, subjects had to be in clinical response at Week 10. Clinical response was defined as a decrease from baseline in CDAI of at least 100 points (i.e., CDAI-100 responders).
A total of 323 subjects were randomized at Week 10 in a double-blind fashion (2:1) to ZYMFENTRA 120 mg as a subcutaneous injection or placebo every 2 weeks.
At the time of randomization into thd double-blind phase (Week 10), 61% were receiving aminosalicylates, 40 % were receiving oral corticosteroids, and 32% were receiving immunomodulators including azathioprine, 6-mercaptopurine, or methotrexate.
A total of 11% of randomized subjects had prior exposure to biological products.
Subjects in the double-blind phase had a mean age of 35 years (range 18 to 75 years); 40% were female; and 91% identified as White, 4% identified as American Indian or Alaska Native, 4% identified as Asian, 0.3% as Black or African American, and 1% identified as another racial group.
The co-primary endopoints were clinical remission (based on CDAI) and endoscopic response at Week 54. Secondary endpoints included endoscopic remission, and corticosteroid-free remission at Week 54 (see Table 4).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS )
5.5 Congestive Heart Failure
Cases of worsening congestive heart failure (CHF) and new onset CHF, with and without identifiable precipitating factors (e.g., pre-existing cardiovascular disease), have been reported with TNF blockers, including infliximab products. Some of these patients have been under 50 years of age, and some cases had a fatal outcome. In several exploratory trials of other TNF blockers in the treatment of CHF, there were greater proportions of TNF-blocker-treated patients who had CHF exacerbations requiring hospitalization or increased mortality. ZYMFENTRA has not been studied in patients with a history of CHF. Avoid ZYMFENTRA in patients with CHF.
If a decision is made to administer ZYMFENTRA to patients with CHF, closely monitor patients during therapy for new or worsening symptoms of heart failure and discontinue ZYMFENTRA if symptoms appear.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of outside of the U.S with a non-U.S approved infliximab product administered subcutaneously. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Infections and infestations: cellulitis, disseminated tuberculosis, lower respiratory tract infection, pneumonia, sepsis
- Neoplasms benign, malignant and unspecified: breast cancer, gastric cancer, lung cancer
- Nervous system disorders: multiple sclerosis
- General disorders and administration site conditions: fatigue, malaise
The following additional adverse ractions have been identified during post-approval use of infliximab products administered intravenously.
- Neutropenia, agranuloxytosis (including infants exposed in utero to infliximab products), idiopathis thrombocytopenic purpura, thrombotic thrombocytopenic purpura.
- Interstitial lung disease (including pulmonary fibrosis/interstitial pneumonitis and rapidly progessive disease).
- Pericardial effusion, systemic and cutaneous vasculitis.
- Erythema multiforme, Stevens-Johnson Syndrome, toxic epidermal necrolysis, linear IgA bullous dermatosis (LABD), acute generalized exanthematous pustulosis (AGEP), new onset and worsening psoriasis (all subtypes including pustular, primarily palmoplantar), lichenoid react.
- Preipheral demyelinating disorders (such as Guillain-Barré syndrome, chronic inflammatory demyelinating polyneuropathy, and multifocal motor neuropathy) transverse myelitis, and neuropathies (additional neurologic reactions have also been observed).
- Acute liver failure, jaundice, hepatitis, and cholestasis.
- Serious infections and vaccine breakthrough infection including bovine tuberculosis (disseminated BCG infection) following vaccination in an infant exposed in utero to infliximab products.
- Malignancies, including leukemia, melanoma, Merkel cell carcinoma, and cervical cancer.
- Anaphylactic reactions, including anaphylactic shock, laryngeal/pharyngeal edema and severe bronchospasm, and seizure.
- Transient visual loss has been reported in association with infliximab products during or within 2 hours of infusion. Cerebrovascular accidents, myocardial ischemia/infarction (some fatal), and arrhythmia occurring within 24 hours of initiation of infusion have also been reported.
14.1 Adult Ulcerative Colitis
The safety and efficacy of ZYMFENTRA were assessed in a randomized, double-blind, placebo-controlled clinical trial (UC Trial I; NCT04205643) in adult subjects with moderately to severely active UC (defined as a modified Mayo score [mMS] between 5 to 9 with an endoscopic subscore [ES] of 2 or 3). The mMS is a 3-component Mayo score (0-9), which consists of following subscores (0 to 3 for each subscore): stool frequency (SFS), rectal bleeding (RBS), and findings on centrally read endoscopy score (ES). An ES of 2 was defined by marked erythema, lack of vascular pattern, any friability, and/or erosions, and a score of 3 was defined by spontaneous bleeding and ulceration.
Subjects had demonstrated an inadequate response or intolerance to treatment with corticosteroids alone or in combination with 6-mercaptopurine or azathioprine. Subjects were permitted to use stable doses of oral aminosalicylates, oral corticosteroids (prednisone ≤20 mg/day or equivalent, budesonide ≤9 mg/day), UC-related antibiotics, and/or immunomodulatory agents (azathioprine, 6-mercaptopurine, or methotrexate). Corticosteroid tapering was permitted after Week 10.
All subjects received three intravenous induction doses of 5 mg/kg of infliximab-dyyb at Weeks 0, 2 and 6. In order to be randomized to treatment in UC Trial I, subjects had to be in clinical response at Week 10. Clinical response was defined as a decrease from baseline in the mMS of at least 2 points and at least 30%, with an accompanying decrease in the RBS of at least 1 point or an aabsolute RBS of 0 or 1 point.
A total of 438 subjects were randomized at Week 10 in a double-blind fashion (2:1) to ZYMFENTRA 120 mg as a subcutaneous injection or placebo every two weeks.
At the time of randomization into the double-blind phase (Week 10), 92% were receiving aminosalicylates, 41% were receiving oral corticosteroids, and 22% were receiving immunomodulators including AZA, 6-MP or MTX.
A total of 10% of randomized subjects had prior exposure to biological poucts or JAK inhibitors.
Subjects in the boule-blind phase had a mean age of 39 years (range 18 to 75 years); 44% were female; and 98% identified as White, and 2% as American Indian or Alaska Native.
The primary endpoint was the proportion of subjects in clinical remission at Week 54. Wecondary endpoints included the proportion of subjects achieving histologic-endoscopic mucosal improvement and corticosteroid-fee remission at Week 54 (see Table 3).
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS )
ZYMFENTRA (infliximab-dyyb) is a clear, colorless to pale brown solution available as follow:
- Injection: 120 mg/mL in a single-dose prefilled syringe.
- Injection: 120 mg/mL in a single-dose prefilled syringe with needle guard.
- Injection: 120 mg/mL in a single-dose prefilled pen.
6.1 Clinical Trials Experience
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
- The data described below reflect exposure to ZYMFENTRA in 518 adult subjects in two 54-weekrandomized, double-blind, placebo-controlled trials in subjects with moderately to severely active ulcerative colitis (UC) or Crohn’s disease (CD) (UC Trial I an CD Trial I). Subject who achieved clinical response following three induction doses of infliximab-dyyb administered as an intravenous infusion at Week 0, 2 and 6 were randomized 2:1 to ZYMFENTRA 120 mg or placebo as a subcutaneous injection every two weeks at Week 10 [see Clinical Studies (14.1 and 14.2)].
7.2 Cytochrome P450 Substrates
The formation of CYP450 enzymes may be suppressed by increased levels of cytokines (e.g., TNFα, IL-1, IL-6, IL-10, IFN) during chronic inflammation. Therefore, ZYMFENTRA, an antagonist of TNFα, could normalize the formation of CYP450 enzymes potentially resulting in a decrease in exposure of CYP450 substrates.
Upon initiation or discontinuation of TNF blockers, including ZYMFENTRA, in patients being treated with CYP450 substrates requiring therapeutic drug monitoring, monitor therpeutic parameters (e.g., INR for warfarin) or drug concentration (e.g., cyclosporine or theophylline). Dosage adjustment may be needed to maintain drug concentrations or parameters within the therapeutic range. See prescribing information for specific drugs.
2.1 Important Dosage Information
- ZYMFENTRA is indicated as maintenance treatment only, starting at Week 10 and thereafter.▫ All patients mush coplete an intravenous induction regimen with an infliximab product before starting ZYMFENTRA. For induction dosing information, see the corresponding full prescribing information for the chosen infliximab product.
- ZYMFENTRA is for subtutaneous use only.
5.3 Hepatitis B Virus Reactivation
Use of TNF blockers has been associated with reactivation of hepatitis B virus (HBV) in patients who are chronic carriers of this virus. In some instances, HBV reactivation occurring in conjunction with TNF blocker therapy has been fatal. The majority of these reports have occurred in patients concomitantly receiving other medications that suppress the immune system, which may also contribute to HBV reactivation.
Test patients for HBV infection before initiating TNF blocker therapy, including ZYMFENTRA. For patients who test positive for hepatitis B surface antigen, consultation with a physician with expertise in the treatment of hepatitis B is recommended. Adequate data are not available on the safety or efficacy of treating patients who are carriers of HBV with antiviral therapy in conjunction with TNF blocker therapy to prevent HBV reactivation. Patients who are carriers of HBV and require treatment with TNF blockers, including ZYMFENTRA, should be closely monitored for clinical and laboratory signs of active HBV infection throughout therapy and for several months following termination of therapy.
In patients who develop HBV reactivation, discontinue ZYMFENTRA and initiate antiviral therapy with appropriate supportive treatment. The safety of resuming TNF blocker therapy after HBV reactivation is controlled is not known. Therefore, prescribers should exercise caution when considering resumption of ZYMFENTRA in this situation and monitor patients closely.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient or thier caregiver to read the FDA-Approved Patient Labeling (Medication Guide and Instructions for Use).
Patients or their caregivers should be advised of the potential benefits and risks of ZYMFENTRA. Healthcare providers should instruct their patients or their caregivers to read the Medication Guide before starting ZYMFENTRA therapy and to reread it each time they receive an injection.
2.3 Subcutaenous Administration Instructions
- ZYMFENTRA is intended for use under the guidance and supervision of a healthcare professional.
- If a healthcare professional determines that it is appropriate, patients may self-injection ZYMFENTRA or caregivers may injection ZYMFENTRA using either the ZYMFENTRA prefilled syringe, ZYMFENTRA prefilled syringe with needle guard, or ZYMFENTRA prefilled pen after proper training in subcutaenous injection technique.
- Parental drug products should be inspected visually for particulate matter and discoloration prior to administration, whether solution and container permit. ZYMFENTRA should be a clear, colorless to pale brwon solution. Do not use if particulates or discoloration is presnet.
- Inject into the front of the tights, the abdomen excepts for the 2 inches around the navel, or the outer area of the upper arms (caregiver only).
- Rotate the injectoin site each time an inejction is given. Allow at least 1.2 inches between the new injection site and the previous inejction site. Never inject into areas where the skin is red, bruised, tender, or indurated.
- Do not use the syringe or pen of it has been dropped or is visibly damaged. A damaged syringe may not function proprely.
- Do not resue or shake the syringe or pen at any time.
Anti Drug Antibody Effects On Pharmacokinetics (Anti-Drug Antibody Effects on Pharmacokinetics)
Subjects who were positive for anti-drug antibodies showed lower infliximab-dyyb trough seum concentrations of infliximab-dyyb by approximately 30 to 40% compared to subjects who were negative for anti-drug antibodies. In some subjects with high titers of anti-drug antibodies and positive neutralizing antibodies, trough seum concentrations of infliximab-dyyb were below the lower limit of quantitation (<0.1 mcg/mL). There was no identified clinically significant effect of anti-drug antibodies on the safety and effectiveness of ZYMFENTRA in Study UC I and Study CD I.
7.3 Live Vaccines/therapeutic Infectious Agents (7.3 Live Vaccines/Therapeutic Infectious Agents)
It is recommended that live vaccines not be given concurrently with ZYMFENTRA. It is also recommended that live vaccines not be given to infants after in utero exposure to infliximab products for 6 months following birth [see Warnings and Precautions (5.12)].
It is recommended that therapeutic infectious agents not be given concurrently with ZYMFENTRA [see Warnings and Precautions (5.12)].
5.7 Hypersensitivity and Other Administration Reactions
In clinical trials of ZYMFENTRA, symptoms compatible with hypersensitivity reactions have been reported including bronchospasm, dyspnea, rash, and edema. In post-marketing experience, serious systemic hypersensitivity reactions (including anaphylaxis, hypotension, and serum sickness) have been reported following administration of infliximab products. If an anaphylactic or other clinically significant hypersensitivity reaction occurs, institute appropriate therapy and discontinue ZYMFENTRA. There are no data on the risks of using ZYMFENTRA in patients who have experienced a severe hypersensitivity reaction towards another TNF blocker; in these patients, caution is needed.
7.1 Other Biological Products Used to Treat Uc and Cd (7.1 Other Biological Products Used to Treat UC and CD )
The concurrent use of ZYMFENTRA with other immunosuppressive biological products used to treat UC and CD may increase the risk of infection and is not recommended [see Warnings and Precautions (5.9)].
Consier the half-life and mode of action of prior biologial products to avoid unintended additive immunosuppressive effects when initiating ZYMFENTRA [see Warnings and Precautions (5.10)].
Principal Display Panel 120 Mg/ml Prefilled Pen Carton (PRINCIPAL DISPLAY PANEL - 120 mg/mL Prefilled Pen Carton)
Zymfentra TM
infliximab-dyyb
Injection
120 mg/mL
FOR SUBCUTANEOUS USE ONLY
ATTENTION: Disponse the enclosed Medication Guide to each patient.
NDC 72606-025-01
Rx ony
1 prefilled pen + 2 alcohol preps
1 Single-Dose Prefilled Pen
CELLTRION USA, Inc.
Principal Display Panel 120 Mg/ml prefilled Pen Label (PRINCIPAL DISPLAY PANEL - 120 mg/mL Prefilled Pen Label)
NDC 72606-025-01
Rx only
ZymfentraTM
infliximab–dyyb
injection
Subcutaneous use
CELLTRION Inc.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal studies of infliximab-dyyb have not been conducted to evaluate the carcinogenic potential or its effect on fertility.
Principal Display Panel 120 Mg/ml Prefilled Syringe Label (PRINCIPAL DISPLAY PANEL - 120 mg/mL Prefilled Syringe Label)
NDC 72606-025-05
Rx only
ZymfentraTM
infliximab–dyyb
injection
Subcutaneous use
CELLTRION, Inc.
Principal Display Panel 120 Mg/ml Prefilled Syringe Carton (PRINCIPAL DISPLAY PANEL - 120 mg/mL Prefilled Syringe Carton)
ZymfentraTM
infliximab-dyyb
injection
120 mg/mL
FOR SUBCUTANEOUS USE ONLY
ATTENTION: Dispense the enclosed Medication Guide to each patient.
NDC x72606-025-05
Rx
1 prefilled syringe + 2 alcohol preps
1 Single-Dose Prefilled Syringe
CELLTRION USA, Inc.
5.12 Vaccinations and Use of Live Vaccines/therapeutic Infectious Agents (5.12 Vaccinations and Use of Live Vaccines/Therapeutic Infectious Agents)
Principal Display Panel 120 Mg/ml Prefilled Syringe With Needle Guard Label (PRINCIPAL DISPLAY PANEL - 120 mg/mL Prefilled Syringe with Needle Guard Label)
NDC 72606-025-09
Rx only
ZymfentraTM
infliximab–dyyb
injection
Subcutaneous use
CELLTRION, Inc.
5.10 Risk of Additive Immunosuppressive Effects From Prior Biological Products (5.10 Risk of Additive Immunosuppressive Effects from Prior Biological Products)
Consider the half-life and mode of action of prior biological products to avoid unintended additive immunosuppressive effects when initiating ZYMFENTRA [see Drug Interactions (7.1)].
Principal Display Panel 120 Mg/ml Prefilled Syringe With Needle Guard Carton (PRINCIPAL DISPLAY PANEL - 120 mg/mL Prefilled Syringe with Needle Guard Carton)
ZymfentraTM
infliximab-dyyb
injection
120 mg/mL
FOR SUBCUTANEOUS USE ONLY
ATTENTION: Dispense the enclosed Medication Guide to each patient.
NDC 72606-025-09
1 prefilled syringe with needle guard
+ 2 alcohol preps
1 Single-Dose Prefilled Syringe with Needle Guard
CELLTRION USA, Inc.
5.9 Risk of Infection With Concurrent Administration With Other Biological Products (5.9 Risk of Infection with Concurrent Administration with Other Biological Products)
Serious infections and neutropenia have been reported with concurrent use of TNF-blockers and other immunosuppressive biological products (e.g., anakinra and abatacept). The concurrent use of ZYMFENTRA with other immunosuppressive biological products used to treat ulcerative colitis and Crohn’s disease may increase the risk of infection and is not recommended [see Drug Interactions (7.1)].
2.2 Recommeneded Dosage for Maintenance Treatment in Ulcerative Colitis Crohn's Disease
- Maintenance dosage starting at Week 10 and thereafter: 120 mg subcutaneously once every two weeks. To switch patients who are responding to maintenance therapy with an infliximab product administered intravenously, administer the first subcutaneous dose of ZYMFNETRA in place of the next scheuled intravenous infusion and every two weeks thereafter.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:19.880953 · Updated: 2026-03-14T22:26:55.770037