These Highlights Do Not Include All The Information Needed To Use Tarpeyo Safely And Effectively. See Full Prescribing Information For Tarpeyo.
938cada4-d6bf-4252-836f-dd40f9eadb4d
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
TARPEYO is indicated to reduce the loss of kidney function in adults with primary immunoglobulin A nephropathy (IgAN) who are at risk for disease progression.
Indications and Usage
TARPEYO is indicated to reduce the loss of kidney function in adults with primary immunoglobulin A nephropathy (IgAN) who are at risk for disease progression.
Dosage and Administration
The recommended duration of therapy is 9 months, with a dosage of 16 mg administered orally once daily [see Clinical Studies ( 14.1 )] . When discontinuing therapy, reduce the dosage to 8 mg once daily for the last 2 weeks of therapy [see Warnings and Precautions ( 5.1 )] . The delayed release capsules should be swallowed whole in the morning, at least 1 hour before a meal. Do not open, crush or chew. If a dose is missed, take the prescribed dose at the next scheduled time. Do not double the next dose. Safety and efficacy of treatment with subsequent courses of TARPEYO have not been established.
Warnings and Precautions
Hypercorticism and Adrenal Axis Suppression: Follow general warnings concerning corticosteroids, patients with hepatic impairment may be at increased risk. Taper upon discontinuation. ( 2 , 5.1 , 8.6 , 12.3 ) Immunosuppression and Increased Risk of Infection: Avoid use in patients with active or quiescent tuberculosis infection, untreated fungal, bacterial, systemic viral or parasitic infections, or ocular herpes simplex. May affect vaccine efficacy. ( 5.2 ) Other Corticosteroid Effects: Monitor patients with concomitant conditions where corticosteroids may have unwanted effects (e.g., hypertension, diabetes mellitus). ( 5.3 )
Contraindications
TARPEYO is contraindicated in patients with hypersensitivity to budesonide or any of the ingredients of TARPEYO. Serious hypersensitivity reactions, including anaphylaxis have occurred with other budesonide formulations.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Hypercorticism and adrenal axis suppression [see Warnings and Precautions ( 5.1 )] Risks of immunosuppression [see Warnings and Precautions ( 5.2 )] Other corticosteroid effects [see Warnings and Precautions ( 5.3 )]
Drug Interactions
Potent CYP3A4 Inhibitors (e.g. ketoconazole, grapefruit juice): Can increase systemic budesonide concentrations: avoid concomitant use. ( 7.1 )
Storage and Handling
TARPEYO (budesonide) delayed release capsules 4 mg, are white opaque- coated capsules marked with “CAL10 4 MG” in black ink on the body of the capsule. They are supplied as follows: NDC 81749-004-01: Bottles of 120 capsules. Child-resistant cap. Store at 20-25°C (68 - 77°F); excursions permitted to 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature]. Keep container tightly closed. Protect from moisture.
How Supplied
TARPEYO (budesonide) delayed release capsules 4 mg, are white opaque- coated capsules marked with “CAL10 4 MG” in black ink on the body of the capsule. They are supplied as follows: NDC 81749-004-01: Bottles of 120 capsules. Child-resistant cap. Store at 20-25°C (68 - 77°F); excursions permitted to 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature]. Keep container tightly closed. Protect from moisture.
Medication Information
Warnings and Precautions
Hypercorticism and Adrenal Axis Suppression: Follow general warnings concerning corticosteroids, patients with hepatic impairment may be at increased risk. Taper upon discontinuation. ( 2 , 5.1 , 8.6 , 12.3 ) Immunosuppression and Increased Risk of Infection: Avoid use in patients with active or quiescent tuberculosis infection, untreated fungal, bacterial, systemic viral or parasitic infections, or ocular herpes simplex. May affect vaccine efficacy. ( 5.2 ) Other Corticosteroid Effects: Monitor patients with concomitant conditions where corticosteroids may have unwanted effects (e.g., hypertension, diabetes mellitus). ( 5.3 )
Indications and Usage
TARPEYO is indicated to reduce the loss of kidney function in adults with primary immunoglobulin A nephropathy (IgAN) who are at risk for disease progression.
Dosage and Administration
The recommended duration of therapy is 9 months, with a dosage of 16 mg administered orally once daily [see Clinical Studies ( 14.1 )] . When discontinuing therapy, reduce the dosage to 8 mg once daily for the last 2 weeks of therapy [see Warnings and Precautions ( 5.1 )] . The delayed release capsules should be swallowed whole in the morning, at least 1 hour before a meal. Do not open, crush or chew. If a dose is missed, take the prescribed dose at the next scheduled time. Do not double the next dose. Safety and efficacy of treatment with subsequent courses of TARPEYO have not been established.
Contraindications
TARPEYO is contraindicated in patients with hypersensitivity to budesonide or any of the ingredients of TARPEYO. Serious hypersensitivity reactions, including anaphylaxis have occurred with other budesonide formulations.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Hypercorticism and adrenal axis suppression [see Warnings and Precautions ( 5.1 )] Risks of immunosuppression [see Warnings and Precautions ( 5.2 )] Other corticosteroid effects [see Warnings and Precautions ( 5.3 )]
Drug Interactions
Potent CYP3A4 Inhibitors (e.g. ketoconazole, grapefruit juice): Can increase systemic budesonide concentrations: avoid concomitant use. ( 7.1 )
Storage and Handling
TARPEYO (budesonide) delayed release capsules 4 mg, are white opaque- coated capsules marked with “CAL10 4 MG” in black ink on the body of the capsule. They are supplied as follows: NDC 81749-004-01: Bottles of 120 capsules. Child-resistant cap. Store at 20-25°C (68 - 77°F); excursions permitted to 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature]. Keep container tightly closed. Protect from moisture.
How Supplied
TARPEYO (budesonide) delayed release capsules 4 mg, are white opaque- coated capsules marked with “CAL10 4 MG” in black ink on the body of the capsule. They are supplied as follows: NDC 81749-004-01: Bottles of 120 capsules. Child-resistant cap. Store at 20-25°C (68 - 77°F); excursions permitted to 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature]. Keep container tightly closed. Protect from moisture.
Description
TARPEYO is indicated to reduce the loss of kidney function in adults with primary immunoglobulin A nephropathy (IgAN) who are at risk for disease progression.
Section 42229-5
Tuberculosis
If TARPEYO is used to treat a condition in patients with latent tuberculosis or tuberculin reactivity, reactivation of tuberculosis may occur. In patients with latent tuberculosis or tuberculin reactivity TARPEYO should be discontinued.
Section 42230-3
|
This Patient Information has been approved by the U.S. Food and Drug Administration. |
Revised: 12/2023 |
|
Patient Information
TARPEYO (tar-PAY-oh) (budesonide) delayed release capsules |
|
|
What is TARPEYO?
TARPEYO is a prescription medicine used to reduce the loss of kidney function in adults with a kidney disease called primary immunoglobulin A nephropathy (IgAN), who are at risk for their disease getting worse. It is not known if TARPEYO is safe and effective in children. |
|
| Do not take TARPEYO ifyou are allergic to budesonide or any of the ingredients in TARPEYO. See the end of this leaflet for a complete list of ingredients in TARPEYO. | |
Before taking TARPEYO, tell your healthcare provider about all of your medical conditions, including if you:
|
|
| Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, vitamins, and herbal supplements. TARPEYO and other medicines may affect each other causing side effects. | |
How should I take TARPEYO?
|
|
|
What should I avoid while taking TARPEYO?
Do noteat grapefruit or drink grapefruit juice during your treatment with TARPEYO. Eating grapefruit or drinking grapefruit juice can increase the level of TARPEYO in your blood. |
|
|
What are the possible side effects of TARPEYO?
TARPEYO may cause serious side effects, including:
|
|
|
|
|
|
|
|
| Tell your healthcare provider if you are under stress or have any symptoms of adrenal suppression during treatment with TARPEYO.
|
|
|
|
|
|
| The most common side effects of TARPEYO include: | |
|
|
| These are not all the possible side effects of TARPEYO.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store TARPEYO?
|
|
| Keep TARPEYO and all medicines out of the reach of children. | |
|
General information about the safe and effective use of TARPEYO.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use TARPEYO for a condition for which it was not prescribed. Do not give TARPEYO to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about TARPEYO that is written for health professionals. |
|
|
What are the ingredients in TARPEYO?
Active ingredient:budesonide Inactive ingredients:sugar spheres (sucrose and starch), hypromellose, polyethylene glycol, citric acid monohydrate, ethyl cellulose, medium chain triglycerides and oleic acid. The capsules contain: hypromellose and titanium oxide (E171). The printing ink on the capsules contain: shellac, propylene glycol and black iron oxide (E172). The enteric coating on the capsules contain: methacrylic acid and methacrylate copolymer, talc and dibutyl sebacate. |
|
| Manufactured for and distributed by: Calliditas Therapeutics AB, Stockholm, Sweden
TARPEYO is a trademark of Calliditas Therapeutics AB, or its affiliates. © 2021 Calliditas Therapeutics AB (publ) Patent: http://www.calliditas.com/patents For more information, go to www.TARPEYOTouchpoints.com or call 1-933-444-8277. |
Section 51945-4
Principal Display Panel - 4 mg Bottle Label
NDC 81749-004-01
TARPEYO™
(budesonide) delayed release capsules
4 mg
120 Capsules
Rx only
Calliditas Therapeutics AB
10 Overdosage
Reports of acute toxicity and/or death following overdosage of corticoids are rare.
In the event of acute overdosage, no specific antidote is available. Treatment consists of supportive and symptomatic therapy.
11 Description
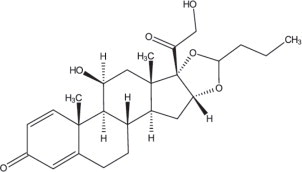
TARPEYO (budesonide) delayed release capsules, for oral administration, contain budesonide, a synthetic corticosteroid, as the active ingredient. Budesonide is designated chemically as 16α, 17α-[(1RS)-Butylidenebis(oxy)]-11β, 21-dihydroxypregna-1,4-diene-3,20-dione.
Budesonide is provided as a mixture of two epimers (22R and 22S). The empirical formula of budesonide is C 25H 34O 6and its molecular weight is 430.5. Its structural formula is:
Budesonide is a white to off-white, tasteless, odorless powder that is practically insoluble in water, sparingly soluble in alcohol, and freely soluble in chloroform.
The beads in each capsule contain the following inactive ingredients: sugar spheres (sucrose and starch), hypromellose, polyethylene glycol, citric acid monohydrate, ethyl cellulose, medium chain triglycerides and oleic acid. The capsule shells contain hypromellose and titanium oxide (E171); and the printing ink on the capsules contain shellac, propylene glycol and black iron oxide (E172). The enteric coating on the capsules contain: methacrylic acid and methacrylate copolymer, talc and dibutyl sebacate.
8.4 Pediatric Use
The safety and efficacy of TARPEYO in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of TARPEYO did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
4 Contraindications
TARPEYO is contraindicated in patients with hypersensitivity to budesonide or any of the ingredients of TARPEYO. Serious hypersensitivity reactions, including anaphylaxis have occurred with other budesonide formulations.
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
7 Drug Interactions
Potent CYP3A4 Inhibitors (e.g. ketoconazole, grapefruit juice): Can increase systemic budesonide concentrations: avoid concomitant use. ( 7.1)
12.2 Pharmacodynamics
Treatment with corticosteroids, including TARPEYO, is associated with a suppression of endogenous cortisol concentrations and an impairment of the hypothalamus-pituitary-adrenal (HPA) axis function.
14.1 Treatment of Igan
TARPEYO was shown to reduce the loss of kidney function in adults with primary IgAN at risk of disease progression in the NefIgArd trial. While the effect on kidney function that was seen during the 9-month treatment period persisted following completion of treatment, TARPEYO did not change the long-term rate of decline in kidney function.
8.6 Hepatic Impairment
Patients with moderate to severe hepatic impairment (Child-Pugh Class B and C, respectively) could be at an increased risk of hypercorticism and adrenal axis suppression due to an increased systemic exposure to budesonide [see Warnings and Precautions ( 5.1) and Clinical Pharmacology ( 12.3)] . Avoid use in patients with severe hepatic impairments (Child-Pugh Class C). Monitor for increased signs and/or symptoms of hypercorticism in patients with moderate hepatic impairment (Child-Pugh Class B).
1 Indications and Usage
TARPEYO is indicated to reduce the loss of kidney function in adults with primary immunoglobulin A nephropathy (IgAN) who are at risk for disease progression.
12.1 Mechanism of Action
Budesonide is a corticosteroid with potent glucocorticoid activity and weak mineralocorticoid activity that undergoes substantial first pass metabolism. Mucosal B-cells present in the ileum, including the Peyer's patches, express glucocorticoid receptors and are responsible for the production of galactose-deficient IgA1 antibodies (Gd-Ag1) causing IgA nephropathy. Through their anti-inflammatory and immunosuppressive effects at the glucocorticoid receptor, corticosteroids can modulate B-cell numbers and activity. It has not been established to what extent TARPEYO's efficacy is mediated via local effects in the ileum vs systemic effects.
5 Warnings and Precautions
- Hypercorticism and Adrenal Axis Suppression:Follow general warnings concerning corticosteroids, patients with hepatic impairment may be at increased risk. Taper upon discontinuation. ( 2, 5.1, 8.6, 12.3)
- Immunosuppression and Increased Risk of Infection:Avoid use in patients with active or quiescent tuberculosis infection, untreated fungal, bacterial, systemic viral or parasitic infections, or ocular herpes simplex. May affect vaccine efficacy. ( 5.2)
- Other Corticosteroid Effects:Monitor patients with concomitant conditions where corticosteroids may have unwanted effects (e.g., hypertension, diabetes mellitus). ( 5.3)
2 Dosage and Administration
The recommended duration of therapy is 9 months, with a dosage of 16 mg administered orally once daily [see Clinical Studies ( 14.1)] . When discontinuing therapy, reduce the dosage to 8 mg once daily for the last 2 weeks of therapy [see Warnings and Precautions ( 5.1)] .
The delayed release capsules should be swallowed whole in the morning, at least 1 hour before a meal. Do not open, crush or chew.
If a dose is missed, take the prescribed dose at the next scheduled time. Do not double the next dose.
Safety and efficacy of treatment with subsequent courses of TARPEYO have not been established.
3 Dosage Forms and Strengths
Delayed release capsule containing 4 mg budesonide. White coated opaque capsules printed with “CAL10 4MG” in black ink.
8 Use in Specific Populations
Lactation: Routine monitoring of linear growth in infants is recommended with chronic use of budesonide in the nursing mother. ( 8.2).
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of TARPEYO was evaluated in 389 patients in the randomized, double-blind, placebo-controlled study, NefIgArd (NCT: 03643965, Phase 3 clinical study in adults with primary IgAN). The data below reflect TARPEYO exposure in 195 patients with a median duration of 41 weeks, compared with comparable exposure to placebo in 194 patients.
The most common adverse reactions, reported in greater than or equal to 5% of TARPEYO-treated patients and greater than or equal to 2% higher than placebo, in the 9-month treatment period are listed in Table 1.
Most adverse reactions that occurred at a greater incidence for TARPEYO compared to placebo were consistent with hypercortisolism and reversible, resolving within 3 months after discontinuation.
| Adverse Reaction |
TARPEYO 16 mg
(N=195) |
Placebo
(N=194) |
| n (%) | n (%) | |
| Peripheral edema | 33 (17) | 10 (5) |
| Hypertension | 23 (12) | 6 (3) |
| Muscle spasms | 23 (12) | 8 (4) |
| Acne | 22 (11) | 2 (1) |
| Headache | 19 (10) | 14 (7) |
| Upper respiratory tract infection | 16 (8) | 12 (6) |
| Face edema | 15 (8) | 1 (0.5) |
| Weight increased | 13 (7) | 6 (3) |
| Dyspepsia | 13 (7) | 4 (2) |
| Dermatitis | 12 (6) | 2 (1) |
| Arthralgia | 12 (6) | 4 (2) |
| White blood cell count increased | 11 (6) | 1 (0.5) |
5.3 Other Corticosteroid Effects
TARPEYO is a systemically available corticosteroid and is expected to cause related adverse reactions. Monitor patients with hypertension, prediabetes, diabetes mellitus, osteoporosis, peptic ulcer, glaucoma or cataracts, or with a family history of diabetes or glaucoma, or with any other condition where corticosteroids may have unwanted effects.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling ( Patient Information).
Advise patients that TARPEYO may cause hypercorticism and adrenal axis suppression and to follow a taper schedule, as instructed by their healthcare provider if discontinuing therapy [see Warnings and Precautions ( 5.1)].
TARPEYO causes immunosuppression. Advise patients to avoid exposure to people with chicken pox or measles and, if exposed, to consult their healthcare provider immediately. There is an increased risk of developing a variety of infections, including worsening of existing tuberculosis, fungal, bacterial, viral or parasitic infections, or ocular herpes simplex, and to contact their healthcare provider if they develop any symptoms of infection [see Warnings and Precautions ( 5.3)] . Provide advice regarding vaccination schedules for immunocompromised patients.
Advise patients that TARPEYO delayed release capsules should be swallowed whole and not chewed, crushed or broken and to take TARPEYO in the morning, at least 1 hour before a meal [See Dosage and Administration ( 2)].
Advise patients to avoid the consumption of grapefruit juice for the duration of their TARPEYO therapy [See Drug Interactions ( 7.1)].
TARPEYO is a registered trademark of Calliditas Therapeutics AB, or its affiliates.
© 2024 Calliditas Therapeutics AB (publ)
Manufactured for and distributed by:
Calliditas Therapeutics AB
Stockholm, Sweden
Patent: http://www.calliditas.com/patents
16 How Supplied/storage and Handling
TARPEYO (budesonide) delayed release capsules 4 mg, are white opaque- coated capsules marked with “CAL10 4 MG” in black ink on the body of the capsule. They are supplied as follows:
NDC 81749-004-01: Bottles of 120 capsules. Child-resistant cap.
Store at 20-25°C (68 - 77°F); excursions permitted to 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature].
Keep container tightly closed. Protect from moisture.
7.1 Interaction With Cyp3a4 Inhibitors
Budesonide is a substrate for CYP3A4. Avoid use with potent CYP3A4 inhibitors; e.g. ketoconazole, itraconazole, ritonavir, indinavir, saquinavir, erythromycin, and cyclosporine [see Clinical Pharmacology ( 12.3)] .
Avoid ingestion of grapefruit juice with TARPEYO. Intake of grapefruit juice, which inhibits CYP3A4 activity, can increase the systemic exposure to budesonide [see Clinical Pharmacology ( 12.3)] .
5.1 Hypercorticism and Adrenal Axis Suppression
When corticosteroids are used chronically, systemic effects such as hypercorticism and adrenal suppression may occur. Corticosteroids can reduce the response of the hypothalamus-pituitary-adrenal (HPA) axis to stress. In situations where patients are subject to surgery or other stress situations, supplementation with a systemic corticosteroid is recommended. When discontinuing therapy [see Dosing and Administration ( 2)] or switching between corticosteroids, monitor for signs of adrenal axis suppression.
Patients with moderate to severe hepatic impairment (Child-Pugh Class B and C respectively) could be at an increased risk of hypercorticism and adrenal axis suppression due to an increased systemic exposure of oral budesonide. Avoid use in patients with severe hepatic impairment (Child-Pugh Class C). Monitor for increased signs and/or symptoms of hypercorticism in patients with moderate hepatic impairment (Child-Pugh Class B) [see Use in Specific Populations ( 8.6), Clinical Pharmacology ( 12.3)] .
5.2 Immunosuppression and Increased Risk of Infection
Corticosteroids, including TARPEYO, suppress the immune system and increase the risk of infection with any pathogen, including viral, bacterial, fungal, protozoan, or helminthic pathogens. Corticosteroids can:
- Reduce resistance to new infections
- Exacerbate existing infections
- Increase the risk of disseminated infections
- Increase the risk of reactivation or exacerbation of latent infections
- Mask some signs of infection
Corticosteroid-associated infections can be mild but can be severe and at times fatal. The rate of infectious complications increases with increasing corticosteroid dosages.
Monitor for the development of infection and consider TARPEYO withdrawal as needed.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies with budesonide were conducted in rats and mice. In a two-year study in Sprague-Dawley rats, budesonide caused a statistically significant increase in the incidence of gliomas in male rats at an oral dose of 50 mcg/kg (approximately 0.03 times the maximum recommended human dose (MRHD) on a body surface area basis). In addition, there were increased incidences of primary hepatocellular tumors in male rats at 25 mcg/kg (approximately 0.015 times the MRHD on a body surface area basis) and above. No tumorigenicity was seen in female rats at oral doses up to 50 mcg/kg (approximately 0.03 times the MRHD on a body surface area basis). In an additional two-year study in male Sprague-Dawley rats, budesonide caused no gliomas at an oral dose of 50 mcg/kg (approximately 0.03 times the MRHD on a body surface area basis). However, it caused a statistically significant increase in the incidence of hepatocellular tumors at an oral dose of 50 mcg/kg (approximately 0.03 times the MRHD of a body surface area basis). The concurrent reference corticosteroids (prednisolone and triamcinolone acetonide) showed similar findings. In a 91-week study in mice, budesonide caused no treatment-related carcinogenicity at oral doses up to 200 mcg/kg (approximately 0.06 times the MRHD on a body surface area basis).
Budesonide was not genotoxic in the Ames test, the mouse lymphoma cell forward gene mutation (TK +/-) test, the human lymphocyte chromosome aberration test, the Drosophila melanogaster sex-linked recessive lethal test, the rat hepatocyte UDS test and the mouse micronucleus test.
In rats, budesonide had no effect on fertility at subcutaneous doses up to 80 mcg/kg (approximately 0.05 times the MRHD on a body surface area basis). However, it caused a decrease in prenatal viability and viability in pups at birth and during lactation, along with a decrease in maternal food consumption and body weight gain, at subcutaneous doses of 20 mcg/kg (approximately 0.012 times the MRHD on a body surface area basis) and above. No such effects were noted at 5 mcg/kg (approximately 0.003 times the MRHD on a body surface area basis).
Structured Label Content
Section 42229-5 (42229-5)
Tuberculosis
If TARPEYO is used to treat a condition in patients with latent tuberculosis or tuberculin reactivity, reactivation of tuberculosis may occur. In patients with latent tuberculosis or tuberculin reactivity TARPEYO should be discontinued.
Section 42230-3 (42230-3)
|
This Patient Information has been approved by the U.S. Food and Drug Administration. |
Revised: 12/2023 |
|
Patient Information
TARPEYO (tar-PAY-oh) (budesonide) delayed release capsules |
|
|
What is TARPEYO?
TARPEYO is a prescription medicine used to reduce the loss of kidney function in adults with a kidney disease called primary immunoglobulin A nephropathy (IgAN), who are at risk for their disease getting worse. It is not known if TARPEYO is safe and effective in children. |
|
| Do not take TARPEYO ifyou are allergic to budesonide or any of the ingredients in TARPEYO. See the end of this leaflet for a complete list of ingredients in TARPEYO. | |
Before taking TARPEYO, tell your healthcare provider about all of your medical conditions, including if you:
|
|
| Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, vitamins, and herbal supplements. TARPEYO and other medicines may affect each other causing side effects. | |
How should I take TARPEYO?
|
|
|
What should I avoid while taking TARPEYO?
Do noteat grapefruit or drink grapefruit juice during your treatment with TARPEYO. Eating grapefruit or drinking grapefruit juice can increase the level of TARPEYO in your blood. |
|
|
What are the possible side effects of TARPEYO?
TARPEYO may cause serious side effects, including:
|
|
|
|
|
|
|
|
| Tell your healthcare provider if you are under stress or have any symptoms of adrenal suppression during treatment with TARPEYO.
|
|
|
|
|
|
| The most common side effects of TARPEYO include: | |
|
|
| These are not all the possible side effects of TARPEYO.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store TARPEYO?
|
|
| Keep TARPEYO and all medicines out of the reach of children. | |
|
General information about the safe and effective use of TARPEYO.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use TARPEYO for a condition for which it was not prescribed. Do not give TARPEYO to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about TARPEYO that is written for health professionals. |
|
|
What are the ingredients in TARPEYO?
Active ingredient:budesonide Inactive ingredients:sugar spheres (sucrose and starch), hypromellose, polyethylene glycol, citric acid monohydrate, ethyl cellulose, medium chain triglycerides and oleic acid. The capsules contain: hypromellose and titanium oxide (E171). The printing ink on the capsules contain: shellac, propylene glycol and black iron oxide (E172). The enteric coating on the capsules contain: methacrylic acid and methacrylate copolymer, talc and dibutyl sebacate. |
|
| Manufactured for and distributed by: Calliditas Therapeutics AB, Stockholm, Sweden
TARPEYO is a trademark of Calliditas Therapeutics AB, or its affiliates. © 2021 Calliditas Therapeutics AB (publ) Patent: http://www.calliditas.com/patents For more information, go to www.TARPEYOTouchpoints.com or call 1-933-444-8277. |
Section 51945-4 (51945-4)
Principal Display Panel - 4 mg Bottle Label
NDC 81749-004-01
TARPEYO™
(budesonide) delayed release capsules
4 mg
120 Capsules
Rx only
Calliditas Therapeutics AB
10 Overdosage (10 OVERDOSAGE)
Reports of acute toxicity and/or death following overdosage of corticoids are rare.
In the event of acute overdosage, no specific antidote is available. Treatment consists of supportive and symptomatic therapy.
11 Description (11 DESCRIPTION)
TARPEYO (budesonide) delayed release capsules, for oral administration, contain budesonide, a synthetic corticosteroid, as the active ingredient. Budesonide is designated chemically as 16α, 17α-[(1RS)-Butylidenebis(oxy)]-11β, 21-dihydroxypregna-1,4-diene-3,20-dione.
Budesonide is provided as a mixture of two epimers (22R and 22S). The empirical formula of budesonide is C 25H 34O 6and its molecular weight is 430.5. Its structural formula is:
Budesonide is a white to off-white, tasteless, odorless powder that is practically insoluble in water, sparingly soluble in alcohol, and freely soluble in chloroform.
The beads in each capsule contain the following inactive ingredients: sugar spheres (sucrose and starch), hypromellose, polyethylene glycol, citric acid monohydrate, ethyl cellulose, medium chain triglycerides and oleic acid. The capsule shells contain hypromellose and titanium oxide (E171); and the printing ink on the capsules contain shellac, propylene glycol and black iron oxide (E172). The enteric coating on the capsules contain: methacrylic acid and methacrylate copolymer, talc and dibutyl sebacate.
8.4 Pediatric Use
The safety and efficacy of TARPEYO in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of TARPEYO did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
4 Contraindications (4 CONTRAINDICATIONS)
TARPEYO is contraindicated in patients with hypersensitivity to budesonide or any of the ingredients of TARPEYO. Serious hypersensitivity reactions, including anaphylaxis have occurred with other budesonide formulations.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
7 Drug Interactions (7 DRUG INTERACTIONS)
Potent CYP3A4 Inhibitors (e.g. ketoconazole, grapefruit juice): Can increase systemic budesonide concentrations: avoid concomitant use. ( 7.1)
12.2 Pharmacodynamics
Treatment with corticosteroids, including TARPEYO, is associated with a suppression of endogenous cortisol concentrations and an impairment of the hypothalamus-pituitary-adrenal (HPA) axis function.
14.1 Treatment of Igan (14.1 Treatment of IgAN)
TARPEYO was shown to reduce the loss of kidney function in adults with primary IgAN at risk of disease progression in the NefIgArd trial. While the effect on kidney function that was seen during the 9-month treatment period persisted following completion of treatment, TARPEYO did not change the long-term rate of decline in kidney function.
8.6 Hepatic Impairment
Patients with moderate to severe hepatic impairment (Child-Pugh Class B and C, respectively) could be at an increased risk of hypercorticism and adrenal axis suppression due to an increased systemic exposure to budesonide [see Warnings and Precautions ( 5.1) and Clinical Pharmacology ( 12.3)] . Avoid use in patients with severe hepatic impairments (Child-Pugh Class C). Monitor for increased signs and/or symptoms of hypercorticism in patients with moderate hepatic impairment (Child-Pugh Class B).
1 Indications and Usage (1 INDICATIONS AND USAGE)
TARPEYO is indicated to reduce the loss of kidney function in adults with primary immunoglobulin A nephropathy (IgAN) who are at risk for disease progression.
12.1 Mechanism of Action
Budesonide is a corticosteroid with potent glucocorticoid activity and weak mineralocorticoid activity that undergoes substantial first pass metabolism. Mucosal B-cells present in the ileum, including the Peyer's patches, express glucocorticoid receptors and are responsible for the production of galactose-deficient IgA1 antibodies (Gd-Ag1) causing IgA nephropathy. Through their anti-inflammatory and immunosuppressive effects at the glucocorticoid receptor, corticosteroids can modulate B-cell numbers and activity. It has not been established to what extent TARPEYO's efficacy is mediated via local effects in the ileum vs systemic effects.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypercorticism and Adrenal Axis Suppression:Follow general warnings concerning corticosteroids, patients with hepatic impairment may be at increased risk. Taper upon discontinuation. ( 2, 5.1, 8.6, 12.3)
- Immunosuppression and Increased Risk of Infection:Avoid use in patients with active or quiescent tuberculosis infection, untreated fungal, bacterial, systemic viral or parasitic infections, or ocular herpes simplex. May affect vaccine efficacy. ( 5.2)
- Other Corticosteroid Effects:Monitor patients with concomitant conditions where corticosteroids may have unwanted effects (e.g., hypertension, diabetes mellitus). ( 5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
The recommended duration of therapy is 9 months, with a dosage of 16 mg administered orally once daily [see Clinical Studies ( 14.1)] . When discontinuing therapy, reduce the dosage to 8 mg once daily for the last 2 weeks of therapy [see Warnings and Precautions ( 5.1)] .
The delayed release capsules should be swallowed whole in the morning, at least 1 hour before a meal. Do not open, crush or chew.
If a dose is missed, take the prescribed dose at the next scheduled time. Do not double the next dose.
Safety and efficacy of treatment with subsequent courses of TARPEYO have not been established.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Delayed release capsule containing 4 mg budesonide. White coated opaque capsules printed with “CAL10 4MG” in black ink.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: Routine monitoring of linear growth in infants is recommended with chronic use of budesonide in the nursing mother. ( 8.2).
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of TARPEYO was evaluated in 389 patients in the randomized, double-blind, placebo-controlled study, NefIgArd (NCT: 03643965, Phase 3 clinical study in adults with primary IgAN). The data below reflect TARPEYO exposure in 195 patients with a median duration of 41 weeks, compared with comparable exposure to placebo in 194 patients.
The most common adverse reactions, reported in greater than or equal to 5% of TARPEYO-treated patients and greater than or equal to 2% higher than placebo, in the 9-month treatment period are listed in Table 1.
Most adverse reactions that occurred at a greater incidence for TARPEYO compared to placebo were consistent with hypercortisolism and reversible, resolving within 3 months after discontinuation.
| Adverse Reaction |
TARPEYO 16 mg
(N=195) |
Placebo
(N=194) |
| n (%) | n (%) | |
| Peripheral edema | 33 (17) | 10 (5) |
| Hypertension | 23 (12) | 6 (3) |
| Muscle spasms | 23 (12) | 8 (4) |
| Acne | 22 (11) | 2 (1) |
| Headache | 19 (10) | 14 (7) |
| Upper respiratory tract infection | 16 (8) | 12 (6) |
| Face edema | 15 (8) | 1 (0.5) |
| Weight increased | 13 (7) | 6 (3) |
| Dyspepsia | 13 (7) | 4 (2) |
| Dermatitis | 12 (6) | 2 (1) |
| Arthralgia | 12 (6) | 4 (2) |
| White blood cell count increased | 11 (6) | 1 (0.5) |
5.3 Other Corticosteroid Effects
TARPEYO is a systemically available corticosteroid and is expected to cause related adverse reactions. Monitor patients with hypertension, prediabetes, diabetes mellitus, osteoporosis, peptic ulcer, glaucoma or cataracts, or with a family history of diabetes or glaucoma, or with any other condition where corticosteroids may have unwanted effects.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling ( Patient Information).
Advise patients that TARPEYO may cause hypercorticism and adrenal axis suppression and to follow a taper schedule, as instructed by their healthcare provider if discontinuing therapy [see Warnings and Precautions ( 5.1)].
TARPEYO causes immunosuppression. Advise patients to avoid exposure to people with chicken pox or measles and, if exposed, to consult their healthcare provider immediately. There is an increased risk of developing a variety of infections, including worsening of existing tuberculosis, fungal, bacterial, viral or parasitic infections, or ocular herpes simplex, and to contact their healthcare provider if they develop any symptoms of infection [see Warnings and Precautions ( 5.3)] . Provide advice regarding vaccination schedules for immunocompromised patients.
Advise patients that TARPEYO delayed release capsules should be swallowed whole and not chewed, crushed or broken and to take TARPEYO in the morning, at least 1 hour before a meal [See Dosage and Administration ( 2)].
Advise patients to avoid the consumption of grapefruit juice for the duration of their TARPEYO therapy [See Drug Interactions ( 7.1)].
TARPEYO is a registered trademark of Calliditas Therapeutics AB, or its affiliates.
© 2024 Calliditas Therapeutics AB (publ)
Manufactured for and distributed by:
Calliditas Therapeutics AB
Stockholm, Sweden
Patent: http://www.calliditas.com/patents
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
TARPEYO (budesonide) delayed release capsules 4 mg, are white opaque- coated capsules marked with “CAL10 4 MG” in black ink on the body of the capsule. They are supplied as follows:
NDC 81749-004-01: Bottles of 120 capsules. Child-resistant cap.
Store at 20-25°C (68 - 77°F); excursions permitted to 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature].
Keep container tightly closed. Protect from moisture.
7.1 Interaction With Cyp3a4 Inhibitors (7.1 Interaction with CYP3A4 Inhibitors)
Budesonide is a substrate for CYP3A4. Avoid use with potent CYP3A4 inhibitors; e.g. ketoconazole, itraconazole, ritonavir, indinavir, saquinavir, erythromycin, and cyclosporine [see Clinical Pharmacology ( 12.3)] .
Avoid ingestion of grapefruit juice with TARPEYO. Intake of grapefruit juice, which inhibits CYP3A4 activity, can increase the systemic exposure to budesonide [see Clinical Pharmacology ( 12.3)] .
5.1 Hypercorticism and Adrenal Axis Suppression
When corticosteroids are used chronically, systemic effects such as hypercorticism and adrenal suppression may occur. Corticosteroids can reduce the response of the hypothalamus-pituitary-adrenal (HPA) axis to stress. In situations where patients are subject to surgery or other stress situations, supplementation with a systemic corticosteroid is recommended. When discontinuing therapy [see Dosing and Administration ( 2)] or switching between corticosteroids, monitor for signs of adrenal axis suppression.
Patients with moderate to severe hepatic impairment (Child-Pugh Class B and C respectively) could be at an increased risk of hypercorticism and adrenal axis suppression due to an increased systemic exposure of oral budesonide. Avoid use in patients with severe hepatic impairment (Child-Pugh Class C). Monitor for increased signs and/or symptoms of hypercorticism in patients with moderate hepatic impairment (Child-Pugh Class B) [see Use in Specific Populations ( 8.6), Clinical Pharmacology ( 12.3)] .
5.2 Immunosuppression and Increased Risk of Infection
Corticosteroids, including TARPEYO, suppress the immune system and increase the risk of infection with any pathogen, including viral, bacterial, fungal, protozoan, or helminthic pathogens. Corticosteroids can:
- Reduce resistance to new infections
- Exacerbate existing infections
- Increase the risk of disseminated infections
- Increase the risk of reactivation or exacerbation of latent infections
- Mask some signs of infection
Corticosteroid-associated infections can be mild but can be severe and at times fatal. The rate of infectious complications increases with increasing corticosteroid dosages.
Monitor for the development of infection and consider TARPEYO withdrawal as needed.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies with budesonide were conducted in rats and mice. In a two-year study in Sprague-Dawley rats, budesonide caused a statistically significant increase in the incidence of gliomas in male rats at an oral dose of 50 mcg/kg (approximately 0.03 times the maximum recommended human dose (MRHD) on a body surface area basis). In addition, there were increased incidences of primary hepatocellular tumors in male rats at 25 mcg/kg (approximately 0.015 times the MRHD on a body surface area basis) and above. No tumorigenicity was seen in female rats at oral doses up to 50 mcg/kg (approximately 0.03 times the MRHD on a body surface area basis). In an additional two-year study in male Sprague-Dawley rats, budesonide caused no gliomas at an oral dose of 50 mcg/kg (approximately 0.03 times the MRHD on a body surface area basis). However, it caused a statistically significant increase in the incidence of hepatocellular tumors at an oral dose of 50 mcg/kg (approximately 0.03 times the MRHD of a body surface area basis). The concurrent reference corticosteroids (prednisolone and triamcinolone acetonide) showed similar findings. In a 91-week study in mice, budesonide caused no treatment-related carcinogenicity at oral doses up to 200 mcg/kg (approximately 0.06 times the MRHD on a body surface area basis).
Budesonide was not genotoxic in the Ames test, the mouse lymphoma cell forward gene mutation (TK +/-) test, the human lymphocyte chromosome aberration test, the Drosophila melanogaster sex-linked recessive lethal test, the rat hepatocyte UDS test and the mouse micronucleus test.
In rats, budesonide had no effect on fertility at subcutaneous doses up to 80 mcg/kg (approximately 0.05 times the MRHD on a body surface area basis). However, it caused a decrease in prenatal viability and viability in pups at birth and during lactation, along with a decrease in maternal food consumption and body weight gain, at subcutaneous doses of 20 mcg/kg (approximately 0.012 times the MRHD on a body surface area basis) and above. No such effects were noted at 5 mcg/kg (approximately 0.003 times the MRHD on a body surface area basis).
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:52:37.983602 · Updated: 2026-03-14T22:44:14.787449